Abstract
Background
Intravenous fluid therapy is important for pediatric and adult patients in intensive care units (ICUs). However, medical professionals continue to struggle to determine the most appropriate fluids to obtain the best possible outcomes for each patient.
Objective
We conducted a meta-analysis involving cohort studies and randomized controlled trials (RCTs) to compare the influence of balanced crystalloid solutions and normal saline among patients in ICUs.
Patients and methods
Studies that compared balanced crystalloid solutions and saline in ICU patients from databases including PubMed, Embase, Web of Science, and Cochrane Library were systematically searched up to July 25, 2022. The primary outcomes were mortality and renal-related outcomes, which included major adverse kidney events within 30 days (MAKE30), acute kidney injury (AKI), new receipt of renal replacement therapy (RRT), maximum creatinine increasing, maximum creatinine level, and final creatinine level ≥ 200% of baseline. Service utilization including length of hospital stay, ICU stay, ICU-free days and ventilator-free days were also reported.
Results
A total of 13 studies (10 RCTs and 3 cohort studies) involving 38,798 patients in ICUs met the selection criteria. Our analysis revealed that each subgroup had no significant difference in mortality outcomes among ICU patients between balanced crystalloid solutions and normal saline. A significant difference was detected between the adult groups (odds ratio [OR], 0.92; 95% confidence interval [CI], [0.86, 1.00]; p = 0.04) indicating that the AKI in the balanced crystalloid solutions group was lower than that in the normal saline group. Other renal-related outcomes, such as MAKE30, RRT, maximum creatinine increasing, maximum creatinine level, and final creatinine level ≥ 200% of baseline showed no significant difference between the two groups. Regarding secondary outcomes, the balanced crystalloid solution group had a longer ICU stay time (WMD, 0.02; 95% CI, [0.01, 0.03]; p = 0.0004 and I2 = 0%; p = 0.96) than the normal saline group among adult patients. Furthermore, children treated with balanced crystalloid solution had a shorter hospital stay time (WMD, − 1.10; 95% CI, [− 2.10, − 0.10]; p = 0.03 and I2 = 17%; p = 0.30) than those treated with saline.
Conclusions
Compared with saline, balanced crystalloid solutions could not reduce the risk of mortality and renal-related outcomes, including MAKE30, RRT, maximum creatinine increasing, maximum creatinine level, and final creatinine level ≥ 200% of baseline, but the solutions may reduce total AKI incidence among adult patients in ICUs. For service utilization outcomes, balanced crystalloid solutions were associated with a longer length of ICU stay in the adult group and shorter length of hospital stay in the pediatric group.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Compared with common hospital wards, the intensive care unit (ICU) is a specialized unit with more necessary support equipment and rescue resources, as it must provide life care and support to critically ill patients, stop or slow down organ failure processes, and ultimately reduce the risk of death [1]. For pediatric and adult patients in critical situations, fluid therapy is essential in resuscitation and maintenance [2], and proper intravenous fluid therapy can maintain vascular volume and improve organ perfusion [3, 4]. Normal 0.9% saline and balanced crystalloid solutions are common medical intravenous fluids that are widely used in ICUs and other hospital departments.
Normal saline is an isotonic fluid containing a high concentration of chloride ions, compared with plasma, while balanced crystalloid solutions have a similar chloride concentration to that of plasma [5]. A growing body of evidence suggests that high chloride concentrations in the circulatory system may increase the risk of adverse outcomes in both adults and children [6, 7]. The possible reason for these outcomes is that compared with balanced crystalloid fluids, 0.9% saline is more likely to lead to an internal electrolyte imbalance that may cause hyperchlorinated metabolic acidosis, a reduction in the glomerular filtration rate and fluid overload in the body [8,9,10]. Previous studies have shown that normal saline is associated with a higher incidence of renal dysfunction and overall mortality than balanced crystalloid solutions [11].
In recent years, a number of studies have evaluated balanced multielectrolyte solutions as a replacement for normal saline for intravenous fluid resuscitation in patients supported in ICUs. Hayes [12] showed that balanced crystalloid fluids were more suitable for critically ill children than saline and could also significantly reduce overall mortality and renal-injury events in adults. Nevertheless, some literature has reported that balanced crystalloid solutions did not effectively improve prognosis in ICU patients, compared with normal saline [5, 13]. Because of physiological differences, fluid therapy in children can be extremely different from that in adults. In addition, various intravenous fluid protocols remain are based on the cause of admission to the ICU. To provide clear guidance on intravenous fluid therapy among patients in ICUs, further research is needed to compare balanced crystalloid solutions and normal saline. Therefore, our systematic review and meta-analysis was conducted to investigate related outcomes.
