Abstract
Generally, urbanisation affects biodiversity negatively; however, some species thrive in urban areas. One mammalian species thriving in the urban forest mosaic landscape is the vervet monkey (Chlorocebus pygerythrus), particularly in Durban, eThekwini Municipality, KwaZulu-Natal Province, South Africa. Vervet monkeys’ spatial use can provide important ecological information to understand their behavioural plasticity in an urban forest mosaic landscape. We, therefore, captured and collared vervet monkeys with Global Positioning System (GPS)-Ultra High Frequency (UHF) transmitters in two sites of varying degrees of urban and natural land-use types within eThekwini. Three home range methods Minimum Convex Polygon (MCP), Kernel Density Estimator (KDE), and Local Convex Hull (LoCoH) were used to determine individual home range size and core area use. Home range (30.7 ha 95% KDE) and core area (6.5 ha 50% KDE) estimates were relatively small for these vervet monkeys. Forest and thicket, and built-up habitat types were most used by vervet monkeys across the urban forest mosaic landscape. Overall, individuals in this study showed that the vervet monkeys, regardless of the environment, can successfully exist across the urban forest mosaic landscape. The spatial ecology of urban vervet monkeys provides valuable information for future education and management interventions, especially where there are negative human-monkey interactions amongst urban communities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The expanding human population in municipalities is altering existing natural environments. Over half of the world’s population lives in urban areas and could reach 2.5 billion by 2050 (UN DESA 2019). Space is a primary concern as land is converted for anthropogenic infrastructure, housing developments and agriculture (Brueckner 2000). Anthropogenic land-use change caused by urbanisation threatens biodiversity and transforms wildlife habitats (Zipperer et al. 2000; McKinney 2006; Seto et al. 2013). Urban areas are becoming highly modified, with complex landscapes and generally threatening wildlife as anthropogenic activities increase (Bar-Massada et al. 2014; Soulsbury and White 2016). The detrimental effects caused within an anthropogenically transformed landscape have led to local extinctions of endemic wildlife and an overall deterioration of ecosystem functioning (Fischer and Lindenmayer 2007; Lindenmayer et al. 2008; Magle and Fidino 2018). Additionally, landscape modifications and disturbances lead to fragmented populations of flora and fauna species, causing direct or indirect changes in community structures and or behavioural modifications (Berger-Tal and Saltz 2019). Studies reveal that the type and degree of responses to anthropogenic disturbances are variable and species-specific (Battin 2004; Villarreal-Barajas and Martorell 2009; Murphy and Romanuk 2012; Valiente‐Banuet 2015; Downs et al. 2021). In urban areas, human-wildlife interactions often occur between the interface of green spaces and natural patches of habitats (Kretser et al. 2008; Soulsbury and White 2016). As urbanisation gradually increases, the relationship and complexity of human-wildlife interactions increase as green spaces (primarily wildlife habitats) shrink (McCleery et al. 2014). Therefore, it is beneficial to scientists, conservation managers and policymakers to examine the spatial ecology of wildlife species existing in urban mosaic landscapes within green spaces (natural and managed) (Skidmore et al. 2011; Magle et al. 2012; Downs et al. 2021).
An animal’s available spatial area for its natural activities, such as foraging, reproductivity and parental care, is defined as its home range and is important in understanding animal ecology (Burt 1943; Börger et al. 2008). An individual’s home range size is influenced by a range of factors, including habitat availability, intrinsic factors (age and sex), its internal state, feeding, biotic interactions (social interactions, group dynamics, territorial behaviour) and other external factors associated with its state and characteristics (Börger et al. 2008; Giuggioli and Kenkre 2014). The direct link between home range size and habitat use is movement (Van Moorter et al. 2016). Habitat use affects home range size and is facilitated by resource availability and distribution, landscape features and climatic conditions (Van Beest et al. 2011). Variation in home range sizes is species-dependent and generally reflects the habitat quality where resource-rich environments result in reduced home ranges (McLoughlin and Ferguson 2000; Wiegand et al. 2005). Home range and habitat use are important aspects of species ecology to understand species’ behavioural modifications in changing urban mosaic landscapes. Radiotelemetry has dramatically improved, facilitating the detailed study of the behaviour and ecology of wild animals such as primates (Cagnacci et al. 2010; Dore et al. 2020).
Among the class Mammalia, various primate species persist in human-modified environments because of their behavioural flexibility (McKinney 2015; McLennan Spagnoletti and Hockings 2017). Their behavioural diversity is enhanced by their use of bipedalism, brachiation or quadrupedalism, depending on habitat type (Blanchard and Crompton 2011). The vervet monkey (Chlorocebus pygerythrus) (F. Cuvier 1821) is a medium-sized, highly flexible, semi-terrestrial primate that persists despite anthropogenic pressures in urban mosaic landscapes with natural and managed green spaces in South Africa (Patterson et al. 2017; Thatcher et al. 2019a, b; Zungu et al. 2020). They are classified as “Least Concern” in the Red List of Mammals of South Africa, Swaziland, and Lesotho (Turner et al. 2016a, b) and the International Union for Conservation of Nature (IUCN) Red List of Threatened Species (Butynski and de Jong 2019).
Vervet monkey habitat is broad with woodland, riverine woodland, savanna, coastal scrub forest, and fragmented habitats, including cultivated areas and rural and urban environments but generally absent from deserts (Smithers 2012; Turner et al. 2016a, b). These arboreal and terrestrial omnivorous foragers are water-dependent, feeding on various vegetables, fruits, flowers, small insects, some invertebrates, chicks, and eggs (Smithers 2012; Turner et al. 2016a, b; Butynski and de Jong 2019). Vervet monkeys live in a social hierarchy of multi-male and multi-female troops, consisting of about 30–70 individuals, having territories with well-defined boundaries and home ranges varying from 5 to 103 ha (Struhsaker 1967; Smithers 2012; Turner et al. 2016a, b). Some urban areas have relatively few natural predatory threats to vervet monkeys. In the wild, they are preyed upon by crowned (Stephanoaetus coronatus) and martial (Polemaetus bellicosus) eagles, African leopards (Panthera pardus pardus), and African rock pythons (Python sebae) (Seyfarth and Cheney 1990; Smithers 2012; McPherson et al. 2016; Grey et al. 2017; Naude et al. 2019).
Despite its widespread nature, the vervet monkey population in KwaZulu-Natal Province, South Africa, has been affected by numerous anthropogenic activities creating human-wildlife interactions, typically human-monkey conflict (Seoraj-Pillai and Pillay 2017; Thatcher et al. 2019a). They are often persecuted for crop-raiding by farmers (Sillero-Zubiri and Switzer 2001; Siljander et al. 2020; Findlay and Hill 2021) or as pests in urban, industrial, and residential gardens where there are direct interactions between humans and monkeys (Healy and Nijman 2014; Patterson et al. 2017; Thatcher et al. 2019a; pers. obs.). Anthropogenic food items and supplemental feeding of vervet monkeys in urban gardens can affect this further (Thatcher et al. 2019b, 2020). Vervet monkeys experience unnatural injuries and deaths initiated by these anthropogenic influences and consequential negative human-wildlife interactions (Wimberger et al. 2010).
