Abstract
Background
Acute kidney injury (AKI) is common in patients with sepsis and septic shock. Urine output and serum creatinine (SCr) levels are the criteria for diagnosing AKI. However, the application of these levels in the diagnosis of AKI has limitations.
Objective
To detect the expression of various adhesion factors in different stages of AKI as defined by Kidney Disease: Improving Global Outcomes (KDIGO) and to analyse their relationship with the prognosis of patients with sepsis-induced AKI (S-AKI).
Methods
Adult patients with sepsis who were admitted to the hospital between June 2019 and May 2020 were included. Of 90 adult patients with sepsis, 58 had S-AKI. Sixty-seven subjects without sepsis were used as controls. Enzyme-linked immunosorbent assay kits were used to measure E-selectin (CD62E), L-selectin (CD62L), P-selectin, intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1), and their relationship with the prognosis of patients with S-AKI patients was analysed. Receiver operating characteristic curves were used to analyse the predictive value of different adhesion factors on renal resistance index and renal function recovery. Multivariate logistic regression analysis was used to identify factors associated with renal recovery.
Results
The expression of CD62L was significantly higher in S-AKI patients than in non-AKI patients with sepsis. Compared with the non-AKI group, Acute Physiology and Chronic Health Evaluation II and Sequential Organ Failure Assessment scores were significantly higher in the AKI group than in the non-AKI group (P < 0.05). Mean blood pressure, SCr levels and procalcitonin levels were higher in the AKI group than in the non-AKI group (P < 0.05 for all). The CD62L levels decreased with increasing S-AKI stage. The CD62E levels were highest in S-AKI stage 2, and the VCAM-1 levels were highest in S-AKI stage 3. All patients with S-AKI were followed up with for 28 days. The results found that VCAM-1 was the best predictor of renal recovery in patients with S-AKI.
Conclusion
CD62L is an indicator of S-AKI stage1, and CD62E is an indicator of S-AKI stage 2. In addition, VCAM-I demonstrated satisfactory performance in predicting early recovery of renal function in patients with S-AKI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute kidney injury (AKI) associated with sepsis (S-AKI) is frequent in patients with sepsis and septic shock. According to earlier research, 57% of critically ill patients in intensive care units (ICUs) have acute kidney injury. Approximately 50% of these patients develop AKI secondary to sepsis, and AKI occurs in 50–70% of patients with sepsis [1,2,3,4]. Most current recommendations, including those from Kidney Disease: Improving Global Outcomes (KDIGO), regard serum creatinine (SCr) levels and urine output as diagnostic indicators of AKI. Even though SCr is easily measured, there are limitations to its use in diagnosing AKI. For example, renal hypoperfusion may result in a higher SCr without significant renal parenchymal damage. Moreover, reliance on increased SCr can delay early diagnosis and treatment of AKI, as approximately 50% of patients with kidney injury do not have an increased SCr [5,6,7,8]. Therefore, it is necessary to identify other sensitive and specific biomarkers for the early diagnosis of AKI.
Previous studies have recommended using several early biomarkers for the diagnosis of AKI based on their varied activities and connections with damage to different regions of the nephron unit. Ischaemic damage raises the levels of kidney injury molecule-1 (KIM-1), neutrophil gelatinase-associated lipocalin (NGAL), monocyte chemoattractant protein-1 and cysteine-rich angiogenic inducer-61. In contrast, hypoxic damage increases the level of liver fatty acid-binding protein. Glomerular injury and a lower estimated glomerular filtration rate led to increased levels of SCr, cystatin C, and NGAL. Proximal tubule injury leads to higher levels of KIM-1, N-acetyl-β-D-glucosaminidase, insulin-like growth factor-binding protein-7 and tissue inhibitor of metalloproteinases-2. In contrast, distal convoluted tubule damage leads to increased levels of NGAL [9].
