Abstract
Lowland temperate rivers provide important habitats for piscivorous fishes, but with their year-round spatial and temporal habitat use is often poorly understood, including their use of off-channel habitats. Here, the movements and habitat use of the piscivorous native Northern pike Esox lucius and invasive pikeperch Sander lucioperca were investigated using acoustic telemetry in the highly regulated (through impoundment) lower River Severn, Western England over a 12-month period, where off-channel habitat availability was limited to a single boat marina. The movements of both species varied with season and temperature, with both species moving greater distances in spring. Increasing water temperatures up to 15 °C resulted in a higher frequency of movements of both species, but movements then decreased at temperatures higher than this. Northern pike detections in the river increased in periods of lower river discharge and warmer temperatures, with the off-channel refuge providing an important habitat all year round (78% of detections occurred there). While 63% of pikeperch detections also occurred in the marina, 89% of these detections occurred between December and April. These results thus emphasise the importance of this limited off-channel habitat as potential spawning locations for invasive pikeperch and foraging areas for native Northern pike.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Large-bodied non-native fishes of high trophic position are often introduced into inland waters to enhance recreational freshwater angling (Hickley & Chare, 2004; Ellender & Weyl, 2014). Should these fishes develop an invasive population then in addition to being a major driver of biological and functional homogenisation, they can have substantial negative effects on native fish diversity through both their consumptive effects on prey species and non-consumptive effects on native trophic analogues (Eby et al., 2006; Sih et al., 2010). The release of these fishes into rivers modified by engineered structures (e.g. dams and weirs) often results in their successful establishment and invasion because of the favourable conditions provided by the more homogeneous and stable hydrological environment (Clavero et al., 2004; Johnson et al., 2008). However, weirs and dams can also act to limit the upstream dispersal of invasive species, thus protecting native species in those areas (Burnett et al. 2023).
These engineered riverine environments often also result in the loss of river-floodplain connectivity, despite the importance of this connectivity for enabling fish to access a range of functional habitats in the floodplain (Bolland et al., 2015). Moreover, engineered rivers are often also subjected to channelisation and artificial levee construction that results in a straightened channel (Brookes et al., 1983). These modifications result in episodic high flow and flood events being of higher severity as the water remains in the main channel throughout, with the conditions often preventing fish from accessing off-channel refugia (Bolland et al., 2012). Accordingly, in temperate lowland rivers, summers often provide homogeneous and stable hydrological environments that are favourable for larval and juvenile fish recruitment (Nunn et al., 2007a), increasing the probability of non-native fish establishing. However, during winter periods, these rivers often provide relatively hostile conditions characterised by elevated flows and large in-channel flood peaks (Death et al., 2015), which can be deleterious for juvenile fishes where off-channel refugia is limited and largely inaccessible (Bolland et al., 2015).
The lower River Severn in western England was first modified in the 1850s through construction of multiple of navigation weirs, with the impounded river also subjected to channel straightening to assist flood prevention (Gutmann Roberts et al., 2019). Prior to modification, the river channel included areas of water that was sufficiently shallow for boats to have to pushed upstream manually (IHBC, 2021) and the river is considered likely to have been a heterogenous habitat for fish including multiple natural off-channel refuges. Today, however, the main river channel is characterised by a largely straight river channel with high flood banks, depths that always exceed 2 m and are usually deeper. There are also minimal off-channel habitats for fish, with the most downstream non-tidal impounded section only having one off-channel macro-habitat present in the form of a boat marina of approximately 3 ha in size. The fish assemblage of the river had also been modified by the presence of alien species including the obligate piscivore pikeperch Sander lucioperca (Linnaeus, 1758), which has been present since 1980 (Hickley, 1986). Pikeperch thus coexist with native piscivore Northern pike Esox lucius Linnaeus, 1758 in the river.
Differences in the biology of Northern pike and pikeperch suggest some potential for differences in their habitat use and movement patterns. For example, Northern pike have greater foraging success than pikeperch in the presence of submerged vegetation (Greenberg et al., 1995), with these habitats also being important Northern pike spawning substrate and nursery habitat, especially in shallow water but with spawning also occurring in water up to 6 m deep (Craig, 2008). Conversely, pikeperch spawning involves nest construction and guarding by males, with nests made on sandy, silty or muddy substrates (Lappalainen et al., 2003). Northern pike in temperate waters tend to spawn in early spring (March, April), with pikeperch spawning generally commencing in April (Craig, 2008). The reproductive cycle is at least in part governed by temperature, with active vitellogenesis commencing in autumn and accelerating as water temperatures reduce in winter (Lenhardt & Cakić, 2002).
In anthropogenically modified rivers, off-channel habitats also provide Northern pike with important areas for refugia and foraging (Pauwels et al., 2016), while pikeperch foraging generally involves active searching in open water (Turesson & Brönmark, 2004). Although both species tend to rely on sight for locating prey, juveniles of both species are capable of foraging successfully in turbid water, although energetic costs might be increased (Skov et al., 2002; Zingel & Paaver, 2010). Both species also tend to move more at twilight as this elevates foraging success (e.g. Horký et al., 2008; Baktoft et al., 2012) and, where pikeperch are invasive, they often share prey resources with Northern pike (Jepsen et al., 2000), especially in similar size classes (Nolan et al., 2019a). Consequently, in channelised and impounded river sections of limited off-channel habitat, the two species could potentially express similar movement patterns and habitat use in relation to foraging and spawning. However, pike movements are also scaled by body size, with larger fish using greater spatial areas than smaller pike, thus potentially avoiding interactions with other piscivores (Dhellemmes et al., 2023), and with floods potentially displacing some individuals from areas of river that do not always return (Chevallier et al., 2023).
The aim of this study was to thus simultaneously assess the habitat use and movement patterns of these two piscivorous fishes in the lower River Severn basin, England, using acoustic telemetry. The objectives were to: (i) determine the spatial and temporal movement patterns (including diel movement patterns) for both species; (ii) test the influence of environmental parameters on these movement patterns; and (iii) quantify the importance of the limited off-channel habitat to each species (as an off-channel residency index) versus the main river channel. We predicted that the two species would show similar movement patterns and habitat use, from diel to seasonal patterns that were temperature related, with the off-river refuge providing important habitats for both species throughout the year. The application of acoustic telemetry, where the individual fish were implanted with an internal acoustic transmitter that enabled their detection on an array of acoustic hydrophones (receivers), enabled the fish movements to be recreated through time and space using these detections and tested versus environmental parameters, as has been completed for other fishes in the study river (e.g. Gutmann-Roberts et al., 2019; Davies et al., 2022). Thus, throughout the study, a movement is defined as a tagged fish being initially detected on one receiver and subsequently detected on another receiver located either up- or downstream. The river distance between the receivers allows for determination of the movement distance, and the detection times of the receiver provide information on the direction and duration of the movement.
