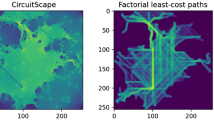
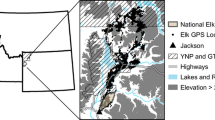
Abstract
Context
Maintaining connectivity is crucial for wildlife conservation in human-occupied landscapes. Structural connectivity modeling (SCM) attempts to quantify the degree to which physical features facilitate or impede movement of individuals and has been widely used to identify corridors, but its accuracy is rarely validated against empirical data.
Objectives
We evaluated SCM’s ability to identify suitable habitat and corridors for onagers (Equus hemionus onager) through a comparison with functional connectivity (i.e., actual movement of individuals) using satellite tracking data.
Methods
We used MaxEnt to predict suitable habitat and evaluated the ability of three SCM approaches: circuit theory, factorial least cost path, and landscape corridors approaches to identify corridors. The performance of the three SCM approaches was validated against independently collected GPS telemetry data.
Results
Onagers selected water sources and dense vegetation while avoiding areas grazed intensely by livestock. The three approaches to SCMs identified similar movement corridors, which were interrupted by roads, affecting major high-flow movement corridors. The SCMs overlapped with functional connectivity by about 21%.
Conclusion
Movement corridors derived from SCMs did not align with the locations or intensity of corridors identified using the functional connectivity model. This finding suggests that SCMs might have a tendency to overestimate landscape resistance in areas with low habitat suitability. Therefore, SCM may not adequately capture individual decisions about habitat selection and movement. To protect corridors linking suitable habitat, data on functional connectivity (i.e., telemetry data) can be coupled with SCM to better understand habitat selection and movements of populations as a consequence of landscape features.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Landscape connectivity is vital in safeguarding biodiversity by facilitating movement, resource access, and genetic exchange among populations (Crooks et al. 2011). The fragmentation of habitat poses a significant threat to connectivity, leading to habitat loss, increased extinction rates, reduced distribution and gene flow, ecological disruption, and edge effects (Rudnick et al. 2012; Synes et al. 2020; Liang et al. 2023). To mitigate these detrimental consequences, it is crucial to prioritize the maintenance of landscape connectivity (Kauffman et al. 2021). Connectivity allows individuals to access resources and avoid risks, and it promotes species survival through dispersal and migration, thereby mitigating inbreeding and extinction risks (Tischendorf and Fahrig 2000; Ranius et al. 2023). Disrupted connectivity leads to decreased gene flow and genetic diversity (Hedrick 2005), highlighting the need for conservation efforts to focus on landscape connectivity (Crooks et al. 2011; Kauffman et al. 2021). In the aftermath of extirpations stemming from habitat fragmentation, wildlife reintroductions (or “rewilding”) offers a potential means by which to restore community structure and ecosystem function (Donadio et al. 2022). The success of such programs requires a mechanistic understanding of how landscape connectivity facilitates gene flow, offsets genetic drift, and boosts population resilience to environmental changes (Fischer and Lindenmayer 2007; Ranius et al. 2023).
Landscape connectivity consists of two main components, structural connectivity (the physical characteristics of a landscape that allow for animal movement) and functional connectivity (the movements of individuals, populations, or genes across a landscape; Taylor et al. 1993; Taylor 2006; Rudnick et al. 2012). Structural connectivity is independent of species biology (Taylor 2006), whereas functional connectivity hinges on habitat preferences, dispersal ability, and other traits that interact with the structural features of the landscape (Rudnick et al. 2012). Structural connectivity modeling (SCM) is a suite of tools used to quantify the locations and intensity of use of potential corridors. This predictive framework quantifies the degree of connectivity among habitat patches by analyzing the sizes and spatial configuration of such patches across landscapes (Urban and Keitt 2001; Fortuna et al. 2008; Keeley et al. 2021). Through SCM, a suite of methods has been proposed to estimate landscape connectivity—each with distinct advantages and disadvantages—such that it often is essential to use several methods to accurately predict the locations and use intensity of corridors (Muratet et al. 2013; Rayfield et al. 2016; Marrotte et al. 2017). Specifically, data on habitat suitability, occupancy, and expert opinion frequently are combined to parameterize “resistance surfaces”, the degree to which landscape features impede or facilitate animal movement (Adriaensen et al. 2003; McGuire et al. 2016; Littlefield et al. 2017; Zeller et al. 2021). However, because SCM is based on habitat selection by individuals within populations, characteristics of movement corridors have only a small impact on the resulting model (Rudnick et al. 2012). As a result, erroneous predictions of functional (actual) corridors are likely, which can potentially mislead conservation efforts aimed at maintaining or restoring landscape connectivity (Rudnick et al. 2012). Until now, there have only been a few quantitative empirical studies directly comparing the ability of SCM to models based on actual movement data (e.g. genetic data: Mateo Sánchez et al. 2015a, b; GPS telemetry: Ziółkowska et al. 2016). Habitat suitability models can predict animal occurrences within their home ranges, although these models may not fully encompass the impact of environmental factors on animals when moving outside of their customary home ranges (Cushman et al. 2013). Because numerous studies have provided conservation implications based solely on SCM, this may result in ineffective safeguarding of movement corridors and, consequently, the failure of conservation programs. Only a single study (Ziółkowska et al. 2016) has compared the outcomes of SCM with telemetry data. This study utilized least-cost paths to predict brown bear (Ursus arctos) movement corridors, but telemetry data were employed for both habitat and movement models. In contrast, our study obtained onager occurrence locations independently of telemetry data.