Understanding how this common generalist primate species navigates a heterogeneous, human-dominated landscape and uses the urban-natural mosaic landscape to its advantage is fundamental. We investigated vervet monkey’s spatial ecology to provide sound scientific knowledge that can be integrated into decision-making, and conservation targets and offer management best practice solutions to reduce human-vervet monkey conflict, particularly in urban areas. Our study used geospatial data and analysis tools to understand spatial-ecological factors of vervets to address human-wildlife interactions that influence the species across the urban forest mosaic landscape. Our study documented the home range and habitat use of vervet monkeys using Global Positioning System (GPS)-Ultra High Frequency (UHF) telemetry transmitters (Dore et al. 2020) in areas with varying degrees of human disturbance across the urban forest mosaic landscape. We also compared the habitat use of vervet monkey troops occurring in the north and south of the eThekwini Municipality. Our study provides baseline data for a rehabilitated landfill site (natural) and a residential area (urban) to supplement previous vervet monkey studies in the municipality (Patterson et al. 2019; Thatcher et al. 2019a). Additionally, limited spatial ecological studies have focused on vervet monkeys in urban-residential, rehabilitated or natural areas; our study aimed to address these gaps. Due to the nature and behavioural plasticity, we predicted that the home ranges of vervet monkeys would be larger in the Buffelsdraai rehabilitation site, which has more natural habitat available than the intensely fragmented urban forest mosaic of the Bluff. Additionally, we predicted that vervet monkeys from both troops’ habitat use would be concentrated in the fragmented forest and thicket habitat types within their respective sites. Therefore, we expected their habitat use to show an uneven distribution across habitat types in their respective home ranges.
Methods
Study area
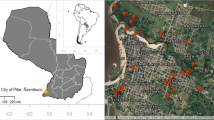
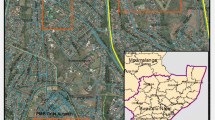
Our study was conducted in Durban, eThekwini Municipality, South Africa, at two study sites: Buffelsdraai Regional Landfill Site and Treasure Beach, the Bluff (Fig. 1). These areas represent the urban forest mosaic landscape that occurs in the region.
The Buffelsdraai Regional Landfill Site (BRLS) (-29.6306, 30.9835) is owned and managed by the eThekwini Municipality. The area of importance in this study is the conservancy area (Conservancy Registration Number C2016/006) of the 2 km buffer zone (787 ha) around the active landfill (116.2 ha). Historically the buffer zone was predominantly used for sugar cane (Saccharum officinarum) plantations (Douwes et al. 2015). In 2008, reforestation activities of the buffer zone began with the planting of sixty-one indigenous tree species at a density of approximately 1300 trees/ha (Roy 2016). Phase two of the forest restoration program took place in 2016 to include existing woodlands, forests, grasslands, wetlands, and riparian areas (Roy 2016). Presently, there are remanent patches of undisturbed indigenous forest interspersed with alien invasive species and old agricultural patches (Roy 2016). The forest type of the site is described as Coastal Scarp and Dry Valley Thicket (MacFarlane et al. 2011), and the vegetation is broadly described as belonging to the KwaZulu-Natal Coastal Belt (Mucina and Rutherford 2006). The summer months (December-February) have the highest precipitation, with an annual rainfall of approximately 766 mm. The daily average temperatures range from 27.4 °C in summer and 22.2 °C in winter (Roy 2016). Although people are present in small numbers all year round, most activities occur in the landfill, the workers’ quarters, the plant nursery, and offices.
Treasure Beach, Bluff (-29.9466, 30.9949) to the south of Durban is a suburban, residential area bordering the second-largest crude oil refinery in South Africa. The Bluff is interspersed with coastal lowland forest patches with the Indian Ocean on its periphery. Precipitation is highest in the summer, with an annual mean rainfall of 975 mm. The mean annual minimum and maximum temperatures are 16.8 and 24.5 °C, respectively (https://en.climate-data.org/africa/south-africa/kwazulu-natal/durban-511/). The approximate population of residents in Treasure Beach are 30 854 (StatSA 2011).
Vervet monkey trapping, collaring and monitoring procedures
We trapped, immobilised, and fitted vervet monkeys with telemetry collars from July 2016 to July 2018 (Table 1). We targeted two individuals from each troop, typically a sub-adult male and an adult female, based on tagging from a previous study (Patterson et al. 2019) to reflect the troop’s spatial ecology. The University of KwaZulu-Natal (no. 020/15/animal) provided animal ethics clearance. Each monkey was captured using a steel walk-in cage trap (70 × 60 × 120 cm) with a pressure pad mechanism that triggered the removable door when a monkey stepped onto it. At dawn, cage traps were baited with commercial fruits (bananas and oranges), peanuts, and raisins placed along a known path that the monkeys frequented (pers. obs.). It was unnecessary to camouflage cage traps as traps were pre-baited seven days before a known trapping event by removing the door - this period allowed the monkeys to become habituated to the traps by providing food. Traps were monitored hourly until sunset or until a monkey was captured and positively identified as suitable for collaring. All captured monkeys were physically assessed and anaesthetised with an intramuscular injection by a veterinarian using a combination of 0.2 ml/2kg Ketamine® and Domitor® (medetomidine hydrochloride). The effects of the anaesthetic were reversed using Antisedan® (atipamezole hydrochloride). Each captured monkey was aged, sexed, weighed, and its body condition was recorded before a transmitter collar was fitted. Additionally, we took photographs for identification, morphological measurements, and faecal, hair and blood samples for DNA. The latter was for a corollary study. We visually identified adult female monkeys (large nipples and canine size), and subadult males were identified by ageing their canines and smaller body size compared with adult males. Monkeys captured that were either too young or physically too small were released. We calculated the body mass index (BMI) for each vervet monkey from the body mass (in kg) divided by length (in meters) squared (Kavanagh et al. 2011).
The GPS-UHF telemeter collar (WW1500AS-TERRESTRIAL, Animal Trackem, Pietermaritzburg, and Wireless Wildlife, Potchefstroom, South Africa) used on each vervet monkey weighed ~ 60 g, < 2% of the body mass of individual monkeys as recommended by Kenward (2001a, b) for animal tracking. We placed a single-finger space gap between the collar and neck of the individual to provide for growth or discomfort if experienced and secured with clear plastic cable ties (100 mm x 2.5 mm), which break away with time. The GPS-UHF telemetry collars were programmed to record a GPS fix (accuracy 5–30 m) at 05h00, 10h00, 15h00 and 20h00, reducing autocorrelation effects (Streicher et al. 2020). Vervet monkeys are diurnal in nature; therefore, the telemeters were switched to ‘sleep mode’ at night to preserve battery life (Baldellou 1998). We downloaded the GPS fixes stored on telemeters using a solar-powered GPS/UHF receiver base station placed at a vantage point (> 6 m) on each study site. Location data were only downloaded when an individual was within range of the base station (5–10 km radius). These data were then uploaded onto a remotely accessed platform, Wireless Wildlife (http://wireless-wildlife.co.za/). Telemeters continued to record GPS fixes until their battery failed. The manufacturer stated that transmitters would function for a minimum of 12 months. However, in our study, all transmitters failed to collect data for a twelve-month cycle, reducing the duration of the tracking to seven months. We downloaded data onto a desktop for statistical analyses.
