Abstract
Mammalian mothers flexibly invest in their offspring to maximize their lifetime fitness. Flexible maternal investment may be particularly important in large-brained species with prolonged maternal care, e.g., in great apes. We investigated the effects of socioecological factors and mother–offspring characteristics on nine maternal behaviors in wild Sumatran orangutans (Pongo abelii; N = 22 mother-offspring pairs; >11,200 hr of focal data from 2007–2022) using generalized linear mixed models. The behaviors fall under four maternal functions: locomotory support (carrying), skill acquisition support (feeding in proximity, food transfer), protective proximity maintenance (body contact and proximity initiation, following), and independence promotion (body contact and proximity termination, avoiding). Mother’s parity was not significantly associated with any maternal behavior. Mothers were more likely to show locomotory support, skill acquisition support, and protective proximity maintenance toward younger than older offspring, whereas they were more likely to promote independence in older than younger offspring. Mothers with male offspring were more likely to show skill acquisition support to their offspring than those with female offspring. With increasing food availability, skill acquisition support reduced. With increasing association size (i.e., the number of individuals within 50 m of each other), mothers were more likely to show protective proximity maintenance and less likely to promote independence. When males were present, mothers were more likely to show locomotory support to their offspring. Sumatran orangutan mothers thus flexibly adjust offspring-directed behavior in response to prevailing socioecological factors and mother–offspring characteristics. Our findings add support to the evolutionary theory that mammalian mothers flexibly invest in their offspring.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The mother–offspring relationship is of particular significance in the life of young mammals. This is especially true of large-brained altricial mammals with complex behavioral repertoires, such as cetaceans, elephants, and human and nonhuman primates, in which physical and behavioral development occurs over an extended postnatal period (Lee & Moss, 2011; Mann, 2019; Revathe et al., 2020; van Noordwijk & van Schaik, 2005). In such species, the developmental period stretches over multiple years, which means that individuals are likely to be confronted with numerous changes in their ecological and social environments. In most mammals, mothers bear the major or the sole responsibility of postnatal care (Clutton-Brock, 1991; Maestripieri, 2018; Nicolson, 1991; van Noordwijk, 2012). Mothers provide support for nutrition (through milk and sharing solid food: Brown et al., 2004; Hinde & Milligan, 2011; Johnson et al., 2010; Lee & Moss, 2011; Nair, 1989), thermoregulation (through maintaining body contact: Lubach et al., 1992; Taber & Thomas, 1982), and locomotion (through carrying and, in certain primates, bridging to cross canopy: Chappell et al., 2015; van Noordwijk & van Schaik, 2005). Furthermore, mothers actively maintain the distance between themselves and their dependents (Förster & Cords, 2002; Szabo & Duffus, 2008); facilitate their dependents’ independence acquisition (through breaking body contact: Maestripieri, 1995), and foster the formation of their social relationships (by choosing association partners and maintaining relationships with them: Fairbanks & McGuire, 1986; Murray et al., 2014). Mothers, along with other social unit members, protect their dependents against predators (Lee & Moss, 2011; Mann, 2019; Nicolson, 1991). Mothers additionally serve as role models for social learning of skill and knowledge repertoires (Inoue-Nakamura & Matsuzawa, 1997; Jaeggi et al., 2008; Lonsdorf, 2006; Schuppli et al., 2016). Consequently, in the absence of mothers and their care, offspring mortality risk increases in mammals (Lahdenperä et al., 2016; Zipple et al., 2021).
Maternal investment is defined in terms of future reproductive costs incurred by the mother for reproductive effort allocated to her current offspring (Trivers, 1972). Because maternal investment, e.g., gestation, lactation, and carrying, is time-consuming and energy-intensive, females are expected to balance the benefits and costs of investing in each of their offspring to maximize their lifetime reproductive success (Clutton-Brock, 1991; Lee et al., 1991). In several species, mothers flexibly adjust the quantity and quality of their investment in response to their and their offspring’s characteristics (e.g., parity, maternal age, offspring age and sex), as well as to their immediate ecological and social conditions (e.g., social unit size and composition), which are studied in detail in many primate species (Lee, 1987; Nicolson, 1987, 1991; van Noordwijk, 2012).
In primates, maternal age and parity often affect maternal investment, wherein offspring of younger/primiparous females have a lower body mass, face more neglect and abandonment, and consequently have reduced survival compared with those of older/multiparous females (Altmann & Alberts, 2005; Arlet et al., 2014; Fairbanks, 1996; but see Silk, 1991). Possible explanations include that younger/primiparous mothers face a trade-off between investing in their own growth versus in their offspring’s (Setchell et al., 2002); therefore, they may produce less milk than older/multiparous mothers (Hinde et al., 2009) or are less experienced and thus less skilled in rearing their offspring than older/multiparous mothers (Mitchell & Stevens, 1968).
In general, maternal investment decreases with increasing offspring age due to the offspring’s increasing ecological and social competence (Dias et al., 2018; Fairbanks, 1996; Jaeggi et al., 2008; Li et al., 2013; Mikeliban et al., 2021; Nishida & Turner, 1996). However, mothers and offspring may disagree over the length of the period of investment because of an underlying genetic conflict (parent–offspring conflict theory; Trivers, 1974). Concerning offspring sex, the Trivers-Willard hypothesis posits that in species in which body condition differentially affects the reproductive success of the sexes, mothers in good body condition should bias their investment in favor of the sex with more variable reproductive success when controlled for investment (males in polygynous species), provided mothers’ investment during dependency translates into offspring’s body condition in adulthood (Trivers & Willard, 1973). Studies on primates found inconsistent results for sex-biased maternal investment (Dettmer et al., 2016; Fairbanks, 1996; Leigh & Shea, 1995; Setchell et al., 2002; Silk, 1991; Tanaka, 1989).
Maternal duration of investment is hypothesized to have an inverse U-shaped relationship with maternal body condition in response to food availability, with mothers in average body condition investing more than mothers in poor or good body condition (Fairbanks & McGuire, 1995; Lee et al., 1991), especially in annual breeders. The quadratic relationship is explained with the rationale that mothers living in relatively poor-quality habitats may not be able to satisfy both their own and their offspring’s needs and thus might reduce the period of investment (such as through early weaning age; Lee et al., 1991) to support their future reproduction at the expense of current offspring’s survival. In contrast, mothers living in high-quality habitats are likely able to support their own and their offspring’s needs. Mothers living in high-quality habitats can reduce the period of investment in their current offspring—without decreasing the quantity of investment or increasing the risk of offspring mortality (Hauser & Fairbanks, 1988)— to ultimately speed up one’s reproductive pace.
Mothers may also adjust their investment in response to the social risk posed by sociodemographic factors, such as social unit size and composition, along with dominance structure (Maestripieri, 1994; Nicolson, 1991). For example, it is hypothesized that in species with frequent aggressive interactions among conspecifics, mothers may increase their investment by staying close to their offspring with increasing social unit size because of the perceived social risk to the offspring (Maestripieri, 1994). Moreover, when there is a risk of infanticide by males, mothers may stay closer to their offspring in the presence of unfamiliar/unrelated males than in their absence in the social unit (Fairbanks & McGuire, 1987; Scott et al., 2019, 2023).
The effects of mother-offspring characteristics, ecological, and sociodemographic variables on maternal investment have been studied in various terrestrial primates (e.g., vervet monkeys (Cercopithecus aethiops sabaeus): Fairbanks & McGuire, 1995; rhesus macaques (Macaca mulatta): Maestripieri, 2001; baboons (Papio cynocephalus): Nguyen et al., 2012; mandrills (Mandrillus sphinx): Setchell et al., 2002). However, most of these studies have looked at one or few factors influencing one or few forms of maternal investment rather than comprehensively investigating the effects of several factors on multiple forms of investment. Simultaneously testing for the effects of multiple known predictors of investment is important, as effects of certain predictors may only become apparent when controlling for the effects of other predictors (e.g., controlling for offspring age). Furthermore, studies on maternal investment in arboreal primates are comparatively rare (but see Arlet et al., 2014; Dias et al., 2018; Mikeliban et al., 2021; van Noordwijk & van Schaik, 2005). As arboreal primates face different ecological challenges compared with terrestrial primates (e.g., learning to cross gaps in the canopy without risking a fall), some arboreal primates show specialized maternal behaviors, such as bridging. Furthermore, the arboreal lifestyle entails high energetic costs, especially for large primates (e.g., climbing costs: Hanna & Schmitt, 2011; Thorpe & Crompton 2009), which may lead to constraints on maternal investment. Thus, the obvious differences in habitat, predation risk, diet composition, energetic constraints, and social characteristics between terrestrial and arboreal primates must be considered to understand the variation found in their patterns of maternal care.
Among arboreal primates, orangutans (Pongo spp.) are particularly interesting to study maternal care, because they have the slowest life history, including the longest dependency period and interbirth intervals of all nonhuman primates (van Adrichem et al., 2006; van Noordwijk et al., 2018). Females give birth to a single offspring at a time and provide care for 6–8 years (van Noordwijk et al., 2009). Mothers invest heavily in each offspring through prolonged nursing, carrying, and nest-sharing (van Noordwijk & van Schaik, 2005). Furthermore, the orangutans’ predominantly arboreal lifestyle requires maternal care in the form of carrying and bridging to cross gaps in the canopy. Males disperse from their natal area and do not provide paternal care (Arora et al., 2012; Morrogh-Bernard et al., 2011; Nietlisbach et al., 2012; van Noordwijk et al., 2023). This, along with a prolonged period of lactation relative to gestation length (van Schaik, 2000), sexual size dimorphism (Utami Atmoko et al., 2009), and potential for contest competition between males for mating opportunities (Kunz et al., 2023; Spillmann et al., 2017; van Schaik & van Hooff, 1996), puts orangutans at a high risk of infanticide by males, even though there are no confirmed cases (Beaudrot et al., 2009; Knott et al., 2019; van Schaik, 2000).