Despite its widespread use, SCM has limitations in accurately predicting functional corridors due to its focus on habitat suitability rather than movement characteristics. Individuals select habitats based on food, thermal and security cover, and mating opportunities in attempt to maximize fitness (Molles and Sher 2019; Brown et al. 2023). Movement corridors link suitable habitats via dispersal, migration, or interpatch movements within a broader landscape (Beier and Loe 1992; Fingesi 2023). However, individuals may select corridors based on characteristics that are distinct from those used to select habitat (Trainor et al. 2013; Gastón et al. 2016; Keeley et al. 2016). For instance, food abundance or quality may not be prioritized when an animal moves among habitat patches across a landscape; instead, minimizing exposure to human activities may drive selection for corridors (Rezvani et al. 2020).Wide-ranging mammals and their habitats are affected by human population growth, exploitation of natural resources, direct persecution, and competition with humans and their livestock (Ripple et al. 2015). The situation is especially precarious for perissodactyls, with nearly 95% of this order classified as threatened or near-threatened (Bowyer et al. 2019; Kaczensky et al. 2015). As a family adapted to arid and semi-arid grasslands, equids in particular have declined in number and geographical range from competition with livestock and over-hunting (Feh et al. 2002; Neumann-Denzau and Denzau 2007). The Persian wild ass or onager (Equus hemionus onager) is an endangered subspecies of the Asiatic wild ass (Equus hemionus). In its native range, populations are confined to two protected areas in central Iran (Kaczensky et al. 2015). The largest onager population occurs in the 310 km2 Qatrouiyeh National Park (QNP), which is encompassed by the larger (3747 km2) Bahram-e-Goor Protected Area (BPA; Kaczensky et al. 2015). Because onagers have been extirpated across much of Iran, it is impractical to quantify functional (i.e., actual) connectivity across the majority of their historical range. Consequently, and by quantifying connectivity and associated barriers to movement based on physical characteristics of landscapes, SCM offers a potential solution to the challenge of identifying sites at which reintroductions have a high probability of success (i.e., self-sustaining populations). However, the utility of SCM for successful reintroductions hinges on the degree to which it can accurately identify actual movement corridors (i.e., functional connectivity).
We compared movement corridors predicted by SCM for onagers with actual movement corridors obtained through satellite tracking to inform reintroduction sites. Specifically, our objectives were threefold: (i) to identify suitable habitats and landscape connectivity for onagers using three approaches to SCM with different algorithms and natures: circuit theory, factorial least cost path, and landscape corridors (LSCorridor); (ii) to estimate functional connectivity of telemetered individuals using a Brownian bridge movement model; and (iii) to validate the three SCM approaches against functional connectivity in attempt to identify which approach (if any) holds the most utility for future reintroductions of onagers across their historical range.
Materials and methods
Study area
With an area of approximately 3747 km2, the BPA is located in the southern region of the Iranian plateau (28° 37′ to 29° 45′ N and 54° 25′ to 55° 16′ E; Fig. 1), surrounding the QNP. This protected area has an elevation range of 1580–2840 m, an average annual temperature of 15 °C, and an average annual precipitation of 150–250 mm. The region has a dry climate and includes desert steppe habitats with perennial shrubs and sparse trees such as wild almond (Prunus scoparia) and wild pistachio (Pistacia atlantica). The dominant plant species in the area are Artemisia sieberi, Zygophylllum eurypterum, Astragalus spp., and Noaea mucronata (Darvishsefat 2006). The QNP consists of three plains, Rig-Jamshid, Deh-Vazir and Einol-Jalal, partially separated by a mountain range. QNP and BPA are home to an increasing population of onagers (ca. 1050 individuals; Iranian Department of Environment 2022), which frequently comes into conflict with pastoralists and farmers in BPA (Esmaeili et al. 2019). Semi-nomadic pastoralists herd their livestock through a grazing lease schedule in BPA throughout the year, but livestock is prohibited within QNP; BPA is a multi-use area in which livestock grazing is permitted. Additionally, the expansion of onagers’ home ranges and their movement from QNP to BPA in the wet season brings them into close proximity with livestock, which may affect onager activity and resource selection (Esmaeili 2020). Over the past decade, a combination of increased enforcement and resource supplementation (water and food additions) have led to a 2.61% increase in the onager population within QNP, leading to increased human-onager conflict in and around BPA (Esmaeili et al. 2019; Mohammadi et al. 2021). Such conflicts are exacerbated by water shortages and high grazing pressure in BPA, highlighting the need for translocations from QNP as part of broader efforts to reintroduce onagers to large swaths of their historical range.
Modeling onager distribution and landscape connectivity
To model the distribution and landscape connectivity for onagers, we used a combination of species occurrence data and telemetry data. From 2012 to 2020, we collected occurrence data by visually identifying onagers and recording presence using a Global Positioning System (GPS; n = 234). To reduce spatial autocorrelation and bias, we performed spatial filtering of our data using the radius of the mean home range (Jennings et al. 2010; Kramer‐Schadt et al. 2013). We calculated the radius of the mean home range based on the home range size reported by Esmaeili (10–12 km2; 2020). Lastly, we used Moran's index to verify the absence of spatial autocorrelation in the spatially-filtered data (Fig. S1). Ultimately, we used 97 locations to model the distribution of onagers within QNP and BPA.
In three attempts, we captured 15 onagers using two baited corral traps with remotely triggered doors in December 2016 and January 2017 in QNP. In each attempt, three mares (total n = 9) were selected from inside the corrals and fitted with GPS collars (Vertex Lite 2 Iridium, Vectronic Aerospace GmbH, Berlin, and Germany) after being anesthetized with remote darting. Males and juveniles were released. As onagers exhibit fission–fusion herds—i.e., collared individuals do not stay with a specific herd and move between herds—(Kaczensky et al. 2008), we assumed that these mares would represent different herds during the telemetry period. We programmed collars to record locations every 2 h. To minimize the impact of capture and handling on animal movement, we excluded the initial 2 weeks of GPS locations after collar deployment (Dechen et al. 2012). We improved accuracy by screening GPS locations with a dilution of precision (DOP) greater than 10, resulting in ca. 72,000 locations from January 2017 to December 2018. We used these data to validate movement corridors identified through SCM. We followed the approved protocol for capture and telemetry outlined by the Iranian Department of Environment.
