Abstract
Background
Diabetic nephropathy (DN), which is a chronic outcome of diabetes mellitus (DM), usually progresses to end-stage renal disease (ESRD). The DN pathophysiology, nevertheless, is not well-defined. Several miRNAs were reported to be either risk or protective factors in DN.
Methods, and results
The present study sought to inspect the potential diagnostic and prognostic value of hsa-miR-221 in DN. The study included 200 participants divided into four groups: Group 1 (50 patients with DN), Group 2 (50 diabetic patients without nephropathy), Group 3 (50 nondiabetic patients with CKD), and Group 4 (50 healthy subjects as a control group). Patients in groups 1 and 3 were further classified based on the presence of macroalbuminuria and microalbuminuria. Hsa-miR-221 expression was measured by RT- qRT-PCR. DN patients had significantly elevated serum hsa-miR-221 levels than the other groups, while diabetic patients without nephropathy exhibited elevated levels compared to both nondiabetic patients with CKD, and the control group. The DN patients with macroalbuminuria revealed significantly higher mean values of hsa-miR-221 relative to the patients with microalbuminuria. Significant positive associations were observed in the DN group between serum hsa-miR-221 and fasting insulin, fasting glucose, HOMA IR, ACR, and BMI. The ROC curve analysis of serum hsa-miR-221 in the initial diagnosis of DN in DM revealed high specificity and sensitivity.
Conclusions
It is concluded that hsa-miR-221 has the potential to be a useful biomarker for prognostic and diagnostic purposes in DN.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Between 1980 and 2021, the number of diabetic patients globally increased from 108 million to 536.6 million, climbing to 783.2 million in 2045. Type 2 diabetes represents about 90–95% of these cases [1, 2].
Micro- and macrovascular complications in diabetes mellitus (DM) affect various systems and organs [3]. Diabetic nephropathy (DN) is among the most serious and common diabetes complications and develops in 20% of diabetic patients [4, 5]. Renal replacement would be necessary for approximately half of all chronic diabetes patients having end-stage renal disease (ESRD) [6].
Diabetes is considered responsible for 80% of ESRD cases worldwide. Type 2 diabetes is responsible for 91% of all new cases of diabetes related ESRD, following the United States Renal Data System (USRDS) [7].
Proteinuria and glomerulopathy are developed gradually with years-long latent periods of DN [5]. Although substantial recent studies, the specific etiology of DN remains unknown. However, the development of DN is known to involve various variables and mechanisms.
The pathophysiology of DN is marked by hypertrophy and hyperplasia of the mesangium of the glomeruli and dysfunction of podocyte [8]. Long-term oxidative stress, advanced glycation end products (AGEs), hyperglycemia, and inflammation are all contributors to the development of DN [9].
MicroRNAs (miRs) have been attributed to the DN pathogenesis in two ways: Binding upregulated miRs to the protective genes in the kidney, repressing them, and downregulated miRNAs, in turn, can cause upregulation of genes that promote the mesangial expansion of or podocyte dysfunction [10].
Hsa-MiRNA-221 (Hsa-miR-221) accounts for a miRNA recognized within human cells of endothelium that regulate angiogenesis [11]. Hsa-miR-221 is an X-chromosome gene associated with metabolic disease [12].
Previous studies on hsa-miR-221 reported that its expression was upregulated and involved in the physiopathology of diabetes and type 2 diabetes (T2D) macrovascular and microvascular complications via targeting specific genes [13].
It has been found that aberrant overexpression of hsa-miR-221 accelerates retinal neovascularization in diabetic retinopathy by promoting cell migration, apoptosis, and proliferation, boosting hyperplasia and retinal artery blockage, and causing retinal ischemia and hypoxia [14].
As we know so far, no research has been accomplished to examine the potential of serum hsa-miR-221 as a biomarker for prognostic and diagnostic purposes in DN. The current study explores the hypothesis based on earlier research: whether serum hsa-miR-221 constitutes an innovative biomarker with significant accuracy for the diagnosis of DN.
Subjects and methods
Study design
This case-control study was conducted on a total of 200 participants chosen from the Internal Medicine Department at Beni-Suef University Hospital. The participants were divided into four groups: Group 1 (50 diabetic nephropathy patients), Group 2 (50 diabetic patients without nephropathy), Group 3 (50 nondiabetic patients having chronic kidney disease (CKD), and Group 4 (50 apparently healthy subjects as a control group). Then, the fifty patients with diabetic nephropathy were subdivided into 27 patients with macroalbuminuria and 23 patients with microalbuminuria. In contrast, the 50 patients with nondiabetic CKD were subdivided into 20 patients with macroalbuminuria and 20 patients with microalbuminuria.
