Abstract
Boreal forests undergo rapid transformation from more or less intact states to even-aged production stands, due to the expanding clearcutting practice. Deepened knowledge on how species diversity and composition vary with local resource availability, such as dead wood, as well with remaining never clearcut forest (having long continuity = ‘continuity forest’) in the surrounding landscape, is essential for the formulation of conservation strategies. In each of six 15 × 15 km study areas in mid-boreal Sweden (12 000 km2) we sampled saproxylic beetles through sieving of bark from Picea abies logs in early stages of decay with bark still attached, within 5 production forests and 5 woodland key habitats (biodiversity hotspots, used as reference; ‘WKH’). A total number of 5147 individuals and 78 taxa were found. Beetle diversity related positively to the local factor of sieved bark area, while the share of continuity forest in the surroundings (5 km buffer; varying 5 − 29% among study stands) was less important, and even showed negative relationships. Species richness was higher in > 60 years old than 0–60 years old production forests but we did not detect a change with age in species composition. The production forests > 60 years old and WKH were similar regarding species richness and composition. WKH had more species of conservation concern than the production forests.
Implications for conservation
An increase in local dead wood amount would benefit saproxylic beetles species richness in north European boreal production forests. Production forests > 60 years old may be of interest to species of conservation concern, although WKH seem considerably more important.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Many boreal forest landscapes have undergone strong anthropogenic transformation through clearcutting forestry resulting in a dramatic reduction in the area of intact forests, with subsequent risk of undermining biodiversity, ecological processes and the delivery of ecosystem services (Watson et al. 2018). Northern Europe is the boreal region with the longest and most intense forestry practices, in many parts with large-scale application of clearcutting since the mid-1900s, resulting in only small areas remaining with limited human impact (Potapov et al. 2017; Svensson et al. 2019). Along with the transformation of forests, the amount of dead wood has decreased drastically, as have many dead wood (saproxylic) species (Stokland et al. 2012; SLU Artdatabanken 2020). In contrast, after natural disturbances, the volume of dead trees can exceed that of living trees, thus forming a large habitat resource for saproxylic species (Seibold and Thorn 2018).
Dead wood is an important habitat for numerous invertebrates, fungi and other taxonomic groups (Seibold and Thorn 2018) but despite much research during the last decades large uncertainty remains on the relative importance of different scales, a knowledge essential for the formulation of conservation strategies. Studies so far show varying results, restricting abilities for generalizations (Sverdrup-Thygeson et al. 2014; Kärvemo et al. 2021) with some studies pointing to large importance of the local scale (e.g. Lassauce et al. 2011) while others have identified positive landscape effects (e.g. Gibb et al. 2006a; Olsson et al. 2012 Häläinen et al. 2023) or lack of or varying effects (e.g. Ranius et al. 2015; Jacobsen et al. 2020). Saproxylic beetles are frequently used to assess status and dynamics of biodiversity in forests under human transformation, due to their high diversity, comparatively well-known taxonomy and possibility for detection (Grove 2002; Stokland et al. 2012). For instance, the number of obligate saproxylic beetles from the Nordic countries is estimated to about 1100 (Stokland et al. 2012).
A large interest in applied conservation research has resulted in a strong emphasis on the oldest and least anthropogenically affected forests (e.g. Kouki et al. 2012; Svensson et al. 2020), although clearcuts have attracted increased attention not the least regarding saproxylic beetles (e.g. Rubene et al. 2017; Johansson et al. 2020), and specific production forest stages like thinned stands have also been studied regarding these organisms (Stenbacka et al. 2010; Gran and Götmark 2021). Occurrence and dynamics of saproxylic beetles are driven by factors acting at different scales, from individual dead wood objects to landscapes and above (Sverdrup-Thygeson et al. 2014). At the local scale, amount of dead wood is a strong driver (Seibold and Thorn 2018), and species communities also vary depending on microclimatic factors, e.g. how much light that reaches the logs or snags (Lindhe et al. 2005; Seibold et al. 2016). Many studies also point to a significance of landscape properties (e.g. Rubene et al. 2017; Ekström et al. 2021) although detailed studies with replicated landscapes are scarce (but see Olsson et al. 2012). Despite this knowledge-base, studies on species diversity and composition of various taxonomic groups of different ages of boreal, even-aged production forests are few (but see López-Bedoya et al. 2021). One evident knowledge gap is the possible importance of so far never clearcut forests, i.e. forests with long continuity (here called ‘continuity forest’) still remaining in the landscapes, to dead wood organisms.
Our main aim is to increase insights into drivers of saproxylic beetle diversity and community composition in landscapes dominated by even-aged, coniferous production forests but with still some continuity forest remaining. We investigate boreal landscapes in Sweden, since long transformed by industrial forestry but where some forests without clearcutting impact still occur (Svensson et al. 2019). Although some of these long-continuity forests have been set-aside as conservation areas most are production forests, managed through selective harvest, first through high-grading (removal of the largest trees) and later through repeated thinning. We specifically want to add knowledge on the relative role of local bark amount versus continuity forest in the landscape. To clearly link to nature conservation, we also include old spruce forests identified as biodiversity hotspots, ‘woodland key habitats’ (abbreviated WKH), mapped in a nation-wide survey (Timonen et al. 2010; Wester and Engström 2016), and we also target species of conservation concern. We survey beetles in logs of Norway spruce in an early stage of decomposition when the bark is still attached to the trunk, a substrate to which a large number of saproxylic beetles are associated (e.g. Djupström et al. 2008). Our assumptions are that:
-
i.
local amount of dead wood is a strong driver of species richness and composition.
-
ii.
amount of never clearcut, long-continuity forest (continuity forest) in the surrounding landscape is also a strong driver.
-
iii.
species richness is higher in old compared to young production forest and species composition differs between forest ages.
