Abstract
Haemodynamic monitoring and management are cornerstones of perioperative care. The goal of haemodynamic management is to maintain organ function by ensuring adequate perfusion pressure, blood flow, and oxygen delivery. We here present guidelines on “Intraoperative haemodynamic monitoring and management of adults having non-cardiac surgery” that were prepared by 18 experts on behalf of the German Society of Anaesthesiology and Intensive Care Medicine (Deutsche Gesellschaft für Anästhesiologie und lntensivmedizin; DGAI).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Background
Haemodynamic monitoring and management are cornerstones of perioperative care. The goal of haemodynamic management is to maintain organ function by ensuring adequate perfusion pressure, blood flow, and oxygen delivery to prevent perioperative complications – that remain as high as almost 20% in patients having elective non-cardiac surgery [1, 2].
We identified clinically important questions on intraoperative haemodynamic monitoring and management of adults having non-cardiac surgery, discussed them in a group of experts, and formulated consensus recommendations based on current evidence.
2 Methods
Initiated by the German Society of Anaesthesiology and Intensive Care Medicine (Deutsche Gesellschaft für Anästhesiologie und lntensivmedizin; DGAI) these guidelines were prepared between April 2022 and October 2023 by four coordinators (BS, DAR, MS, BJ) and 14 experts (all co-authors). We registered this project as S-class 1 guidelines at the Association of the Scientific Medical Societies in Germany (http://www.awmf.org). S-class 1 guidelines are expert recommendations developed by a representative group of experts based on informal consensus without formally assessing the quality of evidence.
We assumed that experts and clinicians may not necessarily consider the same topics clinically important. In May 2022, we, therefore, asked anaesthesiologists at three German university hospitals (Giessen, Hamburg, and Rostock) to submit questions on intraoperative haemodynamic monitoring and management they would expect to get answered in guidelines. The submitted questions were used to formulate 19 core questions. We discussed and answered these 19 questions (a) within groups of 2–3 experts and (b) within the group of all experts in two plenary meetings in September 2022 (virtual) and January 2023 (in-person). The revised final consensus recommendations (considering comments of the board and working groups of the DGAI) were agreed on by the experts and approved by the DGAI in September 2023. The guidelines (including a table detailing each expert’s conflicts of interest) were published online in German on October 4, 2023 (https://register.awmf.org/de/leitlinien/detail/001-049).
The strength of the recommendations is reflected by the wording – with “should”/”should not” reflecting strong recommendations, “ought”/”ought not” reflecting recommendations, and “may be considered” reflecting open recommendations [3].
3 Recommendations
3.1 How should oscillometric arterial pressure monitoring be performed?
3.1.1 Consensus recommendations
-
Oscillometric arterial pressure monitoring should – if possible – be performed on the upper arm.
-
For oscillometric arterial pressure monitoring, a cuff size appropriate for the circumference of the upper arm should be selected and the cuff should be placed tightly around the upper arm without contact with the olecranon.
-
For oscillometric arterial pressure monitoring, the upper arm cuff should be positioned at the level of the heart and external compression or manipulation of the cuff during the measurement should be avoided.
-
For patients under general anaesthesia, oscillometric arterial pressure monitoring ought to be performed every 3 min. The measurement interval should be adapted to the clinical context.
3.1.2 Summary of evidence
Automated oscillometry is the most commonly used method for non-invasive arterial pressure monitoring in perioperative medicine. Selecting a cuff adequately sized in relation to the upper arm circumference is essential for correct measurements. Using a cuff that is too small will provide erroneously high arterial pressures. Using a cuff that is too large will provide erroneously low arterial pressures [4]. The cuff should be placed around the upper arm without contact to the olecranon to allow circular compression. The cuff should be positioned at the level of the heart during the measurement. A cuff positioned above the level of the heart during measurements will provide erroneously low arterial pressure values due to hydrostatic pressure differences [5]. A cuff positioned below the level of the heart during measurements will provide erroneously high arterial pressure values [5]. External compression, vibration, or muscle activity during the measurement will cause artifacts.
In general, oscillometry systematically overestimates low arterial pressures and underestimates high arterial pressures [6,7,8]. This may lead to delayed detection of hypo- or hypertension.
For patients under general anaesthesia, measurements ought to be taken every 3 min. If closer arterial pressure monitoring is deemed necessary, continuous arterial pressure monitoring is indicated. The measurement interval for oscillometric arterial pressure measurement should be adapted to the clinical context.
3.2 In which patients should arterial pressure be measured continuously?
3.2.1 Consensus recommendation
-
Continuous arterial pressure monitoring should be used in all patients who – because of anaesthesiologic or surgical procedures or concomitant diseases – are at risk for complications associated with hypotension or hypertension.
3.2.2 Summary of evidence
Arterial pressure can be continuously measured invasively with an arterial catheter (reference method) or non-invasively (e.g., using a finger cuff method). Continuous arterial pressure monitoring allows detecting arterial pressure changes in real-time and can thus help reduce arterial pressure fluctuations and hypotension [9,10,11,12,13].
Whether continuous arterial pressure monitoring ought to be used depends on surgical and patient-specific risks for arterial pressure fluctuations [14]. Close arterial pressure monitoring is required during intracranial and vascular surgery, surgery in sitting position, and surgery with expected hypotension. Patient-specific factors for continuous arterial pressure monitoring include cardiovascular diseases, high risk for cardiovascular complications, elevated intracranial pressure (with the risk of low cerebral perfusion pressure), and aneurysms at risk of rupture. Additionally, continuous arterial pressure monitoring may also be indicated in patients in whom intermittent arterial pressure monitoring using oscillometry is inaccurate or difficult to perform (e.g., severly obese patients) [15, 16].
3.3 How should continuous arterial pressure monitoring be performed?
3.3.1 Consensus recommendations
-
Continuous arterial pressure monitoring ought to be performed with an arterial catheter.
-
In low or moderate risk patients, non-invasive continuous arterial pressure monitoring may be considered.
3.3.2 Summary of evidence
Direct intraarterial arterial pressure measurement with an arterial catheter is the clinical reference method for measuring arterial pressure [17]. When used correctly, direct intraarterial arterial pressure monitoring is more accurate than non-invasive arterial pressure measurement. When correctly measured intraarterial and non-invasive arterial pressure measurements differ, intraarterial arterial pressure measurements should be used to make treatment decisions.
Non-invasive continuous arterial pressure measurements are not interchangeable with intraarterial measurements [18, 19], but may be considered for continuous arterial pressure monitoring in low or moderate risk patients.
3.4 Should the arterial catheter for intraarterial arterial pressure monitoring be inserted before induction of anaesthesia?
3.4.1 Consensus recommendation
-
In patients with an indication for intraarterial arterial pressure monitoring, the arterial catheter should be inserted before induction of anaesthesia.
3.4.2 Summary of evidence
Hypotension after induction of anaesthesia is common and associated with postoperative organ injury [20, 21]. In contrast to hypotension during surgery, hypotension after induction of anaesthesia is mainly caused by anaesthetic management and can thus be anticipated [22]. Intermittent oscillometric arterial pressure monitoring may miss hypotension – especially because it systematically overestimates low arterial pressures [6, 10, 11, 13]. In a randomised trial of 242 non-cardiac surgery patients, continuous intraarterial – compared to intermittent oscillometric – arterial pressure monitoring significantly reduced the duration and severity of hypotension during induction of anaesthesia [10].
3.5 Which artery should be used for intraarterial arterial pressure monitoring?
3.5.1 Consensus recommendation
-
Arterial catheters should primarily be inserted in the radial artery.
3.5.2 Summary of evidence
In general, arterial catheters used in perioperative and intensive care medicine are associated with a low risk for complications due to catheter insertion [23,24,25]. Insertion of arterial catheters into the radial artery is associated with a lower risk compared to catheter insertion in the brachial or femoral artery (2.7 per 10,000 radial, 9.0 per 10,000 femoral, and 12.3 per 10,000 brachial artery catheters) [24]. The radial artery may also be preferred because of the collateral circulation of the hand, its easy accessibility, and good possibility for compression in case of bleeding. Arterial catheters with a smaller diameter (20 G) are associated with fewer complications than arterial catheters with a larger diameter (18 G) [24]. Arterial catheters ought to be inserted using ultrasound guidance [26,27,28].
3.6 What sources of error must be considered when intraarterial arterial pressure monitoring is used?
3.6.1 Consensus recommendations
-
The pressure transducer should always be checked for correct levelling or zeroing.
-
The dynamic response of the measurement system should be closely checked.
3.6.2 Summary of evidence
The pressure transducer must be levelled or zeroed to ensure correct intraarterial arterial pressure monitoring [29]. Levelling and zeroing procedures differ dependent on whether pressure transducers are used without or with a zero-line (i.e., a fluid-filled tube) [29].
If used without a zero-line, the pressure transducer must be positioned at the reference level (usually the right atrium [30]) to account for hydrostatic pressure differences [29]. A height difference of 10 cm between the transducer and the reference level results in an arterial pressure difference of approximately 7.5 mmHg.
If the pressure transducer is used with a zero-line, the zero-line is attached to the patient with its free end at the reference level and the zeroing function on the monitor is activated. Using a zero-line can be advantageous for patients in whom the pressure transducer cannot be positioned accurately during surgery [31]. Whenever the patient is being moved relative to the height of the pressure transducer, a new zeroing manoeuvre must be performed.
The measurement system should be closely checked for an adequate dynamic response – e.g., by means of a fast-flush test – to avoid damping phenomena [29, 32]. An underdamped system may overestimate systolic arterial pressure and underestimate diastolic arterial pressure [29]. Common reasons for underdamping include stiff tubing or modifications of the measurement system by adding additional tubes and stopcocks [29]. An overdamped system may underestimate systolic arterial pressure and overestimate diastolic arterial pressure [29]. Common reasons for overdamping include low infusion bag pressure, air bubbles in the measurement system, blood clots, lose or open connections, and kinking or obstruction of the catheter [29].
3.7 How must the position of the patient be considered when measuring arterial pressure?
3.7.1 Consensus recommendations
-
Especially during changes in patient position, arterial pressure ought to be measured closely or – even better – continuously.
