Abstract
Background
Daratumumab, an anti-CD38 monoclonal antibody, is used for treatment of multiple myeloma (MM) and light chain amyloidosis at an intravenous dosage of 16 mg/kg or at a subcutaneous fixed dose of 1800 mg. However, the subcutaneous formulation has only recently been approved in Europe, and real-life data on its safety are still few.
Objective
In this multicenter retrospective real-life experience, we provided evidence for the safety of subcutaneous daratumumab in plasma cell disorders.
Patients and Methods
A total of 189 patients diagnosed with MM or light chain amyloidosis were included in this retrospective study, and all subjects were daratumumab-naïve. Primary endpoint was safety of subcutaneous daratumumab, especially for infusion-related reaction (IRR) incidence and severity. All patients received premedication with dexamethasone, paracetamol, and antihistamine, with montelukast usage in 85% of cases.
Results
Eight patients (4%) experienced IRRs, mainly of grade I–II, and other frequent toxicities were: hematological (thrombocytopenia, 4%; neutropenia, 5%; lymphopenia, 6%) and non-hematological (pneumonia, 4%; diarrhea, 2%; and cytomegalovirus reactivation, 0.5%). In our multicenter retrospective real-life experience, subcutaneous daratumumab was well-tolerated with an excellent safety profile with a very low (4%) IRR incidence, even in frailer MM patients with severe renal impairment or increased body weight.
Conclusions
Subcutaneous daratumumab was safe in a real-life setting including patients with severe renal failure and advanced disease. However, further studies on larger and prospective cohorts are required to confirm our real-life observations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Subcutaneous daratumumab, an anti-myeloma monoclonal antibody, is administered at a fixed dose of 1800 mg with similar efficacy compared with the intravenous formulation. |
Subcutaneous daratumumab has very low incidence of infusion-related reactions and hematological toxicities, even in patients with severe comorbidities and advanced disease. |
1 Introduction
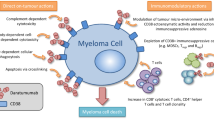
Plasma cell disorders, including multiple myeloma (MM) and light chain amyloidosis, are hematological disorders characterized by bone marrow (BM) proliferation of neoplastic plasma cells producing monoclonal proteins (M-proteins) that accumulate in tissues causing progressive organ damage [1,2,3]. Neoplastic plasma cells express CD38, a 45-kDa type II transmembrane glycoprotein present at low levels in myeloid, lymphoid, and other cell types [4]. In neoplastic cells, CD38 regulates several cell functions, including proliferation, survival, adhesion pathways, and ectoenzyme activity for calcium signaling, thus making this surface marker an ideal therapeutic target for MM treatment [5]. Daratumumab, a fully humanized IgG-k anti-CD38 monoclonal antibody, exerts its anti-neoplastic activity through several mechanisms, such as antibody-dependent cellular cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP), direct cellular apoptosis, and modulation of extracellular ectoenzyme activity and has demonstrated efficacy in MM treatment as a single agent or in combination with other anti-tumor agents [5,6,7,8,9,10]. Daratumumab was first approved as an intravenous drug at a dosage of 16 mg/kg; however, high rates of infusion-related reactions (IRRs) (45–48% of events) have been described in the phase III trials CASTOR and POLLUX, especially during the first administration; most were grade I–II, with grade III–IV being more rare (8.6%) [8, 11,12,13]. Therefore, pre- and postmedication with corticosteroids, acetaminophen, and antihistamines is required for IRR prophylaxis, as well as prolongation of daratumumab infusion time [13]. To decrease IRR rate and severity, a subcutaneous formulation has been developed and tested in MM patients, showing a shorter administration time compared with intravenous daratumumab (3–5 min versus 3.4–7.0 h, subcutaneous versus intravenous administration), and lower IRR rates [13, 14]. This improved toxicity profile is related to the combination of the anti-CD38 monoclonal antibody to a recombinant human hyaluronidase PH20 (rHuPH20), favoring systemic absorption through interstitial skin matrix depolymerization [15]. Non-inferiority and the improved safety profile of subcutaneous daratumumab compared with intravenous formulation has been confirmed in combination with standard treatment regimens and in newly diagnosed and relapsed/refractory (RR) patients with MM in phase Ib PAVO (NCT02519452), phase II PLEIADES (NCT03412565), and phase III COLUMBA (NCT03277105) [16,17,18]. Moreover, subcutaneous daratumumab is administered at a fixed dosage of 1800 mg, regardless of renal function and body weight [16, 17, 19]. The US Food and Drug Administration approved this formulation for use in the USA in 2020, while the European Medicines Agency approved it in late 2021, and the Italian Agency only approved it in early 2022. Therefore, few real-life studies of efficacy and safety of subcutaneous daratumumab are present. In addition, few data are present on safety of this formulation in patients with MM with advanced-stage renal failure and/or disease, as they are underrepresented in clinical trials. In this retrospective multicenter real-world Southern Italian experience, we investigated safety of subcutaneous daratumumab in newly diagnosed patients and patients with RR/MM, both eligible and not eligible for autologous hematopoietic stem cell transplantation (auto-HSCT), and in amyloid light chain (AL) amyloidosis subjects, also including subjects with severe renal failure and advanced disease.