Ethical considerations
The current study’s protocol followed the ethical principles of the Helsinki Declaration [15]. The study was granted approval by the Ethical Committee of the Faculty of Medicine, Beni-Suef University, with approval number (FMBSUREC /09072023/Abd Elazeem). Informed written consent was obtained from all participants.
Patients’ selection
According to the American Diabetes Association, both diabetic patients [16] and patients with diabetic nephropathy [17] were diagnosed. DN criteria of diagnosis were chronic albuminuria (> 200 g/min or > 300 mg/d) demonstrated on at least two occasions at least 3–6 months apart or an albumin-to-creatinine ratio (ACR) > 300 mg/g, with a consistent reduction in the glomerular filtration rate (GFR), without symptoms or signs of other primary renal diseases.
All patients’ clinical and medical histories were obtained. Next, the patients were submitted to a comprehensive clinical evaluation to select individuals with diabetic nephropathy who were over the age of 18. Exclusion criteria were hypertension, congestive heart failure, severe illness, current infection, peripheral artery disease, pregnancy, and malignancy. By measuring weight and height, body mass index (BMI) was estimated [18].
Sampling and laboratory investigations
Five milliliters of venous blood were drawn and put in either plain or fluoride-containing vacutainer tubes. Then, Urine samples were collected to evaluate albuminuria and estimate the urinary ACR and eGFR. Following overnight fasting for 10 h at least, biochemical tests (fasting insulin, fasting glucose, total cholesterol, HDL-c, LDL-c, HbA1c, triglycerides, creatinine, and urea) were conducted. The formula [19] adopted to assess the Homeostasis model assessment for insulin resistance (HOMA-IR) was as follows: HOMA-IR = fasting insulin (mlU/L) X fasting glucose (mg/dL) ÷ 405. Insulin resistance was deemed high if it was ˃ 2 [20]. Additionally, the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation [21] was employed to determine eGFR. LDL-c levels were calculated by Friedewald equation [22]: LDL-c = (Total Cholesterol mg/dL) − (HDL-c mg/dL) − (TGs mg/dL /5).
Kits used for investigations
All laboratory tests were performed according to the instructions of the manufacturer’s protocols for the following kits: Human Insulin ELISA Kit (Cat NO.:SL0933Hu, SUNLONG, China), Human Hb A1c ELISA Kit (Cat No: SL3471Hu, SUNLONG, China), Glucose Colorimetric Assay (Cat No: KitMAK263-1KT, Sigma-Aldrich, Germany), Triglyceride Colorimetric Assay Kit (Cat No: 10,010,303, Cayman, USA), Cholesterol Quantitation Kit Cat No: MAK043-1KT, Sigma-Aldrich, Germany), HDL-c Colorimetric Assay Kit (Cat No: E-BC-K222-S, Elabscience, USA), BUN Colorimetric Assay Kit (Cat No: MAK006-1KT, Sigma-Aldrich, Germany), Creatinine Colorimetric Assay Kit (Cat No: MAK080, Sigma-Aldrich, Germany), and ACR colorimetric Assay Kit (Cat NO: LS-K562-100, LSBio, UK).
Extraction of miRNA
Purified total RNA, comprising miRNA, from serum samples was extracted using the miRNeasy mini kit (cat. no. 217,004, QIAGEN, Germany) following the manufacturer’s instructions. Spectrophotometry (JENWAY, USA) was utilized to measure the isolated RNA at 260 nm.
Real time quantitative reverse-transcription polymerase chain reaction (qRT-PCR)
According to manufacturer’s instructions, Real-time qRT-PCR was accomplished utilizing a single-plex TaqMan two-step stem-loop real-time RT. TaqmanTM miRNA assay and the TaqmanTM miRNA Reverse Transcription Kit (Thermo Fisher Scientific, Waltham, MA, USA) were utilized to produce cDNA. The RT reaction total volume was 15 µL. Primer Premier 6.00 was used to design the primers (Premier Biosoft, Palo Alto, CA, USA). The micro-RNA specific stem loop primer of miRNA-221, and miRNA-16 (a reference gene) were added. TaqMan® Small RNA Assay, and the TaqMan® Universal PCR Master Mix II PCR were used to obtain amplified PCR from cDNA.