Materials and methods
Study area
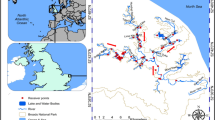
The study area was a section of the lower River Severn, Western England (Fig. 1a) between Diglis Weir (upstream) and Upper Lode Weir (downstream) (Fig. 1b; 52.1819, − 2.2241 to 51.9943, − 2.1735) and encompassing the lower section of the River Teme tributary. The weirs at the up- and downstream limits of the area provided a closed area of 28 km, as neither of the species were assumed to be capable of traversing these weirs because of their height and with no fish passes present at that time; although locks are present next to the weirs to assist navigation, fish have not been detected moving through them in any studies on the river (e.g. Davies et al., 2022, 2024). It was also considered highly improbable that the fish would leave the study area during brief periods of flooding when the weirs were over-topped, and to the best of our knowledge, such events did not occur. The weirs were constructed for navigation and have resulted in the river being highly impounded, with heavy boat traffic in summer (Fig. 1b). The study area is characterised by widths to 40 m, depths to over 4 m (C-MAP, 2019); there is minimal instream vegetation and off-channel areas, with the only exception being a boat marina located at Upton-upon-Severn (Fig. 1).
Map showing a the position of the study area within the UK; b the study area within the River Severn with the receiver and their respective locations shown by circles and the position of the flow gauging station (star) c the off-channel habitat provided by Upton-Upon-Severn marina showing the receiver locations (circles) and the sampling location (triangle). The arrow indicates the direction of water flow, solid lines show the position of weirs, * marks the location of the Saxons Lode gauging station. All receivers were in place for the entire duration of the study with the exception of receiver #2 and #3 which were deployed on the 24 April 2018
An acoustic receiver array was established in the study area prior to fish tagging (on the 21 August 2017), except for receivers #2 and #3 which were deployed on 24 April 2018. The receiver array thus comprised a total of 11 acoustic receivers (VR2, Vemco Ltd) in fixed locations (Table 1; Fig. 1b). These receivers remained in place throughout the remainder of the study period, (Fig. 1b). Range testing revealed a maximum detection range of approximately 100 m across the study area (Gutmann Roberts et al., 2019); this exceeded the river width in all locations and thus the receivers functioned as a gated array. Receiver positions were selected to provide equidistant coverage between the upstream and downstream range of the study area, whilst also enabling detection of movements in and out of the boat marina at Upton-Upon-Severn, and the residency of the fish in this marina (Fig. 1c). Receiver batteries were removed and replaced periodically, enabling the stored data to be downloaded for analysis. A temperature logger (Tinytag; Gemini Data Loggers) was deployed at the site of receiver number 6 and recorded temperature (to 0.1 °C) every three hours. Flow data (m3 s−1) were acquired from the flow gauging station operated by the Environment Agency at Saxons Lode (52.0495, − 2.2005, Fig. 1b), with river flow recorded every 15 min.
Fish sampling and tagging
Fish sampling and tagging was completed on 27 September 2017 in the river and marina at Upton-Upon-Severn (Fig. 1c), selected because of its centrality within the study area. Fish were captured using electric fishing from a boat, coupled with rod and line angling. Following their capture, fish were transferred to an aerated tank. Tagging involved general anaesthesia (tricaine methanesulfonate; MS-222) before an acoustic transmitter (69 kHz V9 or V13; Vemco Ltd) was inserted into the peritoneal cavity through a small incision (less than 2 cm wide) which was then closed with a single suture and the application of surgical adhesive. V9 acoustic transmitters were 9 × 21 mm and 1.6 g, whilst V13 transmitters were 16 × 36 mm and 6 g. All transmitters were set to transmit randomly every 60 to 180 s, providing an overall battery life of approximately 22 months (V9) and 36 months (V13). Random repeat pulse rates allowed multiple individuals to be monitored simultaneously within a given area via the fixed receivers with reduced risk of continuous signal overlap and interference. Tag identification numbers were recorded, with the fish measured (fork length, nearest mm) and transferred to an aerated recovery tank where they were held until normal swimming behaviour resumed. The fish were then released close to their location of capture. All surgical procedures were undertaken by a licensed, competent and experienced practitioner following an ethical approval process, with all regulated procedures completed under UK Home Office licence PPL 70/8063. A total of 17 Northern pike and 8 pikeperch were tagged, Northern pike ranged in size from 574 to 958 mm and pikeperch from 356 to 692 mm (Table 2).
Data analysis
The fish tracking data were analysed for the period 27 October 2017 to 26 October 2018 that provided data over 365 continual days. Data from the period between tagging and 27 October 2017 were not included to avoid movements that might have been subject to behavioural changes caused by the tagging procedure (Pauwels et al., 2014). Although tagging procedures can reduce fish movements for several days post-tagging (e.g. Sonamzi et al., 2020), this was not considered a major concern. Movements during this period would be indicative of under-estimated distances moved rather than unusually high movement patterns. Northern pike ID 51155 was not detected after 09 November 2017 and so was removed from further analyses.
At the end of the tracking period, the detection data (comprising over 1.6 million individual detections) were initially analysed in the package ‘Vtrack’ in R (Campbell et al., 2012) for residency and non-residency events for each individual. A residency event was defined when a transmitter was detected by a receiver (minimum of 2 detections) and terminated when the transmitter was detected at another receiver, or if the transmitter was not detected by the same receiver within a defined timeout window of 10 min. This time was chosen as a conservative estimate of the time it would take an individual Northern pike or pikeperch to move away from the detection range of a receiver (~ 100 m) based on Northern pike mean swimming speed of 0.23 m s−1 (0.45 body length s−1) (Diana, 1980). No data exists for absolute swimming speed in pikeperch, although swimming speed has been recorded as 1.6 body lengths s−1 (Poulet et al., 2005a). A non-residency event was defined as the movement between the detection fields of two receivers and incorporated measurements of the circuitous distance (river distance) between receivers, with river distance used in all distance calculations. Thus a non-residency event is analogous to a fish movement between two receivers in a specific timeframe). Throughout the study, the receivers remained functional and detection efficiency was > 99% (i.e. minimal instances of where the detection of a moving fish was missed on a receiver between two other receivers that did detect it), and thus no action was needed on this. No false detections were apparent in the dataset. The detection fields of receivers never overlapped, ensuring that individual fish detections occurred on a single receiver and were never detected simultaneously on multiple receivers. Consequently, the detection of a fish on adjacent receivers indicated a movement, with the time between detections on the receiver providing information on the speed of movement.