Home range and core area estimation
We imported GPS data into ArcGIS 10.6 (ESRI, Redlands, CA, USA) and projected these in Universal Transverse Mercator (UTM) (WGS 1984 UTM Zone 36 S). We cleaned the data, removing all outliers or odd GPS fixes manually and tested for site fidelity before analyses (Laver and Kelly 2008). The home range and core area size were estimated using three home range estimates: Maximum Convex Polygon (MCP), Kernel Density Estimator (KDE), and Local Convex Hull (LoCoH) used at the 95% and 50% levels (Worton 1989; Getz et al. 2007; Laver and Kelly 2008). We calculated multiple home range estimates using the reproducible home range (RHR) GUI package in statistical software, R Studio (1.2.909) (R Studio 2015; Signer and Balkenhol 2015), with hplug-in bandwidth as an appropriate smoothing parameter used for studying species in a highly fragmented landscape like the urban mosaic (Laver and Kelly 2008; Walter et al. 2011). The 95% MCPs and 95% KDEs were computed onto layers for the two study sites and exported as maps for visual comparisons displaying the overlap of home ranges of individual vervet monkeys of the same troop.
Statistical analyses
The home range and core area estimate at 95% and 50% levels were reported respectively for individual vervet monkeys collared using the three home range measures. The overall home range estimates, overall sexes and locations were also reported. Due to the relatively low sample size of vervet monkeys collared, we could not do any further statistical analysis concerning home range and core area use. Additionally, no seasonal comparisons were possible as vervet monkeys were collared at random times in the year, with collars failing to obtain complete seasonal data. The body mass, total body length, and BMI of individual vervet monkeys caught during trapping were analysed and compared according to sex and location, respectively, using an independent sample t-test. A one-way ANOVA was applied to determine the difference between vervet monkeys’ location and troop size. For all statistical analyses, the means (± SE) were reported and computed as graphs for visual representation using STATISTICA 7.0 (Statsoft Inc. Tulsa USA).
Habitat use
Vervet monkey habitat use was determined using GPS data obtained and habitat types documented in the two study sites. A land-use map with a 20 m resolution from Ezemvelo KZN Wildlife (EKZNW) was used (Wildlife and GeoTerraImage, 2018). Habitat types and land use features were reclassified into five broad land-use types: built-up, rehabilitated, forest and thicket, grassland and landfill and quarry using the South African National Land Cover map (2017). The available habitat type proportions in the respective vervet monkey individual home ranges were determined using ArcGIS 10.6. We added the GPS points from the 95% MCP home range estimates to the reclassified land-use map to determine habitat types for the observed locations (Streicher et al. 2020). We assessed individual vervet monkey habitat use as the observed proportion of locations in the respective land-use types (Gehrt et al. 2009). We compared the habitat use of vervet monkeys with habitat availability in the study sites and determined the preference or avoidance of a habitat type. We calculated the selection of habitat types using the null model of the differences between the observed and the expected based on habitat availability (Byers et al. 1984). To determine whether habitat use occurred in proportion to available habitat, we used the Chi-square goodness-of-fit analysis, with differences at the 95-percentage probability level, taken as indicating a significant difference between use and availability. We considered the frequency of the GPS fixes of each vervet monkey in the respective habitats to represent the actual use of these habitat types. We compared the proportion of habitat use with the proportion of habitat availability with the Bonferroni confidence interval (CI) (Bonferroni Z-statistic) to determine any significant differences for each habitat type (Byers et al. 1984). A significance level of P < 0.05 was used to determine significance. Lastly, we quantified each habitat type’s percentage use and percentage availability for the vervet monkey population in this study.
Results
Body mass and condition
Vervet monkeys were captured in an equal sex ratio (n = 10) (Table 1). The mean body mass of captured vervet monkeys (n = 10) was 3.9 ± 0.39 kg. Male vervet monkeys (4.4 ± 0.91 kg) were, on average, heavier than female vervet monkeys (3.4 ± 0.80 kg); however, the difference was not significant (t (8) = 1.77, P = 0.11; Fig. 2a). The mean total body length for male vervet monkeys were significantly larger to females (t (8) = 2.43, P = < 0.05; Fig. 2b). Also, the mean BMI did not differ significantly between male and female vervet monkeys (t (8) = 3.17, P = 0.41; Fig. 2c). The mean body mass for vervet monkeys located on the Bluff was 4.5 ± 1.07 kg and for Buffelsdraai was 3.5 ± 0.71 kg (Fig. 2a). The mean total body length for vervet monkeys captured on the Bluff was 113.9 ± 4.19 cm, while for Buffelsdraai, 113.9 ± 7.00 cm. The mean BMI for captured vervet monkeys on the Bluff was 3.4 ± 0.71 cm, while at Buffelsdraai, 2.7 ± 0.42 kg/m2. There were no significant differences between study sites for captured vervet monkey body mass (t (8) = 1.72, P = 0.12), total body length (t (8) = 0.01, P = 0.99) and BMI (t (8) = 2.06, P = 0.07).
The overall mean troop size of vervet monkeys from which individuals were captured in the eThekwini Municipality was 29.8 ± 4.35 (Table 1). The troop size of vervet monkeys (n = 10) was dependent on the study site (Table 1). There was a significant difference in troop size according to the study site (F (1,8) = 18.06, P = < 0.05). Post-hoc Tukey HSD indicated that the mean troop size for vervet monkeys on the Bluff (16.5 ± 4.04) was significantly smaller than those at Buffelsdraai (38.7 ± 3.30).
Home range and core area estimation
A total of ten vervet monkeys were captured and fitted with GPS-UHF tracking collars; however, only six had sufficient data to be analysed. Consequently, we determined home range estimates for six collared vervet monkeys (four females and two males) from the two locations in the urban forest mosaic landscape in eThekwini Municipality between November 2016 and March 2018 (Table 2). These included four females (VM2, VM4, VM13, VM14) and two males (VM3, VM5). The number of GPS fixes from collared vervet monkeys ranged from 59 to 446, varying individually. Over time the GPS-UHF tracking collars began to lose power and eventually stopped obtaining GPS fixes (Table 2). A total of 1491 GPS fixes were recorded during the duration of this study for the collared vervet monkeys. The mean number of days for reporting GPS fixes from collared vervet monkeys was 127.3 ± 16.86 days (Table 2).
The three home range estimates for vervet monkeys showed individual variations in home range and core area size (Fig. 3a-b). The overall mean of the 95% home range (± SE) for vervet monkeys (n = 6) were 47.3 ± 16.04 ha (MCP), 30.7 ± 8.10 ha (KDE), and 23.0 ± 7.00 ha (LoCoH). The overall core areas at the mean of 50% core range for vervet monkeys using the three methods were 17.4 ± 5.51 ha (MCP), 6.5 ± 1.44 ha (KDE) and 3.6 ± 1.08 ha (LoCoH) (Fig. 3c). On average, male vervet monkeys had a slightly more extensive home range than female vervet monkeys for all three home range estimates (Supplementary Fig. S1a), whereas the 50% core area for vervet monkeys showed marginally larger sizes for female than male vervet monkeys (Supplementary Fig. S1b). Overall, the means of the 95% home range (Supplementary Fig. S1c) and 50% core area (Supplementary Fig. S1d) for vervet monkeys were larger at Buffelsdraai than the Bluff (Supplementary Fig. S2). The troops that had two individuals tracked revealed data with similar spatial patterns, supporting that the individual reflected the troop’s movements.