Sepsis affects the glomeruli and renal tubules by producing renal haemodynamic abnormalities, activating immune cells, releasing inflammatory chemicals and downregulating endogenous hormones [10]. Many studies have explored renal haemodynamics and biomarkers of tubular injury, but research on the immune system’s interaction with sepsis and AKI is scarce and controversial. According to reports, AKI has anti-inflammatory and inhibitory effects on neutrophil granulocytes [11]. There is additional evidence that renal tubular cells and other significant epithelial cells are associated with adhesion factor release in tissue injury. Renal tubular epithelial cells might express vascular cell adhesion molecules and cell adhesion molecules, resulting in the recruitment and activation of T lymphocytes, subsequently activating the cluster of differentiation-40 and harming epithelial cells [12].
Given the above reasons, this study aims to determine the expression of various adhesion factors in the different phases of AKI defined by KDIGO and examine their relationship with the prognosis of patients diagnosed with S-AKI.
Patients and methods
Participants and groups
In this prospective study, adult patients with sepsis who were admitted between June 2019 and May 2020 and had a 28-day follow-up appointment were included. Sepsis was defined as per the Sepsis-3 criteria [13]. The inclusion criteria were as follows: (1) sequential organ failure assessment (SOFA) score ≥ 2 points in diagnosed or suspected infection; (2) blood lactic acid level above 2.0 mmol/L. The exclusion criteria were as follows: (1) potential chronic kidney disease or patients on maintenance haemodialysis; (2) age less than 18 years; (3) pregnancy; (4) renal vascular abnormalities or previous renal surgery, such as kidney transplant; (5) ICU stay less than three days; or (6) patients unwilling to participate.
The control group included patients without sepsis. The inclusion criteria were: (1) willingness to participate in the entire study. (2) age > 18 years; and (3) no abnormality on physical examination. The exclusion criteria were as follows: (1) history of a genetic type of nephropathy. (2) Previous history of AKI; and (3) pregnancy.
The Ethics Committee of the Third Hospital of Hebei Medical University approved this study (approval number: KE-2019-20-1, KE-2019-19-1), and each participant signed informed consent before participation.
Acute kidney injury was diagnosed according to the 2012 KDIGO criteria [14]: an increase in SCr of more than 26.5 μmoL/L within 48 h, a 50% or greater rise of SCr from the baseline or a urine output of less than 0.5 mL/kg/h for more than six h. Patients with S-AKI were also classified by stage (S-AKI-1, S-AKI-2 and S-AKI-3) and were followed up with for 28 days. According to international consensus criteria [15] and a previous study [16], renal recovery was defined as a restoration of SCr to within 150% of baseline level without requiring renal replacement therapy (RRT). Non-recovery was defined as the need for RRT or no restoration of the SCr level at 28 days after admission.
Intervention measures
Following the Surviving Sepsis Campaign guidelines for achieving resuscitation goals [17], septic patients were given conventional treatment, which included fluid resuscitation and vasoactive drugs to maintain a mean arterial pressure (MAP) of at least 60 mmHg, a superior vena caval blood oxygen saturation of at least 70% and the lowest possible central venous pressure using restrictive fluid management once shock was corrected.
Data collection
Data collected from all subjects at admission were age, gender, underlying disease(s), site(s) of infection, Acute Physiology and Chronic Health Evaluation II (APACHE II) and SOFA scores within 24 h of entering the ICU, systolic blood pressure, diastolic blood pressure and MAP. The following parameters were recorded: laboratory results, including white blood cell (WBC) count, platelet count, lactic acid, procalcitonin (PCT), SCr, blood urea nitrogen, duration of ICU stay and 28-day mortality.
Testing of adhesion factors
Blood and urine samples were collected when patients entered the ICU and after 24 h and 72 h. Samples were centrifuged for 15 min, and the soluble and supernatant fractions were collected and stored at – 80 °C before testing. Blood samples were sent to the laboratory for routine blood and biochemical examination. Enzyme-linked immunosorbent assay (ELISA) kits (Arigo Biolaboratories, China) were used to measure the serum levels of E-selectin (CD62E), L-selectin (CD62L), P-selectin (CD62P), intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1).