Most orangutan habitats are characterized by low and fluctuating food availability (Marshall et al., 2009). Even though fruits are the orangutans’ main energy source, individuals survive on fallback foods during periods of low fruit availability (Morrogh-Bernard et al., 2009). Furthermore, the orangutans’ arboreal lifestyle and large body and brain size result in high energetic requirements. As a likely response to the energetic constraints resulting from a combination of living in low food availability habitats and having high energetic requirements, orangutans have developed behavioral and physiological adaptations to deal with periods of low food availability (e.g., reduced movement when food is scarce and exceptionally low basal metabolic rate (BMR); Morrogh-Bernard et al., 2009; Pontzer et al., 2010; Vogel et al., 2017).
As a result of low forest productivity, which results in feeding competition, and the species’ energetic constraints, associations are costly to maintain for orangutans (Kunz et al., 2021; van Schaik, 1999). They are generally semisolitary (Setia et al., 2009), but mothers with dependent offspring spend up to 50% of their time in association with others (depending on population; Roth et al., 2020; van Noordwijk et al., 2009). Orangutans are large-brained and have broad behavioral repertoires (van Schaik, 2013). They occupy a complex foraging niche with many food items requiring pre-ingestive processing, including tool use in some populations (van Schaik et al., 1996). Dependent offspring begin to consistently feed on solid food from ~1 year of age and acquire skill and knowledge repertoires via a mix of individual and social learning, mainly by closely observing their mothers and soliciting food from her, followed by independent practice (Jaeggi et al., 2010; Mikeliban et al., 2021; Schuppli et al., 2016). Because male orangutans start to disperse during adolescence, they have less time to learn from their mothers than their female peers who stay in their natal area and regularly meet with their mothers throughout their lives (Ashbury et al., 2020; Ehmann et al., 2021).
We investigated the effects of mother–offspring characteristics (mother’s parity and offspring age and sex), an ecological factor (prevailing fruit availability), and social factors (association size, i.e., the number of individuals within 50 m of each other, and the presence of males) on nine maternal behaviors in Sumatran orangutans (Pongo abelii). These behaviors fall under four potential domains of maternal investment, which we refer to as maternal functions: locomotory support (carrying offspring while moving; van Noordwijk, 2012); skill acquisition support (feeding in close proximity, food transfer; Mikeliban et al., 2021; Jaeggi et al., 2008, 2010; Schuppli et al., 2016); protective proximity maintenance (initiating body contact or close proximity with offspring, following offspring; Maestripieri, 2001; Nicolson, 1991); and independence promotion (terminating body contact or close proximity with offspring, avoiding offspring; Maestripieri, 1995; Nicolson, 1991). Following the evolutionary theory of flexible maternal investment (Clutton-Brock, 1991), and based on previous findings on orangutans and other primates detailed above, we hypothesize that Sumatran orangutan mothers will flexibly adjust their investment in these maternal functions in response to mother–offspring characteristics and socioecological factors (given the trade-offs that these factors impose) to increase their lifetime fitness by increasing reproductive pace (via increasing the speed at which their offspring develop), offspring survival, or offspring reproduction while ensuring their own survival.
Predictions
-
1.
Mother’s parity. We predict maternal investment to vary as a function of maternal experience in that primiparous mothers would show more protective proximity maintenance and locomotory and skill acquisition support but less independence promotion toward their offspring than multiparous mothers to compensate for the lack of direct maternal experience.
-
2.
Offspring’s age. As the developing offspring acquires subsistence skills and becomes increasingly competent with age, we predict mothers to show less protective proximity maintenance and provide less locomotory and skill acquisition support but increase offspring independence promotion.
-
3.
Offspring’s sex. We predict mothers to generally invest more in male than female offspring, because males have more variable reproductive success than females.
-
4.
Food availability. As orangutans face significant energetic constraints, we predict maternal investment, especially potentially energy-intensive forms of investment, such as locomotory and skill acquisition support (see discussion), to reduce during periods of extremely low food availability.
-
5.
Association size. As a result of the combination of low forest productivity, general energetic constraints, and the energetic requirements of offspring care, we predict associations at certain times (e.g., during large associations) to be costly for females with dependent offspring. We, therefore, predict mothers to reduce their investment in especially potentially energy-intensive forms of investment, such as locomotory and skill acquisition support, when association sizes are large.
-
6.
Male presence. Because of the potentially high infanticide risk, we predict that mothers will show counterstrategies to infanticide by males in that mothers will increase protective proximity maintenance and reduce independence promotion in the presence of associating males.
Methods
Study Site and Data Collection
For this study, we used data collected on wild Sumatran orangutans from 2007 to 2022 at the Suaq Balimbing research area in the Gunung Leuser National Park (3°42 N, 97°26 E) in South Aceh, Indonesia. Multiple experienced researchers and field assistants collected the data, whose concordance index had reached more than 85% during interobserver reliability testing (computed using simultaneous follow data collected by multiple observers on the same focal individual without any information exchange between the observers). During focal follows, we collected the data through scan sampling (Altmann, 1974) at 2-min intervals by following mother–offspring pairs, when possible, from their morning to evening nest (see ab.mpg.de/571325/standarddatacollectionrules_suaq_detailed_jan204.pdf for data collection protocols). At each scan, we noted down the behavior of the focal animal, the distance to all association partners (measured in classes: contact (0 m), <2 m, 2–5 m, 5–10 m, 10–50 m, no association (>50 m)), the individual responsible for a change in distance class, and whether the offspring was being carried by its mother. We considered individuals to be in association if they were within 50 m of one another during a scan. Because of fission–fusion dynamics (van Schaik, 1999), such associations may last for only a few minutes to several days. We used a total of 1220 focal follows (including 839 follows on mothers and 381 follows on dependent offspring) for a total duration of 11,271.5 follow hours on 22 mother–offspring pairs (Table S1). Each follow represents data on a single focal (minimum duration: 30 min; maximum duration: 13 hr). Because “food transfer” (Table S2) is a relatively rare behavior, aside from scan data, we also used data collected ad libitum to increase the sample size. Because we were interested only in the probability of successful food transfer when the offspring begs, and not in the rate of occurrence of this behavior, including ad libitum data is not expected to affect the observed probability of food transfer. We used data on offspring from birth until 8 years of age, as at Suaq, the period of constant mother–offspring association ends at around 8 years, which marks the end of the offspring dependency period (van Noordwijk et al., 2018). As we followed the orangutans opportunistically, depending on encountering them in the study area, sample sizes vary within mother–offspring pairs across behaviors (depending on whether a behavior was recorded during the follow) and between mother-offspring pairs. Furthermore, the offspring age period during which mothers were sampled varied among the mothers (Table S2).
Measurement of Maternal Investment
We calculated maternal investment as the probability that mothers showed a maternal behavior (i.e., proportion of scans during which the focal mother showed a behavior) (Table S2), representing four maternal functions: locomotory support; skill acquisition support; protective proximity maintenance; and independence promotion. To minimize the influence of offspring behavior on the expression of the maternal investment in different behaviors, where possible, we accounted for the opportunities that the mothers had available to show the respective behaviors, by including them in the denominator of the calculation used to get the probability that the mother showed a behavior (Table S2). For example, because mothers can follow their offspring only when their offspring moves away from them, we calculated the probability that a mother followed her offspring as the number of scans during which the mother followed her offspring divided by the number of scans during which the offspring moved away from its mother (Table S2), instead of using the total number of scans as the denominator. The total number of follows and observation hours for the different maternal behaviors included in our analyses are summarized in Fig. S1. In orangutans, lactation increases the mother’s energy needs by 25% of baseline level (van Noordwijk et al., 2013a, 2013b). Despite nursing being a significant form of maternal investment in orangutans (van Noordwijk et al., 2013a, 2013b), we did not examine suckling behavior in the current study, because in this orangutan population nursing cannot be accurately recorded, which often happens in day or night nests where visibility of the dependent offspring is low.
Predictors of Maternal Investment
We modelled the effects of mother’s parity (primiparous/multiparous), offspring age (in years, Fig. S2), and sex (female/male), prevailing fruit availability (assessed through a fruit availability index, henceforth called FAI, Fig. S3), daily average association size (i.e., the number of association partners apart from the focal individual averaged across the scans taken during a follow, Fig. S4), and any adult males in association (presence/absence). Each month, we monitored all the trees with a diameter at breast height (DBH) of >10 cm for ripe, half-ripe, and unripe fruits along two established phenology transects in the study site. These transects run North-South and West-East, through the home ranges of the frequently sampled focal individuals and comprised approximately 1000 trees (see https://www.aim.uzh.ch/en/research/orangutannetwork/psp.html). We calculated FAI as the percentage of fruit-bearing trees in the phenology plots each month, which is a measure of fruit abundance (Vogel et al., 2017). We included maternal parity instead of maternal age as a proxy for maternal experience as a predictor, because we did not know the ages of all our adult females.
Dependent offspring were, per definition, in constant association with their mothers. We followed mothers of different reproductive status (primiparous, multiparous), and we had data available on one to three dependent offspring per mother (Table S1). The number of male and female offspring, primiparous and multiparous females, and the number of follows with and without a male varied among the different behaviors. In total, there were six primiparous and 16 multiparous mothers and six female and 16 male offspring (Table S1). There was at least one adult male associating with the focal mother–offspring pair in ~42% of the 1220 follows.
Statistical Analysis
All statistical analyses were conducted in R (version 4.2.2; R Core Team, 2023). We fitted Generalized Linear Mixed Effects Models (GLMMs; Baayen, 2008) using the glmer function of the lme4 package (version 1.1.34; Bates et al., 2014). Each full model contained maternal investment in a given behavior as a response variable and all the predictors described above as fixed effects. Based on visual inspection of the raw data and the fact that certain behaviors are not expressed at certain ages (e.g., offspring do not feed on solid food when they are very young), we additionally tested for a quadratic effect of offspring age in the models for the probability of mothers feeding in close proximity with their offspring and the probability of food transfer. We did so by comparing the model with only a linear effect of offspring age to a model with both a linear and quadratic effect of offspring age, while keeping all other fixed effects and the random effects the same between the two models, using likelihood ratio tests (LRT; Dobson & Barnett, 2018) as implemented in the anova function in R. If the quadratic model outperformed the original full model, the quadratic model was then considered as the full model; otherwise the model with only the linear age effect was considered as the full model. Based on visual inspection, we included only a linear effect of FAI.
