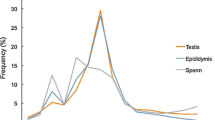
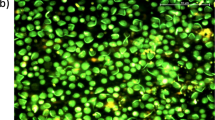
Abstract
Spermatogenesis is a complicated and poorly understood process that relies on the precise regulation of the self-renewal and differentiation of spermatogonia. In many organisms, microRNAs (miRNAs) are involved in multiple developmental processes as critical regulators of transcriptional and post-transcriptional gene silencing. This study investigated the expression pattern of miRNAs in type B spermatogonia cells (BSc) and primary spermatocytes (PSc) of mice, using a high-throughput small RNA sequencing system. The results revealed that the expression levels of Let-7 family miRNAs were remarkably high in both cell types. Furthermore, the expression levels of miR-21, miR-140-3p, miR-103, miR-30a, miR-101b and miR-99b were decreased during the transformation from BSc to PSc. These miRNAs target vital genes that participate in apoptosis, cell proliferation and differentiation, junction assembly and cell cycle regulation. These results highlight the indispensable role of miRNAs in spermatogenesis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
de Rooij DG. Proliferation and differentiation of spermatogonial stem cells. Reproduction, 2001, 121: 347–354
Zhou Q, Griswold MD. Regulation of spermatogonia. Stembook, 2008
Tolia NH, Joshua TL. Slicer and the argonautes. Nat Chem Biol, 2007, 3: 36–43
Maatouk DM, Loveland KL, Mcmanus MT, Moore K, Harfe BD. Dicer1 is required for differentiation of the mouse male germline. Biol Reprod, 2008, 79: 696–703
Lin H. piRNAs in the germ line. Science, 2007, 316: 397
Gan HY, Lin XW, Zhang ZQ, Zhang W, Liao SY, Wang LX, Han CS. piRNA profiling during specific stages of mouse spermato-genesis. RNA, 2011, 17: 1191–1203
Peng H, Shi J, Zhang Y, Zhang H, Liao S, Li W, Lei L, Han C, Ning L, Cao Y, Zhou Q, Chen Q, Duan E. A novel class of tRNA-derived small RNAs extremely enriched in mature mouse sperm. Cell Res, 2012, 22: 1609–1612
Rana TM. Illuminating the silence: Understanding the structure and function of small RNAs. Nat Rev Mol Cell Bio, 2007, 8: 23–36
Kim VN, Nam JW. Genomics of microRNA. Trends Genet, 2006, 22: 165–173
Tijsterman M, Plasterk RH. Dicers at RISC; the mechanism of RNAi. Cell, 2004, 117: 1–3
González-González E, López-Casas PP, Del Mazo J. Gene silencing by RNAi in mouse Sertoli cells. Reprod Biol Endocrinol, 2008, 6: 29
Hayashi K, Chuva de Sousa Lopes SM, Kaneda M, Tang F, Hajkova P, Lao K, O’Carroll D, Das PP, Tarakhovsky A, Miska EA, Surani MA. microRNA biogenesis is required for mouse primordial germ cell development and spermatogenesis. PLoS One, 2008, 3: e1738
Gangaraju VK, Lin HF. MicroRNA: Key regulators of stem cells. Nat Rev Mol Cell Biol, 2009, 10: 116–125
Niu Z, Goodyear SM, Rao S, Wu X, Tobias JW, Avarbock MR, Brinster RL. MicroRNA-21 regulates the self-renewal of mouse spermatogonial stem cells. Proc Natl Acad Sci USA, 2011, 108: 12740–12745
Hofmann MC, Hess RA, Goldberg E, Millan JL. Immortalized germ cells undergo meiosis in vitro. Proc Natl Acad Sci USA, 1994, 91: 5533–5537
Hofmann MC, Narisawa S, Hess RA, Millan JL. Immortalization of germ cells and somatic testicular cells using the SV40 large T antigen. Exp Cell Res, 1992, 201: 417–435
Li R, Li Y, Kristiansen K, Wang J. SOAP: Short oligonucleotide alignment program. Bioinformatics, 2008, 24: 713–714
Audic S, Claverie JM. The significance of digital gene expression profiles. Genome Res, 1997, 7: 986–996
Dweep H, Sticht C, Pandey P, Gretz N. Mirwalk-database: Prediction of possible miRNA binding sites by “walking” the genes of three genomes. J Biomed Inform, 2011, 44: 839–847
Chen C, Ridzon DA, Broomer AJ, Zhou Z, Lee DH, Nguyen JT, Barbisin M, Xu NL, Mahuvakar VR, Andersen MR, Lao KQ, Livak KJ, Guegler KJ. Real-time quantification of microRNAs by stem-loop RT-PCR. Nucleic Acids Res, 2005, 33: e179
Boyerinas B, Park SM, Hau A, Murmann AE, Peter ME. The role of let-7 in cell differentiation and cancer. Endocri Relat Cancer, 2010, 17: 19–36
Houbaviy HB, Murray MF, Sharp PA. Embryonic stem cell-specific microRNAs. Dev Cell, 2003, 5: 351–358
Zhang X, Hao L, Meng L, Liu M, Zhao L, Hu F, Ding C, Wang Y, He B, Pan Y, Fang W, Chen J, Hu S, Jia M. Digital gene expression tag profiling analysis of the gene expression patterns regulating the early stage of mouse spermatogenesis. PLoS One, 2013, 8: e58680
Rougvie AE. Control of developmental timing in animals. Nat Rev Genet, 2001, 2: 690–701
Lee RC, Feinbaum RL, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell, 1993, 75: 843–854
Johnson CD, Esquela-Kerscher A, Stefani G, Byrom M, Kelnar K, Ovcharenko D, Wilson M, Wang X, Shelton J, Shingara J, Chin L, Brown D, Slack FJ. The let-7 microRNA represses cell proliferation pathways in human cells. Cancer Res, 2007, 67: 7713–77122
Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA, Ruotti V, Stewart R, Slukvin II, Thomson JA. Induced pluripotent stem cell lines derived from human somatic cells. Science, 2007, 318: 1917–1920
Viswanathan SR, Daley GQ, Gregory RI. Selective blockade of microRNA processing by lin28. Science, 2008, 320: 97–100
Newman MA, Thomson JM, Hammond SM. Lin-28 interaction with the let-7 precursor loop mediates regulated microRNA processing. RNA, 2008, 14: 1539–1549
Piskounova E, Viswanathan SR, Janas M, LaPierre RJ, Daley GQ, Sliz P, Gregory RI. Determinants of micro processing inhibition by the developmentally regulated RNA-binding protein lin28. J Biol Chem, 2008, 283: 21310–21314
Moss EG, Tang L. Conservation of the heterochronic regulator lin-28, its developmental expression and microRNA complementary sites. Dev Bio, 2003, 258: 432–442
Johnson SM, Grosshans H, Shingara J, Byrom M, Jarvis R, Cheng A, Labourier E, Reinert KL, Brown D, Slack FJ. Ras is regulated by the let-7 microRNA family. Cell, 2005, 120: 635–647
Gillis AJ, Stoop HJ, Hersmus R, Oosterhuis JW, Sun Y, Chen C, Guenther S, Sherlock J, Veltman I, Baeten J, van der Spek PJ, de Alarcon P, Looijenga LH. High-throughput microRNAome analysis in human germ cell tumours. J Pathol, 2007, 213: 319–328
Martins RP, Krawetz SA. RNA in human sperm. Asian J Androl, 2005, 7: 115–120
