Abstract
Context
The Mediterranean basin is characterized by a heterogeneous landscape historically shaped by human activities. Land abandonment and extensive monocultures, however, have led to increasing homogeneity of Mediterranean habitats. Albeit the effects of habitat heterogeneity on wildlife have been widely studied, the available information on how habitat homogenization impacts the organization of mesocarnivore communities is still scant.
Objectives
We investigated the relationship of environmental characteristics with occupancy, activity, community organization, and co-occurrence of mesocarnivore species in space and time. We focused on five key species (Vulpes vulpes, Genetta genetta, Meles meles, Herpestes ichneumon, and Martes foina) widely distributed throughout Mediterranean ecosystems.
Methods
The study was conducted in north-central Portugal between June 2019 and June 2020. We used 300 camera trap sampling points coupled with occupancy, activity, and co-occurrence analysis to assess how different environmental characteristics influence mesocarnivores community.
Results
We found that the occupancy of mesocarnivores is influenced by topography, landscape composition and structure, and human activity. We observed that landscape homogeneity limits the co-occurrence of mesocarnivores. In heterogeneous landscapes we recorded a greater overlap in activity (seven of ten species pairs) and spatial co-occurrence (six of the seven species pairs analyzed) of mesocarnivores species.
Conclusions
We demonstrated the importance of landscape characteristics in the composition of the mesocarnivores community. Our findings support the adoption of management measures able to mitigate the impacts of landscape homogenization. Maximizing the heterogeneity is an important step to benefit the mesocarnivore community in altered Iberian landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Mammalian carnivores play a key role in the structure and dynamics of wildlife communities (Estes et al. 2011; Ritchie et al. 2012). Mesocarnivores [< 15 kg (kg)], in particular, are the most representative and abundant group of the order Carnivora (Prugh et al. 2009; Roemer et al. 2009). These species are ecologically diverse, present a wide range of social and feeding behaviors, and can be found in a wide variety of habitats, including human-shaped landscapes. Mesocarnivores can alter vegetation communities, facilitating nutrient flow, and even occupy unique roles (e.g., dispersers of seed or predators of small mammals) that cannot be filled by larger carnivores (Roemer et al. 2009; Ćirović et al. 2016). The composition and structure of mesocarnivore communities are strongly influenced by a set of environmental filters, both natural and/or of human origin, that may drive the frequency and intensity of competitive interactions (Ritchie and Johnson 2009; Monterroso et al. 2020; Davies et al. 2021; Suárez-Tangil and Rodríguez 2022).
Competitive interactions can act directly on the creation of ecological niches since competing species often reduce the access of subordinate species to optimal resources (e.g., habitat, time of day, optimal prey) (Schmitt and Holbrook 1986; Case and Bolger 1991; Davies et al. 2021). For example, in California, it was observed that more dominant mesocarnivores can occupy the spatial niche of subordinate mesocarnivores and modify resource exploitation when human pressure is higher, resulting in more intense intraguild competition (Smith et al. 2018). Therefore, to reduce the negative impacts of interspecific competition, species usually segregate into three main niche dimensions: trophic dimension, temporal dimension, and spatial dimension (Schoener 1974).
The description of species co-occurrence strategies is relevant for wildlife conservation and management, and the Mediterranean ecosystems of the Iberian Peninsula are important study sites as they are recognized as one of the most important worldwide biodiversity hotspots (Myers et al. 2000). Iberian Mediterranean landscapes have been progressively transformed into an area of intense forest and agriculture production. Yet, depending on the management options and local activities, there are still regions represented by heterogeneous and functional landscapes, supporting a rich and diverse animal community (Rosalino et al. 2009). But even these regions are at risk. The ongoing abandonment of traditional practices, combined with forest and agricultural intensification, can strongly influence the structure of ecosystems, affecting the composition and dynamics of landscapes and wildlife communities (Davies et al. 2021). The most visible impact of land change are the monocultures of maritime pine (Pinus pinaster) and eucalyptus (Eucalyptus globulus) that have transformed the Mediterranean landscapes of Iberia at an unprecedented rate (Nunes et al. 2019).
Landscape homogenization and simplification are known to directly influence the composition and structure of mesocarnivore communities. Sympatric species interact multidimensionally and often adapt niche dimensions to co-exist. Some studies have already assessed the ecological interactions between carnivore species (temporal: Monterroso et al. 2014; Vilella et al. 2020; spatial: Palomares et al. 1996; Sarmento et al. 2011); however, few assessed community interactions using a multidimensional approach. There is still a substantial lack of knowledge on the ecological characteristics driving carnivore communities in Mediterranean landscapes, especially on the effects of landscape composition and structure on mesocarnivore community organization, which limits the adoption of amenable measures to foster species co-occurrence. In competitive systems and complex ecological communities, sympatric species may need to segregate into niches of different dimensions to preserve a minimum overlap, which is more difficult in homogeneous landscapes as the resources to be exploited are limited (Verdade et al. 2011; Davies et al. 2021). Due to the high complexity of mesocarnivores’ responses to environmental and human factors (Gonçalves et al. 2012; Curveira-Santos et al. 2017), the adoption and improvement of habitat-oriented management practices in changing environments are essential.
This study aims to describe how environmental characteristics, including landscape composition and structure, influence the occupancy, community organization, and co-occurrence of mesocarnivore species in space and time. We focus on five key species due to their abundance and ecological role in Mediterranean ecosystems (red fox: Vulpes vulpes, common genet: Genetta genetta, European badger: Meles meles, Egyptian mongoose: Herpestes ichneumon, and beech marten: Martes foina). We hypothesized that species occupancy is driven by the combined effects of five factors (H1, microscale habitat, topography, landscape composition and structure, human activity, and seasonality—Table 1) and that species co-occurrence is fostered by heterogeneous landscapes (H2). Heterogeneous landscapes are characterized by different physical characteristics and by an irregular distribution of resources. These characteristics are often considered key facilitators of species co-occurrence in space and time (Palmer 2003; Tews et al. 2004; Davies et al. 2021). For instance, the availability of habitats that characterizes the heterogeneous landscapes (e.g., microhabitats, shrub and tree cover, outcrops, and tree-related structures) may facilitate niche partitioning in different spatial dimensions, which may foster the co-occurrence of sympatric species in time (Chesson 2000; Stein et al. 2014). Thus, we expect that spatial and temporal avoidance among species will be particularly evident in more homogeneous landscapes, where the risk of agonistic encounters is higher (Monterroso et al. 2020; Davies et al. 2021).
