Abstract
Context
In a conservation context, identifying key habitats suitable for reproduction, foraging, or survival is a useful tool, yet challenging for species with large geographic distributions and/or living in remote regions.
Objectives
The objective of this study is to identify selected habitats at multiple levels and scales of the threatened eastern North American population of golden eagles (Aquila chrysaetos). We studied habitat selection at three levels: landscape (second order of selection), foraging (third order of selection), and nesting (fourth order of selection).
Methods
Using tracking data from 30 adults and 366 nest coordinates spanning over a 1.5 million km2 area in remote boreal and Arctic regions, we modelled the three levels of habitat selection with resource selection functions using seven environmental features (aerial, topographical, and land cover). We then calculated the relative probability of selection in the study area to identify regions with higher probabilities of selection.
Results
Eagles selected more for terrain ruggedness index and relative elevation than land cover (i.e., forest cover, distance to water; mean difference in relative selection strength: 1.2 [0.71; 1.69], 95% CI) at all three levels. We also found that the relative probability of selection at all three levels was ~ 25% higher in the Arctic than in the boreal regions. Eagles breeding in the Arctic travelled shorter foraging distances with greater access to habitat with a high probability of selection than boreal eagles.
Conclusion
Here we found which aerial and topographical features were important for several of the eagles’ life cycle needs. We also identified important areas to monitor and preserve this threatened population. The next step is to quantify the quality of habitat by linking our multi-level, multi-scale approach to population demography and performance such as reproductive success.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Broad geographical species’ distributions are typical of highly mobile species, such as birds of prey, making the protection of essential habitat challenging (Ruth et al. 2003; Engler et al. 2017). Firstly, demographic and habitat surveys are often limited by data collection across a wide range, which is laborious and costly in remote regions (Mallory et al. 2018). Second, sectors and regions of importance for these species often span over large areas, making it difficult to protect enough habitat for all the species life cycle needs (i.e., reproduction, foraging, or survival). For example, birds of prey populations can be composed of nonmigrating (often females) and migrating individuals (males and immatures; Kjellén 1994; Maynard et al. 2022), creating a wider wintering range to protect. Indeed, habitat selection by organisms, i.e., the process of choosing the physical and biological characteristics in an area to use (Hall et al. 1997), occurs at different levels, and implicitly different spatial and temporal scales, and is driven by a variety of behaviour and life cycle needs. Studying multiple selection levels and scales can identify key habitat characteristics common to needs during multiple life cycles, a useful tool in the context of conservation and habitat protection (McGarigal et al. 2016; Bauder et al. 2018; Assandri et al. 2022).
The golden eagle is a highly mobile top predator (Johnson et al. 2022) with a vast breeding range separated into different populations (Katzner et al. 2020); some populations more vulnerable than others (Kochert et al. 2002). Key nesting and foraging habitats during breeding season have been identified in some well-studied populations (Marzluff et al. 1997; Singh et al. 2016; Squires et al. 2020). Golden eagles often establish their nests near a waterbody or stream (Menkens and Anderson 1987; Weber 2015), but water appears to be avoided while foraging (Singh et al. 2016). Low vegetation cover has been identified as a preferred foraging habitat component providing good hunting grounds (Marzluff et al. 1997; Singh et al. 2016; Squires et al. 2020), but nearby forest cover can also offer nesting sites (Katzner et al. 2020). Based on these relationships, Arctic regions may thus include good habitat for golden eagles considering the open landscape of tundra and taiga, which may increase prey detectability (Marzluff et al. 1997; Carrete et al. 2000). High-elevation cliffs are also preferred because they provide better nesting areas sheltered from predators and weather conditions (Tack and Fedy 2015; Weber 2015; Squires et al. 2020) as well as create updrafts to reduce energetic costs of flight during foraging (Bohrer et al. 2012; Katzner et al. 2012; Duerr et al. 2019a). Preferred selection for land cover features (e.g., forest cover, waterbodies) or aerial and topographical conditions (e.g., wind speed, terrain ruggedness index, elevation) by golden eagles may, thus, vary with the level of selection or geographically given the availability of these features within the population’s distribution. Using a multi-level, multi-scale approach to habitat selection in different regions of their range can help better understand regional habitat conservation needs and identify habitat characteristics important for several levels.
Although distributional modeling has been conducted effectively predicting the geographical range of golden eagles across eastern North America (first order of selection sensu Johnson 1980; McCabe et al. 2021), habitats selected over multiple levels for specific lifecycle needs (e.g., reproduction, second to fourth order of selection; Johnson 1980; Meyer and Thuiller 2006) have not been identified. In eastern North America, the golden eagle breeding population ranges over 14° in latitude, spanning several Canadian provinces and territories, and two different bioclimatic zones, i.e., Arctic and boreal. Although its IUCN species status is of Least Concern (BirdLife International 2021), the eastern North American population is considered has special conservation status in six U.S. states and two Canadian provinces; Katzner et al. 2012, Katzner et al. 2020) due to relatively low population numbers for the spatial extent covered (~ 5,000 eagles) relative to other golden eagle populations (western North American population: ~ 40,000 eagles; Kaztner et al. 2020). Studying this low-density population is particularly challenging, especially in remote northern regions of Canada (Mallory et al. 2018; Anctil et al. 2019). Although tracking studies and nesting surveys have recently accumulated key information on breeding ecology and population numbers, many areas are still understudied and data collected mainly comes from areas targeted by anthropogenic developments (Morneau et al. 2015; Équipe de rétablissement des oiseaux de proie du Québec 2020). Therefore, identifying and mapping key habitat characteristics at multiple levels for this population can provide important information for managers on where and how to efficiently protect essential habitat or focus monitoring efforts.
The objective of this study is to identify key breeding habitat characteristics across the breeding distribution of the threatened eastern North American population of golden eagles (hereafter ‘eagles’) within three Canadian provinces (Québec, Newfoundland and Labrador, and New Brunswick) by taking a multi-level, multi-scale habitat selection approach. We used eagle tracking data and breeding survey data to study habitat selection at three hierarchical levels of selection (sensu Johnson 1980; Meyer and Thuiller 2006): landscape (second order of selection), foraging (third order of selection), and nesting area (fourth order of selection). Because of the aerial nature of avian mobility and previously identified features that may benefit several levels of selection, we hypothesized (H1) that strength of selection between aerial/topographical features (i.e., relative elevation, terrain ruggedness index, northness, eastness, and wind speed) and land cover features (i.e., forest cover and distance to water) will be different across the three levels of selection. We predicted that eagles would show stronger selection for aerial and topographical features than for land cover across the three levels of selection. Given the wide latitudinal range of our study area, we also hypothesized that selection would differ between bioclimatic zones (i.e., Arctic vs. boreal; H2). Specifically, the Arctic bioclimatic zones would have more area covered by habitats of higher probability of selection then southern regions (i.e., boreal bioclimatic zone) because eagles seem to prefer low forest cover for foraging (Singh et al. 2016; Katzner et al. 2020) and semi-open taiga and open tundra prevail in Arctic regions compared to boreal regions. Our study aimed to identify breeding habitat characteristics common to multiple levels for a threatened population of golden eagles on an area encompassing more than 1.5 million km2 and three Canadian provinces (Québec, Newfoundland and Labrador, and New Brunswick). Identifying critical aerial and terrestrial characteristics at large spatial extent is essential to conservation of this highly mobile bird of prey (Nielson et al. 2016; Assandri et al. 2022) and could also benefit a whole spectrum of species living in the same habitats such as other cliff-nesting raptors (Sergio et al. 2006; Smits and Fernie 2013). Doing so, we also provide new information for management and conservation measures by identifying areas of high probability of selection, which can host undocumented potential nest sites.