Home range of vervet monkeys (VM) in an urban mosaic landscape where (a) shows individual home range at 95% home range estimates (n = 6); (b) individual home range at 50% home range estimates (n = 6), and (c) the overall mean home range (± SE) of the three home range estimates (Maximum Convex Polygon (MCP), Kernel Density Estimate (KDE) and Local Convex Hull (LCH)) in the present study for all collared vervet monkeys
Habitat use
Overall, vervet monkeys in the eThekwini Municipality showed a significant preference for forest and thicket habitats, as revealed by the Bonferroni Z-statistic computations (Table 3). Built-up and grassland habitats were used by vervet monkeys proportionate to the available habitat. Built-up areas were used by all vervet monkeys regardless of availability. Landfill and quarry appear to be avoided by two vervet monkeys as these were used significantly less than available. However, overall, landfill and quarry did not affect use in proportion to availability by most of the vervet monkeys. Rehabilitated land was used proportionately to the availability and significantly less by vervet monkey VM 4 in Buffelsdraai. Vervet monkey VM 2 used all habitat types proportionally to its availability on the Bluff. Habitat types rehabilitated and landfill and quarry were not present in the Bluff study site. (Table 3). The overall percentage of land-use type used compared with its availability for vervet monkeys were higher for forest and thicket, and built-up, whereas grassland and rehabilitated land-use types were used less even when available. Mines and quarries were significantly used less than available (Fig. 4).
Discussion
Vervet monkeys reported in this study were similar in body mass compared with other studies of urban-living monkeys in KwaZulu-Natal Province, South Africa (Patterson et al. 2019). However, when comparing sexes to previous studies (Turner et al. 2018; Patterson et al. 2019), males and females across an urban forest mosaic in this study had an overall lower body mass. The vervet monkeys from the two study sites differed in body mass by 27%, suggesting that habitat types were partly responsible for the body condition of vervet monkeys. The morphological aspects of vervet monkeys are known to be influenced by many environmental conditions, food consumption, competition, and social dynamics in vervet monkeys (Pasternak et al. 2013; Turner et al. 2016a, b, 2018; Jarrett et al. 2020). The large span of foraging area at Buffelsdraai could have resulted in lower mass and leaner monkeys than those in the urban-residential Bluff site, where monkeys were observed to include anthropogenic food in the diet (pers. obs.). The vervet monkeys in urban areas of the Bluff displayed more aggression for food from bins and waste tips, whereas the Buffelsdraai vervet monkeys generally foraged relatively peacefully in larger troops and fed on more natural foods, which might have impacted their home range sizes (pers. obs.).
Our study reports the home range and habitat use of vervet monkeys using telemeters across an urban forest mosaic landscape of varying degrees of anthropogenic activities in the eThekwini Municipality. Harvey and Clutton-Brock (1981) suggest Cercopithecus species have unusually small home ranges based on their metabolic needs. In the present study, mean home range and core sizes were small when compared with previous vervet monkey studies in South Africa (De Moor and Steffens 1972; Barrett 2004; 2009b; Willems 2007; Pasternak et al. 2013; Patterson et al. 2019). Individual home ranges of vervet monkeys collared in our study illustrated varying sizes but were similar in the size of troops collared in the same region (Patterson et al. 2019). Vervet monkey movements in these contrasting environments indicated different environmental structures and human-induced activities affect these, suggesting that the urban forest mosaic landscape impacted vervet monkeys’ spatial ecology. It is possible that vervet monkey movements are based on their use of cognitive heuristics on available resources (Teichroeb 2015). Additionally, vervet monkey movement data displayed a distinctive home range pattern according to location (Supplementary Fig. S1b).
Our study also showed that vervet monkeys are territorial animals and that troops’ daily movements ranged exclusively in specific areas. The home range for male vervet monkeys presented slightly larger home ranges than females. Male vervet monkeys generally explore their ranges because of dispersal into new troops, which can occur multiple times during their lifespan and throughout the year (Young et al. 2019). However, female vervet monkeys showed a slightly larger range for core area use than their male counterparts. A larger core area use can be explained by female vervets exhibiting tight bonds and philopatry (Seyfarth 1980; Cheney and Seyfarth 1990). In addition, females exhibit familiarity with an area, frequently nursing babies, and demonstrating to juveniles how to forage and protect young, leading to more extensive and longer time spent in core areas (Isbell 2004). Male vervet monkeys typically warn the troop by being vigilant for predators, making alarm calls, or defending territories from neighbouring troops (Henzi 1982; Baldellou and Henzi 1992; Ducheminsky et al. 2014; Snowdon 2020). Although we did not account for the rank of individuals collared in this study, this could have influenced the observed home ranges, and future studies need to investigate such dominance hierarchies. Vervet monkeys had more extensive home range and core area sizes in Buffelsdraai than in the Bluff study site. It was observed in other studies that the primary factor determining vervet monkeys’ spatial use was food resource availability linked to anthropogenic disturbances and infrastructure (Willems et al. 2009; Patterson et al. 2018). The Bluff is a suburban residential area with fragments of natural space, so anthropogenic infrastructure (roads, fences, telephone lines, and residential homes) affected vervet monkeys’ movement. Furthermore, we observed that vervet monkeys on the Bluff were prone to dog attacks, shooting by people, and vehicle collisions (Authors’ unpublished data). In contrast, at Buffelsdraai, large forest corridors, rehabilitated old sugar cane plots, and grasslands with reduced human presence allowed for greater mobility by vervet monkeys (pers. obs.).
Although we did not examine food resources across the urban forest mosaic landscape, the concentration of anthropogenic resources in urban areas also likely contributed to the decrease in home range size, as our result suggests vervet monkeys did need to travel extensively for food. Based on optimal foraging theory, as described by Barrett et al. (2016), vervet monkeys are energy maximisers, selecting resource items in proportion to their energy content irrespective of the variety of resources available in their environment. Regardless of the level of anthropogenic disturbance in both study sites, vervet monkeys showed considerable behavioural flexibility in the present study. Our study suggests that vervet monkey adjusted their use of food resources and intensively used specific areas within their home range where the most nutrient-rich available resources were present and predictably available, as found in other urban primate studies (Riley 2008; Hoffman and O’Riain 2012; McLennan et al. 2017; Thatcher et al. 2020).
In this study, we found that the troop size of vervet monkeys depended on location in urban-residential and the degree of transformed natural areas. The troop sizes on the Bluff were smaller and had smaller home ranges, whereas the Buffelsdraai troops had larger troop sizes and more extensive home ranges. Generally, larger troops require large space to obtain food since intergroup competition forces individuals to travel further and forage over a greater area (Mikula et al. 2018; Teichroeb et al. 2015). This could indicate food and cover resources for vervet monkeys having larger home ranges in areas where woody plant species and larger canopies occur, similarly observed in the vervet monkeys at Buffelsdraai (Barrett et al. 2016). In addition, vervet monkeys live in hierarchical social groups interacting gregariously with various age classes within the troop and communicating about predators and high-quality food resources (Swedell 2012). However, urban vervet monkey troops are at risk of unnatural causes and are exposed to numerous negative human-wildlife interactions, resulting in smaller troop sizes, as seen on the Bluff (Saj 1998; Wimberger and Downs 2010; Pasternak et al. 2013).