Because there were multiple data points for each mother and because six of the 15 mothers were sampled with more than one dependent offspring, mother identity was included as a random factor to avoid pseudo-replication, and offspring identity was nested within mother identity to account for potential differences in maternal investment in behaviors by mothers towards their different offspring, respectively. Because maternal behaviors were calculated in the form of discrete probabilities (numerator and denominator were number of scans; Table S2), we used a logistic model (binomial error distribution and logit link function; McCullagh and Nelder, 1989) to analyze the behaviors and added follow ID as a random factor (we elaborate on the rationale in the supplementary material). To avoid a model being overconfident regarding the precision of fixed effects estimates and to keep the type I error rate at the nominal level of 5%, we included all theoretically identifiable random slopes (Barr et al., 2013; Schielzeth & Forstmeier, 2009). Specifically, we included random slopes of offspring age, FAI, daily average association size, and male presence/absence (the latter manually dummy coded and mean centered) within mother identity and within offspring identity. We did not include correlation parameters between random slopes and intercepts in our final full models (we elaborate on the rationale in the supplementary material). Before fitting the models, we inspected whether all the quantitative predictors were roughly symmetrically distributed. We then z-transformed the quantitative predictors—offspring age, daily average association size, and FAI, for easier interpretable model estimates (Schielzeth, 2010) and to ease model convergence.
After fitting each model, we assessed normality of Best Linear Unbiased Predictors (BLUPs; Harrison et al., 2018; function written by RM), relative model complexity (i.e., the number of observations divided by the number of estimated model parameters; maximum: 46.2 follows; minimum: 14.7 follows), absence of collinearity (using the vif function of the library car (version 3.1.2; Fox and Weisberg, 2011), maximum Variance Inflation Factor in our models was 1.7; Quinn & Keough, 2002), and overdispersion (maximum dispersion parameter in our models was 0.6; function written by RM) for all the models. After fitting the full model, we tested the fit of the full model against the null model (Forstmeier & Schielzeth, 2011), which contained only the random effects, using LRT as implemented in the anova function in R. If the full model produced a significantly better fit than the null model, we tested the significance of the individual fixed effects by using the drop1 function, which drops one fixed effect at a time and compares the fit of the resulting model to that of the full model. We used a cutoff of P < 0.05 for significance. We report the odds ratio (i.e., the exponent of the estimate) for significant fixed effects, which is a measure of effect size in logistic regression (Breaugh, 2003). For a given fixed effect, an odds ratio of <1 indicates a reduction in maternal investment in a behavior, while values >1 indicate an increase in maternal investment in a behavior. We further report marginal coefficients of determination (i.e., the variance explained by the fixed effects; R2m) and conditional coefficients of determination (i.e., the variance explained by the entire model; R2c) obtained using the delta method of the r.squaredGLMM function of the MuMin package (version 1.47.5; Barton & Barton, 2015). To assess the uncertainty in the estimated individual effects, we obtained 95% confidence intervals using bootstrapping (N = 1000 bootstraps; function written by RM). To avoid unreliable estimation of the variance caused by the included random factors, we included only those offspring with data from a minimum of five calendar days (i.e., five focal follows).
Ethical Note
The research protocol of this study was approved by the Indonesian State Ministry for Research, Technology and Higher Education (BRIN RISTEK; Research Permit No.: 152/SIP/FRP/SM/V/2012) and complied with the legal requirements of Indonesia. This study was purely observational and complied with the Principles for the Ethical Treatment of Nonhuman Primates by the American Society of Primatologists (2001). The authors declare that they have no conflict of interest.
Data Availability
The datasets analyzed during the current study and the source codes for all the analyses and graphs presented in the main text and supplementary material of the manuscript are available in the Harvard Dataverse repository, https://doi.org/10.7910/DVN/8WV8XX.
Results
Extent of maternal investment varied from behavior to behavior (Figure S5). For the behavior, “feeding in close proximity,” the model with the linear and quadratic age effects performed significantly better (considered subsequently as the full model) than the model with only the linear age effect (P < 0.001). For the behavior, “food transfer,” the model with only the linear age effect performed equally well (considered subsequently as the full model) as a model with both the linear and quadratic age effects (P = 0.479). The loglikelihoods of final full models without correlation parameters between random intercepts and slopes did not differ much from the models with the correlation parameters (Table S3). The full model explained significantly more variation in maternal investment than the corresponding null model for six of the nine maternal behaviors (Table S4). Below we elaborate on the results of the individual fixed effects on these six behaviors. The full models for the behaviors, “follow,” “avoid,” and “food transfer,” were not significantly different from their respective null models (Table S4).
-
1.
Mother’s parity. Parity did not significantly predict maternal investment in any of the maternal behaviors (Table I).
-
2.
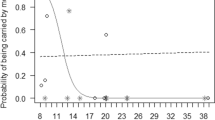
Offspring age. We found that offspring age significantly predicted maternal investment in all six maternal behaviors (Table I). Whereas the probability of mothers initiating body contact and close proximity with their offspring and carrying their offspring decreased with offspring age (odds ratio for an increase in offspring age: 0.18, 0.62, 0.01, respectively; Fig. 1a, b, f), the probability of mothers terminating body contact and terminating close proximity increased with offspring age (odds ratio: 2.81, 2.42, respectively; Fig. 1c, d). The probability of mothers feeding in close proximity with their offspring had a quadratic relationship with offspring age, with the probability being initially low, then increasing with offspring age; subsequently, it reached a peak between 5 and 6 years of age and decreased thereafter (odds ratio: linear age-effect: 11.09; quadratic age-effect: 0.16; Fig. 1e).
Fig. 1 Probability of mothers a) initiating body contact and b) initiating close proximity, c) terminating body contact, d) terminating close proximity, e) feeding in close proximity, and f) carrying their offspring as a function of their offspring’s age in wild Sumatran orangutans (Pongo abelii) analyzed using data collected between 2007 and 2022 at the Suaq Balimbing research area in South Aceh, Indonesia. Each dot represents a follow. They are colored according to offspring identity (color for each offspring is shown in supplementary material, Fig. S1). The solid black line represents the fitted model (offspring age, prevailing fruit availability index, and association size are at their mean; mother’s parity, offspring sex, and male presence/absence were manually dummy coded and then mean centered). The shaded area in grey represents the 95% CI of the fitted model.
-
3.
Offspring sex. We found that the probability of mothers feeding in close proximity with their offspring was significantly higher for mothers with a male offspring than for those with a female offspring (odds ratio: 16.0; Table I; Fig. 2). Offspring sex was not a significant predictor of any of the other maternal behaviors tested (Table I).
Fig. 2 Probability of mothers feeding in close proximity with their offspring as a function of offspring’s sex in wild Sumatran orangutans (Pongo abelii) analyzed using data collected between 2007 and 2022 at the Suaq Balimbing research area in South Aceh, Indonesia. Each dot represents a follow, and they are colored according to offspring identity (color for each offspring is shown in supplementary material, Fig. S1). The horizontal black lines represent the fitted model (offspring age, prevailing fruit availability index, and association size are at their mean; mother’s parity and male presence/absence were manually dummy coded and then mean centered). The vertical black lines represent the 95% CI of the fitted model. The data points are jittered on the x-axis to reduce overlap.
-
4.
FAI. During the study period, FAI ranged between 3.37 and 16.71, with a mean ± SD = 10.035 ± 2.875 (Fig. S3). We found that with an increase in FAI there was a significant reduction in the probability of a mother feeding in close proximity with her offspring (odds ratio: 0.66; Table I; Fig. 3). However, FAI was not a significant predictor of any of the other maternal behaviors tested (Table I).
Fig. 3 Probability of mothers feeding in close proximity with their offspring as a function of monthly prevailing fruit availability index in wild Sumatran orangutans (Pongo abelii) analyzed using data collected between 2007 and 2022 at the Suaq Balimbing research area in South Aceh, Indonesia. Each dot represents a follow, and they are colored according to offspring identity (color for each offspring is shown in supplementary material, Fig. S1). The solid black line represents the fitted model (offspring age, prevailing fruit availability index, and association size are at their mean; mother’s parity, offspring sex, and male presence/absence were manually dummy coded and then mean centered). The shaded grey area represents the 95% CI of the fitted model.
-
5.
Average association size. During the study period, the daily average (here, mean) number of association partners for the focal mother or offspring ranged between 0.89 (excluding the focal individual) to 11.75 association partners (mean ± SD = 1.655 ± 0.823; averaged across all follows). We found that with an increase in average association size, there was a significant increase in the probability of the mother initiating body contact (odds ratio: 1.25; Table 1; Fig. 4a) and a significant decrease in the probability of mothers terminating close proximity (odds ratio: 0.87; Table I; Fig. 4b) with her offspring. Average association size did not significantly predict any of the other maternal behaviors tested (Table I).
Fig. 4 Probability of mothers a) initiating body contact and b) terminating close proximity with their offspring as a function of their daily average association size in wild Sumatran orangutans (Pongo abelii) analyzed using data collected between 2007 and 2022 at the Suaq Balimbing research area in South Aceh, Indonesia. Each dot represents a follow, and they are colored according to offspring’s identity (color for each offspring is shown in supplementary material, Fig. S1). The solid black line represents the fitted model (offspring age, prevailing fruit availability index, and average association size are at their mean; mother’s parity, offspring sex, and male presence/absence were manually dummy coded and then mean centered). The shaded grey area represents the 95% CI of the fitted model.
-
6.
Male presence. Male presence in associations had a significant positive effect on the probability of mothers carrying their offspring while moving (odds ratio: 1.64; Table I; Fig. 5). Male presence/absence did not significantly predict any of the other maternal behaviors (Table I).
Fig. 5 Probability of mothers carrying their offspring as a function of male presence/absence in associations in wild Sumatran orangutans (Pongo abelii) analyzed using data collected between 2007 and 2022 at the Suaq Balimbing research area in South Aceh, Indonesia. Each dot represents a follow, and they are colored according to offspring identity (color for each offspring is shown in supplementary material, Fig. S1). The horizontal black lines represent the fitted model (offspring age, prevailing fruit availability index, and association size are at their mean; mother’s parity and offspring’s sex were manually dummy coded and then mean centered). The vertical black lines represent the 95% CI of the fitted model. The data points are jittered on the x-axis to reduce overlap.
