Abstract
Invertebrates comprise the most diversified animal group on Earth. Due to their long evolutionary history and small size, invertebrates occupy a remarkable range of ecological niches, and play an important role as “ecosystem engineers” by structuring networks of mutualistic and antagonistic ecological interactions in almost all terrestrial ecosystems. Urban forests provide critical ecosystem services to humans, and, as in other systems, invertebrates are central to structuring and maintaining the functioning of urban forests. Identifying the role of invertebrates in urban forests can help elucidate their importance to practitioners and the public, not only to preserve biodiversity in urban environments, but also to make the public aware of their functional importance in maintaining healthy greenspaces. In this review, we examine the multiple functional roles that invertebrates play in urban forests that contribute to ecosystem service provisioning, including pollination, predation, herbivory, seed and microorganism dispersal and organic matter decomposition, but also those that lead to disservices, primarily from a public health perspective, e.g., transmission of invertebrate-borne diseases. We then identify a number of ecological filters that structure urban forest invertebrate communities, such as changes in habitat structure, increased landscape imperviousness, microclimatic changes and pollution. We also discuss the complexity of ways that forest invertebrates respond to urbanisation, including acclimation, local extinction and evolution. Finally, we present management recommendations to support and conserve viable and diverse urban forest invertebrate populations into the future.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Terrestrial invertebrates, including insects, mites, spiders, millipedes, centipedes, snails and earthworms amongst many others, comprise the most diversified animal group on Earth, accounting for as much as 80% of all known terrestrial animal species (Zhang 2011). They are characterised by the lack of an internal bone skeleton, instead showing a remarkable breadth in body plans ranging from hard exoskeletons (e.g., insects, particularly beetles) to hydrostatic skeletons (e.g., earthworms). Invertebrates occupy a vast range of ecological niches and microhabitats across terrestrial ecosystems and play an important role in structuring networks of mutualistic and antagonistic ecological interactions in almost all terrestrial ecosystems (Ings et al. 2009), thus contributing to key ecosystem services including pollination, nutrient recycling and pest control (Noriega et al. 2018). For these reasons alone, terrestrial invertebrates have long been recognised as ‘the little things that run the world’ (Wilson 1987).
Given their role in physically shaping the environment in which they live, and their capacity to change the availability of resources for other species, invertebrates can be considered as “ecosystem engineers” (Jones et al. 1994). While some invertebrates are intentional engineers that directly alter their environment (e.g., ants and termites who create a network of above- and belowground nests), other species are accidental engineers that modify habitat indirectly or as a by-product of their activities (e.g., earthworms that create temporary tunnels as they move through the soil and provide additional nutrients in the form of excretions).
Here we focus on the role of invertebrates in shaping urban forests and in providing key ecosystem functions and underlying services. We define urban forests as self-regenerating communities with a tree canopy that exist as remnant patches of formerly contiguous habitat now surrounded by urban areas, as well as communities composed of planted trees, such as those in gardens, parks and along streets (see Alvey 2006). Urban forests in their most complex form are composed of an upper tree canopy, one or more understory layers, a groundcover layer and soil layers (Jim 2017). They are also one of the dominant types of natural areas in cities ‒ by one estimate, remnant forests account for 68% of parkland area across the most populous cities in the USA (Pregitzer et al. 2021). There is a large amount of variation in the structure and species composition of urban forests, ranging from structurally complex native forest patches to highly simplified systems dominated by a few exotic species (Threlfall et al. 2016). In many contexts, urban forests constitute novel ecosystems (sensu Hobbs et al. 2006), where plants and animals that do not share an evolutionary history now co-exist, including many invasive species. Urban forests provide a plethora of benefits to citizens, including environmental ecosystem services, i.e., the regulation of air, water, soil and climate, the provision of habitat and other goods and services (Roeland et al. 2019) and cultural ecosystem services, including physical, psychological and social health benefits and economic development (Nesbitt et al. 2017). Invertebrates play a critical role in the functioning of urban forests and therefore in providing these benefits to urban inhabitants.
In this review, we describe the many roles invertebrates play in structuring and maintaining the functioning of urban forests and argue for the conservation of urban forests and their invertebrates in the Anthropocene, the current epoch characterised by climate change, urban densification and habitat loss, and the apparent mass loss of invertebrate biomass worldwide (see Hallmann et al. 2017; Sánchez-Bayo and Wyckhuys 2019; Cardoso et al. 2020; Harvey et al. 2020, but see Macgregor et al. 2019; Crossley et al. 2020). The review covers four broad subject areas related to urban forest invertebrates, with the aim of generating a compendium of evidence to be used in the study, planning and management of urban forests. The subject areas are: 1) the diversity and complexity of invertebrate communities in urban forests with a focus on their roles in the provisioning of various ecosystem services and their contributions to people (Díaz et al. 2018); 2) the major ecological filters affecting species assemblages in urban areas; 3) our current understanding of how urban forest invertebrates respond to these filters and the consequences of these responses for ecosystem service provisioning; and 4) how we can incorporate invertebrates into urban design and management to deliver healthier and more taxonomically and functionally resilient urban forests for the future. This review focuses on evidence in the literature of forest invertebrate community composition and change in human-modified landscapes.
Functional roles of invertebrates in urban forests
Urban forests contain terrestrial and aquatic systems that support invertebrates and their complex and varied life cycle requirements (Wilbur 1980). Our focus is primarily on terrestrial and semi-aquatic invertebrates that occupy the different strata within urban forests, from belowground, to ground level, to understory, sub-canopy and canopy. Terrestrial invertebrates contribute to an array of ecosystem functions (Scudder 2009), which translate into a multitude of services for humans (Prather et al. 2013), but also disservices (Dunn 2010), collectively termed nature’s contribution to people (NCP) (Díaz et al. 2018). In this section, we explore the contributions of invertebrates to urban forests and how these forests support invertebrates performing diverse functional roles, recognising that some species may perform different and multiple functions depending on life cycle stage and that their functions in an ecosystem may change over the course of their life. For instance, both holometabolous (complete metamorphosis) and hemimetabolous (partial metamorphosis) insects can experience remarkable ecological niche shifts while transitioning between larval/nymph and adult life stages, e.g., from herbivorous caterpillars to pollinating butterfly and moth adults, or from predacious aquatic nymph to predacious aerial dragonflies.
Pollination
Pollination refers to the exchange of genetic material between plants via reproduction and is a critical process in the ongoing recruitment of new generations for many plant species. Urban forests are a significant habitat resource for pollinators, which primarily include bees (Anthophila), flies (Diptera), and butterflies and moths (Lepidoptera). For example, wild bee communities in remnant forests are stratified vertically in the forest canopy (Urban-Mead et al. 2021) and contain unique species unable to persist in surrounding built-up areas (Harrison et al. 2018; Landsman et al. 2019). Similarly, urban parks that contain patches of remnant forest host more butterfly species, including woodland specialist species, than parks that contain only planted vegetation (e.g., Kitahara and Fujii 1997). However, planted trees (including exotics) can also be important to pollinators (Buchholz and Kowarik 2019), confirmed by the barcoding of pollen sampled from four bee species in five different EU cities (Müller 2021). Additionally, pollinators supported by urban forests provide pollination services both within these forests and to surrounding urban and rural habitats.
Urban forests provide significant nesting resources for social and solitary wild bees. For instance, social bumblebees will forage in private or community gardens, but queens construct nests in the less-disturbed soils of urban parks and forest edges (McFrederick and LeBuhn 2006). Many cavity-nesting solitary bees nest in logs, snags and stumps, and some, for instance leaf-cutting bees, collect leaves from a variety of trees and shrubs to partition their brood cells in the nest (MacIvor 2016). Many bees, such as species of the genus Xylocopa, depend on dead wood for nesting and are potentially limited by these resources in cities, which are found nearly exclusively in remnant urban forests. More generally, many pollinators rely on a variety of urban land covers to complete their complex life cycles, depending on remnant forest for nesting and flower-rich urban greenspaces for foraging. Consequently, ensuring adequate nesting resources in urban forests will improve pollination in nearby urban greenspaces where it is valued (e.g., in residential and community gardens).
In degraded urban forests, many weedy herbaceous species may be present, which often provide foraging resources for generalist pollinators, including non-native honeybees (Threlfall et al. 2015) that might interact with native bees of conservation concern (Colla and MacIvor 2017). However, weeds can have extended flowering periods, or flower at different times than native plants, expanding the foraging season for many groups of pollinators or potentially ‘filling the gap’ brought about by climate warming if flowering and fruiting phenologies shift and thereby create periods of low resource availability (Sherry et al. 2007). It is therefore important to value and appropriately manage a range of urban forest types, even those perceived as lower quality.
Urban forests also contain many tree and shrub species required by moths and butterflies for oviposition and subsequent offspring development, and the structure of the forest resource in the landscape is important for these taxa. Hardy and Dennis (1999) showed that the proportion of forest in the urban matrix was positively correlated with butterfly diversity. Similarly, Kurylo et al. (2020) found that butterfly species richness increased with tree cover across the urban matrix, and Lintott et al. (2014) found that moth diversity in urban forests increased in larger, older, and less fragmented patches.
Predation
Predation is the mechanisms through which populations of more abundant species are regulated by complex top-down trophic interactions. Invertebrate predator–prey interactions are ubiquitous on the forest floor (epigaeic stratum), with the main taxa involved including carabid and rove beetles (Carabidae, Staphylinidae), ants (Formicidae) and spiders (Araneae). These predators exert top-down control on the epigaeic and edaphic (soil) invertebrate communities, including members of their own guild (i.e., intra-guild predation), thus contributing an important top-down ecological process (predation) that structures communities (Niemelä 1993; Vidal and Murphy 2018). Invertebrate community structure in urban landscapes is, however, different from that in rural landscapes, with a general trend of predacious groups shifting towards smaller-sized species (see Merckx et al. 2018), species capable of flight (Niemelä and Kotze 2009) and thermophilic species (Piano et al. 2017). These differences are in line with the general processes operating in urban landscapes, including habitat fragmentation and degradation and the urban heat-island effect. Furthermore, for the largely predacious carabid beetle taxon, Kotze et al. (2012) argued that due to a long history of urban forest fragmentation, forest specialist species have all but disappeared from boreal cities, like Helsinki, although some remain in highly specialized habitats in the city, such as bogs (Noreika et al. 2015).
Research on the effects of the apparent decoupling of interactions between different trophic levels in urban forests (see Samways et al. 2010) is needed to evaluate the functional importance of this dominant epigaeic predatory guild. A non-urban example illustrates the complex effects of epigaeic predators on ecosystem processes: Lawrence and Wise (2000) showed that the removal of spiders from the forest floor in a secondary oak-hickory-maple forest in Madison County, Kentucky, USA, resulted in increased densities of springtails (Collembola). Yet, rather than an increase in the rate of litter decomposition due to a greater number of springtails, the authors later reported lower decomposition rates in the absence of spiders due, in part, to the release of mesopredators of other potentially important decomposer groups, such as mites (Acari) or flies (Diptera) (Lawrence and Wise 2004).
Urban pest populations often flourish when resources such as food or habitat are increased or novel community structures result in decreases in competition and/or predation (Robinson 1996). Changes in the climate of urban areas – as well as a lack of natural enemies in the case of exotic species – can facilitate pest outbreaks (Meineke et al. 2013) and associated economic consequences (Kovacs et al. 2010), such as the northward expansion by the emerald ash borer (Agrilus planipennis) into Canadian cities and towns (Herms and McCullough 2014) and the hemlock woolly adelgid (Adelges tsugae) across the Northeastern USA (Paradis et al. 2008). Arthropod pests such as some species of mosquitoes (Culicidae), cockroaches and termites (Blattodea) and beetles (Coleoptera) require extensive management in cities because they threaten stored products, public health or building structures (Rust 2009). In urban parks and forests, arthropod pests can damage native vegetation (Ciceoi et al. 2017) through elevated levels of herbivory (Christie and Hochuli 2005), or negatively affect native animals through predation or competition. However, arthropod pests may be subject to top-down control in urban areas, as evidenced by decreased foliage loss in large cities across Europe as a result of elevated bird predation (Kozlov et al. 2017). Increases in urban forest pests are also of concern as they can spread to nearby, more natural landscapes, as was shown for the Asian long-horned beetle, Anoplophora glabripennis (Dodds and Orwig 2011).
As indicated above, biological control has the potential to regulate arthropod pests in urban forests, thereby reducing the need for pesticides or other control agents and potentially lowering monetary costs in the long term (Olkowski et al. 1976; Kenis et al. 2017). The success of biological control in urban areas relies on diverse source populations of natural enemies, resource accessibility and the ability of these organisms to permeate through and persist in the urban matrix (Shrewsbury and Leather 2012; Frey et al. 2018). For example, urban vegetation fragments can be an important source for biological control agents such as spiders (Lowe et al. 2018) and parasitoids (Fenoglio et al. 2013), and can increase the diversity of predator communities in nearby urban gardens (Vergnes et al. 2012). Increasing supplementary resources for natural enemies within the urban matrix can also increase biological control services (Ellis et al. 2005; Egerer et al. 2018). However, biological control can be hard to achieve in urban areas as arthropod predator communities are often disrupted, limiting their ability to counter pest populations (Meineke et al. 2014; Gardiner and Harwood 2017).
Herbivory
Herbivory is the process through which the energy plants capture from the sun is transferred to the next level of organisms, and is therefore an essential process for life on Earth. Invertebrate herbivores are a taxonomically diverse and speciose ecological group, dominated by juvenile and adult stages of moths and butterflies (Lepidoptera), beetles (Coleoptera), bugs (Hemiptera), flies (Diptera) and grasshoppers and crickets (Orthoptera). Some are specialist feeders on certain host plants, while others have the capacity to feed on a wide array of hosts (Forister et al. 2019). The sheer diversity and abundance of insect herbivores in urban forests make the interactions between plants and insects a key driver in productivity and nutrient cycling (Hawlena et al. 2012).