Methods
Field work and data collection
Our data are comprised of two sets: telemetry data and nest coordinates. Telemetry data were retrieved from a partnership with two main contributors: the Ministère de l’Environnement, de la Lutte contre les Changements Climatiques, de la Faune et des Parcs, and the Eastern Golden Eagle Working Group. Telemetry units were deployed on golden eagles year-round between 2007 and 2021. The birds were captured with a bow net or a baited net launcher. The nets were set near the nest site to target the breeding eagles (n = 9) or on the wintering grounds and along the migration routes (n = 21). Upon capture, morphometric measurements (i.e., hallux claw length, culmen, wing chord, bill height, and length) and mass were taken to sex individuals following Bortolotti (1984) and Harmata and Montopoli (2013); in some cases, sex was confirmed with DNA from blood samples (Doyle et al. 2016). Telemetry units were attached with a body harness made of Teflon tape (Bally Ribbon Mills, Bally, PA, USA) in a backpack-style harness. Units weighted from 45 to 95 g and were always less than 3% of the body mass of all eagles. Telemetry units were built by Cellular Tracking Technologies (CTT-1100, Rio Grande, NJ, USA) and Microwave Telemetry (PT-100, Columbia, MD, USA) and recorded GPS coordinates (latitude and longitude; ± 18 m) every hour (Microwave Telemetry) or every 15 min (Cellular Tracking Technologies). Eagles were released at the capture site within an hour of handling.
Nest coordinates were retrieved from the Governement of Newfoundland and Labrador and SOS-POP database (SOS-POP 2018), the later collects information on populations of endangered species in Québec, Canada. Amateur and professional ornithologists contribute to this database, operated by QuébecOiseaux, in collaboration with the Ministère de l’Environnement, de la Lutte contre les changements climatiques, de la Faune et des Parcs, and the Canadian Wildlife Service. Nest coordinates (latitude and longitude) were mainly obtained over four decades (1980–2020s) from four different sources: helicopter or on-foot surveys conducted for environmental impact studies (Morneau et al. 2015), the breeding bird atlas (Robert et al. 2019), the Québec Government (McNicholl et al. 1996; Anctil et al. 2019), or by volunteer bird watchers, who reported their sightings.
In our habitat selection models, we tested seven environmental variables (Supplementary Material Table S1). Aerial and topographical features included terrain ruggedness index (change in elevation relative to the surrounding pixels; Riley et al. 1999), northness (− 1: south; 1 : north) and eastness (− 1: west; 1:east) of slope, average annual wind speed (Technical University of Denmark 2021), and relative elevation. Because eagles may select a location relative to its surrounding environmental characteristics, we calculated the relative elevation by subtracting the mean of elevation over a 10 km radius from the elevation at each pixel. Land cover features included forest cover (0 = open cover: tundra, taiga, agricultural fields, wetlands ; 1 = forest cover: deciduous, mixed or coniferous) and the distance of each presence location to the nearest water features (wetlands, rivers, lakes or sea). Details on the source and management of these data are found in the Supplementary Material.
Data management and analysis
We used 30 individuals during the breeding season (April to August) for a total of 81 bird-summers. We removed any locations that did not record at least a two-dimensional fix (latitude and longitude) and standardized the temporal resolution between individual tracks so that individuals may be compared among each other to reflect the population rather than a few individuals with significantly more data. To do so, we used locations only recorded during the first 15-min interval to represent each nominal hour. We removed location data recorded over open marine waters (> 1 km from the coast). Consecutive telemetry locations showing birds had to traveling at > 100 km/h speed were considered erroneous as eagles are unlikely to reach such high speeds of travel during the breeding season (Katzner et al. 2020).
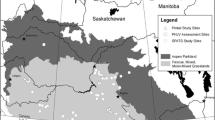
We estimated the behaviour (i.e., travelling or resting versus foraging) at each GPS location with the Residence in Space and Time method (RST) with a radius of 5 km following Torres et al. (2017) and Maynard et al. (2022). We chose this method for its flexibility with spatial scale and individuality of tracks given our unbalanced design (the number of locations recorded differs with birds). The RST method allowed us to discriminate three behaviours, i.e., transit (null values), area-restricted search (ARS; positive values), and rest (negative values; Torres et al. 2017). We were able to remove data from nonbreeders and presumed to be failed reproductive attempts and long-distance nonmigratory movements, i.e., those usually leading away from the nest and the home range (Poessel et al. 2022). We identified these movements with consecutive transit locations paired with an increase in the distance traveled between points (> twice the mean daily distance during the summer months) and general trajectories leading > 100 km away from nests following Miller (2012) and Maynard et al. (2022). Finally, we classified locations from both datasets (telemetry and nests) by bioclimatic zones covering the breeding distribution of eagles in eastern Canada (i.e., Arctic and boreal; Fig. 1).
Binned relative probability of selection of breeding golden eagles (n = 30 individuals) mapped for landscape, foraging, nest models, and all three levels combined in their eastern Canada breeding range. We indicate the abbreviated names and borders of Canadian provinces and territories (NB New Brunswick, NL Newfoundland, NU Nunavut, QC Québec). In the inserts, purple polygons represent a sample (n = 20 individuals) of 95% contours of existing home ranges
To model habitat selection of golden eagles, we chose a use-available design at three hierarchical levels (landscape, foraging, and nesting; Johnson et al. 2006). We standardized the grid resolution by first resampling environmental variable rasters (i.e., terrain ruggedness index, relative elevation, forest cover, distance to water, average annual wind speed, northness, and eastness) to the lowest resolution, i.e., a grid of 25 × 250 m cells. Next, we confirmed a lack of correlation (r < 0.7; Zuur et al. 2010) among environmental variables using the Pearson’s correlation coefficient (Supplementary Material Fig. S2). Our new raster grid meant that our used locations at any level needed to be at least 250 m away from each other to avoid more than one used location (from the same individual) within a cell to avoid spatial autocorrelation (Northrup et al. 2013) and duplicating the positive response from a cell (Guisan et al. 2017). We verified the lack of spatial autocorrelation with a semivariogram for all the environmental variables and the distance between the positive ARS locations (our dataset with the highest spatial and time resolution). We considered 250 m as a biologically significant resolution given the great mobility of the raptor while foraging, the time resolution of our units (1 h between recordings), and the spatial extent of the study.
Next, we overlaid the positive locations (nest and telemetry) with environmental variables and created available locations. For all three levels, we generated 10 available locations (pseudo-absences) for every used location following recommendations by Barbet-Massin et al. (2012) and Northrup et al. (2013) to create the most reliable distribution model. However, we could not reach a ratio of 1:10 for the foraging level given our spatial limitations (see details in Foraging level) and so we chose a used:available location ratio of 1:5. We fit both ratios to our two bioclimatic zones (i.e., Arctic and boreal), meaning that the number of available locations generated in one zone fitted the number of used locations in the same zone. Hereafter, we detail the data management employed to adjust our dataset and create available locations for all three levels.
