Abstract
Brain metastasis (BM) is the most common type of brain tumor and frequently foreshadows disease progression and poor overall survival with patients having a median survival of 6 months. 70,000 new cases of BM are diagnosed each year in the United States (US) and the incidence rate for BM is increasing with improved detection. MicroRNAs (miRNAs) are small non-coding RNAs that serve as critical regulators of gene expression and can act as powerful oncogenes and tumor suppressors. MiRNAs have been heavily implicated in cancer and proposed as biomarkers or therapeutic targets or agents. In this review, we summarize an extensive body of scientific work investigating the role of microRNAs in BM. We discuss miRNA dysregulation, functions, targets, and mechanisms of action in BM and present the current standing of miRNAs as biomarkers and potential therapeutics for BM. We conclude with future directions of miRNA basic and clinical research in BM.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Brain metastasis (BM) is the most common type of brain tumor with approximately 70,000 new cases diagnosed every year in the United States (U.S.) [1]. BM has an estimated incidence rate of 9–45% and has been increasing each year due to improved detection rates [2, 3]. As there are more effective treatments for primary cancers, the incidence of metastases to the central nervous system (CNS) is expected to increase. The most common types of cancers that metastasize to the brain are lung (16–36%), breast (5–30%), and melanoma (6–11%) [1, 4, 5]. Limited understanding of BM pathogenic mechanisms presents a major obstacle to finding better therapies. Although some crucial steps of the brain metastatic cascade are recognized, the full metastatic process remains unclear [6].
MicroRNAs (miRNAs) are small non-coding RNAs that modify cell function by regulating gene expression and protein translation. To date, there are over 75,000 scientific publications on miRNAs in cancer and that number continues to grow each year. miRNAs can be powerful oncogenes and tumor suppressors, disease biomarkers, and attractive therapeutic targets. They play a crucial role in cancer by regulating cellular processes such as proliferation, differentiation, apoptosis, survival, motility, and morphogenesis [7]. miRNAs have been implicated in cancer initiation, progression, and metastasis.
In this review, we review the current standing of miRNAs as it relates to brain metastasis. We also discuss future directions of miRNA research in BM from both basic science and clinical perspectives.
Brain metastases
General concepts
In the US, lung cancer, breast cancer, and melanoma are responsible for almost 80% of all BM with the number of BM cases reflecting the incidence of the primary cancers [8]. 25% of patients with the diagnosis of metastatic non-small cell lung cancer will have BM. In the case of melanoma, almost 70% of those with metastatic disease will have CNS involvement.
Historically, BM heralds disease progression and poor survival due to rapid neurological deterioration. It is a significant clinical problem and is associated with poor prognosis and high morbidity [9]. The contemporary prognosis and treatment of BM depends on various factors, including the number and location of the metastases, the presence of metastases in other organs, and the clinical functional status of the patient [8]. The median survival from BM diagnosis to death is on average less than 6 months and can be as short as 2.3 months despite advances in early detection and new therapeutic modalities [2, 10]. Treatment requires a multidisciplinary approach that includes surgery, radiotherapy, chemotherapy, targeted therapies (small molecules and immunotherapy), and palliative care. Treatment goals include: local control, providing the best level of quality of life, and prolonging survival. Furthermore, morbidity and mortality may also be dictated by the primary cancer.
Pathobiology of brain metastases
Organotropism, the predisposition for particular primary tumors to spread to a specific organ such as the brain, is mostly linked to neoplastic cell intrinsic properties and the metastatic site microenvironment [11]. The metastatic cellular cross-talk is a complex process involving molecular mechanisms that modulate the microenvironment of the metastatic site, migration of primary tumor cells, and eventual infiltration of primary tumor cells into the final invasion site.
The following cellular processes promote BM: (1) formation of the pre-metastatic niche, (2) transendothelial migration across the blood-brain barrier (BBB), and (3) synergistic interaction of the cells and metabolites in the brain microenvironment [11]. In recent years, there has been growing evidence of a role for microRNAs in regulating BM.
miRNAs
General concepts
Non-coding RNAs (ncRNAs) represent a diverse class of functional molecules that comprise up to 98% of the human transcriptome [12]. One of the most well-characterized and significant types of ncRNAs are miRNAs. miRNAs are short, non-coding RNA transcripts that regulate gene expression and protein translation by binding to mRNA (Fig. 1). Since their discovery, miRNAs have been extensively implicated in a wide variety of human cancers [13]. Numerous studies have characterized miRNAs as critical oncogenes and tumor suppressors that regulate a variety of neoplastic processes including brain metastasis.