Collectively, invertebrate herbivores in urban forests are not a homogenous functional group as they employ an extraordinary array of strategies to consume plant material (Strong et al. 1984). This variation in foraging strategy has equally varied impacts on plants. For example, herbivory can result in substantial reductions in photosynthetic area, the destruction of reproductive structures such as flowers or seeds and, in some instances, can promote disease if invertebrates themselves are disease vectors (e.g., Dutch Elm Disease, Ophiostoma ulmi and O. novo-ulmi), or if their herbivory creates entry points for pathogens. In an urban context, herbivory, when out of control (e.g., gypsy moth infestations in Eastern North America [Moeller et al. 1977; Schultz and Baldwin 1982]), defoliates trees and impacts recreation and the overall appreciation of urban forests (see also the “Disservices” section). Therefore, overabundant invertebrate herbivores in urban forests are typically perceived as pests, particularly when the extent of defoliation is severe and the health of the urban forest is compromised (Raupp et al. 2010).
The engineering role of herbivorous insects is most apparent during population outbreaks that threaten the persistence of key plant species, especially when outbreaks interact with other disturbances such as fire (Parker et al. 2006; Halofsky et al. 2020). A range of factors may contribute to elevated levels of herbivorous insects and thus herbivory in urban forests, such as loss of key predators (Hochuli and Threlfall 2018) or parasitoids (Peralta et al. 2011; Nelson and Forbes 2014), changes in landscape structure and configuration (Fenoglio et al. 2012; Rossetti et al. 2017) and microclimate (Meineke et al. 2013; Dale and Frank 2017). Mechanisms driving the population ecology of insect herbivores remain a key frontier in identifying how their impacts in urban forests can be assessed (see “Invertebrate responses to urban environments” section) and managed (see “Managing urban forests for invertebrates” section).
Dispersal of seeds and microorganisms
As plants and microbes are sessile, their main mechanism for movement into new locations is through the dispersal of seeds, spores and other propagules. While some ant species are known for playing an important role in seed dispersal in urban forests (Thompson and Mclachlan 2007), there is emerging evidence that seeds are also dispersed by other insect taxa such as hornets (Vespa spp.) (Chen et al. 2017), crickets (Grylloidea) (Suetsugu 2020) and dung beetles (Scarabaeoidea) (Milotić et al. 2019). Indeed, there are many examples where plants have co-evolved with invertebrates to such an extent that plants develop specialised structures that enable dispersal by specific taxa (e.g., the elaiosomes on seeds of Acacia spp. that enable dispersal by ants). Yet, such ant-seed dispersal relationships can be disrupted in urban areas, as evidenced by elevated rates of seed dispersal after the restoration of ant communities via urban forest restoration efforts in Sydney, Australia (Lomov et al. 2009). Additionally, invertebrates assist with the movement of fungal spores, bacteria and other microorganisms through intentional (e.g., transporting fruiting bodies of fungi) or incidental means (e.g., through digestion and excretion or via surface adhesion) (Bray and Wickings 2019). For instance, some beetles act as transport for fungi, moving and injecting significant quantities and diversity of spores into dead wood and thus improving decomposition and accelerating the creation of hollows that provide habitat for other organisms (Seibold et al. 2019). The movement and foraging of invertebrate taxa such as earthworms (Grant 1983; Milcu et al. 2006), ants (Beattie and Culver 1982; Christian and Stanton 2004; Rowles and O’Dowd 2009) and dung beetles (deCastro-Arrazola et al. 2020) not only facilitate seed dispersal (and fungal dispersal, see next section) but may be important mediators of germination success and seedling recruitment by protecting seeds from predation and locating seeds in nutrient-rich microsites. Although seed dispersal in urban areas can be a significant driver of urban plant community composition, this interaction remains poorly understood (Cheptou et al. 2008; Johnson et al. 2018). Supporting urban forest invertebrate communities that provide seed and microorganism dispersal could be critical for the species and genetic diversity of urban organisms.
Organic matter decomposition and soil development
The decomposition of organic matter closes the nutrient cycle loop in urban forests by reducing the accumulation of dead material and returning nutrients back to the soil to become available to plants once again. There are many soil- and litter-dwelling invertebrates who perform these important functions. Macro-detritivores (e.g., earthworms, woodlice and millipedes) break down leaf litter into smaller pieces (comminution) making it accessible to micro-detritivores (e.g., springtails, oribatid mites) and bacteria and fungi (David and Handa 2010; Ossola et al. 2017). Estimates across various biomes and ecosystems (not including urban forests) show that the presence of complex decomposer communities, including macro-detritivores and their predators, can accelerate both carbon and nitrogen loss on average by 11% (Handa et al. 2014). Studies in urban habitats remain scarce and are much needed, but recent studies have confirmed the importance of soil faunal community complexity for litter decomposition in both urban gardens (Tresch et al. 2019a) and urban forests (Meyer et al. 2020).
Some invertebrates burrow into the soil but feed on the forest floor (e.g., anecic earthworms), which allows for the incorporation of organic detritus and nutrients from the surface deep into the soil profile, while promoting soil gas exchange and water infiltration (Ossola et al. 2015a). In fire-prone urban ecosystems, the removal of large quantities of plant litter from forests by detritivorous invertebrates can decrease fuel loads and fire risk for neighbouring communities (Buckingham et al. 2015). An increase in detritivore species richness significantly enhances the process of decomposition in urban greenspaces and urban forests, as shown in urban gardens in Switzerland (Tresch et al. 2019a, b) and in urban forests in Melbourne, Australia (Ossola et al. 2016), despite the latter being dominated by exotic species from Europe. The dominance of exotic detritivore species, however, is not uncommon and numerous species are now ubiquitous in cities worldwide due to trade and the movement of soil and plant material (Tóth et al. 2020). For example, historic anthropogenic disturbance, over a century old, best explained the intensity of exotic earthworm invasion in a north-eastern North American peri-urban forest (Beauséjour et al. 2014). Exotic detritivorous earthworms in North American forests change plant species composition by favouring non-native plants and reducing the cover of native species (Craven et al. 2017) and by reducing the diversity and density of soil invertebrates (Ferlian et al. 2017).
Decomposing dead wood, including snags/stags (standing dead trees), old roots and fallen branches, is another important forest resource (e.g., Thorn et al. 2020), but not always assessed in urban forest management (Korhonen et al. 2020). Wood decomposition is a long process occurring in different parts of a tree and at different stages of its life, thus providing nursery and refuge resources (i.e., a habitat tree, see Bauerle and Nothdurft 2011) to many taxa and from different trophic levels. For example, dead wood can provide important habitat to springtail (Collembola) communities (Raymond-Leonard et al. 2020). Habitat trees and tree related microhabitats are also particularly important to saproxylic invertebrates, especially jewel beetles (Buprestidae), long-horned beetles (Cerambycidae) and bark beetles (Scolytinae) (Speight 1989; Grove 2002; Kraus et al. 2016) whose larval stage can last up to five years. A specific example is the European stag beetle (Lucanus cervus), which often occurs in warm urban deciduous forests (Harvey et al. 2011). Saproxylic beetles are key actors in ecosystem processes such as wood decomposition and nutrient cycling (Dajoz 2000), and their richness, community composition and genetic diversity depend mainly on tree species identity, decay stage, wood size and volume (Schiegg 2000; Brin et al. 2011) and distribution (Horák 2011, 2018), as well as on the connectivity and management regime of old trees and woody debris (Vandekerkhove et al. 2013). Old trees and woody debris are a critical resource for this group of invertebrates, however these elements are often missing from urban forests due to public safety concerns and aesthetical preferences (Hauru et al. 2014; Le Roux et al. 2014), threatening the persistence of these animals and the functions they perform.
Many saproxylic invertebrates feed on nectar and pollen as adults, thus the distribution and configuration of floral feeding resources (meadows, flowering bushes and trees) outside urban forests are complementary (e.g., Colding 2007) to maintain viable populations within urban forests (Matteson and Langellotto 2010). Since saproxylic invertebrates are generally not highly mobile, such floral resources should be in close proximity to decaying wood in urban forests, or should be well connected through green corridors providing feeding resources and resting places (see also the “Pollination” section).
Disservices
While biodiversity and nature offer many benefits to people, they can also give rise to negative interactions or consequences that can be considered “disservices”. Some examples include property damage by termites or other wood boring insects (e.g., Xylocopa), entomophobia (fear of insects) and major outbreaks of pests, both medical and economic. One of the disservices with the most direct consequences for humans occurs when invertebrates transmit diseases that pose a significant risk to public health (Lyytimäki et al. 2008). Arthropod-borne diseases are of significant concern in urban landscapes (LaDeau et al. 2015), with key groups being mosquitoes (Culicidae) (Lourenço-de-Oliviera et al. 2004; Rochlin et al. 2016; Murdock et al. 2017; Goodman et al. 2018) and ticks (Acari) (Maupin et al. 1991; Stafford and Magnarelli 1993; Frank et al. 1998; Uspensky 2017). The latter rely on vertebrate hosts also being present in forests; therefore, understanding how the interactions between host, tick, and pathogen are affected by characteristics of the urban environment is essential for reducing public health risk (Ostfeld and Keesing 2017). For example, Krystosik et al. (2020) conducted a systematic review and found that solid waste associated with urban landscapes provided a breeding ground for zoonotic disease hosts (often mammals) and invertebrate transmission vectors. Given the potential of public health risks to shape perceptions and management of urban forests, it is vital that risks be assessed and compared against the benefits that these forests provide to nature and humans alike.
Filters acting on urban forest invertebrates
To understand community assembly of urban forest invertebrates and associated ecosystem functions, we must consider how the urban environment acts as an ecological filter of invertebrate traits (Brousseau et al. 2018; Fournier et al. 2020). Filters determining urban pools of species act at different temporal and spatial scales. They include both biophysical and biogeographical constraints, as well as broad-scale human factors such as human-mediated species dispersal at a global scale (Swan et al. 2011; Aronson et al. 2016). Beyond such regional considerations, once a species arrives, they must initially survive the local urban environment to become established. Survival will depend on traits that influence morphology, phenology, physiology and behaviour, enabling individuals to overcome the range of dispersal, abiotic and biotic filters at play in urban landscapes (Brousseau et al. 2018). These include urban landscape configuration, development history and human activity and decision-making, as well as interactions among multiple species (Aronson et al. 2016). For example, invertebrates in urban forests face abiotic filters such as higher temperatures resulting from heat islands (Arnfield 2003; but see Ziter et al. 2019) or pollutants such as noise, light or chemicals (Halfwerk and Slabbekoorn 2015) compared to non-urban forests.
At the bottom strata, soil-dwelling invertebrates in cities are confronted with a soil matrix of diverse origins that may include rubble or other non-native parent material (Pickett et al. 2011). These soils may also exhibit alkaline conditions associated with cement, may be compacted and sealed under impervious surfaces, have contaminants such as salt and heavy metals and be subject to major disruptions such as litter removal (Pickett et al. 2011; Szlavecz et al. 2018). Such soils are associated with changes in the abundance and composition of belowground invertebrate communities (Santorufo et al. 2012) and the composition of aboveground communities (e.g. Do et al. 2014). Altered soil conditions may also lead to invertebrate communities with particular traits that enable survival. For example, in a study of Collembola under urban trees in Italy, species most tolerant to filters such as low organic matter and high heavy metal concentrations were smaller, pigmented, sexually reproducing and had a well-developed jumping apparatus (Santorufo et al. 2014).
Biotic filters may have equally important, if not greater effects on invertebrate communities (see Kraft et al. 2015). The high proportion of non-native plant species in urban areas results in novel resources available to urban forest invertebrates and may influence trophic and non-trophic interactions in these ecosystems (Valentine et al. 2020). For instance, non-native trees as sources of organic matter for invertebrates have the potential to influence the community assembly of collembolans (Raymond-Leonard et al. 2018) and mites (Malloch et al. 2020), and ultimately, litter decomposition rates (Makkonen et al. 2012; but see Finerty et al. 2016).
Cities are thus home to novel community assemblages, including non-native organisms introduced by humans. Indeed, urban areas can be entry points for invasions, for instance for carabid beetles (Spence and Spence 1988) and earthworms (Hendrix et al. 2008). Introduced plants may be intentionally selected for the purposes of urban planning, landscaping or other cultural activities by which they provide ecosystem services or disservices; but plant and invertebrate species may also arrive in cities unintentionally (Padayachee et al. 2017). For example, the harlequin ladybird (Harmonia axyridis) was introduced outside of its native range as a biological control agent, but has now spread to urban areas over several continents (Brown et al. 2011). The following paragraphs explore in further detail how various filters may shape invertebrate communities and their associated ecosystem functions.
Changes in habitat structure and vegetation simplification
Invertebrates inhabiting urban forests can be highly sensitive to changes in habitat structure and composition. For instance, leaf litter and wood detritus are both habitat and trophic resources for detritivores and saproxylic organisms, respectively. As such, the removal of leaf litter and dead wood from urban forests can obliterate communities of these specialised invertebrates (Siitonen 2001; Vandekerkhove et al. 2013; Ossola et al. 2016). In highly frequented forests, soil trampling by humans can cause the reduction of burrows and suitable microhabitats on the forest floor, which negatively affects the cover of understorey forest vegetation (Hamberg et al. 2008). This alters carabid beetle assemblages compared to less trampled areas, yet the responses of individual species may vary, as many forest specialist species have already been lost from urban forests (Kotze et al. 2012). Similarly, when soil becomes compacted or sealed with impervious surfaces, this limits nesting resources for burrowing organisms.
Vegetation structure simplification can lead to a more simplified invertebrate community (Threlfall et al. 2017; Mata et al. 2021). Often, changes in vegetation structure occur as a result of development, management (e.g., mowing), or through invasive species proliferation in city parks (Kühn and Klotz 2006; Cadotte et al. 2017). The latter has been shown to result in a decline in soil micro-invertebrate richness and abundance along an urbanization gradient in Toronto, Canada (Malloch et al. 2020). However, the invasion of urban forests by exotic tree species can accelerate species turnover without decreasing invertebrate richness or abundance (Buchholz et al. 2015). Taxa most affected by the simplification of vegetation structure are phytophagous insects dependent on host plants; for example, butterfly caterpillars feeding on leaves, or bees feeding on pollen and nectar (Bernays and Chapman 1994). Particularly vulnerable are those species that form obligate associations and mutualistic relationships with plants or other invertebrates. For instance, in some urban forests in eastern Australia, the larvae of the imperial hairstreak butterfly (Jalmenus evagoras) feed on a limited set of tree species within the genus Acacia and form a mutualistic relationship with a few ant species of the genus Iridomyrmex that receive nutrient-rich secretions from the larvae in exchange for the protection they provide.