2 Materials and Methods
2.1 Patients
Eight hematology units from Campania and Basilicata regions, Southern Italy, participated in this study: University Hospital “San Giovanni di Dio e Ruggi d'Aragona,” Salerno; Hospital “Antonio Cardarelli,” Naples; Hospital “Andrea Tortora,” Pagani; University Hospital “Federico II,” Naples; Hospital “Sant’Anna e San Sebastiano,” Caserta; Hospital “San Giuseppe Moscati,” Aversa; Hospital “San Carlo,” Potenza; and Hospital “San Giuliano,” Giugliano. A total of 189 patients diagnosed with MM or light chain amyloidosis were included in this retrospective study, since December 2021. Inclusion criteria were: age ≥ 18 years old, diagnosis of MM or light chain amyloidosis according to international guidelines [20, 21], and administration of at least one dose of subcutaneous daratumumab outside clinical trials in absence of previous exposure to the intravenous formulation.
2.2 Subcutaneous Daratumumab Administration and Premedications
Subcutaneous daratumumab was administered at the fixed dose of 1800 mg as single agent or in combination with standard chemotherapy until disease progression or unacceptable toxicity. Premedication was carried out with a bolus of 20 mg dexamethasone and 10 mg chlorphenamine and 1000 mg of oral acetaminophen. Postmedication with 20 mg dexamethasone was administered on the following day of the first administration, or otherwise stated.
2.3 Statistical Analysis and Endpoints
Primary endpoint was safety of subcutaneous daratumumab, especially IRR incidence and severity. Data were collected in spreadsheets and analyzed using R statistical software (v. 4.0.5; RStudio) and SPSS (v. 25; IBM). Logistic regression models were used to investigate effects and relative odds ratios of independent variables on IRR incidence. To reduce the impact of confounding factors in univariate logistic regression analysis, multivariate regression models were carried out, and Nagelkerke’s R-squared test was used to assess model validity. A P value < 0.05 was considered statistically significant.
3 Results
3.1 Clinical Characteristics at Enrollment
Clinical characteristics are summarized in Table 1. Median age at first subcutaneous daratumumab administration was 67 years (range 36–85 years), 97 (51%) patients were males, and 129 (68%) weighed > 65 kg. A total of 9 (5%) patients were diagnosed with AL amyloidosis, and the remaining 180 (95%) patients were diagnosed with MM. The most common heavy and light chain M-proteins were IgG (68%) and kappa (62%), respectively. Median frequency of BM plasma cells were 47% (range, 2–93%), and 43 patients (23%) had BM plasma cell infiltration ≥ 60%. High-risk disease was observed in 14% of cases (N = 26), and 21% (N = 40), 34% (N = 64), and 29% (N = 55) of patients were stratified as revised international staging system (R-ISS) stage I, II, and III, respectively. Regarding renal function, median glomerular filtration rate (GFR) was 73 mL/min (range 4–125 mL/min), and 33 patients showed a GFR < 40 mL/min (17%) with four of them (2%) requiring dialysis. Clinical characteristics of patients with severe renal dysfunction are summarized in Table 1. In this subgroup of patients, subjects more frequently had an IgA or light chain advanced-stage MM with higher levels of β2-microglubulin levels compared with patients with normal renal function (Table 1). In addition, 57%, 70%, and 19% of patients presented with anemia, at least one osteolytic lesion, and hypercalcemia, respectively, while extramedullary disease was observed in 28 cases (15%). Median β2-microglubulin levels were 4 mg/dL (range 0.6–54 mg/dL), and median albumin was 3.8 g/dL (range 1.5–5 g/dL).