-
In all positions where the usual reference level “right atrium” is lower than the cranial base, non-invasively measured mean arterial pressure should be corrected for the difference in hydrostatic pressure or the reference level of the continuous arterial pressure measurement should be set at the level of the cranial base.
3.7.2 Summary of evidence
Various intraoperative positions (e.g., sitting position, prone position, lateral position, and corresponding modifications) can contribute to haemodynamic instability [33,34,35]. In positions with elevated upper body, a major cause of hypotension is the redistribution of blood from central to peripheral compartments with a consecutive decrease in cardiac preload. In flank and prone positions, compression of the inferior vena cava may partially obstruct venous return. Not only positioning itself but also repositioning can lead to a decrease in arterial pressure. Therefore, arterial pressure ought to be monitored closely or even continuously during changes in patient position [36].
Pressure differences between central and cerebral mean arterial pressure occur in all positions in which the head is higher than the usual reference level “right atrium” due to hydrostatic pressure differences. In these positions, mean arterial pressure targets should be corrected for hydrostatic pressure differences or the reference level should be set at the level of the cranial base to correctly monitor arterial pressure in the circle of Willis [37, 38]. Either procedure ought to be documented in the anaesthesia record. In contrast, with head-down positions, arterial pressure targets should not be corrected, and the right atrium should be used as the reference level [39].
3.8 Which arterial pressure component should be used for arterial pressure management?
3.8.1 Consensus recommendation
-
Mean arterial pressure should be used for intraoperative arterial pressure management.
3.8.2 Summary of evidence
A retrospective database study showed that the strength of the association between hypotension and acute kidney or myocardial injury is similarly strong when hypotension is defined using mean or systolic arterial pressure – but substantially weaker when using diastolic arterial pressure [40].
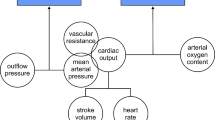
Intraoperative arterial pressure management should be guided by mean arterial pressure, as mean arterial pressure is the inflow pressure for most organ systems. In addition, measurement of mean arterial pressure is less dependent on the measurement location than measurement of systolic and diastolic arterial pressure as mean arterial pressure changes only slightly along the arterial tree – while systolic arterial pressure increases progressively towards the periphery and diastolic arterial pressure decreases [41].
3.9 Which arterial pressure value should be targeted?
3.9.1 Consensus recommendation
-
Mean arterial pressure should be maintained above 65 mmHg.
3.9.2 Summary of evidence
Observational research provides compelling evidence that intraoperative hypotension is associated with organ injury [21, 40, 42,43,44,45,46,47,48,49,50,51]. The association between hypotension and organ injury depends on the severity and duration of hypotension [21, 40, 42,43,44,45,46,47,48,49,50,51]. On a population level, harm thresholds for acute kidney injury and acute myocardial injury are 60–70 mmHg for mean arterial pressure and 90–100 mmHg for systolic arterial pressure [40]. Harm thresholds for individual patients remain unknown. There are only few randomised trials on targeted intraoperative arterial pressure management [52,53,54].
In a trial of 458 high-risk patients, keeping intraoperative mean arterial pressure ≥ 75 mmHg – compared to ≥ 60 mmHg – did not reduce the incidence of a composite outcome of acute myocardial injury, acute kidney injury, other cardiovascular complications, or death [52]. The POISE-3 trial [54] found no clinically important difference in the incidence of severe cardiovascular complications between patients randomised to a hypotension avoidance strategy with an intraoperative mean arterial pressure target of ≥ 80 mmHg and a hypertension avoidance strategy with a mean arterial pressure target of ≥ 60 mmHg.
As preoperative arterial pressure substantially varies among individuals [20], individualised arterial pressure management may reduce hypotension-associated complications. In a multicentre trial of 198 non-cardiac surgery patients, individualised arterial pressure management (based on a single preoperative systolic arterial pressure value) reduced the incidence of a composite outcome of systemic inflammatory response syndrome and organ dysfunction within one week after surgery compared to routine arterial pressure management [53].
Considering the association between intraoperative hypotension and organ injury, clinicians should maintain mean arterial pressure above 65 mmHg or systolic arterial pressure above 90–100 mmHg [17, 55].
There is no clear evidence that intraoperative hypertension is associated with organ injury in non-cardiac surgery patients [56]. A general upper intervention threshold for arterial pressure cannot be recommended.
3.10 Which heart rate value should be targeted?
3.10.1 Consensus recommendations
-
Bradycardia should be treated when it is accompanied by clinically important hypotension, reduced perfusion, or reduced oxygen delivery.
-
If tachycardia is present, hypovolaemia should be excluded.
3.10.2 Summary of evidence
Heart rate, together with stroke volume, is a primary determinant of cardiac output and thus oxygen delivery [57]. In the general adult population, bradycardia is usually defined as a heart rate < 60 beats per minute [58] and tachycardia as a heart rate > 100 beats per minute [59]. These thresholds are usually also considered during surgery. There is insufficient evidence to recommend heart rate target values.
Perioperative bradycardia is common [60] and often caused by general anaesthesia [61,62,63], vasoactive drugs [64], and surgery [65]. Bradycardia can lead to hypotension [66] and a critical decrease in oxygen delivery if the heart rate-related reduction in cardiac output cannot be compensated by an increase in stroke volume [67].
Perioperative tachycardia may be caused by hypovolaemia, inadequate depth of anaesthesia or insufficient analgesia, or a systemic inflammatory response. Critical shortening of diastole may result in insufficient myocardial oxygen delivery with concomitant increased oxygen consumption which should be considered especially in patients with diastolic dysfunction or heart failure [67,68,69].
3.11 In which patients should stroke volume/cardiac output be monitored?
3.11.1 Consensus recommendation
-
Stroke volume/cardiac output monitoring may be considered in patients with a high risk for complications.
3.11.2 Summary of evidence
The decision to monitor stroke volume/cardiac output ought to be based on the risk for intra- and postoperative complications [70]. The risk for complications depends on patient-related and surgery-related risk factors. Patient-related risk factors include the underlying diagnosis requiring surgery, age, and cardiovascular or pulmonary comorbidities [70,71,72]. Surgery-related risk factors include invasiveness of the procedure, risk for high blood loss, or emergency surgery [71, 73].
Stroke volume/cardiac output monitoring may be considered when the individual patient-related or surgery-related risk is high. Especially in patients with a high risk for perioperative complications, stroke volume/cardiac output-guided haemodynamic management may reduce the risk for postoperative complications [74, 75]. Monitoring stroke volume/cardiac output alone will not improve postoperative outcomes [76].
3.12 Which stroke volume/cardiac output value should be targeted?
3.12.1 Consensus recommendations
-
Stroke volume/cardiac output target values should be individually defined for each patient.
-
Stroke volume/cardiac output should be interpreted in the context of clinical and metabolic signs of hypoperfusion.
-
Stroke volume/cardiac output should not be routinely maximised.
3.12.2 Summary of evidence
Stroke volume/cardiac output is regulated to meet metabolic needs. Resting cardiac output depends on age, sex, and comorbidities [77, 78]. Haemodynamic therapy guided by stroke volume/cardiac output thus ought to consider variables of oxygen delivery and oxygen consumption. The targeted stroke volume/cardiac output value ought to be high enough to ensure sufficient oxygen delivery for the individual patient in each situation.
Stroke volume/cardiac output-guided haemodynamic management may reduce postoperative complications [74, 79, 80]. Most blood flow-guided haemodynamic management concepts target maximal stroke volume/cardiac output values for the individual patient [80]. Small randomised trials suggest that cardiac output-guided haemodynamic management targeting preoperative baseline or post-induction cardiac index may reduce postoperative complications compared to routine care [81, 82]. Optimal stroke volume/cardiac output target values remain unclear, and may depend on the degree of surgical trauma and systemic inflammation [83]. We recommend not routinely targeting individual maximal values (OPTIMISE II [84] – results presented at the EBPOM World Congress of Prehabilitation Medicine 2023 in London on July 6, 2023) or „supranormal“ values [85,86,87].
3.13 Which tests should be used to assess fluid responsiveness?
3.13.1 Consensus recommendations
-
To assess fluid responsiveness, dynamic preload variables (e.g., pulse pressure variation or stroke volume variation) should be used in mechanically ventilated patients.
-
If dynamic preload variables cannot be used, stroke volume or cardiac output-based fluid challenge tests ought to be performed to assess fluid responsiveness.
-
Static preload variables (e.g., central venous pressure) should not be used to assess fluid responsiveness.
-
Even in fluid-responsive patients, the indication for fluid administration should be determined individually based on haemodynamics and clinical context.
3.13.2 Summary of evidence
The dynamic preload variables pulse pressure variation (PPV) and stroke volume variation (SVV) can be used to predict fluid responsiveness in mechanically ventilated patients with tidal volumes of at least 8 ml/kg and sinus rhythm [88,89,90,91]. Thresholds to distinguish between fluid responsive and non-responsive patients are 11% (8–15%) for PPV and 11% (7.5–15.5%) for SVV [92]. However, there are grey zones around these thresholds in which the ability of PPV and SVV to predict fluid responsiveness is limited [93]. Limitations for the use of PPV and SVV – such as low heart rate/respiratory rate ratio, irregular heart rhythm, mechanical ventilation with low tidal volumes, increased abdominal pressure, open chest conditions, and spontaneous breathing – need to be considered in clinical practice [94, 95].
If dynamic preload variables cannot be used, a fluid challenge considering fluid-induced changes in stroke volume/cardiac output ought to be performed to assess fluid responsiveness [96,97,98]. During a fluid challenge, a certain amount of fluid is administered over a short time and changes in stroke volume/cardiac output are evaluated [97, 98]. An increase in stroke volume of 10–15% is considered a sign of fluid responsiveness. However, fluid challenges are not only a test but a therapeutic intervention bearing the risk to give fluids to patients who are not fluid responsive [99, 100]. In some patients, an increase in stroke volume may cause a (physiological) reduction in heart rate resulting in no net effect on cardiac output. It is thus useful to evaluate both stroke volume and cardiac output when assessing fluid responsiveness.