Urban forests often have a history of management that includes the planting of trees that resist urban pollutants and other anthropogenic stressors (Roy et al. 2012). Urban afforestation efforts are typically accompanied by the management of herbaceous, often invasive, plants, including by mowing, herbicide application and physical removal (Oldfield et al. 2013). Management interventions such as these, or their absence, influence the type and number of invertebrates in urban forests, which have both positive and negative impacts on the particular ecosystem services invertebrates provide. For example, some ant and butterfly species that are adapted to sparse and more open forests might be impacted by changes in plant communities that occur through plant succession, shrub encroachment or plant invasion (Ossola et al. 2015b). On the other hand, excessive mowing drastically reduces the number of flowers, reducing invertebrate diversity (Watson et al. 2020), while mown parks and urban grasslands result in lower invertebrate abundance compared to un-mown vegetation (Garbuzov et al. 2015; Norton et al. 2019).
Microclimatic changes
Changes in urban habitat structure can affect microclimatic conditions, and thus invertebrate diversity and ecological processes (Ossola et al. 2016). Microclimates available to urban forest invertebrates are influenced by landscape-level changes (e.g., temperature and windflow; Arnfield 2003) as well as the structure and composition of the forest itself. The interior areas of larger forests are buffered against higher temperatures and drier conditions (and edge effects in general) in the surrounding built-up environment (Chang et al. 2007; Chow et al. 2011) and potentially serve as a refuge for edge-sensitive species. However, edge environments and their associated increases in solar radiation, wind and lower moisture levels are common given the very fragmented nature of urban greenspaces. Habitats close to urban forest edges typically support different communities compared to areas away from edges (Kotze et al. 2012). The extent to which the composition and richness of invertebrate communities change is related to the contrast of the forest edge compared to built infrastructure or non-forest vegetation (Noreika and Kotze 2012; Soga et al. 2013; Davis and Gagné 2018). The interior areas of urban forests are also subject to disturbance, such as trampling, that can lead to dramatic changes in habitat structure and microclimatic conditions similar to those at edges. Notably, forest paths have very different conditions from adjacent vegetation, with trails and roadways leading to more open vegetation cover (Lehvävirta et al. 2006) that create movement corridors (or ‘flyways’) for some aerial insects (e.g., Papilio butterflies; Esaki 1949) and compressed leaf litter and soils (Duffey 1975) that can reduce abundances of carabid beetles adapted to more humid conditions (Lehvävirta et al. 2006; Kotze et al. 2012).
Forest management can also have important effects on microclimate. Many urban tree stands, e.g., along roadsides or in parks with mown lawns, are actively managed to avoid an understory from developing (Jorgensen et al. 2002) leading to very dry and sun-exposed conditions similar to those in grasslands, and subsequent changes in species composition (Norton 2011). Urban forest management can also have the opposite effect. For instance, the suppression of fire in built-up areas (Kareiva et al. 2007) and a reduction in historical coppicing (Rackham 2008) that result in a more closed canopy, change the composition of spider communities where species that are dependent on open vegetation decline in number (Košulič et al. 2016). Additionally, the loss of dead wood, leaf litter and tree hollows in urban forests, particularly those managed for public amenity, reduces microclimates available to invertebrates. These are habitats that provide moist and cooler refuge from higher temperatures (Scheffers et al. 2014) and their loss in urban environments might be detrimental to taxa that depend on these conditions (Sebek et al. 2013).
Pollution
Anthropogenic sensory pollution mainly comes in three forms: acoustic noise, artificial light and chemical substances (Halfwerk and Slabbekoorn 2015), all of which may have varied effects on organisms and communities, including the exclusion of maladapted traits, the alteration of acoustic and visual communication or an increase in fitness through adaptation (Swaddle et al. 2015; Henneken and Jones 2017). The effects of light pollution on invertebrates are the most obvious, ranging from a few insects circling a light in the home, to hundreds stuck in the grille or windshield of an automobile and even thousands or more at street lamps, e.g., during termite nuptial flights. However, while light pollution attracts some insects, it also repels other more photophobic species (see examples in Firebaugh and Haynes 2019). Larger urban forest patches may buffer against the effects of light pollution, but this becomes compromised as fragmentation creates ever smaller forest patches in urban environments (Villarroya-Villalba et al. 2021). Consequently, photophobic invertebrates may become locally extinct, while those attracted to light may disperse to a suboptimal environment where they are exposed to elevated predation, physical harm (automobile collisions) and unfavourable habitat conditions.
Artificial light can also have behavioural effects on invertebrates that use light for communication, like fireflies (Lampyridae). Fireflies inhabit ecologically diverse habitats, from wetlands, grasslands, forests to urban parks, but have lost much of this habitat due to urbanisation (Lewis et al. 2020). Owens et al. (2018) showed that male Aquatica ficta fireflies emit brighter signals with decreased frequency when exposed to artificial light at wavelengths below 533 nm, demonstrating light signal plasticity in these fireflies. Firebaugh and Haynes (2019) showed that fireflies lured to artificial light were less likely to engage in courtship dialogues, arguing that these light-polluted areas act as demographic traps. Recent evidence on moths also suggests that artificial light has the potential to disrupt chemical communication resulting in reduced mating opportunities (van Geffen et al. 2015a). The increasing global presence of artificial light at night is linked to shifts and declines in invertebrate diversity (Knop et al. 2017; Grubisic et al. 2018) and related ecosystem processes (e.g., pollination), which may spill over into diurnal insect communities active during the day (Knop et al. 2017).
Little is known about the effects of anthropogenic noise on invertebrates, and even less so in urban forests. In their review, Morley et al. (2014) identified two studies on invertebrates and noise that found: 1) positive correlations between noise levels and both call frequency and chorusing in the cicada Cryptotympana takasagona in urban parks, and 2) a greater low-frequency component in the songs of male grasshoppers (Chorthippus biguttulus) from noisy roadsides compared to paired quiet areas. In addition, a study by Davis et al. (2018) showed that monarch butterfly (Danaus plexippus) larvae exposed to roadside noise for two hours experienced a significant increase in heart rate, which was interpreted as a stress response. Yet, longer exposure to continuous traffic noise did not elevate heart rates at the end of larval development, suggesting desensitisation. Anthropogenic noise (here compressor noise at a natural gas field in New Mexico, USA) has been shown to decrease the abundances of various arthropod taxa collected with pitfall traps (velvet ants and wolf spiders), while some taxa showed no effect and leafhoppers increased (Bunkley et al. 2017).
Chemical disturbances in urban landscapes arise from both indirect sources (e.g., microplastics and heavy metals) and deliberate applications (e.g., pesticides). The effects of active ingredients in some pesticides (e.g., neonicotinoids) have been well documented (Chagnon et al. 2015; van der Sluijs et al. 2015) and led to their ban in areas such as the European Union. In an urban forest context, chemical contamination is likely to be present either as a legacy from past land-uses (e.g., asbestos or heavy metals), current practices (e.g., the direct spraying of or unintentional drift from pesticides, as well as industrial and vehicular emissions), or from novel sources such as microplastics and nanoparticles. While the effects of these first two sources on invertebrates have been relatively well studied (Eggleton 2020), the impacts of novel sources of chemical pollution (e.g., per- and polyfluoroalkyl substances, PFAS) are still largely unknown. Yet regardless of the source, the overwhelming picture is that the presence of chemical contaminants has played a major role in contributing to the massive declines in insect abundance and diversity over the past 20 years (Forister et al. 2019), and even in interfering with the cognitive ability, i.e., learning and memory of honeybees that may have significant consequences for the vital ecosystem service of pollination (Leonard et al. 2019).
Invertebrate responses to urban environments
In the following section, we look at some of the consequences of the filters discussed above for urban forest invertebrates.
Persistence vs. local extinction
Habitat destruction, reduction, fragmentation and transformation act synergistically with urbanisation-derived threats (New 2009; Kotze et al. 2011), such as the heat island and wind tunnel effects and light, noise and chemical pollution (New 2015). Indeed, many studies have shown the negative relationship between urbanisation and invertebrate species richness (McKinney 2008; Faeth et al. 2011; Mata et al. 2014; Fenoglio et al. 2020; Piano et al. 2020) and trait composition change (Merckx et al. 2018; Fournier et al. 2020). While these studies explored response patterns at the community level, recent studies are increasingly highlighting that the response of invertebrates to urbanisation processes and filters are species- and trait-specific – that is, some species and trait values tend to be absent in extreme environmental conditions, whereas others are present across a wider environmental gradient (Magura et al. 2013; Mata et al. 2014, 2017; Threlfall et al. 2017). Species-specific mechanisms driving the responses of invertebrates to urban filters are poorly understood. Specialist species interact with a narrow subset of mutualistic and prey species, and may be acutely susceptible to local extinctions (Dunn 2005; Kotze et al. 2011). For example, specialist insect herbivores, such as the weevil Cydmaea dorsalis, the leafhopper Pogonella bispinus, the lacebug Radinacantha tasmanica and the plant louse Acizzia keithi that depend on specific plant species are unlikely to persist if one or more urban filters result in the local extirpation of their host plant (Moir et al. 2011).
Adaptation and rapid evolution
The urban filters discussed above have the capacity to drive evolutionary as well as phenotypic change in urban-dwelling populations (Johnson and Munshi-South 2017; Alberti 2015; Alberti et al. 2017a, b; Hopkins et al. 2018). To date, the majority of studies assessing adaptation to urban environments are vertebrate and plant focussed (e.g., Kark et al. 2007; McDonnell and Hahs 2015), with little research on invertebrates in urban forests. However, increasing evidence indicates that invertebrates may show comparable biological responses along an urban–rural cline (Altermatt and Ebert 2016; Kotze et al. 2011; Eggenberger et al. 2019). For instance, arthropods in urban environments exhibit increased intraspecific variation in morphology (Weller and Ganzhorn 2004; Magura et al. 2006; Lowe et al. 2014; Eggenberger et al. 2019), key life history traits (Miyashita 1990; Lowe et al. 2016) and behaviour (Kralj-Fišer and Schneider 2012) compared to their rural counterparts. In addition, chemical analyses reveal the presence of human food in the diet of urban ants (Penick et al. 2015).
More recently, the focus has been on exploring the impact of specific anthropogenic stressors on physiological tolerance and behaviour within urban environments, but findings linked to urban forests are scarce. Such studies reveal an increased tolerance for heat in urban ants (Angilletta et al. 2007; Diamond et al. 2018) and carabid beetles (Piano et al. 2017), and higher frequencies in the courtship signals of grasshoppers inhabiting roadside verges (Lampe et al. 2012). Light at night disrupts physiological processes and biological timing (Hopkins et al. 2018), as well as ecosystem functions, such as pollination, carried out by nocturnal and diurnal communities (Knop et al. 2017). Experimental laboratory studies reveal dramatic ecological variation in invertebrates linked directly to the presence of light at night (Durrant et al. 2018; Willmott et al. 2018; van Geffen et al. 2015a, b; van Geffen et al. 2014). The impact of light at night is species-specific and the observed patterns may not reveal the true nature of the ecological trade-offs faced. For example, in spiders, the presence of artificial light at night may disrupt biological timing, affecting growth and development under a resource-controlled laboratory environment (Willmott et al. 2018), but this may be offset by increased nocturnal prey capture rates, as measured in natural environments (Lowe et al. 2014, 2016; Willmott et al. 2019).
Although there are examples of invertebrates that have undergone contemporary evolution to adapt to human activity, there are few documented examples of invertebrate evolution being a direct effect of landscape change (Johnson and Munshi-South 2017). Van’t Hof et al. (2016) demonstrated that industrial pollution accumulating on urban trees in nineteenth century Britain led to greater predation on light-coloured peppered moths (Biston betularia), and so populations underwent mutation for greater melanism. In another study, Jha (2015) showed that gene flow in a bumblebee species (Bombus vosnesenskii) was inhibited by impervious surfaces that replaced natural areas. Reduced flight-to-light behaviour of individuals of the small ermine moth (Yponomeuta cagnagella) from urban (artificial light at night) compared to rural (natural light at night) regions suggests adaptation (Altermatt and Ebert 2016). However, there is limited understanding of how fragmentation via the alteration or removal of urban forests and other greenspaces restricts gene flow and genetic variability in invertebrate species, and whether affected populations are more susceptible to future urbanisation and global change (Santangelo et al. 2018). Novel habitats generated as a result of urbanisation may also increase gene flow for some ubiquitous urban species and pests (Miles et al. 2018), including those that exploit and further damage urban forests. In general, more work is needed to understand how landscape changes in urban areas impact the evolution of urban invertebrates (Miles et al. 2019), both to mitigate evolutionary resistance in pests and foster gene flow within native species populations.
Acknowledging the complexity of responses
There are some general patterns and trends in the responses of urban forest invertebrates to increasing urbanisation that are consistently observed across the diversity of taxa and species. However, there is also an equally diverse array of complex responses that are highly influenced by local context and the specific components of a particular system (see Fournier et al. 2020). For example, at the local scale, insectivorous birds, as well as invertebrate predators, parasites and pathogenic fungi, play an important top-down role in regulating insect numbers in urban tree canopies. Within remnant forests in Sydney, Australia, where small insectivorous birds have been lost from the urban environment, herbivorous insects have been released from predation resulting in higher levels of herbivory (Hochuli et al. 2004). In contrast, recent work from multiple cities in Eastern Europe has found that insectivorous birds are more likely to move into cities, resulting in declines in the density of insects and reduced levels of herbivory (Kozlov et al. 2017). As such, in some cities, urban forests in highly urbanised settings may show enhanced defoliation, whereas in others the impacts of herbivorous insects are diminished through antagonistic trophic interactions. However, changes in bottom-up interactions may be equally at play. Around the world, urban forests are overwhelmingly dominated by very few species (Paquette et al. 2021). Such a simplification of leaf litter traits may lead to a simplification of associated invertebrate consumers. Recent studies in natural forests or plantations have shown a consistent covariation of leaf litter traits and detritivore feeding traits ranging from mesofauna such as springtails (Raymond-Leonard et al. 2019) to detritivorous macrofauna such as millipedes and isopods (Brousseau et al. 2019).