Methods
Sweden is an ideal study system for even-aged forests since clearcutting forestry has been predominant in all parts of the country since the middle of the last century, and consequently, even-aged stands < 60 years old form large parts of the landscapes. Most forests > 60 years old have never been clearcut although this harvest practice also occurred in some regions prior to the 1950–60s (Lundmark et al. 2013). Production forests comprise 86% of the productive forestland of 23 million ha, with the rest set aside for conservation, by the state (6%) or through retention actions mainly within certification commitments (8%) (SCB 2022). The minimum rotation time ranges between 45 and 100 years, increasing towards the north (Roberge and Fries 2020). Clearcuts are usually regenerated with either Norway spruce Picea abies (L.) H.Karst. or Scots pine Pinus sylvestris L., each comprising about 40% of the total wood volume in the country (Skogsdata 2020). The dead wood amount on average per hectare for boreal production forests increases with forest age from 3 m3ha− 1 for the age class 21–40 years to 16 m3ha− 1 for forests > 120 years according to the Swedish National Forest Inventory (Skogsdata 2020). For the youngest age-class, 0–20 years, retention of dead wood has contributed to current levels of approximately 8 m3ha− 1 (Skogsdata 2020).
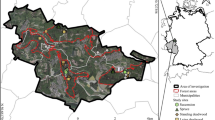
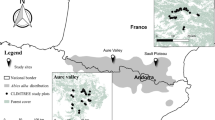
The study was conducted within the counties of Jämtland and Västernorrland, mid-boreal Sweden, covering approximately 12 000 km2 (Fig. 1a, b). In order to capture as large as possible range in landscape proportion of continuity forest, we initially identified 6 15 × 15 km study areas varying in this respect (Fig. 1b). Within each study area we inventoried saproxylic beetles in spruce logs in production forests of different ages, and as a reference in WKH (Fig. 1c, d). A ‘landscape’ was defined as a circular 5 km buffer zone around each production forest stand and key habitat.
Sampling design with the position of the six 15 × 15 km study areas (a) in mid-boreal Sweden within (b) the counties of Jämtland and Västernorrland, (c) one study area including five production forests (grey) and five WKH (black), and (d) five random circular plots in each production forest or WKH, with a radius of 30 m (area 2800 m2), with one sampled log in each for which 0.5 m2 bark (if possible) was sampled
Selection of study areas
To identify study areas, we used a high-resolution mapping of remaining, not previously clearcut forests in boreal Sweden covering production forests as well as areas set aside for conservation (Ahlkrona et al. 2017a). The mapping is based on a semi-automatic change detection analysis of satellite images taken between 1973 and 2015, later updated with a visual detection of aerial photos from the 1950s and 1960s (Ahlkrona et al. 2017b, 2019). However, since clearcutting is an ongoing forest harvesting practice resulting in rapid landscape changes, we used landcover data including clearcuts from 2018 (Swedish Environmental Protection Agency 2019) to correct for changes since 2017. To further compensate for clearcuttings made between 2018 and 2019, we used data from the clearcutting database of the Swedish Forestry Agency. These never clearcut, long-continuity forests (continuity forests) correspond to the pCF category of Svensson et al. (2019). A continuity forest raster and a mask grid covering the entire forestland of the two counties was used, with a 10 m resolution. Based on searches of 15 × 15 km surroundings from each 10 × 10 m cell, we identified the 6 study areas with the largest range in landscape proportion of continuity forest, resulting in a variation between 4% and 25%. (Fig. 1b). All analyses were conducted in ArcMap 10.7.
Selection of production forests and WKH
Within each of the six 15 × 15 km study areas, we selected five production stands and five WKH based on the criteria that they should be spruce-dominated and be on mesic soil (to decrease the environmental variation and to focus on the most common soil type). (Fig. 1c). Production stands were identified from the stand-database of the forestry company SCA and from a free data source showing the approximate forest age (SLU 2010), followed by field inspection (July – September 2019). In each of the 15 × 15 km study areas, one stand in each of the age-classes 0–20, 21–40, 41–60, 61–80, 81 – >120 years was selected. This fine-scale division was made to enable detailed age-class analyses. WKH have in a large national survey been identified as especially valuable to biodiversity and are in the region dominated by old coniferous forest with high structurally diversity (Wester and Engström 2016). From the WKH database of the Swedish Forestry Agency, we selected coniferous-dominated stands on mesic soil. All production forest stands in the oldest age-category (81 - >120 years) and all WKH were in the remote-sensing analysis identified as continuity forest.
Sampling of logs
We used an area-based sampling design with selection of a maximum of 5 logs in each forest stand, within an area of 1.4 ha. This was a deliberate strategy to reflect that availability of logs (and thus habitat available to saproxylic species) vary between forest ages (Skogsdata 2020), and with the expectation that < 5 logs would be available in some of the youngest stands. One log of Norway spruce Picea abies was selected within each of five circular plots per stand with a radius of 30 m (2800 m2). For stands < 1.4 ha in size, the whole stand was searched (Fig. 1d). The first plot was positioned at the stand center, and the following four plots in directions N, E, S, W, at a distance of 120 m from the center point for stands > 5 ha and at 60 m for stands < 5 ha in siz (Fig. 1d). In a few cases, the stand was so difficult to traverse that the first plot was a random point at the part of entry, with enough space for four additional plots. The first log found when searching clockwise from the center point in the plot was selected. Criteria for logs were (i) Norway spruce P. abies, (ii) decomposition category II and III according to Siitonen and Saristo (2000) implying early stages of decay before most of the bark has fallen off but excluding logs dead less than one year (with fresh phloem), (iii) diameter > 10 cm, (iv) mesic soil (the part > 10 cm in diameter), and v) > 10 m from stand edge towards a habitat clearly deviating from the stand structure (e.g. clearcut, lake). Sampling was performed in September 2019. Totally, 6 study areas, 30 WKH, 30 production forests and 242 logs (< 5 logs were found in 22 of the stands) were included in the study.