Static pressure-based and volumetric preload variables, such as central venous pressure or global end-diastolic volume, should not be used to assess fluid responsiveness, but still are important in the assessment of haemodynamics [89].
Fluid administration should always be indicated carefully – also in patients with signs of fluid responsiveness – considering other haemodynamic variables and the clinical context; fluid responsiveness does not equal the need for fluids.
3.14 When should intraoperative echocardiography be performed?
3.14.1 Consensus recommendations
-
Echocardiography should be performed in patients with haemodynamic instability not responding to initial treatment, especially when the cause of haemodynamic instability is unclear.
-
Echocardiography may be considered to guide haemodynamic therapy.
-
Echocardiography images and results should be saved in the patient’s medical records.
3.14.2 Summary of evidence
During surgery, transthoracic echocardiography (TTE) or transoesophageal echocardiography (TEE) can be used to directly assess cardiac anatomy and function [101]. TTE is non-invasive and therefore ought to be preferred [102, 103].
Intraoperative focused echocardiography should be performed in patients with haemodynamic instability not responding to initial treatment, especially when the cause of haemodynamic instability is unclear. Echocardiography may be considered to guide haemodynamic therapy.
Preoperative focused echocardiography ought to be performed in (emergency) patients with suspected or known clinically relevant cardiovascular disease to better interpret intraoperative findings during haemodynamic instability [104,105,106,107,108,109].
Echocardiography, especially TEE, should be performed by experienced examiners [110]. Training may be certified by national [111, 112] or international certificates.
3.15 Should urine output be used to assess haemodynamics?
3.15.1 Consensus recommendation
-
Urine output alone should not be used to diagnose hypovolaemia or to guide haemodynamic management.
3.15.2 Summary of evidence
Normal urine output is often considered a sign of adequate volume status and cardiac output. Oliguria may result from haemodynamic instability with reduced renal perfusion. In addition, increased intra-abdominal pressure, e.g., during laparoscopic procedures, can cause transient oliguria. During surgery, possible causes of oliguria should be considered and treated if necessary. Intraoperative oliguria is associated with postoperative acute kidney injury in patients having non-cardiac surgery [113, 114]. However, intraoperative oliguria is a poor predictor of postoperative acute kidney injury [115]. Targeting urine output does not reduce acute kidney injury [116, 117] or mortality [118].
3.16 Should lactate be used to assess haemodynamics?
3.16.1 Consensus recommendations
-
When hypoperfusion or inadequate tissue oxygenation are suspected, lactate should be measured to assess haemodynamics.
-
Elevated lactate should be interpreted considering possible non-haemodynamic causes.
3.16.2 Summary of evidence
Lactate levels > 2 mmol/l are considered pathological. Perioperative lactataemia may indicate anaerobic metabolism as a result of hypoperfusion, insufficient oxygen delivery or impaired oxygen utilisation [119, 120]. Perioperative lactataemia, therefore, ought to be evaluated as a sign of hypoperfusion and possible risk factor for postoperative complications – considering other diagnoses such as liver failure, medications, or thiamine deficiency [121, 122]. In addition to single lactate measurements, lactate clearance is an important prognostic factor. During haemodynamic instability or lactataemia, lactate measurements ought to be repeated in a regular interval, e.g., at least every two hours as recommended in patients with shock [123]. Elevated lactate is an unspecific marker that always ought to be interpreted in context with haemodynamic variables.
3.17 Should central venous oxygen saturation be used to assess haemodynamics?
3.17.1 Consensus recommendation
-
If hypoperfusion or inadequate tissue oxygenation is suspected, central venous oxygen saturation may be used for additional assessment of haemodynamics.
3.17.2 Summary of evidence
Central venous oxygen saturation (ScvO2) reflects the global ratio of oxygen delivery and oxygen consumption [124]. There is thus a close relationship between ScvO2 and cardiac output. At the same time, ScvO2 also reflects oxygen uptake and metabolism [125]. Normal ScvO2 (> 70%) does not exclude regional hypoperfusion or tissue hypoxemia [126]. ScvO2 is also influenced by other factors, such as arterial oxygen content, body temperature, and level of sedation [124, 127]. Low ScvO2 is associated with complications in patients having non-cardiac surgery [128, 129]. However, in a trial of 241 patients, ScvO2-guided fluid therapy did not reduce postoperative complications compared to routine care in patients having colorectal surgery [130].
3.18 What is the role of monitoring the microcirculation using vital microscopy?
3.18.1 Consensus recommendation
-
Monitoring the microcirculation using vital microscopy should not be used to guide haemodynamic therapy.
3.18.2 Summary of evidence
The functionality of the microcirculation is essential for a sufficient oxygen and nutrition exchange and thus organ function [131]. The regional microcirculation can be visualised and quantified using vital microscopy. Direct monitoring of the microcirculation, however, is technically challenging and analysis is time-consuming [132]. Vital microscopy is usually performed in the sublingual area because it is easily accessible [132]. The sublingual microcirculation not necessarily reflects the microcirculation in other regions of the body [133, 134]. In patients having elective non-cardiac surgery, the microcirculation usually remains intact and preserved [135,136,137,138]. Further studies are needed to investigate whether therapeutic interventions targeting the microcirculation may have positive effects [139].
3.19 Should near-infrared spectroscopy be used to assess haemodynamics?
3.19.1 Consensus recommendation
-
Near-infrared spectroscopy may be considered to complement the hemodynamic assessment.
3.19.2 Summary of evidence
Near-infrared spectroscopy (NIRS) allows measurement of the regional oxygen saturation in vessels as a surrogate for tissue oxygenation. In contrast to pulse oximetry, NIRS cannot differentiate between arterial and venous oxygen saturation. Absolute values of regional oxygen saturation show a high inter-individual variability. Thus, relative changes compared to baseline values are often considered [140,141,142]. NIRS is most commonly used to monitor cerebral oxygen saturation. The effect of using perioperative NIRS monitoring on postoperative cerebral outcomes in adults remains to be elucidated [143]. In addition, NIRS harm thresholds as well as effects of therapeutic interventions on NIRS need to be investigated.
4 Strengths and limitations
The core questions discussed in these guidelines were proposed by clinicians at three German university hospitals and thus represent a broad spectrum of anaesthesiologists with various levels of experience. The practical consensus recommendations thus focus on thematic areas which remained unclear for clinicians.
We carefully selected a representative group of 18 members of the DGAI who have a solid scientific track record in the field of intraoperative haemodynamic monitoring and management. Nevertheless, the consensus process is based on expert interpretation during discussions between a limited number of experts. There thus remains some risk of bias. Furthermore, the consensus recommendations result from unsystematic literature review with expert interpretation. We did not perform a systematic review or meta-analysis. Additionally, the consensus recommendations are based on informal consensus without formally assessing the quality of the evidence.
5 Summary
Haemodynamic monitoring and management are cornerstones of perioperative care. These guidelines on “Intraoperative haemodynamic monitoring and management of adults having non-cardiac surgery” were prepared by 18 experts on behalf of the DGAI and provide expert consensus recommendations addressing 19 core questions around intraoperative haemodynamic monitoring and management.
Data availability
No datasets were generated or analysed.
References
Pearse RM, Moreno RP, Bauer P, Pelosi P, Metnitz P, Spies C, Vallet B, Vincent JL, Hoeft A, Rhodes A, European Surgical Outcomes Study group for the Trials groups of the European Society of Intensive Care Medicine and the European Society of Anaesthesiology. Mortality after surgery in Europe: a 7 day cohort study. Lancet. 2012;380:1059–65. https://doi.org/10.1016/S0140-6736(12)61148-9.
International Surgical Outcomes Study Group. Global patient outcomes after elective surgery: prospective cohort study in 27 low-, middle- and high-income countries. Br J Anaesth. 2016;117:601–9. https://doi.org/10.1093/bja/aew316.
German Society of the Scientific Medical Societies (AWMF) - Standing Guidelines Comission. (2012). AWMF Guidance Manual and Rules for Guidline Development, 1st edition. Available at: http://www.awmf.org/leitlinien/awmf-regelwerk.html (accessed on 22.11.2023).
Pickering TG, Hall JE, Appel LJ, Falkner BE, Graves J, Hill MN, Jones DW, Kurtz T, Sheps SG, Roccella EJ. Recommendations for blood pressure measurement in humans and experimental animals: part 1: blood pressure measurement in humans: a statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on high blood pressure research. Circulation. 2005;111:697–716. https://doi.org/10.1161/01.CIR.0000154900.76284.F6.
Netea RT, Lenders JW, Smits P, Thien T. Both body and arm position significantly influence blood pressure measurement. J Hum Hypertens. 2003;17:459–62. https://doi.org/10.1038/sj.jhh.1001573.
Wax DB, Lin HM, Leibowitz AB. Invasive and concomitant noninvasive intraoperative blood pressure monitoring: observed differences in measurements and associated therapeutic interventions. Anesthesiology. 2011;115:973–8. https://doi.org/10.1097/ALN.0b013e3182330286.
Meidert AS, Dolch ME, Muhlbauer K, Zwissler B, Klein M, Briegel J, Czerner S. Oscillometric versus invasive blood pressure measurement in patients with shock: a prospective observational study in the emergency department. J Clin Monit Comput. 2021;35:387–93. https://doi.org/10.1007/s10877-020-00482-2.
Picone DS, Schultz MG, Otahal P, Aakhus S, Al-Jumaily AM, Black JA, Bos WJ, Chambers JB, Chen CH, Cheng HM, Cremer A, Davies JE, Dwyer N, Gould BA, Hughes AD, Lacy PS, Laugesen E, Liang F, Melamed R, Muecke S, Ohte N, Okada S, Omboni S, Ott C, Peng X, Pereira T, Pucci G, Rajani R, Roberts-Thomson P, Rossen NB, Sueta D, Sinha MD, Schmieder RE, Smulyan H, Srikanth VK, Stewart R, Stouffer GA, Takazawa K, Wang J, Westerhof BE, Weber F, Weber T, Williams B, Yamada H, Yamamoto E, Sharman JE. Accuracy of Cuff-measured blood pressure: systematic reviews and Meta-analyses. J Am Coll Cardiol. 2017;70:572–86. https://doi.org/10.1016/j.jacc.2017.05.064.