3.2 Treatment Description
All patients received premedication with dexamethasone, chlorphenamine, and acetaminophen, and 161 of them (85%) also were premedicated with montelukast as IRR prophylaxis, as per Italian recommendations and based on medical decision. Subcutaneous daratumumab was administered in combination with bortezomib, thalidomide, and dexamethasone in 37% of cases (N = 70), with lenalidomide and dexamethasone in 36% (N = 68), with melphalan and prednisone in 4% (N = 8), with bortezomib and dexamethasone in 7% (N = 13), with cyclophosphamide, bortezomib, and dexamethasone in 5% (N = 9), with pomalidomide and dexamethasone in 1% (N = 2), or as a single agent in 10% of cases (N = 19). Subcutaneous daratumumab was given as first-line therapy in 65% of subjects (N = 123), as second-line treatment in 23% (N = 43), and as a third or greater than a third-line regimen in 12% of patients (N = 23), with a median number of administrations of 11 (range, 1–23) for a single patient (total number of administrations was 2078). Median administration was 4 min (range, 3–5 min). Drug discontinuation rate was 22% (N = 42), and reasons for discontinuation were: disease progression (18%, N = 34), drug intolerance (0.5%, N = 1), death (2%, N = 4), or medical decision (1.6%, N = 3).
3.3 Safety and Risk Factors of IRR Occurrence
Safety features are summarized in Table 2. Subcutaneous daratumumab was well tolerated, with only eight (4%) IRR events, mainly of grade I–II (3.5%; N = 7), and only one severe IRR (0.5%), all occurring during the first administration. The patient with grade III–IV reaction had a complete recovery after high-dose steroids and anti-histamines. Median time for IRR onset was 2.5 h (range 1–7 h). No delayed reactions were reported. Patients who received montelukast together with standard premedication tended to have a lower risk of IRR [3.8% versus 7.6%; odds ratio (OR), 0.43; P = 0.4]. Regarding hematological toxicities, 30 patients (16%) experienced grade I–II toxicities, including thrombocytopenia (N = 8; 4%), neutropenia (N = 10; 5%), and lymphopenia (N = 12; 6%); we observed grade I–II daratumumab-related diarrhea in 4 cases (2%). Other reported toxicities were: pneumonia (grade I–II, 2% and III–IV, 2%) and cytomegalovirus (CMV) reactivation (0.5%) requiring preemptive therapy with antiviral agents. Safety characteristics of patients with severe renal dysfunction are presented in Table 2, and no IRRs were reported while other hematological and non-hematological toxicities were observed, especially in those subjects treated with daratumumab in combination with chemotherapy.
Next, a univariate logistic regression analysis was performed to investigate the effects of several patient-, disease-, and treatment-related features on IRR incidence (Table 3). Significant associations with high-risk cytogenetic disease [OR 6.87; 95% confidential interval (CI), 1.60–29.4; P = 0.009] and extramedullary disease (OR 6.37; 95% CI 1.49–27.2; P = 0.01) were observed; multivariate analysis was also performed (high-risk cytogenetic disease OR 16.36; 95% CI 2.44–109.73; P = 0.005; and extramedullary disease OR 12.25; 95% CI 1.95-76.72; P = 0.007) (Table 4). Conversely, montelukast added to premedication did not decrease IRR incidence (OR 0.5; 95% CI 0.09–2.62; P = 0.41), as well as daratumumab as first-line therapy (OR 1.9; 95% CI 0.46–7.93; P = 0.36).
4 Discussion
Subcutaneous daratumumab at a fixed dosage of 1800 mg has been recently approved for newly diagnosed patients and patients with RR/MM as single agents or in combination with standard therapy based on non-inferiority and high safety observed in PAVO, PLEIADES, and COLUMBA trials [16,17,18,19]. In registrational trials, patients with late-stage disease and severe renal impairment are under-represented; therefore, real-life data are important to accurately assess efficacy and safety in the postmarketing phase. In our multicenter retrospective real-life experience, we investigated safety of subcutaneous daratumumab in MM and AL amyloidosis patients.
In recent years, the introduction of intravenous daratumumab at a dose of 16 mg/kg in combination with proteasome inhibitors (bortezomib or carfilzomib), immunomodulators (thalidomide, lenalidomide, or pomalidomide), and corticosteroids (dexamethasone or prednisone) has revolutionized the prognosis of patients with MM [5]. Despite clinical benefits, the intravenous formulation has high IRR rates, also requiring drug discontinuation, and is associated with several hematological and non-hematological toxicities [22, 23]. To reduce IRR incidence, a subcutaneous daratumumab formulation was introduced with a dramatic decrease in IRR, ranging from less than 10–16% [16,17,18,19]. In our cohort of 65% newly diagnosed patients and 35% of patients with RR/MM, IRR rate was 4%, similar to that reported in pivotal PLEIADES study and significantly lower than those observed in patients treated with intravenous daratumumab. IRR incidence during intravenous daratumumab administration ranges from 25.7% (ALCYONE study including 350 patients with MM treated with intravenous daratumumab, bortezomib, melphalan, and prednisone, ineligible for transplant) to 45.6% (POLLUX study including 286 patients with RR/MM treated with intravenous daratumumab, lenalidomide, and dexamethasone) [8, 9, 19]. Moreover, our particularly low IRR incidence could be related to the use of subcutaneous daratumumab as first-line therapy and the use of a premedication of dexamethasone, chlorphenamine, oral acetaminophen, with or without montelukast, followed by a postmedication with dexamethasone on the following day after the first administration. Indeed, patients in the PLEIADES study treated with subcutaneous daratumumab as first-line therapy displayed the lowest IRR incidence [16,17,18,19].