The diversity of invertebrates themselves, and their complex life cycles that include reliance on multiple terrestrial and aquatic habitat types, also mean that it is challenging to identify a common framework that applies across all cities and contexts, and as such, idiosyncratic responses to urbanisation are likely to be common.
Managing urban forests for invertebrates
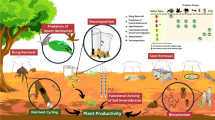
Based on what we have learned from the responses of invertebrates to the urban environment in general, here we make a series of recommendations aimed at maintaining and enhancing invertebrates in urban forests. We believe that the evidence of impacts in urban environments, even if not specific to forests, is sufficient to formulate measures to mitigate the impacts described above. We recommend that the protection and promotion of urban forest invertebrate diversity be a two-pronged approach that seeks to 1) enhance resources for invertebrates, whilst 2) reducing exposure to urban threats (Fig. 1).
Actions to promote the presence and diversity of invertebrates in urban forests. Invertebrate communities are enhanced by maintaining or increasing habitat complexity through the addition of dead and decaying wood, providing areas of exposed soil and clean water bodies, promoting native and indigenous plant species across all strata of the forest and ensuring resources for all life stages. At the same time, actions that reduce deleterious effects of the city include mitigating the urban heat island effect, reducing light, air, water and soil pollution, and managing overabundant, often pest, species. By involving the public in these actions, people are empowered to be stewards of urban forests and the invertebrates they contain
Enhancing resources for invertebrates requires actions such as:
-
Maintaining leaf litter cover and dead and decaying wood to promote a speciose detritivore and saproxylic community (Siitonen 2001; Ossola et al. 2016). Dead wood is a limiting resource in urban environments (Harper et al. 2005; Sebek et al. 2013; Le Roux et al. 2014; Korhonen et al. 2020) but an important refuge from, for instance, increased temperatures (Scheffers et al. 2014). These resources can be reintroduced by actively adding piles of dead wood of different species, sizes and decay stages (Gaston et al. 2005) and in certain configurations to show intent (the ‘cues to care’ concept by Nassauer 1995; Li and Nassauer 2020) together with information posts to explain the benefits of dead wood, changing management to support natural regeneration of dead wood features (Sebek et al. 2013), or introducing artificial structures that mimic these features (Goldingay and Stevens 2009). However, it should be noted that the latter strategy is limited in its benefit as it does not provide feeding resources for target taxa.
-
Maintain areas of exposed soils and soils that have not been overly compacted or otherwise altered by human activities to provide habitat for invertebrates with a soil-dwelling (e.g., earthworms, ants or beetles) or soil-nesting (e.g., some bees and wasps) life stage. Additionally, limiting the sealing and compaction of soils by reducing human use to a small number of trails or areas will benefit ground beetles, ground-nesting bees, and other epigeic and endogeic groups (Cane et al. 2006; Lehvävirta et al. 2006; Galli et al. 2015).
-
Protect, restore and create water bodies, including natural and human-made permanent or semi-permanent standing or running sources of water, for invertebrates with aquatic life stages. These species can be further supported through the presence of vegetation and natural surfaces (e.g., sand or mud) and minimal macro, micro and chemical pollutants (Forister et al. 2019).
-
Promote indigenous plant species (Mata et al. 2021), and undertake management practices that support them, to ensure that food resources are provided in a form that is accessible to co-evolved native invertebrates. For example, open cup flowers are an essential resource if we wish to support short-tongued native bee species in southeastern Australia’s urban forests (Threlfall et al. 2015), while the maintenance or establishment of a native forest understory layer will benefit the detritivore community, as well as provide habitat and resources for bees, beetles, and wasps among others (Ossola et al. 2016; Threlfall et al. 2017; Mata et al. 2021). Note that we are not advocating for the exclusion of exotic, ornamental plant species, which also provide important resources for many urban forest invertebrates, but do promote native plant species that are important for co-evolved invertebrate-plant relationships.
-
Provide resources for all life stages to support a complex assemblage of invertebrates (see Hauck and Weisser 2015) that require a diversity of habitat types (e.g., dragonflies depend on aquatic and terrestrial habitats to complete their life cycle and aerial moths develop from soil-dwelling larvae) and food sources (e.g., herbivorous caterpillars or xylophagous beetle larvae become nectar-feeding butterflies and beetles, respectively). It is also important to consider that threats and degrading processes may change as organisms proceed through their life cycle, and to manage habitat accordingly. Furthermore, the length of different life stages should be considered when managing dead wood ‒ saproxylic beetles such as the European stag beetle (Lucanus cervus) spend up to five years in decaying roots.
-
Maintain or increase vegetation structural complexity to provide a wide range of food, shelter and other resources that support the invertebrates that feed directly on plant leaves, nectar, flowers, seeds and fruits, and the predatory invertebrates that feed on them. For public managers, the benefits of increasing vegetation structural complexity in urban forests may be public preference for the resulting naturalistic character this creates (Heyman 2012; Harris et al. 2018), although this may not be a universal phenomenon. Public support for increasing the complexity of an urban forest may be enhanced by co-management programmes that engage with stakeholders to collaboratively achieve common management goals (Tsuchiya et al. 2013), as illustrated by the co-management approach to woodlands in the neighbourhood of Sletten, Denmark (Fors et al. 2018).
While the previous recommendations highlight the positive actions than can enhance urban forests for invertebrates, the following mitigation actions will additionally benefit them by reducing the impact of broader threats and disturbances (Fig. 1). These actions include:
-
Reduce chemical pollutants by reducing inputs at the source, and by prioritising non-chemical control of pests (e.g., biological control (Kenis et al. 2017)) through Integrated Pest Management.
-
Reduce the urban heat island effect and detrimental disturbances through landscape-scale planning, site-scale design, and appropriate management practices. For example, large greenspaces provide cooling benefits and offer a refuge where invertebrates can reduce their exposure to the urban heat island effect (Ziter et al. 2019). Site and landscape design can also provide local refuges where invertebrates can shelter during disturbances, as can management practices such as creating mosaics of different age stands.
-
Reduce light pollution by: (i) maintaining unlit areas; (ii) reducing the duration of lighting; (iii) minimising the ‘trespass’ of light through improved design; (iv) changing the intensity of lighting; (v) adjusting the spectral composition of lighting (Gaston et al. 2012); and (vi) use new technology and lighting systems (Goddard et al. 2021). While night lighting is an important amenity for people, including for their perceived safety, there is also strong evidence that the associated loss of darkness interrupts natural circadian rhythms with negative consequences for human physical and mental health (Cho et al. 2015).
-
Reduce exposure to natural disasters such as catastrophic bushfires or destructive flooding by distributing areas of urban forest across the landscape, and providing areas where invertebrates can retreat to safety (or repopulate landscapes) such as upland areas in floodplains, or reducing the risk of catastrophic fire through cultural burning and Indigenous land management practices (McKemey et al. 2019).
-
Manage overabundant invasive species and pests to help prevent completely disrupting urban forest ecosystems by altering invertebrate, vertebrate and plant communities and ecological processes, as has happened with extremely successful invaders, such as Argentine and yellow crazy ants (Linepithema humile, Anoplolepis gracilipes) (Silverman and Brightwell 2008). As urban areas are also often primary sites for biological invasions, monitoring invertebrates can complement biosecurity efforts and allow a more rapid response to emerging pests (Hendrix et al. 2008). Supporting diverse invertebrate communities can help reduce the incidence and rate of invasions by providing a broader range of potential natural enemies (Jones and Leather 2012; Gaudon and Smith 2020).
-
Empower humans to become stewards of ecosystems, through actions such as engaging citizens and other urban stakeholders in the sustainable management of pest species (Lowe et al. 2019) and the conservation of wild, forested land in cities. Creative ways to engage and empower people to uphold conservation could include public demonstrations, art installations, guided walks, or education programmes. This, in turn, will help reduce the growing disconnect between people and nature, and generate stronger support for the other actions outlined in this section.
Research gaps and future research
While we know quite a lot about invertebrates in urban environments, several gaps still remain in our knowledge of these organisms in urban forests. To effectively manage and conserve diverse communities of urban forest invertebrates, we need to build a stronger understanding of how they survive, persist and respond to densifying urban landscapes. For a broader perspective on a research agenda for urban biodiversity, see Knapp et al. (2021).
Some of the important questions for future research on invertebrates in urban forests, but also in urban environments in general, include:
-
i.
At what rate are forest invertebrate numbers, biomass and species declining and how strong is the evidence for a sliding baseline of invertebrate numbers based on the experiences of the people who are doing the assessments? This requires long-term research on population sizes, biomass and species richness in multiple cities and biogeographic areas (Hallmann et al. 2017; Macgregor et al. 2019; Sánchez-Bayo and Wyckhuys 2019), and an understanding of the pitfalls in these long-term studies (Didham et al. 2020).
-
ii.
What are the implications of current and future actions on forest invertebrates and the ability of urban areas to support them under climate extremes (e.g., intensified storm events, droughts and heatwaves) and intentional, e.g., the planting of street trees, and unintentional changes in vegetation (Ossola et al. 2020)?
-
iii.
How do forest invertebrates respond to different types of disturbance events (e.g., catastrophic wildfires versus Indigenous cultural burning practices) and what are the underlying mechanisms behind these responses (Erenler et al. 2020; Filazzola et al. 2021)?
-
iv.
What are the metapopulation and metacommunity structures and dynamics present in urban forest invertebrate communities? What can highly fragmented, isolated and dynamic patches of forests in cities reveal with respect to theories on species and community occupancy (Turrini and Knop 2015)?
-
v.
What are the effects of city history and historical landscapes (legacy effects) on contemporary invertebrate communities (e.g., Lindborg and Eriksson 2004; du Toit et al. 2016 for plants; Ossola et al. 2021)? Furthermore, how do invertebrate communities change over time in different urban spatial contexts and are priority effects, i.e., the impacts on a community based on the order or timing of species arrival (Fukami 2015), important in this process?
-
vi.
What are the pre-existing adaptations that enable invertebrates to persist in cities now and in the future, and what are the eco-evolutionary responses that are emerging in invertebrates in response to current and future urban filters and pressures (Alberti 2015)? What are the potential consequences of these changes on future urban forest invertebrate assemblages and their ability to deliver critical ecosystem services?
-
vii.
To what extent do cities act as a conservation refuge for endangered and vulnerable species that might struggle to survive in human-dominated peri-urban landscapes, such as those affected by intensive agriculture, forest logging, or desertification (Hall et al. 2017)?
Conclusion
Invertebrates play a key role in supporting healthy urban environments for people, as reflected in their diverse and varied contributions to the functioning of urban forest ecosystems. While many environmental filters are similar in the urban milieu across the globe, individual responses to these filters are highly variable, reflecting the diversity and complexity of invertebrate ecology and life cycles. Given the current concern of an acute loss of invertebrate biomass, abundance and diversity, the conservation of forests in urban environments, and the invertebrates that support key ecosystem processes and underlying services, is even more critical. Delivering healthier and more resilient urban systems into the future requires urgent action to enhance the role of invertebrates as ecosystem engineers of urban forests. This can be achieved by actively promoting access to essential urban forest resources for a broad variety of organisms, combined with management actions to reduce the negative impacts of urban environments, such as habitat loss and air, noise and light pollution. Our success in safeguarding ecosystem functions and processes in the face of an ever-densifying human population truly does rely on ‘the little things that run the world’.
Availablity of data and material
NA.
Code availability
NA.
