Abstract
Grassland ecosystems are species-rich habitats that are rapidly declining globally posing serious concerns for biodiversity conservation. This situation is particularly relevant in agricultural areas in Europe. As traditional management practices and livestock grazing regimes ceased, rewilding could be a potential avenue to tackle current biodiversity declines. To test this hypothesis, we set up a 3-year experiment where 12 horses were introduced in three 10-hectare enclosure replicates (four horses per enclosure). Horses were kept without supplementary feeding to mimic ecosystem functions of wild horses. We applied Generalized Linear Mixed Effects Models and a backward stepwise model selection procedure to elucidate factors that modulate insect richness induced by grazing. Our results show that plant species richness, the proportion of flowers and plant height play a significant role for butterfly and bumblebee richness, while the opposite effect was detected for grasshoppers. However, the effect on grasshoppers was counterbalanced by increased grasshopper species richness in habitats adjacent to horse latrines.
Implications for insect conservation
Rewilding with horses may offset current biodiversity declines by maintaining important functional links between plants and pollinators in grassland ecosystems. Horse grazing can however have different effects on diverse functional groups of insects. Application of integrative landscape scale approaches may be needed to elucidate the effects of rewilding for certain functional groups such as grasshoppers. With current biodiversity declines, up-scaling rewilding research and practice might be crucial to mitigate the pervasive effects on insects as their services and functions are critical for our existence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The biological diversity of grassland habitats likely evolved symbiotically with large herbivores and was largely determined by climate and other abiotic factors such as fire, flooding and storms (Bond 2005; Bond et al. 2005; Estes et al. 2011). Prehistoric megaherbivore faunas were however drastically depleted as humans expanded globally (Dirzo et al. 2014; Sandom et al. 2014), which resulted in a progressive simplification of megafaunas in ecosystems with cascade effects on plant community composition, vegetation structure and fire regimes (Gill 2014; Rouet-Leduc et al. 2021). Such defaunation processes also produced ecological state shifts in different biomes (Barnosky et al. 2016) including the extinction of ecological interactions (Galetti et al. 2018). Subsequently, landscape changes were driven by human agriculturalists through animal and plant domestication, and agriculture and husbandry practices (Bocherens 2018). These processes were additionally intensified in the last century decimating grassland habitats and extirpating wild and domestic herbivores from landscapes (Cousins et al. 2015). To tackle current biodiversity declines, the concept of trophic rewilding as an ecological restoration strategy has been proposed (Navarro and Pereira 2012; Svenning et al. 2016). It focuses on introducing species to restore top-down interactions and associated trophic cascades to promote self-regulating biodiverse ecosystems (Pedersen et al. 2020; Svenning et al. 2016). In cases where the wild ancestor of a species is extinct, such as the horse, the introduction of an ecologically functional substitute may mitigate current biodiversity declines and restructure plant communities and insect diversity (Garrido et al. 2019, 2021).
Grazing by large herbivores is commonly applied for grassland management worldwide (Sjödin et al. 2008; Tälle et al. 2016; van Klink et al. 2015; Zhu et al. 2012) and can largely affect their community dynamics and ecosystem functioning (Diaz et al. 2007). Biodiversity conservation management strategies for grasslands have primarily been targeting vascular plants (Tälle et al. 2016; WallisDe Vries et al. 2002), while mutualistic plant-pollinator interactions are equally fundamental for the stability and functioning of ecosystems and intrinsically connected to changing land use practices (Aslan et al. 2013; Kremen et al. 2007). Therefore, flower-visiting species such as butterflies and bumblebees, should also be monitored to improve the understanding of how management practices affect biodiversity and ecosystem functioning (van Klink et al. 2015). Butterflies and bumblebees are key functional groups with different co-evolutionary relationships with plants (Alanen et al. 2011). They have additional specific properties that qualify them as suitable complementary taxon for biodiversity conservation assessments. They are well-studied compared to other taxonomic groups (Boggs et al. 2003), have shown to have a rapid response to environmental changes (Goulson et al. 2005; Thomas et al. 2004), and may be used as umbrella species for other insect taxa (Thomas, 2005).
Grasshoppers are another well-studied functional group in relation to presence of large herbivores (van Klink et al. 2020, 2015; Nickell et al. 2018). In addition to butterflies and bumblebees, many grasshoppers and bush crickets (hereafter referred to as ‘grasshoppers’) are herbivorous species associated with grassland habitats and thus directly influenced by grazing pressure. Due to different habitat requirements at larval developmental stages, as well as nutrient requirements determined by their distinct phenologies among grasshopper species, a mosaic of habitats might be required to complete their life cycle (Adu-Acheampong et al. 2016). As such this functional group is a suitable complement for biodiversity research in relation to herbivory. Additionally, grasshopper species richness has been shown to increase in grazed grasslands and with vegetation heterogeneity, while too intensive grazing may disrupt beneficial plant–insect interactions (Kruess and Tscharntke 2002). Since grasshopper species richness might benefit from vegetation heterogeneity, rewilding efforts may offer opportunities for improving grasshopper diversity (cf. van Klink and WallisDe Vries 2018).