Materials and methods
Study area
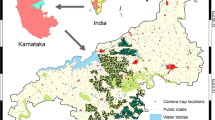
The study was carried out in the north-central of Portugal, in an area of 5504 km2 (Fig. 1). The elevation varies between 130 and 1140 m (Farr et al. 2007). The study area encompasses different subregions that present landscape and socioeconomic particularities. The north-eastern region is mainly focused on agriculture and livestock. The north-western region encompasses broadleaved forests, scrublands, and monocultures, mostly. The central region comprises the largest protected area in Portugal: the Parque Natural da Serra da Estrela, characterized by broadleaved, coniferous, and remnants of oak forests. Finally, the southern region presents a landscape dominated by dense shrub and oak forests, however, monocultures of Pinus sp. are also abundant. During the twentieth century, land use changes caused by rural exodus, the abandonment of traditional agricultural practices, shrub and forest encroachment, and the spatial representativeness of monocultures have fostered an increased landscape homogenization.
Land use and land cover characterization of the study area located in the central-north of Portugal. The white points represent the camera trap stations. The map in the lower left-hand corner represents Portugal (grey) and the location of the study area (black). The land use/land cover information was gathered from the Land Use and Land Cover (LULC) Map of Continental Portugal for 2018 (Direção-Geral do Território 2020)
Field survey and data collection
Field campaigns were conducted from June 2019 to June 2020 by selecting areas that ensured the spatial representativeness of landscape composition and structure. We installed 30 cameras (Browning BTC-5HDPX) per campaign, that remained in the field over a minimum period of 30 days. Based on the home range of the species studied (Cavallini and Lovari 1994; Santos-Reis et al. 2005; Rovero and Zimmermann 2016), cameras were installed 1000–1500 m from each other to ensure sampling independence. The cameras were active 24 h a day and were placed at a height of approximately 30–40 cm from the ground. The locations had good visibility and were free from dense vegetation to avoid false triggers. We did not use bites or attractants to not influence the spatial and temporal behavior of species. The equipment was programmed to take three photos in each detection, with a time interval of 30 s between detections. This choice represents a trade-off between increasing the detectability of individuals and the storage capacity of SD cards. The cameras were continuously relocated to another location to cover our sampling area until June 2020. Our methodology was supported by a set of guidelines aimed at obtaining precise estimates of species occupancy and detection rates (Kays et al. 2020).
Explanatory variables
We considered five factors (F) represented by 28 explanatory variables to assess the drivers of mesocarnivore occupancy (Table 1). We tested the variables that are considered most relevant to mesocarnivore detection (Cruz et al. 2015; Curveira-Santos et al. 2019). We measured in the field five microscale landscape indicators that may influence occupancy and detection probability (F1- the percentage of tree, shrub, herbaceous, and rock cover, and the height of vegetation at the sampling point). The collection of microscale landscape indicators was carried out at the locations where the cameras were installed. The observer analyzed the vegetation surrounding the point for a radius of 100 m. This measure is a trade-off between what the observer can see and its significance for the species under study. The classification was carried out considering the following categories: tree, shrub, herbaceous, and rock cover (0%; 1–25%; 26–50%; 51–75%; 76–100%); and the average height of the vegetation (< 50 cm; 50–150 cm; > 150 cm). The collection was always performed by the same observers to avoid variations between observations.
Regarding the macroscale variables, represented by factors 2, 3, and 4, we defined a 500 m radius buffer around each camera trap to estimate the percentage of the different habitats present within a particular area. The radius of the buffers was consistent with the known home range for some mesocarnivore species (e.g., beech marten (455 m radius; Santos-Reis et al. 2005); common genet (512 m radius; Santos-Reis et al. 2005); red fox (474 m radius; Cavallini and Lovari 1994). Topography (F2) influences, both directly and indirectly, the presence of mesocarnivores, by driving vegetation composition, human pressure, and climate (Curveira-Santos et al. 2019; Reddell et al. 2021). Landscape composition (F3) acts directly on the ecology and dynamics of mesocarnivore populations (Sarmento et al. 2011; Wait et al. 2018), potentially altering the resources available to the mesocarnivore community (Verdade et al. 2011; Davies et al. 2021). Human activity (F4) increases disturbance, which can directly affect mesocarnivores occupancy and modulate the importance of other environmental characteristics, such as land use and landscape composition and structure (Barrull et al. 2014; Cruz et al. 2015). Different seasons present higher or lower resource availability, which is expected to influence mesocarnivores occupancy (F5) (Schuette et al. 2013). Finally, as these factors are not mutually exclusive, we also assessed how their combined effects influence the occupancy of Mediterranean carnivores.
Occupancy
Our data matrix was organized in sampling points (279 points on the y-axis) over the 35 sampling days that the cameras were in the field (x-axis). We installed cameras trap at 300 sites, however, 21 of the sites were not used due to mechanical problems or theft. This matrix was constructed for each species and was filled considering species presence (1), absence (0), and camera inactivity (NA). Our models were built according to the single-season rule (MacKenzie et al. 2002), where species occupancy can be estimated with higher probability, taking into account imperfect detection. The single-season occupancy model collects information on sampling occasions; thus, we arranged the matrix (5 days = 1 occasion) with a total of 7 occasions.
