Abstract
The heterogeneity of microhabitats is an important driver of biodiversity. Therefore, conserving habitat heterogeneity at multiple spatial scales is essential to biodiversity conservation. This study investigates the effect and the conservation value of micromosaic heterogeneity provided by the heather (Calluna vulgaris) patches in an Eastern-European peat bog on the spider communities. We found that the heather patches strongly affect spiders; the species richness and abundance were higher in heather than in open bogs. Heather shrubs form a separate special microhabitat, which differs in structure, microclimate and litter quality, and has a significant positive effect on diversity of spiders. Furthermore, we collected smaller spiders and more web builders in heather sites than in control sites, presumably due to the different microhabitat structures and prey availability. However, habitat type did not affect the vertical microhabitat preference of spiders. The species composition of spider communities differed between the two microhabitat types. We identified seven significant indicator species in heather shrubs, whereas three species were significant indicators of control sites. The species richness and the presence of rare and endangered peat bog specialist spider species in heather patches highlight the high conservation value of peat bogs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Wetlands are critical ecosystems for biodiversity conservation (Dawson et al. 2003; Gopal and Ghosh 2008). About one-third of the wetlands are peat bogs, characterized by accumulated organic matter and acidic, stagnant water with low mineral content. Some are ombrotrophic, as they receive water and minerals primarily from precipitation (Charman 2002).
Although peat bogs cover less than 3% of Earth’s land surface, they provide several essential ecosystem services, including water storage, flood prevention, drinking water provisions, and carbon sequestration. Furthermore, they provide suitable habitats for many specialized and highly endangered plant and animal species (Bain et al. 2011; Evans et al. 2014; Harper et al. 2018). Peat bogs are declining throughout the world, but the most severe losses occurred in Europe, where they are currently considered among the most vulnerable and endangered habitats (Charman 2002; Riecken et al. 2006; Parish et al. 2008; Haase and Balkenhol 2015). Many species of the Red Data Book of Central and Northern European countries live in peat bogs (Gärdenfors 2000; Koponen 2002). Furthermore, the Habitats Directive of the European Union listed peat bogs as priority habitats in Annex 1 (92/43/EEC). The primary conservation threats to these habitats are drainage, peat harvesting, soil erosion and nutrient infiltration due to forest management and grazing, pollution from agriculture, increasing tourism, as well as the spread of invasive and problematic native species (Succow 1990; Buchholz 2016; Grzybowski and Glinska-Lewczuk 2020).
Although natural raised and basin bogs are primarily located in Northern Europe, the Carpathian Mountains also harbour several small, natural and semi-natural peat bogs. They are endangered by habitat loss, fragmentation, isolation and degradation (Pop 1960). Most of them are protected areas, natural or botanical reserves, included in the Natura 2000 ecological network that was created based on the Habitats Directive of the European Union. They are the ultimate refuge for some rare, glacial relict plant species with high conservation value (Rupreht & Szabó 1999, Margóczi et al. 2000). According to recent studies, many other organisms living in peat bogs are considered endangered. The invertebrate communities of peat bogs comprise bog specialist species, the spiders among them being the most representative of the predatory arthropod fauna (Coulson and Butterfield 1986; Gallé and Urák 2001, 2002, 2006; Urák and Samu 2008; Gallé et al. 2017, 2019, 2021). If natural conditions persist, peat bogs are stable ecosystems. However, if the conditions suffer any changes, a significant turnover occurs in species composition (Koponen 1979; Curtis and Corrigan 1990). In most cases, the number of typical bog-inhabiting species with a preference for open habitats decreases, while the number of eurytopic species increases with the appearance and the spreading of shrubs and trees (Buchholz 2016). Canopy closure and the microclimate affect the spider fauna of forested bogs (Gallé et al. 2021).
Habitat structure influences spider abundance and functional diversity (e.g. Lafage et al. 2019). Small-scale structural elements, such as shrubs, may have a disproportionately strong effect on the species composition of arthropods (Barton et al. 2009; Manning et al. 2006). Shrubs increase structural heterogeneity, shelter and web attachment points for spiders. Furthermore, shrubs provide food resources for herbivore arthropods that serve as food items for spiders, therefore, they affect spider species diversity, abundance and trait composition (Lewinsohn et al. 2005; Schirmel et al. 2012).