Materials and methods
Literature research
Our prospective study followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis 2020 statement, Supplement Table 1) and PROSPERO register (Number: CRD42022347375). Databases, including PubMed, Embase, Web of Science (WOS) and Cochrane library, were systematically searched for primary literature that compared saline and balanced crystalloid fluids among ICU patients up to February, 2023. The following items were used in our search: “Balanced crystalloid solutions,” “saline,” and “intensive care units” (the specific search strategy is shown in Supplement Table 2).
Identification of eligible studies
The following studies were considered eligible: (1) study design was randomized controlled trials (RCT) or cohort studies; (2) studies were conducted on ICU patients; (3) studies comparing balanced crystalloid and normal saline; (4) at least one mortality outcome (28-day mortality; 30-day mortality; 60-day mortality; 90-day mortality; or hospital mortality), renal function (major adverse kidney events within 30 days (MAKE30), acute kidney injury (AKI), new receipt of renal replacement therapy(RRT), maximum creatinine increasing, maximum creatinine level and final creatinine level ≥ 200% of baseline) or service utilization outcomes (length of hospital stay, ICU stay, ICU-free days, and ventilator-free days) was evaluated; and (5) sufficient data available to calculate odds ratio (OR) or weighted mean difference (WMD). Reviews, letters, editorial comments, case reports, unpublished articles, study protocols, and non-English articles were excluded. For different primary studies enrolling participants from the same database, we only included the study with the largest sample size if they had same experimental timing and inclusion criteria. In contrast, the literature with dissimilar study periods and inclusion standards were also included.
Data extraction
Two investigators (Puze Wang and Yin Huang) performed data extractions independently, and a third investigator (Liangren Liu) made a final decision if there were any disagreements. Data for the following aspects were recorded: (1) basic literature information: first author, publication year, study period, country of study, study design, sample size, type of balanced crystalloids, center numbers; (2) demographic data: age, sex, comorbidities, renal functional indicators (creatinine, blood urea nitrogen, and estimate glomerular filtration rate, [EGFR]) and critical illness index (SOFA score and APACHE II score); (3) mortality outcomes: 28-day, 30-day, 60-day, 90-day, and hospital mortality; (4) renal-related outcomes: MAKE30, AKI, RRT, maximum creatinine increasing, maximum creatinine level, and final creatinine level ≥ 200% of baseline; (5) service utilization outcomes: length of hospital stay, ICU stay, ICU-free days, and ventilator-free days. The meaning of maximum creatinine increasing was described as “a standard that measuring the degree of change in kidney function” because changes in the creatinine concentration reflect the quality of kidney function indirectly. In addition, AKI was qualitatively defined as “abrupt and sustained decrease in glomerular filtration, urine output, or both” [14], and if possible, we extracted data of AKI, respectively, according to the KDIGO criteria [15].
If studies reported continuous variables such as the median with range or interquartile range, we used the validated mathematical method to obtain a concrete mean ± standard deviation [16, 17].
Quality assessment
The Newcastle–Ottawa Scale (NOS) and Cochrane Risk of Bias Assessment Tool (CRBAT) were utilized to evaluate the quality of the included studies [18, 19]. Studies scoring 7–9 points were considered to be high quality [20]. In addition, the evidence level for every study was also assessed according to the Oxford Centre for Evidence-Based Medicine Levels of Evidence Working Group [21]. Evaluations were launched by the same two investigators independently, and any discrepancies were resolved through discussion. All related information are shown in Supplement Table 3.
Statistical analysis and publication bias
Review Manager version 5.4 (Cochrane Collaboration, Oxford, UK) was used to perform evidence synthesis. We compared dichotomous and continuous variables applied for OR and WMD, and used 95% confidential intervals [CIs] to report on all metrics; the Chi-squared (χ2) test and inconsistency index (I2) were used to assess any potential heterogeneities in our included studies. An χ2 P value < 0.05 or I2 > 50% indicated that significant heterogeneity existed. A random-effect model was used to estimate the combined WMD or OR was used when significant heterogeneity was detected (χ2 P value < 0.05 or I2 > 50%). In addition, one-way sensitivity analyses were performed to estimate the effects of studies on the combined results for outcomes if obvious heterogeneity was detected. Otherwise, publication bias was evaluated visually by funnel plots via Review Manager version 5.4.