Environmental variables
Initially, we extracted climatic variables from the CHELSA database at a resolution of approximately 30 s (https://chelsa-climate.org). To downscale the annual mean temperature and annual precipitation from coarse-to-fine scale, we employed moving window regression. Elevation was used as a covariate to create downscaled bioclimatic variables. We assessed model performance by comparing model outcomes with observational data obtained from climatological and synoptic stations (obtained from the National Meteorological Organization of Iran). Linear regression yielded the best performance and highest accuracy in downscaling annual precipitation and annual mean temperature. The R2 values represent the amount of variability captured by the best fit model as calculated by the ratio of the cross-validated residual sum of squares to the total sum of squares. Climatic variables were then corrected based on the regression model. Next, we extracted the values of all 19 climatic variables and 1000 pseudo-absence points, and calculated the degree of collinearity between them using variance inflation factors (VIF) in R with the usdm package (Naimi 2013). Based on a VIF < 6, we selected seven climatic variables for distribution modeling: annual mean temperature, precipitation of driest month, precipitation seasonality, precipitation of warmest quarter, temperature seasonality, temperature annual range, and mean temperature of driest quarter. In addition to the aforementioned climatic variables, we used 16 additional environmental variables that collectively represented spatial variation in topography, land cover, and human threats (Table S1). We obtained a digital elevation model with a resolution of 30 m from the USGS database (https://earthexplorer.usgs.gov) to extract slope, aspect, and roughness using the surface analysis tool in ArcGIS 10.3 (ESRI 2014). The Euclidian distance to mountains was calculated according to the global mountains layer (Karagulle et al. 2017), which was also acquired from the USGS using the surface analysis tool in ArcGIS 10.3. The roughness variable was calculated using a digital elevation model, vector of ruggedness index (Sappington et al. 2007), and a 3 × 3 moving window. We obtained a soil type layer from the Geological Survey and Mineral Exploration of Iran. Playas were extracted from this layer, and the distance to playas was calculated through the surface analysis tool.
The Modified Soil-Adjusted Vegetation Index (MSAVI) of the study area was generated using MODIS satellite images (mean MSAVI values at a spatial resolution of 250 × 250 m and a temporal resolution of eight days, spanning from 2012 to 2020). Land cover variables, including bareground, poor-quality rangelands (sparse vegetation with ≤ 25% canopy cover), and medium-quality rangelands (mixture of scrubland and grassland with ≥ 25% canopy cover), were obtained from Iranian Natural Resources and Watershed Management Organization based on the total vegetation biomass in the study area.
The locations of water sources were recorded during field surveys using a hand-held GPS unit and marked on a 1/250,000 topography map. These data were subsequently included in the final model as the distance to the nearest water source. Human settlements, agricultural lands, and paved roads were extracted using the land use map acquired from Ghorbanian et al. (2020). The layers were then updated using Google Earth images, and the distance to each land use/land cover variable was calculated using the surface analysis tool in ArcGIS 10.3.
Only paved roads were included in the habitat suitability modeling process, as dirt roads have less traffic and the onagers regularly cross them. To investigate impacts of livestock within our study area, we acquired the land allotments layer from the Provincial Natural Resources Departments of Fars, Yazd, and Kerman provinces. The livestock capacity index (the amount of forage required by one mature sheep for one month) was determined using animal unit month (AUM) provided by Mesdaghi (2010), and based on the condition of the rangeland and the mean annual precipitation. We considered that the grazing period was approximately the same in grazing leases in this area (mean = 152; SD = 7.3); thus, the length of the grazing period was considered uniform for all pastures. Finally, the grazing intensity raster was calculated by multiplying the livestock capacity index by the livestock density at each allotment.
We followed the four-step method employed by Li et al. (2020) to reduce the number of variables with high correlations used to improve the transferability of the habitat suitability model. This approach involved selecting variables based on their percentage contribution to habitat suitability modeling (Fig. S2). However, in cases where predictor variables are correlated, interpreting the contribution of each variable can be challenging (Phillips et al. 2006; Li et al. 2020). Therefore, we excluded variables exhibiting strong collinearity with other variables before examining their percentage of contribution (Fig. S2). This resulted in the retention of 10 variables for modeling onager distribution: digital elevation model (DEM), roughness (ROU), distance to mountains (MOU), distance to bareground (BA), distance to medium rangeland (MR), distance to poor rangeland (PR), distance to the nearest water sources (WS), grazing intensity (INT-LIV), distance to playa (PL), and distance to paved road (PPR).
Habitat suitability modeling
We evaluated the habitat suitability of QNP and BPA for onagers using the maximum entropy algorithm (MaxEnt desktop version 3.3.3k; Phillips et al. 2006), which has been shown to outperform other habitat suitability models such as BioMapper, DOMAIN, FloraMap, and the genetic algorithm GARP (Elith et al. 2006; Ortega-Huerta and Peterson 2008). The MaxEnt algorithm requires presence data of the modeled species and relevant environmental and climatic variables. To evaluate the model's performance, we used a repeated split-sample test (10 times) with training models that were created by eliminating each fold in turn, and measured accuracy with the AUC (area under the receiver operating characteristic curve) and TSS (true skill statistic) metrics. We employed cross-validation in each replicate run and performed 500 iterations, while setting the regularization number to 1 to avoid overfitting of the test data (Phillips et al. 2004).
Structural connectivity modeling
We investigated the connectivity among suitable patches of habitat for onagers using circuit theory, factorial least cost path, and landscape corridors (LSCorridor). The objective was to gain a better understanding of the differences and similarities between these approaches, as outlined below (Table S2):
-
(A)
Circuit theory, which employs the CIRCUITSCAPE (McRae and Shah 2009) software to follow multiple paths taken by individuals. Circuit theory is a movement ecology concept that uses circuit analysis and random walk theory to simulate species movement in a landscape as a conductive surface. The landscape is represented as a network with nodes and resistors, where nodes are specific locations and resistors represent resistance between them (McRae et al. 2008). Resistance distance measures the ease of movement between nodes based on landscape characteristics. By assigning conductivity values to locations and predicting movement using random walks, circuit theory predicts the likelihood of individual movement between habitat patches (Hanks and Hooten 2013). To do so, we transformed the study landscape into a resistant surface using the habitat suitability map, where patches of suitable habitat served as focal nodes.
-
(B)
The resistant kernel approach (Compton et al. 2007), which identifies habitat cores through factorial least-cost path analysis (Landguth et al. 2012) implemented in UNICOR (UNIversal CORridor and network simulation model, Landguth et al. 2010). Our analysis assumed that individual movement between patches would occur along optimal or least-cost paths. To estimate the shortest path from each presence point to the neighboring point (Cushman et al. 2013), we employed Dijkstra’s algorithm (Dijkstra 2022). We converted the suitability map into a resistance map using a negative exponential function and used costs as the dispersal function while considering presence points as sources to predict the primary kernels in the UNICOR model. We generated a connectivity map by combining the least-cost paths using factorial least-cost path analysis (Cushman et al. 2009, 2013).