MiRNA genes are first transcribed as a primary miRNA (pri-miRNA). This pri-miRNA is cleaved into a precursor miRNA (pre-miRNA) before leaving the nucleus. Once in the cytoplasm, the pre-miRNA is cleaved into the mature miRNA. To exert their regulatory functions, mature miRNAs associate with the RNA-induced silencing complex (miRISC) which is composed of an Argonaute and other proteins. The miRNA wraps into the RISC complex, leaving the first 8 nucleotides of the 5’end free. These free nucleotides are termed the seed sequence and are critical for miRNA function. Complementarity between an RNA transcript and the miRNA seed sequence identifies miRNA targets and directs the miRISC complex. Once the miRNA seed sequence is bound to the RNA target, the miRISC complex blocks protein translation or initiates degradation of the RNA target (Fig. 1). Often, miRNAs bind to the 3’-UTR of mRNA targets to regulate their expression. However, miRNA binding to the 5’-UTR, coding region, and gene promoter region has also been observed [12]. miRNAs can target different types of cells including cancer cells and cells in the tumor microenvironment.
miRNA biogenesis and mechanism of action. Schematic illustrating miRNA biogenesis as the miRNA is transcribed and processed in the nucleus before being exported into the cytoplasm as a pre-miRNA. Once in the cytoplasm, the pre-miRNA is further processed into the mature miRNA that associates with the RISC complex to elicit target mRNA regulation. Created in BioRender.com
miRNAs expression and functions in brain metastasis
General concepts
A substantial body of scientific literature has focused on studying miRNA expression profiles and functions in primary tumors and BM. These studies have focused on lung cancers, breast cancers, and melanomas. Subsequent comparison of these miRNA expression profiles has revealed numerous miRNAs that are significantly dysregulated in BM. miRNA signatures outside the tumor and its microenvironment, particularly in blood, plasma, and cerebral spinal fluid (CSF) may be used as surrogate biomarkers. These biomarkers may identify patients with cancer and at high risk of developing BM, detect micrometastasis before it is detected on imaging studies, and characterize BM without requiring pathology and invasive brain surgery. Table 1 presents a summary of miRNAs that are dysregulated in BM of the most common types of cancers that metastasize to the brain.
Lung cancer
Brain metastasis is a frequent complication associated with both small-cell and non-small-cell lung cancer (NSCLC). Many miRNAs in primary cancers have been correlated with the development of BM such as miR-21 in lung cancer patients [14]. Another oncogenic miRNA in lung cancer BM is miR-378 whose activity promotes higher expression of VEGF [15]. Downregulation of the following miRNAs is related to a higher incidence of BMs: miR-217, which is involved in the activation of the oncogenic protein p53, and miR-145, whose activity upregulates EGFR, thus promoting mitosis and cancer invasiveness [16,17,18]. Analysis of paired primary lung cancer and BM tissue biopsy identified upregulation of miR-423-5p, miR-197, miR-184, miR-9, miR-330-3p, miR-328 and downregulation of miR-199a-5p and miR-596-3p in BM [19,20,21,22,23,24,25,26].
The evidence for miRNAs as biomarkers in lung carcinoma BMs is scant. miR-let-7a is correlated with radiosensitivity of BMs, miR-330-3p influences the ability of lung cancer cells to metastasize, miR-375 is correlated with the incidence and prognosis of BMs, and miR-1207-5p inhibition promotes higher BBB permeability [27,28,29,30]. These miRNAs may represent predictive biomarkers.
Breast cancer
Fifteen to 25% of patients with metastatic breast cancer develop BM. Figueira, et al. reported that miR-802-5p and miR-195-5p were downregulated, while miR-92a-1-5p, miR-205-5p, and miR-181a-1-3p were upregulated in both tissue sample and plasma of breast cancer BM patients [31]. miRNA expression profiling of primary breast cancers and BM identified miR-199a-5p as downregulated in BM [32].
Downregulation of the following miRNAs was associated with the development of breast cancer BM: miR-7, miR-146a, miR-509, miR-211 [2]. In terms of BBB dysfunction, miR-181c promotes BBB destruction and miR-503 modifies the BBB microenvironment allowing cancer cells to pass [33, 34]. Additionally, miR-122 primes the BBB microenvironment as a premetastatic niche to support metastatic growth [35].