Statistical analysis
All data were analysed using the IBM SPSS Statistics for Windows, Version 25.0 (Armonk, NY). Continuous variables were expressed as means ± standard deviation (\(\overline{X }\pm S\)) and were compared using either a t-test or a one-way analysis of variance, as appropriate. Categorical variables were expressed as numbers and percentages, and comparisons between groups were performed using the chi-square (χ2) test or Fisher’s exact test. Receiver operating characteristic (ROC) curves were used to analyse the predictive value of different adhesion factors on the renal resistive index and recovery. Multiple logistic regression analysis was used to identify factors associated with renal recovery. A P-value of less than 0.05 was considered statistically significant.
Results
Baseline characteristics of sepsis patients with and without AKI
In total, 90 septic patients and 67 patients without sepsis were enrolled (Table 1). Among the septic patients, 32 had pulmonary infections, 16 had abdominal cavity infections, 6 had blood infections and 28 had soft tissue infections. Seventy-eight of the septic patients had AKI (59%). The APACHE II and SOFA scores were significantly higher in the AKI group as compared with the non-AKI group (P < 0.001 for both). The mean blood pressure (MBP), SCr level and PCT levels were also higher in the AKI group as compared with the non-AKI group (P < 0.001 for all). There were no significant differences in WBC counts between the two groups, but the platelet count was significantly lower in the AKI group (P < 0.001). In addition, patients in the AKI group had a significantly longer length of hospital stay and higher 28-day mortality (P < 0.05 for both).
Biomarkers and prognosis of patients with different stages of AKI
According to the KDIGO criteria [18], 27 patients had AKI-1, 13 had AKI-2 and 18 had AKI-3 (Table 2). A comparison of the three AKI groups indicated significant statistical differences in age (P = 0.013) but not gender (P = 0.432). However, there were other significant differences among these three groups. In particular, the AKI-2 group had a higher APACHE II score and a lower MBP than the AKI-1 and AKI-3 groups. These three groups also had significant differences in PCT levels, with the highest level in the AKI-2 group. A comparison of patients with sepsis with and without AKI indicated that the AKI patients had greater expression of CD62L, CD62E and VCAM-1. Notably, the level of CD62L decreased as the AKI stage increased. The level of CD62E was highest in the AKI-2 group, and the level of VCAM-1 was highest in the AKI-3 group, followed by the AKI-2 and AKI-1 groups but without a statistical difference (P > 0.05).
Univariate analysis of factors associated with renal recovery in patients with S-AKI
All 52 patients with S-AKI were followed up with for 28 days. Fifteen of these patients (28.8%) had recovery of renal function (see Table 3). Univariate analysis identified lower APACHE II and SOFA scores and WBC counts in the renal recovery group, with P < 0.05. The levels of CD62P, CD62L, CD62E, ICAM-1 and VCAM-1 were all lower in the renal recovery group, but the difference was not statistically significant. The renal recovery group’s 28-day mortality rate was also lower (P < 0.05).
Receiver operating characteristic analysis of patients with S-AKI
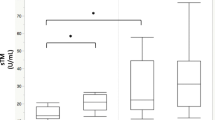
A ROC analysis was performed for each of the four blood markers, using the optimal cut-off values based on Youden’s J statistic, to assess their performance in predicting renal recovery in patients with S-AKI (Table 4). The results indicated that VCAM-1 provided the best diagnostic performance (area under the curve [AUC] = 0.61, sensitivity = 0.867 and specificity = 0.460) and CD62E provided the worst diagnostic performance (AUC = 0.52, sensitivity = 0.667 and specificity = 0.514) (Fig. 1).