Lansdcape level
To establish used habitat, we used telemetry data to estimate the breeding season home range of each bird summer with the 95% contour of a dynamic Brownian bridge kernel density estimator (Kranstauber et al. 2022). To established available habitat, we generated circular shapes randomly spaced across the study area that corresponded to average breeding season home range sizes (794 km2, 16 km radius; Supplementary Material Fig. S2). We clipped the available habitat with the coastal lines (> 1 km from the coast) as golden eagles are not expected to forage above open waters (Watson 2010; Katzner et al. 2020) and confirmed clipped available habitat were still within 95% of the average size of used home ranges (≥ 754 km2). Available habitat intercepting with either (1) used habitat or (2) known nest locations were removed and replaced by a newly generated available habitat to avoid false negatives. Finally, we merged all used home ranges by dissolving boundaries from the same individual across years into a single used home range per individual. We calculated the means of the environmental variables within the used and available habitat for each individual and available home ranges.
Foraging level
To define our used habitat, we selected ARS locations during daylight hours (~ 3 a.m. – 9 p.m. in the south and 2 a.m. to 11 p.m. in the north) from the telemetry data, previously identified with the RST method (see the Data management section). To define our available habitat, we limited our available locations within a buffer around the estimated location of the nest since eagles are central place foragers and are spatially limited in their movement (Watson 2010). We did not have all nest locations for all tracked eagles; therefore, we estimated the nest location by calculating the centroid of a 10% contour of our dynamic Brownian Bridge kernel density estimator. We calculated the accuracy of estimated nest locations with known nest locations, and centroids were less than 1.55 km from known nests (mean: 0.55 km [0.19; 0.91]). Next, we defined foraging area spatial extent based on a buffer of 35.71 km around our estimated nest locations (95% of the maximal distance of ARS locations from the estimated nest location). This distance represents the maximum distance eagles are expected to travel from their nest measured from the tracked animal, and thus me assumed reasonable to use for all individuals. We removed used locations outside the 35.71 km buffer to respect our comparative buffer within this level resulting in 37,884 used locations; Northrup et al. 2013). We clipped buffers around the nest to exclude areas overlapping open water (> 1 km from the coast). We then randomly generated available locations within the buffer. Finally, environmental predictors were associated with each used and available locations.
Nest level
For used nesting habitat, we first, we selected golden eagle nests that had been detected and confirmed at least once during the last 25 years (366 nests) and then calculated the distance to the closest neighbouring nest. When the nests were within 4.75 km of each other, we considered them within the same nesting territory and likely used by the same pair (Morneau et al. 2015; LeBeau et al. 2015; Weber 2015) resulting in 166 territories. For our analyses, we randomly selected one nest per nesting territory because nest locations within the same territory are not independent considering they are used and selected by the same pair, thus violating our model assumption of independence of data. However, we randomly selected a nest per territory for each model iteration (see data analysis below), allowing us to keep the whole dataset and represent an array of used nests. For available nesting habitat, we randomly generated locations throughout the monitored area of study, corresponding to the province of Québec and the southern part of Labrador only (Supplementary Material Fig. S2). The minimal distance between our available nesting locations was set to 4.75 km to respect nesting territory distances. To reduce the potential for false negatives, we also ensured that all available nests were at least 4.75 km from any used nest. Finally, environmental predictors were associated with each used and available nest sites.
Data analysis
Prior to running our models, we scaled environmental variables (predictors) using the z-score \(\left({x}_{i}- \stackrel{-}{x}\right)/SD\left(\stackrel{-}{\text{x}}\right)\) with the mean (\(\stackrel{-}{x}\)) and standard deviation (\(SD\)) for the entire region of the study to reduce biases and improve model performance (Zuur et al. 2013).
We used a resource selection function (RSF) to compare used-available habitat. Our RSFs were built with generalized linear models with a binomial distribution and a logit link function where used = 1 and available = 0. We adjusted the weights of each data point so that the sum of the weight of all available locations equaled the sum of the weight of used locations, as recommended by Barbet-Massin et al. (2012). For the foraging level, given repeated measurements for each individual, we ran a mixed model with individual ID included as a random attribute for slopes and intercept, after verifying with likelihood ratio test (Supplementary Material). Therefore, we could account for both individual variation and an unbalanced design (Muff et al. 2020). Environmental predictors were included as fixed effects. The nest model was the only model to include northness and eastness in addition to the other variables (Table 1).
To select the best models, we used a sequential selection approach with candidate models to avoid testing all possible models and by starting with previous knowledge of biologically significant variables (Arnold 2010). We used Akaike’s Information Criterion (AIC) to discriminate between candidate models (Arnold 2010; Table 1). We calculated the AIC, AIC weight, and Log likelihood of the null model, then sequentially added one parameter at a time. If the addition of one parameter did not improve the AIC value (ΔAIC < 2 per one parameter increase) and log likelihood showed little change, we discarded the most complex model and chose the most parsimonious. We continued to add parameters until the lowest AIC was reached. The model and AIC values are reported in Table 1.
We trained the selected models over 100 iterations with 75% of the used/available habitat locations sampled randomly and stratified among individuals. From there, we averaged the coefficients of the 100 iterations of the selected model to stabilize the inference (Banner and Higgs 2017). For each iteration, we validated the models with the remaining 25% for each iteration as the test dataset and calculated the Boyce index and the total R2 (sum of mixed and fix effects where it applies; Guisan et al. 2017; Boyce et al. 2002). The Boyce index is a calibrating index ranging from − 1 to 1, whereby a value closer to 0 indicates that the predictive capacity of the model is as performant as a random model (Boyce et al. 2002).
To answer our first hypothesis (comparison of selection strength between features), we calculated the relative selection strength and the relative probability of selection using the averaged coefficients of the fixed effects. The relative selection strength allows comparison of predictor coefficients within a model and corresponds to the exponential of the coefficient estimate (Avgar et al. 2017), which allowed us to answer our first hypothesis on the selection difference between terrestrial and land cover features. The relative probability of selection w(x) gives us an indication of how strong the selection is compared to other cells within the same level and w(x) was calculated as follows:
where \(\stackrel{-}{\beta }\)ixi are averaged coefficients of environmental predictors. We calculated the relative probability in the study area using our z-scored environmental predictors at 250 m, which allowed us to create a map of the relative probability of selection in eastern Canada and answer our second hypothesis on bioclimatic differences. For the landscape level, we created new rasters, where each pixel corresponded to the mean for each environmental predictor over a 16 km radius around the pixel to match the spatial reference between the model and the prediction. We created a map of combined levels to explore regions and habitats, which could be selected for at all three levels (therefore of greater interest to protect for eagles) by multiplying the relative probability of selection at each pixel (Johnson et al. 2004; Bauder et al. 2018). For easier interpretation, we reclassified the predicted relative probability of selection into five equally ranged bins from very low (1) to high (5) at each level.
Finally, we answered our first hypothesis, that eagles selected aerial and topographical features more strongly than land cover features, by comparing the mean difference in relative selection strength of the environmental predictors within a level. We answered our second hypothesis, i.e., that relative selection at all levels was higher in the Arctic versus the boreal areas, by comparing the mean relative probability of selection between bioclimatic zones (Arctic vs. boreal; Fig. 1). We also compared the average size of breeding season home ranges (mean area km2) between bioclimatic zones as an indicator of habitat (higher abundance of habitats with high probability of selection when eagles travel less far from the nest, thus smaller home range). We consider statistical effects to be significant when the 95% confidence interval (CI) of the mean did not include 0 or when the CI did not overlap between two levels of comparison. Results are presented as mean [95% CI]. All analyses were performed in R v. 4.1.3 (R Core Development Team 2022) and QGIS v. 3.22.1 (QGIS Development Team 2022).