We grouped the variables into five factors known to influence species detection and occupancy (Table 1). Within each factor, we performed multicollinearity tests using the ‘corvif’ function in the ‘AED’ R package (Zuur et al. 2007). A variable was excluded whenever we recorded a variance inflation factor higher than 5 (VIF > 5, Zuur et al. 2007). In each run, the variable with the highest value was removed and the process was repeated until there were no more variables exceeding the defined threshold.
We tested how microscale habitats could influence species detection (F1). We fitted detection models considering all possible combinations between microscale habitat indicators. All models produced were classified using the Akaike information criteria (AIC, Akaike 1974), and the one that reached the lowest AIC value was considered the most adequate to explain how detectability varies concerning microscale habitat. Then, we produce occupancy models encompassing all the remaining factors (F2–F5). For each factor, we fitted a set of models corresponding to all possible combinations of variables. The models with an ΔAIC < 2 were considered suitable to estimate species-specific occupancy states and to identify the variables per factor that shape these estimates (Burnham and Anderson 2002). Finally, the significant variables within each factor were merged into a final model (FM). The final model with the smallest AICc was selected to calculate the value of the c-hat, an indicator of model dispersion. If the model presented a c-hat value distant from 1, it would be adjusted using the excessive dispersion factor and would be classified based on the Quasi Akaike information criterion corrected for small samples (QAICc) using the limit of ΔQAICc ≤ 2 (MacKenzie and Bailey 2004). The packages ‘unmarked’ and ‘MuMIn’ from the R software (R Core Team 2020) were used to produce the occupancy models.
Space–time co-occurrence in homogeneous and heterogeneous landscapes
The spatial co-occurrence analysis is based on the conditional occupancy of two species, which are composed of a hierarchical model of conditional occupancy and detection probabilities. The model provides several indicators, such as the probability of species occupancy A (ψA); the probability of species B occupancy when species A is present (ψBA); and the probability of species B occupancy when species A is absent (ψBa). Additionally, the model allows to assess the probability of a dominant species being detected when the subordinate species is present (RA); the probability of a dominant species being detected when the subordinate species is absent (PA); the probability of a subordinate species being detected when the dominant species is not present (PB); the probability of a subordinate species being detected when the dominant species is present and detected (RBA); and the probability of a subordinate species being detected when the dominant species is present but not detected (RBa) (Richmond et al. 2010). Based on these indicators, we calculated the Species Interaction Factor (SIF), which indicated if the studied species are presenting a segregation (are avoiding) or an aggregation (are co-occurring) behavior. The SIF was calculated using the formula of Richmond and colleagues (2010). When SIF = 1, the two species are considered to occur independently; if SIF < 1, the species B is less likely to co-occur with the dominant species A (segregation hypothesis); and if SIF > 1, the species B is more likely to co-occur with species A (aggregation hypothesis) (Richmond et al. 2010). To perform the estimates of co-occurrence between subordinate (B) and dominant (A) species, we considered the following species pairs, according to the highest probability of these pairs co-occurring in the spatiotemporal dimension (Vilella et al. 2020; Ferreiro-Arias et al. 2021). We considered the red fox as a dominant species in all pairs except for the European badger, as this mustelid dominance over the canid has been previously documented (Macdonald et al. 2004; Barrull et al. 2014). The interaction between the beech marten and the common genet was also studied, due to their morphological and ecological similarities (Santos-Reis et al. 2005), and based on body size, the common genet was considered dominant. The European badger according to his body mass and behavior, is dominant over the common genet and stone marten (Monterroso et al. 2013; Barrull et al. 2014). Accordingly, we determined: badgerA—foxB; badgerA—martenB; badgerA—genetB; foxA—martenB; foxA—genetB; foxA—mongooseB; genetA—martenB. The Egyptian mongoose was only assessed with the fox, due to the low possibility of a spatiotemporal encounter with the other species. We estimate these metrics for two distinct scenarios: homogeneous and heterogeneous landscapes. The division between the homogeneous and heterogeneous landscapes was carried out by collecting and analyzing the landscape variables extracted for the 300 buffers (500 m radius) of the study area using the most recent land use and land cover layer made available by the Direção-Geral do Território (DGT 2020). We calculated five indices for the characterization of landscape structure: Simpson’s diversity index, number of habitats per buffer, number of fragments per buffer, average shape, and patch cohesion index. These variables can translate in a complex way whether a given area exhibits greater or lesser heterogeneity (Fahrig et al. 2011; Katayama et al. 2014; Grande et al. 2020). The variables were used for the k-means cluster analysis method in R. The test can group the objects into clusters, being a useful tool to organize and visualize the information contained in multivariate spatial data (Long et al. 2010). We performed the division into two groups, where one represents areas of greater heterogeneity and the other areas of greater homogeneity. The landscape indices were computed using the Landscape Ecology Statistics (LecoS) (Jung 2016) plugin of the QGIS® 3.16.11 (QGIS 2018). The co-occurrence patterns between the five mesocarnivore species were analyzed using the ‘occ2sps’ function in the ‘wiqid’ package in R software (R Core Team 2020).
For the activity overlap analysis, we used the records with a minimum interval of 30 min between them (Davis et al. 2011; Ares-Pereira et al. 2022). This option ensures the independence of the gathered information (Wang et al. 2015; O’Connor et al. 2017). We calculated confidence intervals (CI) by performing 1000 bootstraps and considering the overlap coefficient Dhat4, which is advised when we have records greater than 50 (Ridout and Linkie 2009a). The test varies between 0 and 1, where values close to 1 indicate high overlap and values close to 0 indicate low activity overlap. We considered three overlap classes: low (Dhat4 < 0.5), medium (0.5 < Dhat4 > 0.75), or high (Dhat4 > 0.75) (Monterroso et al. 2014). Overlap was calculated for the entire dataset and also tested for the effects of the season (spring/summer and autumn/winter) and landscape composition (heterogeneous and homogeneous landscapes). We used the 'overlap' package version 0.3.3 (Ridout and Linkie 2009b) in R software.