Analysis of data
The cycle threshold (CT) was adopted to quantify the levels of miRNA-221 expression. The comparative CT method (ΔΔ CT) was calculated by the difference between miRNA-221 CT and the reference gene average CT in each sample. Furthermore, miRNA expression fold change was calculated according to the formula 2−ΔΔCt [23] using the controls as calibrators.
Statistical analysis
SPSS (Statistical Package for Social Sciences) version 23 (SPSS Inc., Chicago, IL, USA) was used. The data were presented as mean ± SD. The statistical analysis of group differences involved the use of the Kruskal–Walli test, the Shapiro–Wilk test, and the t test. To compare the discrepancies among groups, Tukey post-hoc testing was employed. To evaluate the linear relationships between the study genes and clinical factors, a simple linear correlation (Pearson correlation coefficient test) (r) was conducted. If p was 0.05, the p value was considered significant. Receiver operating characteristic curves (ROC curves) were employed to appraise the diagnostic performance of the studied parameter.
Results
The Frequency of some demographic, and clinical data in the studied groups were demonstrated in Table 1.
The mean levels of demographic and clinical data in the studied groups
The mean levels of demographic and clinical data in the investigated groups were recorded in Table 2.
The current study was conducted on 121 males (60.5% of subjects) and 79 females (39.5% of subjects). The mean levels of age and LDL-c revealed no significant variations across the studied groups.
In diabetic nephropathy, the mean levels of BMI, serum fasting insulin, serum fasting glucose, HOMA IR, serum HbA1c, serum creatinine, and ACR exhibited significantly higher mean levels. In contrast, the eGFR exhibited significantly lower mean levels than diabetic patients without nephropathy, nondiabetic patients with CKD, and healthy controls. Diabetic nephropathy patients had a significantly longer disease duration than diabetic patients without nephropathy and nondiabetic patients with CKD.
Compared to healthy controls and nondiabetic patients with CKD, both diabetic nephropathy and diabetic patients without nephropathy showed a significant decline in mean serum HDL-c levels along with significant increases in mean serum levels of TG and cholesterol.
The mean levels of serum BUN demonstrated a significant increase in diabetic nephropathy compared to diabetic patients without nephropathy, healthy controls, and nondiabetic patients having CKD.
The mean level of serum hsa-miR-221 in the studied groups
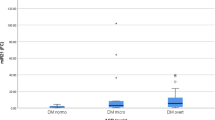
As demonstrated in Fig. 1a, the mean levels of serum hsa-miR-221 were significantly (p value ˂0.001) higher in diabetic nephropathy patients (3.06 ± 0.44) than those in diabetic patients without nephropathy (2.19 ± 0.39), nondiabetic patients with CKD (0.995 ± 0.16), and healthy controls (1 ± 0.043). They were significantly (p-value ˂0.001) higher in diabetic patients without nephropathy (2.19 ± 0.39) than those in nondiabetic patients with CKD (0.995 ± 0.16) and healthy controls (1 ± 0.043); however, they were comparable in nondiabetic patients with CKD (0.995 ± 0.16) and healthy subjects (1 ± 0.043).
The mean levels of serum hsa-mir-221 in studied subjects. a mean levels of serum hsa-mir-221 in studied groups. b mean levels of serum hsa-mir-221 in subjects with different degrees of albuminuria in diabetic nephropathy group. c mean levels of serum hsa-mir-221 in subjects with different degrees of albuminuria in nondiabetic CKD group
The mean levels of hsa-miR-221 in the subgroups
According to Table 3, the mean levels of hsa-miR-221 in males and females in each group were not significantly different. Additionally, they showed no significant variations between patients who used rosiglitazone, glyburide, or metformin in any group of diabetic patients, However, they were significantly higher as obesity developed in patients with diabetic nephropathy.
The relation between hsa-miR-221 and levels of albuminuria in the DN group
As shown in Fig. 1b, in diabetic patients with nephropathy, the mean levels of hsa-miR-221 were significantly higher (P < 0.001) in patients with macroalbuminuria (3.11 ± 0.43) than those with microalbuminuria (2.43 ± 0.14). However, no considerable variation was found in the mean levels of hsa-miR-221 between patients with macroalbuminuria (0.992 ± 0.03) and those with microalbuminuria (0.996 ± 0.2) in the nondiabetic CKD group (Fig. 1c).