Individual maximum upstream and downstream distance moved was calculated from the central position of Upton-Upon-Severn marina (‘0’; Figs. 1 and 2) to the most upstream and downstream receivers with detections. Individual total distance moved was calculated for the 12-month study period as the sum of all movements between receivers. Individual mean daily distance was then calculated for the 12-month study period as the total distance travelled by each individual, divided by the length of the study period (i.e. the time between first and last detections for each individual). Although such movement rates are likely to be an underestimate of total movement, they can provide useful insights (Cooke et al., 2001), and are an attempt to reduce the error associated with differences in the number of days individuals were detected. Spearman’s rank correlation coefficient was used to initially test for a correlation between individual total distance moved and mean daily distance moved across the analytical period; as there was significant correlation for both Northern pike (Spearman’s rho (r) = 0.97, P < 0.01) and pikeperch (r = 0.95, P < 0.01), then following testing for normality and homoscedasticity (Shapiro-Wilks and Levene’s tests, respectively), differences between Northern pike and pikeperch in mean daily distances moved during the 12-month study period were tested using a Mann–Whitney U test.
Continuous upstream and downstream movements (km) of Northern pike and pikeperch from 0 (r representing the marina at Upton-Upon-Severn) on the primary axis (solid line), and the total monthly distance moved (km) on the secondary axis (open circle, dashed line); spawning month is represented by the area between the dotted lines and individuals are identified according to transmitter ID (Table 2). Note the difference in scale between the primary and secondary axis
To assess seasonal differences in total daily distance moved (cf. Objective i), a mean of the total daily distance moved was calculated for each species for each day in the study period. Differences in this mean total daily distance moved across seasons were tested using a generalised linear model (GLM) with a quasi-Poisson distribution to account for over-dispersion, with the significance of the model tested using a likelihood ratio test.
To assess movements in relation to time of year, water flow and temperature for both species (cf. Objective ii), it was first necessary to ensure that all data were comparable across the study period. For Northern pike this meant removing individuals that were not detected for the entire 12-month study period, resulting in the exclusion of 4 fish (Table 2). For pikeperch, because of a lower number of tagged individuals, the removal of fish without a full 12 months data would have excluded 50% of the sample. Consequently, pikeperch ID 43266 was removed, as this individual was last detected on a receiver on the 25 April 2018 and so would not span the entire spawning period for pikeperch. The analysis of movement for the remaining 7 individuals was completed only for months when all individuals were present (to 04 July 2018; Table 2). Then, a mixed effects logistic regression model tested the binary response of daily movement (as a detected daily movement versus no detected daily movement) against daily mean water temperature and river flow for both species, with season as a fixed effect and individual as a random effect in the model. Individual was included as a random effect to mitigate autocorrelation from repeated measures from the same individual (Harrison et al., 2018). Water temperature and flow were entered as quadratic terms to account for potential non-linear relationships and data were scaled for continuous variables before analyses. As season was defined according to the Northern meteorological season then the pikeperch model did not have a complete dataset for summer and autumn.
As range testing revealed that the receiver located within the boat marina could only detect acoustic transmitters within the marina and not the river, then the proportion of time spent in the marina by each fish was assessed as its ‘off-channel residency’ (cf. Objective iii). This was calculated as the total time of individual residency events within the marina for both the length of the entire study period (i.e. the time between first and last detection for each-individual) and weekly. A GLM (Poisson distribution and negative binomial distribution where over-dispersion was apparent) tested the influence of river temperature and discharge (and their interaction) on the presence of each species in the main river channel (as the number of fish present per day and the total number of detections per species per day).
Daily timings of dawn, day, dusk and night were retrieved for each day in the study period, obtained using the package ‘maptools’ with civil twilight definitions, with the package providing the times of day of these diel periods across the year (and thus the seasonal differences) (Bivand & Lewin-Koh, 2019). Diel periods were defined as dawn, day, dusk and night. A fish movement during each of these diel periods was defined when a fish was initially detected on a receiver and then detected on the next receiver upstream or downstream later in the same diel period. As the receivers did not overlap in their detection fields, fish could not be detected simultaneously on adjacent receivers. Therefore, the detection of a fish on two receivers at different times within the same diel period constituted a movement in that period. Detections of a fish on a single receiver in one diel period and its subsequent detection on an adjacent receiver in the next diel period were not counted as movements here. Diel movements between the marina and the main river—in either direction—were only counted when the fish was detected as moving between both receivers in the same diel period. Once the number of diel movements had been determined for the dawn, day, dusk and night period of each day, they were standardised to the number of movements per hour for both species. These measurements were calculated for the entire 12-month study period, by season and by month for individuals with 12 months of data only. A Kruskal–Wallis rank sum test then tested the overall differences in number of movements over the 12-month study period across diel periods, and a Chi-squared (χ2) contingency table analysis was used to test for an association between dawn, day, dusk and night movements with season (cf. Objective i). Monthly movement within each diel period was used for graphical purposes only. Tests were completed for Northern pike and pikeperch separately.
Analyses and graphical outputs were completed in R (Version 4.2.3; R Core Team, 2023). Logistic regression and generalised linear models were analysed for Northern pike and pikeperch separately and were completed using the package lme4 (Bates et al., 2014). Where error is expressed around the mean, it represents the 95% confidence intervals, unless otherwise stated. All fixed factors used in GLM models were predetermined, eliminating the need for a model selection process based on AIC values.
Results
Tag detections and general movements of tagged fish
Across the tracking period, there was a greater total number of detections on receivers around Upton-Upon-Severn (the location of fish capture and tagging) than elsewhere in the array (Table 1), with most detected movements of both species being in this area (Fig. 2). All pikeperch and all but one Northern pike showed some level of residency within the off-channel habitat of the boat marina (Table 2), with 78% of all Northern pike detections and 63% of all pikeperch detections occurring on the receiver located there. Northern pike had a significantly higher mean off-channel residency index across the 12-month tracking period than pikeperch (0.29 ± 0.06 vs. 0.10 ± 0.06; t test: t = 3.88, P < 0.01; Table 2).
Northern pike detections beyond the Upton-Upon-Severn receivers included three fish detected 8.8 km downstream of the marina between 08 March 2018 and 16 March 2018 that returned upstream between 10 March 2018 and 11 April 2018 (Fig. 2). Northern pike detections upstream of Upton-Upon-Severn included two Northern pike that moved 16.5 km to the upper limit of the array, where they were detected between 28 January 2018 and 15 April 2018 (Fig. 2). Only one Northern pike was detected within the River Teme tributary throughout the study period. There were three tagged pikeperch detected beyond the Upton-Upon-Severn receivers, with one fish detected at the upper limit of the array on 15 separate dates between 03 August 2018 and 17 September 2018 (Fig. 2).