The heather (Calluna vulgaris) is a characteristic low-growing evergreen shrub in peat bogs. Peatlands dominated by heather are among the most distinctive and characteristic habitats of North-Western Europe, especially the sub-montane areas of the United Kingdom and the Republic of Ireland. Prescribed, regular and controlled burning, mowing, or cutting to maintain the heather’s density, structure, and age are integral aspects of grouse-moor management in these areas (Tucker 2003; Heinemeyer et al. 2019). In Southern and Central Europe, the heather is heather grows in post-pyrogenic sites of the peat bogs and considered extremely important in developing and persistence of the hummock-hollow pattern (Wallèn 1987).
This study aimed to investigate the role of heather patches in shaping the diversity pattern of spider assemblages and the conservation of bog specialist species of a intact peat bog in Romania. We hypothesized a higher abundance and species richness in heather than in open bog. Furthermore, we expected more web builders, vegetation-dweller species, and larger spiders in heather sites than in the control bog sites.
Materials and methods
Our study was conducted in the Vlășinescu peat bog, situated in the Eastern-Carpathians, on Izvoare volcanic plateau of the Igniş Mountains, close to the tourist complex of Izvoare, in the administrative territory of Desești municipality, Maramureș county (N 47°44,817’, E 023°43.378’, Fig. 1.). The bog was formed in an ancient volcano crater at the altitude of 881 m a.s.l., it has a surface of about 3.4 ha, and it is delimited by the Vlășinescu Stream on the north-west and by the Igniş Stream on the east. The total volume of the peat is about 50,000 m3, and the thickness of the peat layer varies between 3.5 and 5.5 m, with an average thickness of 2.9 m (Pop 1960).
The peat bog is included in a Nature reserve. The main Natura 2000 habitat is the 7110* Active raised bog, characterized by the plant associations Sphagnetum magellanici and Eriophoro vaginato-Sphagnetum. The plant list includes rare species such as S. magelanicum, S. recurvum, Drosera rotundifolia, Andromeda polifolia, Empetrum nigrum, Carex echinata, C. rostrata, Eriophorum vaginatum, Vaccinium uliginosum, V. vitis idaea, V. oxycoccus, Potentilla erecta, Lycopodium inundatum, etc. No invasive alien plant species were identified in the study area (Danci 2016).
Heather forms patches of different sizes, from single shrubs to patches of 4–5 m2. The sampling sites consisted of heather patches between one and two square meters. Furthermore, we sampled control sites between heather. Predominantly sphagnum mosses covered the ground of control sites. We used a ‘D-vac’ suction sampler to survey spiders. We conducted monthly samplings in the vegetation period of 2017, from April to October. We collected 10 samples from heather patches and 10 samples between them (control) (10 samples × two habitat types × 7 sampling periods = 140 samples in total). Each sample consisted of 10 subsamples, i.e. the ‘D-vac’ head was placed on the ground 10 times. We placed the collected spiders in 70:30 alcohol:water solution. All adult spider specimens were identified to species level using standard keys (Nentwig et al. 2019); however, juvenile spiders cannot be identified precisely to species level; therefore, we included only their abundance in further analyses. We pooled the data from the seven collection periods.
We used three functional traits (body size, hunting strategy, and stratum). The mean body size of each spider species was a continuous variable in mm based on literature data following Nentwig et al. (2019). To characterize the feeding trait of species, we used the hunting strategy of spiders (two categories: active hunters (code: 1) and web-builders (code: 2) following Cardoso et al. 2011). Finally, we described the vertical microhabitat preference of spiders with stratum (three categories: ground-dwellers (code: 1), herbaceous vegetation layer dwellers (code: 2), shrubs dwellers (code: 3) based on the classification by Buchar and Ruzicka 2002).
We used generalized linear models to investigate the effect of habitat type on the species richness and abundance of spiders. Our data did not meet normality, therefore we used the Poisson error term for species richness and the negative binomial error term for spiders’ abundance after checking for the data’s overdispersion. We calculated community weighted mean (CWM) values for body size and stratum at each site (Ricotta and Moretti 2011) using the R package ‘FD’ (Laliberte and Legendre 2010) and we calculated the percentage of web builders. We used a series of one-way ANOVAs to test the effect of habitat type on spider CWM values and percentage of web builders.