Multiple logistic analysis of factors associated with death in patients with S-AKI
Multiple logistic regression analysis showed that the levels of CD62L, CD62E, VCAM-1 and ICAM-1 were not significantly associated with 28-day mortality (P > 0.05 for all) (Table 5).
Discussion
This study explored the expression of various adhesion factors in different stages of AKI as defined by KDIGO and evaluated their diagnostic value for predicting the prognosis of patients with S-AKI. The main findings can be summarised as follows: (1) Compared with the non-AKI group, MBP, SCr levels, PCT levels, APACHE II scores and SOFA scores were significantly higher in the AKI group than in the non-AKI group. (2) The expression of CD62L was significantly higher in patients with S-AKI than in patients with sepsis without AKI. (3) Among the multiple adhesion factors, VCAM-1 provided the best predictive value for the 28-day renal recovery in patients with S-AKI. These results suggest that some adhesion factors change significantly during the recovery of renal function and, therefore, might be useful tools for risk stratification and clinical decision-making.
White cells [19] and inflammatory markers [20] were raised in all stages of AKI, including onset, progress and recovery. Due to the functions of CD62E, CD62L, CD62P and integrins, neutrophil granulocytes adhere to the inflammatory site of the kidney [21, 22]. Inflammation can eradicate infections, but it also increases tissue and organ damage. Inflammatory mediators simultaneously stimulate epithelial cells and upregulate the expression of adhesion factors such as CD62E, which accelerate the recruitment of white blood cells to the kidney. The continuance of this process, the increase in vascular permeability and the activation of adhesive factors further promote the inflammatory reaction [23,24,25]. Therefore, inflammation and leukocyte fundraising are at the core of sepsis development, and these processes play a role in a positive feedback cycle that exacerbates severity. In this study, the three adhesion-related factors (CD62L, CD62E and VCAM-1) of patients with S-AKI were higher than those with sepsis. In addition, the ROC analysis showed that CD62L, CD62E and VCAM-1 levels were related to renal function recovery of patients with S-AKI.
The study found that the expression of CD62L, VCAM-1 and CD62E was greater in patients with S-AKI when compared with patients with sepsis alone. Moreover, the CD62L level appeared to decline as AKI progressed, in that its expression was highest in the S-AKI-1 group, followed by the S-AKI-2 and S-AKI-3 groups. The CD62E level appeared to at first increase and then decrease as AKI progressed, in that its expression was highest in the S-AKI-2 group, followed by the S-AKI-3 and S-AKI-1 groups. The level of VCAM-1 was highest in the S-AKI-3 group, followed by the S-AKI-2 and S-AKI-1 groups. The level of ICAM-1 was highest in the S-AKI-2 group, followed by the S-AKI-1 and S-AKI-2 groups. These results suggest that CD62L are indicators of S-AKI-1, CD62E is an indicator of AKI-2 and ICAM-1 is an indicator of AKI-2. Previous AKI research has shown that the peak of neutrophil recruitment in the kidneys occurs approximately 24 h after injury [26]. This finding is consistent with the results in this study in that the levels of the adhesion factor CD62L were higher in patients with S-AKI-1 than in those with S-AKI-2 and S-AKI-3. The results of other studies have shown that cytokine levels declined after neutrophil migration, and cytokine release promoted leukocyte recruitment and increased vascular permeability [27]. Thus, the levels of several adhesion factors increase soon after AKI onset and then decline as AKI progresses.