Mean daily distances, river versus off-channel residency, and diel movements
Differences in mean daily distance moved between species across the tracking period were not significant (Northern pike: 245 ± 108 m; pikeperch: 247 ± 217 m; Mann–Whitney U = 57, P = 0.97). There was a significant difference in mean total daily movement across seasons for both Northern pike (GLM; F = 20.57, df = 3, P < 0.01) and pikeperch (GLM; F = 14.59, df = 3, P < 0.01), with both species showing higher mean total daily movements in spring (Table 3, Fig. 3). Daily movement behaviour (binary response of detected movement vs. no detected movement) for both species revealed that up to a threshold of 15 °C, higher temperatures increased the probability of a detected movement in both species, while at temperatures > 15 °C, this probability was significantly reduced (Table 4, Fig. 4). There was also an increase in the probability of a predicted movement with increasing fork length for Northern pike, but not pikeperch (Table 4, Fig. 4). Seasonally, the probability of a detected movement for Northern pike in winter was significantly reduced (P < 0.05; Table 4), but with no significant seasonal differences in the probability of a detected movement in pikeperch (P > 0.05; Table 4) (See Fig. 5).
Predicted probabilities of daily movement with mean daily water temperature (°C) for a Northern pike and c pikeperch, and with fish length (mm) for b Northern pike and d pikeperch for the lower River Severn during the 12-month study period as predicted from a mixed effects logistic regression where the binary response was daily movement (detected movement vs. no detected movement). Shaded regions represent the 95% confidence intervals
a Weekly mean water temperature (°C) (open circle, dashed line) and water flow (m3/s) (closed circle, solid line) with 95% confidence intervals across the study period; and b weekly mean off-channel residency index for Northern pike (closed circle, solid line) and pikeperch (open circle, dashed line) for the lower River Severn during the 12-month study period
Detections of both species in the main river channel were significantly reduced in periods of lower flows, but with Northern pike detections higher in relatively warm temperatures and pikeperch in relatively cooler temperatures (Table 5). Northern pike showed no seasonal differences in weekly off-channel residency while pikeperch weekly off-channel residency was significantly reduced in summer and autumn compared to spring (P > 0.05; Table 5), with 89% of their detections in the marina occurring between December and April. The diel movements of both species revealed no significant differences in the number of movements per hour within each dawn, day, dusk and night period over the 12 months (Northern pike: Kruskal–Wallis χ2 = 7.26, P = 0.06; pikeperch: Kruskal–Wallis χ2 = 6.27, P = 0.09; Fig. 6). However, when analysed seasonally, Northern pike movement within each diel period were not equally distributed across seasons (χ2 = 24.46, df = 9, P < 0.01; Fig. 6), with increased movements during dawn and dusk in summer and autumn (Fig. 6). This was not apparent in pikeperch, where movements at dawn, day, dusk and night were equally distributed across seasons (χ2 = 6.27, df = 9, P = 0.44) (Fig. 6).
Number of detected movements per hour for a Northern pike and b pikeperch for dawn, day, dusk and night across the tracking period, seasonally, and for each month of the study. Boxplots show the first, median and third quartiles and 95% confidence intervals are shown by the whiskers, and outliers as filled circles
Discussion
The movement and behaviour of the native Northern pike and non-native pikeperch was characterised by within species variability in spatial and temporal habitat use, but with an overall increase in the total daily distance moved during spring for both species. The relationship of their movements with temperature were strongly non-linear, where an increase in temperatures up to 15 °C resulted in more movements of both species, but with there being fewer movements as temperatures then increased above this level. These relationships between movement patterns and temperature were consistent with the prediction. Given these temperature responses, it is considered that neither species is likely to benefit from increased temperatures resulting from climate change (Ruiz-Navarro et al., 2016). Northern pike movements also increased as body length increased, with this consistent with Dhellemmes et al. (2023) who revealed that larger pike consistency use larger spatial areas than smaller pike. Relatively long-distance movements were detected in Northern pike in spring, assumed to be associated with movements to spawning areas, but with these movements not detected for pikeperch, with these species-specific differences not consistent with the prediction. There was some contrasting seasonal variation between species in the use of the off-channel boat marina, with this also contrary to prediction. This marina was an important Northern pike habitat all year round, suggesting it provided an important foraging and refuge location, but was only an important habitat for pikeperch in winter and spring, suggesting it was possibly an important spawning area. There were decreased numbers of detections of both species in the main river channel during elevated flows, suggesting their ability to access the marina during episodic high flow periods was never comprised. The main periods when these species overlapped in habitat use was in the off-channel marina in winter and spring. Although it is highly likely both species consumed similar prey species in these periods, the extent to which this represented competition was unclear, given that sampling of prey fish populations in the marina indicate high abundance (E. Nolan, unpublished data). There was no evidence of any intra-guild predation, although the tagged species were both of sizes capable of consuming juvenile individuals of both species, and despite increased intra-guild predation being apparent in other studies following introduction of a new piscivore (Schulze et al., 2006).
Although these results indicate that the study was able to complete its objectives and test its predictions, it is acknowledged that a potential issue was the limited sample size of pikeperch (n = 8), with this number of tagged fish potentially not capturing the full extent of individual variability present in their population. Nevertheless, this sample size was similar to the number of tracked pikeperch used by Poulet et al. (2005a) (n = 10 tracked fish, maximum tracked per month = 5) and Koed et al. (2002) (n = 13, of which 8 were tracked throughout the study). Accordingly, it is argued that the results here for pikeperch are comparable to these previous studies because of similar sample sizes.