Discussion
Our study showed that mother–offspring characteristics and socioecological variables are associated with variation in maternal investment in several maternal behaviors in Sumatran orangutans. However, we did not investigate the extent to which the analyzed maternal behaviors entail actual costs (e.g., energetic and/or opportunity costs) for the mother and thus represent forms of maternal investment in the classical sense (Clutton-Brock, 1991).
Mother's Parity
Contrary to our first prediction, primiparous and multiparous mothers did not significantly differ in their maternal investment in locomotory and skill acquisition support, protective proximity maintenance, and independence promotion. Our results augment the existing literature showing a lack of difference in maternal investment based on parity in terrestrial primates (cradle, restrain, retrieve: Seay, 1966; different spatial proximity measures: Silk, 1991). However, these findings contrast with studies on primates of varying degree of arboreality in which multiparous mothers seem to invest more into (premasticated) food transfer (e.g., chimpanzee (Pan troglodytes): Bădescu et al., 2020) and independence promotion (e.g., rhesus macaques: Seay, 1966; Japanese macaques (Macaca fuscata): Tanaka, 1989) during the first 6 months of (offspring) age. Because of the extremely long period of offspring dependency, orangutan mothers have the unique opportunity to learn and improve their maternal proficiency during the dependency period of their first offspring, which is equivalent to multiple offspring dependency periods for mothers in other primate species. Although it is possible that primiparous orangutan mothers may have to learn basic maternal care, this may not necessarily take 8 years. To understand the development in maternal proficiency, with a larger sample size, follow-up studies should investigate the interaction effect of mother’s parity and offspring age on maternal investment.
The fact that first time and experienced mothers were similar in their investment through different maternal functions suggests that primiparous Sumatran orangutan females do not need to compensate for a lack of experience and show adequate offspring age-appropriate care without previous direct experience. Given the increasing evidence for social learning of routine skills (such as feeding and nest building) by immature orangutans (Schuppli et al., 2016; Schuppli & van Schaik 2019), it is plausible that daughters learn maternal behaviors from their mothers either during their own dependency period (Maestripieri, 2005) or by watching their mothers’ behavior towards their younger sibling (Berman, 1990). Analyzing developmental speed and survival of offspring of mothers of differing experience would help to pin down whether primiparous mothers are indeed fully competent caregivers.
Offspring Age
Offspring age affected six of the maternal behaviors. In support of our second prediction, mothers decreased locomotory and skill acquisition support and protective proximity maintenance, while they increased independence promotion as their dependents became older. This pattern suggests that mothers adjust their investment to their offspring’s needs and thus optimize their investment in terms of energy and time costs without negatively affecting their offspring’s development. Examples of maternal investment changing with offspring age in terrestrial and arboreal primates include contact termination, rejection, and leaving one’s offspring (Rhesus monkeys: Berman, 1980; vervet monkey: Fairbanks & McGuire, 1987) increasing with offspring age, and time spent carrying (baboons: Altmann and Samuels, 1992; Mantled howler monkeys (Alouatta palliata): Dias et al., 2018) and body contact initiation and restraining (Berman, 1980; Fairbanks & McGuire, 1987; Förster & Cords, 2002) decreasing with offspring age.
The initial low probability of mothers and offspring feeding in close proximity (while they fed on the same food item) is a result of the offspring starting to feed on solid foods only gradually from around 1 year of age onwards (van Noordwijk et al., 2013a, 2013b; van Noordwijk & van Schaik, 2005). This was followed by an increase in the probability of mothers and offspring feeding in close proximity, peaking around 5–6 years of offspring age, which coincides with a steep increase in the immatures’ dietary breadth (Schuppli et al., 2021). Previous chimpanzee and orangutan studies also showed that mothers were increasingly less likely to share food items, as food processing competence of the offspring increases with age (Bădescu et al., 2020; Jaeggi et al., 2008; Mikeliban et al., 2021), indicating that mothers may be attuned to the developing competence of their offspring (Mikeliban et al., 2021). However, our current analysis cannot disentangle the role of mother versus offspring in the probability of feeding in close proximity. With a larger dataset, one could analyze whether the mother or the offspring breaks close proximity while feeding. Such an analysis would help to understand if maternal contribution to feeding skill acquisition changes with offspring age.
Offspring Sex
In line with our prediction, we found that mothers with male offspring showed higher skill acquisition support through feeding in close proximity than those with female offspring. However, compared with their female age-peers, immature males peer at their mothers less frequently (Ehmann et al., 2021). In chimpanzees, there is evidence of mothers sharing nuts more with their male than female offspring (Estienne et al., 2019), but active skill acquisition support cannot be discerned from our study. Furthermore, research on another orangutan population has shown that females reach larger diet repertoires sooner than males (Schuppli et al., 2021). Due to earlier skill acquisition, female offspring can afford to feed further away from their mothers, suggesting that this behavior may be initiated by the offspring more than that by the mothers.
Mothers investing more into male offspring appears to be in line with the Trivers-Willard hypothesis (Trivers & Willard, 1973), given that orangutan males potentially have a more variable reproductive success than females. To properly test the Trivers-Willard hypothesis, we would need to account for maternal body condition and compare the same mother with offspring of both sexes. The latter is important to tease apart interindividual differences in maternal behavior from differences in maternal behavior related to offspring sex. Unfortunately, our dataset currently does not allow for this. Therefore, support for the Trivers-Willard hypothesis by our results is very limited. Furthermore, our analyses cannot rule out that the difference in skill acquisition support are not mostly mediated by the offspring: male and female offspring may differ in how much time they spend at different distances to their mothers, which may lead to differences in close proximity feeding.
Contrary to our third prediction, mothers with male or female offspring did not differ in their investment in protective proximity maintenance, locomotory, and other forms of skill-acquisition support, and independence promotion. These results are in line with other studies on primates, which found little to no overt sex-biased difference in maternal investment in behaviors related to protective proximity maintenance and independence promotion (restraint, rejection, body contact, and various proximity measures: Brown & Dixson, 2000; Silk, 1991; Tanaka, 1989). The absence of sex-biased maternal investment in these behaviors could be a result of these behaviors being vital for the development and safety of both the sexes. It is possible that sex-biased investment occurs during a certain age period rather than throughout offspring development. Our sample size did not allow us to test the interaction between offspring age and sex on maternal investment. Furthermore, we had more than twice the number of male than female offspring in our dataset, which means that our current results, including the absence of sex differences in certain behaviors, should be treated with caution. A larger and a more balanced sample would allow us to interpret the effect of offspring sex more reliably.
Food availability
Contrary to our fourth prediction, locomotory support did not decrease with decreasing food availability. Because our measures of skill acquisition support included either solid food sharing or food patch sharing (in case of feeding in close proximity) with one’s offspring, we expected such support to affect mother’s feeding efficiency, ultimately resulting in lower energy intake for her. However, contrary to our prediction, skill acquisition support through mothers feeding in close proximity to their offspring increased with decreasing food availability. These patterns suggest that food availability may not constrain the time or energy that mothers invest in locomotory or skill acquisition support; however, we had only few data points in the low range of FAI. Hence, the results should be interpreted with caution. At times of low fruit availability, orangutans feed on fallback foods, such as bark, pith, and leaves (Vogel et al., 2017). Even though food availability is generally high at Suaq compared with other orangutan habitats (Marshall et al., 2009), there is an increase in bark and pith feeding during periods of relatively low food availability in Suaq (Schuppli, unpublished data). Some of these fallback foods are more clumped (e.g., bark) and might lead to higher frequency of feeding in close proximity than while feeding on fruits (which at Suaq often are eaten in fruiting trees with large crowns). Furthermore, some of the fallback foods are difficult to process (e.g., bark and pith), i.e., require more intense learning (Schuppli et al., 2016), and may thus favor increased time spent in close proximity.
We further found that food availability did not affect any other maternal behavior. A reason for this could be that food availability is generally high in Suaq. In other populations where individuals face more pronounced fluctuations, punctuated with periods of extremely low food availability (Marshall et al., 2009), maternal investment may be considerably affected by prevailing food availability.
All in all, the results regarding the effects of food availability on maternal investment add to the growing body of literature that shows that resource availability influences certain aspects of maternal care. It is well known that in primates food availability affects maternal body condition (Dias et al., 2018) and that food availability (Hauser, 1993) and maternal body condition influence the quality of maternal care (Clutton-Brock, 1991; Fairbanks & McGuire, 1995; Lee et al., 1991). Mothers living in high-quality habitat and who are in better body condition are more likely to respond to their offspring’s distress calls (Hauser, 1993) and remain spatially closer to their offspring or carry their offspring (Dias et al., 2018) than those living in poor-quality habitat or who are in poor body condition.
Association Size
Contrary to our fifth prediction, energy-intensive forms of maternal behavior, such as locomotory and skill acquisition support, were not affected by daily average association sizes. However, in agreement with studies on terrestrial primates (Berman et al., 1997), protective proximity maintenance with dependents through body contact initiation increased and close proximity termination decreased with an increase in daily average association size, even though notably the effect sizes were small. Average association size did not influence any of the other maternal behaviors. Even though Suaq orangutans are more socially tolerant than Bornean orangutans (Pongo pygmaeus wurmbii), they are semisolitary with low overall rates of close associations and social interactions, most likely because associations come with energetic costs, such as increased duration of the active period and decreased feeding time (Kunz et al., 2021). Unfamiliar/unrelated association partners may pose a risk for young and socially inexperienced offspring, thereby prompting an increase in protective proximity maintenance from their mothers. A previous study on orangutans found a positive relationship between association size and attempts of the mothers to restrain their offspring (Falkner, 2015). This finding provides further support that mothers are actively keeping their dependent offspring close to them during associations. Because mothers invest more with increasing association size, it indicates that the costs of associations do not constrain mothers in their maternal investment, at least during the time of the associations. However, because we did not have dense data on maternal investment during large association sizes, these results should be interpreted with caution. Future studies should investigate how the effects of association size on maternal behavior depend on the age-sex class or the degree of relatedness/familiarity of the associating individuals, with data spanning the entire range of the predictor.