Research on grazing by large herbivores has either been focused on grassland plant diversity (Tälle et al. 2016), or on specific focal insect groups such as butterflies (Öckinger et al. 2006), bumblebees (Redpath et al. 2010) and grasshoppers (Almásy et al. 2021); particularly under seasonal (normally summer) grazing regimes by domestic livestock. Thus, there is a need to investigate the effects of year-round grazing on diverse functional groups of insects under experimental rewilding conditions. The importance of large herbivore grazing on grassland ecosystem functioning, as well as the positive effects on plant species richness and pollinators has already been documented (Garrido et al. 2019). However, grasshopper diversity may primarily be modulated by plant structural diversity in grazed grasslands (Zhu et al. 2012). Indeed, large herbivores can affect the structural diversity of grasslands by exhibiting spatial selectivity in feeding, defecation and wallowing (van Klink et al. 2015).
As grazing has positive effects on plant species richness, and particularly forbs, we hypothesize that plant species richness may favour pollinator richness (butterfly and bumblebee). A reduction in plant height induced by grazing may also have positive effects on pollinators as this may facilitate floral detection opportunities. However, such effects might be detrimental for grasshoppers as they are not dependent on floral resources. A growing body of evidence suggests widespread losses in insect abundance, biomass and species richness, in particular in agricultural areas in Europe (Hallmann et al. 2017; Seibold et al. 2019). Such declines are of paramount importance as insects play critical roles in ecosystems including pollination services and food supply for many other species (Wagner 2020). Thus, further research and implementation of large scale rewilding initiatives is urgent and crucial to mitigate current unprecedented biodiversity declines.
Methods
Study area and experimental design
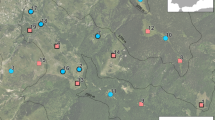
The study area was located at Krusenberg estate, 17 kms south of Uppsala, Sweden (59° 44′ N 17° 40′ E) (see Fig. 1a). The estate contains a total of 204 ha of agricultural land, 72 ha of pasture and grasslands, 510 ha of forest and, 46 ha correspond to other land uses (Päiviö 2008). Here a 3-year experiment was conducted at three different 10 ha wood-pasture enclosures (Fig. 1a), where four one-year old horse stallions of the national breed Gotland Russ (average stocking rate 0.35 horse/ha; average body mass 250 kg/horse) were introduced per enclosure in May 2014 and kept until September 2016. The horses were kept on year-round grazing without supplementary feeding (except a salt- and trace mineral block) in order to mimic ecosystem functions of wild horses. The experimental area is defined as a wood-pasture mosaic where forest dominated areas are interspersed with grasslands. From 2004 to 2014 (until the experiment started), the experimental area was partially harvested and/or grazed (including the forest) by cattle and pasturelands were not tilled (Ryberg, pers.comm.). The area is located within the hemiboreal zone (Ahti et al. 1968) with mean temperature of − 4.8 °C (± 6.5 SD) in January and 17.5 °C (± 4.1 SD) in July. Rainfall ranged from 65 to 123 mm in July.

© Lantmäteriet. Red asterisks in grasslands represent approximate locations of latrines surveyed in 2016. b Vegetation and grasshopper surveys. Plant species were surveyed in seven paired grazed and ungrazed plots (25 dm2) equidistant 2.5 m. For grasshoppers, every second grassland plot was surveyed (green quadrats) equidistant 5 m. c Butterfly and bumblebee surveys were performed in 5 × 5 m plots shaded in grey in both grazed and ungrazed areas. Figure adapted from Garrido et al. (2019). (Color figure online)
a Location of the study area and experimental design at Krusenberg estate, 17 kms south of Uppsala, Sweden (59° 44′ N 17° 40′ E).
Grassland habitat surveys
In each of the three enclosures, three rectangular 20 × 5 m exclosures were surveyed on grassland dominated areas. Within each exclosure, vegetation surveys were performed in seven permanent inventory plots (0.25 m2 [0.282 m radius]) equidistant 2.5 m; these were paralleled with another seven plots on grazed areas (see Fig. 1a, b). Grassland vegetation surveys were performed in July and September 2014, and in May, July and September during 2015 and 2016 (N = 1006). Plot centres were marked with black plastic needles hammered into the soil. For each plot pair (grazed-ungrazed) all plants were identified at species level and their abundance recorded. Grasses were recorded as a group, including Alopecurus pratensis, Festuca rubra, Dactylis glomerata, Phleum pretense, Festuca pratensis, Poa annua, Lolium perenne, Elytrigia repens, Agrostis gigantea, Agrostis capillaris, Deschampsia cespitosa. Additionally, the proportion of flowers per plot, vegetation height, proportion of mosses and lichens, exposed bare ground, number of pellets (dung) and litter was recorded (see Table 1). Vegetation height was measured with a herbometer (Herbometre, AGRO-Systémes, La membrolle sur Chosille, France). Soil compaction was obtained with a penetrometer. Vegetation height and soil compaction were measured one decimetre from the centre of the inventory spot.
Butterfly and bumblebee surveys
For butterflies and bumblebees, a point inventory method was applied [see Swedish Butterfly Monitoring Scheme (www.dagfjarilar.lu.se/english)]. Adjacent equally sized grazed-ungrazed 5 × 5 m plots were simultaneously observed for 20 min twice a day (morning and afternoon) and three times a year (May, July and September) in 2015 and 2016 (N = 216; Fig. 1c). Surveys were performed depending on sunlight and solar time; criteria described in the National Inventory of Landscapes in Sweden, i.e., at least 17 °C and preferably sunshine (Cornvall 2017). For each 20 min observation survey, butterfly and bumblebee species were identified, and total number of species recorded.
Grasshopper surveys
Grasshoppers were surveyed both inside and outside exclosures using a 1 m2 box quadrat with cotton-clothed sides of 60 cm height, centred upon every second grassland plot, equidistant 5 m (method described in Gardiner and Hill 2006, see ESM, Illustration S1; Fig. 1b). Grasshopper species were trapped within the box quadrat, and then flushed from the sward using a pole, which facilitated species identification and count. Surveys were performed in August–September 2015 and 2016, and between 11.00 and 15.30 to avoid low temperatures and maximize sun-exposure and, thus, insect activity. Individuals were identified at species level using a specific key for grasshopper species identification in Sweden (Strid 2010), and their abundance recorded. In 2016 we additionally surveyed latrine habitats, i.e., habitats adjacent to where horses aggregated their faecal droppings. The closest latrine to each exclosure was selected, i.e., three latrines per enclosure, and four plots equidistant 5 m along a 20 m line were sampled (see Fig. 1a for approximate location of surveyed latrines). This mimicked the way of previous grasshopper surveys both inside and outside exclosures (grazed vs ungrazed areas). Such data was not included for modelling purposes as it was just available for 2016. Due to the abundance of latrines created by horses we wanted to investigate their effect on grasshopper species richness.
Statistical analyses
Due to the characteristics of the experimental design, we applied a general hierarchical statistical approach (plot within exclosure and enclosure). We used Generalized Linear Mixed Effects Models (GLMMs) with a nested random structure term (plot within exclosure and enclosure) fitted to a Poisson distribution (log-link) to model butterfly, bumblebee and grasshopper species richness as response variable. For this, we used the function glmer in package “lme4” (Bates et al. 2015). In order to model the vegetation data together with the insect data, vegetation surveys were aggregated by exclosure and merged to the butterfly, bumblebee and grasshopper data in order to be comparable. We first inspected correlations between variables (see ESM, Figure S1a, b, c), setting a Spearman coefficient threshold of 0.6 to avoid the inclusion of correlated factors and thus minimizing the risk of making erroneous ecological conclusions (Zuur et al. 2009, 2010). This resulted in the exclusion of the factors soil compaction, proportion (%) of bare ground and proportion (%) of litter per plot for further analysis. We additionally checked for outliers using a boxplot function. To understand which habitat factors had a significant effect on the richness of butterfly, bumblebee and grasshopper species, we performed a backward stepwise model selection procedure based on parsimony principles (Sober 1981). In addition, we tested whether grasshopper species richness was affected by the experimental treatment (grazed vs ungrazed) and time, fitting a GLMM with a Poisson distribution (log-link) and a nested random structure term (plot within exclosure and enclosure). For analyses and plotting R version 4.0.5 (R Core Team 2021) was used.
Results
The most parsimonious model for butterflies retained the factors plant species richness, proportion of flowers per plot and plant height (see Table 2). Plant species richness and proportion of flowers per plot were positively related to butterfly species richness, while plant height showed no relation to butterfly species richness (Table 2). For bumblebees, the proportion of flowers, and mosses and lichens supported bumblebee species richness, whereas plant height exerted the opposite effect (Table 2).