Results
Literature search and study characteristics
The flowchart of our search and selection process is presented in Fig. 1. In total, 1450 relevant articles in PubMed (n = 218), Embase (n = 630), Web of Science (n = 350) and Cochrane Library (n = 259) were identified through a systematic automatic and artificial search. After reviewing the titles and abstracts, 1, 334 articles were excluded, and 59 studies were selected for the second screening after additionally excluding 64 duplicated studies. Finally, 13 full-text articles involving 38,798 patients were included in this meta-analysis [11, 22,23,24,25,26,27,28,29,30,31,32,33]. Of these studies, 10 were randomized controlled trials (RCTs), and 3 were cohort studies. Patients with sepsis, trauma, and neurological or cardiac situations were recorded separately, and subgroup analysis was performed. Pediatric patients were identified for another subgroup analysis. If studies involved three types of fluids, we repeated the inclusion of the same study with different comparisons.
Table 1 shows the baseline characteristics of the included studies.
Mortality outcomes
Data on mortality outcomes were synthesized from 10 studies. Subgroup analysis was performed according to mortality time. Pooled analysis revealed that each patient subgroup had no significant difference in mortality outcomes based on the use of balanced crystalloid solutions and normal saline, with no apparent heterogeneity, aside from the 30-day mortality group in cohort studies (Fig. 2A). A visual assessment of the funnel plot indicated apparent publication bias in 28-day mortality and hospital mortality groups (Fig. 2B).
A Forest plots of mortality outcomes: (1) 28-day mortality, (2) 30-day mortality, (3) 60-day mortality, (4) 90-day mortality, and (5) hospital mortality. B Funnel plot of mortality outcomes: (1) 28-day mortality, (2) 30-day mortality, (3) 60-day mortality, (4) 90-day mortality, and (5) hospital mortality
Renal function outcomes
MAKE30
MAKE30 was described as the composite of death, new receipt of RRT, or persistent renal dysfunction within 30 days after enrollment. In total, 4 studies (3 RCTs and 1 cohort study) reported the data of MAKE30 for 3,709 patients (1,776 balanced crystalloid group vs. 1,933 normal saline group). After excluding the data from the cohort study, no significant difference was detected between the two groups (OR, 0.92; 95% CI, [0.85, 1.00]; p = 0.06] with no obvious heterogeneity (I2 = 0%, p = 0.53), indicating that the MAKE30 in the balanced crystalloid solutions group was similar to that in the normal saline group. However, the funnel plot indicated publication bias in the primary studies.
AKI
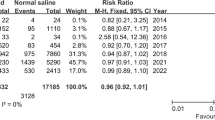
In total, 9 studies involving 3,092 patients (1512 balanced crystalloid group vs. 1,580 normal saline group) were included in the meta-analysis. After grouping studies according to patient age, our results demonstrated that adult patients who accepted balanced crystalloid solutions had a lower total AKI incidence than those who accepted saline (OR, 0.92; 95% CI, [0.86, 1.00]; p = 0.04). However, in children patients and adults with KDIGO ≥ 2, both groups had similar incidences in AKI (children: OR, 1.26; 95% CI, [0.70, 2.25]; p = 0.44; adults with KDIGO ≥ 2: OR, 0.94; 95% CI, [0.88, 1.01]; p = 0.09) with no significant heterogeneity (adults and children: I2 = 0%). In addition, a visual assessment of the funnel plot indicated slight publication bias among the pediatric patients.
RRT
Of the 8 studies reporting RRT, 6 were selected for analysis. Pooled analysis detected no significant difference between the balanced crystalloid and saline in the RRT rate (OR, 0.94; 95% CI, [0.88, 1.01]; p = 0.09), with no significant heterogeneity (I2 = 26%, p = 0.24). In addition, funnel plots revealed no apparent publication bias. After developing subgroups according to ICU admission cause (sepsis), we still reach the same conclusion with significant heterogeneity (Figure S1).
Final creatinine level ≥ 200% of baseline
In total, 3 studies reported 4,095 patients (2,015 balanced crystalloid group vs. 2,080 normal saline group) whose final creatinine levels were double that of the baseline. Similar results were observed between these two groups in our analysis (OR, 0.96; 95% CI, [0.90, 1.03]; p = 0.26), with no statistically significant heterogeneity (I2 = 0%, p = 0.60). The funnel plot did not suggest a publication bias.