Male Presence
In support of our sixth prediction, we found that mothers were more likely to carry their offspring in the presence than in the absence of an associating male. In several terrestrial and arboreal primate species with sexual size dimorphism, mothers were reported to show potential anti-infanticide strategies, such as increased body contact time, initiation of body contact, physical restraint, as well as inspection of offspring, and reduced distance and decreased time away from their offspring in the presence of males (vervets: Fairbanks & McGuire, 1987; chimpanzees: Otali & Gilchrist, 2006; Bornean orangutans: Scott et al., 2023, 2019). Because of their arboreal lifestyle, carrying is likely an additional means by which orangutan mothers protect their offspring in the presence of a male by reducing the time their offspring are out of body contact. This may reduce the risk of an attack by the male on the offspring and/or keep the offspring safe from potential falls when moving away from the male. Our results indicate that Sumatran orangutan females may view at least some males as a potential threat and increase locomotory support as a counter-infanticide strategy. Mothers also might change their behavior in the presence of males depending on their reproductive status, which is closely linked to the age of their current dependent offspring (Fairbanks & McGuire, 1987; Kunz et al., 2022; Scott et al., 2019). Furthermore, it is likely that not all males are a threat, and maternal behavior may vary based on whether a male is a resident (Fairbanks & McGuire, 1987; Kunz et al., 2022; van Noordwijk et al., 2023). With a larger sample size, future studies should test the interaction effect of offspring age and male presence/absence and the effect of associating male’s familiarity on maternal investment in different behaviors.
Unaffected Maternal Behaviors
The probabilities of mothers following and avoiding their offspring and the probability of food transfer were not significantly associated with any of the six predictors that we tested. Previous studies have shown that mothers adjust how much they tolerate food taking by their offspring according to the age of their offspring and based on the properties of the food item that is taken (Jaeggi et al., 2008; Mikeliban et al., 2021). However, the sample size for food transfer was quite low in the current study (N = 264 focal follows), especially in relation to the complexity of our statistical model, which may be the reason for the full model not performing better than the null model. With respect to the behaviors “follow’ and “avoid,” it remains to be investigated whether they do indeed represent forms of maternal investment in orangutans. Notably, these were among the most infrequent behaviors in our data.
Conclusions
Our comprehensive approach of testing the effects of six predictors on nine maternal behaviors showed that Sumatran orangutan mothers invest in locomotory and skill acquisition support, protective proximity maintenance, and independence promotion flexibly in response to mother–offspring characteristics and short-term varying socioecological factors. Offspring age, sex, association size, and male presence had similar effects on maternal investment in Sumatran orangutans as found in terrestrial primates, underscoring the importance of these shared effects across species and the environments they inhabit. In many aspects, these flexible adjustments appear to support offspring development or protect the mothers from facing costs that may endanger their own survival. As such, this study is in line with the evolutionary theory that females allocate investment in a flexible manner to maximize their lifetime reproductive success (Clutton-brock, 1991; Trivers, 1972; Lee et al., 1991). However, whether maternal investment through these behaviors indeed translates into actual fitness benefits for the mother (through increased own or offspring survival, and/or faster reproductive rates) remains to be investigated.
References
Altmann, J. (1974). Observational study of behavior: Sampling methods. Behaviour, 49(3–4), 227–266. https://doi.org/10.1163/156853974X00534
Altmann, J., & Alberts, S. C. (2005). Growth rates in a wild primate population: Ecological influences and maternal effects. Behavioral Ecology and Sociobiology, 57(5), 490–501. https://doi.org/10.1007/s00265-004-0870-x
Altmann, J., & Samuels, A. (1992). Costs of maternal care: Infant-carrying in baboons. Behavioral Ecology and Sociobiology, 29(6), 391–398. https://doi.org/10.1007/BF00170168
American Society of Primatologists (2001). American society of primatologists' policy statement on the ethical treatment of nonhuman primates. https://asp.org/2021/04/20/principles-for-the-ethical-treatment-of-non-human-primates/
Arlet, M. E., Isbell, L. A., Molleman, F., Kaasik, A., Chancellor, R. L., Chapman, C. A., Mänd, R., & Carey, J. R. (2014). Maternal investment and infant survival in Gray-cheeked mangabeys (Lophocebus albigena). International Journal of Primatology, 35(2), 476–490. https://doi.org/10.1007/s10764-014-9754-8
Arora, N., van Noordwijk, M. A., Ackermann, C., Willems, E. P., Nater, A., Greminger, M., Nietlisbach, P., Dunkel, L. P., Utami Atmoko, S. S., Pamungkas, J., Perwitasari-Farajallah, D., van Schaik, C. P., & Krützen, M. (2012). Parentage-based pedigree reconstruction reveals female matrilineal clusters and male-biased dispersal in nongregarious Asian great apes, the Bornean orang-utans (Pongo pygmaeus). Molecular Ecology, 21(13), 3352–3362. https://doi.org/10.1111/j.1365-294X.2012.05608.x
Ashbury, A. M., Willems, E. P., Utami Atmoko, S. S., Saputra, F., van Schaik, C. P., & van Noordwijk, M. A. (2020). Home range establishment and the mechanisms of philopatry among female Bornean orangutans (Pongo pygmaeus wurmbii) at Tuanan. Behavioral Ecology and Sociobiology, 74(4), 42. https://doi.org/10.1007/s00265-020-2818-1
Baayen, R. H. (2008). Analyzing Linguistic Data: A Practical Introduction to Statistics using R. Cambridge University Press. https://doi.org/10.1017/CBO9780511801686
Bădescu, I., Sicotte, P., Sandel, A. A., Desruelle, K. J., Curteanu, C., Watts, D. P., & Sellen, D. W. (2020). Premasticated food transfer by wild chimpanzee mothers with their infants: Effects of maternal parity, infant age and sex, and food properties. Journal of Human Evolution, 143, 102794. https://doi.org/10.1016/j.jhevol.2020.102794
Barr, D. J., Levy, R., Scheepers, C., & Tily, H. J. (2013). Random effects structure for confirmatory hypothesis testing: Keep it maximal. Journal of Memory and Language, 68(3), 255–278. https://doi.org/10.1016/j.jml.2012.11.001
Barton, K., & Barton, M. K. (2015). Package “mumin.” Version, 1(18), 439.
Bates, D., Maechler, M., Bolker, B., & Walker, S. (2014). lme4: Linear mixed-effects models using Eigen and S4. R package version 1, 1–7. https://cran.r-project.org/web/packages/lme4/lme4.pdf
Beaudrot, L. H., Kahlenberg, S. M., & Marshall, A. J. (2009). Why male orangutans do not kill infants. Behavioral Ecology and Sociobiology, 63(11), 1549–1562. https://doi.org/10.1007/s00265-009-0827-1
Berman, C. M. (1980). Mother-infant relationships among free-ranging rhesus monkeys on Cayo Santiago: A comparison with captive pairs. Animal Behaviour, 28(3), 860–873. https://doi.org/10.1016/S0003-3472(80)80146-1
Berman, C. M. (1990). Intergenerational transmission of maternal rejection rates among free-ranging rhesus monkeys. Animal Behaviour, 39(2), 329–337. https://doi.org/10.1016/S0003-3472(05)80878-4
Berman, C. M., Rasmussen, K. L. R., & Suomi, S. J. (1997). Group size, infant development and social networks in free-ranging rhesus monkeys. Animal Behaviour, 53(2), 405–421. https://doi.org/10.1006/anbe.1996.0321
Boesch, C. (1991). Teaching among wild chimpanzees. Animal Behaviour, 41, 530–532. https://doi.org/10.1016/S0003-3472(05)80857-7
Breaugh, J. A. (2003). Effect size estimation: Factors to consider and mistakes to avoid. Journal of Management, 29(1), 79–97. https://doi.org/10.1177/014920630302900106
Brown, G. R., Almond, R. E. A., & Bergen, Y. V. (2004). Begging, stealing, and offering: Food transfer in nonhuman primates. In P. J. B. Slater, J. S. Rosenblatt, C. T. Snowdon, T. J. Roper, H. J. Brockmann, & M. Naguib (Eds.), Advances in the Study of Behavior (pp. 265–295). Elsevier Academic Press. https://doi.org/10.1016/S0065-3454(04)34007-6
Brown, G. R., & Dixson, A. F. (2000). The development of behavioural sex differences in infant rhesus macaques (Macaca mulatta). Primates, 41(1), 63–77. https://doi.org/10.1007/BF02557462
Chappell, J., Phillips, A. C., van Noordwijk, M. A., Mitra Setia, T., & Thorpe, S. K. (2015). The ontogeny of gap crossing behaviour in Bornean orangutans (Pongo pygmaeus wurmbii). PLOS ONE, 10(7), e0130291. https://doi.org/10.1371/journal.pone.0130291
Clutton-Brock, T. H. (1991). The evolution of parental care. Princeton University Press.
Dettmer, A. M., Kaburu, S. S. K., Byers, K. L., Murphy, A. M., Soneson, E., Wooddell, L. J., & Suomi, S. J. (2016). First-time rhesus monkey mothers, and mothers of sons, preferentially engage in face-to-face interactions with their infants. American Journal of Primatology, 78(2), 238–246. https://doi.org/10.1002/ajp.22503
Dias, P. A. D., Cano-Huertes, B., Coyohua-Fuentes, A., Chavira-Ramírez, D. R., Canales-Espinosa, D., & Rangel-Negrín, A. (2018). Maternal condition and maternal investment during lactation in mantled howler monkeys. American Journal of Physical Anthropology, 167(1), 178–184. https://doi.org/10.1002/ajpa.23626
Dobson, A. J., & Barnett, A. G. (2018). An Introduction to Generalized Linear Models. CRC Press.