-
(C)
LSCorridors, which identifies movement corridors between two patches using a “least cost path” approach. This approach is similar to the “least cost corridor” (LCC) method (Adriaensen et al. 2003; Pinto et al. 2012), but LSCorridors identifies the cumulative cost instead (Correa Ayram et al. 2016). Patches were treated as collections of pixels and one pixel per patch was randomly selected in each simulation. This approach also allowed us to assign different resistance costs to pixels in the same patch, accounting for variation due to edge effects or habitat quality in corridor design. We used habitat patches and the resistance layer as modeling inputs (Keeley et al. 2016).
Functional connectivity modeling
Brownian Bridge Models (BBM; Horne et al. 2007) determine high-use movement corridors and core areas—i.e. areas with optimal conditions for a species to survive and reproduce—at the population level. We utilized the Brownian bridge movement model to estimate utilization distributions (UDs) for each of the nine collared female onagers during the study period, following the methods of Horne et al. (2007) and Sawyer et al. (2009). Our calculations employed a grid-cell size of 30 m and incorporated a spatial error of 10 m. The 99% contour polygons of individual-specific UDs were used to quantify the number of overlapping polygons in each grid cell situated within our study area, per the approach outlined by Sawyer et al. (2019) (Fig. 2).
To evaluate the conservation value of the derived movement corridors, we initially converted the output maps of structural connectivity models and functional corridors into binary maps using the 20th percentile of the total current density. Subsequently, these maps were overlaid to determine overlap percentages across all four approaches.
Results
Habitat suitability modeling
The AUC and TSS values for the habitat suitability model were both high (AUC = 0.91; TSS = 0.80), indicating good performance. Habitat suitability (i.e., onager presence probability) was positively correlated with medium-quality rangeland (7.3%) and low grazing intensity (4.2%) by livestock, while it was negatively correlated with distance from water sources (68.9%), mountains (4.7%), and playas (4.4%). The habitat suitability map demonstrates that QNP houses most of the primary suitable habitat in the study area (Fig. 3). Within the central regions of BPA, there exist isolated patches of high suitability. Moreover, a significant patch displaying good suitability occurred in the northwestern part of the region, outside the BPA, which is frequented by onagers during specific periods. Furthermore, there are areas in the southwestern portion of QNP that extend beyond its designated boundaries and remain suitable.
Structural connectivity modeling
The results from the SCMs demonstrated the spatial continuity of onagers’ habitat in BPA (Fig. 4). The circuit model revealed that the eastern region of QNP had the highest probability of onager movement, where high flow density was observed (the route connecting patch 2 to 10, Fig. 4a). Additionally, a relatively wide band with high flow was found in the route connecting patches 1 to 3, which extended from the north of the area to the central areas of BPA (Fig. 4a). The LSCorridors and UNICOR models produced similar results as the circuit model (see Fig. 4b, c). Furthermore, the map obtained from UNICOR identified corridor density and main patches, which closely resembled those acquired from the habitat suitability classes map. The overlap between the habitat continuity maps and the roads map revealed that the northern corridors (Fig. 4a) were bisected by a side road.
Map of the flow intensity of onagers in BPA according to circuit theory (a), LSCorridor (b), and UNICOR (c). Map (d) shows the location of high-use routes (used by > 1 onager) and high-use core areas (used by at least four onagers) resulted from the BBM in 2017 and 2018. Map (e) shows the habitats with high conservation values, which are derived from the combination of habitat suitability map, SCM, and functional connectivity modeling
Functional connectivity modeling
Cell values resulting from the analysis varied from 1 (used by only one animal) to 9 (used by all nine animals), representing low-use and high-use areas, respectively. We defined low-use movement corridors as cells used by one to three animals (> 10% of the collared individuals) and designated high-use core areas as cells used by at least four animals (> 45% of the collared individuals; see Fig. 4d).
The comparison of connectivity outputs revealed that the circuit model overlapped with LSCorridors, UNICOR, and BBM by 82.5%, 48.4%, and 20%, respectively. The LSCorridors showed the highest overlap with UNICOR, with 57% of the common route. On average, the telemetry-based functional connectivity model (BBM) had a 21.4% overlap with SCMs (Table 1). The most substantial overlap of the SCMs occurs in the north-northeast movement routes. The highest overlap of SCMs and the functional connectivity model is observed in the northern and northeastern parts of QNP, as well as the northern areas of BPA. However, it shows the least resemblance to the telemetry data in QNP and its western areas. Figure 4e shows that areas with the highest connectivity overlap have higher conservation value in QNP.
Discussion
Habitat suitability
We examined habitat suitability, core areas, and landscape connectivity for onagers in QNP and BPA using SCM (based on species presence) and functional connectivity (based on data from GPS telemetered individuals). Our study revealed that onagers prefer rangelands of moderate quality (Nowzari et al. 2013; St-Louis and Côté 2014) while actively avoiding areas with intense livestock grazing and proximity to livestock allotments. The presence of livestock at nearly all water-accessible sites compromises onagers’ access to forage and water (Esmaeili 2020), and illegal livestock intrusion into BPA compounds this issue. Illegal livestock encroachment in BPA (but not QNP) could be exacerbated by unstable economic conditions, which could further limit food and water for onagers. Elsewhere, and especially in dry areas across the globe, forage quality constrains habitat suitability and use for wild ungulates in rangelands (Nowzari et al. 2013; Schroeder et al. 2013; St-Louis and Côté 2014; Fynn et al. 2016; Du Toit et al. 2017).
Proximity to mountains further shaped habitat suitability, as hill-valley habitats provide a refuge and thermal cover during severe weather conditions and disturbances (Nowzari et al. 2013). Consistently, we observed that onagers flee towards hill-valleys when disturbed. Lastly, the existence of playa landforms, especially in the southern and southwestern parts of the BPA, plays a critical role in meeting the water requirements of onagers. In areas characterized by high groundwater levels (such as playas), Mongolian kulans can dig up to 60 cm in dry soil to access water (Feh et al. 2002; Stubbe et al. 2007). Consequently, the presence of these landforms is paramount to conserving and safeguarding water resources in the region.
Landscape connectivity
We employed four approaches—three of which generated structural connectivity metrics, one of which generated a functional connectivity metric—to investigate landscape connectivity and identify the most frequent movement corridors and primary habitats at the population level. These tools are widely used to identify conservation corridors (Correa Ayram et al. 2016). Connectivity models like LSCorridors, UNICOR, and circuit theory are effective for identifying optimal routes for individual movement while accounting for landscape structure and habitat parameters (Beier et al. 2008; McRae et al. 2008; Landguth et al. 2012; Ribeiro et al. 2017). Each model generates outputs that complement the others. LSCorridors avoid pixel size bias, and predicts multiple paths based on different pixel sets (Beier et al. 2008). UNICOR focuses on the validity of main habitat cores (Landguth et al. 2012), while circuit models and UNICOR both consider the contribution of each patch to overall connectivity (McRae et al. 2008; Ribeiro et al. 2017). Nevertheless, models that incorporate animal behavior are likely to generate predictions that more accurately reflect movement corridors across seasons, thereby providing valuable insights for targeted conservation strategies. For instance, and in our study, the three SCMs for predicting movement corridors generated similar predictions, but GPS relocations of onagers did not support certain pathways due to distinct characteristics of the models employed. Improving the accuracy of SCMs thus requires a better understanding of how movement corridors are selected by individuals within a population (Rudnick et al. 2012).
Telemetry data provides insights into the temporal dynamics of animal dispersal and other movements, which are fundamental for understanding demography, species interactions, and the establishment of new populations (Kays et al. 2015). In our study, telemetry data revealed that sites occupied by onagers and livestock are temporally separated, as onagers tend to avoid livestock (Esmaeili 2020). This finding is consistent with the results from MaxEnt modeling, which showed a negative influence of livestock density on onager habitat suitability. Our telemetry data further refined this result by revealing the causes of avoidance of livestock by onagers: disturbance (risk of foraging during the day when livestock is present) and the reduction of forage biomass by livestock (Esmaeili 2020), factors that were not identifiable by the structural model.
Our study revealed that, in areas with low habitat suitability, SCMs significantly overestimated resistance relative to the BBM. The SCMs produced movement corridors with much greater effective distances and absolute resistances than the functional connectivity model. These results are in accordance with the study of Ziółkowska et al. (2016) that compared structural and telemetry-based functional connectivity models. Predicted movement corridors from SCMs overlapped the telemetry-based functional connectivity model by only about 21%, indicating that animals frequently do not follow the routes generated by SCMs. Furthermore, the BBM (which was generated using data from nine collared onagers), did not represent the movements of the entire population. Consequently, low overlap between the BBM and SCMs may be partly due to the small number of telemetered individuals. Nonetheless, this limitation is unlikely to cause serious bias in the results due to the gregariousness and fission–fusion social structure of onagers. Additionally, this small sample size still identified important areas and movement corridors used by onagers that were not detected by the SCMs.
It is worth noting that SCMs for different species and habitats can vary in their predictive performance (Baguette et al. 2013). Moreover, these models are generally unable to account for animal behaviors like dispersal, migration, and mating excursions, or demographic events like mortality (Sawyer et al. 2011; Baguette et al. 2013; Mateo Sánchez et al. 2015a; Diniz et al. 2020; Hofmann et al. 2023). As such, they are more appropriate for species with low rates of dispersal that require access to food and other resources during movement. In contrast, onagers are able to move long distances across open habitats without water and food. Consequently, they do not necessarily follow the routes predicted by SCMs.
Habitat suitability models are blind to the density of individuals at a point, while the analysis of telemetry data determines the percentage of use per unit area. Therefore, the map obtained from telemetry data differentiates between areas with high, medium, and low suitability with greater accuracy. Habitat suitability indicates potential structural corridors, while tracking data often reveals that the real (functional) movement routes may not necessarily be located in the desirable habitats. Telemetry data accurately provides spatial records of individual animal movement corridors and dispersal, enabling a direct assessment of the influence of landscape features on these corridors. They hence greatly improve the credibility of habitat suitability models and our understanding of animal movements and dispersal (Manly et al. 2007; Kays et al. 2015). Genetic data may provide additional insights into the connectivity of populations (Beier et al. 2008; Zeller et al. 2021; Mateo Sánchez et al. 2015b; Nakajima et al. 2023).
Onagers exhibit nomadic movements characterized by irregular directionality and timing, leading to temporal variability in their movement patterns (Teitelbaum and Mueller 2019). Such movements are unlikely to be accurately captured by occurrence data derived from static sampling methods, potentially leading to misrepresentation of movement corridors in SCMs. Conversely, telemetry data collected over extended periods provide a more dynamic (and thus more realistic) depiction of the actual paths of onagers, reflecting true habitat use. Consequently, the absence of a species from a particular habitat does not preclude its ability to traverse that area in response to environmental variability, such as during dispersal, migration, or nomadic movements.
Our findings demonstrate that the outcomes of SCMs are highly sensitive to factors determining landscape resistance. While differences in the SCM approaches that we employed generated similar potential movement corridors, the low overlap of commonly-identified routes among approaches suggests that different corridors are key to conservation. It is therefore challenging to prioritize the most critical areas for protection based solely on SCMs. Moreover, movement corridors predicted by telemetry data exhibited minimal overlap with those derived from SCMs. Consequently, and compared to SCMs, we conclude that functional connectivity models derived from telemetry data facilitate a more precise and realistic assessment of movement corridors. Nonetheless, a blend of both telemetry data and SCM-derived resistance levels in tandem should permit more comprehensive understanding of spatiotemporal movement patterns of onagers, and wildlife more generally. Our integrative approach enables effective prioritization and management of wildlife habitats.
To ensure the protection of movement corridors and effective planning of conservation actions, our results hold profound implications. Specifically, our study shows that habitat suitability alone is unlikely to adequately predict movement corridors for highly mobile species. Therefore, we propose prioritizing connectivity analyses that take into account species-specific movement behaviors, rather than relying solely on habitat suitability. Nonetheless, it is worth noting that habitat suitability models and SCMs remain valuable tools in pinpointing suitable patches for resident populations and corridors for the relatively slow, non-migratory movements of the species between them. Furthermore, in situations where conducting telemetry studies is not feasible due to financial or conservation constraints, SCMs can serve as a viable proxy to determine resistance to movement surfaces.
Conclusion
Our study combining SCMs and telemetry-derived metrics of habitat suitability, core areas, and landscape connectivity has provided valuable insights into the habitat preferences and movement patterns of onagers, a globally-endangered equid. Onagers favor moderate-quality rangelands and avoid areas with intense livestock grazing due to disrupted resource access. Proximity to mountains provides essential security and thermal cover during severe weather events, while hill-valleys serve as refugia for disturbances. Our work highlights the limitations of SCMs, which tend to overestimate resistance compared to functional connectivity models based on telemetry data. To improve the rigor of our understanding of animal movements, more nuanced approaches that incorporate species behavior (via telemetry or other methods) are necessary. Although our study focused on onagers, the lessons learned can be applied to other highly mobile species. Our results suggest that the applicability of SCMs may differ for different species depending on their life history traits and movement types and capacities. It is therefore crucial to consider the unique characteristics and ecological requirements of each species when applying these methods to different organisms. We hope that our work will provide a foundation for future research to yield deeper insights about landscape connectivity for conservation purposes.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adriaensen F, Chardon JP, De Blust G, Swinnen E, Villalba S, Gulinck H, Matthysen E (2003) The application of ‘least-cost’modelling as a functional landscape model. Landsc Urban Plan 64(4):233–247
Baguette M, Blanchet S, Legrand D, Stevens VM, Turlure C (2013) Individual dispersal, landscape connectivity and ecological networks. Biol Rev 88(2):310–326
Beier P, Loe S (1992) A checklist for evaluating impacts to wildlife movement corridors. Wildl Soc Bull 20:434–440
Beier P, Majka DR, Spencer WD (2008) Forks in the road: choices in procedures for designing wildland linkages. Conserv Biol 22:836–851
Bowyer RT, Boyce MS, Goheen JR, Rachlow JL (2019) Conservation of mammals: threats, protected areas, and hunting. J Mammal 100:923–941
Brown L, Zedrosser A, Arnemo JM, Fuchs B, Kindberg J, Pelletier F (2023) Landscape of fear or landscape of food? Moose hunting triggers an antipredator response in brown bears. Ecol Appl 33(4):e2840
Compton BW, McGarigal K, Cushman SA, Gamble LR (2007) A resistant-kernel model of connectivity for amphibians that breed in vernal pools. Conserv Biol 21(3):788–799
Correa Ayram CA, Mendoza ME, Etter A, Salicrup DRP (2016) Habitat connectivity in biodiversity conservation: A review of recent studies and applications. Prog Phys Geogr 40(1):7–37
Crooks KR, Burdett CL, Theobald DM, Rondinini C, Boitani L (2011) Global patterns of fragmentation and connectivity of mammalian carnivore habitat. Phil Trans R Soc b: Biol Sci 366(1578):2642–2651
Cushman SA, McKelvey KS, Schwartz MK (2009) Use of empirically derived source-destination models to map regional conservation corridors. Conserv Biol 23(2):368–376
Cushman SA, Landguth EL, Flather CH (2013) Evaluating population connectivity for species of conservation concern in the American Great Plains. Biodivers Conserv 22(11):2583–2605
Darvishsefat A (2006) Atlas of protected areas of Iran [English-Persian]. University of Tehran Press, Tehran, p 157
Dechen Quinn AC, Williams DM, Porter WF (2012) Postcapture movement rates can inform data-censoring protocols for GPS-collared animals. J Mammal 93(2):456–463
Dijkstra EW (2022) A note on two problems in connexion with graphs. Edsger Wybe Dijkstra: his life, work, and legacy. Association for Computing Machinery, New York, pp 287–290
Diniz MF, Cushman SA, Machado RB, De Marco JP (2020) Landscape connectivity modeling from the perspective of animal dispersal. Landsc Ecol 35:41–58
Donadio E, Di Martino S, Heinonen S (2022) Rewilding Argentina: lessons for the 2030 biodiversity targets. Nature 603(7900):225–227
Du Toit JT, Cross PC, Valeix M (2017) Managing the livestock–wildlife interface on rangelands. In: Briske DD (ed) Rangeland systems. Springer, Cham, pp 395–425
Elith JH, Graham CP, Anderson R, Dudík M, Ferrier S, Guisan A et al (2006) Novel methods improve prediction of species’ distributions from occurrence data. Ecography 29(2):129–151
Esmaeili S (2020) Quantifying resource selection and community attitude to inform conservation of the globally-endangered Onager. University of Wyoming, Wyoming
Esmaeili S, Hemami MR, Goheen JR (2019) Human dimensions of wildlife conservation in Iran: assessment of human-wildlife conflict in restoring a wide-ranging endangered species. PLoS ONE 14(8):e0220702
Feh C, Shah N, Rowen M, Reading R, Goyal S. P (2002) Status and action plan for the Asiatic wild ass (Equus hemionus). In: Moehlman PD (ed) Equids: Zebras, Asses and Horses. Status survey and conservation action plan. IUCN, Glan, pp 62–71
Fingesi UI (2023) Habitat corridors use by wildlife in wooded patches of an agricultural landscape in New Bussa. Niger Sci Rep Life Sci 4(2):58–74
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Glob Ecol Biogeogr 16(3):265–280
Fortuna MA, Garcia C, Guimaraes PR, Bascompte J (2008) Spatial mating networks in insect-pollinated plants. Ecol Lett 11:490–498
Fynn RW, Augustine DJ, Peel MJ, de Garine-Wichatitsky M (2016) Strategic management of livestock to improve biodiversity conservation in African savannahs: a conceptual basis for wildlife–livestock coexistence. J Appl Ecol 53(2):388–397
Gastón A, Blázquez-Cabrera S, Garrote G, Mateo-Sánchez MC, Beier P et al (2016) Response to agriculture by a woodland species depends on cover type and behavioral state: insights from resident and dispersing Iberian lynx. J Appl Ecol 53(3):814–824
Ghorbanian A, Kakooei M, Amani M, Mahdavi S, Mohammadzadeh A, Hasanlou M (2020) Improved land cover map of Iran using sentinel imagery within Google earth engine and a novel automatic workflow for land cover classification using migrated training samples. ISPRS J Photogramm Remote Sens 167:276–288