Maximum creatinine increasing and maximum creatinine level
Serum creatinine has been considered as a gold standard determiner for the diagnosis of AKI for approximately 100 years [34]. Creatinine levels are strongly associated with the glomerular filtration rate. Hence, an increase of maximum creatinine increasing may represent the degree of kidney impairment. Data on the maximum creatinine increasing and maximum level after intravenous fluid therapy were extracted from 3 studies with 21,813 patients in the ICU (10,977 balanced crystalloid group vs. 10,836 normal saline group). No significant difference and heterogeneity were detected from the forest plot between balanced crystalloid solutions and 0.9% normal saline (maximum creatinine increasing: WMD, -0.00; 95% CI, [-0.01, 0.01]; p = 0.53 and I2 = 0%; p = 0.99; maximum creatinine level: WMD, 0.00; 95% CI, [-0.02, 0.02]; p = 0.85 and I2 = 0%; p = 0.44). In addition, no significant publication bias was observed from the funnel plot.
All analyses and forest plots of renal-related outcomes are shown in Fig. 3. Figure 4 presents the funnel plots of the above outcomes.
Service utilization outcomes
Length of ICU stay
In total, 8 studies with 15,960 involved patients (7872 balanced crystalloid group vs. 8088 normal saline group) revealed the length of ICU stay data. After conducting subgroup analyses, our study reported that the adults who accepted balanced crystalloids solution had longer lengths of ICU stay with no significant heterogeneity (adults: WMD, 0.02; 95% CI, [0.01, 0.03]; p = 0.0004 and I2 = 0%; p = 0.96), while no difference was found in both two groups of pediatric patients (children: WMD, – 0.05; 95% CI, [– 0.48, 0.39]; p = 0.84 and I2 = 68%; p = 0.02). In addition, for both RCT and cohort studies, the two groups had similar lengths of ICU stay (RCT: WMD, – 0.01; 95% CI, [– 0.13, 0.11]; p = 0.85; cohort studies: WMD, – 0.94; 95% CI, [– 4.71, 2.83]; p = 0.63), with apparent heterogeneity in the cohort group (RCT: I2 = 48%, p = 0.06; cohort studies: I2 = 99%, p < 0.00001). Funnel plots revealed a slight publication bias in the pediatric group.
Length of hospital stay
In total, 7 articles reported the length of hospital stay. The results of the pediatric subgroup indicated patients who accepted balanced crystalloid solutions had shorter hospital stay time compared with those in normal saline group, with no obvious heterogeneity (WMD, – 1.10; 95% CI, [– 2.10, – 0.10]; p = 0.03 and I2 = 17%; p = 0.30). In other subgroups, including the RCT group, cohort group, and adult group, different degrees of heterogeneity were revealed clearly with similar hospital lengths of stay (adults: WMD, − 0.05; 95% CI, [− 0.45, 0.35]; p = 0.85 and I2 = 80%; p = 0.03; RCT: WMD, − 0.35; 95% CI, [− 0.86, 0.15]; p = 0.17 and I2 = 65%; p = 0.009; cohort studies: WMD, − 2.48; 95% CI, [− 6.27, 1.31]; p = 0.20 and I2 = 89%; p = 0.002). Funnel plots also revealed apparent publication bias in pediatric and RCT groups.
ICU-free days
Data on ICU-free days were reported in 2 RCT studies that involved 16,776 patients (8,462 in balanced crystalloids and 8,314 in saline) totally. The forest plot revealed that these two groups had similar ICU-free days with no significant heterogeneity (WMD = 0.00, 95% CI = [– 0.07, 0.07], p = 1.00, I2 = 0%, p = 1.00). In addition, the funnel plot also detected a negative publication bias.
Ventilator-free days
Further, 3 articles reported ventilator-free days of 13,679 patients in the balanced crystalloid group and 13,601 in the normal saline group (27,280 in total). Our study reports that there was no significant difference between those two groups with significant heterogeneity (WMD, 0.41; 95% CI, [− 0.06, 0.88]; p = 0.09 and I2 = 92%; p < 0.00001). Publication bias was not apparent according to the results of the funnel plot.
Related information of service utilization outcomes is shown in Figs. 5 and 6.