The significant correlations of serum hsa-miR-221 in the study
According to Fig. 2, significant (P < 0.001) positive correlations were noticed between serum hsa-miR-221 and fasting insulin (r = 0.504), fasting glucose (r = 0.407), HOMA IR (r = 0.610), ACR (r = 0.782), and BMI (r = 0.645) in diabetic nephropathy group. In diabetic patients without nephropathy, there were significant (P < 0.001) positive correlations between serum hsa-miR-221 and fasting insulin (r = 0.537) and HOMA IR (r = 0.622) only.
Logistic regression of significant predictor parameters for prediction of diabetic nephropathy
Univariate logistic regression revealed hsa-miR-221 to be a significant predictor of the development of diabetic nephropathy in diabetic patients, along with HbA1c, fasting glucose, fasting insulin, HOMA IR, and BMI (Table 4). This result remained in multivariate logistic regression models, whether conventional risk factors (BMI) or the significant variables from the univariate models (hsa-miR-221, HbA1c, fasting glucose, and HOMA IR; Table 4) were specified as covariates. According to Table 4, the combination of hsa-miR-221 and either fasting glucose or HbA1c showed the best performance and explained 70% and 79% (Nagelkerke R2) of the variation, respectively.
ROC curve analysis of serum hsa-miR-221 in the early detection of diabetic nephropathy in diabetes mellitus
The AUC of serum hsa-miR-221 was 0.953 with a sensitivity of 96% and specificity of 88% at the cut-off value ≥ 2.55, with a significantly accurate diagnosis for patients with diabetic nephropathy, as shown in Fig. 3.
Discussion
Early diagnosis and management of diabetic nephropathy are urgent to avoid complications and fatality [24].
Although albuminuria is considered the best available test for the diagnosis of diabetic nephropathy, additional measures, such as serum creatinine, urine ACR, or eGFR, can be used to assess renal damage [25]. However, none of those tests offers a significant sensitivity for early diagnosis of all patients with diabetic renal dysfunction [26, 27].
The current investigations demonstrated that the mean levels of BMI, serum fasting insulin, serum fasting glucose, HOMA IR, serum HbA1c, serum creatinine, and urinary ACR were significantly higher. In contrast, the mean eGFR concentrations were significantly decreased in diabetic nephropathy patients than their corresponding levels in diabetic patients with no nephropathy, nondiabetic patients with CKD, and healthy controls, indicating the increased severity of diabetes and the deterioration of the kidney in patients with diabetic nephropathy.
Although BUN levels were deemed significantly higher in DN than in diabetic patients without nephropathy, and healthy controls, they were comparable to nondiabetic patients with CKD, undistinguishing the kidney injury in both diseases.
In the present study, the hsa-miR-221 mean levels revealed a significant increase in diabetic nephropathy and diabetic patients without nephropathy compared to patients with nondiabetic CKD and healthy controls. At the same time, they were higher in DN than in diabetic patients without nephropathy and comparable between nondiabetic CKD and healthy controls. These findings suggested the significance of hsa-miR-221 for the diabetes pathogenesis and the progression of diabetic nephropathy.
This role was explained by Li et al. [28], and Poliseno et al. [11] who coincided with these results as they reported that longstanding hyperglycemia in diabetes resulted in induction of the expression of hsa-miR-221 and dysfunction of endothelial cells. Also, Fiorentino et al. [29], Coinciding with the present findings Costantino et al. [30], Lightell et al. [13], and Li et al. [31] revealed that hsa-miR-221 expression was highly increased in diabetes and might be included in the physiopathology of macrovascular and diabetes complications via specific genes.
Similar findings were found in diabetic retinopathy (DR), as researchers identified hsa-miR-221 as a biomarker for DR in T2DM and proliferative DR patients, and it was thought to contribute to DM pathogenesis and associated macrovascular complications [32,33,34,35].
Also, Li et al., [28] explained the importance of hsa-miR-221 in the occurrence and progression of T2DM as they reported that under high glucose levels, significantly elevated hsa-miR-221 could promote apoptosis and cell proliferation facilitating endothelial dysfunction and hyperplasia of retinal vessels contributing to diabetic microvascular complications. As a result, hsa-miR-221 seems to be abnormally increased in the serum of T2D patients, rising gradually as DR severity increased, aggravating the damage of retinal vessels and dysfunction exposed to elevated glucose levels suggesting that hsa-miR-221 could be implicated in angiogenesis, proposing an innovative hypothesis for DR in T2D patients.
In contrast to earlier and current findings, Togliatto et al. [36] discovered downregulation of hsa-miR-221 expression related to reduced angiogenesis in hyperglycemia, and Marei et al. [37] found substantial decreases in hsa-miR-221 levels in both placenta and serum in the GDM group relative to the control group. The findings on the effect of glucose levels on hsa-miR-221 expression are contentious. However, this could be attributed to modifications in cell culture procedures. Evidently, more research into the expression of hsa-miR-221 in hyperglycemia is needed.
The current study found that the mean levels of hsa-miR-221 in diabetic patients with nephropathy were greater in patients with macroalbuminuria than those with microalbuminuria but comparable in similar patients in the nondiabetic CKD group. Additionally, a significant positive correlation was noticed between hsa-miR-221 and urinary ACR in diabetic patients with nephropathy.
The current outcomes agree with those of Atya et al. [38], who discovered that the serum hsa-miR-221 expression and ACR ratio had a significant positive correlation in diabetic patients and patients with either proliferative or non-proliferative diabetic retinopathy.
The coincidence between hsa-miR-221 and DN and the level of albuminuria was explained by Su et al. [39], who reported that the proximal tubule was the target of impact in DN. In high-glucose conditions, podocytes triggered the division of epithelial cells of the proximal tubule, and extracellular vehicles mediated the connection. The level of hsa-miR-221, which was extracted from these extracellular vehicles, was significantly higher, promoting the division of the proximal tubule’s epithelial cells. Additionally, in diabetic mice, the suppression of hsa-miR-221 corrected these abnormal changes in the proximal tubule’s epithelial cells.
The present investigation found that in the diabetic patients with the nephropathy group, the hsa-miR-221 concentrations were positively correlated with BMI, fasting insulin, fasting glucose, HOMA IR, and urinary ACR. In contrast, in the diabetic patients without the nephropathy group, these correlations were limited to fasting insulin and HOMA IR.
The current correlations of hsa-miR-221 were similarly reported by Lustig et al. [40] who discovered that hsa-miR-221 was raised in obesity animal models, Atya et al. [38], who discovered that the serum hsa-miR-221 expression was positively correlated with HBA1c and HOMA-IR in diabetic patients without retinopathy and those with different degrees of diabetic retinopathy compared to healthy subjects, Liu et al. [14] who discovered that the serum hsa-miR-221 was significantly positively associated with metabolic HOMA IR in type 2 diabetes patients with diabetic retinopathy.
The explanation of these correlations were demonstrated by previous research [12], which revealed the binding of hsa-miR-221 to important genes and components of the insulin/insulin-like signaling pathway, as well as their relationship with metabolic syndrome, diabetes, and diabetic implications, Lustig et al. [40], who revealed that hsa-miR-221 could have a role in IR regulation via reducing adiponectin expression, and Meerson et al. [41] who showed that hsa-miR-221 expression in subcutaneous adipose tissue was positively linked with BMI in nondiabetic Pima Indians.
In contrast to the present results, Chou et al. [42] found that hsa-miR-221 concentrations in adipocytes were negatively associated with BMI. Additionally, Wang et al. [43] showed that metabolic syndrome-affected females without obesity had greater hsa-miR-221 plasma concentrations than did controls, while Ortega et al. [44] found lower plasma concentrations of hsa-miR-221 in obese patients.
The variance in these outcomes could be attributed to methodologic discrepancies or unknown attributes in the cohorts. Despite contradictory human data, hsa-miR-221 can promote IR development by suppressing adiponectin signaling and has the potential as a circulating biomarker.
This study revealed considerable declines in the HDL-c serum levels along with considerable elevations in the TG and cholesterol serum levels in diabetic nephropathy and diabetic patients without nephropathy relative to nondiabetic patients with CKD and controls.
Similar findings were discovered by Vinod et al. [45], who reported that serum cholesterol and TG levels were significantly elevated while HDL levels were significantly decreased in patients with poor control of hyperglycemia than those with good control, implying that the lipid profile could be used to predict and monitor diabetes mellitus.
The ROC curve analysis for the role of serum hsa-miR-221 in the early identification of diabetic nephropathy in diabetes mellitus revealed high substantial sensitivity and specificity with highly accurate diabetic nephropathy diagnosis.
No previous study investigated the use of serum hsa-miR-221 in the early detection of diabetic nephropathy in diabetes mellitus. Still, Atya et al. [38], and Liu et al. [14] discovered related results in diabetic retinopathy, revealing that hsa-miR-221 had clear diagnostic value in diabetic retinopathy in diabetic patients using ROC curve analysis.
