Abstract
The seroprevalence of Paslahepevirus balayani genotype 3 (hepatitis E virus genotype 3 – HEV-3; Hepeviridae family, genus Paslahepevirus) in pet cats, dogs and rabbits was evaluated. Samples from cats and dogs were collected from three veterinary practices from various parts of Poland: Poznan (wielkopolskie voivodeship), Przemysl (podkarpackie voivodeship) and Lublin (lubelskie voivodeship). Samples from rabbits were collected in Poznan. In total, serum samples from 90 cats, 82 dogs and 71 rabbits were selected and tested for specific anti-HEV-3 immunoglobulin (IgG) antibodies using a commercial ELISA test. Pathogen seroprevalence among rabbits was calculated at a 95% confidence interval (CI) for each gender, age (up to 12 months, 1–3 years, 4–7 years and over 8 years), symptoms group (healthy, gastrointestinal disorders, other disorders) and compared with a chi-squared test. No anti-HEV-3 IgG antibodies were detected in any of the samples from cats and dogs. Anti-HEV-3 IgG antibodies were detected in 2.82% of the serum samples from rabbits (2/71; 95% CI: 0.78–9.70). No significant correlations between seropositivity and gender, age, and symptoms (p > 0.05) were observed in rabbits. Our findings indicate that pet rabbits in Poland are exposed to HEV-3, develop humoral response due to infection and might constitute a source for HEV-3 transmission to humans.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Paslahepevirus balayani genotype 3 (hepatitis E virus genotype 3 – HEV-3) includes variants able to infect humans and several mammalian species. According to the 2021 release of the International Committee on the Taxonomy of Viruses, HEV belongs to the Hepeviridae family, Orthohepevirinae subfamily (divided into four genera: Paslahepevirus, Avihepevirus, Rocahepevirus and Chirohepevirus). Genus Paslahepevirus contains two species, P. balayani (formerly known as Orthohepevirus A) and Paslahepevirus alci (Purdy et al. 2022). Eight distinct genotypes thus far have been proposed within HEV with four major genotypes (1–4) implicated in human disease (Pirani et al. 2023). HEV-1 and HEV-2 infect only humans and cause acute hepatitis, predominantly in developing countries, where are spread orofaecally via contaminated water supplies (Dalton et al. 2014). HEV-3 and HEV-4 are well-recognized zoonotic strains that circulate among a broad spectrum of animal species, including pigs and wild boar, which are considered the main reservoirs (Li et al. 2022). In developed countries, HEV-3 and HEV-4 have been found in all stages of the human food chain and one established route of transmission from pigs to humans is via undercooked or uncooked pig meat products (Dalton et al. 2014). However, HEV-3 has also been found in soft fruits such as strawberries (Brassard et al. 2012). It can also be transmitted iatrogenic with blood transfusions or organ transplants (Dalton et al. 2014). Animal species other than pigs may also contribute to the spread of zoonotic HEV genotypes to humans. HEV-3 and HEV-4 RNA was detected in the liver (Wu et al. 2015; Li et al. 2017; Go et al. 2019) and milk (Huang et al. 2016; Demirci et al. 2019) samples collected from cows, goats and sheep. HEV-1, HEV-3 and HEV-4 were found in ruminants (Demirci et al. 2019). It poses a real risk of infection with zoonotic HEV genotypes after consuming infected unpasteurized milk or edible organs from these animals. Rabbits are the natural host of HEV-3 and are considered the main reservoir of HEV second to pigs (Wang et al. 2017b). Humans can acquire HEV-3 infection from rabbits via consumption of their undercooked meat or via contact with contaminated feces during viral shedding (Wang et al. 2017b). Human HEV infections are generally asymptomatic and self-limiting (Behrendt et al. 2014; Takakusagi et al. 2023). However, severe hepatitis in patients with concurrent hepatic disease and chronic hepatitis in immunocompromised patients may occur (Behrendt et al. 2014; Takakusagi et al. 2023). The mortality rate in humans is approximately 0.2–4.0% (Teshale et al. 2010; Takakusagi et al. 2023), however, rates of greater than 20% have been reported in pregnant women (Singh et al. 2003; Takakusagi et al. 2023). Zoonotic HEV-3 is circulating in Poland among human and pig populations (Grabarczyk et al. 2018; Bigoraj et al. 2021). RNA of HEV-3 was found in 2.4% (6/246) of samples of pig’s blood and liver for human consumption (Bigoraj et al. 2021). According to Grabarczyk et al. (2018), 43.5% (1340/3079) of human blood donors