References
Alberti M (2015) Eco-evolutionary dynamics in an urbanizing planet. Trends Ecol Evol 30:114–126
Alberti M, Correa C, Marzluff JM, Hendry AP, Palkovacs EP, Gotanda KM, Hunt VM, Apgar TM, Zhou Y (2017a) Global urban signatures of phenotypic change in animal and plant populations. PNAS 114:8951–8956
Alberti M, Marzluff J, Hunt VM (2017b) Urban driven phenotypic changes: empirical observations and theoretical implications for eco-evolutionary feedback. Philos Trans R Soc B 372:20160029
Altermatt F, Ebert D (2016) Reduced flight-to-light behaviour of moth populations exposed to long-term urban light pollution. Biol Lett 12:20160111
Alvey AA (2006) Promoting and preserving biodiversity in the urban forest. Urban For Urban Green 5:195–201
Angilletta MJ, Wilson RS Jr, Niehaus AC, Sears MW, Navas CA, Ribeiro PL (2007) Urban physiology: city ants possess high heat tolerance. PLoS ONE 2(2):e258
Arnfield AJ (2003) Two decades of urban climate research: a review of turbulence, exchanges of energy and water, and the urban heat island. Int J Climatol 23:1–26
Aronson MFJ, Nilon CH, Lepczyk CA, Parker TS, Warren PS, Cilliers SS, Goddard MA, Hahs AK, Herzog C, Katti M, La Sorte FA, Williams NSG, Zipperer W (2016) Hierarchical filters determine community assembly of urban species pools. Ecology 97:2952–2963
Bauerle H, Nothdurft A (2011) Spatial modeling of habitat trees based on line transect sampling and point pattern reconstruction. Can J For Res 41:715–727
Beattie AJ, Culver DC (1982) Inhumation: How ants and other invertebrates help seeds. Nature 297:627
Beauséjour R, Handa IT, Lechowicz MJ, Gilbert B, Vellend M (2014) Historical anthropogenic disturbances influence patterns of non-native earthworm and plant invasions in a temperate primary forest. Biol Invasions 17:1267–1281
Bernays EA, Chapman RF (1994) Host-plant selection by phytophagous insects. Chapman & Hall, New York
Bray N, Wickings K (2019) The roles of invertebrates in the urban soil microbiome. Front Ecol Evol Urban Ecol 7:359
Brin A, Bouget C, Brustel H, Jactel H (2011) Diameter of downed woody debris does matter for saproxylic beetle assemblages in temperate oak and pine forests. J Insect Conserv 15:653–669
Brousseau P-M, Gravel D, Handa IT (2018) On the development of a predictive functional trait approach for studying terrestrial arthropods. J Anim Ecol 87:1209–1220
Brousseau PM, Gravel D, Handa IT (2019) Traits of litter-dwelling temperate forest arthropod predators and detritivores covary spatially with traits of their resources. Ecology 100:e02815
Brown PMJ, Thomas CE, Lombaert E, Jeffries DL, Estoup A, Handley LJL (2011) The global spread of Harmonia axyridis (Coleoptera: Coccinellidae): distribution, dispersal and routes of invasion. Biocontrol 52:623–641
Buchholz S, Kowarik I (2019) Urbanisation modulates plant-pollinator interactions in invasive vs. native plant species. Sci Rep 9:6375
Buchholz S, Tietze H, Kowarik I, Schirmel J (2015) Effects of a major tree invader on urban woodland arthropods. PLoS ONE 10(9):e0137723
Buckingham S, Murphy N, Gibb H (2015) The effects of fire severity on macroinvertebrate detritivores and leaf litter decomposition. PLoS ONE 10(4):e0124556
Bunkley JP, McClure CJW, Kawahara AY, Francis CD, Barber JR (2017) Anthropogenic noise changes arthropod abundances. Ecol Evol 7:2977–2985
Cadotte MW, Yasui SLE, Livingstone S, MacIvor JS (2017) Are urban systems beneficial, detrimental, or indifferent for biological invasion? Biol Invasions 19:3489–3503
Cane JH, Minckley RL, Kervin LJ, Roulston TAH, Williams NM (2006) Complex responses within a desert bee guild (Hymenoptera: Apiformes) to urban habitat fragmentation. Ecol Appl 16:632–644
Cardoso P, Barton PS, Birkhofer K, Chichorro F, Deacon C, Fartmann T, Fukushima CS, Gaigher R, Habel JC, Hallmann CA, Hill MJ, Hochkirch A, Kwak ML, Mammola S, Noriega JA, Orfinger AB, Pedraza F, Pryke JS, Roque FO, Settele J, Simaika JP, Stork NE, Suhling F, Vorster C, Samways MJ (2020) Scientists’ warning to humanity on insect extinctions. Biol Conserv 242:108426
Chagnon M, Kreutzweiser D, Mitchell EAD, Morrissey CA, Noome DA, van der Sluijs JP (2015) Risks of large-scale use of systemic insecticides to ecosystem functioning and services. Environ Sci Pollut Res 22:119–134
Chang C-R, Li M-H, Chang S-D (2007) A preliminary study on the local cool-island intensity of Taipei city parks. Landsc Urban Plan 80:386–395
Chen G, Wang ZW, Qin Y, Sun WB (2017) Seed dispersal by hornets: An unusual insect-plant mutualism. J Integr Plant Biol 59:792–796
Cheptou PO, Carrue O, Rouifed S, Cantarel A (2008) Rapid evolution of seed dispersal in an urban environment in the weed Crepis sancta. PNAS 105:3796–3799
Cho Y, Ryu SH, Lee BR, Kim KH, Lee E, Choi J (2015) Effects of artificial light at night on human health: A literature review of observational and experimental studies applied to exposure assessment. Chronobiol Int 32:1294–1310
Chow WTL, Pope RL, Martin CA, Brazel AJ (2011) Observing and modeling the nocturnal park cool island of an arid city: Horizontal and vertical impacts. Theor Appl Climatol 103:197–211
Christian CE, Stanton ML (2004) Cryptic consequences of a dispersal mutualism: Seed burial, elaiosome removal, and seed-bank dynamics. Ecology 85:1101–1110
Christie FJ, Hochuli DF (2005) Elevated levels of herbivory in urban landscapes: are declines in tree health more than an edge effect? Ecol Soc 10:10.
Ciceoi R, Gutue C, Gutue M, Roşca I (2017) Current status of pests associated with urban vegetation in Bucharest area. Acta Zool Bulg Suppl 9:181–190
Colding J (2007) ‘Ecological land-use complementation’ for building resilience in urban ecosystems. Landsc Urban Plan 81:46–55
Colla SR, MacIvor JS (2017) Questioning public perception, conservation policy, and recovery actions for honeybees in North America. Conserv Biol 31:1202–1204
Craven D, Thakur M, Cameron E, Frelich L, Beauséjour R, Blair R, Blossey B, Burtis J, Choi A, Dávalos A, Fahey TJ, Fisichelli NA, Gibson K, Handa IT, Hopfensperger K, Loss SR, Nuzzo V, Maerz JC, Sackett T, Scharenbroch BC, Smith SM, Vellend M, Umek LG, Eisenhauer N (2017) The unseen invaders: introduced earthworms as drivers of change in plant communities in North American forests. Glob Change Biol 23:1065–1074
Crossley MS, Meier AR, Baldwin EM, Berry LL, Crenshaw LC, Hartman GL, Lagos-Kutz D, Nochols DH, Patel K, Varriano S, Snyder WE, Moran MD (2020) No net insect abundance and diversity declines across US Long Term Ecological Research sites. Nat Ecol Evol 4:1368–1376
Dajoz R (2000) Insects and forests: the role and diversity of insects in the forest environment. Intercept Lavoisier, Paris
Dale AG, Frank SD (2017) Warming and drought combine to increase pest insect fitness on urban trees. PLoS ONE 12:e0173844
David JF, Handa IT (2010) The ecology of saprophagous macroarthropods (millipedes, woodlice) in the context of global change. Biol Rev 85:881–895
Davis DE, Gagné SA (2018) Boundaries in ground beetle (Coleoptera: Carabidae) and environmental variables at the edges of forest patches with residential developments. PeerJ 6:e4226
Davis AK, Schroeder H, Yeager I, Pearce J (2018) Effects of simulated highway noise on heart rates of larval monarch butterflies, Danaus plexippus: implications for roadside habitat suitability. Biol Lett 14:20180018
deCastro-Arrazola I, Hortal J, Noriega JA, Sánchez-Piñero F (2020) Assessing the functional relationship between dung beetle traits and dung removal, burial, and seedling emergence. Ecology 101:e03138
Diamond SE, Chick LD, Perez A, Strickler SA, Zhao C (2018) Evolution of plasticity in the city: urban acorn ants can better tolerate more rapid increases in environmental temperature. Conserv Physiol 6:coy030
Díaz S, Pascual U, Stenseke M, Martín-López B, Watson RT, Molnár Z, Hill R, Chan KMA, Baste IA, Brauman KA, Polasky S, Church A, Lonsdale M, Larigauderie A, Leadley PW, van Oudenhoven APE, van der Plaat F, Schröter M, Lavorel S, Aumeeruddy-Thomas Y, Bukvareva E, Davies K, Demissew S, Erpul G, Failler P, Guerra CA, Hewitt CL, Keune H, Lindley S, Shirayama Y (2018) Assessing nature’s contributions to people. Science 359:270–272
Didham RK, Basset Y, Collins CM, Leather SR, Littlewood NA, Menz MHM, Müller J, Packer L, Saunders ME, Schönrogge K, Stewart AJA, Yanoviak SP, Hassall C (2020) Interpreting insect declines: seven challenges and a way forward. Insect Conserv Divers 13:103–114
Do Y, Lineman M, Joo GJ (2014) Carabid beetles in green infrastructures: the importance of management practices for improving the biodiversity in a metropolitan city. Urban Ecosyst 17:661–673
Dodds KJ, Orwig DA (2011) An invasive urban forest pest invades natural environments –Asian longhorned beetle in northeastern US hardwood forests. Can J for Res 41:1729–1742
Duffey E (1975) The effects of human trampling on the fauna of grassland litter. Biol Conserv 7:255–274
Dunn RR (2005) Modern insect extinctions, the neglected majority. Conserv Biol 19:1030–1036
Dunn RR (2010) Global mapping of ecosystem disservices: the unspoken reality that nature sometimes kills us. Biotropica 42:555–557
Durrant J, Botha LM, Green MP, Jones TM (2018) Artificial light at night prolongs juvenile development time in the black field cricket, Teleogryllus commodus. J Exp Zool Part B 330:225–233
du Toit MJ, Kotze DJ, Cilliers SS (2016) Landscape history, time lags and drivers of change: urban natural grassland remnants in Potchefstroom, South Africa. Landsc Ecol 31:2133–2150
Egerer MH, Liere H, Bichier P, Philpott SM (2018) Cityscape quality and resource manipulation affect natural enemy biodiversity in and fidelity to urban agroecosystems. Landsc Ecol 33:985–998
Eggenberger H, Frey D, Pellissier L, Ghazoul J, Fontana S, Moretti M (2019) Urban bumblebees are smaller and more phenotypically diverse than their rural counterparts. J Anim Ecol 88:1522–1533
Eggleton P (2020) The state of the world’s insects. Annu Rev Environ Resour 45:61–82
Ellis JA, Walter AD, Tooker JF, Ginzel MD, Reagel PF, Lacey ES, Bennett AB, Grossman EM, Hanks LM (2005) Conservation biological control in urban landscapes: Manipulating parasitoids of bagworm (Lepidoptera: Psychidae) with flowering forbs. Biol Control 34:99–107
Erenler HE, Gillman MP, Ollerton J (2020) Impact of extreme events on pollinator assemblages. Curr Opin Insect Sci 38:34–39
Esaki T (1949) Records on Papilio flyways. J Lepid Soc 3:62
Faeth SH, Bang C, Saari S (2011) Urban biodiversity: patterns and mechanisms. Ann NY Acad Sci 1223:69–81
Fenoglio MS, Rossetti MR, Videla M (2020) Negative effects of urbanization on terrestrial arthropod communities: A meta-analysis. Glob Ecol Biogeogr 29:1412–1429
Fenoglio MS, Srivastava D, Valladares G, Cagnolo L, Salvo A (2012) Forest fragmentation reduces parasitism via species loss at multiple trophic levels. Ecology 93:2407–2420
Fenoglio MS, Videla M, Salvo A, Valladares G (2013) Beneficial insects in urban environments: Parasitism rates increase in large and less isolated plant patches via enhanced parasitoid species richness. Biol Conserv 164:82–89
Ferlian O, Eisenhauer N, Aguirrebengoa M, Camara M, Ramirez-Rojas I, Santos F, Tanalgo K, Thakur MP (2017) Invasive earthworms erode soil biodiversity: A meta-analysis. J Anim Ecol 87:162–172
Filazzola A, Matter SF, MacIvor JS (2021) The direct and indirect effects of extreme climate events on insects. Sci Total Environ 769:145161
Finerty GE, de Bello F, Bílá K, Berg MP, Dias ATC, Pezzatti GB, Moretti M (2016) Exotic or not, leaf trait dissimilarity modulates the effect of dominant species on mixed litter decomposition. J Ecol 104:1400–1409
Firebaugh A, Haynes KJ (2019) Light pollution may create demographic traps for nocturnal insects. Basic Appl Ecol 34:118–125
Forister ML, Pelton EM, Black SH (2019) Declines in insect abundance and diversity: we know enough to act now. Conserv Sci Pract 8:e80
Fors H, Nielsen AB, Konijnendijk van den Bosch CC, Jansson M (2018) From borders to ecotones – private-public co-management of urban woodland edges bordering private housing. Urban for Urban Green 30:46–55
Fournier B, Frey D, Moretti M (2020) The origin of urban communities: From the regional species pool to community assemblages in cities. J Biogeogr 47:615–629
Frank DH, Fish D, Moy FH (1998) Landscape features associated with lyme disease risk in a suburban residential environment. Landsc Ecol 13:27–36
Frey D, Vega K, Zellweger F, Ghazoul J, Hansen D, Moretti M (2018) Predation risk shaped by habitat and landscape complexity in urban environments. J Appl Ecol 55:2343–2353
Fukami T (2015) Historical contingency in community assembly: Integrating niches, species pools, and priority effects. Annu Rev Ecol Evol Syst 46:1–23
Galli L, Bonacchi A, Capurro M, Conti I, Crovetto F, Ferrari C, Conti FD, Menta C (2015) Assessment of the impact of trampling on soil Arthropoda in a Mediterranean habitat. Acta Societatis Zoologicae Bohemicae 79:193–198
Garbuzov M, Fensome KA, Ratnieks FL (2015) Public approval plus more wildlife: twin benefits of reduced mowing of amenity grass in a suburban public park in Saltdean, UK. Insect Conserv Divers 8:107–119