Results
Our sampling comprised information from 279 camera trapping sites throughout 9947 cameras-night. We obtained a total of 1257 (red fox), 327 (beech marten), 150 (European badger), 146 (common genet), and 59 (Egyptian mongoose) independent records.
Occupancy
For the occupancy analysis, as the matrix is constituted with presence/absence data, we ended up with a total of 1051 mesocarnivores independent records: 620 (red fox), 190 (beech marten), 103 (European badger), 103 (common genet), and 35 (Egyptian mongoose). The Egyptian mongoose was the species with the lowest occupancy probability in our study area, followed by the common genet and the European badger. The red fox and the beech marten recorded an occupation probability higher than 50% (Table 2).
We did not detect multicollinearity issues. Overall, the best final occupancy models (lowest AICc) presented c-hat values distant from 1 (Egyptian mongoose = 3.46; European badger = 2.59; beech marten = 1.58; common genet = 1.40; red fox = 1.45), indicating data overdispersion. We performed all models using the Quasi Akaike information criterion corrected for small samples (QAICc). Finally, except for the Egyptian mongoose, all species’ occupancy was affected by at least one factor (Table 3).
From the initial set of 28 variables, we retained six significant variables (altimetry, slope, number of habitats, coniferous land cover, agriculture, and eucalyptus) from three factors (F2–Topography, F3–Landscape composition and structure, and F4–Human activity). Topography influences the occupancy of beech marten and red fox; altimetry has a positive effect on the occupancy of beech marten and slope has a negative influence on the occupancy of red fox. Landscape composition and structure, in particular, the number of habitats, increases the occupancy of the common genet and red fox, while the presence of coniferous forests harms the occupancy of the common genet and European badger. Finally, human activities, such as agriculture, have a negative influence on the occupancy of the common genet and beech marten (Fig. 2). All the models for each species can be found in Supplementary Material.
Variables with significant influence on each species occupancy (red fox, common genet, European badger, Egyptian mongoose, beech marten). Our modeling approach allowed to retain six significant variables (slope, number of habitats, agriculture, coniferous land cover, eucalyptus and altimetry) from three factors (F2–Topography, F3–Landscape composition and structure, and F4–Human activity)
Space–time co-occurrence in homogeneous and heterogeneous landscapes
Our results show that species tend to occur independently in heterogeneous landscapes, while avoidance behaviors are particularly common in homogeneous landscapes. We observed that six of the seven pairs of species reported in heterogeneous landscapes record SIF values closer to 1 (Table 4). This result indicates that landscape heterogeneity allows species to share the same space, increasing the diversity and balance of the mesocarnivores community.
The analysis of the species activity patterns complements the results focused on the spatial settings. We found a high overlap (Dhat4 > 0.75) between common genet, beech marten, and European badger activities, being the common genet and beech marten the species with the highest overlap (Dhat4 = 0.91) (Fig. 3).
Fox-genet, fox-marten, and fox-badger showed a medium overlap (0.5 < Dhat4 < 0.75). We observed a low overlap between the Egyptian mongoose and the other species evaluated (Fig. 3). We did not record large seasonal changes in activity overlap (Table 5).
We observed greater activity overlap in heterogeneous areas for the nocturnal species (red fox, European badger, beech marten, and common genet), while the Egyptian mongoose seems to decrease the activity overlap with the remaining species in heterogeneous areas (Fig. 4).
Discussion
The co-occurrence of sympatric species can be affected by a myriad of natural and human-induced factors that act through direct or indirect pathways (Monterroso et al. 2014; Zhao et al. 2020). We tested the effects of five complementary factors that act at different spatial scales to shed light on the environmental mechanisms that drive the occupancy and community composition of Mediterranean mesocarnivores. Our results suggest a significant influence of topography, landscape composition and structure, and human activities on the patterns of mesocarnivores occupancy, which corroborates our working hypothesis. Overall, we report a higher occupancy for red fox and beech marten concerning the other mesocarnivores. Red fox occupancy is higher than what was previously recorded in the southern region of our study area (Serra da Malcata: Sarmento et al. 2011). In contrast to the common genet and Egyptian mongoose, beech marten also shows higher occupancy compared to other areas (Sarmento et al. 2011; Curveira-Santos et al. 2019). The European badger shows a similar occupancy to other European regions (Italy: Torretta et al. 2016). Topography influenced the red fox and the beech marten occupancy. The selection of beech marten for higher altitudes was already reported in a previous study (Zabala et al. 2009). Altimetry is probably a key variable that supports the competitive exclusion between genets (lower altitudes) and martens (higher altitudes) (Zabala et al. 2009). The slope negatively influenced red fox occupancy. The effect of the slope in red fox ecology is still poorly understood. The influence of topography should be interpreted carefully as topography brings potentially confounding factors associated (e.g., temperature, human pressure, vegetation structure).
The landscape composition and structure play a key role in species occupancy. For instance, the number of habitats positively influences the occupancy of the red fox and common genet. Our study supports the contention that the common genet mainly occurs in heterogeneous habitats characterized by complex plant strata (Pereira et al. 2012), and shows that the red fox, taking advantage of its generalist and opportunistic behavior (Harrington and Macdonald 2008), is also favored by a landscape composed by a mosaic of habitats. This landscape characteristic provides a greater diversity of refuges and food, acting positively on mesocarnivores occupancy (Cavallini and Lovari 1994; Cagnacci et al. 2004; Castro et al. 2022). The coniferous forests negatively influence the occupancy of the common genet and European badger. This result corroborates what was previously reported in Switzerland, where European badgers also strongly avoided coniferous landscapes (Good et al. 2001). The low occupancy of common genets in coniferous forests may result from the low availability of shelters and resources (Pereira et al. 2012), as coniferous forests in our study area are exploited, as well as in most parts of the country (Águas et al. 2014; ICNF 2019).