were positive for anti-HEV immunoglobulin G (IgG) antibodies. RNA of HEV-3 was identified in three individuals (Grabarczyk et al. 2018). The common prevalence rate of anti-HEV antibodies among cats and dogs in Europe and evidence of zoonotic HEV-3 circulation in the human and pig populations in Poland point to the need to evaluate the presence of anti-HEV-3 antibodies among pet animals in this country. Pets, including dogs, cats, and rabbits, are in close contact with humans and might be accidental hosts for HEV-3. Dogs and cats are carnivores and often eat raw porcine or cattle products intended for human consumption, so they can acquire HEV-3 infection. Rabbits are also likely to become infected by eating contaminated, unwashed fruit or vegetables. These animal species may indicate HEV-3 contamination of food products. Serological studies so far indicate that HEV (mainly HEV-3) circulates among cats, dogs, and rabbit populations worldwide (Di Bartolo et al. 2016; Dahnert et al. 2018; Lyoo et al. 2019; Li et al. 2020; Capozza et al. 2021; Caballero-Gomez et al. 2022; Cagigran et al. 2022). This study is aimed to investigate the evidence of infection with HEV-3 using a serological assay in domestic cats, dogs and rabbits in Poland.
Materials and methods
Samples
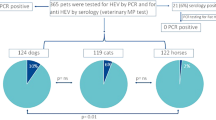
A total of 243 serum samples collected between September 2020 and January 2022 in three veterinary practices located in various parts of Poland (Poznan 52°24′24″N 16°55′47″E (wielkopolskie voivodeship); Przemysl 49°47′05″N 22°46′02″E (podkarpackie voivodeship); Lublin 22°34′E 51°15′N (lubelskie voivodeship); (Fig. 1) were selected for this study. Samples from rabbits were collected in one practice (n = 71). Rabbit sera came from animals from Poznan and their surroundings. Sera were stored at − 70 °C until analyses. Serum samples from 90 cats, 82 dogs and 71 rabbits were used in the study. The following information has been available for each sample: species, gender, age at sampling, health status and location. Information about gender, age and health status was collected during anamnesis and clinical examination performed by veterinarians during the visits to veterinary clinics. The main reasons for the visits to the veterinary clinic other than gastrointestinal disorders were respiratory symptoms, chronic diseases and routine checks during vaccinations or before performing surgical procedures. The detailed information about the structure of a sampled population is presented in Table 1 (cats and dogs) and Table 2 (rabbits).
Detection of anti-HEV-3 IgG antibodies
Serum samples were tested for the presence of anti-HEV-3 IgG antibodies using the ID Screen® Hepatitis E Indirect Multi-species ELISA, (IDvet, Grabels, France) according to the manufacturer’s instructions. This is a duplicate-well test, where even-numbered wells are coated with a recombinant antigen from the capsid of HEV-3, and odd-numbered wells are uncoated. Briefly, 10 µL of sera was diluted with 90 µL ELISA dilution buffer in the test plate and incubated for 45 min at room temperature (21°C +/- 5°C). The plate was washed three times, and the conjugate was added to each well. Then, the plate was incubated at the same temperature for 30 min and washed three times. Next, 100 µL of 3,3’,5,5’-tetramethylbenzidine (TMB) substrate solution was added to each well for 15 min, followed by 100 µL of stop solution. The optical density (OD) was measured at 450 nm in the Infinite® 200 PRO microplate reader (TECAN) immediately after stopping the reaction. The cut-off values (S/P%) that allowed the sample to be considered as positive, doubtful, or negative were calculated using the following formula: (optical density (OD) value of tested sample/OD value obtained for positive control) × 100%. The serum was considered positive when its cut-off value exceeded the borderline seropositivity of 70%. A doubtful result ranged from 60 to 70% and for negative sera, OD values were below 60%. The correct performance of the indirect ELISA was monitored using the results obtained for positive and negative control sera supplied with the assay.
Statistical analysis
The analyses were performed using RStudio (version 4.1.2), except for prevalence (with 95% confidence intervals (CI)), which was available as an online program (https://epitools.ausvet.com.au/ciproportion). CI for prevalence was calculated with the Wilson score method. Pearson’s chi-square (χ2) tests were used to analyse the data in age groups, gender and symptoms in rabbits.