Our study highlighted the habitat use of vervet monkeys across an urban forest mosaic landscape. As predicted, there were differences in the habitat use of vervet monkeys living in this landscape. The forest and thicket, and built-up habitat types were used more frequently than other land-use types in the landscape. Furthermore, habitat use of vervet monkeys in Buffelsdraai showed extensive use of forest and thicket habitat types than rehabilitated, landfill and quarry areas. Buffelsdraai vervet monkeys also used built-up and grassland habitat types in proportion to availability, possibly maximising foraging efforts and supplementing their diets with anthropogenic food. Habitat type for vervet monkeys on the Bluff had minimal effect on habitat use, apart from forest and thicket and grassland being used less than available and built-up used in proportion. The flexibility in habitat use suggests that these vervet monkeys can exploit and potentially persist in urban environments without relying on natural habitats, as they can use managed green spaces like gardens and parks (Patterson et al. 2017, 2018). These results further showed vervet monkey behavioural flexibility in habitat use in an urban mosaic landscape.
South African law requires a buffer zone around an active landfill to obscure the view and shield any odours associated with landfill operations protecting any adjacent community. Rehabilitating transformed areas at Buffelsdraai Regional Landfill Site has been a conservation success by increasing indigenous species richness (flora and fauna) (Roy 2016). MacFarlane et al. (2011) reported substantially less biodiversity in transformed areas when compared with remnant patches of untransformed land. Although a considerable proportion of the land has been converted to rehabilitated land, vervet monkeys still preferred forested and thicket habitats, possibly because of a lack of competitors and accessibility to energy-rich food resources (Isabell et al. 1990; Saj et al. 1999). The vervet monkey collared in the nursery troop had the smallest home range yet spent the most time in the natural forest. With large canopy trees and dense bush, forest productivity is important to primates because it provides food and cover protection from predation (Horwich et al. 1993; Cheyne 2009). The habitat types of forest, thicket, and grassland used by vervet monkeys at Buffelsdraai exhibited the importance of the buffer zone. Although natural food was available, this did not deter the monkeys from foraging in built-up areas, raiding bins and the staff kitchen, as we observed from the two troops that shared the area at separate times and days (pers. obs.).
Two troops at Buffelsdraai (BD_Resident and BD_Mad) displayed considerable overlap in home ranges, likely driven by the availability of anthropogenic food resources present at the offices on the edge of the Buffelsdraai rehabilitation site (Supplementary information Fig. S2). However, at no identical time did these troops overlap (pers. obs.). Vervet monkeys are known to demonstrate absentee ownership of territories. Isbell et al. (2021) defined this as “territories which are nonexclusive but may still be perceived by each troop as sole-owned, as neighbours may only intrude when territory owners are absent from that area”, as was evident in our study. The rehabilitation of the landfill in Buffelsdraai appeared not directly beneficial for the vervet monkeys; however, it provided increased connectivity between habitats in the adjoining landscape, facilitating dispersal and the movement of individuals. Furthermore, increased habitat and food available in the existing forest habitats may increase carrying capacity and troop size. The relative lack of human activity allows these populations to mimic wild troop numbers (Isbell et al. 1990; Barrett et al. 2016). However, with highly modified landscapes, vervet monkeys will exploit and use the environment to fulfil their intuitive nature and behaviour.
In our study, the technical limitations arising from the GPS-UHF tracking collars yielded limited data as none of the collars lasted the proposed twelve months. In addition, the low success of working collars considers it necessary to increase the sample size to understand vervet monkeys’ spatial movements comprehensively. Therefore, we recommend future studies on increasing the number of remote-tracking collars to identify fine-resolution spatial patterns regarding seasonal variation and individual overlapping of this primate species.
Conclusions and recommendations
The home range and habitat use of vervet monkeys living in the urban forest interface alongside humans showed individual variation and differences in movement patterns. These semiarboreal primates use urban and forest habitats while successfully navigating the anthropogenic mosaic landscape. However, vervet monkeys in urban areas need management intervention to reduce negative human-wildlife interactions (Seoraj-Pillai and Pillay 2017; Thatcher et al. 2019a; pers. obs). Possible management methods are suggested to protect the species’ status and limit human-monkey interaction. Including and enforcing national legislation and provincial by-laws to protect the species from human threats is crucial for regulating keeping primates as pets, animal welfare and feeding. Implementing a complete ban on anthropogenic food provision for primates is paramount to avoid unnecessary pain and suffering for vervet monkeys caused by inevitable conflicts (pet attacks, poisoning or shooting). Management practices are also required to conceal waste and human food to avoid possible raiding events. The future of urban planning in tackling negative human-wildlife interactions is justified through environmental education of the above management practices. A requirement of the public involves active engagement and education on the importance and value of co-existing with wildlife, especially with an indigenous primate such as the vervet monkey.
Data Availability
Data for this study are available on request from the authors but belong to the University of KwaZulu-Natal.
References
Baldellou M, Adan A (1998) Diurnal and seasonal variations in vervet monkeys’ activity. Psychol Rep 83:675–685. https://doi.org/10.2466/pr0.1998.83.2.675
Baldellou M, Henzi SP (1992) Vigilance predator detection and the presence of supernumerary males in vervet monkey troops. Anim Behav 43:451–461. https://doi.org/10.1016/S0003-3472(05)80104-6
Bar-Massada A, Radeloff VC, Stewart SI (2014) Biotic and abiotic effects of human settlements in the wildland-urban interface. Bioscience 64:429–437. https://doi.org/10.1093/biosci/biu039
Barrett AS (2004) Foraging ecology of the vervet monkey (Chlorocebus aethiops) in mixed lowveld bushveld and sour lowveld bushveld of the Blydeberg Conservancy Northern Province South Africa. MSc Dissertation, University of South Africa