Grasshopper species richness declined with time (β = − 0.80, SE = 0.39, z value = − 2.04, p value = 0.04) in both grazed and ungrazed conditions (β = 0.22, SE = 0.29, z value = 0.76, p value = 0.45; Fig. 2). However, after 3 years of experimental treatment, grasshopper species richness was double in latrine habitats (compared to grazed and ungrazed habitats; Fig. 3). Two grasshopper species (e.g., Decticus verrucivorus and Metrioptera roeselii) were only observed in latrine habitats. Species richness for grasshoppers was best explained by plant species richness, number of flowers and the amount of dung per plot. The first two factors were negatively related to grasshopper species richness while the amount of dung had a positive effect (Table 2; Fig. 3). This corroborates the observations around latrine habitats in 2016 (Fig. 3).
Discussion
We demonstrate that experimental rewilding with horses can have significantly different effects on diverse functional groups of insects. Our results show that butterfly and bumblebee species seem to be favoured by a higher proportion of flowers and lower plant height, and butterflies also benefited from a higher plant species richness. The very same factors however (plant species richness and number of flowers) have the opposite effect for grasshoppers (see Table 2). Nevertheless, that effect was counterbalanced by the amount of dung provided to the system (see Table 2, Fig. 3). Indeed, surveyed latrine habitats created by horses were found to have a higher species richness, with two grasshopper species only observed in such habitats (Fig. 3). For instance, Decticus verrucivorus is a habitat specialist species that requires bare ground, low herb-rich turf, and taller tussocky grassland occurring in close proximity (Gardiner 2018; Fartmann et al. 2012); such optimal habitat characteristics may have occurred around latrine habitats which might explained the species presence. This calls to broaden our perspectives when assessing the effect of rewilding in ecosystems and to apply landscape scale approaches to fully understand the effect of reintroduced species. These results have to be taken with caution however, as it has been suggested that latrines created by horses may be the result of an enclosure-effect (Lamoot et al. 2004), which impel us to investigate the matter under true rewilding conditions (free ranging animals).
Species used for rewilding, such as the European bison (Bison bonasus L.) and rustic cattle- and horse breeds, have functionally diverse diets and should thus be considered when designing future rewilding actions (Cromsigt et al. 2018). Support for this view was found in a meadow steppe in China, where insect abundance responded differently as function of herbivore species and insect order (Zhu et al. 2015). For instance, Orthoptera and Homoptera species abundance increased under sheep grazing, while Coleoptera and Diptera increased under cattle grazing, and Lepidoptera under goat grazing. Therefore, different feeding guilds profoundly changed the grassland insect community (Zhu et al. 2015). In addition to feeding guild, herbivore density, body mass and digestive physiology are important factors to account for in rewilding interventions as they may determine the effect of the novel herbivore assemblage on insect diversity (Cromsigt et al. 2018).
For different functional groups of pollinators, i.e., butterflies and bumblebees, the proportion of flowers, and plant height as well as plant species richness for butterflies were positively related to pollinator species richness. This might be associated with the significant effects that grazers may have in ecosystems. Indeed, a recent experiment has shown that horses significantly changed the functional composition of grasslands, mitigated plant species declines, in particular bee-dependent plants, and boosted pollinator habitat use (Garrido et al. 2019). This correlates with previous studies by Zhu et al. (2012) who found that flower proportion and plant structural diversity were the most important factors explaining insect diversity in grazed grasslands.
Pollinator species richness decreased with plant height as shorter plants might facilitate flower detection opportunities whereas areas with taller plants, i.e., ungrazed or avoided areas, might be dominated by competitive plant species which may hinder flower detection (Borer et al. 2014). Butterfly species richness increased with plant richness while the opposite effect was detected for grasshoppers. This might be a direct effect of grazing, as grazing with horses may induce a functional change of the grassland community, favouring a richer, more palatable (higher Specific Leaf Area (SLA)) and shorter vegetation, characteristic of ruderal communities (cf. Garrido et al. 2019). This has direct effects on ecosystem functioning as SLA is associated to key ecosystem processes such as litter decomposition and productivity (Lavorel and Garnier 2002; Pérez-Harguindeguy et al. 2000). Moreover, ruderal communities have higher net nitrogen mineralization rates (Mattson 1980) which positively correlates with above-ground net primary productivity (Hunt et al. 1988; Reich et al. 1997). Indeed, nitrogen content in plants, i.e., enhanced plant quality, appears to be an important driver in mediating the negative effect of large herbivore grazing on grasshoppers (Zhu et al. 2019). Such an effect might be modulated not only by grazing-induced changes in plant nutritional content (plant nutritional changes occurred at our experimental site, cf. Ringmark et al. 2019), but also by the unique nutritional niches of different grasshopper species (Zhu et al. 2020). For example, Zhu et al. (2020) found that cattle grazing had a positive effect on the early season grasshopper Euchorthippus cheui, whereas it suppressed the late season grasshopper E. unicolor. These results suggest that insects belonging to the same herbivore guild can have opposite nutrient requirements determined by their distinct phenologies (Zhu et al. 2020), which might explain the general decline of grasshopper species richness in our rewilding experiment, and the enhanced diversity found around nitrogen-rich horse-created latrine habitats. Our results on grasshoppers have to be interpreted with caution however, as the grasshopper community was solely surveyed once a year in August–September (in contrast to the surveys of pollinators in May, July and September), and thus early season species might have been under-represented in our data. Similarly, the surveys were performed near (20 m, see Fig. 1a) forest dominated areas and due to different habitat requirements at larval developmental stages, and distinct species niche breath, might result in the occurrence of grasshopper species outside grassland dominated areas that were beyond the focus of the present study.