Gardiner MM, Harwood JD (2017) Influence of heavy metal contamination on urban natural enemies and biological control. Curr Opin Insect Sci 20:45–53
Gaston KJ, Smith RM, Thompson K, Warren PH (2005) Urban domestic gardens II: experimental tests of methods for increasing biodiversity. Biodivers Conserv 14:395–413
Gaston KJ, Davies TW, Bennie J, Hopkins J (2012) Reducing the ecological consequences of night-time light pollution: options and developments. J Appl Ecol 49:1256–1266
Gaudon JM, Smith SM (2020) Augmentation of the native North American natural enemies for the biological control of the introduced emerald ash borer in central Canada. Biocontrol 65:71–79
Goddard MA et al (2021) A global horizon scan of the future impacts of robotics and autonomous systems on urban ecosystems. Nat Ecol Evol 5:219–230
Goldingay RL, Stevens JR (2009) Use of artificial tree hollows by Australian birds and bats. Wildl Res 36:81–97
Goodman H, Egizi A, Fonseca DM, Leisnham PT, LaDeau SL (2018) Primary blood-hosts of mosquitoes are influenced by social and ecological conditions in a complex urban landscape. Parasites Vectors 11:218
Grant JD (1983) The activities of earthworms and the fates of seeds. In: Satchell JE (ed) Earthworm Ecology: From Darwin to Vermiculture. Springer, Dordrecht, pp 107–122
Grove SJ (2002) The influence of forest management history on the integrity of the saproxylic beetle fauna in an Australian lowland tropical rainforest. Biol Conserv 104:149–171
Grubisic M, van Grunsven RHA, Kyba CCM, Manfrin A, Hölker F (2018) Insect declines and agroecosystems: does light pollution matter? Ann Appl Biol 173:180–189
Halfwerk W, Slabbekoorn H (2015) Pollution going multimodal: the complex impact of the human-altered sensory environment on animal perception and performance. Biol Lett 11:20141051
Hall DM, Camilo GR, Tonietto RK, Ollerton J, Ahrné K, Arduser M, Ascher JS, Baldock KCR, Fowler R, Frankie G, Goulson D, Gunnarsson B, Hanley ME, Jackson JI, Langellotto G, Lowenstein D, Minor ES, Philpott SM, Potts SG, Sirohi MH, Spevak EM, Stone GN, Threlfall CG (2017) The city as a refuge for insect pollinators. Conserv Biol 31:24–29
Hallmann CA, Sorg M, Jongejans E, Siepel H, Hofland N, Schwan H, Stenmans W, Müller A, Sumser H, Hörren T, Goulson D, de Kroon H (2017) More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLoS ONE 12(10):e0185809
Halofsky JE, Peterson DL, Harvey BJ (2020) Changing wildfire, changing forests: The effects of climate change on fire regimes and vegetation in the Pacific Northwest, USA. Fire Ecol 16:4
Hamberg L, Lehvävirta S, Malmivaara-Lämsä M, Rita H, Kotze DJ (2008) The effects of habitat edges and trampling on understorey vegetation in urban forests in Helsinki, Finland. Appl Veg Sci 11:81–96
Handa IT, Aerts R, Berendse F, Berg MP, Bruder A, Butenschoen O, Chauvet E, Gessner MO, Jabiol J, Makkonen M, McKie BG, Malmqvist B, Peeters ETHM, Scheu S, Schmid B, van Ruijven J, Vos VCA, Hättenschwiler S (2014) Consequences of biodiversity loss for litter decomposition across biomes. Nature 509:218–221
Hardy PB, Dennis RL (1999) The impact of urban development on butterflies within a city region. Biodivers Conserv 8:1261–1279
Harper MJ, McCarthy MA, van der Ree R (2005) The abundance of hollow-bearing trees in urban dry sclerophyll forest and the effect of wind on hollow development. Biol Conserv 122:181–192
Harris V, Kendal D, Hahs AK, Threlfall CG (2018) Green space context and vegetation complexity shape people’s preferences for urban public parks and residential gardens. Landsc Res 43:150–162
Harrison T, Gibbs J, Winfree R (2018) Forest bees are replaced in agricultural and urban landscapes by native species with different phenologies and life-history traits. Glob Change Biol 24:287–296
Harvey DJ, Gange AC, Hawes CJ, Rink M (2011) Bionomics and distribution of the stag beetle, Lucanus cervus (L.) across Europe. Insect Conserv Divers 4:23–38
Harvey JA, Heinen R, Armbrecht I, Basset Y, Baxter-Gilbert JH, Bezemer TM, de Kroon H (2020) International scientists formulate a roadmap for insect conservation and recovery. Nat Ecol Evol 4:174–176
Hauck TE, Weisser WW (2015) AAD Animal-Aided Design©. Bei der Projektleitung, Freising (ISBN 978–3–00–047519–1)
Hauru K, Koskinen S, Kotze DJ, Lehvävirta S (2014) The effects of decaying logs on the aesthetic experience and acceptability of urban forests – Implications for forest management. Landsc Urban Plan 123:114–123
Hawlena D, Strickland MS, Bradford MA, Schmitz OJ (2012) Fear of predation slows plant-litter decomposition. Science 336:1434–1438
Hendrix PF, Callaham MA Jr, Drake JM, Huang C-Y, James SW, Snyder BA, Zhang W (2008) Pandora’s box contained bait: the global problem of introduced earthworms. Annu Rev Ecol Evol Syst 39:593–613
Henneken J, Jones TM (2017) Pheromones based sexual selection in a rapidly changing world. Curr Opin Insect Sci 24:84–88
Herms DA, McCullough DG (2014) Emerald ash borer invasion of North America: history, biology, ecology, impacts, and management. Annu Rev Entomol 59:13–30
Heyman E (2012) Analysing recreational values and management effects in an urban forest with the visitor-employed photography method. Urban for Urban Green 11:267–277
Hobbs RJ, Arico S, Aronson J, Baron JS, Bridgewater P, Cramer VA, Epstein PR, Ewel JJ, Klink CA, Lugo AE, Norton D, Ojima D, Richardson DM, Sanderson EW, Valladares F, Vilà M, Zamora R, Zobel M (2006) Novel ecosystems: theoretical and management aspects of the new ecological world order. Glob Ecol Biogeogr 15:1–7
Hochuli DF, Gibb H, Burrows SE, Christie FJ (2004) Ecology of Sydney’s urban fragments: has fragmentation taken the sting out of insect herbivory? In: Lunney D, Burgin S (eds) Urban wildlife: More than meets the eye. Royal Zoological Society of New South Wales, Mosman NSW, pp 63–69
Hochuli DF, Threlfall CG (2018) Planning for protection: Promoting pest suppressing urban landscapes through habitat management. In: Ossola A, Niemelä J (eds) Urban biodiversity: From research to practice. Routledge, London, UK, pp 54–70
Hopkins GR, Gaston KJ, Visser ME, Elgar MA, Jones TM (2018) Artificial light at night as a driver of evolution across urban–rural landscapes. Front Ecol Environ 16:472–479
Horák J (2011) Response of saproxylic beetles to tree species composition in a secondary urban forest area. Urban for Urban Green 10:213–222
Horák J (2018) The Role of urban environments for saproxylic insects. In: Ulyshen MD (ed) Saproxylic Insects. Springer, Singapore, pp 835–846
Ings TC, Montoya JM, Bascompte J, Blüthgen N, Brown L, Dormann CF, Edwards F, Figueroa D, Jacob U, Jones JI, Lauridsen RB, Ledger ME, Lewis HM, Olesen JM, van Veen F, Warren PH, Woodward G (2009) Ecological networks – beyond food webs. J Anim Ecol 78:253–269
Jha S (2015) Contemporary human-altered landscapes and oceanic barriers reduce bumble bee gene flow. Mol Ecol 24:993–1006
Jim CY (2017) Conservation and creation of urban woodlands. In: Tan PY, Jim CY (eds) Greening cities: forms and functions. Springer, Singapore, pp 307–330
Johnson AL, Borowy D, Swan CM (2018) Land use history and seed dispersal drive divergent plant community assembly patterns in urban vacant lots. J Appl Ecol 55:451–460
Johnson MT, Munshi-South J (2017) Evolution of life in urban environments. Science 358:eaam8327
Jones CG, Lawton JH, Shachak M (1994) Organisms as ecosystem engineers. Oikos 68:373–386
Jones EL, Leather SR (2012) Invertebrates in urban areas: A review. Eur J Entomol 109:463–478
Jorgensen A, Hitchmough J, Calvert T (2002) Woodland spaces and edges: their impact on perception of safety and preference. Landsc Urban Plan 60:135–150
Kareiva P, Watts S, McDonald R, Boucher T (2007) Domesticated nature: shaping landscapes and ecosystems for human welfare. Science 316:1866–1869
Kark S, Iwaniuk A, Schalimtzek A, Banker E (2007) Living in the city: can anyone become an ‘urban exploiter’? J Biogeogr 34:638–651
Kenis M, Hurley BP, Hajek AE, Cock MJW (2017) Classical biological control of insect pests of trees: facts and figures. Biol Invasions 19:3401–3417
Kitahara M, Fujii K (1997) An island biogeographical approach to the analysis of butterfly community patterns in newly designed parks. Res Popul Ecol 39:23–35
Knapp S, Aronson MFJ, Carpenter E, Herrera-Montes A, Jung K, Kotze DJ, La Sorte FA, Lepczyk CA, MacGregor-Fors I, MacIvor JS, Moretti M, Nilon CH, Piana MR, Rega Brodsky CC, Salisbury A, Threlfall CG, Trisos C, Williams NSG, Hahs AK (2021) A research agenda for urban biodiversity in the global extinction crisis. BioScience 71:268–279
Knop E, Zoller L, Ryser R, Gerpe C, Hörler M, Fontaine C (2017) Artificial light at night as a new threat to pollination. Nature 548:206–209
Korhonen A, Siitonen J, Kotze DJ, Immonen A, Hamberg L (2020) Stand characteristics and dead wood in urban forests: Potential biodiversity hotspots in managed boreal landscapes. Landsc Urban Plan 201:103855
Košulič O, Michalko R, Hula V (2016) Impact of canopy openness on spider communities: implications for conservation management of formerly coppiced oak forests. PLoS ONE 11(2):e0148585
Kotze DJ, Lehvävirta S, Koivula M, O’Hara RB, Spence JR (2012) Effects of habitat edges and trampling on the distribution of ground beetles (Coleoptera, Carabidae) in urban forests. J Insect Conserv 16:883–897
Kotze DJ, Venn S, Niemela J, Spence J (2011) Effects of urbanization on the ecology and evolution of arthropods. In: Niemela J (ed) Urban ecology, patterns processes and applications. Oxford University Press, New York, pp 159–166
Kovacs KF, Haight RG, McCullough DG, Mercader RJ, Siegert NW, Liebhold AM (2010) Cost of potential emerald ash borer damage in U.S. communities, 2009–2019. Ecol Econ 69:569–578
Kozlov MV, Lanta V, Zverev V, Rainio K, Kunavin MA, Zvereva EL (2017) Decreased losses of woody plant foliage to insects in large urban areas are explained by bird predation. Glob Change Biol 23:4354–4364
Kraft NJB, Adler PB, Godoy O, James EC, Fuller S, Levine JM (2015) Community assembly, coexistence and the environmental filtering metaphor. Funct Ecol 29:592–599
Kralj-Fišer S, Schneider JM (2012) Individual behavioural consistency and plasticity in an urban spider. Anim Behav 84:197–204
Kraus D, Bütler R, Krumm F, Lachat T, Larrieu L, Mergner U, Paillet Y, Rydkvist T, Schuck A, Winter S (2016) Catalogue of tree microhabitats – Reference field list. https://informar.eu/tree-microhabitats
Krystosik A, Njoroge G, Odhiambo L, Forsyth JE, Mutuku F, LaBeaud AD (2020) Solid wastes provide breeding sites, burrows, and food for biological disease vectors, and urban zoonotic reservoirs: a call to action for solutions-based research. Front Public Health 7:405
Kurylo JS, Threlfall CG, Parris KM, Ossola A, Williams NSG, Evans KL (2020) Butterfly richness and abundance along a gradient of imperviousness and the importance of matrix quality. Ecol Appl 30:e02144
Kühn I, Klotz S (2006) Urbanization and homogenization–comparing the floras of urban and rural areas in Germany. Biol Conserv 127:292–300
LaDeau SL, Allan BF, Leisnham PT, Levy MZ (2015) The ecological foundations of transmission potential and vector-borne disease in urban landscapes. Funct Ecol 29:889–901
Lampe U, Schmoll T, Franzke A, Reinhold K (2012) Staying tuned: grasshoppers from noisy roadside habitats produce courtship signals with elevated frequency components. Funct Ecol 26:1348–1354
Landsman AP, Ladin ZS, Gardner D, Bowman JL, Shriver G, D’Amico V, Delaney DA (2019) Local landscapes and microhabitat characteristics are important determinants of urban–suburban forest bee communities. Ecosphere 10:e02908
Lawrence KL, Wise DH (2000) Spider predation on forest-floor Collembola and evidence for indirect effects on decomposition. Pedobiologia 44:33–39
Lawrence KL, Wise DH (2004) Unexpected indirect effect of spiders on the rate of litter disappearance in a deciduous forest. Pedobiologia 48:149–157
Lehvävirta S, Kotze DJ, Niemelä J, Mäntysaari M, O’Hara B (2006) Effects of fragmentation and trampling on carabid beetle assemblages in urban woodlands in Helsinki, Finland. Urban Ecosyst 9:13–26
Leonard RJ, Pettit TJ, Irga P, McArthur C, Hochuli DF (2019) Acute exposure to urban air pollution impairs olfactory learning and memory in honeybees. Ecotoxicology 28:2056–2062
Le Roux DS, Ikin K, Lindenmayer DB, Blanchard W, Manning AD, Gibbons P (2014) Reduced availability of habitat structures in urban landscapes: Implications for policy and practice. Landsc Urban Plan 125:57–64
Lewis SM, Wong CH, Owens ACS, Fallon C, Jepsen S, Thancharoen A, Wu C, De Cock R, Novák M, López-Palafox T, Choo V, Reed JM (2020) A global perspective on firefly extinction threats. Bioscience 70:157–167
Li J, Nassauer JI (2020) Cues to care: a systematic analytical review. Landsc Urban Plan 201:103821
Lindborg R, Eriksson O (2004) Historical landscape connectivity affects present plant species diversity. Ecology 85:1840–1845
Lintott PR, Bunnefeld N, Fuentes-Montemayor E, Minderman J, Blackmore LM, Goulson D, Park KJ (2014) Moth species richness, abundance and diversity in fragmented urban woodlands: implications for conservation and management strategies. Biodivers Conserv 23:2875–2829