Other documented subcutaneous daratumumab-induced toxicities are: lymphopenia, thrombocytopenia, and neutropenia (Table 5) [16,17,18,19]. In our real-life study, hematological toxicities were 16% without grade III–IV events, and were more frequently lymphopenia (6%), neutropenia (5%), and thrombocytopenia (4%); however, incidence was lower than that reported in other pivotal studies, supporting the great tolerability of the drug even in a real-life setting. Other non-hematological toxicities reported in pivotal studies are: arthralgia, back pain, diarrhea, and upper and/or lower respiratory tract infections [16,17,18,19]. In our series, no episodes of arthralgia and back pain were reported; we observed diarrhea (2%) and CMV reactivation (0.5%), while pneumonia occurred in 4% of cases with half of them grade III–IV, similar to that reported in PLEIADES study for the group of patients that were transplant-eligible treated with daratumumab, bortezomib, lenalidomide, and dexamethasone [17].
In pivotal studies, most enrolled patients are in R-ISS stage I (52.2% in PAVO study; 31.2 % in COLUMBA study; 44.8%, 32.8%, and 41.5% in PLEIADES study for the three subgroups) [16,17,18,19], while we included a high proportion of subjects with R-ISS stages II and III (34% and 29%, respectively), with extramedullary disease (15%) and renal impairment (17%, with 2% of patients under dyalisis treatment). Patients with MM with severe renal impairment are usually under-represented in clinical trials, while comprising the majority of subjects in real-life clinical practice. In our study, we enrolled consecutive patients with MM regardless of disease stage and renal function, and 63% of them had advanced disease and 17% had end-stage renal failure, also requiring dialisis in 2% of them. Surprisingly, none of the patients with severe renal impairment experienced IRR, and hematologic and non-hematologic toxicities were more frequently observed in combinatorial regimens, rather than daratumumab as single agent, suggesting a manageable safety profile of this monoclonal antibody.
Despite low IRR incidence in our cohort, patients with high-risk disease and/or with extramedullary locations had an increased risk of IRR occurrence by uni- and multivariate analysis, while renal impairment and body weight were not associated with this type of toxicity. Montelukast, a selective antagonist of leukotriene D4 (LTD4) receptor has been proposed as an additional premedication agent prior to intravenous daratumumab administration [24]; however, no significant IRR incidence reduction was observed in our study, although 15% of our patients did not receive montelukast premedication and showed a slight increase in IRR rates (7.6% versus 3.8% of montelukast-premedicated patients). Moreover, median administration time was 4 min and patients were observed for 3 h after administration as per COLUMBA trial recommendations [18, 19], only after the first dose, while they were immediately discharged after the following cycles.
Strengths of our multicenter real-life experience are: our patients have never been treated with daratumumab before enrollment, in contrast with other studies [16,17,18,19]; therefore, to the best of our knowledge, this is the first real-life experience on safety of subcutaneous daratumumab formulation in a population completely naïve to this anti-CD38 agent; and we also included patients with high-risk disease and severe renal failure. Limitations of our study are: assessment of patients’ quality of life and real benefit of montelukast premedication, as this is not a randomized controlled trial, and a low number of total reactions for an accurate identification of IRR predictors.
In conclusion, our real-life results confirmed an excellent safety profile of subcutaneous daratumumab even in frailer MM patients with severe renal impairment or increased body weight, while subjects with high-risk and/or extramedullary disease might be at higher risk of IRR occurrence, and might likely benefit more from montelukast addition to premedication. However, further studies on larger and prospective cohorts are required to confirm our real-life observations.
References
Gavriatopoulou M, Musto P, Caers J, Merlini G, Kastritis E, van de Donk N, et al. European myeloma network recommendations on diagnosis and management of patients with rare plasma cell dyscrasias. