Abstract
Ambient fine particulate matter of 2.5 μm or less in diameter (PM2.5) of environment contamination is deemed as a risk factor of cerebrovascular diseases. Yet there is still no explicit evidence strongly supporting that PM2.5 with per unit increment can increase the risk of hemorrhagic stroke (HS). Literatures were searched from PubMed, Cochrane, and Embase. After the systemic review of relevant studies, random effects model was used to perform meta-analysis and to evaluate the association between PM2.5 and risk of HS. Seven cohort studies were finally included, involving more than 6 million people and 37,667 endpoint events (incidence or mortality of HS). Total scores of quality assessment were 50. Pooled hazard ratio (HR) for crude HRs was 1.13 (95%CI: 1.09–1.17) (CI for confidence interval). Pooled HR of subgroup analysis for current smoking with exposure to growing PM2.5 was 1.14 (95%CI: 0.92–2.15) and for never and former smoking was 1.04 (95%CI: 0.74–1.46). Ambient PM2.5 level is significantly associated with the risk of HS, which might be a potential risk factor of HS. Smoking does not further increase the risk of HS under exposure of PM2.5.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ambient air pollution is a major and significant environmental risk to the health of people in both cities and rural areas (https://www.who.int/). According to the data from the World Health Organization, 58% of premature deaths were related to outdoor air pollution. Especially, the fatal effects of air pollution were presented as ischemic heart diseases and strokes resulted from exposure to fine particulate matter of 2.5 μm or less in diameter (PM2.5). Moreover, hemorrhagic stroke (HS) accounted for one-third of strokes. Therefore, PM2.5 might be closely related to risk of HS. Recently, Sheng Yuan (Yuan et al. 2019) and other collaborators conducted a meta-analysis and concluded that the long-term exposure to PM2.5 was an important risk factor for stroke. However, the relationship between risk of HS and ambient PM2.5 exposure has not been accurately confirmed. We supposed that some risk factors of ischemic stroke such as smoking were not associated with risk of HS. Thus, we searched recent cohort studies from open medical database to conduct a meta-analysis and to elucidate the relationship between risk of HS and increase of PM2.5.
Methods
This systemic review was performed according to the protocol published on the database of International Platform of Registered Systematic Review and Meta-analysis Protocols (INPLASY, https://inplasy.com/, registration number: INPLASY202050022, DOI number: https://doi.org/10.37766/inplasy2020.5.0022).
Literature search
Literature search was conducted from PubMed, Cochrane, and Embase databases. While making the strategy of literature search, publication time, regions, language, and human species were not restricted. The MeSH term was defined as “stroke” combined with “particulate matter.” Moreover, types of literature were not limited. Titles, keywords, abstracts, and relevant information of publication were downloaded to the software Endnote X9 (BId 12062) for article management. All literatures were independently reviewed and analyzed by two authors (Kai Zhao and Yu Guo). If there is inconsistency between them, another author (Mingfei Yang) would be consulted or the literature search strategy would be modified to reach a consensus.
The full strategy of searching literature in PubMed and Cochrane was as follows: (((((((((Stroke[MeSH Terms]) OR Strokes[Title/Abstract]) OR “Cerebrovascular Accident*”[Title/Abstract]) OR CVA*[Title/Abstract]) OR Apoplexy[Title/Abstract]) OR “Vascular Accident*, Brain”[Title/Abstract]) OR “Brain Vascular Accident*”[Title/Abstract]) OR stroke[Title/Abstract])) AND (((((((((((“Air Pollution”[MeSH Terms]) OR “Pollution, Air”[Title/Abstract]) OR “Air Quality”[Title/Abstract]) OR “Particulate Matter”[MeSH Terms]) OR “Airborne Particulate Matter”[Title/Abstract]) OR “Particulate Matter, Airborne”[Title/Abstract]) OR “Air Pollutant*, Particulate”[Title/Abstract]) OR “Particulate Air Pollutant*”[Title/Abstract]) OR “Pollutant*, Particulate Air”[Title/Abstract]) OR “Ambient Particulate Matter”[Title/Abstract]) OR “Particulate Matter, Ambient”[Title/Abstract]).
