Abstract
Background
Concurrent training (CT) is a common method used in health-oriented training programs. However, its design needs to be explored in order to inform practitioners about the most effective methods to improve different health-related markers (e.g., maximal fat oxidation [MFO]). Thus, this study aims to determine the order effect of an 8 week CT on the MFO of recreationally trained young adults.
Methods
Twenty participants were allocated in two different groups to perform the CT differing only in the exercise sequence. The endurance training (ET) consisted of 4 to 6 repetitions of 30 s all-out running sprints with 4 min of active recovery. The resistance training (RT) consisted of 4 to 6 sets at 60 to 80% of the one-repetition maximum with 5 to 1 repetition in reserve of back squat and bench press exercises. 15 min of rest were established between exercise modes. Previous and after the CT program, participants performed a graded exercise test where MFO was determined.
Results
There was a significant time effect on MFO (p = 0.044). A moderate increase was observed in both ET + RT (Mean change: 0.11 [− 0.02 to 0.25] g/min; Effect size: 0.61 [− 0.12 to 1.35]) and RT + ET (Mean change: 0.07 [− 0.01 to 0.16] g/min; Effect size: 0.62 [− 0.12 to 1.36]) groups. No significant interaction was observed (p = 0.658).
Conclusions
The 8 week CT program improved the muscle oxidative capacity of recreationally trained young adults regardless of the exercise sequence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The capacity to oxidize fat (i.e., fat oxidation [FO]) during exercise has been a used in clinical settings as an impairment in its maximum rate (i.e., maximal fat oxidation [MFO]) is associated with the development of metabolic syndrome [1]. FO is regulated through different steps. It begins with the adipose tissue lipolysis, followed by the transport of free fatty acids to muscle and across the cell membrane, and ends through the beta-oxidation process carried out by the enzymes that regulate the Krebs cycle and the electron transport chain [2]. Thus, health training programs should consider those exercises that maximize the protein expression of those elements involved in FO.
Traditionally, moderate-intensity continuous training (MICT) has been used for improving FO. However, it has been established that high-intensity interval training (HIIT) or sprint interval training (SIT) can be as effective as MICT in almost half of the training time [3]. The mechanism by which SIT improves FO relies on the activation of the signaling pathways of mitochondrial biogenesis [4, 5]. The glycolytic metabolism required during the high-intensity bouts involves the accumulation of free radicals, ions and metabolites (i.e., lactate, creatine, AMP, H+), resulting in the phosphorylation of the AMP-activated protein kinase (AMPK) and, thus, the expression of the peroxisome proliferator-activated receptor coactivator-1α (PGC-1α), the main transcriptional cofactor that mediates mitochondrial biogenesis [4]. Of note, it seems that the AMPK activation is also bound to the on–off pattern of interval training [6].
Albeit in a lesser degree, it has been shown that resistance training (RT) can also have an impact on FO through the phosphorylation of the AMPK [7, 8]. As it occurs during endurance training (ET), the AMPK is activated through markers of low cellular energy and, thus, the load and the volume established on RT can modulate the signaling of the biogenesis pathways. Porter et al. [7] observed an improvement in mitochondrial respiration after 12 weeks of whole-body RT conducted 3 times per week at an intensity of 60–80% of the one-repetition maximum (1RM), with multiple sets performed to failure. Thus, it seems that in order to stimulate mitochondrial biogenesis, RT should be focused on inducing a high metabolic stress [9].
A high metabolic stress could be also induced by combining ET and RT within the same training program (i.e., concurrent training [CT]). However, the synergistic effect of both exercise modes is determined by the exercise principles employed. The exercise sequence has been postulated as one of the potential factors that mediates mitochondrial biogenesis. Wang et al. [10] observed an enhanced expression of marker genes related to mitochondrial biogenesis (i.e., PGC-1α) and substrate regulation (i.e., PDK4) when ET (i.e., one hour cycling at 65% of the maximum oxygen uptake [VO2max]) was followed by RT (i.e., six sets of leg press at 70–80% 1RM up to 15 repetitions) compared to ET alone in recreationally active adults. Likewise, Coffey et al. [11] observed that PGC-1α raised after CT (ET: 30 min of cycling at 70% of VO2max; RT: eight sets of leg extension at 80% 1RM), being slightly higher when ET was followed by RT in regular training adults.