One study by Li et al. investigated the role of miRNAs in breast cancer cell migration and metastasis. The study found that the ratio of miR-196s to HOXC8 mRNA correlated with breast cancer cell migration and metastasis. Furthermore, the study demonstrated that knockdown of HOXC8 suppressed cell migration and metastasis, and ectopic expression of HOXC8 prevented the anti-tumoral effects of miR-196s on cell migration and metastasis [36]. These results suggest miR-196s as potential diagnostic biomarkers for BM.
miRNAs modulate the expression of cell adhesion molecules (CAMs) in cancer cells and endothelial cells, thereby facilitating the establishment of metastasis at distant metastatic sites. For example, miR-126 has been shown to suppress metastatic colonization by inhibiting pro-angiogenic genes and biomarkers of human metastasis [37]. Silencing of miR-126 in breast cancer cells leads to increased endothelial recruitment and metastatic brain colonization. Another miRNA, miR-21-3p, has been identified as a positive regulator of L1CAM expression, which is involved in the co-option of brain capillaries by cancer cells during metastasis [37]. Debeb et al. also noted that the miR-200 family was involved in the formation of breast BMs through higher expression of E-cadherin [38].
Melanoma
Melanoma is a malignant skin cancer that is notorious for metastasizing to the brain and causing intracranial bleeding and significant mortality. Downregulation of miR-224-5p, miR-452, and miR-542-3p promoted epithelial-to-mesenchymal transition (EMT) induction, thus increasing the invasiveness of melanoma cancer cells [39, 40].
miRNA expression profiling of primary melanomas with and without BM identified increased expression of miR-518a-5p, miR-527, miR-575, miR-622, miR-4501, miR-4654, miR-4698, miR-6759-5p, and miR-8078, and decreased expression of miR-150-5p, miR-34a-3p, miR-548f-4, miR-1270, miR-1290, miR-4278, and miR-4528 was noted [41, 42]. Bustos, et al. identified six miRNAs that were specifically increased in melanoma BMs compared to other types of BMs: miR-671-5p, miR-4664-3p, miR-4665-3p, miR-5694, miR-6741- 3p, and miR-6796-3p [43].
miRNA targets and mechanisms of action in brain metastasis
miRNAs promoting metastatic spread to the brain
miRNAs are important regulators of the tumor microenvironment as described in Table 2. Expression of miR-1258 in breast cancer BM cells inhibited the activity of heparanase (HPSE), a prometastatic enzyme that degrades components of the extracellular matrix (ECM) to increase cell motility, migration, and invasion. HPSE is a direct target of miR-1258; thus, miR-1258 is a tumor-suppressive miRNA in breast cancer BM that functions by upholding ECM integrity [44].
Additionally, miRNAs modulate BBB permeability and therefore are important regulators of tumor cell extravasation into the brain microenvironment. In NSCLC, miR-1207-5p targets EPB41L5. EPB41L5 is an oncogene that induces EMT, disrupts tight junctions, and increases BBB permeability. Thus, miR-1207-5p plays an important tumor suppressive role by maintaining BBB integrity and preventing metastatic spread to the brain [30]. miR-596-3p targets YAP1 in NSCLC and inhibits YAP1-induced MMP-2 activity which degrades tight junctions involved in BBB permeability [23]. Likewise, miR-509 targets TNFα and RhoC in breast cancer BM which inhibits RhoC-induced MMP-9 activity, disrupting tight junctions in the BBB [45]. In glioma vascular endothelial cells, miR-144 targets HSF2, miR-34c targets MAZ, miR-181d-5p targets SOX5, and miR-140 targets NFYA. All of these targets are crucial for promoting the expression of tight junction proteins such as ZO-1, occludin, and claudin-5 [46,47,48,49]. Lastly, miR-105 was shown to directly target ZO-1, an important tight junction protein, in breast cancer BM [50].
miRNAs remodeling the brain microenvironment
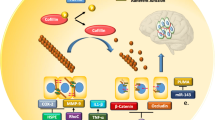
In addition to promoting metastatic spread to the brain, miRNAs remodel the brain microenvironment to foster BM growth. miRNAs significantly facilitate interactions between tumor cells and astrocytes, microglia, and endothelial cells to support BM as illustrated in Fig. 2.
miRNA functions in the brain microenvironment. Figure 2 illustrates miRNA functions and mechanisms of action in the brain microenvironment. miR-142-3p is shown downregulating metastasis progression through FGFR2 inactivation, miR-503-5p is shown downregulating immune activation, and miR-301a-3p is shown down regulating ECM integrity. Red boxes and red arrows indicate oncogenic functions. Blue boxes and blue arrows indicate tumor suppressive functions. Created in BioRender.com
Upregulated miR-345 then targets KISS1, a prominent tumor suppressor [53]. Thus, interactions between astrocytes and BM cells are mediated by miRNAs and significantly contribute to the remodeling of the brain microenvironment (See Table 2).
miRNAs also mediate BM by promoting interactions between tumor cells and microglia. The loss of the lncRNA XIST in breast cancer cells increases the secretion of exosomal miR-503 which triggers M1–M2 polarization of microglia. This polarization upregulates immune suppressive cytokines, ultimately leading to the suppression of T-cell proliferation in the brain microenvironment [34]. miRNAs are implicated in treatment-related remodeling of the brain microenvironment in BM. Irradiation of a BM NSCLC mouse model induced M1 microglial activation. Active M1 microglia increased secretion of miR-9, a miRNA that targets CDH1 and efficiently blocks mesenchymal to epithelial transition to reduce BM [54].