Results
From 2007 to 2021, we tracked 30 adult eagles (12 females, 17 males, 1 unknown sex; Table 1) for a total of 81 bird-summers and 40,446 ARS locations. After processing and filtering, we used a total of 30 home ranges (from the tracked eagles) and 238 nesting locations from theQuébec breeding surveys and 31 locations from Newfoundland and Labrador.
For the landscape and nesting candidate models, many models had ΔAIC < 2 with one parameter increase between each model (Table 1). Adding land cover (forest and distance to water) and wind speed to the landscape model did not inform the model further and therefore were discarded to choose the most parsimonious model. The model includind all the predictors was the candidate model selected for both the foraging and nesting levels, but some predictors were not significant since their confidence interval (CI) crossed 0 (Tables 1 and 2). For the foraging level, the variance of the random slope ranged from 0.08 to 0.28 and the variance of the random intercept ranged from 0.10 to 0.48. For all our models, the average Boyce index was > 0 and ranged from 0.58 to 0.99 (Table 2) and average total R2 of iterations ranged from 0.20 to 0.59.
For all three levels of selection, the most important predictors were terrain ruggedness index and elevation. Terrain ruggedness index had a relative selection strength ranging from 2.03 to 5.00 (Table 2), the highest among predictors. Eagles selected (> 50% probability of selection) more rugged terrain, with terrain ruggedness index of over 50–187 m difference in elevation (~ 38° slopes; Fig. 1). For elevation, the eagles selected mean elevations < 248 m at the landscape level and an elevation of 55–500 m lower than the surrounding area at the nesting level (Fig. 1). Wind speed was included in the nesting and foraging levels; Table 1) best candidate models, but it was not a significant predictor. At the nesting level, slope orientation (northness and eastness) was a significant predictor, indicating eagle selected for south- and east-facing slopes. However, northness and eastness had relatively weaker slopes (relative selection strength < 1.5) compared to terrain ruggedness index and elevation.
Across all levels, land cover features (i.e., forest cover and distance to water) were absent (landscape) or had relatively weaker slopes (foraging and nesting levels; relative selection strength: 1.01–2.41; Table 2). When comparing the relative selection strength of all aerial and topographical features with land cover ones, the difference was not significant at the foraging level (0.23 [− 0.47; 1.17]) or the nesting level (0.24 [− 1.15; 1.63]; Table 2), but all land cover variables were absent at the landscape-level analysis (Table 1). However, when only looking at terrain ruggedness index and relative elevation, the two topographical features with the strongest slopes, the mean difference with land cover features was 1.2 [0.71; 1.69] on average. Forest cover, while absent from the landscape level, was negative at both the foraging and nesting levels, suggesting that eagles avoided forest (Table 2; Fig. 1). Distance to water was not significant at any level and was absent from the landscape model.
Maps and bioclimatic differences
The Gaspé Peninsula, northern New Brunswick, the north coast of the Saint Lawrence, the coast of Hudson Bay, and the northeast coast of Labrador were highlighted as regions of very high relative probability of selection for all levels (Fig. 2). The mean relative probability of selection was higher in the Arctic than in the boreal zone for all levels (mean difference: 0.93 [0.76; 1.11]). In fact, in all models, the mean relative probability of selection in the Arctic ranged from 3.44 [3.43; 3.45] (landscape) to 3.70 [3.69; 3.71] (foraging; medium-high), while in the boreal zone, the mean relative probability of selection ranged from 2.59 [2.58; 2.60] (foraging) to 2.70 [2.69; 2.71] (landscape; low-medium). During the breeding season, the size of the home ranges was smaller in the Arctic (477 [301; 653] km2) than in the boreal zone (1,014 [710; 1,318] km2). The mean environmental characteristics also differed by bioclimatic zones. Elevation (boreal: 397.44 m [397.33; 397.55]; Arctic: 288.79 m [288.71; 288.87]), distance to water (boreal: 784.73 m [784.34; 785.12]; Arctic: 558.66 m [558.38; 558.95) and forest cover (boreal: 80.70% [80.68; 80.72]; Arctic: 20.84% [20.82; 20.86) were lower on average in the Arctic than in boreal regions. The opposite was observed for terrain ruggedness index (boreal: 34.80 m [34.78; 34.81]; Arctic: 36.71 m [36.69; 36.73) where it was higher on average in the Arctic than in the boreal zone.
Discussion
Multi-level, multi-scale habitat selection modelling is arguably one of the best approaches to evaluate how species perceive and select environmental characteristics as the framework incorporates hierarchical ordering of habitat select and behavior mechanisms to meet several life cycle needs (Thompson and McGarigal 2002; Meyer and Thuiller 2006; McGarigal et al. 2016). Our models performed well in predicting habitat selection across three levels given the high Boyce indexes and indicated high importance of topographical features and differences in selection between bioclimate areas. The landscape level had several competing models. However, the predictors present in the competing models but absent in the selected one (wind speed, distance to water, and forest cover) were either weak or not significant in the other two levels, which suggests that these predictors are less important in general and uninformative for the landscape level. With these three models, we were able to identify habitat characteristics at levels meaningful for golden eagles’ foraging, and reproduction during the breeding season, which is important when selecting areas to protect and monitor (Bauder et al. 2018; Macdonald et al. 2018). Our study area also covered most of the summer breeding distribution of the eastern population of golden eagles, i.e., ~ 1.5 million km2, and identified areas of interest in under-monitored and difficult to survey regions.
Aerial and topographical features
Eagles strongly selected more rugged landscape and higher relative elevations compared to land cover features (i.e., forest cover and distance to water) as indicated by importance of terrain ruggedness index and relative elevation in top models. Our findings support the importance of topographical features for cliff nesting raptors (Booms et al. 2010; Galipeau et al. 2020) and hunting from a perch or while flying (Atuo and O’Connell 2017), including studies on golden eagles (Morneau et al. 1994; Weber 2015; Singh et al. 2016). Cliffs and steep terrain are used by eagles for perching (Duerr et al. 2019b), gaining flight altitude (Katzner et al. 2012), reducing the costs of flying by slope soaring (Duerr et al. 2012), or enabling different hunting strategies (Dekker 1985; Duerr et al. 2019a). Furthermore, rugged terrain could create semi-open habitats, increasing prey detectability within forests (Cramp and Simmons 1980; Brodeur and Morneau 1999). Additionally, when nesting on cliffs, rugged terrain may be important to limit terrestrial predators from entering eagle nests (Morneau et al. 1994; Weber 2015; Katzner et al. 2020).
Contrary to our expectations and other studies, eagles selected lower elevations relative to the surrounding environment at the landscape and nesting level. Eagles were expected to select higher nesting sites (Morneau et al. 1994; Weber 2015; Katzner et al. 2020) away from predators. However, the distribution of golden eagles in eastern Canada is primarily over the Canadian shield: a relatively flat landscape (highest peaks < 2,000 m; Natural Resource Canada 2016) eroded by glaciers over millennia (Canada 2011). High-quality nesting cliffs, with lower exposure to harsh weather conditions, are likely positioned at elevations relatively lower than the mean elevation of the surrounding areas (Morneau et al. 1994; Anctil et al. 2019). Eagles also selected for home ranges with lower average elevations. Higher elevations, e.g., mountain summits, may also not be good hunting areas because of lower prey density, stronger winds, or flatter topography (Hoover 2002; McIntyre and Schmidt 2012), driving selection for lower elevations.