Spatial utilisation of the river for both Northern pike and pikeperch across the 12-month study period was focused to an area of less than 5 km upstream and downstream of the sampling location. Patterns of movement could be characterised as long stationary periods followed by movements that were either infrequent or frequent but across short distances only. In Northern pike, long distance movements of greater than 5 km tended to be abrupt and primarily occurred during the spawning season. Northern pike are generally considered sedentary because of their sit-and-wait predator ambush behaviour, but they can shift positions regularly to enhance predation success and avoid conspecifics (Nilsson et al., 2006; Knight et al., 2008). Indeed, Northern pike populations have been suggested as comprising a range of different behavioural types, including individuals that are primarily sedentary and those that move relatively frequently (Vehanen et al., 2006; Sandlund et al., 2016), while others have suggested that Northern pike are characterised by a continuum of behavioural types (Masters et al., 2005) or across three broad behavioural groups, including individuals that stay in restricted areas, those that move between favoured areas, and those that are more opportunistic and exploratory in their resource use (Jepsen et al., 2001; Kobler et al., 2009). Although the weirs in the lower reaches of the River Severn, including the study area, have been fitted with fish passes in recent years to increase longitudinal connectivity (Antognazza et al. 2022), there is no evidence to suggest that this will benefit the Northern pike and pikeperch populations in the study area, as fish rarely moved upstream into the study area from downstream of the modified weirs, as indicated by only 468 detections on the receiver at Diglis (in the vicinity of the most downstream fish pass) versus over 1.3 M detections on a receiver in the middle of the study area. While the upstream movements of pike in spring were assumed to be for spawning, it was not clear where they actually spawned, but the absence of off-river refugia in those areas mean it must have been in littoral areas, presumably over emerging macrophytes (Craig, 2008). For pikeperch, alternative spawning areas to the boat marina could not be easily identified from the acoustic telemetry data and thus if the marina was not present or inaccessible, it remains unclear as to where this species would spawn in the river. It is also unknown where they spawned prior to the river being impounded and channelised.
For pikeperch, individual variability in resource use has rarely been reported, with synchronised movements associated with spawning or foraging activities more common (Koed, 2000; Koed et al., 2002). However, pikeperch movement from a mark-recapture study on the Great Ouse River catchment in Eastern England in the 1980s (Fickling & Lee, 1985), suggested they might comprise of two distinct behavioural types, active and sedentary. Distinct differences in movement patterns within each species were apparent in this study, but the combination of a relatively limited sample size and the spatial distribution of receivers that prevented the measurement of fine-scale movements meant that it was not possible to categorise the tagged fish into distinct behavioural groupings.
The timing of onset of spawning for Northern pike in rivers in England tends to be in March or April (Mann, 1976; Masters et al., 2005), with optimum spawning temperatures of between 6 and 14 °C (Frost & Kipling, 1967). In mature individuals, movements of Northern pike increase around the spawning period (Baktoft et al., 2012), with migratory spawning movements in spring accounting for differences in seasonal movement patterns (Ovidio & Philippart, 2005; Koed et al., 2006; Pauwels et al., 2014). Indeed, evidence is rare of seasonal differences in movement patterns beyond their migratory spawning behaviour (Kobler et al., 2008). Temperature also usually triggers spawning migration in both sexes (Pauwels et al., 2014), and although Northern pike often show homing to natal spawning sites (Engstedt et al., 2014; Sandlund et al., 2016), this might not be evident across the entire population (Vehanen et al., 2006). It is possible, therefore, that Northern pike making the relatively long-distance movements in spring in this study were moving to specific spawning locations that were suitable for spawning, such as over vegetation in shallow water (Casselman & Lewis, 1996), especially given that these areas were limited given the river engineering and impounded environment. However, a short-coming of acoustic telemetry is that while it generates movement data, it does not currently indicate the activity of the fish in the different habitats. Accordingly, while Northern pike movements in spring have been assumed to relate to their spawning activities, this is an assumption that could not be tested further.
For pikeperch, spawning occurs between 8 and 16 °C and, as it is expected to occur predictably across latitudes, tends to commence in mid-April in central England in most years (Lappalainen et al., 2003). Increased movements of pikeperch in spring is also associated with reproductive behaviour and spawning migrations (Lappalainen et al., 2003), but this can vary between sexes, with male movements being reduced because of their nest guarding behaviours, where males can remain in position for between 2 and 6 weeks (Jepsen et al., 1999; Poulet et al., 2005b). However, it is also common to see seasonal peaks in pikeperch movements that are unrelated to spawning migrations, such as peaks in autumn, summer or winter movements that are linked to feeding migrations, particularly in systems where prey resources are limited (Jepsen et al., 1999; Koed, 2000; Horkỳ et al., 2006). However, the pikeperch movement patterns were relatively consistent across the study, with no apparent peaks of movement. As pikeperch prefer to spawn in still or slow flowing waters (Lappalainen et al., 2003), including in low velocity areas in rivers (Koed et al., 2002), their higher residency index in the marina in spring was likely through them using the area for spawning. Again, as the acoustic telemetry data could not indicate the activity of the fish in the different locations where they were detected then this spawning in the marina can only be assumed. However, exposed pikeperch nests were observed in the marina during a receding flood in April 2018, indicating that some pikeperch did indeed spawn there.
Temperature is an important factor in explaining increased movement for both Northern pike and pikeperch. For example, relative changes in temperature can stimulate movement in pikeperch during the pre-spawning and spawning season, with the extent of temperature changes potentially a more important predictor of movement than actual water temperature (Saulamo & Lappalainen, 2007). However, the relationships of the movement of both species with temperature appear to be strongly nonlinear. In pikeperch, decreased movements with reducing temperature, and elevated movements with increasing temperature, have been detected (Jepsen et al., 1999). In Northern pike, elevated winter temperatures of up to 5 °C can also trigger increased movements (Jepsen et al., 2001; Koed et al., 2006), and with both very high and low temperatures decreasing overall movement rates (Kobler et al., 2008). Northern pike in a Finnish river decreased their movements when water temperatures exceeded 20 °C (Vehanen et al., 2006). The tagged fishes in the River Severn thus had movement patterns that were affected by temperature in a similar non-linear manner to these studies.
On a daily basis, many piscivorous fishes have peaks in their movements at dawn and dusk because of the potential for higher foraging success during these periods (Helfman, 1986). Here, Northern pike had only increased their movements during twilight periods in summer and autumn, with pikeperch having no similar peaks in movements. For Northern pike, these results were similar to those of Kobler et al. (2008), who suggested increased movement in summer twilight periods was a short-term behavioural response to maximise food intake during this period. For pikeperch, a measured increase in their movements in twilight periods has also been found (Horký et al., 2008), but other factors such as water temperature, individual behaviour and sex also influencing their diel movement patterns (Poulet et al., 2005a).
These results suggested that whilst there was some intra-specific variation in the movement patterns and habitat use of both Northern pike and pikeperch in the river, there were also some important inter-specific differences. In particular, the boat marina—the only off-channel macro-habitat available to the fish—was important to Northern pike all year round, where it was assumed it provided an important foraging habitat and, potentially, a key spawning area. In contrast, pikeperch primarily used this off-channel refuge in winter and spring, with it again likely to provide an important spawning area. However, the actual activity of both species could not be measured directly by acoustic telemetry and hence why activities such as spawning have to be assumed. These results suggest that the increased provision of off-channel refugia in this river section would provide both species with increased access to important functional habitats, coupled with their provision of new nursery areas for larval and juvenile fish more generally, given the importance of such refugia for supporting fish recruitment success in temperate lowland rivers (Nunn et al., 2007b; Bolland et al., 2012, 2015). Increasing this habitat provision could thus result in the increased abundance of both piscivorous species, which would be considered as ecologically beneficial for Northern pike. Pikeperch, however, are non-native to the river, albeit they are now established and the feasibility of their eradication is considered as minimal (Britton et al., 2011). Pikeperch increased abundance is thus potentially detrimental to the ecological status of the river. Notwithstanding, the recreational fishery they support is increasing in popularity (Hickley & Chare, 2004) with catch-and-release practices used despite these being contrary to extant legislation (Nolan et al., 2019b). As such, increased pikeperch abundance in this river section would potentially support an increase in the socio-economic value of the fishery.