To explore and visualize the effect of heather shrubs on the species composition of spiders, we used non-metric multidimensional scaling with Bray-Curtis dissimilarity measure, and a maximum number of 25 random starts were used to search for a stable solution.
The species abundance data were transformed into relative species values by a Hellinger transformation before the ordination of the species abundance data. The Hellinger transformation is known to reduce the importance of species with high abundances (Legendre and Gallagher 2001). Permutational Multivariate Analysis of variances (PerMANOVA), based on Bray-Curtis dissimilarity matrices with 5000 permutations, was performed to test for differences among spider assemblages. To detect characteristic species for each habitat type, we calculated the indicator values of species (IndVal; Dufrene and Legendre 1997) with the ‘labdsv’ package (Roberts 2012).
Results
The arachnological material sampled in Vlășinescu peat bog comprised 6102 spider specimens, from which 1505 were adults (461 males and 1044 females), belonging to 93 species from 17 families. Out of these species Agyneta orites (Thorell, 1875), Glyphesis servulus (Simon, 1881) and Mermessus trilobatus (Emerton, 1882) from family Linyphiidae were new for the Romanian fauna (Weiss and Urák 2000).
We collected 4152 specimens (1046 adults) from 70 species from heather sites and 1950 specimens (459 adults) belonging to 59 species from control sites. Of these species, 36 were present in both studied sites, and we collected 57 species only in one of them: 34 in the heather sites and 23 in the control sites. The dominant species in heather sites was Minicia marginella (Wider, 1834) (480 individuals), Agyneta affinis (Kulczyński, 1898) (90 individuals), Pocadicnemis pumila (Blackwall, 1841) (76 individuals), from family Linyphiidae and Neon reticulatus (Blackwall, 1853) (54 individuals) from family Salticidae. In the control site, the dominant species were Pardosa pullata (Clerck, 1757) (111 individuals) from family Lycosidae and Minicia marginella (Wider, 1834) (71 individuals) from family Linyphiidae (Appendix 1.).
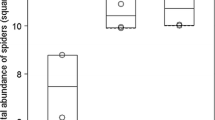
We detected a significant positive effect of heather shrubs on species richness (z = 47.52, p < 0.001, Fig. 2a) and on the abundance of spiders (z = 139.40, p < 0.001, Fig. 2b). We collected smaller spiders (F = 47.06, p < 0.001, Fig. 3a) and more web builders (F = 49.78, p < 0.001, Fig. 3b) in the heather than in control sites. However, habitat type did not affect vertical microhabitat preference of spiders, we did not detect more shrub-dweller species in heather than in control sites (F = 1.23, p = 0.281, Fig. 3c).
Species traits of spider communities. (A) CWM of body size (continuous; in mm), (B) CWM hunting strategy (active hunter: 1, web-builder: 2), (C) CWM stratum (ground-dwellers: 0, herbaceous vegetation layer: 1, shrubs: 2). Horizontal line is the median, boxes are the interquartile ranges, the whiskers extend to the extreme data point
Heather and control sites habitat types were well separated on the NMDS scatterplot (Fig. 4). Furthermore, this difference in species composition was significant according to the PerMANOVA (F = 22.70, p < 0.001). We identified seven significant indicator species in heather shrubs (Aculepeira ceropegia, Agyneta rurestris, Agyneta affinis, Clubiona trivialis, Evarcha arcuata, Minicia marginella, Pocadicnemis pumila), whereas three species were significant indicators of control sites (Araeoncus crassipes, Pardosa pullata, Hypsosinga albovittata; Appendix 1).
Discussion
Peat bogs of the Carpathians are only small fragments; however, they have a significant conservation value. European peat bogs harbour distinct spider species assemblages and specialist species (Buchholz 2016). The faunistic results in the Vlașinescu peat bog revealed a rich and diverse spider fauna and led to the report of some new and rare species to the Romanian fauna. The 93 species identified from our one-vegetation-period sampling of a relatively small peat bog (3.4 ha) represent a high species richness compared to the results of the other peatland surveys in Transylvania. In Mohos peat bog (80 ha), 96 species were identified. Spiders were sampled by pitfall traps, by hand, by sweep net, by sieving and by suction sampling in four different habitat types (Urák and Samu 2008). In Lucs peat bog (120 ha), 67 species were identified, collected by pitfall traps from three habitat types (Urák and Máthé 2013). In Poiana Stampei peat bog (695 ha), 71 species were collected from two habitat types (Gallé et al. 2021). Furthermore, Rėlys and Dapkus (2002) collected 55 species in an open bog and 57 species in a pine bog in southern Lithuania. Buchholz (2016) identified 105 species from a German bog forest. Therefore, the spider fauna of our bogs is rich, and Vlașinescu peat bog represents a high nature conservation value habitat despite its small size. Our study emphasizes that small peat bogs would conserve this specialized fauna when the habitats are undisturbed, and the hydrological properties and water quality are not affected (Gallé and Urák 2001, 2002, 2006).