Results
All of the serum samples from dogs and cats were negative for anti-HEV-3 IgG antibodies. The seroprevalence among rabbits was 2.82% (2/71; 95% CI: 0.78–9.70). No significant gender difference in seroprevalence between females and males and no significant differences regarding age group or symptoms exhibited by rabbits were found (Table 2).
Discussion
Cats, dogs and rabbits are animal species commonly kept as pets worldwide. Moreover, these species are often kept at home together and usually have close contact in the same household. Several studies suggest that pets are frequently exposed to HEV-3 or HEV-4 and thus are potentially infectious for humans in close contact (Lyoo et al. 2019; Mrzljak et al. 2021). The sporadic acute hepatitis E of a 47-year-old man whose pet cat was positive for the antibody to hepatitis E virus was reported in 2003 and it was the first suggestion that HEV has a zoonotic potential (Kuno et al. 2003). Veterinarians, who have close contact with cats, dogs and rabbits, are supposed to have higher rates of anti-HEV antibody positivity compared to the general population (Lyoo et al. 2019; Mrzljak et al. 2021). Moreover, having a pet at home was identified as a risk factor for HEV seropositivity in several studies (Hriskova et al. 2021).
HEV was identified for the first time in a pet house rabbit (an adult 7 years old female) in 2015. Rabbit liver tested positive by real-time PCR (Caruso et al. 2015). Rabbits may have become infected with HEV-3 in several ways: consumption of contaminated vegetables and fruits or contaminated water. The hay provided to pet owners by an organic farm could also be considered a primary source of infection (Caruso et al. 2015). Laboratory rabbits infected with HEV-3 or HEV-4 could present similar signs of acute and chronic hepatitis E in humans (Wang et al. 2018). After experimental infection of specific-pathogen-free rabbits with HEV-3, Wang et al. (2017b) observed viremia and fecal shedding. Fecal shedding lasts up to 40 weeks. The infection led to kidney and liver injury (Wang et al. 2017b). It cannot be excluded that humans may become infected with HEV-3 or HEV-4 when cleaning litter boxes or through contact with infected feces from rabbits. There are several cases of humans infected with a strain of HEV that is genetically similar to those found in rabbits (Kaiser et al. 2018; Sooryanarain and Meng 2019).
The available data on HEV seroprevalence among pet rabbits is scarce. The seroprevalence among farm and pet rabbits in Italy reached 3.40% (7/206) and 6.56% (8/122) respectively (Di Bartolo et al. 2016). However, in a recent study conducted in Italy, 328 hares and 59 farmed rabbits were screened for anti-HEV antibodies and none of them were positive (De Sabato et al. 2023). In Poland, antibodies against HEV were detected in 6% (29/482) of breeding rabbits (Bigoraj et al. 2020). The seroprevalence in our study reached 2.82% (95% CI: 0.78–9.70). Presumably, the older age of animals could increase the likelihood of exposure to HEV-3 and seroconversion; however, statistical analysis of our results showed that the correlation between the age of rabbits and the presence of anti-HEV-3 IgG antibodies was not significant (Table 2). Animals examined in our study were assigned to five age groups. Unfortunately, the age of 5 rabbits was not known. Two seropositive rabbits were found in two age groups: one in the age group below 12 months and the second in the age group of 4–7 years. Seventy-one rabbit sera were tested, which is relatively a small size sample. These samples were also collected from one region and our results may not fully represent the real prevalence in Poland. However, our results indicate that HEV-3 circulates among pet rabbits in Poland and these animals may play a potential role in the epidemiology of HEV-3 infection. To the author’s best knowledge, this is the first study concerning HEV seroprevalence among pet rabbits in Poland.
According to serological studies conducted in Europe, the prevalence of anti-HEV antibodies in stray and pet Spanish cats was 2.8% (4/144) (Caballero-Gomez et al. 2022). The seroprevalence in pet cats in Europe was 32.3% (21/65) in Germany (Dahnert et al. 2018), 14.9% (7/47) in the Netherlands (Li et al. 2020), 3.1% (10/324) in Italy (Capozza et al. 2021), and 5.4% (5/91) in Türkiye (Cagigran et al. 2022). The seroprevalence in stray and pet Spanish dogs was 9.9% (15/152) (Caballero-Gomez et al. 2022). In pet dogs, the seroprevalence reached 18.52% (30/162) in the Netherlands (Li et al. 2020) and 56.6% (47 of 83) in Germany (Dahnert et al. 2018). No anti-HEV-3 antibodies were detected in either cats or dogs in our study. Our results suggest that HEV-3 infections in these populations are rare, although our results may not reflect real prevalence throughout the country.
This study has some limitations that should be kept in mind. We have searched for evidence of HEV-3 infection via one serological method (indirect ELISA) and did not use species-specific HEV-3 immunoassays. Cross-reactivity to the related hepevirus cannot be ruled out despite the high specificity of the ELISA used in the present study. However, serological assays are preferable to a broad screening of potential animal reservoirs of HEV. HEV-specific antibodies are detectable over a much longer period of time than viral RNA, hence the detection window is wide (Panda et al. 2007). Further studies testing serum, including western blot, and molecular analyses of the sera and fecal samples should be carried out to increase the sensitivity of HEV-3 detection in these species. Additionally, a full understanding of the epidemiology of HEV-3 infection in humans and other animals requires a full-length HEV-3 genome isolated from pets.
We have not found evidence of exposure to HEV-3 in dogs and cats. The results of our study suggest that rabbits can become infected and develop a humoral response in response to infection with HEV-3. These results are beneficial to researchers, health workers, and veterinary practitioners. Considering the routes of infection in rabbits, good hygiene practices, washing fruits and vegetables, and giving hay and water from a verified source are the primary ways to prevent HEV-3 infection. The knowledge of good hygiene practices for the pet owner is crucial in the prevention of zoonotic hepatitis E. Even if the true potential of HEV-3 transmission from rabbits to humans is not known and further issues remain to be clarified, our data proved that pet rabbits should be considered in the epidemiological scenario of HEV-3 infection and this issue requires more in-depth research.
Data availability
The data that support the findings of this study are not openly available but are available from the corresponding author upon reasonable request.
References
Behrendt P, Steinmann E, Manns MP, Wedemeyer H (2014) The impact of hepatitis E in the liver transplant setting. J Hepatol 61:1418–1429. https://doi.org/10.1016/j.jhep.2014.08.047
Bigoraj E, Kozyra I, Kwit E, Rzeżutka A (2020) Detection of hepatitis E virus (rabbit genotype) in farmed rabbits entering the food chain. Int J Food Microbiol 319:108507. https://doi.org/10.1016/j.ijfoodmicro.2020.108507
Bigoraj E, Paszkiewicz W, Rzeżutka A (2021) Porcine blood and liver as sporadic sources of Hepatitis E Virus (HEV) in the production chain of Offal-Derived Foodstuffs in Poland. Food Environ Virol 13(3):347–356. https://doi.org/10.1007/s12560-021-09475-z
Brassard J, Gagné MJ, Généreux M, Côté C (2012) Detection of human food-borne and zoonotic viruses on irrigated, field-grown strawberries. Appl Environ Microbiol 78(10):3763–3766. https://doi.org/10.1128/AEM.00251-12
Caballero-Gómez J, Rivero-Juarez A, Jurado-Tarifa E, Jiménez-Martín D, Jiménez-Ruiz E, Castro-Scholten S, Ulrich RG, López-López P, Rivero A, García-Bocanegra I (2022) Serological and molecular survey of hepatitis E virus in cats and dogs in Spain. Transbound Emerg Dis 69(2):240–248. https://doi.org/10.1111/tbed.14437
Cagirgan AA, Yildirim Y, Okulmus C (2022) The first evidence of zoonotic hepatitis E virus (HEV) exposure in domestic cats in Türkiye. Comp Immunol Microbiol Infect Dis 86:101820. https://doi.org/10.1016/j.cimid.2022.101820
Capozza P, Martella V, Lanave G, Beikpour F, Di Profio F, Palombieri A, Sarchese V, Marsilio F, La Rosa G, Suffredini E, Camero M, Buonavoglia C, Di Martino B (2021) A surveillance study of hepatitis E virus infection in household cats. Res Vet Sci 137:40–43. https://doi.org/10.1016/j.rvsc.2021.04.022
Caruso C, Modesto P, Prato R, Scaglione FE, De Marco L, Bollo E, Acutis PL, Masoero L, Peletto S (2015) Hepatitis E Virus: first description in a Pet House rabbit. A New Transmission Route for Human? Transbound Emerg Dis 62(3):229–232. https://doi.org/10.1111/tbed.12348
Dahnert L, Conraths FJ, Reimer N, Groschup MH, Eiden M (2018) Molecular and serological surveillance of hepatitis E virus in wild and domestic carnivores in Brandenburg, Germany. Transbound Emerg Dis 65(5):1377–1380. https://doi.org/10.1111/tbed.12877
Dalton HR, Kamar N, Izopet J (2014) Hepatitis E in developed countries: current status and future perspectives. Future Microbiol 9(12):1361–1372. https://doi.org/10.2217/fmb.14.89
De Sabato L, Ianiro G, Filipello V, Arnaboldi S, Righi F, Ostanello F, Giammarioli M, Lavazza A, Di Bartolo I (2023) Absence of Hepatitis E Virus (HEV) in italian lagomorph species sampled between 2019 and 2021. Anim (Basel) 13(3):545. https://doi.org/10.3390/ani13030545
Demirci M, Yiğin A, Ünlü Ö, Kılıç Altun S (2019) Detection of HEV RNA amounts and genotypes in raw milk obtained from different animals. Mikrobiyol Bul 53(1):43–52. https://doi.org/10.5578/mb.67468
Di Bartolo I, De Sabato L, Marata A, Martinelli N, Magistrali CF, Monini M, Ponterio E, Ostanello F, Ruggeri FM (2016) Serological survey of hepatitis E virus infection in farmed and pet rabbits in Italy. Arch Virol 161(5):1343–1346. https://doi.org/10.1007/s00705-016-2778-y
Go HJ, Park BJ, Ahn HS, Lyoo EL, Kim DH, Lee JB, Park SY, Song CS, Lee SW, Choi IS (2019) Identification of Hepatitis E virus in bovine and porcine raw livers. J Microbiol Biotechnol 29:2022–2025. https://doi.org/10.4014/jmb.1910.10059
Grabarczyk P, Sulkowska E, Gdowska J, Kopacz A, Liszewski G, Kubicka-Russel D, Baylis SA, Corman VM, Noceń E, Piotrowski D, Antoniewicz-Papis J, Łętowska M (2018) Molecular and serological infection marker screening in blood donors indicates high endemicity of hepatitis E virus in Poland. Transfusion 58(5):1245–1253. https://doi.org/10.1111/trf.14531
Hriskova K, Marosevic D, Belting A, Wenzel JJ, Carl A, Katz K (2021) Epidemiology of Hepatitis E in 2017 in Bavaria, Germany. Food Environ Virol 13(3):337–346. https://doi.org/10.1007/s12560-021-09474-0
Huang F, Li Y, Yu W, Jing S, Wang J, Long F, He Z, Yang C, Bi Y, Cao W, Liu C, Hua X, Pan Q (2016) Excretion of infectious hepatitis E virus into milk in cows imposes high risks of zoonosis. Hepatology 64(2):350–359. https://doi.org/10.1002/hep.28668
Kaiser M, Delaune D, Chazouillères O, Blümel J, Roque-Afonso AM, Baylis SA (2018) A World Health Organization Human Hepatitis E virus reference strain related to similar strains isolated from rabbits. Genome Announc 6(16):e00292–e00218. https://doi.org/10.1128/genomeA.00292-18
Kuno A, Ido K, Isoda N, Satoh Y, Ono K, Satoh S, Inamori H, Sugano K, Kanai N, Nishizawa T, Okamoto H (2003) Sporadic acute hepatitis E of a 47-year-old man whose pet cat was positive for antibody to hepatitis E virus. Hepatol Res 26(3):237–242. https://doi.org/10.1016/s1386-6346(03)00197-9
Li S, Liu M, Cong J, Zhou Y, Miao Z (2017) Detection and characterization of Hepatitis E Virus in Goats at Slaughterhouse in Tai’an Region, China. Biomed Res Int 2017:3723650. https://doi.org/10.1155/2017/3723650
Li Y, Qu C, Spee B, Zhang R, Penning LC, de Man RA, Peppelenbosch MP, Fieten H, Pan Q (2020) Hepatitis E virus seroprevalence in pets in the Netherlands and the permissiveness of canine liver cells to the infection. Ir Vet J 73:6. https://doi.org/10.1186/s13620-020-00158-yn
Lyoo KS, Yang SJ, Na W, Song D (2019) Detection of antibodies against hepatitis E virus in pet veterinarians and pet dogs in South Korea. Ir Vet J 72:8. https://doi.org/10.1186/s13620-019-0146-4
Mrzljak A, Balen I, Barbic L, Ilic M, Vilibic-Cavlek T (2021) Hepatitis E virus in professionally exposed: a reason for concern? World J Hepatol 13(7):723–730. https://doi.org/10.4254/wjh.v13.i7.723
Panda SK, Thakral D, Rehman S (2007) Hepatitis E virus. Rev Med Virol 17(3):151–180. https://doi.org/10.1002/rmv.522
Pirani S, Pierini I, Manuali E, Bazzucchi M, De Mia GM, Giammarioli M (2023) Paslahepevirus balayani (Hepatitis E Virus) in italian Nonungulate Wildlife: molecular detection and characterization of an isolate from a Crested Porcupine (Hystrix cristata). J Wildl Dis 59(3):000–000. https://doi.org/10.7589/JWD-D-22-00087
Purdy MA, Drexler JF, Meng XJ, Norder H, Okamoto H, Van der Poel WHM, Reuter G, de Souza WM, Ulrich RG, Smith DB (2022) ICTV Virus Taxonomy Profile: Hepeviridae 2022. J Gen Virol 103:001778. https://doi.org/10.1099/jgv.0.001778
Singh S, Mohanty A, Joshi YK, Deka D, Mohanty S, Panda SK (2003) Mother-to-child transmission of hepatitis E virus infection. Indian J Pediatr 70:37–39. https://doi.org/10.1007/BF02722743
Sooryanarain H, Meng XJ (2019) Hepatitis E virus: reasons for emergence in humans. Curr Opin Virol 34:10–17. https://doi.org/10.1016/j.coviro.2018.11.006
Takakusagi S, Kakizaki S, Takagi H (2023) The diagnosis, pathophysiology, and treatment of chronic Hepatitis E virus Infection-A Condition Affecting Immunocompromised Patients. Microorganisms 11(5):1303. https://doi.org/10.3390/microorganisms11051303
Teshale EH, Hu DJ, Holmberg SD (2010) The two faces of Hepatitis E Virus. Clin Infect Dis 51(3):328–334. https://doi.org/10.1086/653943
Wang L, Xia J, Wang L, Wang Y (2017b) Experimental infection of rabbits with genotype 3 hepatitis E virus produced both chronicity and kidney injury. Gut 66(3):561–562. https://doi.org/10.1136/gutjnl-2016-312023
Wang L, Liu L, Wang L (2018) An overview: rabbit hepatitis E virus (HEV) and rabbit providing an animal model for HEV study. Rev Med Virol 28(1). https://doi.org/10.1002/rmv.1961
Wu J, Si F, Jiang C, Li T, Jin M (2015) Molecular detection of hepatitis E virus in sheep from southern Xinjiang, China. Virus Genes 50(3):410–417. https://doi.org/10.1007/s11262-015-1194-9
Acknowledgements
The scientific activity was supported by statutory funding 506.514.05.00 of the Department of Preclinical Sciences and Infectious Diseases, Faculty of Veterinary Medicine and Animal Science, Poznan University of Life Sciences, Poland and grant 506.514.05.00 of the Young Researcher Program of the Faculty of Veterinary Medicine and Animal Science, Poznan University of Life Sciences, financed by the Polish Ministry of Science and Higher Education.
Author information
Authors and Affiliations
Contributions
H. T. P. worked on the conceptualization, sampling, investigation, and methodology and prepared tables and figure. M. P. M. worked on supervision and has reviewed the manuscript. J. J. R. and J. W. worked on sampling. The first draft of the manuscript was written by H. T. P. and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Consent to participate
Informed consent was obtained from the owners of the animals included in the study.
Consent for publication
Not applicable.
Statement of Animal Ethics
The authors declare no animal ethics conflict. According to the Act on the Protection of Animals Used for Scientific or Educational Purposes in Poland adopted on 15th January 2015 and according to earlier regulations (Act on the Protection of Animals Used for Scientific or Educational Purposes in Poland adopted on 21th January 2005) the study described in this manuscript did not require the permission of the Local Ethical Commission for Investigations on Animals. The reported study is in compliance with the ARRIVE guidelines.
Conflict of interest
The authors declare no conflict of interest.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Turlewicz-Podbielska, H., Ruszkowski, J.J., Wojciechowski, J. et al. No evidence of hepatitis E virus (HEV) infection among pet cats and dogs, and low seroprevalence of hepatitis E virus among pet rabbits in Poland. Vet Res Commun 48, 597–602 (2024). https://doi.org/10.1007/s11259-023-10223-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-023-10223-w