We also observed a significant influence of human activities (F4) on mesocarnivores occupancy. The variable related to eucalyptus monocultures showed a positive influence on Egyptian mongoose occupancy. The establishment of Eucalyptus sp. plantations became common in many regions and Portugal is ranked among the countries with the largest areas of E. globulus planted in the world (Potts et al. 2004). Introduced in Portugal in the mid-nineteenth century, Eucalyptus sp. is quickly replacing the Mediterranean maquis (Águas et al. 2014). These changes in land use, accompanied by the shrub encroachment that characterizes poorly managed plantations, seem to favor the presence of the Egyptian mongoose, which is usually associated with areas of eucalyptus plantations (Palomares and Delibes 1993; Pita et al. 2009; Barros et al. 2015). However, attention is needed since landscape homogenization ended up destroying several other niches, which can have a large impact on other mesocarnivores species (Pereira et al. 2012; Cruz et al. 2015; Castro et al. 2022). We emphasized that these results regarding Egyptian mongoose should be interpreted carefully, as the species was detected only sporadically in our study. Finally, agriculture showed a negative influence on the occupancy of two species, the beech marten and the common genet. Both species benefit from canopy cover, using areas covered by dense trees that provide refuge and are important feeding resources (Virgós et al. 2001; Galantinho and Mira 2009). Agricultural areas are mostly homogeneous and usually characterized by low forest cover, which have direct impacts on the beech marten and common genet occupancy, as the species, particularly the beech marten, is not able to survive in landscapes with forest cover below a minimum required threshold (Virgós et al. 2002).
Landscape homogeneity decreases the spatiotemporal co-occurrence of mesocarnivores
Landscape composition is rarely considered when analyzing the spatiotemporal co-occurrence patterns of mesocarnivores. However, our study highlights the negative effect that landscape homogenization may have on mesocarnivore community organization. Our results demonstrate that all nocturnal species showed greater overlap in heterogeneous areas. As carnivore species exhibit strong agonistic interactions, co-occurrence mechanisms (e.g., temporal segregation) may be fundamental in preventing intraguild predation behaviors (Manlick et al. 2017). Seven of the ten pairs of species showed greater temporal overlap in heterogeneous landscapes, corroborating our working hypothesis that landscape heterogeneity can act as a facilitator of species co-occurrence through the availability of nonuniform resource distributions (Fisher et al. 2013). Seasonality, which is known to have a strong influence on carnivore species activity (Barrull et al. 2014; Torretta et al. 2016; Vilella et al. 2020) did not show any relevant effect in our study, suggesting that, in our area, species adjust their activities much more as a function of landscape composition than seasonality.
The temporal patterns also demonstrated variability between pairs, being possible to detect temporal segregation among species. In general, the activity overlap reported is similar to the results of studies developed in other Mediterranean areas (Curveira-Santos et al. 2017; Ferreiro-Arias et al. 2021). The pairs with both nocturnal species showed considerable temporal overlap between species, while the Egyptian mongoose exhibited low temporal overlap with all species except the red fox. This greater overlap between mongoose-fox is expected and possibly caused by the facultative nocturnal activity and moderate crepuscular activity of the red fox (Barrull et al. 2014; Curveira-Santos et al. 2017; Ferreiro-Arias et al. 2021). A high temporal overlap observed between common genet, beech marten, and European badger may be linked to niche partitioning factors (Curveira-Santos et al. 2017; Vilella et al. 2020), aspects of the species’ ecology, or even stochastic factors.
In spatial co-occurrence analysis, all mesocarnivores pairs analyzed (except fox-mongoose) had their co-occurrence values closer to SIF = 1 in heterogeneous landscapes, indicating a more independent co-occurrence of mesocarnivore species in these scenarios. The European badger, red fox, common genet, and beech marten, for example, share the available food resources to a considerable extent, converging or diverging the diet depending on food availability (Barrientos and Virgós 2006; Donadio and Buskirk 2006; Verdade et al. 2011; Barrull et al. 2014). Accordingly, our results reinforce that multifunctional landscapes can allow a greater possibility of space sharing among mesocarnivores, even facilitating co-occurrence among species that are known to present agonistic encounters (e.g., badger-fox) (Palomares and Caro 1999; Macdonald et al. 2004).
To our best knowledge, this study represents a relevant contribution that sheds light on the influence of landscape heterogeneity on mesocarnivore community organization in Mediterranean ecosystems, an area facing the unpredictable ecological consequences of rural exodus and landscape homogenization. Our results are supported by previous findings on the impacts of landscape homogenization on the wildlife assemblages of Mediterranean ecosystems, and beyond. A recent study carried out in southwestern Spain showed that homogeneous landscapes, resulting from the conversion of traditional farms into intensive agriculture, have a strong and negative impact on the presence of mammal species, including mesocarnivores (Suárez-Tangil and Rodríguez 2022). The authors highlight the importance of preserving native woody communities within certain thresholds to foster habitat heterogeneity and to ensure the maintenance of wild mammal communities. In Africa, it was demonstrated that the species were not able to avoid competitors in highly homogeneous landscapes, which may affect the establishment of permanent populations (Davies et al. 2021). Importantly, the heterogeneity of natural landscapes should not be confused with fragmented landscapes resulting from anthropogenic activities, as these can act detrimentally on the co-occurrence of the carnivore community (Manlick et al. 2020).