Ehmann, B., van Schaik, C. P., Ashbury, A. M., Mörchen, J., Musdarlia, H., Utami Atmoko, S. S., van Noordwijk, M. A., & Schuppli, C. (2021). Immature wild orangutans acquire relevant ecological knowledge through sex-specific attentional biases during social learning. PLOS Biology, 19(5), e3001173. https://doi.org/10.1371/journal.pbio.3001173
Estienne, V., Cohen, H., Wittig, R. M., & Boesch, C. (2019). Maternal influence on the development of nut‐cracking skills in the chimpanzees of the Taï forest, Côte d’Ivoire ( Pan troglodytes verus). American Journal of Primatology, 81(7). https://doi.org/10.1002/ajp.23022
Fairbanks, L. A. (1996). Individual differences in maternal style. Advances in the Study of Behavior, 25, 579–611. https://doi.org/10.1016/S0065-3454(08)60343-5
Fairbanks, L. A., & McGuire, M. T. (1984). Determinants of fecundity and reproductive success in captive vervet monkeys. American Journal of Primatology, 7(1), 27–38. https://doi.org/10.1002/ajp.1350070106
Fairbanks, L. A., & McGuire, M. T. (1986). Age, reproductive value, and dominance-related behaviour in vervet monkey females: Cross-generational influences on social relationships and reproduction. Animal Behaviour, 34(6), 1710–1721. https://doi.org/10.1016/S0003-3472(86)80258-5
Fairbanks, L. A., & McGuire, M. T. (1987). Mother-infant relationships in vervet monkeys: Response to new adult males. International Journal of Primatology, 8(4), 351–366. https://doi.org/10.1007/BF02737388
Fairbanks, L. A., & McGuire, M. T. (1995). Maternal condition and the quality of maternal care in vervet monkeys. Behaviour, 132(9/10), 733–754. https://www.jstor.org/stable/4535296. Accessed 16 Nov 2023.
Falkner, S. (2015). Mother-offspring conflict in orangutans. Master's thesis, University of Zurich
Forss, S. (2009). Social learning and independent exploration in immature Sumatran orangutans, Pongo abelii. Doctoral dissertation, University of Zurich.
Förster, S., & Cords, M. (2002). Development of mother-infant relationships and infant behavior in wild blue monkeys (Cercopithecus mitis stuhlmanni). In M. E. Glenn & M. Cords (Eds.), The Guenons: Diversity and Adaptation in African Monkeys (pp. 245–272). Springer. https://doi.org/10.1007/0-306-48417-X_18
Forstmeier, W., & Schielzeth, H. (2011). Cryptic multiple hypotheses testing in linear models: Overestimated effect sizes and the winner’s curse. Behavioral Ecology and Sociobiology, 65(1), 47–55. https://doi.org/10.1007/s00265-010-1038-5
Fox, J., & Weisberg, S. (2011). An R Companion to Applied Regression. SAGE Publications.
Hanna, J. B., & Schmitt, D. (2011). Locomotor energetics in primates: Gait mechanics and their relationship to the energetics of vertical and horizontal locomotion. American Journal of Physical Anthropology, 145(1), 43–54. https://doi.org/10.1002/ajpa.21465
Harrison, X. A., Donaldson, L., Correa-Cano, M. E., Evans, J., Fisher, D. N., Goodwin, C. E. D., Robinson, B. S., Hodgson, D. J., & Inger, R. (2018). A brief introduction to mixed effects modelling and multi-model inference in ecology. PeerJ, 6, e4794. https://doi.org/10.7717/peerj.4794
Hauser, M. D. (1993). Do vervet monkey infants cry wolf? Animal Behaviour, 45(6), 1242–1244. https://doi.org/10.1006/anbe.1993.1148
Hauser, M. D., & Fairbanks, L. A. (1988). Mother-offspring conflict in vervet monkeys: Variation in response to ecological conditions. Animal Behaviour, 36(3), 802–813. https://doi.org/10.1016/S0003-3472(88)80163-5
Hinde, K., & Milligan, L. A. (2011). Primate milk: Proximate mechanisms and ultimate perspectives. Evolutionary Anthropology: Issues, News, and Reviews, 20(1), 9–23. https://doi.org/10.1002/evan.20289
Hinde, K., Power, M. L., & Oftedal, O. T. (2009). Rhesus macaque milk: Magnitude, sources, and consequences of individual variation over lactation. American Journal of Physical Anthropology, 138(2), 148–157. https://doi.org/10.1002/ajpa.20911
Inoue-Nakamura, N., & Matsuzawa, T. (1997). Development of stone tool use by wild chimpanzees (Pan troglodytes). Journal of Comparative Psychology, 111(2), 159–173. https://doi.org/10.1037/0735-7036.111.2.159
Jaeggi, A. V., van Noordwijk, M. A., & van Schaik, C. P. (2008). Begging for information: Mother–offspring food sharing among wild Bornean orangutans. American Journal of Primatology, 70(6), 533–541. https://doi.org/10.1002/ajp.20525
Jaeggi, A. V., Dunkel, L. P., van Noordwijk, M. A., Wich, S. A., Sura, A. A. L., & van Schaik, C. P. (2010). Social learning of diet and foraging skills by wild immature Bornean orangutans: Implications for culture. American Journal of Primatology, 72(1), 62–71. https://doi.org/10.1002/ajp.20752
Johnson, G., Frantzis, A., Johnson, C., Alexiadou, V., Ridgway, S., & Madsen, P. T. (2010). Evidence that sperm whale (Physeter macrocephalus) calves suckle through their mouth. Marine Mammal Science, 26(4), 990–996. https://doi.org/10.1111/j.1748-7692.2010.00385.x
Knott, C. D., Scott, A. M., O’Connell, C. A., Scott, K. S., LamanRiyandi, T. G., & Susanto, T. W. (2019). Possible male infanticide in wild orangutans and a re-evaluation of infanticide risk. Scientific Reports, 9(1), 7806. https://doi.org/10.1038/s41598-019-42856-w
Kunz, J. A., Duvot, G. J., van Noordwijk, M. A., Willems, E. P., Townsend, M., Mardianah, N., Utami Atmoko, S. S., Vogel, E. R., Nugraha, T. P., Heistermann, M., Agil, M., Weingrill, T., & van Schaik, C. P. (2021). The cost of associating with males for Bornean and Sumatran female orangutans: A hidden form of sexual conflict? Behavioral Ecology and Sociobiology, 75(1), 6. https://doi.org/10.1007/s00265-020-02948-4
Kunz, J. A., van Noordwijk, M. A., & van Schaik, C. P. (2022). Orangutan sexual behavior. In T. K. Shackleford (Ed.), The Cambridge handbook of evolutionary perspectives on sexual psychology (pp. 401–425). Cambridge University Press.
Kunz, J. A., Duvot, G. J., Ashbury, A. M., Willems, E. P., Spillmann, B., Dunkel, L. P., bin Abdullah, M., Schuppli, C., Vogel, E. R., Utami Atmoko, S. S., van Noordwijk, M. A., & van Schaik, C. P. (2023). Alternative reproductive tactics of unflanged and flanged male orangutans revisited. American Journal of Primatology, 85(9), e23535. https://doi.org/10.1002/ajp.23535
Lahdenperä, M., Mar, K. U., & Lummaa, V. (2016). Short-term and delayed effects of mother death on calf mortality in Asian elephants. Behavioral Ecology, 27(1), 166–174. https://doi.org/10.1093/beheco/arv136
Lee, P. C. (1987). Nutrition, fertility and maternal investment in primates. Journal of Zoology, 213(3), 409–422. https://doi.org/10.1111/j.1469-7998.1987.tb03717.x
Lee, P., Majluf, P., & Gordon, I. (1991). Growth, weaning and maternal investment from a comparative perspective. Journal of Zoology, 225, 99–114. https://doi.org/10.1111/j.1469-7998.1991.tb03804.x
Lee, P. C., & Moss, C. J. (2011). Calf development and maternal rearing strategies. In C. J. Moss, H. Croze, & P. C. Lee (Eds.), The Amboseli Elephants: A Long-Term Perspective on a Long-Lived Mammal (pp. 224–237). University of Chicago Press.
Leigh, S. R., & Shea, B. T. (1995). Ontogeny and the evolution of adult body size dimorphism in apes. American Journal of Primatology, 36(1), 37–60. https://doi.org/10.1002/ajp.1350360104
Li, T., Ren, B., Li, D., Zhu, P., & Li, M. (2013). Mothering Style and infant behavioral development in Yunnan snub-nosed monkeys (Rhinopithecus bieti) in China. International Journal of Primatology, 34(4), 681–695. https://doi.org/10.1007/s10764-013-9687-7
Lonsdorf, E. V. (2006). What is the role of mothers in the acquisition of termite-fishing behaviors in wild chimpanzees (Pan troglodytes schweinfurthii)? Animal Cognition, 9(1), 36–46. https://doi.org/10.1007/s10071-005-0002-7
Lubach, G. R., Kittrell, E. M. W., & Coe, C. L. (1992). Maternal influences on body temperature in the infant primate. Physiology & Behavior, 51(5), 987–994. https://doi.org/10.1016/0031-9384(92)90082-D
Lukas, D., & Huchard, E. (2014). The evolution of infanticide by males in mammalian societies. Science, 346(6211), 841–844. https://doi.org/10.1126/science.1257226
Maestripieri, D. (1994). Social structure, infant handling, and mothering styles in group-living old world monkeys. International Journal of Primatology, 15(4), 531–553. https://doi.org/10.1007/BF02735970
Maestripieri, D. (1995). First steps in the macaque world: Do rhesus mothers encourage their infants’ independent locomotion? Animal Behaviour, 49(6), 1541–1549. https://doi.org/10.1016/0003-3472(95)90075-6
Maestripieri, D. (2001). Intraspecific variability in parenting styles of rhesus macaques (Macaca mulatta): The role of the social environment. Ethology, 107(3), 237–248. https://doi.org/10.1046/j.1439-0310.2001.00661.x
Maestripieri, D. (2005). Early experience affects the intergenerational transmission of infant abuse in rhesus monkeys. Proceedings of the National Academy of Sciences, 102(27), 9726–9729. https://doi.org/10.1073/pnas.0504122102
Maestripieri, D. (2018). Maternal influences on primate social development. Behavioral Ecology and Sociobiology, 72(8), 130. https://doi.org/10.1007/s00265-018-2547-x
Mann, J. (2019). Maternal care and offspring development in Odontocetes. In B. Würsig (Ed.), Ethology and Behavioral Ecology of Odontocetes (pp. 95–116). Springer International Publishing. https://doi.org/10.1007/978-3-030-16663-2_5
Marshall, A., Ancrenaz, M., Brearley, F., Fredriksson, G., Ghaffar, N., Heydon, M., Husson, S., Leighton, M., Mcconkey, K., Morrogh-Bernard, H., Proctor, J., Schaik, C., Yeager, C., & Wich, S. (2009). The effects of forest phenology and floristics on populations of Bornean and Sumatran orangutans: Are Sumatran forests more productive than Bornean forests? In S. A. Wich, S. S. Utami Atmoko, T. Mitra Setia, & C. P. van Schaik (Eds.), Geographic variation in behavioral ecology and conservation (pp. 97–117). Oxford University Press. https://doi.org/10.1093/acprof:oso/9780199213276.003.0007
McCullagh, P., & Nelder, J. A. (1989). Generalized Linear Models (2nd ed.). Chapman and Hall.