Therapeutic role of miRNAs in brain metastasis
Brain metastasis is a complex and challenging condition to treat, and there is growing interest in exploring the role of miRNAs in its treatment. In theory, there are two general strategies for treating metastasis using miRNAs. The first is to decrease expression of oncogenic miRNAs using miRNA inhibitors, which are antisense oligonucleotides that block the pro-metastatic miRNAs. The second is to increase the expression of tumor suppressor miRNAs, which results in suppression of oncogenic pathways and cancer cell growth [3].
Synthetic anti-miRNA oligonucleotides (AMOs), locked nucleic acid (LNA)-anti-miRNAs, and miRNA sponges have been used for miRNA tehrapeutics [3, 55]. While these nucleic acid classes improve miRNA stability and regulation, they poorly penetrate the BBB. Viral and non-viral delivery methods have been used to address the challenges in miRNA therapeutic delivery (i.e. stability in blood and crossing the BBB). Using viruses to deliver anti-oncogenic miRNAs or specific tumor suppressor miRNAs shows potential for therapeutic use [56]. Additionally, non-viral delivery methods using liposomes in the form of neutral lipid emulsion through the Trojan Horse Liposome (THL) system, nanoparticles, microspheres, and hydrogels are advantageous as stable formulations [57, 58]. While non-viral delivery mechanisms have lower toxicity, viral delivery mechanisms have higher delivery efficiency [37]. The use of pharmacological agents like Resveratrol and Longevinex to alter miRNA levels has also been tested [59]. While these agents have reduced miRNA specificity, they offer an alternative miRNA targeting strategy compared to nucleic acid methodologies. These nucleic acid therapeutic mechanisms, delivery approaches, and pharmacologic methodologies are shown in Fig. 3.
miRNAs therapeutics for brain metastasis. Schematic summarizing the therapeutic capacity of miRNAs for brain metastasis. This image lists the two main objectives of miRNA therapeutics and describes nucleic acid mechanisms, delivery approaches, and pharmacologic methodologies used to achieve these objectives. Created in BioRender.com
A review by Jafri in 2017 identified miR-96, miR-182, miR-21, miR-181-3p, miR-503-3p, miR-135a/b, miR-155, and miR-520c as oncogenic miRNAs that may be targeted for inhibition. While miR-200, miR-655, miR-101, miR-205, miR-132, miR-21, miR-135b, miR-155, miR-182, miR10b, miR-148a, miR-215, miR-612, miR-191 and miR-192 have potential for miRNA replacement in anti-metastasis therapy [60]. Currently, only a few miRNAs have good evidence to support their use for therapy for metastasis from melanoma (miR-1258) and breast cancer (miR-33) [56]. Altogether, miRNA-based therapeutics that directly target BM are currently lacking.
Future directions
While there is a significant amount of work depicting the mechanisms of action for many miRNAs in BM, there are numerous dysregulated miRNAs for which no functional understanding exists. Further research is needed to fully elucidate how miRNAs strengthen primary cancer cells for metastatic spread, prime the brain microenvironment for metastatic seeding, and promote BM growth. A comprehensive understanding of the mechanisms and pathways underlying pro-metastatic miRNA dysregulation in primary tumor cells may uncover novel therapeutic targets for preventative treatment.
In recent years, the research community has begun to understand the complex relationships that exist between tumor cells and resident cells of the brain. Cells such as astrocytes and neurons have been shown to communicate with tumor cells to promote or inhibit tumor growth. In this review, we discussed the few studies that have begun to investigate how this communication may occur within BM cells and the role of miRNAs in this communication. Further studies dedicated to revealing these mechanisms of communication in the brain microenvironment are crucial for understanding the roles of miRNAs and developing successful treatments for BM. Additionally, how miRNAs may be promoting cell state switching between the primary tumor environment and the brain microenvironment is a promising area of future research.
miRNAs as biomarkers for BM may be used for early detection of cancer progression. Prophylactic treatment of brain micrometastasis (i.e. absence of overt imaging evidence and non-symptomatic) usually with whole-brain radiation is controversial and may not be beneficial in BM due to the high morbidity related to radiation treatment effects. Validation of data on the classification of tissue origin of BMs through miRNAs will be beneficial in the clinical setting in cases of unknown primary tumors. The role of miRNAs in angiogenesis and EMT for prevention of BM, as well as their ability to regulate the tumor microenvironment to halt the progression and sensitize the cancer cells to chemotherapy are areas that need further investigation. Lastly, trends in miRNA expression during treatment can be potentially used as prognostic markers for predicting treatment response. Further translational research is needed to explore miRNAs as potential diagnostic markers and therapeutic agents or targets for patients with BM. Robust data regarding the type of sample (CSF, blood, or urine), standardized cut-off levels, and effective delivery routes are crucial for this translation.