Although eagles selected strongly for steep and rugged terrain at all levels, not all aerial and topographical features showed this strong difference with land cover features, i.e., northness, eastness and wind speed, suggesting that not all topographical characteristics are key habitat characteristics. Indeed, the eagles also showed weak selection for nest sites on south-facing and east-facing slopes. South-facing slopes can increase the chances of successful breeding by providing exposure to the sun’s heat and by reducing costs of thermoregulation for nestlings (Bradley et al. 1997; Burton 2006), especially in polar regions (Burton 2007; Landler et al. 2014; Galipeau et al. 2020). Morneau et al. (1994) found that more than 85% of nests along the east side of Hudson Bay faced south or southwest. In contrast, some studies indicate that northness is irrelevant to golden eagle nest site selection (Brodeur and Morneau 1999; Weber 2015) and our data tend to show only a slight preference for slope orientation. Several nests were located in crevices or had overhangs above, which would have limited the impacts of rain and thermoregulation costs (Anctil et al. 2019); therefore, benefits similar to south-facing slopes could already have been present. However, these data were not systematically documented in the database and could not be tested.
Finally, the average annual wind speed was included in the model for nesting and foraging, but the relationships were not significant. Eagles can use winds to increase foraging success (Collopy 1983) and reduce the energetic costs of certain hunting techniques (Bakaloudis 2010; Safi et al. 2013; Cecere et al. 2020) or launching from a perch (Duerr et al. 2019b). Nielson et al. (2016) found that eagles selected locations with higher wind speed, even though their analyses ran at a very large and coarse spatial scale. It is likely that the temporal and spatial scale of our study was too coarse to register the expected relationship with wind speed and even slope orientation. Other studies have used finer-scale metrics of wind speed (e.g., orographic lift) paired with higher tracking resolution of eagles.
Land cover features
While topographical features were important predictors at all three levels and presented a similar relationship among levels, land cover features (forest cover and distance to water) showed weak (forest cover) or no (distance to water) relation. Yet, our data still showed a significant selection for open habitats for foraging and nest locations. At the nesting level, selection for open cover is not surprising since eastern North American eagles are not tree-nesters (Katzner et al. 2020), At the foraging level, golden eagles are often associated with low forest cover in many regions (Marzluff et al. 1997; Tack and Fedy 2015; Singh et al. 2016) because an open landscape may increase prey detectability (Marzluff et al. 1997; Carrete et al. 2000). Eagles in the eastern North American population, however, are known to use forested areas in boreal and mixed temperate zones (Brodeur and Morneau 1999; Miller et al. 2017), but these studies did not model habitat selection for foraging. Strong relationships for land cover features may not be apparent in populations where multiple selection strategies are used (Maynard et al. 2021, 2022; Assandri et al. 2022). McCabe et al. (2021) showed that there were two contrasting selection patterns existed at the individual level, with some individuals selecting forested landscapes while others selected semi-open landscapes; yet, a neutral relationship existed at the population level. Although we accounted for individual variation at the foraging level, opposing relationships may still temper the estimates at a population scale (Maynard et al. 2021; McCabe et al. 2021). Our foraging models support the results of these studies in eastern Canada, in which eagles only weakly select for open areas (forest cover = 0), however, more individual-based studies on habitat use may show that these features are of certain importance to individual eagles.
Eagles did not select for habitats close to water features at any level. Some raptor species are known to nest near waterbodies (Galipeau et al. 2020) including other populations of the golden eagle (Menkens and Anderson 1987; Weber 2015). However, these golden eagle studies were descriptive and did not quantify selection nor discuss the potential correlation between cliffs and water features (cliffs created by water erosion over time). Despite the prevalence of waterbirds in the diet for the few studies on the eastern North American population (Spofford 1971; Brodeur and Morneau 1999), we did not find any significant foraging selection for waterbodies, suggesting that eagles may not forage extensively in coastal areas or wetlands. We could expect eagles to select habitats away from coastal areas or large waterbodies (Watson 2010) where other top predators like the bald eagle (Haliaeetus leucocephalus) might compete for resources (Joseph 1977; Watson et al. 2019), resulting in home ranges usually leading away from large waterbodies.
These results do not preclude the use of water features altogether. In fact, eagles can still use these habitats to forage, but the 1-h temporal resolution of the transmitter data may hide eagles that depredate waterbirds and transport the prey farther away for consumption. At a broad spatial grid, physical features often take over finer-scaled variables (Fortin and Dale 2005; Booms et al. 2010; Guisan et al. 2017). In our study, we had to trade-off spatial resolution for fine-scale information such as the edge of vegetation or water against semi-open habitats (Guisan et al. 2017). However, without very high temporal resolution, we may never be able to truly untangle direct use and selection of fine-scale land cover characteristics with GPS data. We suggest that future studies should monitor the diet of nesting golden eagles to provide a more complete picture of drivers of foraging habitat selection.
Bioclimatic zones
The Arctic zone had a higher relative probability of selection at all levels supporting our second hypothesis. We also tested differences in home range size and home ranges were smaller in the Arctic zone than in the boreal zone, indicating that eagles travel farther distances during the breeding season in the boreal zone. Longer travels may indicate poor habitat or prey conditions, forcing a longer and farther search for food. Despite prey densities declining poleward (Schemske et al. 2009), higher prey detectability in an open landscape such as tundra and semi-open taiga may provide enough opportunities for eagles to forage closer to the nest during the breeding season (Moss et al. 2014; Miller et al. 2017). However, topographical features could be the driving cause since they were slightly more favorable in the Arctic. While we cannot confirm if habitat suitability is better poleward, monitoring foraging or breeding success in the Arctic would help understand how suitability changes with bioclimatic zones and confirms the Arctic as an area of higher value for this vulnerable population.
While most of the study area indicated a relative probability of selection from very low to moderate for the species, northeast Labrador, the north shore of the Saint Lawrence River, the Hudson Bay coast, the Gaspé Peninsula and northern New Brunswick had a high relative probability of selection. Although some opportunistic nesting surveys have been conducted in eastern Labrador (Morneau et al. 2015), no avian-focused or systematic surveys have been reported to quantify the density of golden eagle nests and no protection measures are currently in place. Similarly, northern New Brunswick has never been officially surveyed for golden eagle nests, although some eagles nesting in the Gaspé Peninsula are known to wander in New Brunswick in late summer (MELCCFP, unpublished data). Additionally, nests reported on the north shore of the Saint Lawrence and the Hudson Bay coast come from environmental impact assessments for hydroelectric dams, mining, roads, or other development projects (Équipe de rétablissement des oiseaux de proie du Québec 2020) and could use increased monitoring to confirm the potential of this region. Our model predicts a high probability of selection in these three regions despite their low monitoring effort to follow the population or breeding outcome. Continue or increase monitoring could confirm the importance of these regions to eastern North American eagles.
Conclusion
We used a multi-level, multi-scale approach to identify habitat characteristics and areas with a high probability of selection across the vast geographical range of a threatened golden eagle (Aquila chrysaetos) population to be used as a tool to inform institutions in charge of conservation. Some regions identified as high probability of selection, such as the Gaspé Peninsula, are also the most anthropogenically disturbed (Équipe de rétablissement des oiseaux de proie du Québec 2005; St-Louis et al. 2015) and may benefit for protection of key habitats. Because eagles are top predators and have a large geographical distribution (Watson 2010), protecting their habitat may also benefit many other threatened and least concerned raptor species relying on the same habitats and areas in eastern Canada (the so-called “umbrella effect”; Sergio et al. 2006; Smits and Fernie 2013) such as the peregrine falcon (Falco peregrinus) and the gyrfalcon (F. rusticolus) that nest within eagle territories (Anctil et al. 2019).