Endothelial cells are crucial in the control of coagulation. Under normal circumstances, the endothelium controls blood volume, electrolyte balance and blood coagulation, preventing microcirculatory diseases such as thrombotic microangiopathy and disseminated intravascular coagulation [28]. Due to their ability to be detected even before conventionally recognised signs, endothelial biomarkers may be used as indicators of endothelial damage [29]. This study demonstrated that among the various adhesion factors, VCAM-1 had the best diagnostic value for predicting the recovery of patients with S-AKI. Additionally, VCAM-1 has been investigated as a diagnostic and prognostic agent for illness since it is only detectable after cytokine activation of endothelial cells [29, 30]. In the study by Mota et al. [31], serum VCAM-1 in the AKI group was considerably higher than in the control and non-AKI groups, suggesting that endothelial cells in the AKI group had been more activated and injured. When used to predict AKI within 24 h of a two-headed snake bite, VCAM-1 performed very well, which is consistent with the findings of this study; VCAM-1 also demonstrated remarkable accuracy when identifying AKI within 24 h of moderate envenomation after a two-headed snake bite. Thus, VCAM-1 might provide valuable information for risk stratification in practice.
The follow-up of S-AKI patients found that the CD62P, CD62L, CD62E, ICAM-1 and VCAM-1 levels in patients who did not recover were low, but the difference was statistically significant only for VCAM-1. The 28-day mortality rate in the renal recovery group was significantly lower than in the non-resilient group. Therefore, these results indicate that low levels of adhesion factors indicate early recovery of renal function in patients with S-AKI and that the recovery of renal function increases survival [32]. These results are like the results of other studies, and patients with early kidney recovery have the same long-term survival as patients with sepsis but no AKI. Therefore, early diagnosis and treatment of AKI and early kidney recovery seem essential to improving the prognosis of patients with S-AKI.
The damage to vascular endothelial cells and the activation, adhesion and infiltration of leukocytes play crucial roles in the progression of sepsis [33]. Both ICAM-1 and VCAM-1 are adhesion molecules of the immunoglobulin family, expressed by endothelial cells. Their primary function is to induce leukocytes to adhere to endothelial cells firmly, thus promoting an increase in circulating levels of ICAM-1 and VCAM-1 [34]. This study’s ROC analysis showed that VCAM-1 demonstrated high sensitivity and specificity in predicting an early renal recovery in patients with S-AKI and that CD62E had high sensitivity but low specificity in predicting an early renal recovery in these patients. Several recent studies have attempted to identify factors associated with early renal recovery in patients with AKI, but the results have been inconclusive. For example, Fiorentino et al. showed that the APACHE II score and the baseline SCr were independent predictors of recovery of renal function in patients with S-AKI [32]. The Biological Markers of Recovery for the Kidney study, BioMaRK, showed that a baseline clinical model that included age, MBP, mechanical ventilation and bilirubin could predict renal recovery and mortality similar to plasma biomarkers. However, the clinical model also considered plasma IL-8 to have a higher predictive value for renal recovery and mortality with AUCs of 0.76 and 0.78, respectively [35]. The prediction model of this study examined several additional biomarkers: CD62L, CD62E, CD62P and VCAM-1, and found that CD62L and VCAM-1 demonstrated a high value for predicting a renal recovery in patients with S-AKI. This study thus suggests a new approach for predicting the prognosis of patients with S-AKI, although studies with larger samples are needed for confirmation.
The multiple logistic regression analysis showed that adhesion factors and PCT showed no significant correlation with 28-day mortality in patients with S-AKI. In contrast, other research [34] has reported that the ICAM-1 and VCAM-1 levels correlated significantly with organ dysfunction and mortality in patients with sepsis. This difference may be due to the small sample size in this study, a low number of deaths or differences in the study populations. Extensive prospective studies are needed to confirm the correlation between serum levels of adhesion factors and deaths in patients with S-AKI.
This study has several limitations. This is a single-centre, prospective study with a relatively small sample size. The results may be vulnerable to selection bias, raising concerns about generalisability. In addition, ELISA was used to determine the level of adhesive factors in plasma. However, the level of these factors in the kidney and other tissues could not be determined. Further, some of the baseline data in this study were not comparable. Therefore, further large and multicentre studies are needed to reveal the underlying mechanism of these adhesion factors in the pathogenesis and recovery of patients with S-AKI.