Juri T, Suehiro K, Kimura A, Mukai A, Tanaka K, Yamada T, Mori T, Nishikawa K. Impact of continuous non-invasive blood pressure monitoring on hemodynamic fluctuation during general anesthesia: a randomized controlled study. J Clin Monit Comput. 2018;32:1005–13. https://doi.org/10.1007/s10877-018-0125-4.
Kouz K, Wegge M, Flick M, Bergholz A, Moll-Khosrawi P, Nitzschke R, Trepte CJC, Krause L, Sessler DI, Zollner C, Saugel B. Continuous intra-arterial versus intermittent oscillometric arterial pressure monitoring and hypotension during induction of anaesthesia: the AWAKE randomised trial. Br J Anaesth. 2022;129:478–86. https://doi.org/10.1016/j.bja.2022.06.027.
Maheshwari K, Khanna S, Bajracharya GR, Makarova N, Riter Q, Raza S, Cywinski JB, Argalious M, Kurz A, Sessler DI. A randomized trial of continuous noninvasive blood pressure monitoring during noncardiac surgery. Anesth Analg. 2018;127:424–31. https://doi.org/10.1213/ANE.0000000000003482.
Naylor AJ, Sessler DI, Maheshwari K, Khanna AK, Yang D, Mascha EJ, Suleiman I, Reville EM, Cote D, Hutcherson MT, Nguyen BM, Elsharkawy H, Kurz A. Arterial Catheters for Early Detection and Treatment of Hypotension during major noncardiac surgery: a Randomized Trial. Anesth Analg. 2020;131:1540–50. https://doi.org/10.1213/ANE.0000000000004370.
Kouz K, Weidemann F, Naebian A, Lohr A, Bergholz A, Thomsen KK, Krause L, Petzoldt M, Moll-Khosrawi P, Sessler DI, Flick M, Saugel B. Continuous finger-cuff versus intermittent oscillometric arterial pressure monitoring and hypotension during induction of anesthesia and noncardiac surgery: the DETECT Randomized Trial. Anesthesiology. 2023;139:298–308. https://doi.org/10.1097/ALN.0000000000004629.
Meidert AS, Briegel J, Saugel B. [Principles and pitfalls of arterial blood pressure measurement]. Anaesthesist. 2019;68:637–50. https://doi.org/10.1007/s00101-019-0614-y.
Hansen J, Pohlmann M, Beckmann JH, Klose P, Gruenewald M, Renner J, Lorenzen U, Elke G. Comparison of oscillometric, non-invasive and invasive arterial pressure monitoring in patients undergoing laparoscopic bariatric surgery - a secondary analysis of a prospective observational study. BMC Anesthesiol. 2022;22:83. https://doi.org/10.1186/s12871-022-01619-3.
Schumann R, Meidert AS, Bonney I, Koutentis C, Wesselink W, Kouz K, Saugel B. Intraoperative blood pressure monitoring in obese patients. Anesthesiology. 2021;134:179–88. https://doi.org/10.1097/ALN.0000000000003636.
Saugel B, Sessler DI. Perioperative blood pressure management. Anesthesiology. 2021;134:250–61. https://doi.org/10.1097/ALN.0000000000003610.
Saugel B, Hoppe P, Nicklas JY, Kouz K, Korner A, Hempel JC, Vos JJ, Schon G, Scheeren TWL. Continuous noninvasive pulse wave analysis using finger cuff technologies for arterial blood pressure and cardiac output monitoring in perioperative and intensive care medicine: a systematic review and meta-analysis. Br J Anaesth. 2020;125:25–37. https://doi.org/10.1016/j.bja.2020.03.013.
Kim SH, Lilot M, Sidhu KS, Rinehart J, Yu Z, Canales C, Cannesson M. Accuracy and precision of continuous noninvasive arterial pressure monitoring compared with invasive arterial pressure: a systematic review and meta-analysis. Anesthesiology. 2014;120:1080–97. https://doi.org/10.1097/ALN.0000000000000226.
Saugel B, Reese PC, Sessler DI, Burfeindt C, Nicklas JY, Pinnschmidt HO, Reuter DA, Sudfeld S. Automated ambulatory blood pressure measurements and intraoperative hypotension in patients having noncardiac surgery with General Anesthesia: a prospective observational study. Anesthesiology. 2019;131:74–83. https://doi.org/10.1097/ALN.0000000000002703.
Maheshwari K, Turan A, Mao G, Yang D, Niazi AK, Agarwal D, Sessler DI, Kurz A. The association of hypotension during non-cardiac surgery, before and after skin incision, with postoperative acute kidney injury: a retrospective cohort analysis. Anaesthesia. 2018;73:1223–8. https://doi.org/10.1111/anae.14416.
Saugel B, Bebert EJ, Briesenick L, Hoppe P, Greiwe G, Yang D, Ma C, Mascha EJ, Sessler DI, Rogge DE. Mechanisms contributing to hypotension after anesthetic induction with sufentanil, propofol, and rocuronium: a prospective observational study. J Clin Monit Comput. 2022;36:341–7. https://doi.org/10.1007/s10877-021-00653-9.
Scheer B, Perel A, Pfeiffer UJ. Clinical review: complications and risk factors of peripheral arterial catheters used for haemodynamic monitoring in anaesthesia and intensive care medicine. Crit Care. 2002;6:199–204. https://doi.org/10.1186/cc1489.
Nuttall G, Burckhardt J, Hadley A, Kane S, Kor D, Marienau MS, Schroeder DR, Handlogten K, Wilson G, Oliver WC. Surgical and Patient Risk factors for severe arterial line complications in adults. Anesthesiology. 2016;124:590–7. https://doi.org/10.1097/ALN.0000000000000967.
Singh A, Bahadorani B, Wakefield BJ, Makarova N, Kumar PA, Tong MZ, Sessler DI, Duncan AE. Brachial arterial pressure monitoring during cardiac surgery rarely causes complications. Anesthesiology. 2017;126:1065–76. https://doi.org/10.1097/ALN.0000000000001626.
Moussa Pacha H, Alahdab F, Al-Khadra Y, Idris A, Rabbat F, Darmoch F, Soud M, Zaitoun A, Kaki A, Rao SV, Kwok CS, Mamas MA, Alraies MC. Ultrasound-guided versus palpation-guided radial artery catheterization in adult population: a systematic review and meta-analysis of randomized controlled trials. Am Heart J. 2018;204:1–8. https://doi.org/10.1016/j.ahj.2018.06.007.
Cho SA, Jang YE, Ji SH, Kim EH, Lee JH, Kim HS, Kim JT. Ultrasound-guided arterial catheterization. Anesth Pain Med (Seoul). 2021;16:119–32. https://doi.org/10.17085/apm.21012.
Wu G, Chen C, Gu X, Yao Y, Yuan D, Lv J, Zhao B, Wang Q. Ultrasound-guided dynamic needle-tip positioning method is Superior to Conventional Palpation and Ultrasound Method in arterial catheterization. J Clin Med. 2022;11:6539. https://doi.org/10.3390/jcm11216539.
Saugel B, Kouz K, Meidert AS, Schulte-Uentrop L, Romagnoli S. How to measure blood pressure using an arterial catheter: a systematic 5-step approach. Crit Care. 2020;24:172. https://doi.org/10.1186/s13054-020-02859-w.
Magder S. Invasive intravascular hemodynamic monitoring: technical issues. Crit Care Clin. 2007;23. https://doi.org/10.1016/j.ccc.2007.07.004. :401 – 14.
Oh C, Lee S, Jeon S, Park H, Chung W, Shim MS, Yoon SH, Kim YH, Lee SY, Hong B. Errors in pressure measurements due to changes in pressure transducer levels during adult cardiac surgery: a prospective observational study. BMC Anesthesiol. 2023;23:8. https://doi.org/10.1186/s12871-023-01968-7.
Gardner RM. Direct blood pressure measurement–dynamic response requirements. Anesthesiology. 1981;54. https://doi.org/10.1097/00000542-198103000-00010. 227 – 36.
Rains DD, Rooke GA, Wahl CJ. Pathomechanisms and complications related to patient positioning and anesthesia during shoulder arthroscopy. Arthroscopy. 2011. https://doi.org/10.1016/j.arthro.2010.09.008. 27:532 – 41.
Papadonikolakis A, Wiesler ER, Olympio MA, Poehling GG. Avoiding catastrophic complications of stroke and death related to shoulder surgery in the sitting position. Arthroscopy. 2008;24:481–2. https://doi.org/10.1016/j.arthro.2008.02.005.
Friedman DJ, Parnes NZ, Zimmer Z, Higgins LD, Warner JJ. (2009) Prevalence of cerebrovascular events during shoulder surgery and association with patient position. Orthopedics 32:orthosupersite.com/view.asp?rID = 38058.
Benes J, Simanova A, Tovarnicka T, Sevcikova S, Kletecka J, Zatloukal J, Pradl R, Chytra I, Kasal E. Continuous non-invasive monitoring improves blood pressure stability in upright position: randomized controlled trial. J Clin Monit Comput. 2015;29:11–7. https://doi.org/10.1007/s10877-014-9586-2.
Fukada T, Tsuchiya Y, Iwakiri H, Ozaki M, Nomura M. Forehead Regional Oxygen Saturation (rSO2)-Related ear-level arterial pressure and lower thigh rSO2 in the Steep Trendelenburg position with CO2 pneumoperitoneum and the Beach Chair position. Cureus. 2021;13:e15687. https://doi.org/10.7759/cureus.15687.
Moss L, Shaw M, Piper I, Arvind DK, Hawthorne C. Automatic calculation of hydrostatic pressure gradient in patients with Head Injury: a pilot study. Acta Neurochir Suppl. 2016;122:263–6. https://doi.org/10.1007/978-3-319-22533-3_52.
Kalmar AF, Foubert L, Hendrickx JF, Mottrie A, Absalom A, Mortier EP, Struys MM. Influence of steep Trendelenburg position and CO(2) pneumoperitoneum on cardiovascular, cerebrovascular, and respiratory homeostasis during robotic prostatectomy. Br J Anaesth. 2010;104:433–9. https://doi.org/10.1093/bja/aeq018.