Our models identified key habitat characteristics that can be integrated into monitoring and conservation activities for golden eagles in eastern Canada., especially in under-monitored regions, such as Labrador and northern New Brunswick. Management decisions are often based on landcover features for many species, but our results indicate golden eagles may benefit from shifting habitat focus to topographical features. While being of conservation concern, the eastern North American golden eagle population may be increasing (Farmer et al. 2008; Dumas et al. 2022) and identifying areas of high selection probability may help in monitoring population growth, or update estimates on current population numbers. However, a low relative probability of selection may not indicate true absence of breeding eagles, as they may still use these areas for other behaviours or life cycle needs (e.g., migration, wintering). Further investigation of habitat selection during these periods are needed and would require to quantify the quality of those habitats and areas by integrating our multi-level, multi-scale approach to population demography and performance measures (e.g., reproductive success, foraging success, survival).
Data availability
Additional information supporting this article has been uploaded as part of the electronic supplementary material. However, the golden eagles we study are both protected and persecuted, within the USA and globally. Nest locations are one of the primary sites of eagle persecution, and the raw biotelemetry data we analyzed show those nest locations. The Québec Government prohibits the release of sensitive wildlife data including locations of the golden eagle that is protected under the Act respecting threatened or vulnerable species (E-12.01). Consequently, we do not make those data publicly available. Data can be made available for research upon request to authors. Computer code will be made available upon acceptance of the manuscript.
References
Anctil A, Potvin-Leduc D, Lemaître J (2019) Suivi de l’aigle royal dans le Nord-du-Québec - Connaître la population pour mieux la protéger, 2018. Ministère De l’Environnement, de la lutte contre les changements climatiques. de la Faune et des Parcs, Gouvernement du Québec
Arnold TW (2010) Uninformative parameters and model selection using akaikes information criterion. J Wildl Manag 74:1175–1178
Assandri G, Cecere JG, Sarà M et al (2022) Context-dependent foraging habitat selection in a farmland raptor along an agricultural intensification gradient. Agric Ecosyst Environ. https://doi.org/10.1016/j.agee.2021.107782
Atuo FA, O’Connell TJ (2017) Spatial heterogeneity and scale-dependent habitat selection for two sympatric raptors in mixed-grass prairie. Ecol Evol 7:6559–6569
Avgar T, Lele SR, Keim JL, Boyce MS (2017) Relative selection strength: quantifying effect size in habitat- and step‐selection inference. Ecol Evol 7:5322–5330
Bakaloudis DE (2010) Hunting strategies and foraging performance of the short-toed eagle in the Dadia‐Lefkimi‐Soufli National Park, north‐east Greece. J Zool 281:168–174
Banner KM, Higgs MD (2017) Considerations for assessing model averaging of regression coefficients. Ecol Appl 27:78–93
Barbet-Massin M, Jiguet F, Albert CH, Thuiller W (2012) Selecting pseudo-absences for species distribution models: how, where and how many? Methods Ecol Evol 3:327–338
Bauder JM, Breininger DR, Bolt MR et al (2018) Multi-level, multi-scale habitat selection by a wide-ranging, federally threatened snake. Landsc Ecol 33:743–763
BirdLife International (2021) Aquila chrysaetos. The IUCN Red List of Threatened Species 2021: e.T22696060A202078899
Bohrer G, Brandes D, Mandel JT et al (2012) Estimating updraft velocity components over large spatial scales: contrasting migration strategies of golden eagles and Turkey vultures. Ecol Lett 15:96–103
Booms TL, Huettmann F, Schempf PF (2010) Gyrfalcon nest distribution in Alaska based on a predictive GIS model. Polar Biol 33:347–358
Boyce MS, Vernier PR, Nielsen SE, Schmiegelow FKA (2002) Evaluating resource selection functions. Ecol Model 157:281–300
Bortolotti GR (1984) Age and sex size variation in golden eagles. J Field Ornithol 55:54–66
Bradley M, Court G, Duncan T (1997) Influence of weather on breeding success of peregrine falcons in the arctic. The Auk 114:786–791. https://doi.org/10.2307/408930
Brodeur S, Morneau F (1999) Rapport sur la situation de l’aigle royal (Aquila chrysaetos) au Québec
Burton NHK (2006) Nest orientation and hatching success in the tree pipit Anthus trivialis. J Avian Biol 37:312–317. https://doi.org/10.1111/j.2006.0908-8857.03822.x
Burton NHK (2007) Intraspecific latitudinal variation in nest orientation among ground-nesting passerines: a Study using published data. The Condor 109:441–446. https://doi.org/10.1093/condor/109.2.441
Canada H (2011) Canadian Shield. The Canadian Encyclopedia
Carrete M, Sánchez-Zapata JA, Calvo JF (2000) Breeding densities and habitat attributes of golden eagles in Southeastern Spain. J Raptor Res 34:48–52
Cecere JG, De Pascalis F, Imperio S et al (2020) Inter-individual differences in foraging tactics of a colonial raptor: consistency, weather effects, and fitness correlates. Mov Ecol 8:28
Collopy MW (1983) Foraging behavior and success of golden eagles. Auk 100:747–749
R Core Development Team (2022) R: A language and environment for statistical computing
Cramp S, Simmons KEL (1980) Handbook of the birds of Europe, the Middle East and North Africa. Oxford University Press, New York
Dekker D (1985) Hunting behaviour of golden eagles, Aquila chrysaetos, migrating in southwestern Alberta. Can Field-Naturalist 993:383–385
Doyle JM, Katzner TE, Roemer GW et al (2016) Genetic structure and viability selection in the golden eagle (Aquila chrysaetos), a vagile raptor with a holarctic distribution. Conserv Genet 17:1307–1322
Duerr AE, Miller TA, Lanzone M et al (2012) Testing an emerging paradigm in migration ecology shows surprising differences in efficiency between flight modes. PLoS ONE 7:1–7
Duerr AE, Miller TA, Dunn L et al (2019a) Topographic drivers of flight altitude over large spatial and temporal scales. Auk 136:ukz002
Duerr AE, Braham MA, Miller TA et al (2019b) Roost- and perch-site selection by golden eagles (Aquila chrysaetos) in eastern North America. Wilson J Ornithol 131:310
Dumas P-A, Côté P, Lemaître J (2022) Analyse des tendances des populations d’oiseaux de proie diurnes du Québec. Ministère De l’Environnement, de la lutte contre les changements climatiques, de la Faune et des Parcs, Direction De l’expertise sur la faune terrestre, l’herpétofaune et l’avifaune. Service de la conservation de la biodiversité et des milieux humides, gouvernement du Québec, Québec, QC, Canada
Engler JO, Stiels D, Schidelko K et al (2017) Avian SDMs: current state, challenges, and opportunities. J Avian Biol 48:1483–1504
Équipe de rétablissement des oiseaux de proie du Québec (2005) Plan de rétablissement de l’aigle royal (Aquila chrysaetos) au Québec 2005–2010. Ministère des Ressources naturelles et de la Faune du Québec, Secteur Faune Québec