Conclusions and practical implications
Our work stresses the importance of landscape structure in the composition of mesocarnivore community. It also suggests that landscape structure and human activities may buffer or strengthen the effects of mesocarnivore interactions, such as competition. We recognize, however, that the mechanisms driving species occupancy, activity, and co-occurrence seldom lead to simple and definitive answers. For instance, we were not able to test the direct effects of apex predators and resource availability on the mesocarnivores community (but see Rossa et al. 2021). Contrary to our expectations, we also did not detect any influence of shrub cover, water availability, and human footprint index on the parameters analyzed, even considering that these variables were previously identified as relevant for the studied species (Wait et al. 2018; Suárez-Tangil and Rodríguez 2022). As technology and new analytical methods are readily available, the time is right to generate new fundamental and applied ecological knowledge. The combination of sampling and remote sensing methods to estimate the carrying capacity of an ecosystem/habitat, coupled with the use of GPS or reverse GPS techniques to describe the movement ecology of the target species, will expand the potential of camera trap data and will allow us to move from correlational to mechanistic evidence on how wildlife communities respond and adapt to ongoing changes.
Despite some limitations, our study has important practical implications and could support the adoption of conservation and management measures aimed at mitigating and counteracting the impacts of landscape homogenization, mainly caused by forestry (e.g., monoculture) and agriculture intensification, but also by land abandonment and shrub encroachment. These measures can be set at two levels with different spatial and temporal scales. First, we recommend limiting or carefully planning intensive forestry and agriculture practices. We are not able to define thresholds of native vegetation that should be kept to guarantee the stability of mesocarnivores communities (but see Suárez-Tangil and Rodríguez 2022), however, we recommend the promotion of mosaic landscapes by retaining native woody vegetation, such as forest and shrub areas, interspersed by natural grasslands. The promotion of forestry and agricultural schemes able to accommodate habitat enhancement initiatives will benefit the co-occurrence of mesocarnivores in space and time while promoting other biodiversity indicators. The conservation and management budgets aimed at restoring landscape functionality are limited. Therefore, the selection and prioritization of restoration areas aimed at maximizing heterogeneity and connectivity is an important step to overcoming homogenization. The re-establishment of historical disturbance regimes, the protection of restoration sites, and the adoption of national policies to halt desertification are also decisive measures to revert the homogenization of Mediterranean landscapes.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Águas A, Ferreira A, Maia P et al (2014) Natural establishment of Eucalyptus globulus Labill. in burnt stands in Portugal. For Ecol Manag 323:47–56
Akaike H (1974) A new look at the statistical model identification. IEEE Trans Automat Contr 19:716–723
Ares-Pereira G, Rosalino LM, Teixeira D et al (2022) Eucalyptus plantations alter spatiotemporal relationships of wild ungulates. Agric Ecosyst Environ 340:108174
Barrientos R, Virgós E (2006) Reduction of potential food interference in two sympatric carnivores by sequential use of shared resources. Acta Oecologica 30:107–116
Barros T, Carvalho J, Pereira MJR et al (2015) Following the trail: factors underlying the sudden expansion of the Egyptian mongoose (Herpestes ichneumon) in Portugal. PLoS ONE 10:1–18
Barrull J, Mate I, Ruiz-Olmo J et al (2014) Factors and mechanisms that explain coexistence in a Mediterranean carnivore assemblage: an integrated study based on camera trapping and diet. Mamm Biol 79:123–131
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Cagnacci F, Meriggi A, Lovari S (2004) Habitat selection by the red fox vulpes vulpes (L. 1758) in an alpine area. Ethol Ecol Evol 16:103–116
Case TJ, Bolger DT (1991) The role of interspecific competition in the biogeography of island lizards. Trends Ecol Evol 6:135–139
Castro G, Teixeira D, Ares-Pereira G et al (2022) Drivers of occupancy patterns for the red fox, Vulpes vulpes, in Mediterranean Eucalyptus plantations. For Ecol Manag 519:120293
Cavallini P, Lovari S (1994) Home range, habitat selection and activity of the red fox in a Mediterranean coastal ecotone. Acta Theriol 39:279–287
Chesson P (2000) of S Pecies D Iversity. Annu Rev Ecol Syst 31:343–366
Ćirović D, Penezić A, Krofel M (2016) Jackals as cleaners: ecosystem services provided by a mesocarnivore in human-dominated landscapes. Biol Conserv 199:51–55
Cruz J, Sarmento P, White PCL (2015) Influence of exotic forest plantations on occupancy and co-occurrence patterns in a mediterranean carnivore guild. J Mammal 96:854–865
Curveira-Santos G, Marques TA, Björklund M, Santos-Reis M (2017) Mediterranean mesocarnivores in spatially structured managed landscapes: community organisation in time and space. Agric Ecosyst Environ 237:280–289
Curveira-Santos G, Pedroso NM, Barros AL, Santos-Reis M (2019) Mesocarnivore community structure under predator control: unintended patterns in a conservation context. PLoS ONE 14:1–18
Davies AB, Tambling CJ, Marneweck DG et al (2021) Spatial heterogeneity facilitates carnivore coexistence. Ecology 102:1–13
Davis ML, Kelly MJ, Stauffer DF (2011) Carnivore co-existence and habitat use in the Mountain Pine Ridge Forest Reserve, Belize. Anim Conserv 14:56–65
Direção-Geral do Território (2020) Carta de Uso e Ocupação do Solo (COS)