Barrett AS (2009) Spatial and temporal patterns in resource dispersion and the structure of range use and coexistence in a social omnivore (Chlorocebus aethiops). PhD Dissertation, University of South Africa
Barrett AS, Barrett L, Henzi P, Brown LR (2016) Resource selection on woody plant species by vervet monkeys (Chlorocebus pygerythrus) in mixed-broad leaf savanna. Afr J Wildl Res 46:14–21. https://hdl.handle.net/10520/EJC186959
Battin J (2004) When good animals love bad habitats: ecological traps and the conservation of animal populations. Conserv Biol 18:1482–1491. https://doi.org/10.1111/j.1523-1739.2004.00417.x
Berger-Tal O, Saltz D (2019) Invisible barriers: anthropogenic impacts on inter- and intra-specific interactions as drivers of landscape-independent fragmentation. Phil T Royal Soc B 374:1–9. https://doi.org/10.1098/rstb.2018.0049
Blanchard ML, Crompton RH (2011) Field study methods for primate locomotor ecology and biomechanics. In: D’Août K, Vereecke EE (eds) Primate Locomotion. Springer, New York, pp 183–199
Börger L, Dalziel BD, Fryxell JM (2008) Are there general mechanisms of animal home range behaviour? A review and prospects for future research. Ecol Lett 11:637–650. https://doi.org/10.1111/j.1461-0248.2008.01182.x
Brueckner JK (2000) Urban sprawl: diagnosis and remedies. Int Reg Sci Rev 23:160–171. https://doi.org/10.1177/016001700761012710
Burt WH (1943) Territoriality and home range concepts as applied to mammals. J Mammal 24:346–352. https://www.jstor.org/stable/1374834
Butynski TM, de Jong YA (2019) Chlorocebus pygerythrus the IUCN Red list of threatened Species: e-T136271A17957823. https://doi.org/10.2305/IUCN.UK.2019-3.RLTS.T136271A17957823.en
Byers CR, Steinhorst RK, Krausman PR (1984) Clarification of a technique for analysis of utilization-availability data. J Wildl Manage 48:1050–1053. https://doi.org/10.2307/3801467
Cagnacci F, Boitani L, Powell RA, Boyce MS (2010) Animal ecology meets GPS-based radiotelemetry: a perfect storm of opportunities and challenges. Phil T Royal Soc B 365:2157–2162. https://doi.org/10.1098/rstb.2010.0107
Cheney DL, Seyfarth RM (1990) How monkeys see the world: inside the mind of another species. University of Chicago Press, United States of America
De Moor PP, Steffens FE (1972) The movements of vervet monkeys (Cercopithecus aethiops) within their ranges as revealed by radio-tracking. J Anim Ecol 41:677–687. https://doi.org/10.2307/3202
Dore KM, Hansen MF, Klegarth AR et al (2020) Review of GPS collar deployments and performance on nonhuman primates. Primates 61:1–15. https://doi.org/10.1007/s10329-020-00793-7
Douwes E, Roy KE, Diederichs-Mander N et al (2015) The Buffelsdraai Landfill Site Community Reforestation Project: leading the way in community ecosystem-based adaptation to climate change. eThekwini Municipality, South Africa
Downs CT, Alexander J, Brown M, Chibesa M, Ehlers Smith YC, Gumede ST, Hart LA, Kalle R, Maphalala M, Maseko M, McPherson S, Ngcobo SP, Patterson L, Pillay K, Price C, Raji IA, Ramesh T, Schmidt W, Senoge ND, Shivambu TC, Shivambu N, Singh N, Singh P, Streicher J, Thabethe V, Thatcher H, Widdows C, Wilson AL, Zungu MM, Ehlers Smith D (2021) Modification of the third phase in the framework for vertebrate species persistence in connected urban environments based on a review of case studies from KwaZulu-Natal Province, South Africa. Ambio 50:1866–1878. https://doi.org/10.1007/s13280-021-01501-5
Ducheminsky N, Henzi SP, Barrett L (2014) Responses of vervet monkeys in large troops to terrestrial and aerial predator alarm calls. Behav Ecol 25:1474–1484. https://doi.org/10.1093/beheco/aru151
Findlay LJ, Hill RA (2021) Baboon and vervet monkey crop-foraging behaviors on a commercial south african farm: preliminary implications for damage mitigation. Hum Wildl Inter 14:505–518
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Global Ecol Biogeogr 16:265–280. https://doi.org/10.1111/j.1466-8238.2007.00287.x
Gehrt SD, Anchor C, White LA (2009) Home range and landscape use of coyotes in a metropolitan landscape: conflict or coexistence? J Mammal 90:1045–1057. https://doi.org/10.1644/08-MAMM-A-277.1
Getz WM, Fortmann-Roe S, Cross PC, Lyons AJ, Ryan SJ, Wilmers CC (2007) LoCoH: nonparameteric kernel methods for constructing home ranges and utilization distributions. PLoS ONE 2:1–11. https://doi.org/10.1371/journal.pone.0000207
Giuggioli L, Kenkre VM (2014) Consequences of animal interactions on their dynamics: emergence of home ranges and territoriality. Mov Ecol 2:1–22. https://doi.org/10.1186/s40462-014-0020-7
Grey JNC, Bell S, Hill RA (2017) Leopard diets and landowner perceptions of human wildlife conflict in the Soutpansberg Mountains South Africa. J Nat Conserv 37:56–65. https://doi.org/10.1016/j.jnc.2017.03.002
Harvey P, Clutton-Brock T (1981) Primate home-range size and metabolic needs. Behav Ecol Sociobiol 8:151–155. https://www.jstor.org/stable/4599372
Healy A, Nijman V (2014) Pets and pests: vervet monkey intake at a specialist south african rehabilitation centre. Anim Welf 23:353–360. https://doi.org/10.7120/09627286.23.3.353
Henzi SP (1982) Some aspects of visual signalling and social organization in the vervet monkey (Cercopithecus aethiops pygerythrus). University of KwaZulu-Natal. Dissertation
Hoffman TS, O’Riain MJ (2012) Landscape requirements of a primate population in a human-dominated environment. Front Zool 9:1–17. https://doi.org/10.1186/1742-9994-9-1
Isbell LA (2004) Is there no Place Like Home? Ecological bases of female dispersal and philopatry and their consequences for the formation of Kin Groups. In: Chapais B, Berman CM (eds) Kinship and behavior in Primates. Oxford University Press, New York, pp 71–108
Isbell LA, Bidner LR, Loftus JC, Kimuyu DM, Young TP (2021) Absentee owners and overlapping home ranges in a territorial species. Behav Ecol Sociobiol 75:1–14. https://doi.org/10.1007/s00265-020-02945-7
Isbell LA, Cheney DL, Seyfarth RM (1990) Costs and benefits of home range shifts among vervet monkeys (Cercopithecus aethiops) in Amboseli National Park Kenya. Behav Ecol Sociobiol 27:351–358. https://doi.org/10.1007/BF00164006
Jarrett JD, Bonnell T, Jorgensen MJ, Schmitt CA, Young C, Dostie M, Barrett L, Henzi SP (2020) Modeling variation in the growth of wild and captive juvenile vervet monkeys in relation to diet and resource availability. Am J Phys Anthropol 171:89–99. https://doi.org/10.1002/ajpa.23960
Kavanagh K, Dozier BL, Chavanne TJ, Fairbanks LA, Jorgensen MJ, Kaplan JR (2011) Fetal and maternal factors associated with infant mortality in vervet monkeys. J of Med Primatol 40:27–36. https://doi.org/10.1111/j.1600-0684.2010.00441.x
Kenward RE (2001a) Historical and practical perspectives. In: Millspaugh JJ, Marzluff JM (eds) Radio Tracking and Animal populations. Academic Press, London, pp 3–12
Kenward RE (2001b) A Manual for Wildlife Tagging. Academic Press, New York