Équipe de rétablissement des oiseaux de proie du Québec (2020) Bilan du rétablissement de l’aigle royal (Aquila chrysaetos) pour la période 2005–2018. Ministère des Ressources naturelles et de la Faune du Québec, Secteur Faune Québec
Farmer CJ, Goodrich LJ, Ruelas IE, Smith JP (2008) Conservation Status of North America.s Birds of Prey. In: Bildstein KL, Smith JP, Ruelas I. E, Veit RR (eds) State of North America.s Birds of Prey. Nuttall Ornithological Club and American Ornithologists. Union Series in Ornithology No. 3, Cambridge, Massachusetts, and Washington, D.C., pp 303–420
Fortin M-J, Dale MRT (2005) Spatial Analysis: a Guide for Ecologists, 1st edn. Cambridge University Press
Galipeau P, Franke A, Leblond M, Bêty J (2020) Multi-scale selection models predict breeding habitat for two arctic-breeding raptor species. Arct Sci 6:24–40
Guisan A, Thuiller W, Zimmerman NE (2017) Habitat suitability and distribution models with applications in R. Cambridge University Press, Cambridge, UK
Hall LS, Krausman PR, Morrison ML (1997) The habitat concept and a plea for standard terminology. Wildl Soc Bull 25:173–182
Hoover S (2002) The response of red-tailed hawks and golden eagles to topographical features, weather, and abundance of a dominant prey species at the Altamont Pass Wind Resource Area, California: April 1999 - December 2000. Subcontractor Report NREL/SR-500-30868 76 pp
Johnson DH (1980) The comparison of usage and availability measurements for evaluating resource. Ecol Soc Am 61:65–71
Johnson CJ, Seip DR, Boyce MS (2004) A quantitative approach to conservation planning: using resource selection functions to map the distribution of mountain caribou at multiple spatial scales. J Appl Ecol 41:238–251
Johnson CJ, Nielsen SE, Merrill EH et al (2006) Resource selection functions based on use–availability data: theoretical motivation and evaluation methods. J Wildl Manag 70:347–357
Johnson DL, Henderson MT, Anderson DL et al (2022) Isotopic niche partitioning and individual specialization in an Arctic raptor guild. Oecologia 198:1073–1084
Joseph RA (1977) Behavior and age class structure of wintering northern bald eagles (Haliaeetus leucocephalus alascanus) in western Utah. Brigham Young University. MSc Thesis. 61 p
Katzner TE, Brandes D, Miller T et al (2012) Topography drives migratory flight altitude of golden eagles: implications for on-shore wind energy development. J Appl Ecol 49:1178–1186
Katzner TE, Kochert MN, Steenhof K et al (2020) Golden Eagle (Aquila chrysaetos). In: Rodewald PG, Keeney BK (eds) Birds of the World. Cornell Lab of Ornithology, Ithaca, NY
Kjéllen N (1994) Differences in age and sex ratio among migrating and wintering raptors in southern Sweden. Auk 111:274–284
Kochert MN, Steenhof K, Kociiert M (2002) Golden eagles in the US and Canada: Status, trends, and conservation challenges. J Raptor Res 36:32–40
Kranstauber B, Smolla M, Scharf A (2022) move: Visualizing and Analyzing Animal Track Data
Landler L, Jusino MA, Skelton J, Walters JR (2014) Global trends in woodpecker cavity entrance orientation: latitudinal and continental effects suggest regional climate influence. Acta Ornithologica 49:257–266. https://doi.org/10.3161/173484714X687145
LeBeau CW, Nielson RM, Hallingstad EC et al (2015) Daytime habitat selection by resident golden eagles (Aquila chrysaetos) in Southern Idaho, U.S.A. J Raptor Res 49:29–42
Macdonald DW, Bothwell HM, Hearn AJ et al (2018) Multi-scale habitat selection modeling identifies threats and conservation opportunities for the Sunda clouded leopard (Neofelis diardi). Biol Conserv 227:92–103
Mallory ML, Grant Gilchrist H, Janssen M et al (2018) Financial costs of conducting science in the Arctic: examples from seabird research. Arct Sci 4:624–633
Marzluff JM, Knick ST, Vekasy TKMS et al (1997) Spatial use and habitat selection of golden eagles in southwestern Idaho. Auk 114:673–687
Maynard LD, Gulka J, Jenkins E, Davoren GK (2021) Different individual-level responses of great black-backed gulls (Larus marinus) to shifting local prey availability. PLoS ONE 16:e0252561–e0252561
Maynard LD, Therrien J-F, Lemaître J et al (2022) Interannual consistency of migration phenology timing is season- and breeding region-specific in North American golden eagles. Ornithology. https://doi.org/10.1093/ornithology/ukac029
McCabe JD, Clare JD, Miller TA et al (2021) Resource selection functions based on hierarchical generalized additive models provide new insights into individual animal variation and species distributions. Ecography 44:1756–1768
McGarigal K, Wan HY, Zeller KA et al (2016) Multi-scale habitat selection modeling: a review and outlook. Landscape Ecol 31:1161–1175
McIntyre CL, Schmidt JH (2012) Ecological and environmental correlates of territory occupancy and breeding performance of migratory golden eagles Aquila chrysaetos in Interior Alaska. Ibis 154:124–135
McNicholl R, Desrosiers A, Faubert R (1996) Inventaire aérien du faucon pèlerin et de l’aigle royal en Gaspésie. Ministère de l’Environnement et de la Faune, Québec, QC, Canada
Menkens G, Anderson SH (1987) Nest site characteristics of a predominantly tree-nesting populations of golden eagles. 58:22–25
Meyer CB, Thuiller W (2006) Accuracy of resource selection functions across spatial scales. Divers Distrib 12:288–297
Miller TA (2012) Movement ecology of golden eagles (Aquila chrysaetos) in eastern North America. Pennsylvania State University
Miller TA, Brooks RP, Lanzone MJ et al (2017) Summer and winter space use and home range characteristics of golden eagles (Aquila chrysaetos) in eastern North America. The Condor 119:697–719
Morneau F, Brodeur S, Decarie R et al (1994) Abundance and distribution of nesting golden eagles in Hudson-Bay, Quebec. J Raptor Res 28:220–225
Morneau F, Tremblay JA, Todd C et al (2015) Known breeding distribution and abundance of golden eagles in Eastern North America. Northeastern Naturalist 22:236–247
Moss EHR, Hipkiss T, Ecke F et al (2014) Home-range size and examples of post-nesting movements for adult golden eagles (Aquila chrysaetos) in boreal Sweden. J Raptor Res 48:93–105
Muff S, Signer J, Fieberg J (2020) Accounting for individual-specific variation in habitat-selection studies: efficient estimation of mixed-effects models using bayesian or frequentist computation. J Anim Ecol 89:80–92
Nielson RM, Murphy RK, Millsap BA, Howe WH, Gardner G (2016) Modeling late-summer distribution of Golden Eagles (Aquila chrysaetos) in the western United States. PLoS ONE 11:e0159271
Northrup JM, Hooten MB, Anderson CR, Wittemyer G (2013) Practical guidance on characterizing availability in resource selection functions under a use-availability design. Ecology 94:1456–1463
Natural Resource Canada (2016) High resolution digital elevation model (HRDEM) - canElevation series. OpenCanada, Government of Canada. Accessible at: https://open.canada.ca/data/en/dataset/957782bf-847c-4644-a757-e383c0057995
Poessel SA, Woodbridge B, Smith BW et al (2022) Interpreting long-distance movements of non-migratory golden eagles: prospecting and nomadism? Ecosphere. https://doi.org/10.1002/ecs2.4072
QGIS Development Team (2022) QGIS Geographic Information System
Riley S, DeGloria SD, Elliot R (1999) A terrain ruggedness index that quantifies topographic heterogeneity. Intermt J Sci 5:23–27