Donadio E, Buskirk SW (2006) Diet, morphology, and interspecific killing in carnivora. Am Nat 167:524–536
Estes JA, Terborgh J, Brashares JS et al (2011) Trophic downgrading of planet earth. Science 333:301–306
Fahrig L, Baudry J, Brotons L et al (2011) Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol Lett 14:101–112
Farr TG, Rosen PA, Caro E et al (2007) The shuttle radar topography mission. Rev Geophys 45:1–33
Ferreiro-Arias I, Isla J, Jordano P, Benítez-López A (2021) Temporal and trophic partitioning promote coexistence between mesocarnivores in a Mediterranean landscape. bioRxiv. https://doi.org/10.1101/2020.11.05.370403
Fisher JT, Anholt B, Bradbury S et al (2013) Spatial segregation of sympatric marten and fishers: the influence of landscapes and species-scapes. Ecography 36:240–248
Galantinho A, Mira A (2009) The influence of human, livestock, and ecological features on the occurrence of genet (Genetta genetta): a case study on Mediterranean farmland. Ecol Res 24:671–685
Gonçalves P, Alcobia S, Simões L, Santos-Reis M (2012) Effects of management options on mammal richness in a Mediterranean agro-silvo-pastoral system. Agrofor Syst 85:383–395
Good TC, Hindenlang K, Imfeld S, Nievergelt B (2001) A habitat analysis of badger (Meles meles L.) setts in a semi-natural forest. Zeitschrift fur Saugetierkd 66:204–214
Grande TO, Alencar RM, Ribeiro PP, Melo FR (2020) Fragment shape and size, landscape permeability and fragmentation level as predictors of primate occupancy in a region of Brazilian Cerrado. Acta Sci-Biol Sci 42:1–15
Harrington LA, Macdonald DW (2008) Spatial and temporal relationships between invasive American mink and native European polecats in the southern United Kingdom. J Mammal 89:991–1000
ICNF (2019) IFN6—Anexo Técnico
Jung M (2016) LecoS—a python plugin for automated landscape ecology analysis. Ecol Inform 31:18–21
Katayama N, Amano T, Naoe S et al (2014) Landscape heterogeneity-biodiversity relationship: effect of range size. PLoS ONE 9:1–8
Kays R, Arbogast BS, Baker-Whatton M et al (2020) An empirical evaluation of camera trap study design: how many, how long and when? Methods Ecol Evol 11:700–713
Long J, Nelson T, Wulder M (2010) Regionalization of landscape pattern indices using multivariate cluster analysis. Environ Manag 46:134–142
Macdonald DW, Buesching CD, Stopka P et al (2004) Encounters between two sympatric carnivores: red foxes (Vulpes vulpes) and European badgers (Meles meles). J Zool 263:385–392
MacKenzie DI, Bailey LL (2004) Assessing the fit of site-occupancy models. J Agric Biol Environ Stat 9:300–318
MacKenzie DI, Nichols JD, Lachman GB et al (2002) Estimating site occupancy rates when detection probabilities are less than one. Ecology 83:2248–2255
Manlick PJ, Woodford JE, Zuckerberg B, Pauli JN (2017) Niche compression intensifies competition between reintroduced American martens (Martes americana) and fishers (Pekania pennanti). J Mammal 98:690–702
Manlick PJ, Windels SK, Woodford JE, Pauli JN (2020) Can landscape heterogeneity promote carnivore coexistence in human-dominated landscapes? Landsc Ecol 35:2013–2027
Monterroso P, Alves PC, Ferreras P (2013) Catch me if you can: diel activity patterns of mammalian prey and predators. Ethology 119:1044–1056
Monterroso P, Alves PC, Ferreras P (2014) Plasticity in circadian activity patterns of mesocarnivores in Southwestern Europe: implications for species coexistence. Behav Ecol Sociobiol 68:1403–1417
Monterroso P, Díaz-Ruiz F, Lukacs PM et al (2020) Ecological traits and the spatial structure of competitive coexistence among carnivores. Ecology 101:1–16
Myers N, Mittermeier RA, Mittermeier CG et al (2000) Biodiversity hotspots for conservation priorities. Nature 403:853–858
Nunes LJR, Meireles CIR, Gomes CJP, de Ribeiro NMC (2019) Socioeconomic aspects of the forests in Portugal: recent evolution and perspectives of sustainability of the resource. Forests 10:1–11
O’Connor KM, Nathan LR, Liberati MR, Tingley MW, Vokoun JC, Rittenhouse TA (2017) Camera trap arrays improve detection probability of wildlife: Investigating study design considerations using an empirical dataset. PLOS ONE 12(4):e0175684. https://doi.org/10.1371/journal.pone.0175684
Palmer TM (2003) Spatial habitat heterogeneity influences competition and coexistence in an African acacia ant guild. Ecology 84:2843–2855
Palomares F, Caro TM (1999) Interspecific killing among mammalian carnivores. Am Nat 153:492–508
Palomares F, Delibes M (1993) Social organization in the Egyptian mongoose: group size, spatial behaviour and inter-individual contacts in adults. Anim Behav 45:917–925
Palomares F, Ferreras P, Fedriani JM, Delibes M (1996) Spatial relationships between Iberian lynx and other carnivores in an area of South-Western Spain. J Appl Ecol 33:5–13
Pereira P, da Silva AA, Alves J et al (2012) Coexistence of carnivores in a heterogeneous landscape: habitat selection and ecological niches. Ecol Res 27:745–753
Pita R, Mira A, Moreira F et al (2009) Influence of landscape characteristics on carnivore diversity and abundance in Mediterranean farmland. Agric Ecosyst Environ 132:57–65
Potts BM, Vaillancourt RE, Jordan G, et al (2004) Exploration of the eucalyptus globulus gene pool brad. In: Eucalyptus in a changing world, pp 11–15
Prugh LR, Stoner CJ, Epps CW et al (2009) The rise of the mesopredator. Bioscience 59:779–791
QGIS Geographic Information System., Open Source Geospatial Foundation (2018) QGIS Development Team
Reddell CD, Abadi F, Delaney DK et al (2021) Urbanization’s influence on the distribution of mange in a carnivore revealed with multistate occupancy models. Oecologia 195:105–116
Richmond OMW, Hines JE, Beissinger SR (2010) Two-species occupancy models: a new parameterization applied to co-occurrence of secretive rails. Ecol Appl 20:2036–2046