Conclusion
This review illustrates the crucial roles that miRNAs play in brain metastasis. We summarized the dysregulation of miRNAs in brain metastasis that originates from a wide variety of primary tumors and the potential use of miRNAs as biomarkers. We discussed how miRNAs can promote metastatic spread to the brain and remodel the brain microenvironment to support or inhibit brain metastasis progression. More research is needed before miRNAs can be exploited for BM therapy.
Data availability
This manuscript did not generate, analyze, or store any primary data.
References
Ostrom QT, Wright CH, Barnholtz-Sloan JS (2018) Brain metastases: epidemiology. Handb Clin Neurol. 149:27–42. https://doi.org/10.1016/B978-0-12-811161-1.00002-5
Siegl F, Vecera M, Roskova I et al (2022) The significance of MicroRNAs in the molecular pathology of brain metastases. Cancers (Basel) 14(14):1–23. https://doi.org/10.3390/cancers14143386
Alsidawi S, Malek E, Driscoll JJ (2014) MicroRNAs in brain metastases: potential role as diagnostics and therapeutics. Int J Mol Sci 15(6):10508–10526. https://doi.org/10.3390/ijms150610508
Barnholtz-Sloan JS, Sloan AE, Davis FG, Vigneau FD, Lai P, Sawaya RE (2004) Incidence proportions of brain metastases in patients diagnosed (1973 to 2001) in the metropolitan detroit cancer surveillance system. J Clin Oncol 22(14):2865–2872. https://doi.org/10.1200/JCO.2004.12.149
Schouten LJ, Rutten J, Huveneers HAM, Twijnstra A (2002) Incidence of brain metastases in a cohort of patients with carcinoma of the breast, colon, kidney, and lung and melanoma. Cancer 94(10):2698–2705. https://doi.org/10.1002/cncr.10541
Kim MM, Mehta MP, Smart DK et al (2023) National cancer institute collaborative workshop on shaping the landscape of brain metastases research: challenges and recommended priorities. Lancet Oncol 24(8):e344–e354. https://doi.org/10.1016/S1470-2045(23)00297-8
Liu X, Chen Z, Yu J, Xia J, Zhou X (2009) MicroRNA profiling and head and neck cancer. Comp Funct Genomics. https://doi.org/10.1155/2009/837514
Fadul CE, Sarai G, Bovi JA et al (2023) Relevance of the updated recursive partitioning analysis (U-RPA) classification in the contemporary care of patients with brain metastases. Cancers (Basel) 15(12):3255. https://doi.org/10.3390/cancers15123255
Qin F, Zhang H, Ma L et al (2015) Low expression of slit2 and robo1 is associated with poor prognosis and brain-specific metastasis of breast cancer patients. Sci Rep. https://doi.org/10.1038/srep14430
Li H, Wu X, Cheng X (2016) Advances in diagnosis and treatment of metastatic cervical cancer. J Gynecol Oncol 27(4):301–310. https://doi.org/10.3802/jgo.2016.27.e43
Campbell BK, Gao Z, Corcoran NM, Stylli SS, Hovens CM (2022) Molecular mechanisms driving the formation of brain metastases. Cancers (Basel) 14(19):1–17. https://doi.org/10.3390/cancers14194963
Djebali S, Davis CA, Merkel A et al (2012) Landscape of transcription in human cells. Nature 489(7414):101–108. https://doi.org/10.1038/nature11233
Bhaskaran M, Mohan M (2014) MicroRNAs: history, biogenesis, and their evolving role in animal development and disease. Vet Pathol 51(4):759–774. https://doi.org/10.1177/0300985813502820
Zhu Z, Li Q, Xu M, Qi Z (2020) Effect of whole-brain and intensity-modulated radiotherapy on serum levels of miR-21 and prognosis for lung cancer metastatic to the brain. Med Sci Monit 26:1–6. https://doi.org/10.12659/MSM.924640
Chen LT, Xu SD, Xu H, Zhang JF, Ning JF, Wang SF (2012) MicroRNA-378 is associated with non-small cell lung cancer brain metastasis by promoting cell migration, invasion and tumor angiogenesis. Med Oncol 29(3):1673–1680. https://doi.org/10.1007/s12032-011-0083-x
Jiang W, Hou L, Wei J et al (2020) Hsa-miR-217 inhibits the proliferation, migration, and invasion in non-small cell lung cancer cells via targeting SIRT1 and p53/KAI1 signaling. Balkan Med J 37(4):208–214. https://doi.org/10.4274/balkanmedj.galenos.2020.2019.9.91
Donzelli S, Mori F, Bellissimo T et al (2015) Epigenetic silencing of miR-145-5p contributes to brain metastasis. Oncotarget 6(34):35183–35201. https://doi.org/10.18632/oncotarget.5930
Zhao C, Xu Y, Zhang Y et al (2013) Downregulation of miR-145 contributes to lung adenocarcinoma cell growth to form brain metastases. Oncol Rep 30(5):2027–2034. https://doi.org/10.3892/or.2013.2728
Tsakonas G, Koulouris A, Kazmierczak D et al (2022) Matched analyses of brain metastases versus primary non-small cell lung cancer reveal a unique microRNA signature. Int J Mol Sci 24(1):193. https://doi.org/10.3390/ijms24010193
Arora S, Ranade AR, Tran NL et al (2011) MicroRNA-328 is associated with (non-small) cell lung cancer (NSCLC) brain metastasis and mediates NSCLC migration. Int J Cancer 129(11):2621–2631. https://doi.org/10.1002/ijc.25939
Lu Y, Govindan R, Wang L et al (2012) MicroRNA profiling and prediction of recurrence/relapse-free survival in stage I lung cancer. Carcinogenesis 33(5):1046–1054. https://doi.org/10.1093/carcin/bgs100
Liu X, Shi H, Liu B, Li J, Liu Y, Yu B (2015) miR-330-3p controls cell proliferation by targeting early growth response 2 in non-small-cell lung cancer. Acta Biochim Biophys Sin (Shanghai) 47(6):431–440. https://doi.org/10.1093/abbs/gmv032
Li C, Zheng H, Xiong J et al (2022) miR-596-3p suppresses brain metastasis of non-small cell lung cancer by modulating YAP1 and IL-8. Cell Death Dis 13(8):699. https://doi.org/10.1038/s41419-022-05062-7
Remon J, Alvarez-Berdugo D, Majem M et al (2016) miRNA-197 and miRNA-184 are associated with brain metastasis in EGFR-mutant lung cancers. Clin Transl Oncol 18(2):153–159. https://doi.org/10.1007/s12094-015-1347-2
Sun G, Ding X, Bi N et al (2018) MiR-423-5p in brain metastasis: potential role in diagnostics and molecular biology. Cell Death Dis 9(10):936–10.1038/s41419-018-0955–5
Zhang L, Liang J, Han Z et al (2022) Micro-ribonucleic acids (miRNAs) and a proteomic profile in lung adenocarcinoma cases with brain metastasis. Ann Transl Med 10(24):1389–10.21037/atm-22-5703
Liu JK, Liu HF, Ding Y, Gao GD (2018) Predictive value of microRNA let-7a expression for efficacy and prognosis of radiotherapy in patients with lung cancer brain metastasis A case–control study. Medicine (United States). https://doi.org/10.1097/MD.0000000000012847
Wei C, Zhang R, Cai Q et al (2019) MicroRNA-330-3p promotes brain metastasis and epithelial-mesenchymal transition via GRIA3 in non-small cell lung cancer. Aging (Albany NY) 11(17):6734–6761. https://doi.org/10.18632/aging.102201
Chen Ljuan, Li X, ya, Zhao Yqiu et al (2017) Down-regulated microRNA-375 expression as a predictive biomarker in non-small cell lung cancer brain metastasis and its prognostic significance. Pathol Res Pract 213(8):882–888. https://doi.org/10.1016/j.prp.2017.06.012
Wu D, Deng S, Li L et al (2021) TGF-β1-mediated exosomal lnc-MMP2-2 increases blood–brain barrier permeability via the miRNA-1207-5p/EPB41L5 axis to promote non-small cell lung cancer brain metastasis. Cell Death Dis 12(8):1–10. https://doi.org/10.1038/s41419-021-04004-z
Figueira I, Godinho-Pereira J, Galego S et al (2021) Micrornas and extracellular vesicles as distinctive biomarkers of precocious and advanced stages of breast cancer brain metastases development. Int J Mol Sci 22(10):1–21. https://doi.org/10.3390/ijms22105214
Giannoudis A, Clarke K, Zakaria R, Varešlija D et al (2019) A novel panel of differentially-expressed microRNAs in breast cancer brain metastasis may predict patient survival. Sci Rep 9(1):18518. https://doi.org/10.1038/s41598-019-55084-z
Tominaga N, Kosaka N, Ono M et al (2015) Brain metastatic cancer cells release microRNA-181c-containing extracellular vesicles capable of destructing blood-brain barrier. Nat Commun. https://doi.org/10.1038/ncomms7716