Seike M, Goto A, Okano T, Bowman ED, Schetter AJ, Horikawa I, Mathe EA, Jen J, Yang P, Sugimura H, Gemma A, Kudoh S, Croce CM, Harris CC. miR-21 is an EGFR-regulated anti-apoptotic factor in lung cancer in never-smokers. Proc Natl Acad Sci USA, 2009, 106: 12085–12090
Sayed D, Rane S, Lypowy J, He M, Chen IY, Vashistha H, Yan L, Malhotra A, Vatner D, Abdellatif M. MicroRNA-21 targets sprouty2 and promotes cellular outgrowths. Mol Bio Cell, 2008, 19: 3272–3282
Selcuklu SD, Donoghue MT, Spillane C. miR-21 as a key regulator of oncogenic processes. Biochem Soc Trans, 2009, 37: 918–925
Miyaki S, Sato T, Inoue A, Otsuki S, Ito Y, Yokoyama S, Kato Y, Takemoto F, Nakasa T, Yamashita S, Takada S, Lotz MK, Ueno-Kudo H, Asahara H. MicroRNA-140 plays dual roles in both cartilage development and homeostasis. Genes Dev, 2010, 24: 1173–1185
Herrera BM, Lockstone HE, Taylor JM, Ria M, Barrett A, Collins S, Kaisaki P, Argoud K, Fernandez C, Travers ME, Grew JP, Randall JC, Gloyn AL, Gauguier D, McCarthy MI, Lindgren CM. Global microRNA expression profiles in insulin target tissues in a spontaneous rat model of type 2 diabetes. Diabetologia, 2010, 53: 1099–1109
Liu SP, Fu RH, Yu HH, Li KW, Tsai CH, Shyu WC, Lin SZ. MicroRNAs regulation modulated self-renewal and lineage differentiation of stem cells. Cell Transplant, 2009, 18: 1039–1045
Baraniskin A, Birkenkamp-Demtroder K, Maghnouj A, Zöllner H, Munding J, Klein-Scory S, Reinacher-Schick A, Schwarte-Waldhoff I, Schmiegel W, Hahn SA. miR-30a-5p suppresses tumor growth in colon carcinoma by targeting DTL. Carcinogenesis, 2012, 33: 732–739
Buechner J, Tømte E, Haug BH, Henriksen JR, Løkke C, Flægstad T, Einvik C. Tumour-suppressor microRNAs let-7 and mir-101 target the proto-oncogene mycn and inhibit cell proliferation in MYCN-amplified neuroblastoma. Br J Cancer, 2011, 105: 296–303
Kottakis F, Polytarchou C, Foltopoulou P, Sanidas I, Kampranis SC, Tsichlis PN. FGF-2 regulates cell proliferation, migration, and angiogenesis through an NDY1/KDM2B-MiR-101-EZH2 pathway. Cell Molecular, 2011, 43: 285–298
Singh SK, Kagalwala MN, Parker-Thornburg J, Adams H, Majumder S. Rest maintains self-renewal and pluripotency of embryonic stem cells. Nature, 2008, 453: 223–227
Georgantas RW 3rd, Hildreth R, Morisot S, Alder J, Liu CG, Heimfeld S, Calin GA, Croce CM, Civin CI. CD34+ hematopoietic stem-progenitor cell microRNA expression and function: A circuit diagram of differentiation control. Proc Natl Acad Sci USA, 2007, 104: 2750–2755
Phillips Bt, Gassei K, Orwig Ke. Spermatogonial stem cell regulation and spermatogenesis. Philos Trans R Soc Lond B Biol Sci, 2010, 365: 1663–1678
Author information
Authors and Affiliations
Corresponding authors
Additional information
Contributed equally to this work
This article is published with open access at springerlink.fh-diploma.de
Electronic supplementary material
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Luo, M., Hao, L., Hu, F. et al. MicroRNA profiles and potential regulatory pattern during the early stage of spermatogenesis in mice. Sci. China Life Sci. 58, 442–450 (2015). https://doi.org/10.1007/s11427-014-4737-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-014-4737-8