The acute molecular signaling is the first adaptive response to training. However, the exercise-training-induced adaptions are the result of the summed effects of repeated training sessions that require to be analyzed in the long term in order to determine the efficacy of a training program on improving the muscle oxidative capacity. Likewise, the adaptive response to CT requires the analysis of further combinations of exercise modes, intensities, and volumes, as well as the population recruited [12]. Therefore, this study aims to determine the order effect of an 8 week CT program composed of SIT and lower- and upper-body resistance exercises on the MFO of recreationally trained young adults.
Materials and methods
Experimental design
A longitudinal pre-post design was used to compare the effect of altering the exercise sequence of a CT program (i.e., ET followed by RT [ET + RT] vs. RT followed by ET [RT + ET]) on MFO. Before starting the training program, participants attended to the pre-tests where body composition, back squat and bench press 1RM, MFO, and VO2max were determined. Standardized groups were created considering VO2max. Then, participants began with an 8 week CT program composed of three sessions per week of 60 to 90 min duration with all-out running SIT, back squat, and bench press resistance exercises. After checking their compliance with the training program, participants followed the post-test sessions. All testing and training sessions were conducted in a research center with at least 48–72 h of rest, at a consistent time of day for each subject (± 1 h) and under similar environmental conditions (~ 20 °C and ~ 60% humidity).
Participants
A group of 20 recreationally trained healthy young adults were initially enrolled. All participants were required to meet the following inclusion criteria: (i) being between 18 and 30 years old, (ii) free from any injuries within the six months before data collection, and (iii) being physically active according to the guidelines of the American College of Sports Medicine (ACSM) [13]. After assessing for eligibility and determining the baseline level of fitness of participants, they were allocated in two groups differing only in the exercise sequence: ET + RT (n = 10) or RT + ET (n = 10) to conduct an 8 week CT program. However, 3 participants dropped out from the training intervention for reasons unrelated to the study and 3 participants were discarded due to the lack of compliance with training. Thus, 7 participants (3 males and 4 females) in the ET + RT group (age: 21.0 [2.0] years; height: 170 [5.9] cm; body mass: 64.1 [8.0] kg; fat mass: 10.9 [3.2] kg; fat-free mass [FFM]: 53.2 [9.9] kg; back squat one-repetition maximum [1-RM]: 1.40 ± 0.31 kg/kgBM; bench press 1-RM: 0.90 ± 0.30 kg/kgBM; training experience: 5 ≥ years; training frequency: 2 ≥ sessions per week) and 7 participants (5 males and 2 females) in the RT + ET group (age: 21 [1.5] years; height: 178 [11] cm; body mass: 77.5 [10.9] kg; fat mass: 15.5 [6.9] kg; FFM: 62.2 [10.7] kg; back squat 1-RM: 1.50 ± 0.25 kg/kgBM; bench press 1-RM: 0.75 ± 0.20 kg/kgBM; training experience: 5 ≥ years; training frequency: 2 ≥ sessions per week) completed the entire intervention process (Table 1). A post hoc analysis of the achieved power for this sample size was conducted using G*power (version 3.1), given α = 0.05, ES = 0.60, total sample size = 14, statistical tests = means: difference between dependent means. This analysis revealed a power of 0.55 for the time effect. All participants were informed about the research purpose and procedures of the study prior to signing a written informed consent form. The study protocol adhered to the tenets of the declaration of Helsinki and was approved by the institutional review board (ref. 2546/CEIH).
Body composition
The anthropometric characteristics of the participants (body mass [kg], fat mass [kg] and FFM [kg]) were obtained using the bioimpedanciometer Inbody 230 (Inbody, Seoul, Korea), which has been previously validated by a dual-energy X-ray system [14]. Participants were encouraged to follow the recommendations of the American Society of Exercise Physiologists before the test [15].