Mikeliban, M., Kunz, B., Rahmaeti, T., Uomini, N., & Schuppli, C. (2021). Orangutan mothers adjust their behaviour during food solicitations in a way that likely facilitates feeding skill acquisition in their offspring. Scientific Reports, 11(1), 23679. https://doi.org/10.1038/s41598-021-02901-z
Mitchell, G., & Stevens, C. W. (1968). Primiparous and multiparous monkey mothers in a mildly stressful social situation: First three months. Developmental Psychobiology, 1(4), 280–286. https://doi.org/10.1002/dev.420010411
Mörchen, J., Luhn, F., Wassmer, O., Kunz, J., Kulik, L., van Noordwijk, M., van Schaik, C., Rianti, P., Atmoko, S. S. U., Widdig, A., & Schuppli, C. (2023). Migrant orangutan males use social learning to adapt to new habitat after dispersal. Frontiers Ecology And Evolution, 11. https://doi.org/10.3389/fevo.2023.1158887
Morrogh-Bernard, H. C., Husson, S. J., Knott, C. D., Wich, S. A., van Schaik, C. P., van Noordwijk, M. A., Lackman-Ancrenaz, I., Marshall, A. J., Kanamori, T., Kuze, N., & bin Sakong R. (2009). Orangutan activity budgets and diet: A comparison between species, populations and habitats. In S. A. Wich, S. S. Utami Atmoko, T. Mitra Setia, & C. P. van Schaik (Eds.), Orangutans: geographic variation in behavioral ecology and conservation (pp. 119–133). UK: Oxford University Press.
Morrogh-Bernard, H. C., Morf, N. V., Chivers, D. J., & Krützen, M. (2011). Dispersal patterns of orang-utans (Pongo spp.) in a Bornean peat-swamp forest. International Journal of Primatology, 32(2), 362–376. https://doi.org/10.1007/s10764-010-9474-7
Murray, C. M., Lonsdorf, E. V., Stanton, M. A., Wellens, K. R., Miller, J. A., Goodall, J., & Pusey, A. E. (2014). Early social exposure in wild chimpanzees: Mothers with sons are more gregarious than mothers with daughters. Proceedings of the National Academy of Sciences, 111(51), 18189–18194. https://doi.org/10.1073/pnas.1409507111
Nair, P. V. (1989). Development of nonsocial behaviour in the Asiatic elephant. Ethology, 82(1), 46–60. https://doi.org/10.1111/j.1439-0310.1989.tb00486.x
Nash, L. T. (2003). Sex differences in the behavior and the social interactions of immature Galago Senegalensis braccatus. Folia Primatologica, 74(5–6), 285–300. https://doi.org/10.1159/000073315
Nguyen, N., Gesquiere, L., Alberts, S. C., & Altmann, J. (2012). Sex differences in the mother–neonate relationship in wild baboons: Social, experiential and hormonal correlates. Animal Behaviour, 83(4), 891–903. https://doi.org/10.1016/j.anbehav.2012.01.003
Nicolson, N. (1987). Infants, mothers, and other females. In B. B. Smuts, D. L. Cheney, R. M. Seyfarth, R. W. Wrangham, & T. T. Struhsaker (Eds.), Primate Societies (pp. 330–342). University of Chicago Press https://cir.nii.ac.jp/crid/1571417125314995200. Accessed 10 Nov 2023.
Nicolson, N. (1991). Maternal behavior in human and nonhuman primates. In J. D. Loy & C. B. Peters (Eds.), Understanding behavior: What primate studies tell us about human behavior (pp. 17–50). Oxford University Press.
Nietlisbach, P., Arora, N., Nater, A., Goossens, B., van Schaik, C. P., & Krützen, M. (2012). Heavily male-biased long-distance dispersal of orang-utans (genus: Pongo), as revealed by Y-chromosomal and mitochondrial genetic markers. Molecular Ecology, 21(13), 3173–3186. https://doi.org/10.1111/j.1365-294X.2012.05539.x
Nishida, T., & Turner, L. A. (1996). Food transfer between mother and infant chimpanzees of the Mahale Mountains National Park, Tanzania. International Journal of Primatology, 17(6), 947–968. https://doi.org/10.1007/BF02735296
Otali, E., & Gilchrist, J. S. (2006). Why chimpanzee (Pan troglodytes schweinfurthii) mothers are less gregarious than nonmothers and males: The infant safety hypothesis. Behavioral Ecology and Sociobiology, 59(4), 561–570. https://doi.org/10.1007/s00265-005-0081-0
Pereira, M. E. (1989). Agonistic interactions of juvenile savanna baboons. Ethology, 80(1–4), 152–171. https://doi.org/10.1111/j.1439-0310.1989.tb00736.x
Pontzer, H., Raichlen, D. A., Shumaker, R. W., Ocobock, C., & Wich, S. A. (2010). Metabolic adaptation for low energy throughput in orangutans. Proceedings of the National Academy of Sciences, 107(32), 14048–14052. https://doi.org/10.1073/pnas.1001031107
Quinn, G. P., & Keough, M. J. (2002). Experimental design and data analysis for biologists. Cambridge University Press.
R Core Team (2023). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Revathe, T., Anvitha, S., & Vidya, T. N. C. (2020). Development of motor control and behaviour in Asian elephants in the Kabini elephant population, southern India. The International Journal of Developmental Biology, 64(4 5 6), 367–382. https://doi.org/10.1387/ijdb.190274tv
Roth, T. S., Rianti, P., Fredriksson, G. M., Wich, S. A., & Nowak, M. G. (2020). Grouping behavior of Sumatran orangutans (Pongo abelii) and Tapanuli orangutans (Pongo tapanuliensis) living in forest with low fruit abundance. American Journal of Primatology, 82(5), e23123. https://doi.org/10.1002/ajp.23123
Schielzeth, H. (2010). Simple means to improve the interpretability of regression coefficients. Methods in Ecology and Evolution, 1(2), 103–113. https://doi.org/10.1111/j.2041-210X.2010.00012.x
Schielzeth, H., & Forstmeier, W. (2009). Conclusions beyond support: Overconfident estimates in mixed models. Behavioral Ecology, 20(2), 416–420. https://doi.org/10.1093/beheco/arn145
Schino, G., & Troisi, A. (2005). Neonatal abandonment in Japanese macaques. American Journal of Physical Anthropology, 126(4), 447–452. https://doi.org/10.1002/ajpa.20078
Schuppli, C., & van Schaik, C. (2019). Social learning among wild orang-utans. Is it affective? In D. Dukes & F. Clement (Eds.), Foundations of affective social learning: conceptualizing the social transmission of value (pp. 25–40). Cambridge University Press.
Schuppli, C., Meulman, E. J. M., Forss, S. I. F., Aprilinayati, F., van Noordwijk, M. A., & van Schaik, C. P. (2016). Observational social learning and socially induced practice of routine skills in immature wild orang-utans. Animal Behaviour, 119, 87–98. https://doi.org/10.1016/j.anbehav.2016.06.014
Schuppli, C., Atmoko, S. S. U., Vogel, E. R., van Schaik, C. P., & van Noordwijk, M. A. (2021). The development and maintenance of sex differences in dietary breadth and complexity in Bornean orangutans. Behavioral Ecology and Sociobiology, 75(5), 81. https://doi.org/10.1007/s00265-021-03014-3
Scott, A. M., Knott, C. D., & Susanto, T. W. (2019). Are male orangutans a threat to infants? Evidence of mother–offspring counterstrategies to infanticide in Bornean orangutans (Pongo pygmaeus wurmbii). International Journal of Primatology, 40(3), 435–455. https://doi.org/10.1007/s10764-019-00097-8
Scott, A. M., Susanto, T. W., Setia, T. M., & Knott, C. D. (2023). Mother-offspring proximity maintenance as an infanticide avoidance strategy in Bornean orangutans (Pongo pygmaeus wurmbii). American Journal of Primatology, 85(6), e23482. https://doi.org/10.1002/ajp.23482
Seay, B. (1966). Maternal Behavior in primiparous and multiparous rhesus monkeys. Folia Primatologica, 4(2), 146–168. https://doi.org/10.1159/000155049
Setchell, J. M., Lee, P. C., Wickings, E. J., & Dixson, A. F. (2002). Reproductive parameters and maternal investment in mandrills (Mandrillus sphinx). International Journal of Primatology, 23(1), 51–68. https://doi.org/10.1023/A:1013245707228
Setia, T. M., Delgado, R. A., Utami Atmoko, S. S., Singleton, I., & van Schaik, C. P. (2009). Social organization and male–female relationships. In S. A. Wich, S. S. Utami Atmoko, T. Mitra Setia, & C. P. van Schaik (Eds.), Orangutans geographic variation in behavioral ecology and conservation (pp. 245–253). Oxford University Press.