Hanks EM, Hooten MB (2013) Circuit theory and model based inference for landscape connectivity. J Am Stat Assoc 108:22–33
Hedrick PW (2005) A standardized genetic differentiation measure. Evolution 59(8):1633–1638
Hofmann DD, Cozzi G, McNutt JW, Ozgul A, Behr DM (2023) A three-step approach for assessing landscape connectivity via simulated dispersal: African wild dog case study. Landsc Ecol 38:981–998
Horne JS, Garton EO, Krone SM, Lewis JS (2007) Analyzing animal movements using Brownian bridges. Ecology 88(9):2354–2363
Jennings AP, Zubaid A, Veron G (2010) Ranging behaviour, activity, habitat use, and morphology of the Malay civet (Viverra tangalunga) on Peninsular Malaysia and comparison with studies on Borneo and Sulawesi. Mamm Biol 75(5):437–446
Kaczensky P, Ganbaatar O, Von Wehrden H, Walzer C (2008) Resource selection by sympatric wild equids in the Mongolian Gobi. J Appl Ecol 45:1762–1769
Kaczensky P, Lkhagvasuren B, Pereladova O, Hemami M, Bouskila A (2015) Equus hemionus. The IUCN Red list of threatened species 2015. 2015 International Union for Conservation of Nature and Natural Resources 8235:1–24
Karagulle D, Frye C, Sayre R, Breyer S, Aniello P, Vaughan R, Wright D (2017) Modeling global Hammond landform regions from 250-m elevation data. Trans GIS 21(5):1040
Kauffman MJ, Aikens EO, Esmaeili S, Kaczensky P, Middleton A, Monteith KL et al (2021) Causes, consequences, and conservation of ungulate migration. Annu Rev Ecol Evol Syst 52:453–478
Kays R, Crofoot MC, Jetz W, Wikelski M (2015) Terrestrial animal tracking as an eye on life and planet. Science 348(6240):aaa2478
Keeley AT, Beier P, Gagnon JW (2016) Estimating landscape resistance from habitat suitability: effects of data source and nonlinearities. Landsc Ecol 31(9):2151–2162
Keeley ATH, Beier P, Jenness JS (2021) Connectivity metrics for conservation planning and monitoring. Biol Cons 255:109008
Kramer-Schadt S, Niedballa J, Pilgrim JD, Schröder B, Lindenborn J, Reinfelder V, Wilting A (2013) The importance of correcting for sampling bias in MaxEnt species distribution models. Divers Distrib 19(11):1366–1379
Landguth EL, Cushman SA, Schwartz MK, McKelvey KS, Murphy M, Luikart G (2010) Quantifying the lag time to detect barriers in landscape genetics. Mol Ecol 19(19):4179–4191
Landguth EL, Hand BK, Glassy J, Cushman SA, Sawaya MA (2012) UNICOR: a species connectivity and corridor network simulator. Ecography 35(1):9–14
Li Y, Li M, Li C, Liu Z (2020) Optimized maxent model predictions of climate change impacts on the suitable distribution of cunninghamia lanceolata in China. Forests 11(3):302
Liang G, Niu H, Li Y (2023) A multi-species approach for protected areas ecological network construction based on landscape connectivity. Global Ecol Conserv 46:e02569
Littlefield CE, McRae BH, Michalak JL, Lawler JJ, Carroll C (2017) Connecting today’s climates to future climate analogs to facilitate movement of species under climate change. Conserv Biol 31(6):1397–1408
Manly BFL, McDonald L, Thomas DL, McDonald TL, Erickson WP (2007) Resource selection by animals: statistical design and analysis for field studies, 2nd edn. Kluwer, Amsterdam
Marrotte RR, Bowman J, Brown MG, Cordes C, Morris KY, Prentice MB, Wilson PJ (2017) Multi-species genetic connectivity in a terrestrial habitat network. Mov Ecol 5(1):1–11
Mateo Sánchez MC, Balkenhol N, Cushman SA, Perez T, Dominguez A, Saura S (2015a) Estimating effective landscape distances and movement corridors: comparison of habitat and genetic data. Ecosphere 6(4):1–16
Mateo Sánchez MC, Balkenhol N, Cushman SA, Perez T, Dominguez A, Saura S (2015b) A comparative framework to infer landscape effects on population genetic structure: are habitat suitability models effective in explaining gene flow? Landsc Ecol 30:1405–1420
McGuire JL, Lawler JJ, McRae BH, Nuñez TA, Theobald DM (2016) Achieving climate connectivity in a fragmented landscape. Proc Natl Acad Sci 113:7195–7200
McRae BH, Shah VB (2009) Circuitscape user’s guide. The University of California, Santa Barbara
McRae BH, Dickson BG, Keitt TH, Shah VB (2008) Using circuit theory to model connectivity in ecology, evolution, and conservation. Ecology 89(10):2712–2724
Mesdaghi M (2010) Range management in Iran, 6th edn. Publications of Imam Reza University, Mashhad
Mohammadi A, Almasieh K, Wan HY, Nayeri D, Alambeigi A, Ransom JI, Cushman SA (2021) Integrating spatial analysis and questionnaire survey to better understand human-onager conflict in Southern Iran. Sci Rep 11(1):1–12
Molles MC Jr, Sher AA (2019) Ecology: concepts and applications, 8th edn. McGrow Hill, New York
Muratet A, Lorrilliere R, Clergeau P, Fontaine C (2013) Evaluation of landscape connectivity at community level using satellite-derived NDVI. Landsc Ecol 28(1):95–105
Naimi B (2013) usdm: Uncertainty analysis for species distribution models. R Package. Version 1: 1-12
Nakajima S, Suzuki H, Nakatsugawa M, Matsuo A, Hirota SK, Suyama Y, Nakamura F (2023) Inferring future changes in gene flow under climate change in riverscapes: a pilot case study in fluvial sculpin. Landsc Ecol 38(5):1351–1362
Neumann-Denzau G, Denzau H (2007) Remarks on the social system of the Mongolian wild ass (Equus hemionus hemionus).
Nowzari H, Hemami M, Karami M, Kheirkhah Zarkesh MM, Riazi B, Rubenstein DI (2013) Habitat use by the Persian onager, Equus hemionus onager (Perissodactyla: Equidae) in Qatrouyeh National Park, Fars, Iran. J Nat Hist 47(43–44):2795–2814
Ortega-Huerta MA, Townsend Peterson A (2008) Modeling ecological niches and predicting geographic distributions: a test of six presence-only methods. Rev Mexic Biodivers 79(1):205–216
Phillips SJ, Anderson RP, Schapire RE (2006) Maximum entropy modeling of species geographic distributions. Ecol Model 190(3–4):231–259
Phillips SJ, Dudík M, Schapire RE (2004) A maximum entropy approach to species distribution modeling. In Proceedings of the twenty-first international conference on Machine learning 83.