Sensitivity analysis
Sensitivity analysis was conducted to resolve the existence of heterogeneity in outcomes of ventilator-free days by excluding studies in the series through Review Manager 5.4. Results revealed that significant heterogeneity could not be eliminated by excluding any involved primary studies. Moreover, all analyses in subgroups of cohort studies report apparent heterogeneity that could not be resolved by removing any study.
Discussion
As the cornerstone treatment of critically ill patients, intravenous fluid administration is necessary for management in ICUs [35, 36]. Normal saline is the earliest and most common crystalloid solution that has been widely used in fluid resuscitation, while balanced crystalloid solutions have a different composition, as chloride anions are replaced with bicarbonate or buffers [37]. To maintain the homeostasis of patients, balanced crystalloid solutions are classified based on their elements and have different indications, contraindications, and prognoses. The balanced crystalloids used in our study mainly include Plasma-Lyte-A, Sterofundin, sodium bicarbonate Ringer’s solution, and lactated Ringer’s solution. The safety of organ function with intravenous fluids among patients in the ICU is prioritized by monitoring arterial blood gas. The influence of a high chloride concentration in saline on acidosis has long been reported in existing literature [38, 39], and its specific mechanisms may be related to plasma dilution and changes in blood carbonic acid concentrations [40, 41]. One retrospective cohort study involving 60,374 patients divided intravenous fluids into three groups (saline, saline with crystalloid, and saline with colloform) and investigated the mortality of each group. The study found that the saline group and the balanced crystalloid group had the highest and lowest mortality, rates, respectively [42]. However, the severity of acidosis and its impact on patients’ internal environments and organ functions remain uncertain. Some studies suggests that as patients can develop a strong tolerance to acidosis [43, 44], it cannot be the definitive cause of kidney dysfunction [45]. In addition, although several studies have reported that normal saline can reduce the glomerular filtration rate and renal artery flow velocity [12], these adverse effects can be reduced through comprehensive treatments including vasopressor drugs, respiratory stimulant drugs, or mechanical ventilation in the ICU. Moreover, balanced crystalloid solution can potentially be unsafe. For example, Plasma-Lyte-A can increase the risk of hyperkalemia and hypermagnesemia in patients.
Our study focused on the following three important indicators of prognosis: mortality, renal-related, and service utilization outcomes. The mortality outcomes are divided into several subgroups including 28-day, 30-day, 60-day, 90-day, and hospital mortality. Some previous studies reported that saline administration can increase the mortality of critically ill patients [7, 11], while other studies report no significant improvement in mortality after administration of balanced crystalloid solutions [25, 29]. Our analysis found that the mortality rate was similar after administration of balanced crystalloid solutions and normal saline in critically ill patients and further found that balanced crystalloid solutions did not have an advantage in reducing mortality in ICU patients, both children and adults. However, by contrast, a recent meta-analysis involving 13 RCTs showed that balanced crystalloid solutions can reduce mortality in critically ill adults. Our analysis concluded that there was no evident difference in MAKE30 between these two groups, similar to previous studies [46, 47]. It was detected that the balanced crystalloid solutions could not decrease the MAKE30 rate of ICU patients. However, publication bias was present in our study, as we combined adult and pediatric patients, suggesting that additional research is necessary to reduce publication bias. In addition, for other renal-related outcomes, our analysis found that all outcomes, except AKI, between balanced crystalloid solutions and normal saline had similar results. We revealed that balanced crystalloid solutions could reduce AKI incidence among adults, but not among children. This result resembles that of a recent study that reported relative outcomes among adults in critical conditions and further verified the previous confirmations. Furthermore, regarding service utilization outcomes, length of ICU and hospital stay, ICU-free days, and ventilator-free days were also included in our analysis. Primary analysis showed that although length of ICU stay among all patients was similar between the two groups, the subgroup of both adults and children revealed that balanced crystalloid solution yielded a longer ICU stay than saline, different from previous studies. No significant difference between balanced crystalloid solution and normal saline was present in other service utilization outcomes.
Moreover, our study found similar RRT rates among adult patients in both groups, although the risk of AKI was lower in patients treated with balanced crystalloid solutions. The AKI Network (AKIN) criteria classify AKI into risk, injury, and failure categories [48, 49]. In general, only patients with end-stage renal failure need RRT, and it is unnecessary for patients classified under AKIN Stage 1 or 2. However, although we had conducted subgroup analysis according to KDIGO criteria, we still could not include more detailed subgroups based on the AKIN classification because of the limitation of data in primary studies. Hence, more analyses, including the correlations between categories of AKI and different intravenous fluid infusion therapy are needed.