Conclusion
These results provided important insights into the movements of these co-existing native and non-native piscivorous fishes and can assist the management of this temperate lowland river and the fishery it supports. Both species used the limited off-channel habitat at key times of the year, with Northern pike using it all year round, and thus this outcome can be applied to the consideration of increasing the provision of off-channel habitats, given their highly limited availability in this lowland and highly fragmented river.
Data availability
Data are available from the corresponding author on reasonable request.
References
Antognazza, C. M., S. J. Sabatino, R. J. Britton, R. J. Hillman, M. Aprahamian, E. A. Hardouin & D. Andreou, 2022. Hybridization and genetic population structure of Alosa population in the United Kingdom. Journal of Fish Biology 101: 408–413.
Baktoft, H., K. Aarestrup, S. Berg, M. Boel, L. Jacobsen, N. Jepsen & C. Skov, 2012. Seasonal and diel effects on the activity of northern pike studied by high-resolution positional telemetry: seasonal and diel activity of pike. Ecology of Freshwater Fish 21: 386–394. https://doi.org/10.1111/j.1600-0633.2012.00558.x.
Bates, D., M. Mächler, B, Bolker & S. Walker, 2014. Fitting linear mixed-effects models using lme4. ArXiv Preprint ArXiv:1406.5823.
Bivand, R. & N. Lewin-Koh, 2019. Maptools: Tools for Handling Spatial Objects. R package (Version 0.9–5). Retrieved from https://CRAN.R-project.org/package=maptools
Bolland, J. D., A. D. Nunn, M. C. Lucas & I. G. Cowx, 2012. The importance of variable lateral connectivity between artificial floodplain waterbodies and river channels. River Research and Applications 28: 1189–1199.
Bolland, J. D., A. D. Nunn, M. C. Lucas & I. G. Cowx, 2015. The habitat use of young-of-the-year fishes during and after floods of varying timing and magnitude in a constrained lowland river. Ecological Engineering 75: 434–440.
Britton, J. R., R. E. Gozlan & G. H. Copp, 2011. Managing non-native fish in the environment. Fish and Fisheries 12: 256–274.
Brookes, A., K. J. Gregory & F. H. Dawson, 1983. An assessment of river channelization in England and Wales. Science of the Total Environment 27: 97–111.
Burnett, M. J., C. Hanzen, A. Whitehead, G. C. O’Brien & C. T. Downs, 2023. The lesser of two evils: the role of an instream barrier to control the distribution and invasive potential of non-native fish species in the Mooi River, South Africa. Marine and Freshwater Research 74: 1211–1223.
Campbell, H. A., M. E. Watts, R. G. Dwyer & C. E. Franklin, 2012. V-Track: software for analysing and visualising animal movement from acoustic telemetry detections. Marine and Freshwater Research 63: 815–820. https://doi.org/10.1071/MF12194.
Casselman, J. M. & C. A. Lewis, 1996. Habitat requirements of northern pike (Esox lucius). Canadian Journal of Fisheries and Aquatic Sciences 53: 161–174. https://doi.org/10.1139/f96-019.
Chevallier, E., G. P. Denys, R. Marlot, M. Duntze, A. Mierral, A. Fasquel, M. Dhainaut & J. Boucault, 2023. Behaviour of two predator fishes Esox lucius Linnaeus, 1758 and Silurus glanis Linnaeus, 1758 during two successive floods in the French Aisne River. Cybium: Revue Internationale d’Ichtyologie, 47: 79–99.
Clavero, M., F. Blanco-Garrido & J. Prenda, 2004. Fish fauna in Iberian Mediterranean river basins: biodiversity, introduced species and damming impacts. Aquatic Conservation: Marine and Freshwater Ecosystems 14: 575–585. https://doi.org/10.1002/aqc.636.
Cooke, S. J., C. M. Bunt, J. F. Schreer & D. H. Wahl, 2001. Comparison of several techniques for mobility and activity estimates of smallmouth bass in lentic environments. Journal of Fish Biology 58: 573–587. https://doi.org/10.1111/j.1095-8649.2001.tb02273.x.
Craig, J. F., 2008. A short review of pike ecology. Hydrobiologia 601: 5–16. https://doi.org/10.1007/s10750-007-9262-3.
Davies, P., J. R. Britton, A. D. Nunn, J. R. Dodd, C. Bainger, R. Velterop & J. D. Bolland, 2022. Individual movement variation in upstream-migrating sea lamprey Petromyzon marinus in a highly fragmented river. Freshwater Biology 67: 43–656.
Davies, P., J. R. Britton, D. Andreou, C. Crundwell, J. R. Dodd, O. Lepais, A. D. Nunn, S. Sabatino, R. Velterop & J. D. Bolland, 2024. Tracking repeat spawning anadromous fish migrations over multiple years in a fragmented river suggests philopatry and sex-linked variation in space use. Aquatic Sciences 86: 34.
Death, R. G., I. C. Fuller & M. G. Macklin, 2015. Resetting the river template: the potential for climate-related extreme floods to transform river geomorphology and ecology. Freshwater Biology 60: 2477–2496.
Dhellemmes, F., E. Aspillaga, T. Rittweg, J. Alós, P. Möller & R. Arlinghaus, 2023. Body size scaling of space use in coastal pike (Esox lucius) in brackish lagoons of the southern Baltic Sea. Fisheries Research 260: 106560.
Diana, J. S., 1980. Diel activity pattern and swimming speeds of northern pike (Esox lucius) in Lac Ste. Anne, Alberta. Canadian Journal of Fisheries and Aquatic Sciences 37: 1454–1458. https://doi.org/10.1139/f80-187.
Eby, L. A., W. J. Roach, L. B. Crowder & J. A. Stanford, 2006. Effects of stocking-up freshwater food webs. Trends in Ecology and Evolution 21: 576–584. https://doi.org/10.1016/j.tree.2006.06.016.
Ellender, B. R. & O. L. Weyl, 2014. A review of current knowledge, risk and ecological impacts associated with non-native freshwater fish introductions in South Africa. Aquatic Invasions 9: 117–132. https://doi.org/10.3391/ai.2014.9.2.01.
Engstedt, O., R. Engkvist & P. Larsson, 2014. Elemental fingerprinting in otoliths reveals natal homing of anadromous Baltic Sea pike (Esox lucius L.). Ecology of Freshwater Fish 23: 313–321. https://doi.org/10.1111/eff.12082.
Fickling, N. J. & R. L. G. Lee, 1985. A study of the movements of the zander, Lucioperca lucioperca L., population of two lowland fisheries. Aquaculture Research 16: 377–393. https://doi.org/10.1111/j.1365-2109.1985.tb00080.x.
Frost, W. E. & C. Kipling, 1967. A Study of Reproduction, early life, weight-length relationship and growth of pike, Esox lucius L., in windermere. The Journal of Animal Ecology 36: 651. https://doi.org/10.2307/2820.
Greenberg, L. A., C. A. Paszkowski & W. M. Tonn, 1995. Effects of prey species composition and habitat structure on foraging by two functionally distinct piscivores. Oikos. https://doi.org/10.2307/3545998.
Gutmann Roberts, C., A. M. Hindes & J. R. Britton, 2019. Factors influencing individual movements and behaviours of invasive European barbel Barbus barbus in a regulated river. Hydrobiologia 830: 213–228.
Harrison, X. A., L. Donaldson, M. E. Correa-Cano, J. Evans, D. N. Fisher, C. E. Goodwin & R. Inger, 2018. A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ 6: e4794. https://doi.org/10.7717/peerj.4794.
Helfman, G. S., 1986. Fish behaviour by day, night and twilight. In Pitcher, Tony J. (ed), The Behaviour of Teleost Fishes Springer, Boston: 366–387.
Hickley, P., 1986. Invasion by zander and the management of fish stocks. Philosophical Transactions of the Royal Society of London b: Biological Sciences 314: 571–582. https://doi.org/10.1098/rstb.1986.0073.
Hickley, P. & S. Chare, 2004. Fisheries for non-native species in England and Wales: angling or the environment? Fisheries Management and Ecology 11: 203–212. https://doi.org/10.1111/j.1365-2400.2004.00395.x.
Horkỳ, P., O. Slavík, L. Bartoš, J. Kolářová & T. Randák, 2006. The effect of the moon phase and seasonality on the behaviour of pikeperch in the Elbe River. Folia Zoologia 55: 411–417.
Horký, P., O. Slavík & L. Bartoš, 2008. A telemetry study on the diurnal distribution and activity of adult pikeperch, Sander lucioperca (L.), in a riverine environment. Hydrobiologia 614: 151–157. https://doi.org/10.1007/s10750-008-9503-0.
IHBC, 2021. https://www.designingbuildings.co.uk/wiki/The_Severn_navigation
Jepsen, N., A. Koed & F. Økland, 1999. The movements of pikeperch in a shallow reservoir. Journal of Fish Biology 54: 1083–1093. https://doi.org/10.1111/j.1095-8649.1999.tb00859.x.
Jepsen, N., S. Pedersen & E. Thorstad, 2000. Behavioural interactions between prey (trout smolts) and predators (pike and pikeperch) in an impounded river. Regulated Rivers Research and Management 16: 189–198.
Jepsen, N., S. Beck, C. Skov & A. Koed, 2001. Behavior of pike (Esox lucius L.)> 50 cm in a turbid reservoir and in a clearwater lake. Ecology of Freshwater Fish 10: 26–34. https://doi.org/10.1034/j.1600-0633.2001.100104.x.
Johnson, P. T., J. D. Olden & M. J. Vander Zanden, 2008. Dam invaders: impoundments facilitate biological invasions into freshwaters. Frontiers in Ecology and the Environment 6: 357–363. https://doi.org/10.1890/070156.
Knight, C. M., R. E. Gozlan & M. C. Lucas, 2008. Can seasonal home-range size in pike Esox lucius predict excursion distance? Journal of Fish Biology 73: 1058–1064. https://doi.org/10.1111/j.1095-8649.2008.01989.x.
Kobler, A., T. Klefoth, C. Wolter, F. Fredrich & R. Arlinghaus, 2008. Contrasting pike (Esox lucius L.) movement and habitat choice between summer and winter in a small lake. Hydrobiologia 601: 17. https://doi.org/10.1007/s10750-007-9263-2.
Kobler, A., T. Klefoth, T. Mehner & R. Arlinghaus, 2009. Coexistence of behavioural types in an aquatic top predator: a response to resource limitation? Oecologia 161: 837–847. https://doi.org/10.1007/s00442-009-1415-9.
Koed, A., 2000. Annual movement and migration of adult pikeperch in a lowland river. Journal of Fish Biology 57: 1266–1279. https://doi.org/10.1006/jfbi.2000.1389.
Koed, A., K. Balleby & P. Mejlhede, 2002. Migratory behaviour of adult pikeperch (Stizostedion lucioperca) in a lowland river. Hydrobiologia 483: 175–184. https://doi.org/10.1023/A:1021331629628.
Koed, A., K. Balleby, P. Mejlhede & K. Aarestrup, 2006. Annual movement of adult pike (Esox lucius L.) in a lowland river. Ecology of Freshwater Fish 15: 191–199. https://doi.org/10.1111/j.1600-0633.2006.00136.x.
Lappalainen, J., H. Dörner & K. Wysujack, 2003. Reproduction biology of pikeperch (Sander lucioperca (L.)) – a review. Ecology of Freshwater Fish 12: 95–106. https://doi.org/10.1034/j.1600-0633.2003.00005.x.
Lenhardt, M. & P. Cakić, 2002. Seasonal reproductive cycle of pike, Esox lucius L., from the River Danube. Journal of Applied Ichthyology 18: 7–13.
Mann, R. H. K., 1976. Observations on the age, growth, reproduction and food of the pike Esox lucius (L.) in two rivers in southern England. Journal of Fish Biology 8: 179–197. https://doi.org/10.1111/j.1095-8649.1976.tb03930.x.
Masters, J. E. G., K. H., Hodder, W. R. C. Beaumont, R. E. Gozlan, A. C. Pinder, R. E. Kenward & J. S. Welton, 2005. Spatial Behaviour of Pike Esox lucius L. in the River Frome, UK. Aquatic Telemetry: Advances and Applications. 179–190. Ustica, Italy, 9–13 June 2003.: Rome, FAO/COISPA.
Nilsson, P. A., T. Hakan & C. Brönmark, 2006. Friends and foes in foraging: intraspecific interactions act on foraging-cycle stages. Behaviour 143: 733–746. https://doi.org/10.1163/156853906777791379.
Nolan, E. T., C. Gutmann Roberts & J. R. Britton, 2019a. Predicting the contributions of novel marine prey resources from angling and anadromy to the diet of a freshwater apex predator. Freshwater Biology 64: 1542–1554.
Nolan, E. T., J. R. Britton & S. Curtin, 2019b. Angler behaviors and motivations for exploiting invasive and native predatory fishes by catch-and-release: a case study on the river Severn catchment, Western England. Human Dimensions of Wildlife 24: 463–479.
Nunn, A. D., J. P. Harvey, J. R. Britton, P. A. Frear & I. G. Cowx, 2007a. Fish, climate and the Gulf Stream: the influence of abiotic factors on the recruitment success of cyprinid fishes in lowland rivers. Freshwater Biology 52: 1576–1586.
Nunn, A. D., J. P. Harvey & I. G. Cowx, 2007b. Benefits to 0+ fishes of connecting man-made waterbodies to the lower River Trent, England. River Research and Applications 23: 361–376.
Ovidio, M. & J. C. Philippart, 2005. Long range seasonal movements of northern pike (Esox lucius L.) in the barbel zone of the River Ourthe (River Meuse basin, Belgium). Aquatic Telemetry: Advances and Applications 191–202.
Pauwels, I. S., P. L. M. Goethals, J. Coeck & A. M. Mouton, 2014. Movement patterns of adult pike (Esox lucius L.) in a Belgian lowland river. Ecology of Freshwater Fish 23: 373–382. https://doi.org/10.1111/eff.12090.
Pauwels, I. S., P. L. M. Goethals, J. Coeck & A. M. Mouton, 2016. Habitat use and preference of adult pike (Esox lucius L.) in an anthropogenically impacted lowland river. Limnologica 62: 151–160. https://doi.org/10.1016/j.limno.2016.10.001.
Poulet, N., C. Arzel, S. Messad, S. Lek & C. Argillier, 2005a. Diel activity of adult pikeperch Sander lucioperca (L.) in a drainage canal in the Mediterranean basin during spring. Hydrobiologia 543: 79–90. https://doi.org/10.1007/s10750-004-6823-6.
Poulet, N., S. Lek & C. Argillier, 2005b. Pikeperch habitat use within a canal network in spring. Journal of Fish Biology 67: 1460–1474. https://doi.org/10.1111/j.0022-1112.2005.00855.x.
R Core Team, 2023. R: A language and environment for statistical computing. R Foundation for statistical Computing, Vienna, Austria.
Ruiz-Navarro, A., P. K. Gillingham & J. R. Britton, 2016. Predicting shifts in the climate space of freshwater fishes in Great Britain due to climate change. Biological Conservation 203: 33–42.
Sandlund, O. T., J. Museth & S. Øistad, 2016. Migration, growth patterns, and diet of pike (Esox lucius) in a river reservoir and its inflowing river. Fisheries Research 173: 53–60. https://doi.org/10.1016/j.fishres.2015.08.010.
Saulamo, K. & J. Lappalainen, 2007. Effects of abiotic factors on movements of pikeperch during pre-spawning and spawning season in a Baltic archipelago. Hydrobiologia 579: 271–277. https://doi.org/10.1007/s10750-006-0410-y.
Schulze, T., U. Baade, H. Dörner, R. Eckmann, S. S. Haertel-Borer, F. Hölker & T. Mehner, 2006. Response of the residential piscivorous fish community to introduction of a new predator type in a mesotrophic lake. Canadian Journal of Fisheries and Aquatic Sciences 63: 2202–2212.
Sih, A., D. I. Bolnick, B. Luttbeg, J. L. Orrock, S. D. Peacor, L. M. Pintor, E. Preisser, J. S. Rehage & J. R. Vonesh, 2010. Predator–prey naïveté, antipredator behavior, and the ecology of predator invasions. Oikos 119: 610–621. https://doi.org/10.1111/j.1600-0706.2009.18039.x.
Skov, C., S. Berg, L. Jacobsen & N. Jepsen, 2002. Habitat use and foraging success of 0+ pike (Esox lucius L.) in experimental ponds related to prey fish, water transparency and light intensity. Ecology of Freshwater Fish 11: 65–73.
Sonamzi, B., M. Burnett, R. Petersen, G. O’Brien & C.T. Downs, 2020. Assessing the effect of tagging and the vulnerability to predation in tigerfish (Hydrocynus viattus, Castelnau 1861) in a water-stressed system using telemetry methods. Koedoe: African Protected Area Conservation and Science 62: 1–12.
Turesson, H. & C. Brönmark, 2004. Foraging behaviour and capture success in perch, pikeperch and pike and the effects of prey density. Journal of Fish Biology 65: 363–375. https://doi.org/10.1111/j.0022-1112.2004.00455.x.
Vehanen, T., P. Hyvarinen, K. Johansson & T. Laaksonen, 2006. Patterns of movement of adult northern pike (Esox lucius L.) in a regulated river. Ecology of Freshwater Fish 15: 154–160. https://doi.org/10.1111/j.1600-0633.2006.00151.x.
Zingel, P. & T. Paaver, 2010. Effects of turbidity on feeding of the young-of-the-year pikeperch (Sander lucioperca) in fishponds. Aquaculture Research 41: 189–197.
Acknowledgements
The authors would like to thank the anglers of the River Severn predator study for their assistance in capturing fish by angling and to the Environment Agency, particularly the team at Calverton fish farm and Tewkesbury for their help with electric-fishing, transportation of fish and use of acoustic telemetry equipment.
Funding
ETN was funded by a studentship from Bournemouth University, the Environment Agency and the Severn Rivers Trust. AST was supported by the TÜBİTAK BİDEB (2219 Program).
Author information
Authors and Affiliations
Contributions
JRB and ETN: conceived the study, ETN, JRB, JDB, AH, CGR and PD: collected field data, ETN, CGR, JRB and AST: analysed data, ETN and JRB: wrote the manuscript, and all authors contributed to editing the manuscript. All authors approve manuscript submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Ethical approval
The ethical approval process and all regulated procedures were completed under UK Home Office licence PPL 70/8063.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guest editors: Sidinei M. Thomaz, Cécile Fauvelot, Lee B. Kats, Jonne Kotta & Fernando M. Pelicice / Aquatic Invasive Species IV
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nolan, E.T., Hindes, A.M., Bolland, J.D. et al. Movements and habitat use of native and invasive piscivorous fishes in a temperate and channelized lowland river. Hydrobiologia (2024). https://doi.org/10.1007/s10750-024-05533-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10750-024-05533-2