Beetle sampling and identification
The sampling procedure was modified after Djupström et al. (2008). A piece of bark maximum 0.5 m2) was collected from each sample log, its area was recorded, and was sieved for one minute through a metal wire net mesh. This method captures beetles that with certainty have colonized the bark, in contrast to flight traps which catch beetles during their dispersal phase (Djupström et al. 2008). Beetles were extracted by placing the finer fractions from the sieving under a lamp in Tullgren funnels (Southwood and Henderson 2000) at the same day as the collection of bark. The nomenclature followed the Swedish taxonomic database Dyntaxa (www.dyntaxa.se). Classification of red-listed species follows SLU ArtDatabanken (2020).
Statistical analysis
All statistical analyses were performed using the R software, version 4.4.0 (R Core Team 2024). To estimate how large proportion of the total species pool we captured, we constructed a species accumulation curve and extrapolated the curve to its asymptote (using the R package iNEXT, Hsieh et al. 2024).
To ensure statistical power, the age-classes were merged into 0–60 years old (‘young production forest’; 18 stands), and > 60 years old (‘old production forest’; 12 stands). For these two production forest age-classes and the WKH (in the following ‘stand categories’), we first calculated (for each stand) the total number of sampled logs, total bark amounts, and the landscape proportion of continuity forest within a 5 km buffer (all three later used as explanatory variables), as well species richness and the Shannon diversity index (both later used as response variables). We then tested the correlation between all these variables and their relation with stand category using Spearman rank correlations and Kruskal-Wallis tests with associated post-hoc test (for comparisons with stand category). After initial testing, we removed the number of logs from further analysis since it was strongly correlated with the total amount of sieved bark (ρ = 0.96, p < 0.001, Supplementary Materials Table A1).
To analyze species richness and the Shannon diversity index in relation to the remaining explanatory variables mentioned above, we built generalized linear mixed effect models (GLMMs) using the glmmTMB package (Brooks et al. 2017), with study area identity (6 study areas) as random effect. For species richness, we used poisson distributions and for the Shannon diversity index we used a normal distribution. For bark amounts we also tested the squared term as we expect an asymptotic relationship with increasing bark amounts. Models were built based on AIC, and the final (best) model was the one with the lowest AIC. To test if the species richness of all stand categories was explained by the landscape proportion of continuity forest within 5 km, we also fitted separate models for each stand category.
As a complement to the analysis of all species jointly we also tested species richness of a sub-group of species of conservation concern comprising red-listed species (SLU ArtDatabanken 2020) and indicator species of forests with high conservation value (Nitare 2019): Agathidium nigrinum, Atomaria alpina, Atomaria subangulata, Cis dentatus, Corticaria lapponica, Ernobius explanatus, Eudectus giraudi, Hylurgops glabratus, Olisthaerus substriatus, Orchesia fasciata, Peltis ferruginea and Zilora ferruginea.
To describe the general beetle species composition, we performed a nonmetric multidimensional scaling (NMDS) analysis on the total number of individuals in each stand. The NMDS was performed with the R package vegan (Oksanen et al. 2022) using the Bray–Curtis dissimilarity index. We had to use three dimensions to reduce the stress value to acceptable levels (< 0.2, McCune and Grace 2002). We only included species that were present in at least eight stands (14 in total) to improve model convergence. Significant relationships between species composition and explanatory variables (stand category, amount of continuity forest in the surrounding landscape, and bark amount) were assessed with Permutational Multivariate Analysis of Variance (function adonis2 in vegan; Oksanen et al. 2022).
To compare beetle dissimilarity among stands (β-diversity) between stand categories we used the Bray-Curtis similarity indices obtained with the betapart package in R (Beselga and Orme 2020). The distributions within each category were then compared using ANOVA and pairwise differences were assessed with Tukeys HSD (post hoc) test.
To assess how dominant species were associated with stand categories, the amount of continuity forest in the surrounding landscape, and the amount of bark sieved we used non-parametric tests (Kruskal-Wallis for stand category and Spearman rank correlations for continuity forest and amount of bark). We used the total number of individuals and tested all species occurring in at least eight stands (14 species in total).
Results
General characterization
In total we recorded 5147 saproxylic beetles distributed among 78 species. The total number of logs sampled was 50 for young production stands, 54 for old production stands, and 138 for WKH, and the corresponding numbers for total bark amounts were: 22.1 m2, 26.7 m2, and 68.3 m2. The number of sampled logs and amount of sieved bark increased with stand age (Supplementary Materials Fig. A1). The rarefaction analysis indicated that about 62% of the total species pool was captured in our sample (Supplementary Materials Fig. A2). The most common species was the bark beetle Crypturgus hispidulus with 3038 individuals (59.0% of the total number), followed by Leptusa pulchella (440, 8.5%), Polygraphus punctifrons (373, 7.2%), Dryocoetes autographus (310, 6.0%), and Rhizophagus dispar (220, 4.3%) (Supplementary Materials Table A2). These five species, hence, comprised almost 85% of all species records, while most species had very few individuals with a median of 3.
Species diversity
The number of species per stand ranged between 0 and 17 (mean = 8.6, SE = 0.52), and the number of individuals ranged between 0 and 381 (mean = 91.9, SE = 12.2). Only 7 individuals of species of conservation concern were found in production forests (one in young and 6 in old), while 47 were found in WKH. The average number of species of conservation concern per stand was 1.57 (11 species in total) for WKH, 0.5 (3 species in total) for old production forest and 0.06 (1 species in total) for young production forest. The number of logs found in a stand and the total amount of sieved bark were clearly lower in young production forests compared to old production forests and WKH (Supplementary Materials Table A1). The proportion of continuity forest within 5 km did not differ between stand categories (Supplementary Materials Table A1) and was not correlated to any of the other explanatory variables (p > 0.5). Species richness and the Shannon diversity index correlated significantly to both the number of logs, bark amounts, and were clearly lower in young production forests while there were no differences between the old production forests and the WKH (Supplementary Materials Table A1, Fig. 2). For species of conservation concern the number of species was significantly higher in the reference forests (WKH) compared to both young (p < 0.001) and old (p = 0.025) production forests, while the latter two did not differ significantly.
The distribution of a) the species richness (total number of beetle species per stand, α-diversity) and b) the Shannon diversity index for each stand category. Letters (a or b) above each boxplot indicate significant differences among stand categories based on Dunn’s post hoc test (the same letter signifies non-significant differences). Horizontal black line shows the median for each stand category
According to the GLMMs total species richness increased with increasing bark amounts (where the negative squared term gives an asymptotic relationship, Fig. 3), while it decreased with increasing proportion of continuity forest within 5 km (Table 1; Fig. 3), although this relationship was weaker. When tested separately, only the young production forests showed a significant negative relationship with proportion of continuity forest within 5 km (p < 0.001), even if there was a tendency also for WKH (p = 0.07). The Shannon diversity index increased with bark amount (Table 1). When bark amount was a significant predictor in the model, stand category was unimportant to total species richness as well as Shannon diversity index. In contrast, the richness of species of conservation concern was mainly explained by stand category (Table 1), where WKH clearly had more species of conservation concern than production forests (regardless of age). Species of conservation concern, however, showed a tendency to decrease with the proportion of continuity forest within 5 km, but the change in AIC, when this variable was included in the model, indicated a weak relationship (Table 1).
Species richness (total number of beetle species per stand) in relation to a) the amount of sieved bark per sample stand, and b) the proportion continuity forest within 5 km from the focal stand (‘‘andscape’) in young production forests (0–60 years, n = 18), old production forests (> 61 years, n = 12), and for WKH (n = 30). Broken lines signify model predictions for each of the two explanatory variables when the other variable was kept constant at its mean value
Species composition, stand similarities and species abundances
The species composition was explained by the amount of bark (R2 = 0.11, F = 6.27, p = 0.001; Fig. 4) while stand category and the proportion of continuity forest within 5 km were insignificant (R2 = 0.03, F = 0.90, p = 0.52, and R2 = 0.01, F = 0.42, p = 0.89, respectively). Also, when stand category was analyzed separately it remained insignificant (R2 = 0.04, F = 1.04, p = 0.38).
The stand dissimilarity in species composition was significantly larger among young production stands compared to the old production forests (p = 0.029), while there was no difference compared to WKH (Fig. 5). There was no significant difference between the latter two.
The abundances of eight species were positively correlated with the amount of bark (Table 2) and one species Cis punctulatus showed a negative correlation with the proportion continuity forest within 5 km. Four species were affected by stand category, with three most common in old production forests (Cis punctulatus, Crypturgus hispidulus, and Leptusa pulchella) and one in WKH (Dropephylla liniaris).
The species composition of saproxylic beetles (occurring in at least eight stands) in young production forests (black dots), old production forests (red dots), and in WKH (green dots). The ordination was 3-dimentional to reach convergence (stress = 0.18). However, for simplicity only the 2D-plots (of axes 1 and 2) are shown here. The polygons show the outer limits of each stand category and the triangles the corresponding centroids
The dissimilarity between stands (β-diversity) within each stand category. Letters (a and b) above each boxplot indicate significant differences among stand categories based on Tukey’s post hoc test. Distance to centroid (the distance between each stand and the group centers based on principal coordinates) gives a measure of variation in species composition for each group, where larger values signify larger variation
Discussion
Local amount of bark was the strongest driver of the saproxylic beetle diversity, and contrary to our expectations, landscape amount of continuity forest was considerably less important and even showed a negative, although weak, relationship with species richness. In line with one of our expected outcomes, species richness was higher in old than young production forests but on the other hand, species composition did not vary with age or between production forests and WKH. WKH hosted more species of conservation concern than old production forest.
Local and landscape factors
Earlier studies including the landscape scale point to variable outcomes regarding the relative role of local and landscape factors (Sverdrup-Thygeson et al. 2014). We suggest three possible explanations for this variation in results among studies. First, mapping of old forest is based on different data sources such as snapshot satellite images, forest-company and authority maps or a combination of remote sensing and forest inventory data. One advantage with our study is that we used a new dataset with high-resolution, temporal analysis of aerial photographs covering > 50 years, identifying forests that likely have never been clearcut, with considerable old-growth legacies, i.e. such that we named ‘continuity forests’ (Svensson et al. 2019). Second, different trapping methods such as flight traps, eclector traps, and as in our study sieving of bark, vary greatly in their effectiveness to capture saproxylic species (e.g. Martikainen and Kouki 2003). There are pros and cons with respective method with bark sieving and eclector traps capturing saproxylic species from a specific piece of wood, while the local species pool of saproxylics as well as non-saproxylics are captured with window traps. Evaluations point to bark sieving having higher shares of saproxylic species than eclector traps while eclector traps have the advantage of capturing species also inside the wood (Alinvi et al. 2007). Third, species richness and composition vary with decay stage, as shown by Jonsell et al. (2019) that in a time-series only found an effect of surrounding forest on saproxylic beetles in dead wood in late stages of decomposition. Since we used only slightly decayed spruce logs, we may have missed important variation. Further, our landscape proportion of continuity forests within 5 km ranged between 5% and 29% which might be too narrow to capture effects. Unfortunately, in northern Europe, the boreal region with the longest history of clearcutting globally, continuity forests are today very uncommon, and thus higher proportions are difficult to find. Taken together, the current evidence-base seems too weak to enable general conclusions regarding landscape effects.
Forest age
The finding that young production forests (< 60 years old) had lower total species richness than old forest (> 60 years old) agrees with Stenbacka et al. (2010); one of few other studies including the full range of boreal production forest ages. As they did, we interpret this to be linked to habitat availability, in our case to the lower amount of logs and bark in the young production forests. The sampling that we applied was designed to capture logs and bark in relation to their density, and we expected (and found) lower density in younger stands. Had we used a design with sampling of the same number of logs over areas varying in size (larger areas searched in low-density stands), the number of saproxylic beetles in the youngest age stages would have been overestimated. The low amount of dead wood and the increase with forest age is typical for clearcutting forestry in north Europe while it may be different in other regions, such as Central Europe where young coniferous stands can host a high richness of saproxylic beetles (Zumr et al. 2022). Also in some cases, such as following extensive tree die-back caused by bark beetle outbreaks, the situation may be different with very high amounts of dead wood and a rich saproxylic fauna (e.g. Vrba et al. 2024), although absence of salvage logging then is a condition (Thorn et al. 2018). The lack of effect of forest age on species composition in our study was surprising since such could be expected from the change in environmental conditions during forest succession. For instance, light is a strong determinant of saproxylic species composition, with more species in open conditions (Lindhe et al. 2005; Seibold et al. 2016; Lettenmaier et al. 2022) and thus, a specific species composition could be expected in the young, comparatively open forests with a higher light availability compared to closed forest (Chen et al. 1993). Several studies that compare clearcuts to old forests (excluding intermediate age-classes) also point to clearcuts having a different saproxylic beetle composition than forests mature to be harvested (e.g. Gibb et al. 2006b; Johansson et al. 2007; Jacobsen et al. 2020). The compositional similarity between young production forests, old production forests and WKH point to common, generalist species strongly driving species composition. The fact that five species comprised almost 90% of all captured individuals, and with a strong dominance of the bark beetle the Crypturgus hispidulus, indicate that a few species have a decisive influence on the diversity patterns.
The higher variation in species composition in the < 60 year old than > 60 year old stands (beta diversity) still point to the younger age-class having larger environmental variation. This is not surprising since this younger age-class comprises recently harvested, open, treeless areas as well as regenerating forests. Our data did not allow analyses with a finer age-class resolution but such a division will be essential for further studies. For the future, ideally, the youngest, open age-classes (about 0–30 years old) should be separated due to its deviating microclimatic conditions.
Production forests, WKH and species of conservation concern
Our reference stands, WKH, did not differ from the > 60 year old production forests regarding species diversity and composition of saproxylic beetles. This is surprising since WKH were mapped using structural and species indicators in a nation-wide inventory to identify hotspots for biodiversity (Wester and Engström 2016). Nevertheless, in boreal Sweden, Hjältén et al. (2012) also failed to detect compositional differences for saproxylic beetles between old, managed forests and protected areas, and explained this with many old stands never been clearcut in the past and thus having long tree continuity. In contrast to saproxylic species as a group, species of conservation concern were in our data-set significantly more common in WKH than in production forests. This result should be interpreted with some caution, due to the low total number of individuals; 54 for production forests and key habitats together, with almost 90% found in WKH. Still the rarefaction curve revealed that we had sampled a comparatively large proportion of the total species pool (Supplementary Materials Fig. A2). Both WKH and production forests > 80 years old (comprising half of the stands in our age-category > 60 years old) were in the remote sensing analysis found to be continuity (never clearcut), possibly indicating that they might be overall similar also regarding uncommon species, as has been found by Hjältén et al. (2012). Although the continuity forests have been selectively harvested at several occasions and also thinned, they have had a continuous tree-cover, and thus have a widely different background compared with stands emerging from clearcutting, although they may look surprisingly structurally similar. In the future, it will be important to, through a larger sample size, divide the age-class > 60 years old further, and compare production forests > 80 years old and WKH, especially regarding their content of species of conservation concern. This is especially urgent since there has been an intense debate on how much production forests, especially in the western part of boreal Sweden, qualify as WKH (Bjärstig et al. 2019; Hallberg-Sramek et al. 2020). Further, the so far never clearcut forests will soon disappear, if they are not protected (Ahlström et al. 2022). To fully evaluate the diversity patterns of saproxylic beetles, extended studies including a wider range of decay stages are also needed (Martikainen and Kouki 2003; see also Burner et al. 2022).
Conclusions
In agreement with many other studies, we show that dead wood availability at local level is a strong driver of saproxylic beetle richness and composition in forests (e.g. Lassauce et al. 2011; Seibold and Thorn 2018). The conservation implication is that an increase in dead wood in production forests would benefit the saproxylic beetle fauna. Unexpectedly, we found a negative, although weak, relationship between amount of continuity forests and species richness of saproxylic beetles, indicating so far little known and unexplored mechanisms for patterns and dynamics of saproxylic species. More studies at the landscape scale are needed to understand interactions between production forests and the small proportion continuity forests remaining in the since long heavily transformed boreal forests of north Europe. Such knowledge is also relevant for other parts of the boreal region with expanding industrial forestry where strategies for maintenance of remnant old forests will be an important issue. For conservation, deeper analyses of the oldest, never clearcut production forests is also essential, including comparisons with the mapped biodiversity hotspots (WKH).
Data availability
No datasets were generated or analysed during the current study.