Rewilding has the potential to mitigate biodiversity declines (Bakker and Svenning 2018). However, scientific rewilding experiments are still scarce (cf. Svenning et al. 2016) albeit crucial to advance rewilding-related science and implementation (see Garrido et al. 2019). It is important to note that horses in this experiment were not truly rewilded, rather experimentally kept on year-round grazing without supplementary feeding. They were additionally inspected daily, and provided with water, salt- and mineral blocks and shelter. The effect of reintroducing an ecological replacement of an extinct large herbivore may vary among different functional groups of insects as well as herbivore feeding guilds, body mass, herbivore densities, and digestive physiology. This calls for implementing more integrative landscape scale research approaches to better understand the complex interactions between large herbivores and insect communities as grazing might modulate functional compositional changes of grasslands which may affect plant phenology and therefore mediate insect physiological needs to local resource availabilities. Up-scaling rewilding actions may become crucial to advance our knowledge on the effects of rewilding initiatives and to palliate current unprecedented biodiversity declines in the Anthropocene.
References
Adu-Acheampong S, Bazelet CS, Samways MJ (2016) Extent to which an agricultural mosaic supports endemic species-rich grasshopper assemblages in the Cape Floristic Region biodiversity hotspot. Agric Ecosyst Environ 227:52–60. https://doi.org/10.1016/j.agee.2016.04.019
Ahti T, Hämet-Ahti L, Jalas J (1968) Vegetation zones and their sections in northwestern Europe. In: Annales Botanici Fennici, pp 169–211
Alanen E, Hyvönen T, Lindgren S, Härmä O, Kuussaari M (2011) Differential responses of bumblebees and diurnal Lepidoptera to vegetation succession in long-term set-aside. J Appl Ecol 48(5):1251–1259
Almásy J, Essl F, Berger A, Schulze CH (2021) To graze or to mow? The influence of grassland management on grasshoppers (Orthoptera) on a flood protection embankment in the Donau-Auen National Park (Austria). J Insect Conserv 25(4):707–717. https://doi.org/10.1007/s10841-021-00337-4
Aslan CE, Zavaleta ES, Tershy B, Croll D (2013) Mutualism disruption threatens global plant biodiversity: a systematic review. PLoS ONE 8(6):e66993
Bakker ES, Svenning J-C (2018) Trophic rewilding: Impact on ecosystems under global change. Philos Trans R Soc B 373(1761):20170432. https://doi.org/10.1098/rstb.2017.0432
Barnosky AD, Lindsey EL, Villavicencio NA, Bostelmann E, Hadly EA, Wanket J, Marshall CR (2016) Variable impact of late-Quaternary megafaunal extinction in causing ecological state shifts in North and South America. Proc Natl Acad Sci 113(4):856–861
Bates D, Maechler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48. https://doi.org/10.18637/jss.v067.i01
Bocherens H (2018) The rise of the Anthroposphere since 50,000 years: an ecological replacement of megaherbivores by humans in terrestrial ecosystems? Front Ecol Evol 6:3. https://doi.org/10.3389/fevo.2018.00003
Boggs CL, Watt WB, Ehrlich PR (2003) Butterflies: ecology and evolution taking flight. The University of Chicago Press, Chicago
Bond WJ (2005) Large parts of the world are brown or black: a different view on the ‘Green World’ hypothesis. J Veg Sci 16(3):261–266
Bond WJ, Woodward FI, Midgley GF (2005) The global distribution of ecosystems in a world without fire. New Phytol 165(2):525–538. https://doi.org/10.1111/j.1469-8137.2004.01252.x
Borer ET, Seabloom EW, Gruner DS, Harpole WS, Hillebrand H, Lind EM, Adler PB, Alberti J, Anderson TM, Bakker JD, Biederman L, Blumenthal D, Brown CS, Brudvig LA, Buckley YM, Cadotte M, Chu C, Cleland EE, Crawley MJ, Yang LH (2014) Herbivores and nutrients control grassland plant diversity via light limitation. Nature 508:517. https://doi.org/10.1038/nature13144
Cornvall E (2017) Fältinstruktion för fjärilar och humlor i ängs- och betesmarker 2017 [Instructions for monitoring butterflies and bumble bees in pasturelands] (p 48). Institutionen för skoglig resurshushållning