Conclusion
CD62L is an indicator of S-AKI-1 and CD62E is an indicator of S-AKI-2. In addition, VCAM-I showed satisfactory performance in predicting early recovery of renal function in patients with S-AKI.
Abbreviations
- S-AKI:
-
Sepsis-induced acute kidney injury
- ICU:
-
Intensive care unit
- CD62E:
-
E-selectin
- CD62L:
-
L-selectin
- CD62P:
-
P-selectin
- ICAM-1:
-
Intercellular adhesion molecule 1
- VCAM-1:
-
Vascular cell adhesion molecule 1
- KDIGO:
-
Kidney disease: improving global outcomes
- SCr:
-
Serum creatinine
- VCAMs:
-
Vascular cell adhesion molecules
- ICAMs:
-
Intercellular adhesion molecules
- RRT:
-
Renal replacement therapy
- MAP:
-
Mean arterial pressure
- APACHE II:
-
Acute physiology and chronic health evaluation II
- WBC:
-
White blood cell
- PCT:
-
Procalcitonin
- MBP:
-
Mean blood pressure
- ELISA:
-
Enzyme-linked immunosorbent assay
- SOFA:
-
Sepsis-related organ failure assessment
- ROC:
-
Receiver operating characteristic
References
Schrier RW, Wang W (2004) Acute renal failure and sepsis. N Engl J Med 351(2):159–169
Kim WY, Huh JW, Lim CM et al (2012) Analysis of progression in risk, injury, failure, loss, and end-stage renal disease classification on outcome in patients with severe sepsis and septic shock. J Crit Care 27(1):104.e1–7
Marino R, Struck J, Hartmann O et al (2015) Diagnostic and short-term prognostic utility of plasma pro-enkephalin (pro-ENK) for acute kidney injury in patients admitted with sepsis in the emergency department. J Nephrol 28(6):717–724
Medeiros P, Nga HS, Menezes P et al (2015) Acute kidney injury in septic patients admitted to emergency clinical room: risk factors and outcome. Clin Exp Nephrol 19(5):859–866
Ichai C, Vinsonneau C, Souweine B et al (2016) Acute kidney injury in the perioperative period and in intensive care units (excluding renal replacement therapies). Ann Intens Care 6(1):48
Mishra J, Dent C, Tarabishi R et al (2005) Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 365(9466):1231–1238
Bellomo R, Kellum JA, Ronco C (2004) Defining acute renal failure: physiological principles. Intens Care Med 30(1):33–37
Alge JL, Arthur JM (2015) Biomarkers of AKI: a review of mechanistic relevance and potential therapeutic implications. Clin J Am Soc Nephrol 10(1):147–155
Wang KF, Xie SL, Xiao K et al (2018) Biomarkers of Sepsis-Induced Acute Kidney Injury. Biomed Res Int 2018:6937947
Dellepiane S, Marengo M, Cantaluppi V (2016) Detrimental cross-talk between sepsis and acute kidney injury: new pathogenic mechanisms, early biomarkers and targeted therapies. Crit Care 20:61
Singbartl K, Formeck CL, Kellum JA (2019) Kidney-Immune System Crosstalk in AKI. Semin Nephrol 39(1):96–106
Cantaluppi V, Quercia AD, Dellepiane S et al (2014) Interaction between systemic inflammation and renal tubular epithelial cells. Nephrol Dial Transplant 29(11):2004–2011
Singer M, Deutschman CS, Seymour CW et al (2016) The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 315(8):801–810
Khwaja A (2012) KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 120(4):c179-184
Chawla LS, Bellomo R, Bihorac A et al (2017) Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol 13(4):241–257
Kellum JA (2014) How can we define recovery after acute kidney injury? Considerations from epidemiology and clinical trial design. Nephron Clin Pract 127(1–4):81–88
Rhodes A, Evans LE, Alhazzani W et al (2017) Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med 43(3):304–377
Kidney Disease; Improving Global Outcomes (KDIGO). Kidney Disease: improving global outcomes (KDIGO) acute kidney injury work group. Kidney Int. 2012;2:1–138