References
Ahlkrona E, Giljam C, Wennberg S (2017a) Kartering av kontinuitetsskog i boreal region. Metria AB på uppdrag av Naturvårdsverket (In Swedish) https://www.skogsstyrelsen.se/globalassets/aga-skog/skydda-skog/bilaga-3-kartering_av_kontinuitetsskog_boreal_region.pdf
Ahlkrona E, Giljam C, Kesketalo C, Klein J, Naumov V (2017b) Preciserad kartering av kontinuitetsskog i Västernorrlands län. Metria AB på Uppdrag Av Naturvårdsverket (In Swedish) https://geodata.naturvardsverket.se/nedladdning/Kartering_av_kontinuitetsskog/Preciserad_kskog_2016_Y/Precisera%20karteringen%20av%20kontinuitetsskog%20Vasternorrlands%20lan.pdf
Ahlkrona E, Giljam C, Klein J, Eriksson T, Lindevall H (2019) Preciserad kartering av kontinuitetsskog i Jämtlands län. Metria AB på uppdrag av Naturvårdsverket (In Swedish) https://geodata.naturvardsverket.se/nedladdning/Kartering_av_kontinuitetsskog/Preciserad_kskog_2017_Z/Preciserad%20karteringen%20av%20Kskog%20Jamtland.pdf
Ahlström A, Canadell JG, Metcalfe DB (2022) Widespread unquantified conversion of old boreal forests to plantations. Earths Future 10(11). https://doi.org/10.1029/2022EF003221. e2022EF003221
Alinvi O, Ball JP, Danell K, Hjältén J, Pettersson RB (2007) Sampling saproxylic beetle assemblages in dead wood logs: comparing window and eclector traps to traditional bark sieving and a refinement. J Insect Conserv 11:99–112. https://doi.org/10.1007/s10841-006-9012-2
Beselga A, Orme CDL (2020) Betapart: an R package for the study of beta diversity. Methods Ecol Evol 3:808–812. https://doi.org/10.1111/j.2041-210X.2012.00224.x
Bjärstig T, Sandström C, Sjögren J, Soneson J, Nordin A (2019) A struggling collaborative process–revisiting the woodland key habitat concept in Swedish forests. Scand J for Res 34:699–708. https://doi.org/10.1080/02827581.2019.1674916
Brooks ME, Kristensen K, van Benthem KJ, Magnusson A, Berg CW, Nielsen A et al (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized Linear mixed modeling. R J 9:378–400. https://doi.org/10.32614/RJ-2017-066
Burner RC, Birkemoe T, Åström J, Sverdrup-Thygeson A (2022) Flattening the curve: approaching complete sampling for diverse beetle communities. Insect Conserv Divers 15:157–167. https://doi.org/10.1111/icad.12540
Chen J, Franklin JF, Spies TA (1993) Contrasting microclimates among clearcut, edge, and Interior of old-growth Douglas-fir forest. Agric for Meteorol 63:219–237. https://doi.org/10.1016/0168-1923(93)90061-L
R Core Team (2023) R: a language and environment for statistical computing
Djupström LB, Weslien J, Schroeder LM (2008) Dead wood and saproxylic beetles in set-aside and non set-aside forests in a boreal region. For Ecol Manag 255:3340–3350. https://doi.org/10.1016/j.foreco.2008.02.015
Ekström AL, Bergmark P, Hekkala AM (2021) Can multifunctional forest landscapes sustain a high diversity of saproxylic beetles? For Ecol Manag 490:119107. https://doi.org/10.1016/j.foreco.2021.119107
Gibb H, Hjältén J, Ball JP, Atlegrim O, Pettersson RB, Hilszczański J, Johansson T, Danell K (2006a) Effects of landscape composition and substrate availability on saproxylic beetles in boreal forests: a study using experimental logs for monitoring assemblages. Ecography 29:191–204. https://doi.org/10.1111/j.2006.0906-7590.04372.x
Gibb H, Pettersson RB, Hjältén J, Hilszczański J, Ball JP, Johansson T, Atlegrim O, Danell K (2006b) Conservation-oriented forestry and early successional saproxylic beetles: responses of functional groups to manipulated dead wood substrates. Biol Conserv 129:437–450. https://doi.org/10.1016/j.biocon.2005.11.010
Gran O, Götmark F (2021) Saproxylic beetles in pre-commercially thinned Norway spruce stands and woodland key habitats: how do conservation values differ? For Ecol Manag 479:118584. https://doi.org/10.1016/j.foreco.2020.118584
Grove SJ (2002) Saproxylic insect ecology and the sustainable management of forests. Annu Rev Ecol Syst 33:1–23. https://doi.org/10.1146/annurev.ecolsys.33.010802.150507
Hallberg-Sramek I, Bjärstig T, Nordin A (2020) Framing woodland key habitats in the Swedish media–how has the framing changed over time? Scand J for Res 35:198–209. https://doi.org/10.1080/02827581.2020.1761444
Hämäläinen A, Fahrig L, Strengbom J, Ranius T (2023) Effective management for deadwood-dependent lichen diversity requires landscape‐scale habitat protection. J Appl Ecol. https://doi.org/10.1111/1365-2664.14429
Hjältén J, Stenbacka F, Pettersson RB, Gibb H, Johansson T, Danell K, Ball JP, Hilszczański J (2012) Micro and macro-habitat associations in saproxylic beetles: implications for biodiversity management. PLoS ONE e41100. 7https://doi.org/10.1371/journal.pone.0041100
Hsieh T, Ma K, Chao A (2024) iNEXT: Interpolation and Extrapolation for Species Diversity. R package
Jacobsen RM, Burner RC, Olsen SL, Skarpaas O, Sverdrup-Thygeson A (2020) Near-natural forests harbor richer saproxylic beetle communities than those in intensively managed forests. For Ecol Manag 466:118124. https://doi.org/10.1016/j.foreco.2020.118124
Johansson T, Gibb H, Hjältén J, Pettersson RB, Hilszczański J, Alinvi O, Ball JP, Danell K (2007) The effects of substrate manipulations and forest management on predators of saproxylic beetles. For Ecol Manag 242:518–529. https://doi.org/10.1016/j.foreco.2007.01.064
Johansson V, Gustafsson L, Andersson P, Hylander K (2020) Fewer butterflies and a different composition of bees, wasps and hoverflies on recently burned compared to unburned clear-cuts, regardless of burn severity. For Ecol Manag 463:118033. https://doi.org/10.1016/j.foreco.2020.118033
Jonsell M, Abrahamsson M, Widenfalk L, Lindbladh M (2019) Increasing influence of the surrounding landscape on saproxylic beetle communities over 10 years succession in dead wood. For Ecol Manag 440:267–284. https://doi.org/10.1016/j.foreco.2019.02.021
Kärvemo S, Jönsson M, Hekkala AM, Sjögren J, Strengbom J (2021) Multi-taxon conservation in northern forest hot-spots: the role of forest characteristics and spatial scales. Landscape Ecol 36:989–1002. https://doi.org/10.1007/s10980-021-01205-x
Kouki J, Hyvärinen E, Lappalainen H, Martikainen P, Similä M (2012) Landscape context affects the success of habitat restoration: large-scale colonization patterns of saproxylic and fire‐associated species in boreal forests. Divers Distrib 18:348–355. https://doi.org/10.1111/j.1472-4642.2011.00839.x
Lassauce A, Paillet Y, Jactel H, Bouget C (2011) Deadwood as a surrogate for forest biodiversity: meta-analysis of correlations between deadwood volume and species richness of saproxylic organisms. Ecol Ind 11:1027–1039. https://doi.org/10.1016/j.ecolind.2011.02.004
Lettenmaier L, Seibold S, Bässler C, Brandl R, Gruppe A, Müller J, Hagge J (2022) Beetle diversity is higher in sunny forests due to higher microclimatic heterogeneity in deadwood. Oecologia 198:825–834. https://doi.org/10.1007/s00442-022-05141-8
Lindhe A, Lindelöw Å, Åsenblad N (2005) Saproxylic beetles in standing dead wood density in relation to substrate sun-exposure and diameter. Biodivers Conserv 14:3033–3053. https://doi.org/10.1007/s10531-004-0314-y
López-Bedoya PA, Magura T, Edwards FA, Edwards DP, Rey‐Benayas JM, Lövei GL, Noriega JA (2021) What level of native beetle diversity can be supported by forestry plantations? A global synthesis. Insect Conserv Divers 14:736–747. https://doi.org/10.1111/icad.12518
Lundmark H, Josefsson T, Östlund L (2013) The history of clear-cutting in northern Sweden–Driving forces and myths in boreal silviculture. For Ecol Manag 307:112–122. https://doi.org/10.1016/j.foreco.2013.07.003
Martikainen P, Kouki J (2003) Sampling the rarest: threatened beetles in boreal forest biodiversity inventories. Biodivers Conserv 12:1815–1831. https://doi.org/10.1023/A:1024132829581
McCune B, Grace JB (2002) Analysis of Ecological communities. MjM Software Design, Gleneden Beach
Nitare J (2019) Skyddsvärd skog - Naturvårdsarter och andra kriterier för naturvärdesbedömning. Skogsstyrelsen (In Swedish)
Oksanen J, Blanchet FG, Kindt R, Legendre P, Minchin PR, O’Hara RB et al (2022) Package ‘vegan’. Community ecology package
Olsson J, Johansson T, Jonsson BG, Hjältén J, Edman M, Ericson L (2012) Landscape and substrate properties affect species richness and community composition of saproxylic beetles. For Ecol Manag 286:108–120. https://doi.org/10.1016/j.foreco.2012.08.033
Potapov P, Hansen MC, Laestadius L, Turubanova S, Yaroshenko A, Thies C, Esipova E (2017) The last frontiers of wilderness: Tracking loss of intact forest landscapes from 2000 to 2013. Sci Adv 3:e1600821. https://doi.org/10.1126/sciadv.1600821
Ranius T, Johansson V, Schroeder M, Caruso A (2015) Relative importance of habitat characteristics at multiple spatial scales for wood-dependent beetles in boreal forest. Landscape Ecol 30:1931–1942. https://doi.org/10.1007/s10980-015-0221-5
Roberge JM, Fries C, Normark E, Mårald E, Sténs A, Sandström C, Sonesson J, Appelqvist C, Lundmark T (2020) Forest management in Sweden. Current practice and historical background. Swedish Forest Agency Report 4 2020. https://www.skogsstyrelsen.se/globalassets/om-oss/rapporter/rapporter-20222021202020192018/rapport-2020-4-forest-management-in-sweden.pdf
Rubene D, Schroeder M, Ranius T (2017) Effectiveness of local conservation management is affected by landscape properties: species richness and composition of saproxylic beetles in boreal forest clearcuts. For Ecol Manag 399:54–63. https://doi.org/10.1016/j.foreco.2017.05.025
SCB (2022) Formellt skyddad skogsmark, frivilliga avsättningar, hänsynsytor samt improduktiv skogsmark 2022. MI 41 2022A02 Statistikmyndigheten (In Swedish) https://www.scb.se/hitta-statistik/statistik-efter-amne/miljo/markanvandning/formellt-skyddad-skogsmark-frivilliga-avsattningar-hansynsytor-samt-improduktiv-skogsmark/
Seibold S, Thorn S (2018) The Importance of Dead-Wood Amount for Saproxylic insects and How It Interacts with Dead-Wood Diversity and Other Habitat Factors. In: Ulyshen M (ed) Saproxylic insects. Zoological Monographs, vol 1. Springer, Cham. https://doi.org/10.1007/978-3-319-75937-1_18
Seibold S, Bässler C, Brandl R, Büche B, Szallies A, Thorn S, Ulyshen MD, Müller J (2016) Microclimate and habitat heterogeneity as the major drivers of beetle diversity in dead wood. J Appl Ecol 53:934–943. https://doi.org/10.1111/1365-2664.12607