MFO, fatmax, VO2max
The graded exercise test (GXT) was conducted on a treadmill (WOODWAY Pro XL, Woodway, Inc., Waukesha, WI, USA). Participants warmed up for 5 minutes at a self-selected speed with the premise of not exceeding an intensity at which they could not talk. Then, participants were fitted with the validated portable metabolic analyzer (PNOE, ENDO Medical, Palo Alto, CA) [16], which was previously calibrated according to the manufacturer’s instructions. The GXT was customized to determine MFO and VO2max in the same session and according to the experience and level of the participants. From a starting velocity of 5 km/h, the velocity was increased by 1 km/h every 3 min until the respiratory gas exchange rate reached 1.0. After that, the velocity remained constant, and the incline was increased by 2% every minute until volitional exhaustion.
The breath-by-breath data of each record were exported into an Excel spreadsheet. To exclude errant breaths, values outside the 95% confidence interval of the local mean were removed. Then, breath-by-breath data were linearly interpolated to give 1 s values. Average values of oxygen uptake (O2; L/min) and dioxide production (CO2; L/min) for the last 60 s in each 3 min step were used to determine FO (gr/min) through the Frayn equation [17] with the assumption that the urinary nitrogen excretion rate (n) was negligible:
FO = 1.67*O2 − 1.67*CO2 − 1.92*n.
For each subject, the calculated values for FO in each step were depicted graphically as a function of exercise intensity (%VO2max) and a 2nd polynomial curve with an intersection in (0,0) was constructed to determine MFO (g/min) and the intensity that elicits the MFO (Fatmax) (% VO2max). VO2max was determined as the highest 30 s rolling mean value.
CT program
Both groups completed an 8 week CT program composed of three sessions of 60 to 90 min per week with running SIT, as well as back squat and bench press resistance exercises, differing only in their exercise sequences (ET + RT vs. RT + ET) (Table 2). Relative loads were applied according to the 1RM pre-test (Table 1). Back squat and bench press 1RM were estimated from the individual and exercise-specific load-velocity profile [18]. The training program progressed every 2 weeks in volume and load for running SIT (i.e., 4–6 intervals) and resistance exercises (i.e., 60–80% 1RM), respectively. Since sessions were created with a time-efficient purpose, only 15 min of rest was established between exercise modes. To minimize the potential effect of fatigue on the subsequent exercise to be performed, the ET + RT group conducted the bench press exercise first after the running SIT, and the RT + ET group began with the back squat before the bench press exercise and the running SIT. An instructor assisted and encouraged the participants in each session of the training program. Sessions were conducted with at least 48 h of recovery in between. Participants were encouraged to follow their habitual dietary patterns over the entire training program.
Statistical analysis
Data are presented as mean (SD) or (95% confidence interval). The normal distribution and homogeneity of variance were confirmed through the Shapiro–Wilk test and Levene’s tests, respectively. The baseline characteristics of each training group were compared through an independent sample student’s t test. A one-way mixed model (group x time) analysis of variance (ANOVA) was used to determine the effect of altering the exercise sequence (i.e., ET + RT or RT + ET) on the MFO, MFO relativized to FFM (MFOrel), Fatmax, and VO2max. The normality of the residuals was confirmed through the inspection of Q–Q plots. The homogeneity of variances was also confirmed through the Levene test. Pairwise effect size (ES) was determined as (mean change/SD change) [19], and interpreted as follows: trivial (< 0.2), small (0.2–0.59), moderate (0.60–1.19), large (1.20–2.0), and extremely large (> 2.0) [20]. Statistical analyses were performed using the software package SPSS (IBM SPSS, version 25.0; IBM, Chicago, IL). Alpha was set at 0.05.
Results
Training groups presented similar baseline characteristics regarding MFO (ET + RT: 0.60 [0.15] g/min; RT + ET: 0.68 [0.12] g/min; p = 0.579), MFOrel (ET + RT: 0.011 [0.003] g/min/kg FFM; RT + SIT: 0.011 [0.002] g/min/kg FFM; p = 0.793), Fatmax (ET + RT: 53.9 [7.5] % VO2max; RT + ET: 55.7 (5.5) % VO2max; p = 0.620), and VO2max (ET + RT: 52.1 [5.1] ml/kg/min; RT + ET: 49.6 [5.0] ml/kg/min; p = 0.367) (Table 1).