Xing F, Liu Y, Wu SY et al (2018) Loss of XIST in breast cancer activates MSN-c-Met and reprograms microglia via exosomal miRNA to promote brain metastasis. Cancer Res 78(15):4316–4330. https://doi.org/10.1158/0008-5472.CAN-18-1102
Weiying YFM et al (2015) Breast cancer-secreted miR-122 reprograms glucose metabolism in pre-metastatic niche to promote metastasis. Nat Cell Biol 17(2):183–194. https://doi.org/10.1038/ncb3094.Breast
Li Y, Zhang M, Chen H et al (2010) Ratio of miR-196s to HOXC8 messenger RNA correlates with breast cancer cell migration and metastasis. Cancer Res 70(20):7894–7904. https://doi.org/10.1158/0008-5472.CAN-10-1675
Kanchan RK, Siddiqui JA, Mahapatra S, Batra SK, Nasser MW (2020) MicroRNAs orchestrate pathophysiology of breast cancer brain metastasis: advances in therapy. Mol Cancer 19(1):1–16. https://doi.org/10.1186/s12943-020-1140-x
Debeb BG, Lacerda L, Anfossi S et al (2016) miR-141-mediated regulation of brain metastasis from breast cancer. J Natl Cancer Inst 108(8):2–11. https://doi.org/10.1093/jnci/djw026
Knoll S, Fürst K, Kowtharapu B et al (2014) E2F1 induces miR-224/452 expression to drive EMT through TXNIP downregulation. EMBO Rep 15(12):1315–1329. https://doi.org/10.15252/embr.201439392
Rang Z, Yang G, Wang YW, Cui F (2016) MIR-542-3p suppresses invasion and metastasis by targeting the proto-oncogene serine/threonine protein kinase, PIM1, in melanoma. Biochem Biophys Res Commun 474(2):315–320. https://doi.org/10.1016/j.bbrc.2016.04.093
Mikkelsen LH, Andersen MK, Andreasen S et al (2019) Global microRNA profiling of metastatic conjunctival melanoma. Melanoma Res 29(5):465–473. https://doi.org/10.1097/CMR.0000000000000606
Hanniford D, Zhong J, Koetz L et al (2015) A miRNA-based signature detected in primary melanoma tissue predicts development of brain metastasis. Clin Cancer Res 21(21):4903–4912. https://doi.org/10.1158/1078-0432.CCR-14-2566
Bustos MA, Tran KD, Rahimzadeh N et al (2020) Integrated assessment of circulating cell-free microRNA signatures in plasma of patients with melanoma brain metastasis. Cancers (Basel) 12(6):1–24. https://doi.org/10.3390/cancers12061692
Zhang L, Sullivan PS, Goodman JC et al (2011) MicroRNA-1258 suppresses breast cancer brain metastasis by targeting heparanase. Cancer Res 71(3):645–654. https://doi.org/10.1158/0008-5472.CAN-10-1910
Xing F, Sharma S, Liu Y et al (2015) miR-509 suppresses brain metastasis of breast cancer cells by modulating RhoC and TNF-α. Oncogene 34(37):4890–4900. https://doi.org/10.1038/onc.2014.412
Cai H, Xue Y, Wang P et al (2015) The long noncoding RNA TUG1 regulates blood-tumor barrier permeability by targeting miR-144. Oncotarget 6(23):19759–19779. https://doi.org/10.18632/oncotarget.4331
Zhao L, Wang P, Liu Y, Ma J, Xue Y (2015) miR-34c regulates the permeability of blood-tumor barrier via MAZ-mediated expression changes of ZO-1, occludin, and claudin-5. J Cell Physiol 230(3):716–731. https://doi.org/10.1002/jcp.24799
Guo J, Cai H, Zheng J, Liu X et al (2017) Long non-coding RNA NEAT1 regulates permeability of the blood-tumor barrier via miR-181d-5p-mediated expression changes in ZO-1, occludin, and claudin-5. Biochim Biophys Acta Mol Basis Dis 1863(9):2240–2254. https://doi.org/10.1016/j.bbadis.2017.02.005
Ma J, Wang P, Yao Y, Liu Y et al (2016) Knockdown of long non-coding RNA MALAT1 increases the blood-tumor barrier permeability by up-regulating miR-140. Biochim Biophys Acta 1859(2):324–338. https://doi.org/10.1016/j.bbagrm.2015.11.008
Zhou W, Fong MY, Min Y et al (2014) Cancer-secreted miR-105 destroys vascular endothelial barriers to promote metastasis. Cancer Cell 25(4):501–515. https://doi.org/10.1016/j.ccr.2014.03.007