There is a strong relationship between spider community structure and habitat properties. In peat bogs, the unique environmental conditions probably represent an environmental filter, which may exclude many generalist species, resulting in a spider fauna comprising specialists (Gallé et al. 2019). Therefore, spiders are excellent indicators of peatland habitats (McGeoch 1998, Scott 2006, Urák & Samu 2008, Samu & Urák 2014, Gallé et al. 2019), and they can also indicate the subtle and long-term changes in peat bog conditions (Haase and Balkenhol 2015). Previous studies in Transylvanian peat bogs revealed that the heterogeneity of the habitat increase species richness and abundance of spiders. Peatbog habitat types (e.g. open bog area, forested bog with various tree species: Picea abies, Pinus silvestris, Betula sp.) has well-distinguishable spider communities with a considerable number of rare, specialist species. Microhabitat diversity (e.g. hummock-hollow mosaic complex) further increases spider diversity (Samu and Urák 2014; Gallé et al. 2019).
The Vlașinescu peat bog is an open bog, with only sporadic dwarf growth spruce on the margins, but without a coherent forested area. Our results showed that many species were non-randomly distributed. The heather sites and the control sites were well separated on the NMDS scatterplot (Fig. 4), indicating a significant difference in species composition. This emphasized that both microhabitat types are essential for biodiversity conservation.
The presence of heather patches provides a structural heterogeneity in the open bog, they offer web attaching points for the web builder species (Uetz 1979). This might be the reason why the web-builder species and a more diverse spider community is hosted by the heather shrubs than open bogs. The physical structure and spatial geometry of the 20–40 cm high heather shrubs are suitable sites for the sheet webs of linyphiid spiders. Indeed, we found more linyphiids in the heather sites than in the control sites (Fig. 4., Appendix A.). Presumably the small body size of these species shifted the CWM size values of the community.
Spiders are the major predators in terrestrial ecosystems; they prey on herbivores, detritivores and fungivores (Wise 1993). Springtails (Collembola) are among the most abundant detritivorous and fungivorous arthropods in peat bogs (Sławski et al. 2022). They provide a large part of the diet of ground-dwelling spiders (Wise 2004). Microclimate (temperature and moisture conditions) and substrate quality affect the vertical stratification and community composition of springtails in Sphagnum peatlands (Krab et al. 2010). In addition, heather shrubs might influence ground temperature. During the day, the leaves absorb or reflect a significant fraction of solar radiation. Night-time temperatures are presumably warmer as a consequence of the thermal insulating layer of the shrubs, resulting in cooler and more uniform near-ground thermal conditions (Vanneste et al. 2020). The microscale temperature pattern of Sphagnum mosses influences springtails’ density and species composition and therefore, affects spiders.
In addition to the structural complexity and microclimate, the physical and chemical properties of the heather litter quality may affect potential prey items of spiders (Bolger et al. 2013; Kováč et al. 2005). For instance, many species and a diverse community of springtails are associated with the leaf litter of heather shrubs (Furgoł et al. 2019). Furthermore, heather patches support a high diversity of insect herbivores, such as Hemiptera-Auchenorrhyncha, potential prey items for spiders (Sanderson et al. 2020).
We did not detect more shrub- and canopy-dweller species in heather sites. These shrubs were not tall enough to affect the vertical microhabitat preference of spiders (Fig. 3c). In the boreal region, homogeneous open bogs are relatively species-poor habitats compared to the heterogeneous mesotrophic bogs (Kamayev 2012), emphasizing the importance of microhabitat heterogeneity in shaping species richness and composition of peat bog spider assemblages.