Cousins SAO, Auffret AG, Lindgren J, Tränk L (2015) Regional-scale land-cover change during the 20th century and its consequences for biodiversity. Ambio 44(1):17–27. https://doi.org/10.1007/s13280-014-0585-9
Cromsigt JPGM, Kemp YJM, Rodriguez E, Kivit H (2018) Rewilding Europe’s large grazer community: how functionally diverse are the diets of European bison, cattle, and horses? Restor Ecol 26(5):891–899. https://doi.org/10.1111/rec.12661
Diaz S, Lavorel S, McIntyre S, Falczuk V, Casanoves F, Milchunas DG, Skarpe C, Rusch G, Sternberg M, Noy-Meir I (2007) Plant trait responses to grazing–a global synthesis. Glob Change Biol 13(2):313–341
Dirzo R, Young HS, Galetti M, Ceballos G, Isaac NJ, Collen B (2014) Defaunation in the Anthropocene. Science 345(6195):401–406
Estes JA, Terborgh J, Brashares JS, Power ME, Berger J, Bond WJ, Carpenter SR, Essington TE, Holt RD, Jackson JBC, Marquis RJ, Oksanen L, Oksanen T, Paine RT, Pikitch EK, Ripple WJ, Sandin SA, Scheffer M, Schoener TW, Wardle DA (2011) Trophic downgrading of planet earth. Science 333(6040):301–306. https://doi.org/10.1126/science.1205106
Fartmann T, Krämer B, Stelzner F, Poniatowski D (2012) Orthoptera as ecological indicators for succession in steppe grassland. Ecol Indic 20:337–344. https://doi.org/10.1016/j.ecolind.2012.03.002
Galetti M, Moleón M, Jordano P, Pires MM, Guimaraes PR Jr, Pape T, Nichols E, Hansen D, Olesen JM, Munk M (2018) Ecological and evolutionary legacy of megafauna extinctions. Biol Rev 93(2):845–862
Gardiner T (2018) Grazing and Orthoptera: a review. J Orthoptera Res 27:3–11
Gardiner T, Hill J (2006) A comparison of three sampling techniques used to estimate the population density and assemblage diversity of Orthoptera. J Orthoptera Res 15(1):45–51. https://doi.org/10.1665/1082-6467(2006)15[45:ACOTST]2.0.CO;2
Garrido P, Mårell A, Öckinger E, Skarin A, Jansson A, Thulin C-G (2019) Experimental rewilding enhances grassland functional composition and pollinator habitat use. J Appl Ecol 56(4):946–955. https://doi.org/10.1111/1365-2664.13338
Garrido P, Edenius L, Mikusiński G, Skarin A, Jansson A, Thulin C-G (2021) Experimental rewilding may restore abandoned wood-pastures if policy allows. Ambio 50(1):101–112. https://doi.org/10.1007/s13280-020-01320-0
Gill JL (2014) Ecological impacts of the late Quaternary megaherbivore extinctions. New Phytol 201(4):1163–1169
Goulson D, Hanley ME, Darvill B, Ellis J, Knight ME (2005) Causes of rarity in bumblebees. Biol Conserv 122(1):1–8
Hallmann CA, Sorg M, Jongejans E, Siepel H, Hofland N, Schwan H, Stenmans W, Müller A, Sumser H, Hörren T, Goulson D, de Kroon H (2017) More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 12(10):e0185809. https://doi.org/10.1371/journal.pone.0185809
Hunt H, Ingham E, Coleman D, Elliott E, Reid C (1988) Nitrogen limitation of production and decomposition in prairie, mountain meadow, and pine forest. Ecology 69(4):1009–1016
Kremen C, Williams NM, Aizen MA, Gemmill-Herren B, LeBuhn G, Minckley R, Packer L, Potts SG, Steffan-Dewenter I, Vázquez DP (2007) Pollination and other ecosystem services produced by mobile organisms: a conceptual framework for the effects of land-use change. Ecol Lett 10(4):299–314
Kruess A, Tscharntke T (2002) Contrasting responses of plant and insect diversity to variation in grazing intensity. Biol Conserv 106(3):293–302. https://doi.org/10.1016/S0006-3207(01)00255-5
Lamoot I, Callebaut J, Degezelle T, Demeulenaere E, Laquière J, Vandenberghe C, Hoffmann M (2004) Eliminative behaviour of free-ranging horses: do they show latrine behaviour or do they defecate where they graze? Appl Anim Behav Sci 86(1):105–121. https://doi.org/10.1016/j.applanim.2003.12.008
Lavorel S, Garnier É (2002) Predicting changes in community composition and ecosystem functioning from plant traits: Revisiting the Holy Grail. Funct Ecol 16(5):545–556
Mattson WJ Jr (1980) Herbivory in relation to plant nitrogen content. Annu Rev Ecol Syst 11(1):119–161