Lomov B, Keith DA, Hochuli DF (2009) Linking ecological function to species composition in ecological restoration: Seed removal by ants in recreated woodland. Austral Ecol 34:751–760
Lourenço-de-Oliveira R, Castro MG, Braks MA, Lounibos LP (2004) The invasion of urban forest by dengue vectors in Rio de Janeiro. J Vector Ecol 29:94–100
Lowe EC, Latty T, Webb CE, Whitehouse MEA, Saunders ME (2019) Engaging urban stakeholders in the sustainable management of arthropod pests. J Pest Sci 92:987–1002
Lowe EC, Threlfall CG, Wilder SM, Hochuli DF (2018) Environmental drivers of spider community composition at multiple scales along an urban gradient. Biodivers Conserv 27:829–852
Lowe EC, Wilder SM, Hochuli DF (2014) Urbanisation at multiple scales is associated with larger size and higher fecundity of an orb-weaving spider. PLoS ONE 9(8):e105480
Lowe EC, Wilder SM, Hochuli DF (2016) Persistence and survival of the spider Nephila plumipes in cities: do increased prey resources drive the success of an urban exploiter? Urban Ecosyst 19:705–720
Lyytimäki J, Petersen LK, Normander B, Bezák P (2008) Nature as a nuisance? Ecosystem services and disservices to urban lifestyle. Environ Sci 5:161–172
Macgregor CJ, Williams JH, Bell JR, Thomas CD (2019) Moth biomass increases and decreases over 50 years in Britain. Nat Ecol Evol 3:1645–1649
MacIvor JS (2016) DNA barcoding to identify leaf preference of leafcutting bees. Royal Soc Open Sci 3:150623
Magura T, Nagy D, Tóthmérész B (2013) Rove beetles respond heterogeneously to urbanization. J Insect Conserv 17:715–724
Magura T, Tóthmérész B, Lövei GL (2006) Body size inequality of carabids along an urbanisation gradient. Basic Appl Ecol 7:472–482
Makkonen M, Berg MP, Handa IT, Hättenschwiler S, van Ruijven J, van Bogedom P, Aerts R (2012) Highly consistent effects of plant litter identity and functional traits on decomposition across a latitudinal gradient. Ecol Lett 15:1033–1041
Malloch B, Tatsumi S, Seibold S, Cadotte MW, MacIvor JS (2020) Urbanization and plant invasion alter the structure of litter microarthropod communities. J Anim Ecol 89:2496–2507
Mata L, Andersen AN, Morán-Ordóñez A, Hahs AK, Backstrom A, Ives CD, Bickel D, Duncan D, Palma E, Thomas F, Cranney K, Walker K, Shears I, Semeraro L, Malipatil M, Moir ML, Plein M, Porch N, Vesk PA, Smith TR, Lynch Y (2021) Indigenous plants promote insect biodiversity in urban greenspaces. Ecol Appl 31:e02309
Mata L, Goula M, Hahs AK (2014) Conserving insect assemblages in urban landscapes: accounting for species-specific responses and imperfect detection. J Insect Conserv 18:885–894
Mata L, Threlfall CG, Williams NSG, Hahs AK, Malipatil M, Stork NE, Livesley SJ (2017) Conserving herbivorous and predatory insects in urban green spaces. Sci Rep 7:40970
Matteson KC, Langellotto GA (2010) Determinates of inner city butterfly and bee species richness. Urban Ecosyst 13:333–347
Maupin GO, Fish D, Zultowsky J, Campos EG, Piesman J (1991) Landscape ecology of lyme disease in a residential area of Westchester County, New York. Am J Epidemiol 133:1105–1113
McDonnell MJ, Hahs AK (2015) Adaptation and adaptedness of organisms to urban environments. Annu Rev Ecol Evol Syst 46:261–280
McFrederick QS, LeBuhn G (2006) Are urban parks refuges for bumble bees Bombus spp. (Hymenoptera: Apidae)? Biol Conserv 129:372–382
McKemey MB, Patterson ML, Rangers B, Ens EJ, Reid N, Hunter JT, Costello O, Ridges M, Miller C (2019) Cross-cultural monitoring of a cultural keystone species informs revival of Indigenous burning of country in South-Eastern Australia. Hum Ecol 47:893–904
McKinney M (2008) Effects of urbanization on species richness: a review of plants and animals. Urban Ecosyst 11:161–176
Meineke EK, Dunn RR, Frank SD (2014) Early pest development and loss of biological control are associated with urban warming. Biol Lett 10:20140586
Meineke EK, Dunn RR, Sexton JO, Frank SD (2013) Urban warming drives insect pest abundance on street trees. PLoS ONE 8(3):e59687
Merckx T, Souffreau C, Kaiser A, Baardsen LF, Backeljau T, Bonte D, Brans KI, Cours M, Dahirel M, Debortoli N, De Wolf K, Engelen JMT, Fontaneto D, Gianuca AT, Govaert L, Hendrickx F, Higuti J, Lens L, Martens K, Matheve H, Matthysen E, Piano E, Sablon R, Schön I, Van Doninck K, De Meester L, Van Dyck H (2018) Body-size shifts in aquatic and terrestrial urban communities. Nature 558:113–116
Meyer S, Rusterholz H-P, Salamon J-A, Baur B (2020) Leaf litter decomposition and litter fauna in urban forests: Effect of the degree of urbanisation and forest size. Pedobiologia 78:150609
Milcu A, Schumacher J, Scheu S (2006) Earthworms (Lumbricus terrestris) affect plant seedling recruitment and microhabitat heterogeneity. Funct Ecol 20:261–268
Miles LS, Breitbart ST, Wagner HH, Johnson MT (2019) Urbanization shapes the ecology and evolution of plant-arthropod herbivore interactions. Front Ecol Evol 7:310
Miles LS, Dyer RJ, Verrelli BC (2018) Urban hubs of connectivity: contrasting patterns of gene flow within and among cities in the Western black widow spider. Proc Royal Soc B 285:20181224
Milotić T, Baltzinger C, Eichberg C, Eycott AE, Heurich M, Müller J, Noriega JA, Menendez R, Stadler J, Ádám R, Bargmann T, Bilger I, Buse J, Calatayud J, Ciubuc C, Boros G, Jay-Robert P, Kruus M, Merivee E, Miessen G, Must A, Ardali E, Preda E, Rahimi I, Rohwedder D, Rose R, Slade EM, Somay L, Tahmasebi P, Ziani S, Hoffmann M (2019) Functionally richer communities improve ecosystem functioning: Dung removal and secondary seed dispersal by dung beetles in the Western Palaearctic. J Biogeogr 46:70–82
Miyashita T (1990) Decreased reproductive rate of the spider Nephila clavata, inhabiting small woodlands in urban areas. Ecol Res 5:341–351
Moeller GH, Marler R, McCay RE, White WB (1977) Economic Analysis of the Gypsy Moth Problem in the Northeast: III. Impacts on homeowners and managers of recreation areas. Forest Service, US Department of Agriculture, Northeastern Forest Experiment Station (No. 360)
Moir ML, Vesk PA, Brennan KE, Keith DA, McCarthy MA, Hughes L (2011) Identifying and managing threatened invertebrates through assessments of coextinction risk. Conserv Biol 25:787–796
Morley EL, Jones G, Radford AN (2014) The importance of invertebrates when considering the impacts of anthropogenic noise. Proc Royal Soc B 281:20132683
Murdock CC, Evans MV, McClanahan TD, Miazgowicz KL, Tesla B (2017) Fine-scale variation in microclimate across an urban landscape shapes variation in mosquito population dynamics and the potential of Aedes albopictus to transmit arboviral disease. PLoS Negl Trop Dis 11:e0005640
Müller MS (2021) Feeding behavior of urban bees - A study on the feeding behavior of solitary bees in urban areas across five European cities. Department of Evolutionary Biology and Environmental Studies, University of Zurich (MSc thesis)
Nassauer JI (1995) Messy ecosystems, orderly frames. Landsc J 14:161–169
Nelson AE, Forbes AA (2014) Urban land use decouples plant-herbivore-parasitoid interactions at multiple spatial scales. PLoS ONE 9:e102127
Nesbitt L, Hotte N, Barron S, Cowan J, Sheppard SRJ (2017) The social and economic value of cultural ecosystem services provided by urban forests in North America: A review and suggestions for future research. Urban for Urban Green 25:103–111
New TR (2009) Insect species conservation. Cambridge University Press, Cambridge
New TR (2015) Insect conservation and urban environments. Springer, Cham
Niemelä J (1993) Interspecific competition in ground-beetle assemblages (Carabidae): what have we learned? Oikos 66:325–335
Niemelä J, Kotze DJ (2009) Carabid beetle assemblages along urban to rural gradients: A review. Landsc Urban Plan 92:65–71
Noriega JA, Hortal J, Azcárate FM, Berg MP, Bonada N, Briones MJI, Del Toro I, Goulson D, Ibanez S, Landis DA, Moretti M, Potts SG, Slade EM, Stout JC, Ulyshen MD, Wackers FL, Woodcock BA, Santos AMC (2018) Research trends in ecosystem services provided by insects. Basic Appl Ecol 26:8–23
Noreika N, Kotze DJ (2012) Forest edge contrasts have a predictable effect on the spatial distribution of carabid beetles in urban forests. J Insect Conserv 16:867–881
Noreika N, Pajunen T, Kotze DJ (2015) Urban mires as hotspots of epigaeic arthropod diversity. Biodivers Conserv 24:2991–3007
Norton BA (2011) The sanitisation of urban ecosystems: simplification of the ground layer in eucalypt woodlands and the effects on arthropod communities. Science Botany, The University of Melbourne (PhD thesis)
Norton BA, Bending GD, Clark R, Corstanje R, Dunnett N, Evans KL, Grafius DR, Gravestock E, Grice SM, Harris JA, Hilton S, Hoyle H, Lim E, Mercer TG, Pawlett M, Pescott OL, Richards JP, Southon GE, Warren PH (2019) Urban meadows as an alternative to short mown grassland: effects of composition and height on biodiversity. Ecol Appl 29:e01946
Oldfield EE, Warren RJ, Felson AJ, Bradford MA (2013) Challenges and future directions in urban afforestation. J Appl Ecol 50:1169–1177
Olkowski W, Olkowski H, van den Bosch R, Hom R (1976) Ecosystem management: A framework for urban pest control. Bioscience 26:384–389
Ossola A, Aponte C, Hahs AK, Livesley SJ (2017) Contrasting effects of urban habitat complexity on metabolic functional diversity and composition of litter and soil bacterial communities. Urban Ecosyst 20:595–607
Ossola A, Cadenasso ML, Meineke EK (2021) Valuing the role of time in urban ecology. Front Ecol Evol. https://doi.org/10.3389/fevo.2021.620620
Ossola A, Hahs AK, Livesley SJ (2015a) Habitat complexity influences fine scale hydrological processes and the incidence of stormwater runoff in managed urban ecosystems. J Environ Manag 159:1–10
Ossola A, Hahs AK, Nash MA, Livesley SJ (2016) Habitat complexity enhances comminution and decomposition processes in urban ecosystems. Ecosystems 19:927–941
Ossola A, Hoeppner JM, Burley H, Gallagher RV, Beaumont LJ, Leishman MR (2020) The Global Urban Tree Inventory: A database of the diverse tree flora that inhabits the world’s cities. Glob Ecol Biogeogr 29:1907–1914
Ossola A, Nash M, Christie F, Hahs A, Livesley S (2015b) Urban habitat complexity affects species richness but not environmental filtering of morphologically-diverse ants. PeerJ 3:e1356
Ostfeld RS, Keesing F (2017) Is biodiversity bad for your health? Ecosphere 8:e01676
Owens ACS, Meyer-Rochow VB, Yang E-C (2018) Short- and medium-wavelength artificial light influences the flash signals of Aqutica ficta fireflies (Coleoptera: Lampyridae). PLoS ONE 13(2):e0191576
Padayachee AL, Irlich UM, Faulkner KT, Gaertner M, Procheş S, Wilson JRU, Rouget M (2017) How do invasive species travel to and through urban environments? Biol Invasions 19:3557–3570
Paquette A, Sousa-Silva R, Maure F, Cameron E, Belluau M, Messier C (2021) Praise for diversity: a functional approach to reduce risks in urban forests. Urban for Urban Green 62:127157
Paradis A, Elkinton J, Hayhoe K, Buonaccorsi J (2008) Role of winter temperature and climate change on the survival and future range expansion of the hemlock woolly adelgid (Adelges tsugae) in eastern North America. Mitig Adapt Strateg Glob Chang 13:541–554
Parker TJ, Clancy KM, Mathiasen RL (2006) Interactions among fire, insects and pathogens in coniferous forests of the interior western United States and Canada. Agric for Entomol 8:167–189
Penick CA, Savage AM, Dunn RR (2015) Stable isotopes reveal links between human food inputs and urban ant diets. Proc Royal Soc B 282:20142608
Peralta G, Fenoglio MS, Salvo A (2011) Physical barriers and corridors in urban habitats affect colonisation and parasitism rates of a specialist leaf miner. Ecol Entomol 36:673–679
Piano E, De Wolf K, Bona F, Bonte D, Bowler DE, Isaia M, Lens L, Merckx T, Mertens D, Van Kerckvoorde M, De Meester L, Hendrickx F (2017) Urbanization drives community shifts towards thermophilic and dispersive species at local and landscape scales. Glob Change Biol 23:2554–2564
Piano E, Souffreau C, Merckx T, Baardsen LF, Backeljau T, Bonte D, Brans KI, Cours M, Dahirel M, Debortoli N, Decaestecker E, De Wolf K, Engelen JMT, Fontaneto D, Gianuca AT, Govaert L, Hanashiro FTT, Higuti J, Lens L, Martens K, Matheve H, Matthysen E, Pinseel E, Sablon R, Schön I, Stoks R, Van Doninck K, Van Dyck H, Vanormelingen P, Van Wichelen J, Vyverman W, De Meester L, Hendrickx F (2020) Urbanization drives cross-taxon declines in abundance and diversity at multiple scales. Glob Change Biol 26:1196–1211
Pickett ST, Cadenasso ML, Grove JM, Boone CG, Groffman PM, Irwin E, Kaushal SS, Marshall V, McGrath BP, Nilon CH, Poyat RV, Szlavecz K, Troy A, Warren P (2011) Urban ecological systems: Scientific foundations and a decade of progress. J Environ Manage 92:331–362
Prather CM, Pelini SL, Laws A, Rivest E, Woltz M, Bloch CP, Del Toro I, Ho CK, Kominoski J, Newbold TAS, Parsons S, Joern A (2013) Invertebrates, ecosystem services and climate change. Biol Rev 88:327–348
Pregitzer CC, Charlop-Powers S, Bradford MA (2021) Natural area forests in US cities: Opportunities and challenges. J Forest 119:141–151
Raupp MJ, Shrewsbury PM, Herms DA (2010) Ecology of herbivorous arthropods in urban landscapes. Annu Rev Entomol 55:19–38