Conclusions.
The spider fauna of the Vlașinescu peat bog is species-rich, with many rare, specialist species, with high conservation value. We collected different characteristic spider species and we identified different trait state compositions in the heather patches and the surrounding control bog sites. The species richness and the presence of rare and endangered peat bog specialist spider species of heather patches highlight the high conservation relevance of microhabitat heterogeneity in peat bogs. Therefore the preservation of dwarf shrubs in Central European peat bogs is of crucial importance for biodiversity conservation.
Data availability
Raw data was included in supplementary material of the article. Voucher specimens of the collected species were placed in the arachnological colletion of Sapientia Hungarian University of Transylvania.
References
Bain C, Bonn A, Stoneman R, Chapman S, Coupar A, Evans M, Geary B, Howat M, Joosten H, Keenleyside C, Labadz J, Lindsay R, Littlewood N, Lunt P, Miller CJ, Moxey A, Orr H, Reed M, Smith P, Swales V, Thompson DBA, Thompson PS, Van de Noort R, Wilson JD, Worrall F (2011) Commission of inquiry on UK peatlands. IUCN UK Peatland Programme, Edinburgh
Barton PS, Manning AD, Gibb H, Lindenmayer DB, Cunningham SA (2009) Conserving ground-dwelling beetles in an endangered woodland community: multi-scale habitat effects on assemblage diversity. Biol Cons 142:1701–1709
Bolger T, Kenny J, Arroyo J (2013) The Collembola fauna of irish forests – a comparison between forest type and microhabitats within the forests. Soil Organisms 85:61–67
Buchar J, Ruzicka V (2002) Catalogue of spiders of the Czech Republic. Peres, Prague
Buchholz S (2016) Natural peat bog remnants promote distinct spider assemblages andhabitat specific traits. Ecol Indic 60:774–780
Cardoso P, Pekár S, Jocqué R, Coddington JA (2011) Global patterns of guild composition and functional diversity of spiders. PLoS ONE 6:e21710
Charman DJ (2002) Peatlands and environmental change. John Wiley & Sons Ltd., Chichester, p 301
Coulson JC, Butterfield J (1986) The spider communities on peat and upland grasslands in northern England. Holarctic Ecol 9:229–239
Curtis DJ, Corrigan H (1990) Peatland spider communities and land management on a scottish island. C.R. XIIe Colloque européen d’Arachnologie, pp 97–102
Danci O (2016) Conservation status of some peat bogs in Maramureș county. Transylv Rev Systematical Ecol Res 18:69–82
Dawson TP, Berry PM, Kampa E (2003) Climate change impact on freshwater wetland habitats. J Nat Conserv 11:25–30
Dufrene M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–367
Evans CD, Bonn A, Holden J, Reed M, Evans M, Worrall F, Parnell M (2014) Relationships between anthropogenic pressures and ecosystem functions in UK blanket bogs: linking process understanding to ecosystem service valuation. Ecosyst Serv 9:5–19
Furgoł M, Piwnik A, Wiśniewsk K (2019) Five springtail (Collembola) species inhabiting heathlands in Poland. Entomol Fenn 30:186–195
Gallé R, Galle-Szpisjak N, Zsigmond AR, Könczey B, Urák I (2021) Tree species and microhabitat affect forest bog spider fauna. Eur J For Res 140:691–702
Gallé R, Samu F, Zsigmond AR, Gallé-Szpisjak N, Urák I (2019) Even the smallest habitat patch matters: on the fauna of peat bogs. J Insect Conserv 23:699–705
Gallé R, Urák I (2001) Contribution to the spiders (Arachnida: Araneae) of upper Mureş river valley with some new data for the romanian fauna. Entomol Rom 6:141–145
Gallé R, Urák I (2002) Faunistical data ont he spiders (Arachnida: Araneae) of the Nemira Montain’s bog complex with two new species for the romanian fauna. Entomol Rom 7:85–88
Gallé R, Urák I, Gallé-Szpisjak N, Hartel T (2017) Sparse trees and shrubs confers a high biodiversity to pastures: Case study on spiders from Transylvania. PLoS ONE 12(9):1–12
Gallé R, Urák I (2006) Faunistical data on the spiders (Arachnida: Araneae) of the Lacul Dracului bog complex with new data for the romanian fauna. Sci Ann “Danube Delta” Inst 12:29–32