Navarro LM, Pereira HM (2012) Rewilding abandoned landscapes in Europe. Ecosystems 15(6):900–912. https://doi.org/10.1007/s10021-012-9558-7
Nickell Z, Varriano S, Plemmons E, Moran MD (2018) Ecosystem engineering by bison (Bison bison) wallowing increases arthropod community heterogeneity in space and time. Ecosphere 9(9):e02436. https://doi.org/10.1002/ecs2.2436
Öckinger E, Eriksson AK, Smith HG (2006) Effects of grassland abandonment, restoration and management on butterflies and vascular plants. Biol Conserv 133(3):291–300. https://doi.org/10.1016/j.biocon.2006.06.009
Päiviö E-L (2008) Det agrara landskapet på vinst och förlust. Biologiska och historiska värden inom lantbrukets nya uppdrag. In: Faculty of natural Resources and Agricultural Sciences: vol Doctoral thesis No. 2008:95. Swedish University of Agricultural Sciences
Pedersen PBM, Ejrnæs R, Sandel B, Svenning J-C (2020) Trophic Rewilding Advancement in Anthropogenically Impacted Landscapes (TRAAIL): a framework to link conventional conservation management and rewilding. Ambio 49(1):231–244. https://doi.org/10.1007/s13280-019-01192-z
Pérez-Harguindeguy N, Díaz S, Cornelissen JH, Vendramini F, Cabido M, Castellanos A (2000) Chemistry and toughness predict leaf litter decomposition rates over a wide spectrum of functional types and taxa in central Argentina. Plant Soil 218(1):21–30
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Redpath N, Osgathorpe LM, Park K, Goulson D (2010) Crofting and bumblebee conservation: the impact of land management practices on bumblebee populations in northwest Scotland. Biol Conserv 143(2):492–500. https://doi.org/10.1016/j.biocon.2009.11.019
Reich PB, Grigal DF, Aber JD, Gower ST (1997) Nitrogen mineralization and productivity in 50 hardwood and conifer stands on diverse soils. Ecology 78(2):335–347
Ringmark S, Skarin A, Jansson A (2019) Impact of year-round grazing by horses on pasture nutrient dynamics and the correlation with pasture nutrient content and fecal nutrient composition. Animals 9(8):500
Rouet-Leduc J, Peer G, Moreira F, Bonn A, Helmer W, Shahsavan Zadeh SAA, Zizka A, van der Plas F (2021) Effects of large herbivores on fire regimes and wildfire mitigation. J Appl Ecol 58(12):2690–2702. https://doi.org/10.1111/1365-2664.13972
Sandom C, Faurby S, Sandel B, Svenning J-C (2014) Global late quaternary megafauna extinctions linked to humans, not climate change. Proc R Soc B 281(1787):20133254
Seibold S, Gossner MM, Simons NK, Blüthgen N, Müller J, Ambarlı D, Ammer C, Bauhus J, Fischer M, Habel JC, Linsenmair KE, Nauss T, Penone C, Prati D, Schall P, Schulze E-D, Vogt J, Wöllauer S, Weisser WW (2019) Arthropod decline in grasslands and forests is associated with landscape-level drivers. Nature 574(7780):671–674. https://doi.org/10.1038/s41586-019-1684-3
Sjödin NE, Bengtsson J, Ekbom B (2008) The influence of grazing intensity and landscape composition on the diversity and abundance of flower-visiting insects. J Appl Ecol 45(3):763–772. https://doi.org/10.1111/j.1365-2664.2007.01443.x
Sober E (1981) The principle of parsimony. Br J Philos Sci 32(2):145–156. https://doi.org/10.1093/bjps/32.2.145
Strid T (2010) Gräshoppor i Sverige: En fälthandbok. Entomologiska föreningen
Svenning J-C, Pedersen PBM, Donlan CJ, Ejrnæs R, Faurby S, Galetti M, Hansen DM, Sandel B, Sandom CJ, Terborgh JW, Vera FWM (2016) Science for a wilder Anthropocene: synthesis and future directions for trophic rewilding research. Proc Natl Acad Sci 113(4):898–906. https://doi.org/10.1073/pnas.1502556112
Tälle M, Deák B, Poschlod P, Valkó O, Westerberg L, Milberg P (2016) Grazing vs. Mowing: a meta-analysis of biodiversity benefits for grassland management. Agric Ecosyst Environ 222:200–212. https://doi.org/10.1016/j.agee.2016.02.008
Thomas JA (2005) Monitoring change in the abundance and distribution of insects using butterflies and other indicator groups. Philos Trans R Soc Lond B 360:339–357
Thomas JA, Telfer MG, Roy DB, Preston CD, Greenwood J, Asher J, Fox R, Clarke RT, Lawton JH (2004) Comparative losses of British butterflies, birds, and plants and the global extinction crisis. Science 303(5665):1879–1881