Robert M, Hachey M-H, Lepage D, Couturier AR (2019) Deuxième Atlas des Oiseaux Nicheurs du Québec méridional. Regroupement Québec Oiseaux. Environnemental et Changement Climatique Canada et Oiseaux Canada, Montréal, Québec
Ruth JM, Petit DR, Sauer JR et al (2003) Science for avian conservation: priorities for the new millennium. Auk 120:204–211
Safi K, Kranstauber B, Weinzierl R et al (2013) Flying with the wind: scale dependency of speed and direction measurements in modelling wind support in avian flight. Mov Ecol 1:4
Schemske DW, Mittelbach GG, Cornell HV et al (2009) Is there a latitudinal gradient in the importance of biotic interactions? Annu Rev Ecol Evol Syst 40:245–269
Sergio F, Newton I, Marchesi L, Pedrini P (2006) Ecologically justified charisma: preservation of top predators delivers biodiversity conservation: top predators and biodiversity. J Appl Ecol 43:1049–1055
Singh NJ, Moss E, Hipkiss T et al (2016) Habitat selection by adult golden eagles Aquila chrysaetos during the breeding season and implications for wind farm establishment. Bird Study 63:233–240
Smits JEG, Fernie KJ (2013) Avian wildlife as sentinels of ecosystem health. Comp Immunol Microbiol Infect Dis 36:333–342
Spofford WR (1971) The breeding status of the golden eagle in the appalachians. Am Birds 25:3–7
Squires JR, Olson LE, Wallace ZP et al (2020) Resource selection of apex raptors: implications for siting energy development in sagebrush and prairie ecosystems. Ecosphere. https://doi.org/10.1002/ecs2.3204
St-Louis A, Gauthier I, Beaudet S et al (2015) L’EROP: 10 ans pour le rétablissement des oiseaux de proie Au Québec. Le Naturaliste Canadien 139:90–95
Tack JD, Fedy BC (2015) Landscapes for energy and wildlife: conservation prioritization for golden eagles across large spatial scales. PLoS ONE 10:1–18
Technical University of Denmark (2021) Global Wind Atlas 3.1, a free, web-based application developed, owned and operated by the Technical University of Denmark (DTU). https://globalwindatlas.info
Thompson CM, McGarigal K (2002) The influence of research scale on bald eagle habitat selection along the lower Hudson River, New York (USA). Landscape Ecol 17:659–586
Torres LG, Orben RA, Tolkova I, Thompson DR (2017) Classification of animal movement behavior through residence in space and time. PLoS ONE 12:1–18
Watson J (2010) The Golden Eagle. Yale University Press, London
Watson JW, Vekasy MS, Nelson JD, Orr MR (2019) Eagle visitation rates to carrion in a winter scavenging guild. J Wildl Manage 83:1735–1743
Weber SA (2015) Golden eagle nest site selection and habitat suitability. Dissertation, Texas State University
Zuur AF, Ieno EN, Elphick CS (2010) A protocol for data exploration to avoid common statistical problems. Methods Ecol Evol 1:3–14
Zuur AF, Hilbe JM, Ieno EN (2013) A beginner’s guide to generalized additive mixed models with R. Highland Statistics Ltd, Newburgh
Harmata A, Montopoli G (2013) Morphometric sex determination of north american golden eagles. JRR 47:108–116. https://doi.org/10.3356/JRR-12-28.1
Acknowledgements
We would like to acknowledge the contribution of many who made this study possible; in particular, we thank Adam Durocher at the Atlantic Canada Conservation Data Centre, David Hanni, Tennessee Wildlife Resource Agency, Christine Kelly and Sara Schweitzer at North Carolina Wildlife Resources Commission, and Mercedes Maddox, Eric Soehren, Carrie Threadgill, Daniel Toole, Frank Allen, and Mitchell Marks at Alabama Department of Conservation and Natural Resources (ADCNR). We also thank the biologist aids, wildlife technicians and laborers at the ADCNR Freedom Hills Wildlife Management Area (WMA), ADCNR Skyline WMA, Philippe Beaupré, Guillaume Tremblay, and Alexandre Anctil at the Ministère de l’Environnement, de la Lutte contre les changements climatiques, de la Faune et des Parcs du Québec (MELCCFP), West Virginia Department of Natural Resources, Tufts Wildlife Clinic, and Cellular Tracking Technologies, and Melissa Braham at Conservation Science Global for formatting the data. We thank the MELCCFP who provided telemetry data for 18 golden eagles (Source des données utilisées: Ministère de l’Environnement, de la Lutte contre les changements climatiques, de la Faune et des Parcs © Gouvernement du Québec) and members of the Eastern Golden Eagle Working Group who provided telemetry data for 10 golden eagles. Any use of trade, firm, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Funding
Field work and data collection were financed by: MECCLFP at the Gouvernement du Québec, from the Charles A. and Anne Morrow Lindbergh Foundation, the Virginia Department of Game and Inland Fisheries through a Federal Aid in Wildlife Restoration Grant from the U.S. Fish and Wildlife Service, the Friends of Talladega National Forest, the Tennessee Wildlife Resources Agency, the Alabama Department of Natural Resources, the Georgia Department of Natural Resources, North Carolina Wildlife Resources Commission through a Federal Aid in Wildlife Restoration Grant from the U.S. Fish and Wildlife Service, Allegheny Plateau Audubon Society. ADCNR monitoring efforts were made possible through Wildlife Restoration Funds through grant AL-W-48: Segments 30 through 34. L.D.M. was financed by the Canada Research Chair in Polar and Boreal Ecology, the Northern Scientific Training Program, Université de Moncton, MELCCFP Qc, Canadian Heritage PhD grants, New Brunswick Sciences, Technologies, Engineering, Mathematics et Social Innovations and an NSERC Alexander Graham Bell award. This project is part of a PhD supervised by N. L. and co-supervised by J. L. and J. F. T. at the Canada Research Chair in Polar and Boreal Ecology, Université de Moncton.
Author information
Authors and Affiliations
Contributions
LDM, JL, and NL conceived the study. LDM drafted the manuscript and analyzed the data with feedback from NL; J-FT, JL, and NL supervised the work and research. JL, TAM, TK, SS, RS, and JC collected and provided data and gave feedbacks on the study and the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The use of golden eagles for this research was approved by the West Virginia University Institutional Animal Care and Use Committee (IACUC): protocol #11-0304 for eagles in the eastern US, by the MELCCFP: CPA-FAUNE 20-08; CPA-FAUNE 19-05; CPA-FAUNE 18-05; CPA-FAUNE 2017-08; CPA-FAUNE 14-35.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This draft manuscript is distributed solely for purposes of scientific peer review. Its content is deliberative and predecisional, so it must not be disclosed or released by reviewers. Because the manuscript has not yet been approved for publication by the U.S. Geological Survey (USGS), it does not represent any official USGS finding or policy.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Maynard, L.D., Lemaître, J., Therrien, JF. et al. Key breeding habitats of threatened golden eagles across Eastern Canada identified using a multi-level, multi-scale habitat selection approach. Landsc Ecol 39, 91 (2024). https://doi.org/10.1007/s10980-024-01835-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10980-024-01835-x