Silk, J. B. (1991). Mother-infant relationships in bonnet macaques: Sources of variation in proximity. International Journal of Primatology, 12(1), 21–38. https://doi.org/10.1007/BF02547556
Spillmann, B., Willems, E. P., van Noordwijk, M. A., Setia, T. M., & van Schaik, C. P. (2017). Confrontational assessment in the roving male promiscuity mating system of the Bornean orangutan. Behavioral Ecology and Sociobiology, 71, 20. https://doi.org/10.1007/s00265-016-2252-6
Surbeck, M., Mundry, R., & Hohmann, G. (2010). Mothers matter! Maternal support, dominance status and mating success in male bonobos (Pan paniscus). Proceedings of the Royal Society B: Biological Sciences, 278(1705), 590–598. https://doi.org/10.1098/rspb.2010.1572
Szabo, A., & Duffus, D. (2008). Mother–offspring association in the humpback whale, Megaptera novaeangliae: Following behaviour in an aquatic mammal. Animal Behaviour, 75(3), 1085–1092. https://doi.org/10.1016/j.anbehav.2007.08.019
Taber, S., & Thomas, P. (1982). Calf development and mother-calf spatial relationships in Southern right whales. Animal Behaviour, 30(4), 1072–1083. https://doi.org/10.1016/S0003-3472(82)80197-8
Tanaka, I. (1989). Variability in the development of mother-infant relationships among free-ranging Japanese macaques. Primates, 30(4), 477–491. https://doi.org/10.1007/BF02380875
Thorpe, K. S., & Crompton, R. H. (2009). Orangutan positional behaviour. In S. A. Wich, S. S. Utami Atmoko, T. Mitra Setia, & C. P. van Schaik (Eds.), Orangutans: Geographic variation in behavioral ecology and conservation (pp. 33–47). Oxford University Press. https://doi.org/10.1093/acprof:oso/9780199213276.003.0015
Trivers, R. L. (1972). Parental investment and sexual selection. In B. G. Campbell (Ed.), Sexual Selection and the Descent of Man (pp. 52–95). Routledge.
Trivers, R. L. (1974). Parent-offspring conflict. American Zoologist, 14(1), 249–264. https://doi.org/10.1093/icb/14.1.249
Trivers, R. L., & Willard, D. E. (1973). Natural selection of parental ability to vary the sex ratio of offspring. Science, 179(4068), 90–92. https://doi.org/10.1126/science.179.4068.90
Utami Atmoko, S. S., Singleton, I., van Noordwijk, M. A., van Schaik, C. P., & Mitra Setia, T. (2009). Male-male relationships in orangutans. In S. A. Wich, S. S. Utami Atmoko, T. Mitra Setia, & C. P. van Schaik (Eds.), Orangutans: Geographic variation in behavioral ecology and conservation (pp. 225–233). Oxford University Press. https://doi.org/10.1093/acprof:oso/9780199213276.003.0015
van Adrichem, G. G. J., Utami, S. S., Wich, S. A., van Hooff, J. A. R. A. M., & Sterck, E. H. M. (2006). The development of wild immature Sumatran orangutans (Pongo abelii) at Ketambe. Primates, 47(4), 300–309. https://doi.org/10.1007/s10329-006-0193-9
van Noordwijk, M. A. (2012). From maternal investment to lifetime maternal care. In J. C. Mitani, J. Call, P. M. Kappeler, R. A. Palombit, & J. B. Silk (Eds.), The evolution of primate societies (pp. 321–42). University of Chicago Press. https://doi.org/10.5167/UZH-70319
van Noordwijk, M. A., & van Schaik, C. P. (2005). Development of ecological competence in Sumatran orangutans. American Journal of Physical Anthropology, 127(1), 79–94. https://doi.org/10.1002/ajpa.10426
van Noordwijk, M. A., Kuzawa, C. W., & van Schaik, C. P. (2013a). The evolution of the patterning of human lactation: A comparative perspective. Evolutionary Anthropology: Issues, News, and Reviews, 22(5), 202–212. https://doi.org/10.1002/evan.21368
van Noordwijk, M. A., Willems, E. P., Utami Atmoko, S. S., Kuzawa, C. W., & van Schaik, C. P. (2013b). Multi-year lactation and its consequences in Bornean orangutans (Pongo pygmaeus wurmbii). Behavioral Ecology and Sociobiology, 67(5), 805–814. https://doi.org/10.1007/s00265-013-1504-y
van Noordwijk, M. A., Sauren, S. E. B., Nuzuar, Abulani, A., Morrogh-Bernard, H. C., Utami Atmoko, S. S., & van Schaik, C. P. (2009). Development of independence: Sumatran and Bornean orangutans compared. In S. A. Wich, S. S. Utami Atmoko, T. Mitra Setia, & C. P. van Schaik (Eds.), Geographic variation in behavioral ecology and conservation (pp. 189–203). Oxford University Press. https://doi.org/10.1093/acprof:oso/9780199213276.003.0012
van Noordwijk, M. A., Utami Atmoko, S. S., Knott, C. D., Kuze, N., Morrogh-Bernard, H. C., Oram, F., Schuppli, C., van Schaik, C. P., & Willems, E. P. (2018). The slow ape: High infant survival and long interbirth intervals in wild orangutans. Journal of Human Evolution, 125, 38–49. https://doi.org/10.1016/j.jhevol.2018.09.004
van Noordwijk, M. A., LaBarge, L. R., Kunz, J. A., Marzec, A. M., Spillmann, B., Ackermann, C., Rianti, P., Vogel, E. R., Atmoko, S. S. U., Kruetzen, M., & van Schaik, C. P. (2023). Reproductive success of Bornean orangutan males: Scattered in time but clustered in space. Behavioral Ecology and Sociobiology, 77(12), 134. https://doi.org/10.1007/s00265-023-03407-6
van Schaik, C. P. (1999). The socioecology of fission-fusion sociality in Orangutans. Primates, 40(1), 69–86. https://doi.org/10.1007/BF02557703
van Schaik, C. P. (2000). Vulnerability to infanticide by males: patterns among mammals. In C. P. van Schaik & C. H. Janson (Eds.), Infanticide by Males and Its Implications (pp. 61–72). Cambridge University Press. https://doi.org/10.1017/CBO9780511542312.005
van Schaik, C. P. (2013). The costs and benefits of flexibility as an expression of behavioural plasticity: A primate perspective. Philosophical Transactions of the Royal Society B: Biological Sciences, 368(1618), 20120339. https://doi.org/10.1098/rstb.2012.0339
van Schaik, C. P., & van Hooff, J. A. R. A. M. (1996). Toward an understanding of the orangutan’s social system. In W. C. McGrew, L. F. Marchant, & T. Nishida (Eds.), Great ape societies (pp. 3–15). Cambridge University Press. https://doi.org/10.1017/CBO9780511752414.003
van Schaik, C. P., Fox, E. A., & Sitompul, A. F. (1996). Manufacture and use of tools in wild Sumatran orangutans. Naturwissenschaften, 83(4), 186–188. https://doi.org/10.1007/BF01143062
Vogel, E. R., Alavi, S. E., Utami-Atmoko, S. S., van Noordwijk, M. A., Bransford, T. D., Erb, W. M., Zulfa, A., Sulistyo, F., Farida, W. R., & Rothman, J. M. (2017). Nutritional ecology of wild Bornean orangutans (Pongo pygmaeus wurmbii) in a peat swamp habitat: Effects of age, sex, and season. American Journal of Primatology, 79(4), e22618. https://doi.org/10.1002/ajp.22618
Zipple, M. N., Altmann, J., Campos, F. A., Cords, M., Fedigan, L. M., Lawler, R. R., Lonsdorf, E. V., Perry, S., Pusey, A. E., Stoinski, T. S., Strier, K. B., & Alberts, S. C. (2021). Maternal death and offspring fitness in multiple wild primates. Proceedings of the National Academy of Sciences, 118(1), e2015317118. https://doi.org/10.1073/pnas.2015317118
Acknowledgments
We thank the Editor-in-Chief, Joanna Setchell, and Andreas Berghänel and an anonymous reviewer for the helpful comments they provided in improving the manuscript. We thank all the dedicated field staff, students, and research assistants (in particular, Armas Fitra, Adami Khairul, Fikar Zulkarnean, Julia Kunz, Lara Nellissen, Mudin, Natasha Bartolotta, Saidi Agan, Toni M. Sunil, and Ulil Azhari) who contributed to the data collected for this study at Suaq. We are grateful to the Indonesian State Ministry for Research and Technology (BRIN-RISTEK), the Directorate General of Natural Resources and Ecosystem Conservation–Ministry of Environment and Forestry of Indonesia (KSDAE-KLHK), the Ministry of Internal Affairs, Indonesia, the Sumatran orangutan conservation Program (SOCP), and Balai Besar Taman Nasional Gunung Leuser (BBTNGL) in Medan for their permission and support to conduct this study. We thank Universitas Nasional (UNAS) for support and collaboration. We thank the Max Planck Institute of Animal Behavior, University of Zurich, A.H. Schultz Foundation, Leakey Foundation (Primate Research Fund and project grant), SUAQ Foundation, and the Volkswagen Stiftung (Freigeist fellowship to CS) for financial support. Open access funding was provided by the Max Planck Institute of Animal Behavior.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Conceptualization: TR, MvN, CS.
Data curation: TR, CS.
Data collection: DP, CS
Formal analysis: TR, RM, PB, CS.
Funding acquisition: CS.
Investigation: TR, RM, SSUA, PB, MvN, CS.
Field project administration: SSUA, DP, CS.
Supervision: RM, CS.
Writing—original draft: TR.
Writing—review & editing: TR, RM, SSUA, PB, MvN, CS.
Corresponding author
Ethics declarations
Inclusion and Diversity Statement
The author list includes contributors from the location where the research was conducted, who participated in data collection, interpretation of the findings, and long-term project administration.
Additional information
Handling Editor: Joanna (Jo) M. Setchell


Badge earned for open practices: Open Data Badge and Open Code Badge. Experiment materials and data are available in the repository at https://doi.org/10.7910/DVN/8WV8XX.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Revathe, T., Mundry, R., Utami-Atmoko, S.S. et al. Maternal Behavior in Sumatran Orangutans (Pongo abelii) is Modulated by Mother-Offspring Characteristics and Socioecological Factors. Int J Primatol (2024). https://doi.org/10.1007/s10764-024-00435-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10764-024-00435-5