van Klink R, van der Plas F, Van Noordwijk C, WallisDeVries MF, Olff H (2015) Effects of large herbivores on grassland arthropod diversity. Biol Rev 90(2):347–366
van Klink R, WallisDeVries MF (2018) Risks and opportunities of trophic rewilding for arthropod communities. Philos Trans R Soc B 373(1761):20170441. https://doi.org/10.1098/rstb.2017.0441
van Klink R, van Laar-Wiersma J, Vorst O, Smit C (2020) Rewilding with large herbivores: positive direct and delayed effects of carrion on plant and arthropod communities. PLoS ONE 15(1):e0226946. https://doi.org/10.1371/journal.pone.0226946
Wagner DL (2020) Insect declines in the Anthropocene. Annu Rev Entomol 65(1):457–480. https://doi.org/10.1146/annurev-ento-011019-025151
WallisDeVries MF, Poschlod P, Willems JH (2002) Challenges for the conservation of calcareous grasslands in northwestern Europe: integrating the requirements of flora and fauna. Biol Conserv 104(3):265–273. https://doi.org/10.1016/S0006-3207(01)00191-4
Zhu H, Wang D, Wang L, Bai Y, Fang J, Liu J (2012) The effects of large herbivore grazing on meadow steppe plant and insect diversity. J Appl Ecol 49(5):1075–1083. https://doi.org/10.1111/j.1365-2664.2012.02195.x
Zhu H, Wang D, Guo Q, Liu J, Wang L (2015) Interactive effects of large herbivores and plant diversity on insect abundance in a meadow steppe in China. Agric Ecosyst Environ 212:245–252. https://doi.org/10.1016/j.agee.2015.07.008
Zhu Y, Zhong Z, Pagès JF, Finke D, Wang D, Ma Q, Hassan N, Zhu H, Wang L (2019) Negative effects of vertebrate on invertebrate herbivores mediated by enhanced plant nitrogen content. J Ecol 107(2):901–912. https://doi.org/10.1111/1365-2745.13100
Zhu Y, Veen GF, Wang D, Wang L, Zhong Z, Ma Q, Li H, Li X, Pan D, Bakker ES (2020) Herbivore phenology can predict response to changes in plant quality by livestock grazing. Oikos 129(6):811–819. https://doi.org/10.1111/oik.07008
Zuur AF, Ieno EN, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer-Verlag, New York
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1(1):3–14. https://doi.org/10.1111/j.2041-210X.2009.00001.x
Acknowledgements
We thank Cecilia Rätz for floral inventories and Sofie Fröberg for butterfly and bumblebee inventories. The World Wildlife Fund (WWF) Sweden, Helge Ax:son Johnsons Foundation, Knut and Alice Wallenberg Foundation and, a crowd funding project at Swedish University of Agricultural Sciences (SLU) funded the project. We are also grateful to the Division of Estate Management at SLU for enclosure management in kind. The manuscript was drafted during a writing week at the Sigtuna Foundation financed with a grant from the Faculty of Veterinary Medicine and Animal Science, SLU. We additionally thank two anonymous reviewers for suggestions that improved the manuscript. The experiment with horses was approved by Uppsala Ethical Committee, Approval Number C28/14.
Funding
Open access funding provided by Swedish University of Agricultural Sciences. The research was funded by grants from WWF Sweden, Helge Ax:son Johnsons Foundation, Knut and Alice Wallenberg Foundation and a crowd funding project at Swedish University of Agricultural Sciences (SLU). The Division of Estate Management at SLU supported in kind with enclosure management. The manuscript was drafted during a writing week at the Sigtuna Foundation financed with a grant from the Faculty of Veterinary Medicine and Animal Science, SLU.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
Authors have no competing interest(s).
Ethical approval
The experiment with horses was approved by the Uppsala Ethical Committee, Approval Number C28/14. All authors consent to publication and submission to Journal of Insect Conservation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Garrido, P., Naumov, V., Söderquist, L. et al. Effects of experimental rewilding on butterflies, bumblebees and grasshoppers. J Insect Conserv 26, 763–771 (2022). https://doi.org/10.1007/s10841-022-00420-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-022-00420-4