Gärdenfors U (2000) Rödlistade arter i sverige 2000 - the 2000 red list of swedish species. ArtDatabanken, SLU, Uppsala, p 397
Gopal B, Ghosh D (2008) Natural wetlands. In: Jørgensen SE, Fath BD (eds) Encyclopedia of Ecology. Elsevier Science B.V., Amsterdam, pp 2492–2504
Grzybowski M, Glinska-Lewczuk K (2020) The principal threats to the peatlands habitats, in the continental bioregion of Central Europe – a case study of peatland conservation in Poland. J Nat Conserv 53:125778
Haase H, Balkenhol B (2015) Spiders (Araneae) as subtle indicators for successional stages in peat bogs. Wetl Ecol Manag 23:453–466
Harper AR, Doerr S, Santin C, Froyd CA, Sinnadurai P (2018) Prescribed fire and its impacts on ecosystem services in UK. Sci Total Environ 624:691–703
Heinemeyer A, Vallack HW, Morton PA, Pateman R, Dytham C, Ineson P, McClean C, Bristow C, Pearce-Higgins JW, Thom T (2019) Restoration of heather-dominated blanket bog vegetation on grouse moors for biodiversity, carbon storage, greenhouse gas emissions and water regulation: comparing burning to alternative mowing and uncut management. Final report to Defra on Project BD5104. Stockholm Environment Institute at the University of York, York, UK
Kamayev IO (2012) Spider (Aranei) assemblages in bog-forest ecotone of the northern taiga subzone (Eastern Fennoscandia). Entomol Rev 92:471–485
Krab EJ, Oorsprong H, Berg MP, Cornelissen JH (2010) Turning northern peatlands upside down: disentangling microclimate and substrate quality effects on vertical distribution of Collembola. Funct Ecol 24:1362–1369
Koponen S (1979) Differences of spider fauna in natural and man – made habitats in a raised bog. In: The use of ecological variables in enviromental monitoring. The National Swedish Environment Protection Board, Report PM 1151, Uppsala, 104–108
Koponen S (2002) Ground-living spiders in bogs in Northern Europe. J Arachnol 30:262–267
Kováč LU, Kostúrová N, Miklisová D (2005) Comparison of collembolan assemblages (Hexapoda, Collembola) of thermophilous oak woods and Pinus nigra plantations in the Slovak Karst (Slovakia). Pedobiologia 49:29–40
Lafage D, Djoudi EA, Perrin G, Gallet S, Pétillon J (2019) Responses of ground-dwelling spider assemblages to changes in vegetation from wet oligotrophic habitats of Western France. Arthropod-Plant Inte 13:653–662
Laliberte E, Legendre P (2010) A distance-based framework for measuring functional diversity from multiple traits. Ecology 91:299–305
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280
Lewinsohn TM, Novotny V, Basset Y (2005) Insects on plants: diversity of herbivore assemblages revisited. Annu Rev Ecol Evol Syst 36:597–620
McGeoch MA (1998) The selection, testing and application of terrestrial insects as bioindicators. Biol Rev 73:181–201
Manning AD, Fischer J, Lindenmayer DB (2006) Scattered trees are keystone structures–implications for conservation. Biol Cons 132:311–321
Margóczi K, Drăgulescu C, Macalik K (2000) Vegetation description of representative habitat complexes along the Maros (Mureş) River I. The upper section (Vasláb/Voşlobeni). In: Gallé L, Körmöczi L (eds) Ecology of River Valleys. TISCIA monograph series. Department of Ecology, University of Szeged
Nentwig W, Blick T, Gloor D, Hanggi A, Kropf C (2019) Spiders of Europe. Retrieved from www.araneae.unibe.ch
Parish F, Sirin A, Charman D, Joosten H, Minayeva T, Silvius M, Stringer L (2008) Assessment on Peatlands, Biodiversity and Climate Change: Main Report. Global Environment Centre, Kuala Lumpur, Malaysia / Wetlands International, Wageningen, The Netherlands, 179 pp
Pop E (1960) Mlaștinile de turbă din România, edit. Academiei Republicii Populare România, București
Relys V, Dapkus D (2000) Similarities between epigeic spider communities in a peatbog and surrounding pine forest: a study from southern Lithuania. Eur Arachnology 19:207–214