Ridout MS, Linkie M (2009a) Estimating overlap of daily activity patterns from camera trap data
Ridout M, Linkie M (2009b) Package “overlap”: estimates of coefficient of overlapping for animal activity
Ritchie EG, Johnson CN (2009) Predator interactions, mesopredator release and biodiversity conservation. Ecol Lett 12:982–998
Ritchie EG, Elmhagen B, Glen AS et al (2012) Ecosystem restoration with teeth: what role for predators? Trends Ecol Evol 27:265–271
Roemer GW, Gompper ME, Van VB (2009) The ecological role of the mammalian mesocarnivore. Bioscience 59:165–173
Rosalino LM, do Rosário J, Santos-Reis M (2009) The role of habitat patches on mammalian diversity in cork oak agroforestry systems. Acta Oecologica 35:507–512
Rossa M, Lovari S, Ferretti F (2021) Spatiotemporal patterns of wolf, mesocarnivores and prey in a Mediterranean area. Behav Ecol Sociobiol 75:32
Rovero F, Zimmermann F (2016) Camera trapping for wildlife research
R Core Team (2020) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.r-project.org/
Santos-Reis M, Santos MJ, Lourenço S et al (2005) Relationships between stone martens, genets and cork oak woodlands in Portugal. In: Martens and Fishers (Martes) in Human-altered environments: an international perspective, pp 147–172
Sarmento PB, Cruz J, Eira C, Fonseca C (2011) Modeling the occupancy of sympatric carnivorans in a Mediterranean ecosystem. Eur J Wildl Res 57:119–131
Schmitt RJ, Holbrook SJ (1986) Seasonally fluctuating resources and temporal variability of interspecific competition. Oecologia 69:1–11
Schoener TW (1974) Resource partitioning in ecological communities. Science 185:27–39
Schuette P, Wagner AP, Wagner ME, Creel S (2013) Occupancy patterns and niche partitioning within a diverse carnivore community exposed to anthropogenic pressures. Biol Conserv 158:301–312
Smith JA, Thomas AC, Levi T et al (2018) Human activity reduces niche partitioning among three widespread mesocarnivores. Oikos 127:890–901
Stein A, Gerstner K, Kreft H (2014) Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scales. Ecol Lett 17:866–880
Suárez-Tangil BD, Rodríguez A (2022) Environmental filtering drives the assembly of mammal communities in a heterogeneous Mediterranean region. Ecol Appl 33:e2801. https://doi.org/10.1002/eap.2801
Tews AJ, Brose U, Grimm V et al (2004) Animal species diversity driven by habitat heterogeneity/diversity : the importance of keystone structures. J Biogeogr 31:79–92
Torretta E, Serafini M, Puopolo F, Schenone L (2016) Spatial and temporal adjustments allowing the coexistence among carnivores in Liguria (N-W Italy). Acta Ethol 19:123–132
Venter O, Sanderson EW, Magrach A et al (2016) Sixteen years of change in the global terrestrial human footprint and implications for biodiversity conservation. Nat Commun 7:1–11
Verdade LM, Rosalino LM, Gheler-costa C et al (2011) Adaptation of mesocarnivores (Mammalia: Carnivora) to agricultural landscapes in Mediterranean Europe and Southeastern Brazil: a trophic perspective. In: Rosalino LM, Gheler-Costa C (eds) Middle-sized carnivores in agricultural landscapes. Nova Science Publishers, New York, pp 1–38
Vilella M, Ferrandiz-Rovira M, Sayol F (2020) Coexistence of predators in time: Effects of season and prey availability on species activity within a Mediterranean carnivore guild Marc. Ecol Evol 10(20):11408–11422
Virgós E, Romero T, Mangas JG (2001) Factors determining “gaps” in the distribution of a small carnivore, the common genet (Genetta genetta), in central Spain. Can J Zool 79:1544–1551
Virgós E, Tellería JL, Santos T (2002) A comparison on the response to forest fragmentation by medium-sized Iberian carnivores in central Spain. Biodivers Conserv 11:1063–1079
Wait KR, Ricketts AM, Ahlers AA (2018) Land-use change structures carnivore communities in remaining tallgrass prairie. J Wildl Manag 82:1491–1502
Wang Y, Allen ML, Wilmers CC (2015) Mesopredator spatial and temporal responses to large predators and human development in the Santa Cruz Mountains of California. Biological Conservation 190:23–33. https://doi.org/10.1016/j.biocon.2015.05.007
Zabala J, Zuberogoitia I, Martínez-Climent JA (2009) Testing for niche segregation between two abundant carnivores using presence-only data. Folia Zool 58:385–395
Zhao G, Yang H, Xie B et al (2020) Spatio-temporal coexistence of sympatric mesocarnivores with a single apex carnivore in a fine-scale landscape. Glob Ecol Conserv 21:1–11
Zuur AF, Ieno EN, Smith GM (2007) Analysing ecological data. Springer, New York
Acknowledgements
We acknowledge Sara Aliácar and Raquel Martins who were involved in fieldwork and with the installation of the camera traps. PL was supported by a PhD grant (2022.12536.BD) and JC was supported by a research contract (CEECIND/01428/2018) from Fundação para a Ciência e a Tecnologia, I.P. We acknowledge the financial support to CESAM by FCT/MCTES (UIDP/50017/2020+UIDB/50017/2020+LA/P/0094/2020), through national funds.
Funding
Open access funding provided by FCT|FCCN (b-on). The work was supported by the LIFE WolFlux Project (LIFE17-NAT/PT/000554).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study. Study conception and methodology were performed by PL, FP, NN, CF, and JC. Data collection and processing were carried out by PL, MR, AC, and JC. Statistical analyses, figures, and charts were performed by PL. The writing and revision were carried out by PL, FP, NN, MR, AC, and JC. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests or personal relationships that could influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Linck, P., Palomares, F., Negrões, N. et al. Increasing homogeneity of Mediterranean landscapes limits the co-occurrence of mesocarnivores in space and time. Landsc Ecol 38, 3657–3673 (2023). https://doi.org/10.1007/s10980-023-01749-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-023-01749-0