The full strategy of searching literature in Embase was as follows: (exp Stroke or Stroke$:ab,ti or 'Cerebrovascular Accident$':ab,ti or CVA$:ab,ti or Apoplexy:ab,ti or 'Vascular Accident$, Brain':ab,ti or 'Brain Vascular Accident$':ab,ti or stroke:ab,ti) and (exp 'Air Pollution' or 'Pollution, Air':ab,ti or 'Air Quality':ab,ti or exp 'Particulate Matter' or 'Airborne Particulate Matter':ab,ti or 'Particulate Matter, Airborne':ab,ti or 'Air Pollutant$, Particulate':ab,ti or 'Particulate Air Pollutant$':ab,ti or 'Pollutant$, Particulate Air':ab,ti or 'Ambient Particulate Matter':ab,ti or 'Particulate Matter, Ambient':ab,ti).
Inclusion and exclusion criteria
Articles from three electronic databases were pooled to complete the preparation of screening. After deleting the duplications, we established two initial criteria for inclusion and exclusion, respectively. Inclusion criteria included cohort studies, “stroke” and “particulate matter” found in title or abstract. Exclusion criteria: studies without cohort design, animals, or special people enrolled as subjective of studies, and no MeSH term in abstract. After completion of initial screening, final criterion was used to select studies for meta-analysis. Final inclusion criterion for full texts of article: “PM2.5” and “hemorrhagic stroke.” Final exclusion criterion: absence of hazards ratio (HR) or “increase of PM2.5.” Screening of articles was conducted by one author (Kai Zhao) and examined and verified by another author (Jing Li). The third author (Mingfei Yang) would settle disagreement.
Data extraction
The data of first author’s name, publication year, study region, exposure, period, total numbers of participants, gender ratio (percent of male in all of the participants), definition of endpoints, increase extent of PM2.5, and HR with 95% confidence interval (CI) were extracted. When there were different results of HR with 95%CI for different models adjusted for various covariates, data and relative information of all models were extracted.
Quality assessment
All enrolled studies were independently assessed by two authors (Kai Zhao and Yu Guo) according to Newcastle-Ottawa Quality Assessment Scale Cohort Studies (NOS). All eligible studies were assessed from three main aspects: representativeness of participants included, levels of ambient PM2.5, and other factors that could influence the outcome, objectivity, and accuracy of determination of endpoint.
Data standardization
There were various factors that were adjusted to different proportional hazard models. If the coefficient of each factor was supplied in full text, crude data would be restored by conversion formula. The data adjusted by the least factors would not be selected to perform data processing, unless there was no way to acquire crude data. Likewise, data adjusted by different factors would be processed in the same way.
Statistical analysis
The pooled HR and 95%CI was calculated by random effects model. I-square (I2) was used to test the heterogeneity. Funnel plot asymmetry and Egger’s regression were used for detecting publication bias. Sensitivity analysis was used to attenuate heterogeneity, in which each study was omitted one by one and the pooled HR of the rest studies was retrieved or the random effects model was switched to fixed effects model. Meta-analysis was completed through corresponding modules in Software for Statistics and Data Science (version 15.1; College Station, TX 77845 USA). All p values were two-sided with a significant level at 0.05.
Results
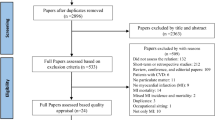
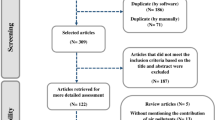
Overall, 2557 publications were obtained from the literature search (780 from PubMed, 15 from Cochrane, and 1762 from Embase). After removing duplicates, 1975 articles were initially included. After excluding reviews, researches of animals, and titles and abstracts that were not conformed to the theme of “PM2.5” and “Stroke,” 17 articles were retained for final screening. After excluding full texts without “PM2.5” and “Hemorrhagic Stroke” or other types of studies except for cohort studies, ultimately, 7 studies were included in this meta-analysis. Of these 7 studies, 6 studies (Huang et al. 2019; Cai et al. 2018; Downward et al. 2018; Qiu et al. 2017; Shin et al. 2019; Noh et al. 2019) reported the HR between exposure to the circumstance contaminated by growing PM2.5 and occurrence of HS or hemorrhagic cerebrovascular accident except that one study (Kim et al. 2018) mentioned the mortality caused by HS and used it as the endpoint. Detailed information of screening is exposed to Fig. 1. The characteristics of eligible studies are shown in Table 1.
Quality assessment for included studies
All the studies were assessed by NOS. Six studies were scored 7 points and one study was scored 8 points. All studies were scored in representativeness of the exposed cohort, ascertainment of exposure, comparability of cohorts on the basis of the design or analysis, assessment of outcome, and a long follow-up duration. Only one study (Huang et al. 2019) was scored in adequacy of follow-up of cohorts. Detailed information of screening is shown in Table 2.
Bias from characteristics of population
Cohorts of the 7 studies selected average located residents for studies’ participants which covered people in urban and rural regions, men and women, adults of all ages, and other aspects; thereinto, 3 studies (Downward et al. 2018; Shin et al. 2019; Noh et al. 2019) included the occurrence of immigration or the inhabitants’ movement in the research areas. One study (Kim et al. 2018) did not report the detailed information or characteristics of research population, and one study (Qiu et al. 2017) selected the elder whose age was over 60 years; moreover, these 2 studies were solely scored in reports of the non-exposed cohort.
Bias from exposure measurement
Data for ambient PM2.5 was acquired through satellite-based model in 5 studies, and data was collected from outdoor-automated monitoring stations in other 2 studies (Noh et al. 2019; Downward et al. 2018). With regard to extent of ambient PM2.5 increasing, the least level (Cai et al. 2018) was 1.4 μg/m3 and the highest one (Huang et al. 2019; Qiu et al. 2017; Kim et al. 2018; Noh et al. 2019) was 10 μg/m3. The range of exposure period was 3 (Cai et al. 2018) to 20 years (Qiu et al. 2017). The earliest observation (Kim et al. 2018) began in 1990, and the last cohort study (Huang et al. 2019; Shin et al. 2019) began in 2015.
Bias from covariate adjustment
Data processing for all 7 cohort studies was performed via proportional hazards models adjusted by different covariates. Mutual covariates were age (4 studies), gender (5 studies), education (4 studies), body mass index (4 studies), and smoking status (5 studies). Besides, 2 studies exhibited crude HR (Cai et al. 2018; Noh et al. 2019) and the data in a sole study (Kim et al. 2018) did not adjust any covariate.
Bias from definition of endpoint
All endpoints of included studies were defined according to items of International Classification of Diseases (ICD). Nevertheless, different editions and ranges of items in detail might result in bias of the meta-analysis. Three studies (Kim et al. 2018; Noh et al. 2019; Huang et al. 2019) only adopted content in ICD-10. Among them, one study (Kim et al. 2018) reported the most detailed items. Both ICD-9 and ICD-10 were used in 2 studies (Cai et al. 2018; Shin et al. 2019). Yet one study (Downward et al. 2018) did not mention editions or items of ICD and the last one (Qiu et al. 2017) only used ICD-9.
Increase of PM2.5 and HS
Total outcome
The crude HRs or HRs adjusted for the least factors of all 7 studies were shown in Fig. 2a, including exposure to PM2.5 increase and incidence or mortality of HS. The pooled HR for each 1.4–10 μg/m3 increase in PM2.5 was 1.10 (95%CI: 1.04–1.16), which indicated a positive association between exposure to growing PM2.5 and incidence or mortality of HS. Yet the heterogeneity of these studies was significant (I2 = 65.4%, p = 0.008). There was no publication bias according to Funnel plot and Egger’s regression (p = 0.225, Fig. 2b). Therefore, to attenuate the heterogeneity, sensitivity analysis was performed. Crude HRs were omitted one by one, and pooled HRs of the rest studies were calculated. All the pooled HRs, 95%CI, I2, and p are shown in Table 3. The difference between I2 was readily discernible. The heterogeneity did not disappear (p = 0.599) until the study (Shin et al. 2019) would be deleted. This might be the HR of the study was from a proportional hazards models adjusted by the least covariates. After a comprehensive survey of covariates in other studies, recent immigrants, income quintile, urban/rural area, and northern/southern Ontario might be special covariates that led to the bias. As the study was omitted, the pooled HR was 1.13 (95%CI: 1.09–1.17).
Subgroup analysis
Adjusted HRs for the most factors of all 7 enrolled studies are shown in Fig. 3a. The pooled HR for each study was 1.16 (95%CI: 1.03–1.30), which indicated a positive association between exposure to growing PM2.5 with different covariates and incidence or mortality of HS. Yet the heterogeneity of these studies was significant (I2 = 87.9%, p = 0.000). Publication bias was not evident according to Funnel plot and Egger’s regression (p = 0.401, Fig.. 3b). Therefore, sensitivity analysis was conducted to attenuate the heterogeneity. Adjusted HRs were omitted one by one, and pooled HRs of the rest studies were re-calculated. All the pooled HRs, 95%CI, I2, and p are shown in Table 4; however, the I2 was not attenuated.
According to the definite fact that smoking could damage the vascular endothelium, smoking might aggravate the effect of PM2.5 increment to brain vessels. Thus, subgroup analysis was performed. For individuals who never smoke and those who were former smokers versus individuals who were currently smoking, significant association between exposure to growing PM2.5 and incidence or mortality of HS was found (never or former smoking, pooled HR = 1.04, 95%CI: 0.74–1.46; current smoking, HR = 1.41, 95%CI: 0.92–2.15). Subgroup analysis and its sensitivity analysis carried out through switching the random effects model to fixed effects model is shown in Fig. 4 a and b.
Discussion
PM2.5 is a kind of the inhaled particulates that could enter the circulatory system via permeating alveolar epithelium and vascular endothelium. Therefore, PM2.5 might be transmitted to the arteries in the brain and deposited on the surface of vascular endothelium, further leading to HS. Thus, effect of deposition or particulates trundle might result in inflammation and injury of vascular endothelium. Finally, the suffered arteries might have corrosion damaging the vessel wall.
Chronic diseases (To et al. 2015) including angina, asthma, congestive heart failure, and diabetes are closely related to PM2.5. Many articles reported relationship between long-term exposure to PM2.5 and increased risks of incident stroke (Atkinson et al. 2013; Lipsett et al. 2011; Ljungman et al. 2019; Lin et al. 2016; Korek et al. 2015; Hoffmann et al. 2015; Dirgawati et al. 2019) and ischemic heart disease (Carey et al. 2016; Hartiala et al. 2016; Katsoulis et al. 2014; Loop et al. 2018; Ueda et al. 2012; Villeneuve et al. 2015; Stockfelt et al. 2017). A case-control study (Qian et al. 2019) reported that fatal intracranial hemorrhage incidence was associated with PM2.5 exposure and diabetes might increase the risk for intracerebral hemorrhage incidence in relation to PM2.5. In this meta-analysis, we found the evidence that exposure to different levels of ambient PM2.5 per unit was related to risk of HS in cohort studies. Sheng Yuan et al. (Yuan et al. 2019) concluded that pooled HR for stroke and long-term exposure to 5 μg/m3 increment of PM2.5 was 1.20 (95%CI: 0.79–1.80; I2 = 64.2%, p = 0.039) and the subgroup analysis outcomes of smoking status were 1.08 (95%CI: 1.03–1.13; I2 = 12.3%, p = 0.334) for never smoking, 1.11 (95%CI: 1.01–1.22; I2 = 0, p = 0.898) for former smoking, and 1.08 (95%CI: 0.94–1.25; I2 = 0, p = 0.462) for current smoking. This meta-analysis included less literatures than previous studies and had un-unified PM2.5 increment levels. Besides, due to limited data of included studies, the subgroups in this meta-analysis were divided into the current smoking subgroup and the subgroup of never smoking and former smoking.
In perspective of recommendations for further research about relationship between PM2.5 and risk of HS, a prospective cohort study is the first choice of study design selection. In the environment exposed by participants, the different levels of PM2.5 should preferably be obvious and balanced. All participants do not present the endpoint (such as HS) at the beginning period of a study. Exposure measurement is related to the sensitivity and accuracy of the outcome; therefore, the third scientific institutions and proper mathematical models would be used. As to adjustment for covariates, according to this meta-analysis, educated levels, classification of body mass index, and different age grade might be new researchable variables, which can be arranged to appropriate scales for subgroup analysis in the future. Definition of endpoints would be standardized and detailed according to professional items in the world; especially, adequacy of follow-up of cohorts will be manifested in articles.
As previously mentioned, there were several limitations of this systemic review. Only 7 articles from 3 open electronic databases during 2017 to 2019 were included in meta-analysis. Therefore, those not published in open platform were lost. Moreover, increment of PM2.5 was not unified, which ranged from 1.4 μg/m3 to 10 μg/m3. Numerous covariates were concealed in data synthesis, which might result in bias or heterogeneity of the meta-analysis and subgroup analysis.
Conclusion
Ambient PM2.5 level was significantly associated with the risk of HS, which might be a potential risk factor of HS. Additionally, under exposure of PM2.5, smoking does not further increase the risk of HS.
Availability of data and materials
All the data and material pertinent to this manuscript are included and have been reviewed by all authors.
References
Atkinson RW, Carey IM, Kent AJ, van Staa TP, Anderson HR, Cook DG (2013) Long-term exposure to outdoor air pollution and incidence of cardiovascular diseases. Epidemiology 24(1):44–53. https://doi.org/10.1097/EDE.0b013e318276ccb8
Cai Y-T, Hodgson S, Blangiardo M, Gulliver J, Morley D, Fecht D, Vienneau D, Hoogh K, Key T, Hveem K, Elliott P, Hansell AL (2018) Road traffic noise, air pollution and incident cardiovascular disease: a joint analysis of the HUNT, EPIC-Oxford and UK Biobank cohorts. Environ Int 114. https://doi.org/10.1016/j.envint.2018.02.048
Carey IM, Anderson HR, Atkinson RW, Beevers S, Cook DG, Dajnak D, Gulliver J, Kelly FJ (2016) Traffic pollution and the incidence of cardiorespiratory outcomes in an adult cohort in London. Occup Environ Med 73(12):849–856. https://doi.org/10.1136/oemed-2015-103531
Dirgawati M, Hinwood A, Nedkoff L, Hankey GJ, Yeap BB, Flicker L, Nieuwenhuijsen M, Brunekreef B, Heyworth J (2019) Long-term exposure to low air pollutant concentrations and the relationship with all-cause mortality and stroke in older men. Epidemiology 30:S82–S89. https://doi.org/10.1097/EDE.0000000000001034
Downward GS, van Nunen EJHM, Kerckhoffs J, Vineis P, Brunekreef B, Boer JMA, Messier KP, Roy A, Verschuren WMM, van der Schouw YT, Sluijs I, Gulliver J, Hoek G, Vermeulen R (2018) Long-term exposure to ultrafine particles and incidence of cardiovascular and cerebrovascular disease in a prospective study of a Dutch cohort. Environ Health Perspect 126(12):127007. https://doi.org/10.1289/EHP3047
Hartiala J, Breton CV, Tang WHW, Frederick Lurmann F, Hazen SL, Gilliland FD, Hooman Allayee H (2016) Ambient air pollution is associated with the severity of coronary atherosclerosis and incident myocardial infarction in patients undergoing elective cardiac evaluation. J Am Heart Assoc 5(8):e003947. https://doi.org/10.1161/jaha.116.003947
Hoffmann B, Weinmayr G, Hennig F, Fuks K, Moebus S, Weimar C, Dragano N, Hermann DM, Kälsch H, Mahabadi AA, Erbel R, Jöckel K-H (2015) Air quality, stroke, and coronary events. Dtsch Arztebl Int 112(12):195–201. https://doi.org/10.3238/arztebl.2015.0195
Huang K-Y, Liang F-C, Yang X-L, Liu F-C, Li J-X, Xiao Q-Y, Chen J-C, Liu X-Q, Cao J, Shen C, Yu L, Lu F-X, Wu X-P, Zhao L-C, Wu X-G, Li Y, Hu D-S, Huang J-F, Liu Y, Lu X-F, Gu D-F (2019) Long term exposure to ambient fine particulate matter and incidence of stroke: prospective cohort study from the China-PAR project. BMJ 367. https://doi.org/10.1136/bmj.l6720
Katsoulis M, Dimakopoulou K, Pedeli X, Trichopoulos D, Gryparis A, Trichopoulou A, Katsouyanni K (2014) Long-term exposure to traffic-related air pollution and cardiovascular health in a Greek cohort study. Sci Total Environ 490:934–940. https://doi.org/10.1016/j.scitotenv.2014.05.058
Kim J-H, Oh I-H, Park J-H, Cheong H-K (2018) Premature deaths attributable to long-term exposure to ambient fine particulate matter in the Republic of Korea. J Korean Med Sci 33(37). https://doi.org/10.3346/jkms.2018.33.e251
Korek MJ, Bellander TD, Lind T, Bottai M, Eneroth KM, Caracciolo B, de Faire UH, Fratiglioni L, Hilding A, Leander K, Magnusson PKE, Pedersen NL, Östenson C-G, Pershagen G, Penell JC (2015) Traffic-related air pollution exposure and incidence of stroke in four cohorts from Stockholm. J Expo Sci Environ Epidemiol 25(5):517–523. https://doi.org/10.1038/jes.2015.22
Lin H, Tao J, Du Y, Liu T, Qian Z-M, Tian L-W, Di Q, Zeng W-L, Xiao J-P, Guo L-C, Li X, Xu Y-J, Ma W-J (2016) Differentiating the effects of characteristics of PM pollution on mortality from ischemic and hemorrhagic strokes. Int J Hyg Environ Health 219(2):204–211. https://doi.org/10.1016/j.ijheh.2015.11.002
Lipsett MJ, Ostro BD, Reynolds P, Goldberg D, Hertz A, Jerrett M, Smith DF, Garcia C, Chang ET, Bernstein L (2011) Long-term exposure to air pollution and cardiorespiratory disease in the California Teachers Study cohort. Am J Respir Crit Care Med 184(7):828–835. https://doi.org/10.1164/rccm.201012-2082oc
Ljungman PLS, Andersson N, Stockfelt L, Andersson EM, Sommar JN, Eneroth K, Gidhagen L, Johansson C, Lager A, Leander K, Molnar P, Pedersen NL, Rizzuto D, Rosengren A, Segersson D, Wennberg P, Barregard L, Forsberg B, Sallsten G, Bellander T, Pershagen G (2019) Long-term exposure to particulate air pollution, black carbon, and their source components in relation to ischemic heart disease and stroke. Environ Health Perspect 127(10):107012. https://doi.org/10.1289/EHP4757
Loop MS, Mcclure LA, Levitan EB, Al-Hamdan MZ, Crosson WL, Safford MM (2018) Fine particulate matter and incident coronary heart disease in the REGARDS cohort. Am Heart J 197:94–102. https://doi.org/10.1016/j.ahj.2017.11.007
Noh J, Sohn J-W, Han M-K, Kang D-Y, Choi Y-J, Kim H-C, Suh I, Kim C-S, Shin D-C (2019) Long-term effects of cumulative average PM2.5 exposure on the risk of hemorrhagic stroke. Epidemiology 30(Suppl 1):S90–S98. https://doi.org/10.1097/EDE.0000000000001001
Qian Y-F, Yu H-T, Cai B-X, Fang B, Wang C-F (2019) Association between incidence of fatal intracerebral hemorrhagic stroke and fine particulate air pollution. Environ Health Prev Med 24(1):38. https://doi.org/10.1186/s12199-019-0793-9
Qiu H, Sun S-Z, Tsang H, Wong C-M, Lee RS-y, Schooling M, Tian L-W (2017) Fine particulate matter exposure and incidence of stroke: a cohort study in Hong Kong. Neurology 88(18):1709–1717. https://doi.org/10.1212/WNL.0000000000003903
Shin S, Burnett RT, Kwong JC, Hystad P, van Donkelaar A, Brook JR, Goldberg MS, Tu K, Copes R, Martin RV, Liu Y, Kopp A, Chen H (2019) Ambient air pollution and the risk of atrial fibrillation and stroke: a population-based cohort study. Environ Health Perspect 127(8):087009. https://doi.org/10.1289/EHP4883
Stockfelt L, Andersson EM, Molnár P, Gidhagen L, Segersson D, Rosengren A, Barregard L, Sallsten G (2017) Long-term effects of total and source-specific particulate air pollution on incident cardiovascular disease in Gothenburg, Sweden. Environ Res 158:61–71. https://doi.org/10.1016/j.envres.2017.05.036
To T, Zhu J-Q, Villeneuve PJ, Simatovic J, Feldman L, Gao C-W, Williams D, Chen H, Weichenthal S, Wall C, Miller AB (2015) Chronic disease prevalence in women and air pollution — a 30-year longitudinal cohort study. Environ Int 80:26–32. https://doi.org/10.1016/j.envint.2015.03.017
Ueda K, Nagasawa SY, Nitta H, Miura K, Ueshima H (2012) Exposure to particulate matter and long-term risk of cardiovascular mortality in Japan: NIPPON DATA80. J Atheroscler Thromb 19(3):246–254. https://doi.org/10.5551/jat.9506
Villeneuve PJ, Weichenthal SA, Crouse D, Miller AB, To T, Martin RV, van Donkelaar A, Wall C, Burnett RT (2015) Long-term exposure to fine particulate matter air pollution and mortality among Canadian women. Epidemiology 26(4):1–545. https://doi.org/10.1097/EDE.0000000000000294
Yuan S, Wang J-X, Jiang Q-Q, He Z-Y, Huang Y-C, Li Z-Y, Cai L-Y, Cao S-Y (2019) Long-term exposure to PM2.5 and stroke: a systematic review and meta-analysis of cohort studies. Environ Res 177. https://doi.org/10.1016/j.envres.2019.108587
Funding
This study was funded by the Project of Science and Technology Department of Qinghai Province (Grant Numbers 2020-ZJ-774) and National Key R&D Program of China (Nos. 2018YFC1312600 and 2018YFC1312601).
Author information
Authors and Affiliations
Contributions
Kai Zhao contributed to the majority of systemic review and manuscript draft preparation, Jing Li contributed to inclusion and exclusion criteria and manuscript writing, Yu Guo contributed to literature search and quality assessment, Chaonan Du contributed to data management and statistical analysis, and Mingfei Yang supervised all of work.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Responsible Editor: Lotfi Aleya
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Kai Zhao and Jing Li are co-first authors.
Appendix
Appendix
The crude data extracted from literature included.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, K., Li, J., Du, C. et al. Ambient fine particulate matter of diameter ≤ 2.5 μm and risk of hemorrhagic stroke: a systemic review and meta-analysis of cohort studies. Environ Sci Pollut Res 28, 20970–20980 (2021). https://doi.org/10.1007/s11356-021-13074-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-13074-7