Ricotta C, Moretti M (2011) CWM and Rao’s quadratic diversity: a unified framework for functional ecology. Oecologia 167:181–188
Riecken U, Finck P, Raths U, Schröder E, Ssymank A (2006) Rote Liste dergefährdeten Biotoptypen Deutschlands, Zweite fortgeschriebene Fassung. Natursch Biol Vielfalt 34:1–318
Roberts DW (2012) Package “labdsv.” http://cran.r-project.org/web/packages/labdsv/labdsv.pdf. Accessed 23 Jan 2021
Scott AG, Oxford GS, Selden PA (2006) Epigeic spiders as ecological indicators of conservation value for peat bogs. Biol Conserv 127:420–428
Samu F, Urák I (2014) Are more bogs better? Comparative studies into Transylvanian peat bog spider (Arachnida: Araneae) assemblages from a conservation biological perspective. North-West J Zool 10:94–101
Sanderson R, Newton S, Selvidge J (2020) Effects of vegetation cutting on invertebrate communities of high conservation value Calluna upland peatlands. Insect Conserv Diver 13:239–249
Schirmel J, Blindow I, Buchholz S (2012) Life-history trait and functional diversity patterns of ground beetles and spiders along a coastal heathland successional gradient. Basic Appl Ecol 13:606–614
Sławski M, Stebel A, Sławska M (2022) Spontaneous regeneration of Collembola assemblages in a raised bog after human-induced disturbance. Appl Soil Ecol 169:104233
Succow M (1990) Moore in der Landschaft. Entstehung Haushalt Lebewelt Verbreitung Nutzung und Erhaltung der Moore, 2nd edn. Urania-Verlag, Leipzig, Jena, Berlin
Tucker G (2003) Review of the impacts of Heather and Grassland Burning in the Uplands on Soils, Hydrology and Biodiversity. English Nature Research Reports, p 550
Uetz GW (1979) The influence of variation in litter habitats on spider communities. Oecologia 40:29–42
Urák I, Máthé I (2013) A Lucs-tőzegláp pókjainak (Arachnida: Araneae) faunisztikai és ökológiai vizsgálata. Acta Siculica: 59–74
Urák I, Samu F (2008) Contribution to the spider fauna of the Mohoş peat bog from Transylvania, with some new data for Romania. North-West J Zool 4:50–60
Vanneste T, Govaert S, Spicher F, Brunet J, Cousins SA, Decocq G, …, De Frenne P (2020) Contrasting microclimates among hedgerows and woodlands across temperate Europe. Agr For Meteorol 281:107818
Wallèn B (1987) Growth pattern and distribution of Biomass of Calluna vulgaris on an Ombrotrophic Peat Bog. Holarctic Ecol 10:73–79
Weiss I, Urák I (2000) Faunenliste der Spinnen Rumäniens. Checklist of the Romanian spiders (Arachnida: Araneae). www.arachnologie.info/fauna.htm, accessed at: 2022.01.02
Wise DH (1993) Spiders in ecological webs. Cambridge University Press, Cambridge
Wise DH (2004) Wandering spiders limit densities of a major microbi-detritivore in the forest-floor food web. Pedobiologia 48:181–188
Funding
This study was financially supported by the Domus Hungarica Scientiarum et Artium research grant and the Institute of Research Programmes of Sapientia Hungarian University of Transylvania.
Open access funding provided by ELKH Centre for Ecological Research.
Open access funding provided by ELKH Centre for Ecological Research.
Author information
Authors and Affiliations
Contributions
IU, SLI and AZ designed the study. IU, AZ collected spiders on the field. IU, NGS and RG identified the spiders. RG analysed the data. UI and GR wrote the manuscript with a significant input from all authors.
Corresponding author
Ethics declarations
Ethical approval
To obtain the data presented in the manuscript it has been necessary to involve animals, however this study did not involve protected species and no human participants were involved in the work. All authors consent to submission of this manuscript.
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Urák, I., Gallé-Szpisjak, N., Szigyártó, IL. et al. Heather (Calluna vulgaris) supports spider diversity of oligotrophic peat bogs. J Insect Conserv 27, 415–422 (2023). https://doi.org/10.1007/s10841-023-00465-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10841-023-00465-z