Our study further contributes to the understanding of the efficacy of balanced crystalloid solution and normal saline. Compared with previous studies, our results included as many indicators as possible to develop an overall assessment of the two groups. To reduce the bias caused by age, both adults and children were involved in our analysis. Subgroups were also created based on the type of study and age. In addition, the causes of ICU admission among patients were also recorded because some causes have different influences on the volume of infusion. However, we also acknowledge that there are several limitations to our study. For example, special situations like sepsis or brain trauma need more intravenous fluids to resuscitate, but we did not develop additional subgroups to analyze patients with different admission causes to compensate for the unavailability of data in primary studies, which may impact the final result with discrepancy of infusion volumes. Further, due to the small size of primary studies, the types of balanced crystalloid solutions were not allocated to subgroups, creating potential bias of our results, especially for renal-related outcomes. Moreover, some outcomes in our analysis, like all outcomes of cohort studies, have significant heterogeneity that cannot be eliminated, decreasing the reliability of our results. Thus, although our analysis revealed some meaningful results comparing balanced crystalloid solutions and normal saline, more primary studies, especially surrounding fluids therapy among children in the ICU are still needed in the future.
Conclusion
Compared with saline, balanced crystalloid solutions could not reduce the risk of mortality and renal-related outcomes, including MAKE30, RRT, maximum creatinine increasing, maximum creatinine level, and final creatinine level ≥ 200% of baseline, but could reduce total AKI incidence in adults. Regarding service utilization outcomes, balanced crystalloid solutions are associated with a longer length of ICU stay among adult patients and a shorter length of hospital stay among pediatric patients than saline. To address existing limitations, more high-quality studies are necessary in the future.
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Ervin JN, Kahn JM, Cohen TR, Weingart LR (2018) Teamwork in the intensive care unit. Am Psychol 73(4):468–477
Malbrain MLNG, Langer T, Annane D, Gattinoni L, Elbers P, Hahn RG et al (2020) Intravenous fluid therapy in the perioperative and critical care setting: executive summary of the International Fluid Academy (IFA). Ann Intensive Care 10(1):64
Hoste EA, Maitland K, Brudney CS, Mehta R, Vincent JL, Yates D, Kellum JA, Mythen MG, Shaw AD, ADQI XII Investigators Group (2014) Four phases of intravenous fluid therapy: a conceptual model. Br J Anaesth 113(5):740–747
Centre NCG (2015) IV fluids in children: intravenous fluid therapy in children and young people in hospital. National Institute for Health and Care Excellence, London
Reddy S, Weinberg L, Young P (2016) Crystalloid fluid therapy. Crit Care 15(20):59
Stenson EK, Cvijanovich NZ, Anas N, Allen GL, Thomas NJ, Bigham MT et al (2018) Hyperchloremia is associated with complicated course and mortality in pediatric patients with septic shock. Pediatr Crit Care Med 19(2):155–160
Raghunathan K, Shaw A, Nathanson B, Stürmer T, Brookhart A, Stefan MS, Setoguchi S et al (2014) Association between the choice of IV crystalloid and in-hospital mortality among critically ill adults with sepsis*. Crit Care Med 42(7):1585–1591
Bampoe S, Odor PM, Dushianthan A, Bennett-Guerrero E, Cro S, Gan TJ et al (2017) Perioperative administration of buffered versus non-buffered crystalloid intravenous fluid to improve outcomes following adult surgical procedures. Cochrane Database Syst Rev 9(9):CD004089
Chowdhury AH, Cox EF, Francis ST, Lobo DN (2012) A randomized, controlled, double-blind crossover study on the effects of 2-L infusions of 0.9% saline and plasma-lyte® 148 on renal blood flow velocity and renal cortical tissue perfusion in healthy volunteers. Ann Surg 256(1):18–24
Reid F, Lobo DN, Williams RN, Rowlands BJ, Allison SP (2003) (Ab)normal saline and physiological Hartmann’s solution: a randomized double-blind crossover study. Clin Sci 104(1):17–24