Kretser HE, Sullivan PJ, Knuth BA (2008) Housing density as an indicator of spatial patterns of reported human-wildlife interactions in Northern New York. Landsc Urban Plan 84:282–292. https://doi.org/10.1016/j.landurbplan.2007.08.007
Laver PN, Kelly MJ (2008) A critical review of home range studies. J Wildl Manage 72:290–298. https://doi.org/10.2193/2005-589
Lindenmayer DB, Fischer J, Felton A, Crane M, Michael D, MacGregor C, Montague-Drake R, Manning A, Hobbs RJ (2008) Novel ecosystems resulting from landscape transformation create dilemmas for modern conservation practice. Conserv Lett 1:129–135. https://doi.org/10.1111/j.1755-263X.2008.00021.x
MacFarlane D, Harvey J, Hamer M (2011) Biodiversity Assessment of the Buffelsdraai Landfill Site Community Reforestation Programme, Report No E P 08 – 01. eThekwini Municipality, South Africa
Magle SB, Fidino M (2018) Long-term declines of a highly interactive urban species. Biodivers Conserv 27:3693–3706. https://doi.org/10.1007/s10531-018-1621-z
Magle SB, Hunt VM, Vernon M, Crooks KR (2012) Urban wildlife research: past present and future. Biol Conserv 155:23–32. https://doi.org/10.1016/j.biocon.2012.06.018
McCleery RA, Moorman CE, Peterson MN (2014) Urban wildlife conservation: theory and practice. Springer, New York
McKinney ML (2006) Urbanization as a major cause of biotic homogenization. Biol Conserv 127:247–260. https://doi.org/10.1016/j.biocon.2005.09.005
McKinney T (2015) A classification system for describing anthropogenic influence on nonhuman primate populations. Am J Primatol 77:715–726. https://doi.org/10.1002/AJP.22395
McLennan MR, Spagnoletti N, Hockings KJ (2017) The implications of primate behavioral flexibility for sustainable human-primate coexistence in anthropogenic habitats. Int J Primatol 38:105–121. https://doi.org/10.1007/s10764-017-9962-0
McLoughlin PD, Ferguson SH (2000) A hierarchical pattern of limiting factors helps explain variation in home range size. Ecoscience 7:123–130. https://doi.org/10.1080/11956860.2000.11682580
McPherson SC, Brown M, Downs CT (2016) Diet of the crowned eagle (Stephanoaetus coronatus) in an urban landscape: potential for human-wildlife conflict? Urban Ecosyst 19:383–396. https://doi.org/10.1007/s11252-015-0500-6
Mikula P, Šaffa G, Nelson E, Tryjanowski P (2018) Risk perception of vervet monkeys Chlorocebus pygerythrus to humans in urban and rural environments. Behav Processes 147:21–27. https://doi.org/10.1016/j.beproc.2017.12.011
Mucina L, Rutherford MC (2006) The vegetation of South Africa Lesotho and Swaziland Strelitizia 19. South African National Biodiversity Institute, Pretoria
Murphy GE, Romanuk TN (2012) A meta-analysis of community response predictability to anthropogenic disturbances. Am Nat 180:316–327. https://www.jstor.org/stable/10.1086/666986
Naude VN, Smyth LK, Weideman EA, Krochuk BA, Amar A (2019) Using web-sourced photography to explore the diet of a declining african raptor, the Martial Eagle (Polemaetus bellicosus). Condor 121:1–9. https://doi.org/10.1093/condor/duy015
Pasternak G, Brown LR, Kienzle S, Fuller A, Barrett L, Henzi SP (2013) Population ecology of vervet monkeys in a high latitude, semi-arid riparian woodland. Koedoe 55(1):1–9. https://doi.org/10.4102/koedoe.v55i1.1078
Patterson L, Kalle R, Downs CT (2017) A citizen science survey: perceptions and attitudes of urban residents towards vervet monkeys. Urban Ecosyst 20:617–628. https://doi.org/10.1007/s11252-016-0619-0
Patterson L, Kalle R, Downs CT (2018) Factors affecting the presence of vervet monkey troops in a suburban matrix in KwaZulu-Natal South Africa Landscape Urban Plan. 169:220–228. https://doi.org/10.1016/j.landurbplan.2017.09.016
Patterson L, Kalle R, Downs CT (2019) Living in the suburbs: space use by vervet monkeys (Chlorocebus pygerythrus) in an eco-estate South Africa. Afr J Ecol 57:539–551. https://doi.org/10.1111/aje.12629
Riley EP (2008) Ranging patterns and habitat use of Sulawesi Tonkean macaques (Macaca tonkeana) in a human-modified habitat. Am J Primatol 70:670–679. https://doi.org/10.1002/ajp.20543
Roy KE (2016) Seeing the wood for the trees: an evaluation of the Buffelsdraai Landfill Community Reforestation Project. Dissertation, University of KwaZulu-Natal
Saj TL (1998) The ecology and behavior of vervet monkeys in a human-modified environment. Dissertaion, University of Calgary
Saj T, Sicotte P, Paterson JD (1999) Influence of human food consumption on the time budget of vervets. Int J Primatol 20:977–994. https://doi.org/10.1023/A:1020886820759
Seoraj-Pillai N, Pillay N (2017) A meta-analysis of human-wildlife conflict: south african and global perspectives. Sustainability 9:1–21. https://doi.org/10.3390/su9010034
Seto KC, Parnell S, Elmqvist T (2013) A global outlook on urbanization. In: Elmqvist T, Frakias M, Goodness J et al (eds) Urbanization, Biodiversity and Ecosystem Services: Challenges and Opportunities. Springer, Dordrecht, pp 1–12
Seyfarth RM (1980) The distribution of grooming and related behaviors among adult female vervet monkeys. Anim Behav 28:798–813. https://doi.org/10.1016/S0003-3472(80)80140-0
Seyfarth R, Cheney D (1990) The assessment by vervet monkeys of their own and another species’ alarm calls. Anim Behav 40:754–764. https://doi.org/10.1016/S0003-3472(05)80704-3
Signer J, Balkenhol N (2015) Reproducible home ranges (rhr): A new user-friendly R package for analyses of wildlife telemetry data. Wildl Soc Bull 39:358–363. https://doi.org/10.1002/wsb.539
Siljander M, Kuronen T, Johansson T, Munyao mn, Pellikka PKE (2020) Primates on the farm-spatial patterns of human-wildlife conflict in forest-agricultural landscape mosaic in Taita Hills Kenya. Appl Geogr 117:1–13. https://doi.org/10.1016/j.apgeog.2020.102185
Sillero-Zubiri C, Switzer D (2001) Crop raiding primates: searching for alternative humane ways to resolve conflict with farmers in Africa. Wildlife Conservation Research Unit, Oxford University, Oxford
Skidmore AK, Franklin J, Dawson TP, Pilesjö P (2011) Geospatial tools address emerging issues in spatial ecology: a review and commentary on the Special Issue. Int J Geogr Inf Sci 25:337–365. https://doi.org/10.1080/13658816.2011.554296
Smithers RH (2012) Smithers Mammals of Southern Africa. Penguin Random House, South Africa
Snowdon CT (2020) Vervet monkey alarm calls: setting the historical context. Anim Behav Cogn 7:87–94. https://doi.org/10.26451/abc.07.02.02.2020
Soulsbury CD, White PC (2016) Human-wildlife interactions in urban areas: a review of conflicts benefits and opportunities. Wildl Res 42:541–553. https://doi.org/10.1071/WR14229
StatsSA Census 2011: Statistics South Africa South African Population Census 2011 https://wazimap.co.za/profiles/ward-59500066-ethekwini-ward-66-59500066 Accessed 30 January 2020
Streicher JP, Ramesh T, Downs CT (2020) Home range and core area utilisation of three co-existing mongoose species: large grey water and white-tailed in the fragmented landscape of the KwaZulu-Natal Midlands South Africa. Mamm Biol 100:273–283. https://doi.org/10.1007/s42991-020-00028-8