By using the logistic regression, hsa-miR-221 was found to be a significant predictor of the development of diabetic nephropathy in diabetic patients, along with HbA1c, fasting glucose, fasting insulin, HOMA IR, and BMI. The combination of hsa-miR-221 and either fasting glucose or HbA1c showed the best performance and explained 70% and 79% of the variation, respectively assisting in an early diagnosis of the diabetic nephropathy, before kidney damage progresses or even before albuminuria develops.
Conclusion
According to the current study, diabetic nephropathy patients had elevated levels of hsa-miR-221 in their serum than diabetic patients without nephropathy and controls. In contrast, all diabetic patients had higher levels than nondiabetic individuals. Additionally, diabetic nephropathy patients with macroalbuminuria had increased levels of hsa-miR-221 than those with microalbuminuria. The levels of hsa-miR-221 positively correlated with the BMI, serum fasting insulin, serum fasting glucose, HOMA IR, and urinary ACR in the diabetic nephropathy group. The early detection of diabetic nephropathy in diabetes mellitus using serum hsa-miR-221 exhibited significant sensitivity and specificity with an extremely accurate diagnosis of diabetic nephropathy. This study points to hsa-miR-221 as a possible biomarker for diagnostic and prognostic proposes in diabetic nephropathy, contributing to diagnose the condition early before kidney damage worsens or even before albuminuria occurs. Future studies should pay greater focus to investigate novel methods to suppress the negative miRNAs. Prior to kidney structural pathological changes, therapeutic targeting of miRNAs would try to reverse these earlier changes.
Data availability
The corresponding author will provide the datasets produced and/or analysed during the current work upon reasonable request.
Change history
20 December 2023
A Correction to this paper has been published: https://doi.org/10.1007/s11033-023-09085-x
Abbreviations
- Hsa :
-
Homo sapiens
- miR :
-
microRNA
- CKD:
-
Chronic kidney diseases
- HOMA IR:
-
Homeostatic Model Assessment for Insulin Resistance
- ACR:
-
Albumin to creatinine ratio
- BMI:
-
Body mass index
References
Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JC, Mbanya JC, Pavkov ME (2022) IDF diabetes atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract 183:109119
Zhou B, Lu Y, Hajifathalian K, Bentham J, Di Cesare M, Danaei G, Bixby H, Cowan MJ, Ali MK, Taddei C, Lo WC (2016) Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4.4 million participants. Lancet 387:1513–1530
Harding JL, Pavkov ME, Magliano DJ, Shaw JE, Gregg EW (2019) Global trends in diabetes complications: a review of current evidence. Diabetologia 62:3–16
Zhang XX, Kong J, Yun K (2020) Prevalence of diabetic nephropathy among patients with type 2 diabetes mellitus in China: a meta-analysis of observational studies. J Diabetes Res 2020
Samsu N (2021) Diabetic nephropathy: challenges in pathogenesis, diagnosis, and treatment. BioMed Res Int 2021
Yarahmadi A, Shahrokhi SZ, Mostafavi-Pour Z, Azarpira N (2021) MicroRNAs in diabetic nephropathy: from molecular mechanisms to new therapeutic targets of treatment. Biochem Pharmacol 189:114301
United States Renal Data System (2022) 2022 USRDS annual data report: epidemiology of kidney disease in the United States. https://usrds-adr.niddk.nih.gov/2022/chronic-kidney-disease
Dronavalli S, Duka I, Bakris GL (2008) The pathogenesis of diabetic nephropathy. Nat Clin Pract Endocrinol Metab 4:444–452
Wada J, Makino H (2013) Inflammation and the pathogenesis of diabetic nephropathy. Clin Sci 124:139–152
Kiyanpour F, Abedi M, Gheisari Y (2020) A systematic integrative approach reveals novel microRNAs in diabetic nephropathy. J Res Med Sci: Off J Isfahan Univ Med Sci 25
Poliseno L, Tuccoli A, Mariani L, Evangelista M, Citti L, Woods K, Mercatanti A, Hammond S, Rainaldi G (2006) MicroRNAs modulate the angiogenic properties of HUVECs. Blood 108:3068–3071