Semler MW, Self WH, Wanderer JP, Ehrenfeld JM, Wang L, Byrne DW, SMART Investigators and the Pragmatic Critical Care Research Group et al (2018) Balanced crystalloids versus saline in critically ill adults. N Engl J Med 378(9):829–839
Hayes W (2019) Ab-normal saline in abnormal kidney function: risks and alternatives. Pediatr Nephrol 34(7):1191–1199
Hammond DA, Lam SW, Rech MA, Smith MN, Westrick J, Trivedi AP et al (2020) Balanced crystalloids versus saline in critically ill adults: a systematic review and meta-analysis. Ann Pharmacother 54(1):5–13
Kellum JA, Levin N, Bouman C, Lameire N (2002) Developing a consensus classification system for acute renal failure. Curr Opin Crit Care 8(6):509–514
Khwaja A (2012) KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 120(4):c179–c184
Luo D, Wan X, Liu J, Tong T (2018) Optimally estimating the sample mean from the sample size, median, mid-range and/or mid-quartile range. Stat Methods Med Res 27:1785–1805
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol 14:135
Wells G, Shea B, O’Connell D, et al. (2011) The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies inMeta-Analyses. Available at:http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed 20 Jul 2022
Higgins JP, Altman DG, Gøtzsche PC et al (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928
Huang Y, Cao D, Chen Z, Chen B, Li J, Guo J et al (2021) Comparison of perioperative, renal functional, and oncological outcomes between off-clamp and on-clamp robot-assisted partial nephrectomy for renal tumors: an updated evidence-based analysis. Front Oncol 21(11):730662
Howick J, Chalmers I, Glasziou P, et al. Explanation of the 2011 Oxford Centre for Evidence-Based Medicine(OCEBM) Levels of Evidence (Background Document):Oxford Centre for Evidence-Based Medicine. Available at: https://www.cebm.ox.ac.uk/resources/levels-of-evidence/explanation-of-the-2011-ocebm-levels-of-evidence/.
Raman S, Gibbons KS, Mattke A, Schibler A, Trnka P et al (2023) Effect of saline vs gluconate/acetate-buffered solution vs lactate-buffered solution on serum chloride among children in the pediatric intensive care unit: the SPLYT-P randomized clinical trial. JAMA Pediatr 177(2):122–131
Shephali S, Singh Y, Mishra M, Sachan S, Pathak NM, Shankar R (2022) Comparison of normal saline and balanced salt solution as a maintenance fluid on acids-base and electrolyte status in traumatic brain injury patients a prospective randomized double-blind study. Anaesth Pain Intensive Care. 26(3):326–331
Scioscia A, Horvat C, Moritz ML, Fuhrman D (2022) Balanced crystalloids versus normal saline in children with critical asthma. Children 9(10):1480
Bian Y, Xu T, Le Y, Li S (2022) The efficacy and safety of sodium bicarbonate ringer’s solution in critically Ill patients: a retrospective cohort study. Front Pharmacol 30(13):829394
Tseng CH, Chen TT, Chan MC, Chen KY, Wu SM, Shih MC et al (2021) Impact of comorbidities on beneficial effect of lactated ringers vs saline in sepsis patients. Front Med 13(8):758902
Trepatchayakorn S, Sakunpunphuk M, Samransamruajkit R (2021) Balanced salt solution versus normal saline in resuscitation of pediatric sepsis: a randomized. Controlled Trial Indian J Pediatr 88(9):921–924
Semler MW, Wanderer JP, Ehrenfeld JM, Stollings JL, Self WH, Siew ED, SALT Investigators and the Pragmatic Critical Care Research Group; SALT Investigators et al (2017) Balanced crystalloids versus saline in the intensive care unit the SALT randomized trial. Am J Respir Crit Care Med 195(10):1362–1372
Finfer S, Micallef S, Hammond N, Navarra L, Bellomo R, Billot L, Delaney A, Gallagher M, Gattas D, Li Q, Mackle D, Mysore J, Saxena M, Taylor C, Young P, Myburgh J, PLUS Study Investigators; Australian New Zealand Intensive Care Society Clinical Trials Group (2022) Balanced multielectrolyte solution versus saline in critically ill adults. N Engl J Med 386(9):815–826
Williams V, Jayashree M, Nallasamy K, Dayal D, Rawat A (2020) 0.9% saline versus Plasma-Lyte as initial fluid in children with diabetic ketoacidosis (SPinK trial): a double-blind randomized controlled trial. Crit Care 24(1):1