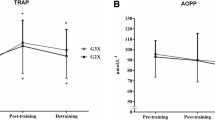
There was a significant time effect on MFO (F(1,12) = 5.07; p = 0.044) (Fig. 1). A moderate increase was observed in both ET + RT (p = 0.08; mean change: 0.11 [− 0.02 to 0.25] g/min; ES: 0.61 [− 0.12 to 1.35]) and RT + ET (p = 0.227; mean change: 0.07 [[− 0.01 to 0.16] g/min; ES: 0.62 [− 0.12 to 1.36]) groups. MFOrel did not reach a significant time effect (F(1,12) = 3.79; p = 0.075) although a small increase was observed in both ET + RT (p = 0.107; mean change: 0.002 [− 0.001 to 0.004] g/min/kg FFM; ES: 0.54 [ − 0.20 to 1.28]) and RT + ET (p = 0.333; mean change: 0.001 [0.000 to 0.002] g/min/kg FFM; ES: 0.53 [− 0.21 to 1.27]) groups. A non-significant time effect was also obtained on Fatmax (F(1,12) = 2.38; p = 0.148), although a small increase was observed in both ET + RT (p = 0.145; mean change: 2.86 [− 1.41 to 7.12] % of VO2max; ES: 0.50 [− 0.24 to 1.24]) and RT + ET (p = 0.544; mean change: 1.14 [− 1.61 to 3.89] % of VO2max; ES: 0.31 [− 0.43 to 1.05]) groups. There was a significant time effect on VO2max (F(1,12) = 11.04; p = 0.006). A large increase was observed in ET + RT group (p = 0.012; mean change: 5.69 [2.46 to 8.92] ml/kg/min; ES: 1.30 [0.56 to 2.04]) and a small increase in RT + ET group (p = 0.107; mean change: 3.35 [− 0.89 to 7.59] ml/kg/min; ES: 0.58 [− 0.15 to 1.32]). Non-significant interactions were observed in the aforementioned variables (F(1,12) < 0.740; p > 0.406).
Discussion
This study aimed to determine the order effect of an 8 week CT program composed of SIT and low and upper-body resistance exercises on the MFO of recreationally trained young adults. The results revealed that the CT program improved the MFO in both training groups in a similar manner, and thus, no interferences were observed regarding the exercise sequences on improving the muscle oxidative capacity.
Both training groups improved MFO in a similar magnitude (ET + RT: 0.11 [− 0.02 to 0.25] g/min; RT + ET: 0.07 [− 0.01 to 0.16] g/min). While training-induced increases of 1-MET have been considered of clinical relevance [21], there are no consolidated thresholds for interpreting MFO changes. In this regard, Atakan et al. [3] have recently proposed that any increases greater than 0.07 g/min would be meaningful, being such changes observed in HIIT and SIT interventions of 4 to 12 weeks (0.05 to 0.13 g/min). These MFO changes are derived from the molecular signaling that the high-intensity exercise induces, activating the AMPK and, thus, the expression of PGC1-α, the main mediator of mitochondrial biogenesis [4]. Using the same SIT protocol of 30 s all-out, Gibala et al. [22] observed an acute twofold increase in PGC1-α mRNA above resting values. However, the AMPK phosphorylation can be also achieved through RT as long as it involves an overload stimulus [23]. Therefore, several studies support that the molecular signaling of mitochondrial biogenesis is enhanced with CT compared to each exercise mode alone [10, 24,25,26]. This is of particular interest for practitioners since it has been observed that CT (RT: 4 × 8 leg extension at 70% 1RM; ET: 20 min cycling at 55% of peak aerobic power output) can elicit a similar response to the double ET time (40 min cycling at 55% of peak aerobic power output) [25], which could give variety to the sessions of a training program with the same aim.
Given the effectiveness of CT compared to isolated exercises, it is necessary to determine if such benefits are due to the effect of an additional exercise or due to a synergistic effect. In line with the results of the present study, Coffey et al. [11] determined the acute molecular response to a CT (RT: 8 × 5 leg extension at 80% 1RM; ET: 30 min cycling at 70% of VO2max), observing a significant time effect for PGC-1α, but not an interaction between both sequences. However, when the CT was composed of the same RT and a cycling SIT (10 × 6 s), there was a moderate exercise order effect in favor of RT followed by SIT [27].
The potential benefits derived from CT or a particular exercise sequence appear to reside in the metabolic environment that the previous exercise leaves in the other. It has been shown that a low carbohydrate availability augments the early signaling responses that mediate mitochondrial biogenesis [28, 29]. This enhanced response is well-established in ET [29]. However, this has been also observed in RT [30], which in conjunction with its glycogen depletion effects [31], reinforces its role in enhancing the muscle oxidative capacity and its synergistic effect with ET.
Conclusion
The results obtained revealed that the 8 week CT program composed of SIT and lower- and upper-body resistance exercises improved the muscle oxidative capacity of recreationally trained young adults regardless of the exercise sequence.
Data availability
No datasets were generated or analysed during the current study.
References
Storlien L, Oakes ND, Kelley DE (2004) Metabolic flexibility. Proc Nutr Soc 63(2):363–368
Spriet LL (2002) Regulation of skeletal muscle fat oxidation during exercise in humans. Med Sci Sports Exerc 34(9):1477–1484
Atakan MM, Guzel Y, Shrestha N, Kosar SN, Grgic J, Astorino TA, Pedisic Z (2022) Effects of high-intensity interval training (HIIT) and sprint interval training (SIT) on fat oxidation during exercise: a systematic review and meta-analysis. Br J Sports Med 56(17):988–996. https://doi.org/10.1136/bjsports-2021-105181
MacInnis MJ, Gibala MJ (2017) Physiological adaptations to interval training and the role of exercise intensity. J Physiol 595(9):2915–2930
Islam H, Townsend LK, Hazell TJ (2017) Modified sprint interval training protocols.Part I physiological responses. Appl Physiol Nutr Metab 42(4):339–346
Combes A, Dekerle J, Webborn N, Watt P, Bougault V, Daussin FN (2015) Exercise-induced metabolic fluctuations influence AMPK, p38-MAPK and Ca MKII phosphorylation in human skeletal muscle. Physiol Rep 3(9):e12462
Porter C, Reidy PT, Bhattarai N, Sidossis LS, Rasmussen BB (2015) Resistance exercise training alters mitochondrial function in human skeletal muscle. Med Sci Sports Exerc 47(9):1922
Dreyer HC, Fujita S, Cadenas JG, Chinkes DL, Volpi E, Rasmussen BB (2006) Resistance exercise increases AMPK activity and reduces 4E-BP1 phosphorylation and protein synthesis in human skeletal muscle. J Physiol 576(2):613–624
Groennebaek T, Vissing K (2017) Impact of resistance training on skeletal muscle mitochondrial biogenesis, content, and function. Front physiol 8:713
Wang L, Mascher H, Psilander N, Blomstrand E, Sahlin K (2011) Resistance exercise enhances the molecular signaling of mitochondrial biogenesis induced by endurance exercise in human skeletal muscle. J Appl Physiol 111(5):1335–1344
Coffey VG, Pilegaard H, Garnham AP, O’Brien BJ, Hawley JA (2009) Consecutive bouts of diverse contractile activity alter acute responses in human skeletal muscle. J Appl Physiol 106(4):1187–1197
Panissa VLG, Greco CC, Ribeiro N, Julio UF, Tricoli V, Franchini E (2022) Concurrent training and the acute interference effect on strength: reviewing the relevant variables. Strength Cond J 44(3):46–57
American College of Sports Medicine (2013) ACSM’s guidelines for exercise testing and prescription. Lippincott Williams & Wilkins
Ling CH, de Craen AJ, Slagboom PE, Gunn DA, Stokkel MP, Westendorp RG, Maier AB (2011) Accuracy of direct segmental multi-frequency bioimpedance analysis in the assessment of total body and segmental body composition in middle-aged adult population. Clin Nutr 30(5):610–615
Heyward V 2001 ASEP methods recommendation: body composition assessment J Exerc Physiol 4:1–2.
Tsekouras YE, Tambalis KD, Sarras SE, Antoniou AK, Kokkinos P, Sidossis LS (2019) Validity and reliability of the new portable metabolic analyzer PNO E. Front Sports Act Living 1:24
Frayn KN (1983) Calculation of substrate oxidation rates in vivo from gaseous exchange. J Appl Physiol 55(2):628–634
Weakley J, Mann B, Banyard H, McLaren S, Scott T, Garcia-Ramos A (2021) Velocity-based training: From theory to application. Strength Cond J 43(2):31–49
Dankel SJ, Loenneke JP (2021) Effect sizes for paired data should use the change score variability rather than the pre-test variability. J Strength Cond Res 35(6):1773–1778
Hopkins W, Marshall S, Batterham A, Hanin J (2009) Progressive statistics for studies in sports medicine and exercise science. Med Sci Sports Exerc 41(1):3
Lee DC, Sui X, Artero EG, Lee IM, Church TS, McAuley PA, Nm BS (2011) Long-term effects of changes in cardiorespiratory fitness and body mass index on all-cause and cardiovascular disease mortality in men: the aerobics center longitudinal study. Circulation 124(23):2483–2490
Gibala MJ, McGee SL, Garnham AP, Howlett KF, Snow RJ, Hargreaves M (2009) Brief intense interval exercise activates AMPK and p38 MAPK signaling and increases the expression of PGC-1α in human skeletal muscle. J Appl Physiol 106(3):929–934
Hawley JA (2009) Molecular responses to strength and endurance training: are they incompatible? Appl Physiol Nutr Metab 34(3):355–361
Pugh JK, Faulkner SH, Jackson AP, King JA, Nimmo MA (2015) Acute molecular responses to concurrent resistance and high-intensity interval exercise in untrained skeletal muscle. Physiol Rep 3(4):e12364
Donges CE, Burd NA, Duffield R, Smith GC, West DW, Short MJ, Edge JA (2012) Concurrent resistance and aerobic exercise stimulates both myofibrillar and mitochondrial protein synthesis in sedentary middle-aged men. J Appl Physiol 112(12):1992–2001
Lundberg TR, Fernandez-Gonzalo R, Gustafsson T, Tesch PA (2012) Aerobic exercise alters skeletal muscle molecular responses to resistance exercise. Med Sci Sports Exerc 44(9):1680–1688
Coffey VG, Jemiolo B, Edge J, Garnham AP, Trappe SW, Hawley JA (2009) Effect of consecutive repeated sprint and resistance exercise bouts on acute adaptive responses in human skeletal muscle. Am J Physiol Cell Physiol 297(5):R1441–R1451
Fyfe JJ, Bishop DJ, Stepto NK (2014) Interference between concurrent resistance and endurance exercise: molecular bases and the role of individual training variables. Sports Med 44:743–762
Knuiman P, Hopman MT, Mensink M (2015) Glycogen availability and skeletal muscle adaptations with endurance and resistance exercise. Nutr Metab 12(1):1–11
Camera DM, Hawley JA, Coffey VG (2015) Resistance exercise with low glycogen increases p53 phosphorylation and PGC-1α mRNA in skeletal muscle. Eur J Appl 115:1185–1194
Robergs RA, Pearson DR, Costill DL, Fink WJ, Pascoe DD, Benedict MA, Zachweija JJ (1991) Muscle glycogenolysis during differing intensities of weight-resistance exercise. J Appl Physiol 70(4):1700–1706
Acknowledgements
The authors would like to thank all the participants involved in the intervention. This work is related to the EDUSport Research Project (ref. PID2020-115600RB-C21) which has been supported by MCIN/AEI/https://doi.org/10.13039/501100011033. In addition, Santiago A. Ruiz-Alias is supported by the Spanish Ministry of Education under grant [FPU19/00542].
Funding
Funding for open access publishing: Universidad de Granada/CBUA.
Author information
Authors and Affiliations
Contributions
All authors have contributed equally in the elaboration of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Human rights
The study protocol adhered to the tenets of the Declaration of Helsinki and was approved by the institutional review board (ref. 2546/CEIH).
Informed consent
All participants were informed about the research purpose and procedures of the study prior to signing a written informed consent form.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ruiz-Alias, S.A., Pérez-Castilla, A., Jaén-Carrillo, D. et al. Order effect of an 8-week concurrent training program on the maximal fat oxidation. Sport Sci Health (2024). https://doi.org/10.1007/s11332-024-01222-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11332-024-01222-8