Bonavia A, Singbartl K (2018) A review of the role of immune cells in acute kidney injury. Pediatr Nephrol 33(10):1629–1639
Angus DC, van der Poll T (2013) Severe sepsis and septic shock. N Engl J Med 369(9):840–851
Singbartl K, Green SA, Ley K (2000) Blocking P-selectin protects from ischemia/reperfusion-induced acute renal failure. FASEB J 14(1):48–54
Herter JM, Rossaint J, Spieker T et al (2014) Adhesion molecules involved in neutrophil recruitment during sepsis-induced acute kidney injury. J Innate Immun 6(5):597–606
Gomez H, Ince C, De Backer D et al (2014) A unified theory of sepsis-induced acute kidney injury: inflammation, microcirculatory dysfunction, bioenergetics, and the tubular cell adaptation to injury. Shock 41(1):3–11
Brown KA, Brain SD, Pearson JD et al (2006) Neutrophils in development of multiple organ failure in sepsis. Lancet 368(9530):157–169
Zarbock A, Ley K (2008) Mechanisms and consequences of neutrophil interaction with the endothelium. Am J Pathol 172(1):1–7
Singbartl K, Ley K (2000) Protection from ischemia-reperfusion induced severe acute renal failure by blocking E-selectin. Crit Care Med 28(7):2507–2514
Awad AS, Rouse M, Huang LP et al (2009) Compartmentalization of neutrophils in the kidney and lung following acute ischemic kidney injury. Kidney Int 75(7):689–698
Rajendran P, Rengarajan T, Thangavel J et al (2013) The vascular endothelium and human diseases. Int J Biol Sci 9(10):1057–1069
Page AV, Liles WC (2013) Biomarkers of endothelial activation/dysfunction in infectious diseases. Virulence 4(6):507–516
Reitsma S, Slaaf DW, Vink H, van Zandvoort MA, oude Egbrink MG. The endothelial glycocalyx: composition, functions, and visualization. Pflugers Arch. 2007;454(3):345–359
Mota SMB, Albuquerque PLMM, Meneses GC, da Silva Junior GB, Martins AMC, De Francesco DE (2021) Role of endothelial biomarkers in predicting acute kidney injury in Bothrops envenoming. Toxicol Lett 345:61–66
Fiorentino M, Tohme FA, Wang S et al (2018) Long-term survival in patients with septic acute kidney injury is strongly influenced by renal recovery. PLoS One 13(6):e0198269
Zonneveld R, Martinelli R, Shapiro NI et al (2014) Soluble adhesion molecules as markers for sepsis and the potential pathophysiological discrepancy in neonates, children and adults. Crit Care 18(2):204
Ley K (2003) The role of selectins in inflammation and disease. Trends Mol Med 9(6):263–268
Pike F, Murugan R, Keener C et al (2015) Biomarker enhanced risk prediction for adverse outcomes in critically Ill patients receiving RRT. Clin J Am Soc Nephrol 10(8):1332–1339
Funding
This study was funded by a Grant for key scientific research projects in Hebei Provincial Health Commission (Grant No: 20160138; 20160592).
Author information
Authors and Affiliations
Contributions
Project administration: LY and GJY; conceptualization: HQS; funding acquisition: HQS; supervision: FMX and WZY; investigation and data curation: LMY; writing—original draft: LY. Writing—review and editing: LY. Approval of the final manuscript: all authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of The Ethics Committee of the Third Hospital of Hebei Medical University (approval number: KE-2019-20-1, KE-2019-19-1) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Li, Y., Huang, Q., Fang, M. et al. Prognostic value of serum levels of multiple adhesion factors in patients with sepsis-induced acute kidney injury. Int Urol Nephrol 55, 1229–1237 (2023). https://doi.org/10.1007/s11255-022-03394-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-022-03394-z