Struhsaker TT (1967) Social structure among vervet monkeys (Cercopithecus aethiops). Behaviour 29:83–121
Swedell L (2012) Primate sociality and Social Systems. Nat Educ Knowl 3:84
Teichroeb JA (2015) Vervet monkeys use paths consistent with context-specific spatial movement heuristics. Ecol Evol 5:4706–4716. https://doi.org/10.1002/ece3.1755
Teichroeb JA, White MMJ, Chapman CA (2015) Vervet (Chlorocebus pygerythrus) intragroup spatial positioning: dominants trade-off predation risk for increased food acquisition. Int J Primatol 36:154–176. https://doi.org/10.1007/s10764-015-9818-4
Thatcher HR, Downs CT, Koyama NF (2019a) Positive and negative interactions with humans concurrently affect vervet monkey (Chlorocebus pygerythrus) ranging behavior. Int J Primatol 40:496–510. https://doi.org/10.1007/s10764-019-00099-6
Thatcher HR, Downs CT, Koyama NF (2019b) Anthropogenic influences on the time budgets of urban vervet monkeys. Landsc Urban Plan 181:38–44. https://doi.org/10.1016/j.landurbplan.2018.09.014
Thatcher HR, Downs CT, Koyama NF (2020) Understanding foraging flexibility in urban vervet monkeys Chlorocebus pygerythrus for the benefit of human-wildlife coexistence Urban Ecosyst 23:1349–1357. https://doi.org/10.1007/s11252-020-01014-1
Turner TR, Cramer JD, Nisbett A, Patrick Gray J (2016a) A comparison of adult body size between captive and wild vervet monkeys (Chlorocebus aethiops sabaeus) on the island of St Kitts. Primates: J Primatol 57:211–220. https://doi.org/10.1007/s10329-015-0509-8
Turner T, Hill R, Coetzer WG, Patterson L (2016b) A conservation assessment of Chlorocebus pygerythrus. In: Child MF, Roxburgh L, Do Linh San E, Raimondo D, Davies-Mostert HT (eds) The Red List of Mammals of South Africa Swaziland and Lesotho. South African National Biodiversity Institute and Endangered Wildlife Trust, South Africa
Turner TR, Schmitt CA, Cramer JD, Lorenz J, Grobler JP, Jolly CJ, Freimer NB (2018) Morphological variation in the genus Chlorocebus: Ecogeographic and anthropogenically mediated variation in body mass postcranial morphology and growth. Am J of Phys Anthropol 166:682–707. https://doi.org/10.1002/ajpa.23459
United Nations Department of Economic and Social Affairs Population Division (2019) World urbanization prospects: the 2018 Revision (ST/ESA/SER A/420). United Nations, New York
Valiente-Banuet A, Aizen MA, Alcántara JM et al (2015) Beyond species loss: the extinction of ecological interactions in a changing world. Funct Ecol 29:299–307. https://doi.org/10.1111/1365-2435.12356
van Beest FM, Rivrud IM, Loe LE, Milner JM, Mysterud A (2011) What determines variation in home range size across spatiotemporal scales in a large browsing herbivore? J Anim Ecol 80:771–785. https://doi.org/10.1111/j.1365-2656.2011.01829.x
Van Moorter B, Rolandsen CM, Basille M, Gaillard JM (2016) Movement is the glue connecting home ranges and habitat selection. J Anim Ecol 85:21–31. https://doi.org/10.1111/1365-2656.12394
Villarreal-Barajas T, Martorell C (2009) Species‐specific disturbance tolerance competition and positive interactions along an anthropogenic disturbance gradient. J Veg Sci 20:1027–1040. https://doi.org/10.1111/j.1654-1103.2009.01101.x
Walter WD, Fischer JW, Baruch-Mordo S, VerCauteren KC (2011) In: Krejcar O (ed) What is the proper method to delineate home range of an animal using today’s advanced GPS telemetry systems: the initial step. Modern Telemetry, InTech, Croatia
Wiegand T, Revilla E, Moloney KA (2005) Effects of habitat loss and fragmentation on population dynamics. Conserv Biol 19:108–121. https://doi.org/10.1111/j.1523-1739.2005.00208.x
Willems EP (2007) From space to species: integrating remotely sensed information on primary productivity into investigations and systems models of vervet monkey (Cercopithecus aethiops) socio ecology. Dissertation, Durham University
Willems EP, Barton RA, Hill RA (2009) Remotely sensed productivity regional home range selection and local range use by an omnivorous primate. Behav Ecol 20:985–992. https://doi.org/10.1093/beheco/arp087
Wimberger K, Downs CT (2010) Annual intake trends of a large urban animal rehabilitation centre in South Africa: a case study. Anim Welf 19:501–513
Wimberger K, Downs CT, Boyes RS (2010) A survey of wildlife rehabilitation in South Africa: is there a need for improved management? Anim Welf 19:481–499
Worton BJ (1989) Kernel methods for estimating the utilization distribution in home-range studies. Ecology 70:164–168. https://doi.org/10.2307/1938423
Young C, McFarland R, Ganswindt A et al (2019) Male residency and dispersal triggers in a seasonal breeder with influential females. Anim Behav 154:29–37. https://doi.org/10.1016/j.anbehav.2019.06.010
Zipperer WC, Wu J, Pouyat RV, Pickett STA (2000) The application of ecological principles to urban and urbanizing landscapes. Ecol Appl 10:685–688. https://doi.org/10.2307/2641038
Zungu MM, Maseko MS, Kalle R, Ramesh T, Downs CT (2020) Factors affecting the occupancy of forest mammals in an urban-forest mosaic in eThekwini Municipality Durban South Africa. Urban For Urban Gree 48:1265. https://doi.org/10.1016/j.ufug.2019.126562
Acknowledgements
Special thanks to F. Moolla and R. Winn at Buffelsdraai Landfill Conservancy; and WESSA Treasure Beach Educational Centre for providing permission and access to the study sites. Dr S. Singh (veterinarian) is thanked for enabling capturing and collaring of vervet monkeys. We thank V. Hugo for technical assistance during the tracking of the vervet monkeys and K. Josiah for creating graphic representations of maps. We thank M. Machawe for his comments on the draft manuscript. Ethical clearance was obtained from the University of KwaZulu-Natal to catch and fit transmitters on the vervet monkeys. We thank the reviewers for their constructive comments that improved our manuscript.
Funding
We are grateful for financial support from the National Research Foundation (ZA, Grant 98404) and Ethekwini Durban Research Action Partnership (DRAP, ZA), and the University of KwaZulu-Natal (ZA). We thank the Ford Wildlife Foundation (ZA) for vehicle support.
Open access funding provided by University of KwaZulu-Natal.
Author information
Authors and Affiliations
Contributions
CTD, KP and JPS conceptualised the research. CTD sought funding. KP and JPS collected and analysed the data. KP drafted the manuscript. CTD and JPS provided comments and revisions to the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest nor competing interests.
Ethical approval
Approval was granted by the University of KwaZulu-Natal, Animal Ethics committee (no. 020/15/animal).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pillay, K.R., Streicher, J.P. & Downs, C.T. Home range and habitat use of vervet monkeys in the urban forest mosaic landscape of Durban, eThekwini Municipality, KwaZulu-Natal, South Africa. Urban Ecosyst 26, 1769–1782 (2023). https://doi.org/10.1007/s11252-023-01396-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-023-01396-y