Abstract
Actin is a major component of all sarcomere and cytoskeletal systems and thus plays a crucial role in muscle and cellular movement. The molecular dynamics of actomyosin complexes are obtaining the central machinery of force generation during contraction. Nalidixic acid, discovered in 1962—the primary compound of the quinolone, fluoroquinolone-type antibiotics—in the forms of its improved derivatives are still widely used today. One of the most commonly used drugs in this class is ciprofloxacin (CF). It was observed that after the use of these drugs—during longer or shorter administration or even after their use—several side effects such as muscle pain (myalgia), muscle spasms, and muscle atrophy also occur, and muscle damage and rupture may be associated with muscle cell death (rhabdomyolysis). In medical cases, muscle weakness after CF treatment can be related to the CF-actin interaction. Here, we studied the effect of CF on skeletal muscle actin using differential scanning calorimetry (DSC). We obtained evident data that CF binding increases the structural stability of G actin and slightly weakens the F actin filaments. The stability change of actin in myofilaments can lead to the adverse effect of CF.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
With the discovery of penicillin (Fleming 1928), the golden age of antibiotics began, and today, antibiotics are the most commonly prescribed drugs worldwide. Nalidixic acid, discovered in 1962—the primary compound of the quinolone, fluoroquinolone-type antibiotics—in the forms of its improved derivatives are still widely used today [1, 2]. These antibiotics, available in the European Union, help treat millions of patients with a variety of bacterial infections, including serious or life-threatening ones and bacterial infections resistant to other treatments. In 1972, the medical literature first reported on the adverse locomotor effects of nalidixic acid, the ancestor of quinolone acid [3], and eleven years later on the rheumatic diseases following the use of fluoroquinolones (norfloxacin) [4]. The most commonly used drugs in this class are ciprofloxacin (CF), levofloxacin, moxifloxacin, norfloxacin, and ofloxacin [5]. It has become apparent that after the use of these drugs—during longer or shorter administration or even after their use—several side effects may develop [6]. In 1996, the US FDA (Food and Drug Administration) issued an alert in a medical communication and ordered changes to fluoroquinolone instructions. It called for the risk of several side effects, e.g., severe musculoskeletal side effects such as tendonitis, rupture of tendons, muscle pain, muscle weakness, joint pain, and gait disturbance. Serious peripheral and central nervous system side effects include peripheral neuropathy and impairment of vision, hearing, smell, and taste. The risk of aortic aneurysm and dissection has recently been added to the information document [7]. It subsequently reviewed the safety of quinolone and fluoroquinolone antibiotics to assess the potential for severe, long-lasting adverse reactions that mainly affect the musculoskeletal system (tendinopathy, rupture) and the nervous system [8, 9]. Muscle pain (myalgia), muscle spasms, and muscle atrophy also occur, and muscle damage and rupture may be associated with muscle cell death (rhabdomyolysis), accompanied by an increase in creatine phosphokinase (CPK) [10,11,12]. However, even though very effective and widely used anti-inflammatory agents, such as ciprofloxacin, are excellent in practice and monitored for patient safety and benefit-risk profile, there is no data in the literature on whether there is an in vitro model of detectable lesions underlying tendon and muscle pain.
Due to its properties, the CF has biological applications as an adjuvant in cancer treatment. CF provides topoisomerase II inhibition [13, 14]. Only a few studies were focused on in vitro molecular and cellular cytoplasmic effects of CF [15, 16]. Surprisingly, CF derivatives can inhibit proliferation and induce apoptosis of HeLa cells; likely, it has an anti-metastatic impact [17]. Actin and actin-based microfilaments are essential parts of the cytoskeleton. They play a crucial role in muscle contraction and motility of eukaryotic cells [18,19,20,21,22,23,24]. The intracellular state of actin is mainly the filamentous form (F actin), but for their dynamics, they need to keep on a cytoplasmic monomer (G actin) pool. The structure of G actin contains a nucleotide-binding cleft, which is localized between the two main domains and can bind ATP in a complex with divalent cations [25]. The G actin hydrolyses ATP during the polymerization [26,27,28,29,30,31,32,33]. Previously, we have studied the effect of various types of toxins on skeletal muscle actin by DSC [34,35,36]. It was shown that the thermal stability of the microfilament system was changed by the cytotoxic drug treatment [36,37,38,39,40,41]. However, the CF can deplete and disassemble F actin and stress fibers and enhance their phalloidin binding [14]. Using all the above data, we have planned the recent investigation to look for a possible effect of CF on the thermal stability of G actin and its polymerization properties, supposing that antibiotic binding can cause similar effects in their structure as cytotoxic agent binding.
Results
Figure 1 shows the data of the intrinsic tryptophan emission change by the polymerization of G actin in the absence or presence of CF. The main goal of the measurements was to see the effect of CF treatment on the polymerization rate of G actin (see Table 1).
Changes of intrinsic tryptophan emission of actin in the presence or absence of CF A. The emission was dropped by adding polymerizing salt (1 mM MgCl2, 100 mM KCl) and then decreased slowly in the absence of CF (black line). The emission of tryptophan was remarkably reduced in the presence of CF (red line), but B followed a similar rate in the absence of CF. Data are the average of three independent measurements; the error bars indicate mean ± SD
Curves were fitted with double exponential to calculate the polymerization speed (kpol) by the time-dependent emission change. The intensity was dropped suddenly by the addition of polymerizing salt and then decreased slowly in the absence of CF (Fig. 1A). The resulting first-rate component of the exponential fitting resulted in 2.1 ± 0.01 nM s−1 which is two times higher, and the second component 0.18 ± 0.015 nM s−1 is five times less than the literature data 0.9 nM s−1 of actin polymerizing speed (measured by pyrene-actin polymerization assay [42]). However, in the presence of CF, the tryptophan emission was remarkably reduced (Fig. 1A) and jumped by 10% immediately after the salt addition. The part of slow change followed the first quick decay of emission. The exponential fitting showed that in the presence of CF, the first component was 4.1 ± 0.021 nM s−1, two times higher than in the absence of CF. The second component was 0.17 ± 0.013 nM s−1, identical to the rate in the absence of CF (Fig. 1B).
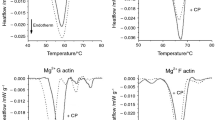
Figure 2 shows the thermal denaturation curves of G and F actin in the presence or absence of CF.
DSC studies of G and F actin in the presence or absence of CF. A Thermal denaturation of G actin in the absence (black line) and in the presence of CF (red line). B Thermal denaturation of F actin in the absence (blue line) and in the presence of CF (magenta line) (endotherm effect is deflected downwards)
Generally, in the case of thermal denaturation of actin, the change of melting temperature (Tm) by 1 °C is a remarkable difference [38]. In DSC studies, G and F actin were applied in a 2 mg mL−1 concentration. The average melting temperature of G actin was at 58.45 °C, then increased to 63.75 °C, and the enthalpy was decreased by 37% by the CF addition (Table 2.). However, in the case of F actin, Tm is shifted from 66.62 °C to 65.3 °C, and the enthalpy was increased by 15% by the CF addition (Table 2.).
Discussion
CF is a frequently applied chemical with antibiotic and cytotoxic effects [13]; however, the side effects of the treatment can be tendon and muscle weakness [10,11,12]. Besides that, F actin and stress fibers can be disassembled by the intracellular and cytoskeletal effects of CF, but they need more investigation [15, 16]. As we observed, the melting temperatures of G and F actin, in the absence of CF, are identical to the former results [43,44,45,46,47]. After CF treatment, the melting temperature of actin monomers was increased. We can interpret that the G actin underwent a structural change and became more rigid by the CF binding. The reduced calorimetric enthalpy in the presence of CF can be assumed as the CF binding decreasing the structural dynamics of the actin monomers. Besides that, the CF binding can stabilize the actin monomers and destabilize the filamentous form, which seems to be justified by the decreased melting temperature of F actin. Under the polymerizing salt conditions (1 mM MgCl2, 100 mM KCl), CF binding decreased the emission of intrinsic tryptophan and, followed by an increased polymerization rate, formed less stable actin filaments. However, the flattening of monomer structure can be lacking by CF binding, causing a subtle conformational relaxation of the filaments [48], and/or the more fragmented structure of filaments obtained an increased twisting feature [49]. The releasing monomers possibly caused the calorimetric enthalpy increment at the end of filaments and by their increased structural entropy due to intermonomer rearrangement. However, it is suggested that the CF treatment changed the monomer: filament ratio.
Conclusions
Actin is a major component of all sarcomere and cytoskeletal systems and thus plays a crucial role in muscle and cellular movement. The molecular dynamics of actomyosin complexes are obtaining the main machinery of force generation during contraction. Refers to medical cases of muscle weakness after CF treatment, we can state that its binding increases the structural stability of actin in myofilaments, which can lead to adverse effects of CF. These observations propose the potential that the application of CF will need more consideration in the medical routine.