Ahuja S, Mascha EJ, Yang D, Maheshwari K, Cohen B, Khanna AK, Ruetzler K, Turan A, Sessler DI. Associations of Intraoperative Radial arterial systolic, Diastolic, Mean, and pulse pressures with myocardial and acute kidney Injury after noncardiac surgery: a retrospective cohort analysis. Anesthesiology. 2020;132:291–306. https://doi.org/10.1097/ALN.0000000000003048.
Nichols WW, Edwards DG. Arterial elastance and wave reflection augmentation of systolic blood pressure: deleterious effects and implications for therapy. J Cardiovasc Pharmacol Ther. 2001;6:5–21. https://doi.org/10.1177/107424840100600102.
Walsh M, Devereaux PJ, Garg AX, Kurz A, Turan A, Rodseth RN, Cywinski J, Thabane L, Sessler DI. Relationship between intraoperative mean arterial pressure and clinical outcomes after noncardiac surgery: toward an empirical definition of hypotension. Anesthesiology. 2013;119:507–15. https://doi.org/10.1097/ALN.0b013e3182a10e26.
Mascha EJ, Yang D, Weiss S, Sessler DI. Intraoperative Mean arterial pressure variability and 30-day mortality in patients having noncardiac surgery. Anesthesiology. 2015;123:79–91. https://doi.org/10.1097/ALN.0000000000000686.
Sun LY, Wijeysundera DN, Tait GA, Beattie WS. Association of intraoperative hypotension with acute kidney injury after elective noncardiac surgery. Anesthesiology. 2015;123:515–23. https://doi.org/10.1097/ALN.0000000000000765.
Monk TG, Bronsert MR, Henderson WG, Mangione MP, Sum-Ping ST, Bentt DR, Nguyen JD, Richman JS, Meguid RA, Hammermeister KE. Association between Intraoperative Hypotension and Hypertension and 30-day postoperative mortality in noncardiac surgery. Anesthesiology. 2015;123:307–19. https://doi.org/10.1097/ALN.0000000000000756.
van Waes JA, van Klei WA, Wijeysundera DN, van Wolfswinkel L, Lindsay TF, Beattie WS. Association between Intraoperative Hypotension and Myocardial Injury after vascular surgery. Anesthesiology. 2016;124:35–44. https://doi.org/10.1097/ALN.0000000000000922.
Salmasi V, Maheshwari K, Yang D, Mascha EJ, Singh A, Sessler DI, Kurz A. Relationship between intraoperative hypotension, defined by either reduction from baseline or Absolute Thresholds, and Acute kidney and myocardial Injury after noncardiac surgery: a retrospective cohort analysis. Anesthesiology. 2017;126:47–65. https://doi.org/10.1097/ALN.0000000000001432.
Stapelfeldt WH, Yuan H, Dryden JK, Strehl KE, Cywinski JB, Ehrenfeld JM, Bromley P. The SLUScore: a Novel Method for detecting hazardous hypotension in adult patients undergoing Noncardiac Surgical procedures. Anesth Analg. 2017;124:1135–52. https://doi.org/10.1213/ANE.0000000000001797.
Wesselink EM, Kappen TH, Torn HM, Slooter AJC, van Klei WA. Intraoperative hypotension and the risk of postoperative adverse outcomes: a systematic review. Br J Anaesth. 2018;121:706–21. https://doi.org/10.1016/j.bja.2018.04.036.
Mathis MR, Naik BI, Freundlich RE, Shanks AM, Heung M, Kim M, Burns ML, Colquhoun DA, Rangrass G, Janda A, Engoren MC, Saager L, Tremper KK, Kheterpal S, Aziz MF, Coffman T, Durieux ME, Levy WJ, Schonberger RB, Soto R, Wilczak J, Berman MF, Berris J, Biggs DA, Coles P, Craft RM, Cummings KC, Ellis TA 2nd, Fleishut PM, Helsten DL, Jameson LC, van Klei WA, Kooij F, LaGorio J, Lins S, Miller SA, Molina S, Nair B, Paganelli WC, Peterson W, Tom S, Wanderer JP, Wedeven C, Multicenter Perioperative Outcomes Group Investigators. Preoperative risk and the Association between Hypotension and postoperative acute kidney Injury. Anesthesiology. 2020;132:461–75. https://doi.org/10.1097/ALN.0000000000003063.
Gregory A, Stapelfeldt WH, Khanna AK, Smischney NJ, Boero IJ, Chen Q, Stevens M, Shaw AD. Intraoperative hypotension is Associated with adverse clinical outcomes after noncardiac surgery. Anesth Analg. 2020;132:1654–65. https://doi.org/10.1213/ANE.0000000000005250.
Wanner PM, Wulff DU, Djurdjevic M, Korte W, Schnider TW, Filipovic M. Targeting higher intraoperative blood pressures does not reduce adverse Cardiovascular events following noncardiac surgery. J Am Coll Cardiol. 2021;78:1753–64. https://doi.org/10.1016/j.jacc.2021.08.048.
Futier E, Lefrant JY, Guinot PG, Godet T, Lorne E, Cuvillon P, Bertran S, Leone M, Pastene B, Piriou V, Molliex S, Albanese J, Julia JM, Tavernier B, Imhoff E, Bazin JE, Constantin JM, Pereira B, Jaber S, INPRESS Study Group. Effect of individualized vs standard blood pressure management strategies on postoperative organ dysfunction among high-risk patients undergoing major surgery: a Randomized Clinical Trial. JAMA. 2017;318:1346–57. https://doi.org/10.1001/jama.2017.14172.
Marcucci M, Painter TW, Conen D, Lomivorotov V, Sessler DI, Chan MTV, Borges FK, Leslie K, Duceppe E, Martinez-Zapata MJ, Wang CY, Xavier D, Ofori SN, Wang MK, Efremov S, Landoni G, Kleinlugtenbelt YV, Szczeklik W, Schmartz D, Garg AX, Short TG, Wittmann M, Meyhoff CS, Amir M, Torres D, Patel A, Ruetzler K, Parlow JL, Tandon V, Fleischmann E, Polanczyk CA, Lamy A, Jayaram R, Astrakov SV, Wu WKK, Cheong CC, Ayad S, Kirov M, de Nadal M, Likhvantsev VV, Paniagua P, Aguado HJ, Maheshwari K, Whitlock RP, McGillion MH, Vincent J, Copland I, Balasubramanian K, Biccard BM, Srinathan S, Ismoilov S, Pettit S, Stillo D, Kurz A, Belley-Cote EP, Spence J, McIntyre WF, Bangdiwala SI, Guyatt G, Yusuf S, Devereaux PJ. POISE-3 Trial investigators and study groups (2023) hypotension-avoidance Versus hypertension-avoidance strategies in noncardiac surgery: an International Randomized Controlled Trial. Ann Intern Med 176:605–14. https://doi.org/10.7326/M22-3157.
Sessler DI, Bloomstone JA, Aronson S, Berry C, Gan TJ, Kellum JA, Plumb J, Mythen MG, Grocott MPW, Edwards MR, Miller TE, Perioperative Quality Initiative-3 workgroup, POQI chairs, Miller TE, Mythen MG, Grocott MP, Edwards MR. Physiology group, Preoperative blood pressure group, Intraoperative blood pressure group, Postoperative blood pressure group (2019) Perioperative Quality Initiative consensus statement on intraoperative blood pressure, risk and outcomes for elective surgery. Br J Anaesth 122:563–574. https://doi.org/10.1016/j.bja.2019.01.013.
Shimada T, Mascha EJ, Yang D, Bravo M, Rivas E, Ince I, Turan A, Sessler DI. Intra-operative hypertension and myocardial injury and/or mortality and acute kidney injury after noncardiac surgery: a retrospective cohort analysis. Eur J Anaesthesiol. 2022;39:315–23. https://doi.org/10.1097/eja.0000000000001656.
Saugel B, Vincent JL, Wagner JY. Personalized hemodynamic management. Curr Opin Crit Care. 2017;23:334–41. https://doi.org/10.1097/MCC.0000000000000422.
Kusumoto FM, Schoenfeld MH, Barrett C, Edgerton JR, Ellenbogen KA, Gold MR, Goldschlager NF, Hamilton RM, Joglar JA, Kim RJ, Lee R, Marine JE, McLeod CJ, Oken KR, Patton KK, Pellegrini CN, Selzman KA, Thompson A, Varosy PD. 2018 ACC/AHA/HRS Guideline on the evaluation and management of patients with Bradycardia and Cardiac Conduction Delay: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice guidelines and the Heart Rhythm Society. Circulation. 2019;140:e382–e482. https://doi.org/10.1161/CIR.0000000000000628.
Page RL, Joglar JA, Caldwell MA, Calkins H, Conti JB, Deal BJ, Estes NA 3rd, Field ME, Goldberger ZD, Hammill SC, Indik JH, Lindsay BD, Olshansky B, Russo AM, Shen WK, Tracy CM, Al-Khatib SM, Evidence Review Committee Chair. 2015 ACC/AHA/HRS Guideline for the management of adult patients with supraventricular tachycardia: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice guidelines and the Heart Rhythm Society. Circulation. 2016;133:e506–74. https://doi.org/10.1161/CIR.0000000000000311.
Kouz K, Hoppe P, Reese P, Burfeindt C, Flick M, Briesenick L, Nitzschke R, Pinnschmidt H, Saugel B. Relationship between intraoperative and preoperative ambulatory Nighttime Heart Rates: a secondary analysis of a prospective observational study. Anesth Analg. 2021;133:406–12. https://doi.org/10.1213/ANE.0000000000005625.
Sato M, Tanaka M, Umehara S, Nishikawa T. Baroreflex control of heart rate during and after propofol infusion in humans. Br J Anaesth. 2005;94:577–81. https://doi.org/10.1093/bja/aei092.
DeSouza G, Lewis MC, TerRiet MF. Severe bradycardia after remifentanil. Anesthesiology. 1997;87:1019–20. https://doi.org/10.1097/00000542-199710000-00061.
Poterman M, Scheeren TWL, van der Velde MI, Buisman PL, Allaert S, Struys M, Kalmar AF. Prophylactic atropine administration attenuates the negative haemodynamic effects of induction of anaesthesia with propofol and high-dose remifentanil: a randomised controlled trial. Eur J Anaesthesiol. 2017;34:695–701. https://doi.org/10.1097/EJA.0000000000000639.
Sear JW. Role of cardiac reflexes in the control of heart rate: what does the anesthesiologist need to know? Anesth Analg. 2012;114:491–3. https://doi.org/10.1213/ANE.0b013e318241be2a.
Doyle DJ, Mark PW. Reflex bradycardia during surgery. Can J Anaesth. 1990;37:219–22. https://doi.org/10.1007/BF03005473.
Kouz K, Brockmann L, Timmermann LM, Bergholz A, Flick M, Maheshwari K, Sessler DI, Krause L, Saugel B. Endotypes of intraoperative hypotension during major abdominal surgery: a retrospective machine learning analysis of an observational cohort study. Br J Anaesth. 2023;130:253–61. https://doi.org/10.1016/j.bja.2022.07.056.
Soar J, Bottiger BW, Carli P, Couper K, Deakin CD, Djarv T, Lott C, Olasveengen T, Paal P, Pellis T, Perkins GD, Sandroni C, Nolan JP. European Resuscitation Council guidelines 2021: adult advanced life support. Resuscitation. 2021;161:115–51. https://doi.org/10.1016/j.resuscitation.2021.02.010.
McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Bohm M, Burri H, Butler J, Celutkiene J, Chioncel O, Cleland JGF, Coats AJS, Crespo-Leiro MG, Farmakis D, Gilard M, Heymans S, Hoes AW, Jaarsma T, Jankowska EA, Lainscak M, Lam CSP, Lyon AR, McMurray JJV, Mebazaa A, Mindham R, Muneretto C, Francesco Piepoli M, Price S, Rosano GMC, Ruschitzka F, Kathrine Skibelund A, ESC Scientific Document Group. 2021 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J. 2021;42:3599–726. https://doi.org/10.1093/eurheartj/ehab368.
Brugada J, Katritsis DG, Arbelo E, Arribas F, Bax JJ, Blomstrom-Lundqvist C, Calkins H, Corrado D, Deftereos SG, Diller GP, Gomez-Doblas JJ, Gorenek B, Grace A, Ho SY, Kaski JC, Kuck KH, Lambiase PD, Sacher F, Sarquella-Brugada G, Suwalski P, Zaza A, ESC Scientific Document Group. 2019 ESC guidelines for the management of patients with supraventricular tachycardiaThe Task Force for the management of patients with supraventricular tachycardia of the European Society of Cardiology (ESC). Eur Heart J. 2020;41:655–720. https://doi.org/10.1093/eurheartj/ehz467.
Whitlock EL, Feiner JR, Chen LL. Perioperative Mortality, 2010 to 2014: a Retrospective Cohort Study using the National Anesthesia Clinical outcomes Registry. Anesthesiology. 2015;123:1312–21. https://doi.org/10.1097/aln.0000000000000882.
Talmor D, Kelly B. How to better identify patients at high risk of postoperative complications? Curr Opin Crit Care. 2017;23:417–23. https://doi.org/10.1097/mcc.0000000000000445.
Pearse RM, Harrison DA, James P, Watson D, Hinds C, Rhodes A, Grounds RM, Bennett ED. Identification and characterisation of the high-risk surgical population in the United Kingdom. Crit Care. 2006;10:R81. https://doi.org/10.1186/cc4928.
Vascular Events in Noncardiac Surgery Patients Cohort Evaluation Study Investigators, Spence J, LeManach Y, Chan MTV, Wang CY, Sigamani A, Xavier D, Pearse R, Alonso-Coello P, Garutti I, Srinathan SK, Duceppe E, Walsh M, Borges FK, Malaga G, Abraham V, Faruqui A, Berwanger O, Biccard BM, Villar JC, Sessler DI, Kurz A, Chow CK, Polanczyk CA, Szczeklik W, Ackland G, X GA, Jacka M, Guyatt GH, Sapsford RJ, Williams C, Cortes OL, Coriat P, Patel A, Tiboni M, Belley-Cote EP, Yang S, Heels-Ansdell D, McGillion M, Parlow S, Patel M, Pettit S, Yusuf S, Devereaux PJ. Association between complications and death within 30 days after noncardiac surgery. CMAJ. 2019;191:E830–7. https://doi.org/10.1503/cmaj.190221.
Chong MA, Wang Y, Berbenetz NM, McConachie I. Does goal-directed haemodynamic and fluid therapy improve peri-operative outcomes? A systematic review and meta-analysis. Eur J Anaesthesiol. 2018;35:469–83. https://doi.org/10.1097/EJA.0000000000000778.
Cecconi M, Corredor C, Arulkumaran N, Abuella G, Ball J, Grounds RM, Hamilton M, Rhodes A. Clinical review: goal-directed therapy-what is the evidence in surgical patients? The effect on different risk groups. Crit Care. 2013;17:209. https://doi.org/10.1186/cc11823.
Vincent JL, Rhodes A, Perel A, Martin GS, Della Rocca G, Vallet B, Pinsky MR, Hofer CK, Teboul JL, de Boode WP, Scolletta S, Vieillard-Baron A, De Backer D, Walley KR, Maggiorini M, Singer M. Clinical review: update on hemodynamic monitoring–a consensus of 16. Crit Care. 2011;15:229. https://doi.org/10.1186/cc10291.
Cioccari L, Luethi N, Glassford NJ, Bellomo R. The normal cardiac index in older healthy individuals: a scoping review. Crit Care Resusc. 2019;21:9–17.
Carlsson M, Andersson R, Bloch KM, Steding-Ehrenborg K, Mosen H, Stahlberg F, Ekmehag B, Arheden H. Cardiac output and cardiac index measured with cardiovascular magnetic resonance in healthy subjects, elite athletes and patients with congestive heart failure. J Cardiovasc Magn Reson. 2012;14:51. https://doi.org/10.1186/1532-429X-14-51.
Jessen MK, Vallentin MF, Holmberg MJ, Bolther M, Hansen FB, Holst JM, Magnussen A, Hansen NS, Johannsen CM, Enevoldsen J, Jensen TH, Roessler LL, Lind PC, Klitholm MP, Eggertsen MA, Caap P, Boye C, Dabrowski KM, Vormfenne L, Hoybye M, Henriksen J, Karlsson CM, Balleby IR, Rasmussen MS, Paelestik K, Granfeldt A, Andersen LW. Goal-directed haemodynamic therapy during general anaesthesia for noncardiac surgery: a systematic review and meta-analysis. Br J Anaesth. 2022;128:416–33. https://doi.org/10.1016/j.bja.2021.10.046.
Pearse RM, Harrison DA, MacDonald N, Gillies MA, Blunt M, Ackland G, Grocott MP, Ahern A, Griggs K, Scott R, Hinds C, Rowan K, OPTIMISE Study Group. Effect of a perioperative, cardiac output-guided hemodynamic therapy algorithm on outcomes following major gastrointestinal surgery: a randomized clinical trial and systematic review. JAMA. 2014;311:2181–90. https://doi.org/10.1001/jama.2014.5305.
Salzwedel C, Puig J, Carstens A, Bein B, Molnar Z, Kiss K, Hussain A, Belda J, Kirov MY, Sakka SG, Reuter DA. Perioperative goal-directed hemodynamic therapy based on radial arterial pulse pressure variation and continuous cardiac index trending reduces postoperative complications after major abdominal surgery: a multi-center, prospective, randomized study. Crit Care. 2013;17:R191. https://doi.org/10.1186/cc12885.
Nicklas JY, Diener O, Leistenschneider M, Sellhorn C, Schon G, Winkler M, Daum G, Schwedhelm E, Schroder J, Fisch M, Schmalfeldt B, Izbicki JR, Bauer M, Coldewey SM, Reuter DA, Saugel B. Personalised haemodynamic management targeting baseline cardiac index in high-risk patients undergoing major abdominal surgery: a randomised single-centre clinical trial. Br J Anaesth. 2020;125:122–32. https://doi.org/10.1016/j.bja.2020.04.094.
Parker T, Brealey D, Dyson A, Singer M. Optimising organ perfusion in the high-risk surgical and critical care patient: a narrative review. Br J Anaesth. 2019;123:170–6. https://doi.org/10.1016/j.bja.2019.03.027.
Edwards MR, Forbes G, MacDonald N, Berdunov V, Mihaylova B, Dias P, Thomson A, Grocott MP, Mythen MG, Gillies MA, Sander M, Phan TD, Evered L, Wijeysundera DN, McCluskey SA, Aldecoa C, Ripolles-Melchor J, Hofer CK, Abukhudair H, Szczeklik W, Grigoras I, Hajjar LA, Kahan BC, Pearse RM, OPTIMISE II investigators. Optimisation of Perioperative Cardiovascular Management to Improve Surgical Outcome II (OPTIMISE II) trial: study protocol for a multicentre international trial of cardiac output-guided fluid therapy with low-dose inotrope infusion compared with usual care in patients undergoing major elective gastrointestinal surgery. BMJ Open. 2019;9:e023455. https://doi.org/10.1136/bmjopen-2018-023455.
Arulkumaran N, Corredor C, Hamilton MA, Ball J, Grounds RM, Rhodes A, Cecconi M. Cardiac complications associated with goal-directed therapy in high-risk surgical patients: a meta-analysis. Br J Anaesth. 2014;112:648–59. https://doi.org/10.1093/bja/aet466.
Takala J, Meier-Hellmann A, Eddleston J, Hulstaert P, Sramek V. Effect of dopexamine on outcome after major abdominal surgery: a prospective, randomized, controlled multicenter study. European Multicenter Study Group on Dopexamine in Major Abdominal surgery. Crit Care Med. 2000;28:3417–23. https://doi.org/10.1097/00003246-200010000-00007.
Shoemaker WC, Appel PL, Kram HB, Waxman K, Lee TS. Prospective trial of supranormal values of survivors as therapeutic goals in high-risk surgical patients. Chest. 1988;94:1176–86. https://doi.org/10.1378/chest.94.6.1176.