Berrout J, Kyriakopoulou E, Moparthi L et al (2017) TRPA1-FGFR2 binding event is a regulatory oncogenic driver modulated by miRNA-142-3p. Nat Commun 8(1):947. https://doi.org/10.1038/s41467-017-00983-w
Morad G, Daisy CC, Otu HH et al (2020) Cdc42-dependent transfer of mir301 from breast cancer-derived extracellular vesicles regulates the matrix modulating ability of astrocytes at the blood-brain barrier. Int J Mol Sci 21(11):3851. https://doi.org/10.3390/ijms21113851
Kaverina N, Borovjagin AV, Kadagidze Z et al (2017) Astrocytes promote progression of breast cancer metastases to the brain via a KISS1-mediated autophagy. Autophagy 13(11):1905–1923. https://doi.org/10.1080/15548627.2017.1360466
Jin Y, Kang Y, Peng X et al (2021) Irradiation-induced activated microglia affect brain metastatic colonization of NSCLC cells via miR-9/CDH1 Axis. Onco Targets Ther 14:1911–1922. https://doi.org/10.2147/OTT.S301412
Li Y, Zhao S, Zhen Y et al (2011) A miR-21 inhibitor enhances apoptosis and reduces G 2-M accumulation induced by ionizing radiation in human glioblastoma U251 cells. Brain Tumor Pathol 28(3):209–214. https://doi.org/10.1007/s10014-011-0037-1
McDermott R, Gabikian P, Sarvaiya P et al (2013) MicroRNAs in brain metastases: big things come in small packages. J Mol Med 91(1):5–13. https://doi.org/10.1007/s00109-012-0971-3
Babar IA, Cheng CJ, Booth CJ et al (2012) Nanoparticle-based therapy in an in vivo microRNA-155 (miR-155)-dependent mouse model of lymphoma. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1201516109
Zhang Y, Zhang YF, Bryant J et al (2004) Intravenous RNA interference gene therapy targeting the human epidermal growth factor receptor prolongs survival in intracranial brain cancer. Clin Cancer Res 10(11):3667–3677. https://doi.org/10.1158/1078-0432.CCR-03-0740
Mukhopadhyay P, Das S, Ahsan MK, Otani H, Das DK (2012) Modulation of microRNA 20b with resveratrol and longevinex is linked with their potent anti-angiogenic action in the ischaemic myocardium and synergestic effects of resveratrol and γ-tocotrienol. J Cell Mol Med 16(10):2504–2517. https://doi.org/10.1111/j.1582-4934.2011.01480.x
Jafri MA, Al-Qahtani MH, Shay JW (2017) Role of miRNAs in human cancer metastasis: Implications for therapeutic intervention. Semin Cancer Biol 44:117–131. https://doi.org/10.1016/j.semcancer.2017.02.004
Acknowledgements
This review was supported by the National Institutes of Health grants UO1 CA220841, R21NS122136-01, RO1NS122222 (all to Roger Abounader), the National Cancer Institute Cancer Center Support Grant 5P30CA044579, and the Translational Adult Glioma Award from the Ben and Catherine Ivy Foundation (to Roger Abounader).
Funding
This review was supported by the National Institutes of Health grants UO1 CA220841, R21NS122136-01, RO1NS122222 (all to Roger Abounader), the National Cancer Institute Cancer Center Support Grant 5P30CA044579, and the Translational Adult Glioma Award from the Ben and Catherine Ivy Foundation (to Roger Abounader).
Author information
Authors and Affiliations
Contributions
Material preparation was performed by K.H. and M.W.M. Manuscript design and manuscript writing were performed by K.H., M.W.M., C.F., and R.A. All authors (K.H., M.W.M., Y.Z., S.S., M.K.G., C.D., Y.S.,P.M, C.F., R.A) contributed significantly to manuscript draft editing and have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
This is a review manuscript of which no ethical approval is required.
Consent to participate
No individual participants were involved in this review manuscript.
Consent for Publication
No human research participants were involved in this review manuscript.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hudson, K., Mondia, M.W., Zhang, Y. et al. The role of microRNAs in brain metastasis. J Neurooncol 166, 231–241 (2024). https://doi.org/10.1007/s11060-023-04541-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-023-04541-x