Deiuliis JA (2016) MicroRNAs as regulators of metabolic disease: pathophysiologic significance and emerging role as biomarkers and therapeutics. Int J Obes 40:88–101
Lightell DJ Jr, Moss SC, Woods TC (2018) Upregulation of miR-221 and-222 in response to increased extracellular signal-regulated kinases 1/2 activity exacerbates neointimal hyperplasia in diabetes mellitus. Atherosclerosis 269:71–78
Liu HN, Li X, Wu N, Tong MM, Chen S, Zhu SS, Qian W, Chen XL (2018) Serum microRNA-221 as a biomarker for diabetic retinopathy in patients associated with type 2 diabetes. Int J Ophthalmol 11:1889
Ashcroft RE (2008) The declaration of Helsinki. The Oxford textbook of clinical research ethics. Oxford University Press, New York, pp 141–148
Chamberlain JJ, Rhinehart AS, Shaefer CF Jr, Neuman A (2016) Diagnosis and management of diabetes: synopsis of the 2016 American diabetes association standards of medical care in diabetes. Ann Intern Med 164:542–552
American Diabetes Association (2016) 9. Microvascular complications and foot care. Diabetes Care 39(Supplement1):S72–S80
Keys A, Fidanza F, Karvonen MJ, Kimura N, Taylor HL (1972) Indices of relative weight and obesity. J Chronic Dis 25:329–343
Bonora E, Targher G, Alberiche M, Bonadonna RC, Saggiani F, Zenere MB, Monauni TI, Muggeo M (2000) Homeostasis model assessment closely mirrors the glucose clamp technique in the assessment of insulin sensitivity: studies in subjects with various degrees of glucose tolerance and insulin sensitivity. Diabetes Care 23:57–63
Romero-Gómez M, Viloria MD, Andrade RJ, Salmerón J, Diago M, Fernández-Rodríguez CM, Corpas R, Cruz M, Grande L, Vázquez L, Muñoz-de-Rueda P (2005) Insulin resistance impairs sustained response rate to peginterferon plus ribavirin in chronic hepatitis C patients. Gastroenterology 128:636–641
Levey AS, Stevens LA, Schmid CH, Zhang Y, Castro AF III, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J (2009) A new equation to estimate glomerular filtration rate. Ann Intern Med 150:604–612
Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–502
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2 –. ∆∆CT method 25:402–408
American Diabetes Association (2020) 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2020. Diabetes Care 43(1):S14–S31
Liu R, Li G, Cui XF, Zhang DL, Yang QH, Mu XY, Pan WJ (2011) Methodological evaluation and comparison of five urinary albumin measurements. J Clin Lab Anal 25:324–329
Zhou B, Zou H, Xu G (2016) Clinical utility of serum cystatin c in predicting diabetic nephropathy among patients with diabetes mellitus: a meta-analysis. Kidney Blood Press Res 41:919–928
Levey AS, Coresh J, Tighiouart H, Greene T, Inker LA (2019) Strengths and limitations of estimated and measured GFR. Nat Rev Nephrol 15:784–784
Li Y, Song YH, Li F, Yang T, Lu YW, Geng YJ (2009) MicroRNA-221 regulates high glucose-induced endothelial dysfunction. Biochem Biophys Res Commun 381:81–83
Fiorentino L, Cavalera M, Mavilio M, Conserva F, Menghini R, Gesualdo L, Federici M (2013) Regulation of TIMP3 in diabetic nephropathy: a role for microRNAs. Acta Diabetol 50:965–969
Costantino S, Paneni F, Lüscher TF, Cosentino F (2016) MicroRNA profiling unveils hyperglycaemic memory in the diabetic heart. Eur Heart J 37:572–576
Li MY, Pan SR, Qiu A (2016) Roles of microRNA-221/222 in type 2 diabetic patients with post-menopausal breast cancer. Genet Mol Res 15:10–4238
Martinez B, Peplow PV (2019) MicroRNAs as biomarkers of diabetic retinopathy and disease progression. Neural Regen Res 14:1858
Mammadzada P, Bayle J, Gudmundsson J, Kvanta A, André H (2019) Identification of diagnostic and prognostic microRNAs for recurrent vitreous hemorrhage in patients with proliferative diabetic retinopathy. J Clin Med 8:2217
Chen S, Yuan M, Liu Y, Zhao X, Lian P, Chen Y, Liu B, Lu L (2019) Landscape of microRNA in the aqueous humour of proliferative diabetic retinopathy as assessed by next-generation sequencing. Clin Exp Ophthalmol 47:925–936
Smit-McBride Z, Nguyen KN, Lai AW, Elliott GW, Nguyen JD, Nguyen AT, Morse LS (2018) The effect of DR circulatory microRNAs on VEGF secretion in human retinal pigment epithelial cells. Investig Ophthalmol Vis Sci 59:5370–5370