Michard F. Changes in arterial pressure during mechanical ventilation. Anesthesiology. 2005;103:419–28. https://doi.org/10.1097/00000542-200508000-00026. quiz 449-5.
Marik PE, Cavallazzi R, Vasu T, Hirani A. Dynamic changes in arterial waveform derived variables and fluid responsiveness in mechanically ventilated patients: a systematic review of the literature. Crit Care Med. 2009;37:2642–7. https://doi.org/10.1097/CCM.0b013e3181a590da.
Perel A, Pizov R, Cotev S. Respiratory variations in the arterial pressure during mechanical ventilation reflect volume status and fluid responsiveness. Intensive Care Med. 2014;40:798–807. https://doi.org/10.1007/s00134-014-3285-9.
Perel A. Using dynamic variables to Guide Perioperative Fluid Management. Anesthesiology. 2020;133:929–35. https://doi.org/10.1097/ALN.0000000000003408.
Messina A, Pelaia C, Bruni A, Garofalo E, Bonicolini E, Longhini F, Dellara E, Saderi L, Romagnoli S, Sotgiu G, Cecconi M, Navalesi P. Fluid challenge during anesthesia: a systematic review and Meta-analysis. Anesth Analg. 2018;127:1353–64. https://doi.org/10.1213/ANE.0000000000003834.
Cannesson M, Le Manach Y, Hofer CK, Goarin JP, Lehot JJ, Vallet B, Tavernier B. Assessing the diagnostic accuracy of pulse pressure variations for the prediction of fluid responsiveness: a gray zone approach. Anesthesiology. 2011;115:231–41. https://doi.org/10.1097/ALN.0b013e318225b80a.
Michard F, Chemla D, Teboul JL. Applicability of pulse pressure variation: how many shades of grey? Crit Care. 2015;19:144. https://doi.org/10.1186/s13054-015-0869-x.
Fischer MO, Dechanet F, du Cheyron D, Gerard JL, Hanouz JL, Fellahi JL. Evaluation of the knowledge base of French intensivists and anaesthesiologists as concerns the interpretation of respiratory arterial pulse pressure variation. Anaesth Crit Care Pain Med. 2015;34:29–34. https://doi.org/10.1016/j.accpm.2014.06.001.
Biais M, de Courson H, Lanchon R, Pereira B, Bardonneau G, Griton M, Sesay M, Nouette-Gaulain K. Mini-fluid challenge of 100 ml of Crystalloid predicts Fluid responsiveness in the operating room. Anesthesiology. 2017;127:450–6. https://doi.org/10.1097/ALN.0000000000001753.
Vincent JL, Cecconi M, De Backer D. The fluid challenge. Crit Care. 2020;24:703. https://doi.org/10.1186/s13054-020-03443-y.
Cecconi M, Parsons AK, Rhodes A. What is a fluid challenge? Curr Opin Crit Care. 2011;17:290–5. https://doi.org/10.1097/MCC.0b013e32834699cd.
Aya HD, Rhodes A, Chis Ster I, Fletcher N, Grounds RM, Cecconi M. Hemodynamic effect of different doses of fluids for a fluid challenge: a quasi-randomized controlled study. Crit Care Med. 2017;45:e161–8. https://doi.org/10.1097/CCM.0000000000002067.
Messina A, Calabro L, Pugliese L, Lulja A, Sopuch A, Rosalba D, Morenghi E, Hernandez G, Monnet X, Cecconi M. Fluid challenge in critically ill patients receiving haemodynamic monitoring: a systematic review and comparison of two decades. Crit Care. 2022;26:186. https://doi.org/10.1186/s13054-022-04056-3.
Trauzeddel RF, Nordine M, Groesdonk HV, Michels G, Pfister R, Reuter DA, Scheeren TWL, Berger C, Treskatsch S. [Perioperative optimization using hemodynamically focused echocardiography in high-risk patients-A practice guide]. Anaesthesist. 2021;70:772–84. https://doi.org/10.1007/s00101-021-00934-7.
American Society of Anesthesiology, Society of Cardiovascular Anesthesiologists Task Force on Transesophageal Echocardiography. Practice guidelines for perioperative transesophageal echocardiography. An updated report by the American Society of Anesthesiologists and the Society of Cardiovascular Anesthesiologists Task Force on Transesophageal Echocardiography. Anesthesiology. 2010;112:1084–96. https://doi.org/10.1097/ALN.0b013e3181c51e90.
Steeds RP, Garbi M, Cardim N, Kasprzak JD, Sade E, Nihoyannopoulos P, Popescu BA, Stefanidis A, Cosyns B, Monaghan M, Aakhus S, Edvardsen T, Flachskampf F, Galiuto L, Athanassopoulos G, Lancellotti P, EACVI Scientific Documents Committee. EACVI appropriateness criteria for the use of transthoracic echocardiography in adults: a report of literature and current practice review. Eur Heart J Cardiovasc Imaging. 2017;18:2014–6. https://doi.org/10.1093/ehjci/jew333.
Halvorsen S, Mehilli J, Cassese S, Hall TS, Abdelhamid M, Barbato E, De Hert S, de Laval I, Geisler T, Hinterbuchner L, Ibanez B, Lenarczyk R, Mansmann UR, McGreavy P, Mueller C, Muneretto C, Niessner A, Potpara TS, Ristic A, Sade LE, Schirmer H, Schupke S, Sillesen H, Skulstad H, Torracca L, Tutarel O, Van Der Meer P, Wojakowski W, Zacharowski K, ESC Scientific Document Group. 2022 ESC guidelines on cardiovascular assessment and management of patients undergoing non-cardiac surgery. Eur Heart J. 2022;43:3826–924. https://doi.org/10.1093/eurheartj/ehac270.
Loxdale SJ, Sneyd JR, Donovan A, Werrett G, Viira DJ. The role of routine pre-operative bedside echocardiography in detecting aortic stenosis in patients with a hip fracture. Anaesthesia. 2012;67:51–4. https://doi.org/10.1111/j.1365-2044.2011.06942.x.
Sheth A, Dabo-Trubelja A. Perioperative focused cardiac ultrasound: a brief report. J Anesth Crit Care. 2021;13:55–60.
Chang HY, Chang WT, Liu YW. Application of transthoracic echocardiography in patients receiving intermediate- or high-risk noncardiac surgery. PLoS ONE. 2019;14:e0215854. https://doi.org/10.1371/journal.pone.0215854.
Canty DJ, Royse CF, Kilpatrick D, Williams DL, Royse AG. The impact of pre-operative focused transthoracic echocardiography in emergency non-cardiac surgery patients with known or risk of cardiac disease. Anaesthesia. 2012;67:714–20. https://doi.org/10.1111/j.1365-2044.2012.07118.x.
Heiberg J, El-Ansary D, Canty DJ, Royse AG, Royse CF. Focused echocardiography: a systematic review of diagnostic and clinical decision-making in anaesthesia and critical care. Anaesthesia. 2016;71:1091–100. https://doi.org/10.1111/anae.13525.
Greim CA, Göpfert M, Treskatsch S, Ender J, Kim SC, Weber SU. Zertifizierung für die sonographische Ausbildung in der Anästhesiologie und Intensivmedizin - Übersicht mit Einführung von SONO∘ZERT und PFE-Zertifikat. Anästh Intensivmed. 2022;63:112–5.
Greim CA, Weber S, Göpfert M, Groesdonk H, Treskatsch S, Wolf B, Zahn P, Müller M, Zenz S, Rauch H, Molitoris U, Ender J. Perioperative fokussierte Echokardiographie in der Anästhesiologie und Intensivmedizin. Anästh Intensivmed. 2017;58:616–48.
DGAI Verbandsmitteilung. TEE-Zertifizierung nach den Richtlinien der DGAI: Aktueller Stand und zukünftige Regelung. Anästh Intensivmed. 2008;49:97–104.
Milder DA, Liang SS, Ong SGK, Kam PCA. Association between intraoperative oliguria and postoperative acute kidney injury in non-cardiac surgical patients: a systematic review and meta-analysis. J Anesth. 2023;37:219–33. https://doi.org/10.1007/s00540-022-03150-8.
Pang Z, Liang S, Xing M, Zhou N, Guo Q, Zou W. The correlation of intraoperative oliguria with acute kidney injury after noncardiac surgery: a systematic review and meta-analysis. Int J Surg. 2023;109:449–57. https://doi.org/10.1097/JS9.0000000000000284.
Myles PS, McIlroy DR, Bellomo R, Wallace S. Importance of intraoperative oliguria during major abdominal surgery: findings of the restrictive versus liberal fluid therapy in major abdominal surgery trial. Br J Anaesth. 2019;122:726–33. https://doi.org/10.1016/j.bja.2019.01.010.
Egal M, Erler NS, de Geus HR, van Bommel J, Groeneveld AB. Targeting Oliguria reversal in goal-Directed hemodynamic management does not reduce renal dysfunction in Perioperative and critically ill patients: a systematic review and Meta-analysis. Anesth Analg. 2016;122:173–85. https://doi.org/10.1213/ANE.0000000000001027.
Egal M, de Geus HR, van Bommel J, Groeneveld AB. Targeting oliguria reversal in perioperative restrictive fluid management does not influence the occurrence of renal dysfunction: a systematic review and meta-analysis. Eur J Anaesthesiol. 2016;33:425–35. https://doi.org/10.1097/EJA.0000000000000416.
van der Zee EN, Egal M, Gommers D, Groeneveld AB. Targeting urine output and 30-day mortality in goal-directed therapy: a systematic review with meta-analysis and meta-regression. BMC Anesthesiol. 2017;17:22. https://doi.org/10.1186/s12871-017-0316-4.
Kraut JA, Madias NE. Lactic acidosis. N Engl J Med. 2014;371:2309–19. https://doi.org/10.1056/NEJMra1309483.
Vincent JL, De Backer D. Oxygen transport-the oxygen delivery controversy. Intensive Care Med. 2004. https://doi.org/10.1007/s00134-004-2384-4. 30:1990-6.
Smith ZR, Horng M, Rech MA. Medication-Induced Hyperlactatemia and Lactic Acidosis: a systematic review of the literature. Pharmacotherapy. 2019;39:946–63. https://doi.org/10.1002/phar.2316.
Andersen LW, Mackenhauer J, Roberts JC, Berg KM, Cocchi MN, Donnino MW. Etiology and therapeutic approach to elevated lactate levels. Mayo Clin Proc. 2013;88:1127–40. https://doi.org/10.1016/j.mayocp.2013.06.012.
Cecconi M, De Backer D, Antonelli M, Beale R, Bakker J, Hofer C, Jaeschke R, Mebazaa A, Pinsky MR, Teboul JL, Vincent JL, Rhodes A. Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2014;40:1795–815. https://doi.org/10.1007/s00134-014-3525-z.
Shepherd SJ, Pearse RM. Role of central and mixed venous oxygen saturation measurement in perioperative care. Anesthesiology. 2009. https://doi.org/10.1097/ALN.0b013e3181af59aa. 111:649 – 56.
Walley KR. Use of central venous oxygen saturation to guide therapy. Am J Respir Crit Care Med. 2011. https://doi.org/10.1164/rccm.201010-1584CI. 184:514 – 20.
van Beest P, Wietasch G, Scheeren T, Spronk P, Kuiper M. Clinical review: use of venous oxygen saturations as a goal - a yet unfinished puzzle. Crit Care. 2011;15:232. https://doi.org/10.1186/cc10351.
Ho KM, Harding R, Chamberlain J. The impact of arterial oxygen tension on venous oxygen saturation in circulatory failure. Shock. 2008;29:3–6. https://doi.org/10.1097/SHK.0b013e318067dec1.
Collaborative Study Group on Perioperative ScvO2 Monitoring. Multicentre study on peri- and postoperative central venous oxygen saturation in high-risk surgical patients. Crit Care. 2006;10:R158. https://doi.org/10.1186/cc5094.
Futier E, Robin E, Jabaudon M, Guerin R, Petit A, Bazin JE, Constantin JM, Vallet B. Central venous O2 saturation and venous-to-arterial CO2 difference as complementary tools for goal-directed therapy during high-risk surgery. Crit Care. 2010;14:R193. https://doi.org/10.1186/cc9310.
Jammer I, Ulvik A, Erichsen C, Lødemel O, Ostgaard G. Does central venous oxygen saturation-directed fluid therapy affect postoperative morbidity after colorectal surgery? A randomized assessor-blinded controlled trial. Anesthesiology. 2010;113:1072–80. https://doi.org/10.1097/ALN.0b013e3181f79337.
Popel AS, Johnson PC. Microcirculation and Hemorheology. Annu Rev Fluid Mech. 2005;37:43–69. https://doi.org/10.1146/annurev.fluid.37.042604.133933.
Ince C, Boerma EC, Cecconi M, De Backer D, Shapiro NI, Duranteau J, Pinsky MR, Artigas A, Teboul JL, Reiss IKM, Aldecoa C, Hutchings SD, Donati A, Maggiorini M, Taccone FS, Hernandez G, Payen D, Tibboel D, Martin DS, Zarbock A, Monnet X, Dubin A, Bakker J, Vincent JL, Scheeren TWL. Second consensus on the assessment of sublingual microcirculation in critically ill patients: results from a task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2018;44:281–99. https://doi.org/10.1007/s00134-018-5070-7. Cardiovascular Dynamics Section of the ESICM.
den Uil CA, Bezemer R, Miranda DR, Ince C, Lagrand WK, Hartman M, Bogers AJ, Spronk PE, Simoons ML. Intra-operative assessment of human pulmonary alveoli in vivo using Sidestream Dark Field imaging: a feasibility study. Med Sci Monit. 2009;15:MT137–141.
Nilsson J, Eriksson S, Blind PJ, Rissler P, Sturesson C. Microcirculation changes during liver resection–a clinical study. Microvasc Res. 2014;94:47–51. https://doi.org/10.1016/j.mvr.2014.05.002.
Bansch P, Flisberg P, Bentzer P. Changes in the sublingual microcirculation during major abdominal surgery and post-operative morbidity. Acta Anaesthesiol Scand. 2014;58:89–97. https://doi.org/10.1111/aas.12204.
Stens J, de Wolf SP, van der Zwan RJ, Koning NJ, Dekker NA, Hering JP, Boer C. Microcirculatory perfusion during different perioperative hemodynamic strategies. Microcirculation. 2015;22:267–75. https://doi.org/10.1111/micc.12194.
Flick M, Briesenick L, Peine S, Scheeren TWL, Duranteau J, Saugel B. The effect of moderate intraoperative blood loss and norepinephrine therapy on sublingual microcirculatory perfusion in patients having open radical prostatectomy: an observational study. Eur J Anaesthesiol. 2021;38:459–67. https://doi.org/10.1097/EJA.0000000000001434.
Flick M, Schreiber TH, Montomoli J, Krause L, de Boer HD, Kouz K, Scheeren TWL, Ince C, Hilty MP, Saugel B. Microcirculatory tissue perfusion during general anaesthesia and noncardiac surgery: an observational study using incident dark field imaging with automated video analysis. Eur J Anaesthesiol. 2022;39:582–90. https://doi.org/10.1097/EJA.0000000000001699.
Astapenko D, Benes J, Pouska J, Lehmann C, Islam S, Cerny V. Endothelial glycocalyx in acute care surgery - what anaesthesiologists need to know for clinical practice. BMC Anesthesiol. 2019;19:238. https://doi.org/10.1186/s12871-019-0896-2.
Bickler PE, Feiner JR, Rollins MD. Factors affecting the performance of 5 cerebral oximeters during hypoxia in healthy volunteers. Anesth Analg. 2013;117:813–23. https://doi.org/10.1213/ANE.0b013e318297d763.
Henson LC, Calalang C, Temp JA, Ward DS. Accuracy of a cerebral oximeter in healthy volunteers under conditions of isocapnic hypoxia. Anesthesiology. 1998;88:58–65. https://doi.org/10.1097/00000542-199801000-00011.
Heringlake M, Garbers C, Käbler JH, Anderson I, Heinze H, Schön J, Berger KU, Dibbelt L, Sievers HH, Hanke T. Preoperative cerebral oxygen saturation and clinical outcomes in cardiac surgery. Anesthesiology. 2011;114:58–69. https://doi.org/10.1097/ALN.0b013e3181fef34e.
Nielsen HB. Systematic review of near-infrared spectroscopy determined cerebral oxygenation during non-cardiac surgery. Front Physiol. 2014;5:93. https://doi.org/10.3389/fphys.2014.00093.
Acknowledgements
None.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
Coordination of the guideline project: BS, DAR, MS, BJ. Drafting of the manuscript: all authors. Critical revision of the article for important intellectual content: all authors. Final approval of the version to be published: all authors. Agreement to be accountable for all aspects of the work thereby ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved: all authors.
Corresponding author
Ethics declarations
Competing interests
The experts declared their conflicts of interest in writing using a form sheet (https://register.awmf.org/de/leitlinien/detail/001-049). BS is a consultant for Edwards Lifesciences, Philips North America, Baxter, GE Healthcare, Maquet Critical Care, Pulsion Medical Systems, Vygon, and Retia Medical. BS has received honoraria for giving lectures from Edwards Lifesciences, Philips Medizin Systeme, Baxter, GE Healthcare, CNSystems Medizintechnik, Getinge, Pulsion Medical Systems, Vygon. BS was a consultant for Tensys Medical. BS has received institutional restricted research grants from Edwards Lifesciences, Baxter, GE Healthcare, CNSystems Medizintechnik, Pulsion Medical Systems, Retia Medical, Osypka Medical, and Tensys Medical. BS is an Editor of the British Journal of Anaesthesia. TA has received honoraria for giving lectures from FomF Deutschland, Anästhesiologie Forschung und Fortbildung e.V., and CSL Behring. TA has received institutional restricted research grants from Corpuls and Cytosorbents. BB is a consultant for Edwards Lifesciences, Gruenenthal, CSL Behring, and Pharmacosmos. MF has received honoraria for giving lectures from Edwards Lifesciences and CNSystems Medizintechnik. MF has received institutional restricted research grants from CNSystems Medizintechnik. MGo is a consultant for Ebnet Medical. MGr is a consultant for GE Healthcare and Johnson & Johnson. MGr has received honoraria for giving lectures from Edwards Lifesciences, Medtronic, and Gruenenthal. MH is a consultant for Edwards Lifesciences and Baxter. BJ has received institutional restricted research grants from Löwenstein Medical Innovation. TK is a consultant for ratiopharm GmbH. KK is a consultant for Edwards Lifesciences and Vygon. KK has received honoraria for giving lectures from Edwards Lifesciences. ASM has received honoraria for giving lectures from Edwards Lifesciences. GP has received institutional restricted research grants from Nihon-Kohden, Edwards Lifesciences, CNSystems Medizintechnik, Masimo, and Deltex Medical. JR has received honoraria for giving lectures from Edwards Lifesciences. SGS is a consultant for Maquet and Getinge. MS is a consultant for Edwards Lifesciences. MS has received honoraria for giving lectures from Edwards Lifesciences, BTG, and Orion Pharma. MS has received institutional restricted research grants from Edwards Lifesciences and Medtronic. ST has received honoraria for giving lectures from Edwards Lifesciences, Orion Pharma, Philips, Amomed, and Cytosorbents. ST has received institutional restricted research grants from Edwards Lifesciences, Orion Pharma, and Cytosorbents. AZ has received honoraria for giving lectures from Edwards Lifesciences. DAR is a consultant for Edwards Lifesciences. DAR has received honoraria for giving lectures from Löwenstein Medical Innovation. DAR has received institutional restricted research grants from Edwards Lifesciences, Getinge, Sentec, Artcline, and Cytosorbents.
Appendices
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Saugel, B., Annecke, T., Bein, B. et al. Intraoperative haemodynamic monitoring and management of adults having non-cardiac surgery: Guidelines of the German Society of Anaesthesiology and Intensive Care Medicine in collaboration with the German Association of the Scientific Medical Societies. J Clin Monit Comput (2024). https://doi.org/10.1007/s10877-024-01132-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10877-024-01132-7