Materials and methods
Actin preparation from rabbit skeletal muscle
Actin was prepared from acetone powder of rabbit skeletal muscle as described earlier by Spudich and Watt [50] and stored in MOPS buffer (2 mM MOPS, 0.2 mM ATP, 0.1 mM CaCl2, 0.1 mM β-mercaptoethanol, pH 7.4). Actin concentration was determined from the absorption spectra (Jasco V-550 spectrophotometer) (as the average concentration by ε = 1.11 mL mg−1 cm−1 at 280 nm and ε = 0.63 mL mg−1 cm−1 at 290 nm). We applied 2 mM EGTA and then 1 mM MgCl2 treatment to exchange calcium for magnesium, and polymerization was initialized with 100 mM KCl addition on 2 mg mL−1 actin.
Ciprofloxacin treatment
For in vitro measurements, the applied dosage of ciprofloxacin (CF) is comparable to the human dosage (400 mg) administered in vivo. in patients [9]. CF can reach all kinds of cells by the circulation system; thus, we need to count also with the rest of the body. The average blood volume is 5.5 L, resulting in a 72.72 μg mL−1 CF concentration. We carried out DSC scans of 2 mg mL−1 actin in the presence of 72.72 μg mL−1 CF, which resulted in the actin to CF ratio having to be \(\frac{27.5}{1}\) as a single dose.
DSC measurements
The samples were freshly prepared before all measurements. The analysis was made by a SETARAM Micro-DSC III (Caluire, France) calorimeter between 0 and 100 °C with a heating rate of 0.3 Kmin−1. Conventional Hastelloy batch vessels (Vmax = 1 mL) were used for the experiment to investigate denaturation with 950 µL sample volume (sample + buffer) on average. Sample masses were between 920 and 970 mg. MOPS buffer was used as a reference. The reference and sample vessels were equilibrated with a precision of ± 0.1 mg; this way, it was not necessary to do any correction between the vessels’ heat capacity. With the help of a two-point SETARAM peak integration setting, calorimetric enthalpy was calculated from the area under the heat absorption curve. Then, the results [denaturation or melting temperature (Tm) and calorimetric enthalpy (ΔHcal) data of samples] were compared.
Time-dependent intrinsic tryptophan fluorescence measurement
The rabbit skeletal muscle actin contains four tryptophans (W79, W86, W340, W356). These tryptophans were available for application as intrinsic probes for the fluorescence emission coupled polymerization time rate measurements. The structural change of chains neighboring tryptophan can affect their time-dependent emission intensity [51]. Fluorescence spectra were measured with a Perkin Elmer LS 50B fluorimeter (λex = 292 nm, λem = 338 nm). The emission was measured before and after adding polymerizing salt (1 mM MgCl2, 100 mM KCl) in the absence or presence of CF. Curves were fitted with double exponential to obtain the emission decay's time constants (kpol). The first component belongs to the lag phase, followed by the second to the elongation phase of polymerization.
Data availability
There is no additional data available to upload.
References
Andriole VT. The quinolones: past, present, and future. Clin Infect Dis. 2005;41(2):S113–9.
Fábrega A, Madurga S, Giralt E, Vila J. Mechanism of action of and resistance to quinolones. Microb Biotechnol. 2009;2(1):40–61.
Bailey RR, Natale R, Linton AL. Nalidixic acid arthralgia. Can Med Assoc J. 1972;107(7):604.
Bailey RR, Kirk JA, Peddie BA. Norfloxacin-induced rheumatic disease. N Z Med J. 1983;96(736):590.
Ciprofloxacin LM. Chemistry, mechanism of action, resistance, antimicrobial spectrum, pharmacokinetics, clinical trials, and adverse reactions. Pharmacotherapy. 1988;8(1):3–33.
Pham Thu DM, Ziora ZM, Blaskovich Mark AT. Quinolone antibiotics. Med Chem Commun. 2019;10(10):1719–39.
Baggio D, Ananda-Rajah MR. Fluoroquinolone antibiotics and adverse events. Aust Prescr. 2021;44(5):161–4.
Singh S, Nautiyal A. Aortic dissection and aortic aneurysms associated with fluoroquinolones: a systematic review and meta-analysis. Am J Med. 2017;30(12):1449–57.
European Medicines Agency: https://www.ema.europa.eu/en/medicines/human/referrals/ciprofloxacin-bayer
Tsai WC, Yang YM. Fluoroquinolone-associated tendinopathy. Rev Chang Gung Med J. 2011;34(5):461–7.
Michalak K, Sobolewska-Wtodarczyk A, Wtodarczyk M, Sobolewska J, Wozniak P, Sobolewski B. Treatment of the fluoroquinolone-associated disability: the pathobiochemical implications. Oxid Med Cell Longev. 2017;2017:8023935.
Alves C, Mendes D, Marques FB. Fluoroquinolones and the risk of tendon injury: a systematic review and meta-analysis. Eur J Clinical Pharmacol. 2019;75:1431–43.
Alhaj-Suliman SO, Naguib YW, Wafa EI, Saha S, Ebeid K, Meng X, Mohammed HH, Abuo-Rahma GEA, Yang S, Salem AK. A ciprofloxacin derivative with four mechanisms of action overcomes paclitaxel resistance in p53-mutant and MDR1 gene-expressing type II human endometrial cancer. Biomaterials. 2023;296:122093.
Kloskowski T, Gurtowska N, Olkowska J, Nowak JM, Adamowicz J, Tworkiewicz J, Dębski R, Grzanka A, Drewa T. Ciprofloxacin is a potential topoisomerase II inhibitor for the treatment of NSCLC. Int J of Oncology. 2012;41(6):1943–9.
Knoll KE, Lindeque Z, Adeniji AA, Oosthuizen CB, Lall N, Loots DT. We are elucidating the antimycobacterial mechanism of action of ciprofloxacin using metabolomics. Microorganisms. 2021;9(6):1158.
Marquez B, Caceres NE, Mingeot-Leclercq MP, Tulkens PM, Van Bambeke F. Identification of the efflux transporter of the fluoroquinolone antibiotic ciprofloxacin in murine macrophages: studies with ciprofloxacin-resistant cells. Antim A Chemother. 2009;53(6):2410–6.
Fathy M, Sun S, Zhao QL, Abdel-Aziz M, Abuo-Rahma GEA, Awale S, Nikaido T. A new ciprofloxacin-derivative inhibits proliferation and suppresses the migration ability of HeLa cells. Anticancer Res. 2020;40(9):5025–33.
Cossart P. Actin-based bacterial motility. Curr Opin Cell Biol. 1996;7:94–101.
Steinmetz MO, Stoffler D, Hoenger A, Bremer A, Aebi U. Actin: from cell biology to atomic detail. J Struct Biol. 1997;119:295–320.
Pollard TD, Blanchoin L, Mullins RD. Molecular mechanisms controlling actin filament dynamics in nonmuscle cells. Ann Rev Biophys Biomol Struc. 2000;29:545–76.
Pollard TD, Borisy GG. Cellular motility driven assembly and disassembly of actin filaments. Cell. 2003;112:453–65.
Carlier M-F, Le Clainche C, Wiesner S, Pantolini D. Actin-based motility: from molecules to movement. BioEssays. 2003;25:336–45.
Pantolini D, Le Clainche C, Carlier M-F. Mechanism of actin-based motility. Science. 2001;292:1502–6.
Hehnly H, Stamnes M. Regulating cytoskeleton-based vesicle motility. FEBS Lett. 2007;581:2112–8.
Sheterline P, Clayton J, Sparrow J. Actin. Protein Profile. 1995;2:1–103.
Feuer G, Molnár F, Pettko E, Straub FB. Studies on the composition and polymerisation of actin. Hung Acta Physiol. 1948;1(4–5):150–63.
Pollard TD. Rate constants for the reactions of ATP- and ADP-actin with the ends of actin filaments. J Cell Biol. 1986;103:2747–54.
Carlier M-F, Pantolini D. Direct evidence for ADP-Pi-F-actin as the major intermediate in ATP-actin polymerisation rate of dissociation of Pi from actin filaments. Biochemistry. 1986;25:7789–92.
Korn ED, Carlier MF, Pantaloni D. Actin polymerization and ATP hydrolysis. Science. 1987;238(4827):638–44.
Carlier M-F. Role of nucleotide hydrolysis in the polymerisation of actin and tubulin. Cell Biophys. 1988;12:105–17.
Carlier M-F, Pantolini D. Binding of phosphate to F-ADP-actin and role of F-ADP-P(i)-actin in ATP-actin polymerisation. J Biol Chem. 1988;263:817–25.
Janmey PA, Hvidt S, Oster GF, Lamb J, Stossel TP, Hartwig JH. Effect of ATP on actin filament stiffness. Nature. 1990;347:95–9.
Pollard TD, Goldberg I, Schwarz WH. Nucleotide exchange, structure, and mechanical properties of filaments assembled from ATP-actin and ADP-actin. J Biol Chem. 1992;267:20339–45.
Visegrády B, Lőrinczy D, Hild G, Somogyi B, Nyitrai M. The effect of phalloidin and jasplakinolide on actin filaments’ flexibility and thermal stability. FEBS Lett. 2004;565:163–6.
Visegrády B, Lőrinczy D, Hild G, Somogyi B, Nyitrai M. A simple model for the cooperative stabilisation of actin filaments by phalloidin and jasplakinolide. FEBS Lett. 2005;579:6–10.
Farkas P, Szatmári D, Könczöl F, et al. Cyclophosphamide treatment evoked side effect on skeletal muscle actin, monitored by DSC. J Therm Anal Calorim. 2022;147:3609–14.
Farkas P, Könczöl F, Lőrinczy D. Examination of the peripheral nerve and muscle damage in cyclophosphamide monotherapy with DSC in animal models. J Thermal Anal Calorim. 2016;126:47–53.
Lőrinczy D. Investigation of side effects in polyneuropathy on skeletal muscle by DSC caused by cyclophosphamide treatment. Eur Biophys J. 2019;48(1):S238.
Lőrinczy D. Cyclophosphamide treatment evoked side effects on skeletal muscle monitored by DSC. J Therm Anal Calorim. 2020;142:1897–901.
Szatmári D, Lőrinczy D. Alterations of inter-domain flexibility in actin monomers during cyclophosphamide treatment. J Therm Anal Calorim. 2022;147:7799–810.
Szatmári D, Bugyi B, Pintér R, et al. Cyclophosphamide treatment modifies the thermal stability of profilin bound monomeric and leiomodin2 bound filamentous actin. J Therm Anal Calorim. 2023;148:837–44.
Mu A, Fung TS, Francomacaro LM, Huynh T, Kotila T, Svindrych Z, Higgs HN. Regulation of INF2-mediated actin polymerization through site-specific lysine acetylation of actin itself. Proc Natl Acad Sci. 2020;117(1):439–47. https://doi.org/10.1073/pnas.1914072117.
Tatunashvili LV, Privalov PL. Calorimetric investigation of G-actin denaturation. Biofizika. 1984;29:583–5.
Bertazzon A, Tian GH, Lamblin A, Tsong TY. Enthalpic and entropic contributions to actin stability: calorimetry, circular dichroism, and fluorescence study and effects of calcium. Biochemistry. 1990;29:291–8.
Le Bihan T, Gicquaud C. Kinetic study of the thermal denaturation of G actin using differential scanning calorimetry and intrinsic fluorescence spectroscopy. BBRC. 1993;194:1065–73.
Lőrinczy D, Belágyi J. Scanning calorimetric and EPR studies on thermal stability of actin. Thermochim Acta. 1995;259:153–64.
Levitsky DI. Structural and functional studies of muscle proteins by using differential scanning calorimetry. In: Lörinczy Dénes, editor. The nature of biological systems as revealed by thermal methods. Dordrecht: Kluwer Academic Publishers; 2005. p. 127–58. https://doi.org/10.1007/1-4020-2219-0_6.
Chou SZ, Pollard TD. Mechanism of actin polymerization revealed by cryo-EM structures of actin filaments with three different bound nucleotides. Proc Natl Acad Sci U S A. 2019;116(10):4265–74.
Bibeau JP, Pandit NG, Gray S, ShateryNejad N, Sindelar CV, Cao W, De La Cruz EM. Twist response of actin filaments. Proc Natl Acad Sci U S A. 2023;120(4):e2208536120.
Spudich JA, Watt S. The regulation of rabbit skeletal muscle contraction: I. Biochemical studies of the interaction of the tropomyosin-troponin complex with actin and the proteolytic fragments of myosin. J Biol Chem. 1971;246(15):4866–71.
Szatmári D, Bugyi B, Ujfalusi Z, Grama L, Dudás R, Nyitrai M. Cardiac leiomodin2 binds to the sides of actin filaments and regulates the ATPase activity of myosin. PLoS ONE. 2017;12(10):e0186288.
Funding
Open access funding provided by University of Pécs. This research was funded by a CO-272 (OTKA) grant (D.L.).
Author information
Authors and Affiliations
Contributions
Conceptualization contributed by FK, DL; methodology contributed by PF, DS; software contributed by DS; validation contributed by DL, DS; formal analysis contributed by DS; investigation contributed by FK, DL; resources contributed by DL; data curation contributed by DS; writing—original draft preparation contributed by DS, DL; writing—review and editing contributed by DL; visualization contributed by DS; supervision contributed by FK, DL; project administration contributed by DS; funding acquisition contributed by DL. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflicts of interest.
Informed consent statement
Not applicable.
Institutional review board statement
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Szatmári, D., Farkas, P., Könczöl, F. et al. The adverse effect of ciprofloxacin treatment can be related to the increased thermal stability of monomeric actin. J Therm Anal Calorim (2024). https://doi.org/10.1007/s10973-024-13613-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10973-024-13613-7