Togliatto G, Trombetta A, Dentelli P, Rosso A, Brizzi MF (2011) RETRACTED ARTICLE: MIR221/MIR222-driven post-transcriptional regulation of P27KIP1 and P57KIP2 is crucial for high-glucose-and AGE-mediated vascular cell damage. Diabetologia 54:1930–1940
Marei E, Gabr Youssef H (2020) Evaluation of MicroRNA-16 and MicroRNA-221 in serum and placenta in gestational diabetes mellitus: correlation with macrosomia. Egypt J Radiat Sci Appl 33:107–118
Atya SG, Elmohamady SN, Zidan MA, Fallah AA (2021) MicroRNA-221 in serum as a biomarker for diabetic retinopathy in type 2 diabetes Egyptian patients. Ann Rom Soc Cell Biol 25:14087–14097
Su H, Qiao J, Hu J, Li Y, Lin J, Yu Q, Zhen J, Ma Q, Wang Q, Lv Z, Wang R (2020) Podocyte-derived extracellular vesicles mediate renal proximal tubule cells dedifferentiation via microRNA-221 in diabetic nephropathy. Mol Cell Endocrinol 518:111034
Lustig Y, Barhod E, Ashwal-Fluss R, Gordin R, Shomron N, Baruch-Umansky K, Hemi R, Karasik A, Kanety H (2014) RNA-binding protein PTB and microRNA-221 coregulate AdipoR1 translation and adiponectin signaling. Diabetes 63:433–445
Meerson A, Traurig M, Ossowski V, Fleming JM, Mullins M, Baier LJ (2013) Human adipose microRNA-221 is upregulated in obesity and affects fat metabolism downstream of leptin and TNF-α. Diabetologia 56:1971–1979
Chou WW, Wang YT, Liao YC, Chuang SC, Wang SN, Juo SH (2013) Decreased microRNA-221 is associated with high levels of TNF-α in human adipose tissue-derived mesenchymal stem cells from obese woman. Cell Physiol Biochem 32:127–137
Wang YT, Tsai PC, Liao YC, Hsu CY, Juo SH (2013) Circulating microRNAs have a sex-specific association with metabolic syndrome. J Biomed Sci 20:1–10
Ortega FJ, Mercader JM, Catalan V, Moreno-Navarrete JM, Pueyo N, Sabater M, Gomez-Ambrosi J, Anglada R, Fernández-Formoso JA, Ricart W, Frühbeck G (2013) Targeting the circulating microRNA signature of obesity. Clin Chem 59:781–792
VinodMahato R, Gyawali P, Raut PP, Regmi P, Singh KP, Pandeya DR, Gyawali P (2011) Association between glycaemic control and serum lipid profile in type 2 diabetic patients: glycated haemoglobin as a dual biomarker. Biomed Res 22(3):375–380
Acknowledgements
The researchers would like to express their gratitude to all the volunteers and other healthcare professionals who contributed during the research procedure.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). No particular grant from a public funding organization was given to this research.
Author information
Authors and Affiliations
Contributions
MSA-T played a vital role in the research study’s design, data analysis, and article writing. The biochemical work and biochemical analysis were completed by MGM and HRR. The analysis of the clinical data was done by NAD and EEMR. The statistical analysis was carried out by MGM. The final review of the manuscript was completed by all authors, who also read and approved the finished product.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
The current study’s protocol followed the ethical principles of the Helsinki Declaration and was granted approval by the ethical committee of Faculty of Medicine at Beni-Suef University with approval number (FMBSUREC /09072023/Abd Elazeem). Informed written consents were obtained from all participants.
Consent to publish
Our raw data, and manuscript did not contain any individual details, images, or videos. The authors used to number the cases to maintain confidentiality of patient data.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this article was revised: In table 1, the values in the rows “Macro-albuminuria” and “Normal” are corrected.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Abdel-Tawab, M.S., Mohamed, M.G., Doudar, N.A. et al. Circulating hsa-miR-221 as a possible diagnostic and prognostic biomarker of diabetic nephropathy. Mol Biol Rep 50, 9793–9803 (2023). https://doi.org/10.1007/s11033-023-08846-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08846-y