Pinto N, Keitt TH, Wainright M (2012) LORACS: JAVA software for modeling landscape connectivity and matrix permeability. Ecography 35(5):388–392
Ranius T, Widenfalk LA, Seedre M, Lindman L, Felton A, Hämäläinen A, Öckinger E (2023) Protected area designation and management in a world of climate change: a review of recommendations. Ambio 52(1):68–80
Rayfield B, Pelletier D, Dumitru M, Cardille JA, Gonzalez A (2016) Multipurpose habitat networks for short-range and long-range connectivity: a new method combining graph and circuit connectivity. Methods Ecol Evol 7(2):222–231
Rezvani A, Malakoutikhah S, Fakheran S, Soffianian A, Hemami MR, Senn J (2020) Comparing landscape suitability and permeability with and without migration data: the influence of species movement behavior. Turk J Zoolo 44(4):335–345
Ribeiro JW, Silveira dos Santos J, Dodonov P, Martello F, Brandão Niebuhr B, Ribeiro MC (2017) LandScape Corridors (lscorridors): a new software package for modelling ecological corridors based on landscape patterns and species requirements. Methods Ecol Evol 8(11):1425–1432
Ripple WJ, Newsome TM, Wolf C, Dirzo R, Everatt KT, Galetti M, Hayward MW, Kerley GI, Levi T, Lindsey PA, Macdonald DW (2015) Collapse of the world’s largest herbivores. Sci Adv 1(4):e1400103
Rudnick D, Ryan SJ, Beier P, Cushman SA, Dieffenbach F, Epps C et al (2012) The role of landscape connectivity in planning and implementing conservation and restoration priorities. Issues Ecol 16:1–23
Sappington JM, Longshore KM, Thompson DB (2007) Quantifying landscape ruggedness for animal habitat analysis: a case study using bighorn sheep in the Mojave Desert. J Wildl Manag 71(5):1419–1426
Sawyer H, Kauffman MJ, Nielson RM, Horne JS (2009) Identifying and prioritizing ungulate migration routes for landscape-level conservation. Ecol Appl 19(8):2016–2025
Sawyer SC, Epps CW, Brashares JS (2011) Placing linkages among fragmented habitats: do least-cost models reflect how animals use landscapes? J Appl Ecol 48(3):668–678
Sawyer H, LeBeau CW, McDonald TL, Xu W, Middleton AD (2019) All routes are not created equal: an ungulate’s choice of migration route can influence its survival. J Appl Ecol 56(8):1860–1869
Schroeder NM, Ovejero R, Moreno PG, Gregorio P, Taraborelli P, Matteucci SD, Carmanchahi PD (2013) Including species interactions in resource selection of guanacos and livestock in Northern Patagonia. J Zool 291(3):213–225
St-Louis A, Côté SD (2014) Resource selection in a high-altitude rangeland equid, the kiang (Equus kiang): influence of forage abundance and quality at multiple spatial scales. Can J Zool 92:239–249
Stubbe A, Stubbe M, Batsaikhan N (2007) Morphology, reproduction and mortality of Equus hemionus hemionus in Mongolia
Synes NW, Ponchon A, Palmer SC, Osborne PE, Bocedi G, Travis JM, Watts K (2020) Prioritising conservation actions for biodiversity: lessening the impact from habitat fragmentation and climate change. Biol Cons 252:108819
Taylor PD (2006) Landscape connectivity: a return to the basics. In: Crooks KR, Sanjayan M (eds) Connectivity conservation. Cambridge University Press, Cambridge
Taylor PD, Fahrig L, Henein K, Merriam G (1993) Connectivity is a vital element of landscape structure. Oikos 68:571–573
Teitelbaum CS, Mueller T (2019) Beyond migration: causes and consequences of nomadic animal movements. Trends Ecol Evol 34(6):569–581
Tischendorf L, Fahrig L (2000) On the usage and measurement of landscape connectivity. Oikos 90(1):7–19
Trainor AM, Walters JR, Morris WF, Sexton J, Moody A (2013) Empirical estimation of dispersal resistance surfaces: a case study with red-cockaded woodpeckers. Landsc Ecol 28(4):755–767
Urban D, Keitt T (2001) Landscape connectivity: a graph-theoretic perspective. Ecology 82:1205–1218
Zeller KA, Schroeder CA, Wan HY, Collins G, Denryter K, Jakes AF, Cushman SA (2021) Forecasting habitat and connectivity for pronghorn across the Great Basin ecoregion. Divers Distrib 27(12):2315–2329
Ziółkowska E, Ostapowicz K, Radeloff VC, Kuemmerle T, Sergiel A, Zwijacz-Kozica T et al (2016) Assessing differences in connectivity based on habitat versus movement models for brown bears in the Carpathians. Landsc Ecol 31:1863–1882
Acknowledgements
We express our sincere gratitude to the Iranian Department of Environment and the provincial Natural Resources Departments of Fars, Yazd, and Kerman provinces for their invaluable coordination and cooperation in facilitating our fieldwork and providing livestock data. We are also grateful to Gilda Shahnaseri and three anonymous reviewers for their insightful and constructive feedback.
Funding
The Iranian Department of Environment and the Mohamed Bin Zayed Species Conservation Fund, supported studies on onager conservation in Iran.
Author information
Authors and Affiliations
Contributions
MRH and AZ conceptualized and designed the study; MRH conducted field data collection with contribution from AZ, SE, PK, and JG. The data were managed and analyzed by AZ, with contribution from SE and advice from other authors. AZ took the lead in writing the initial draft of the manuscript, with contributions from MRH. All authors contributed to revisions of the final version and gave approval for its publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare they have no financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rezvani, A., Hemami, MR., Goheen, J.R. et al. Rethinking connectivity modeling for high-mobility ungulates: insights from a globally endangered equid. Landsc Ecol 39, 73 (2024). https://doi.org/10.1007/s10980-024-01873-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10980-024-01873-5