Siitonen J, Saaristo L (2000) Habitat requirements and conservation of Pytho Kolwensis, a beetle species of old-growth boreal forest. Biol Conserv 94:211–220. https://doi.org/10.1016/S0006-3207(99)00174-3
Skogsdata (2020) Aktuella uppgifter om de svenska skogarna från SLU Riksskogstaxeringen Tema: Den döda veden. Dept. of Forest Resource Management, Swedish University of Agricultural Sciences, Umeå (In Swedish with English summary) https://www.slu.se/globalassets/ew/org/centrb/rt/dokument/skogsdata/skogsdata_2020_webb.pdf
SLU (2010) Skogskartan. Dept. of Forest Resource Management, Swedish University of Agricultural Sciences, Umeå https://www.slu.se/en/Collaborative-Centres-and-Projects/the-swedish-national-forest-inventory/foreststatistics/slu-forest-map/
SLU ArtDatabanken (2020) Rödlistade arter i Sverige 2020. Swedish University of Agricultural Sciences (In Swedish with English summary) https://www.artdatabanken.se/publikationer/bestall-rodlista-2020/
Southwood TRE, Henderson PA (2000) Ecological methods. Blackwell Science, Oxford
Stenbacka F, Hjältén J, Hilszczański J, Dynesius M (2010) Saproxylic and non-saproxylic beetle assemblages in boreal spruce forests of different age and forestry intensity. Ecol Appl 20:2310–2321. https://doi.org/10.1890/09-0815.1
Stokland J, Jonsson BG, Siitonen J (2012) Biodiversity in dead wood. Cambridge University Press, Cambridge
Svensson J, Andersson J, Sandström P, Mikusiński G, Jonsson BG (2019) Landscape trajectory of natural boreal forest loss as an impediment to green infrastructure. Conserv Biol 33:152–163. https://doi.org/10.1111/cobi.13148
Svensson J, Bubnicki JW, Jonsson BG, Andersson J, Mikusiński G (2020) Conservation significance of intact forest landscapes in the Scandinavian Mountains Green Belt. Landscape Ecol 35:2113–2131. https://doi.org/10.1007/s10980-020-01088-4
Sverdrup-Thygeson A, Gustafsson L, Kouki J (2014) Spatial and temporal scales relevant for conservation of dead-wood associated species: current status and perspectives. Biodivers Conserv 23:513–535. https://doi.org/10.1007/s10531-014-0628-3
Swedish Environmental Protection Agency (2019) Public national database on spatial environmental data, Miljödataportalen, administrated by the Swedish Environmental Protection Agency, with data on ground cover data & proxy for continuity forests
Thorn S, Bässler C, Brandl R, Burton PJ, Cahall R, Campbell JL, Müller J (2018) Impacts of salvage logging on biodiversity: a meta-analysis. J Appl Ecol 55:279–289. https://doi.org/10.1111/1365-2664.12945
Timonen J, Siitonen J, Gustafsson L, Kotiaho JS, Stokland JN, Sverdrup-Thygeson A, Mönkkönen M (2010) Woodland key habitats in northern Europe: concepts, inventory and protection. Scand J for Res 25:309–324. https://doi.org/10.1080/02827581.2010.497160
Vrba P, Beneš J, Čížek L, Filippov P, Fric ZF, Hauck D, Konvičk M, Spitzer L (2024) Bark beetle outbreak and biodiversity in commercial spruce plantations: responses of four model groups. For Ecol Manag 555:121700. https://doi.org/10.1016/j.foreco.2024.121700
Watson JE, Evans T, Venter O, Williams B, Tulloch A, Stewart C, Thompson I, Ray JC, Murray K, Salazar A, McAlpine C (2018) The exceptional value of intact forest ecosystems. Nat Ecol Evol 2:599–610. https://doi.org/10.1038/s41559-018-0490-x
Wester J, Engström A (2016) Nulägesbeskrivning om nyckelbiotoper. Rapport 7/2016 Skogsstyrelsen. (In Swedish) https://shop.skogsstyrelsen.se/sv/publikationer/rapporter/nulagesbeskrivning-om-nyckelbiotoper.html
Zumr V, Nakládal O, Remeš J, Brestovanská T (2022) Diversity of click beetles in managed nonnative coniferous and native beech stands: consequences of changes in the structural and species composition of tree stands in Central Europe. For Ecosyst 9:100057. https://doi.org/10.1016/j.fecs.2022.100057
Acknowledgements
We thank the staff at Calluna AB and especially Joel Hallqvist for sampling and determination of beetles, and Sofie Wikberg for GIS-design for plot location.
Funding
Open access funding provided by Swedish University of Agricultural Sciences. This work was supported by the Formas grant 2018 − 00577 to LG and MattJ.
Author information
Authors and Affiliations
Contributions
LG initiated the study, coordinated the beetle sampling, and wrote substantial parts of the manuscript. JA performed the remote sensing analyses, was responsible for the selection of study sites incl. field visits, and wrote parts of the manuscript. MattJ took part in the initiation of the study and in the interpretation of results. MJ, TL, JS, JW contributed to the interpretation of results. VJ took part in the initiation of the study, performed the statistical analyses, contributed to the interpretation of results, and wrote important parts of the manuscript. All authors contributed to the study design and examined the manuscript critically.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gustafsson, L., Andersson, J., Jonsson, M. et al. Saproxylic beetles in Swedish boreal production forests in relation to local and landscape factors. J Insect Conserv (2024). https://doi.org/10.1007/s10841-024-00624-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10841-024-00624-w