Rackham O (2008) Ancient woodlands: modern threats. New Phytol 180:571–586
Raymond-Leonard LJ, Bouchard M, Handa IT (2020) Dead wood provides habitat for springtails across a latitudinal gradient of forests in Quebec Canada. For Ecol Manag 472:118237
Raymond-Léonard LJ, Gravel D, Handa IT (2019) A novel set of traits to describe Collembola mouthparts: Taking a bite out of the broad chewing mandible classification. Soil Biol Biochem 138:107608
Raymond-Leonard LJ, Gravel D, Reich PB, Handa IT (2018) Springtail community structure is influenced by functional traits but not biogeographic origin of leaf litter in soils of novel forest ecosystems. Proc Royal Soc B 285:20180647
Robinson WH (1996) Urban entomology: insect and mite pests in the human environment. Chapman & Hall
Rochlin I, Faraji A, Ninivaggi DV, Barker CM, Kilpatrick AM (2016) Anthropogenic impacts on mosquito populations in North America over the past century. Nat Commun 7:13604
Roeland S, Moretti M, Amorim JH, Branquinho C, Fares S, Morelli F, Niinemets U, Paoletti E, Pinho P, Sgrigna G, Stojanovski V, Tiwary A, Sicard P, Calfapietra C (2019) Towards an integrative approach to evaluate the environmental ecosystem services provided by urban forest. J for Res 30:1981–1996
Rossetti MR, Tscharntke T, Aguilar R, Batáry P (2017) Responses of insect herbivores and herbivory to habitat fragmentation: A hierarchical meta-analysis. Ecol Lett 20:264–272
Rowles AD, O’Dowd DJ (2009) New mutualism for old: indirect disruption and direct facilitation of seed dispersal following Argentine ant invasion. Oecologia 158:709–716
Roy S, Byrne J, Pickering C (2012) A systematic quantitative review of urban tree benefits, costs, and assessment methods across cities in different climatic zones. Urban for Urban Green 11:351–363
Rust MK (2009) Urban habitats. In Encyclopedia of Insects, 2nd edn. Academic Press, pp 1025–1027
Samways MJ, McGeoch MA, New TR (2010) Insect Conservation. A Handbook of Approaches and Methods. Oxford University Press, UK
Sánchez-Bayo F, Wyckhuys KAG (2019) Worldwide decline of the entomofauna: A review of its drivers. Biol Conserv 232:8–27
Santangelo JS, Rivkin LR, Johnson MT (2018) The evolution of city life. Proc Royal Soc B 285:20181529
Santorufo L, Cortet J, Arena C, Goudon R, Rakoto A, Morel JL, Maisto G (2014) An assessment of the urban environment on collembolan communities in soils using taxonomy and trait based approaches. App Soil Ecol 78:48–56
Santorufo L, Van Gestel CA, Rocco A, Maisto G (2012) Soil invertebrates as bioindicators of urban soil quality. Environ Pollut 161:57–63
Scheffers BR, Edwards DP, Diesmos A, Williams SE, Evans TA (2014) Microhabitats reduce animal’s exposure to climate extremes. Glob Change Biol 20:495–503
Schiegg K (2000) Effects of dead wood volume and connectivity on saproxylic insect species diversity. Écoscience 7:290–298
Schultz JC, Baldwin IT (1982) Oak leaf quality declines in response to defoliation by gypsy moth larvae. Science 217:149–151
Scudder G (2009) The importance of insects. In: Foottit RG, Adler PH (eds) Insect biodiversity– Science and society. Wiley-Blackwell, Chichester, United Kingdom
Sebek P, Altman J, Platek M, Cizek L (2013) Is active management the key to the conservation of saproxylic biodiversity? Pollarding promotes the formation of tree hollows. PLoS ONE 8:e60456
Seibold S, Müller J, Baldrian P, Cadotte MW, Štursová M, Biedermann PHW, Krah F-S, Bässler C (2019) Fungi associated with beetles dispersing from dead wood - Let’s take the beetle bus! Fungal Ecol 39:100–108
Sherry RA, Zhou X, Gu S, Arnone JA III, Schimel DS, Verburg PS, Wallace LL, Luo Y (2007) Divergence of reproductive phenology under climate warming. PNAS 104:198–202
Shrewsbury PM, Leather SR (2012) Using biodiversity for pest suppression in urban landscapes: Biodiversity and insect pests. John Wiley & Sons, Ltd, pp 293–308
Siitonen J (2001) Forest management, coarse woody debris and saproxylic organisms: Fennoscandian boreal forests as an example. Ecol Bull 49:11–41
Silverman J, Brightwell RJ (2008) The argentine ant: Challenges in managing an invasive unicolonial pest. Annu Rev Entomol 53:231–252
Soga M, Kanno N, Yamaura Y, Koike S (2013) Patch size determines the strength of edge effects on carabid beetle assemblages in urban remnant forests. J Insect Conserv 17:421–428
Speight MCD (1989) Saproxylic invertebrates and their conservation. Council of Europe, Strasburg, p 81
Spence JR, Spence DH (1988) Of ground beetles and men: Introduced species and the synanthropic fauna of Western Canada. Mem Ent Soc Can 144:151–168
Stafford KC, Magnarelli LA (1993) Spatial and temporal patterns of Ixodes scapularis (Acari: Ixodidae) in Southeastern Connecticut. J Med Entomol 30:762–771
Strong DR, Lawton JH, Southwood R (1984) Insects on plants: Community patterns and mechanisms. Blackwell Scientific, Oxford
Suetsugu K (2020) A novel seed dispersal mode of Apostasia nipponica could provide some clues to the early evolution of the seed dispersal system in Orchidaceae. Evol Lett 4:457–464
Swaddle JP, Francis CD, Barber JR, Cooper CB, Kyba CCM, Dominoni DM, Shannon G, Aschehoug E, Goodwin SE, Kawahara AY, Luther D, Spoelstra K, Voss M, Longcore T (2015) A framework to assess evolutionary responses to anthropogenic light and sound. Trends Ecol Evol 30:550–560
Swan CM, Pickett STA, Szlavecz K, Warren P, Willey KT (2011) Biodiversity and community composition in urban ecosystems: Coupled human, spatial, and metacommunity processes. In: Niemelä J, Breuste JH, Guntenspergen G, McIntyre NE, Elmqvist T, James P (eds) Urban Ecology: Patterns, Processes, and Application. Oxford University Press, pp 179–186
Szlavecz K, Yesilonis I, Pouyat R (2018) Soil as a foundation to urban biodiversity. In: Ossola A, Niemelä J (eds) Urban biodiversity- from research to practice. Routledge Taylor & Francis Group
Thompson B, Mclachlan SM (2007) The effects of urbanization on ant communities and myrmecochory in Manitoba, Canada. Urban Ecosyst 10:43–52
Thorn S, Seibold S, Leverkus AB, Michler T, Müller J, Noss RF, Stork N, Vogel S, Lindenmayer DB (2020) The living dead: acknowledging life after tree death to stop forest degradation. Front Ecol Environ 18:505–512
Threlfall CG, Mata L, Mackie JA, Hahs AK, Stork NE, Williams NS, Livesley SJ (2017) Increasing biodiversity in urban green spaces through simple vegetation interventions. J Appl Ecol 54:1874–1883
Threlfall CG, Ossola A, Hahs AK, Williams NSG, Wilson L, Livesley SJ (2016) Variation in vegetation structure and composition across urban green space types. Front Ecol Evol Urban Ecology 4:66
Threlfall CG, Walker K, Williams NSG, Hahs AK, Mata L, Stork N, Livesley SJ (2015) The conservation value of urban green space habitats for Australian native bee communities. Biol Conserv 187:240–248
Tóth Z, Szlavecz K, Schmidt DJE, Hornung E, Setälä H, Yesilonis ID, Kotze DJ, Dombos M, Pouyat R, Mishra S, Cilliers S, Yarwood S, Csuzdi C (2020) Earthworm assemblages in urban habitats across biogeographical regions. App Soil Ecol 151:103530
Tresch S, Frey D, Le Bayon R-C, Zanetta A, Rasche F, Fliessbach A, Moretti M (2019a) Litter decomposition driven by soil fauna, plant diversity and soil management in urban gardens. Sci Total Environ 658:1614–1629
Tresch S, Frey D, Le Bayon R-C, Mäder P, Stehle B, Fliessbach A, Moretti M (2019b) Direct and indirect effects of urban gardening on aboveground and belowground diversity influencing soil multifunctionality. Sci Rep 9:9769
Tsuchiya K, Okuro T, Takeuchi K (2013) The combined effects of conservation policy and co-management alter the understory vegetation of urban woodlands: a case study in the Tama Hills area, Japan. Landsc Urban Plan 110:87–98
Turrini T, Knop E (2015) A landscape ecology approach identifies important drivers of urban biodiversity. Glob Change Biol 21:1652–1667
Urban-Mead KR, Muñiz P, Gillung J, Espinoza A, Fordyce R, van Dyke M, McArt SH, Danforth BN (2021) Bees in the trees: Diverse spring fauna in temperate forest edge canopies. For Ecol Manage 482:118903
Uspensky IV (2017) Blood-sucking ticks (Acarina, Ixodoidea) as an essential component of the urban environment. Entomol Rev 97:941–969 (Zoologicheskii Zhurnal 96:871–898 (in Russian))
Valentine L, Ramalho C, Mata L, Craig M, Kennedy P, Hobbs R (2020) Novel resources: opportunities for and risks to species conservation. Front Ecol Environ 18:558–566
Vandekerkhove K, Thomaes A, Jonsson BG (2013) Connectivity and fragmentation: island biogeography and metapopulation applied to old-growth elements. In: Kraus D, Krumm F (eds) Integrative approaches as an opportunity for the conservation of forest biodiversity. European Forest Institute, pp 104–115
van der Sluijs JP, Amaral-Rogers V, Belzunces LP, Bijleveld van Lexmond MFIJ, Bonmatin J-M, Chagnon M, Downs CA, Furlan L, Gibbons DW, Giorio C, Girolami V, Goulson D, Kreutzweiser DP, Krupke C, Liess M, Long E, McField M, Mineau P, Mitchell EAD, Morrissey CA, Noome DA, Pisa L, Settele J, Simon-Delso N, Stark JD, Tapparo A, Van Dyck H, van Praagh J, Whitehorn PR, Wiemers M (2015) Conclusions of the Worldwide Integrated Assessment on the risks of neonicotinoids and fipronil to biodiversity and ecosystem functioning. Environ Sci Pollut Res 22:148–154
van Geffen KG, Groot AT, Van Grunsven RHA, Donners M, Berendse F, Veenendaal EM (2015a) Artificial night lighting disrupts sex pheromone in a noctuid moth. Ecol Entomol 40:401–408
van Geffen KG, van Eck E, de Boer RA, van Grunsven RHA, Salis L, Berendse F, Veenendaal EM (2015b) Artificial light at night inhibits mating in a Geometrid moth. Insect Conserv Divers 8:282–287
van Geffen KG, van Grunsven RHA, Ruijven J, Berendse F, Veenendaal EM (2014) Artificial light at night causes diapause inhibition and sex-specific life history changes in a moth. Ecol Evol 4:2082–2089
Van’t Hof AE, Campagne P, Rigden DJ, Yung CJ, Lingley J, Quail MA, Hall N, Darby AC, Saccheri IJ (2016) The industrial melanism mutation in British peppered moths is a transposable element. Nature 534:102–105
Vergnes A, Viol IL, Clergeau P (2012) Green corridors in urban landscapes affect the arthropod communities of domestic gardens. Biol Conserv 145:171–178
Vidal MC, Murphy SM (2018) Bottom-up vs. top-down effects on terrestrial insect herbivores: a meta-analysis. Ecol Lett 21:138–150
Villarroya-Villalba L, Casenelles-Abella J, Moretti M, Pinho P, Samson R, Van Mensel A, Chiron F, Zellweger F, Obrist MK (2021) Response of bats and nocturnal insects to urban green areas in Europe. Basic Appl Ecol 51:59–70
Watson CJ, Carignan-Guillemette L, Turcotte C, Maire V, Proulx R (2020) Ecological and economic benefits of low-intensity urban lawn management. J Appl Ecol 57:436–446
Weller B, Ganzhorn JU (2004) Carabid beetle community composition, body size, and fluctuating asymmetry along an urban-rural gradient. Basic Appl Ecol 5:193–201
Wilbur HM (1980) Complex life cycles. Annu Rev Ecol Syst 11:67–93
Willmott NJ, Henneken J, Selleck CJ, Jones TM (2018) Artificial light at night alters life history in a nocturnal orb-web spider. PeerJ 6:e5599
Willmott NJ, Henneken J, Elgar MA, Jones TM (2019) Guiding lights: foraging responses of juvenile nocturnal orb-weavers to the presence of artificial light at night. Ethology 125:289–297
Wilson EO (1987) The little things that run the world - The importance and conservation of invertebrates. Conserv Biol 1:344–346
Zhang Z (2011) Animal biodiversity: An introduction to higher-level classification and taxonomic richness. Zootaxa 3148:7–12
Ziter CD, Pedersen EJ, Kucharik CJ, Turner MG (2019) Scale-dependent interactions between tree canopy cover and impervious surfaces reduce daytime urban heat during summer. PNAS 116:7575–7580
Acknowledgements
Icons in Fig. 1 are licenced through creative commons by Noun Project creators: Luis Prado (US), H Alberto Gongora (CO), Eucalyp, Insticon (IE), Free Fair & Healthy, Poultry National Park, Priyanka (IN), Maxicons (TH), wahyakup (ID), Sundar Yadhav (IN), Made (AU), Creative Mania (IN), supalerk laipawat (TH), Aline Escobar (MX).
Funding
Open Access funding provided by University of Helsinki including Helsinki University Central Hospital. CGT was supported by the Clean Air and Urban Landscapes Hub, funded by the Australian Government’s National Environmental Science Program and an Australian Research Council Discovery Early Career Researcher Fellowship (DE200101226).
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to the writing of this review.
Corresponding author
Ethics declarations
Ethics approval
NA.
Concent to participate
NA.
Consent for publication
The authors give concent to publish this work in Urban Ecosystems.
Conflicts of interest/Competing interests
The authors declare that they have no conflict of interest.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kotze, D.J., Lowe, E.C., MacIvor, J.S. et al. Urban forest invertebrates: how they shape and respond to the urban environment. Urban Ecosyst 25, 1589–1609 (2022). https://doi.org/10.1007/s11252-022-01240-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-022-01240-9