Ratanarat R, Sanguanwit PCA et al (2017) The effects of normal saline versus balanced crystalloid solution as a resuscitation fluid on acute kidney injury in shock patients: a randomized opened label-controlled trial. Intensive Care Med Exp 5:400
Zampieri FG, Machado FR, Biondi RS, Freitas FGR, Veiga VC, Figueiredo RC et al (2021) Effect of intra-venous fuid treatment with a balanced solution vs 0.9% saline solution on mortality in critically Ill patients: the BaSICS randomized clinical trial. JAMA 326(9):1–12
Young P, Bailey M, Beasley R, Henderson S, Mackle D, McArthur C, SPLIT Investigators; ANZICS CTG et al (2015) Effect of a buffered crystalloid solution vs saline on acute kidney injury among patients in the intensive care unit: the SPLIT randomized clinical trial. JAMA 314(16):1701–1710
Slocum JL, Heung M, Pennathur S (2012) Marking renal injury: can we move beyond serum creatinine? Transl Res 159(4):277–289
Hoorn EJ (2017) Intravenous fluids: balancing solutions. J Nephrol 30(4):485–492
Awad S, Allison SP, Lobo DN (2008) The history of 0.9% saline. Clin Nutr 27(2):179–188
Semler MW, Kellum JA (2019) Balanced crystalloid solutions. Am J Respir Crit Care Med 199(8):952–960
Kellum JA, Bellomo R, Kramer DJ, Pinsky MR (1998) Etiology of metabolic acidosis during saline resuscitation in endotoxemia. Shock 9(5):364–368
Kellum JA (2002) Fluid resuscitation and hyperchloremic acidosis in experimental sepsis: improved short-term survival and acid-base balance with Hextend compared with saline. Crit Care Med 30(2):300–305
Gattinoni L, Carlesso E, Maiocchi G, Polli F, Cadringher P (2009) Dilutional acidosis: where do the protons come from? Intensive Care Med 35(12):2033–2043
Astrup P, Jorgensen K, Andersen OS, Engel K (1960) The acid-base metabolism. A new approach. Lancet 1(7133):1035–1039
Raghunathan K, Bonavia A, Nathanson BH, Beadles CA, Shaw AD, Brookhart MA et al (2015) Association between initial fluid choice and subsequent in-hospital mortality during the resuscitation of adults with septic shock. Anesthesiology 123(6):1385–1393
Bangsbo J, Madsen K, Kiens B, Richter EA (1996) Effect of muscle acidity on muscle metabolism and fatigue during intense exercise in man. J Physiol 495(Pt2):587–596
Sjøgaard G, Adams RP, Saltin B (1985) Water and ion shifts in skeletal muscle of humans with intense dynamic knee extension. Am J Physiol 248(2 Pt 2):R190–R196
Schieve JF, Wilson WP (1953) The changes in cerebral vascular resistance of man in experimental alkalosis and acidosis. J Clin Invest 32(1):33–38
Zhu Y, Guo N, Song M, Xia F, Wu Y, Wang X et al (2021) Balanced crystalloids versus saline in critically ill patients: the PRISMA study of a meta-analysis. Medicine 100(38):e27203
Zwager CL, Tuinman PR, de Grooth HJ, Kooter J, Ket H, Fleuren LM et al (2019) Why physiology will continue to guide the choice between balanced crystalloids and normal saline: a systematic review and meta-analysis. Crit Care 23(1):366
Mehta RL, Kellum JA, Shah SV, Molitoris BA, Ronco C, Warnock DG et al (2007) Acute Kidney Injury Network. Acute kidney injury network: report of an initiative to improve outcomes in acute kidney injury. Crit Care 11(2):R31
Xiong J, Tang X, Hu Z, Nie L, Wang Y, Zhao J (2015) The RIFLE versus AKIN classification for incidence and mortality of acute kidney injury in critical ill patients: a meta-analysis. Sci Rep 7(5):17917
Acknowledgements
We thank all the authors of the included articles and all the editors who reviewed this article.
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
PW: main author, literature research and quality score evaluation. YH: literature research and quality score evaluation. LL: manuscript review and literature research. BC, DC, ZC, JL, JL, RW: manuscript review.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical approval
An ethics statement is not required because this study was based exclusively on published literature.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, P., Huang, Y., Li, J. et al. Balanced crystalloid solutions versus normal saline in intensive care units: a systematic review and meta-analysis. Int Urol Nephrol 55, 2829–2844 (2023). https://doi.org/10.1007/s11255-023-03570-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-023-03570-9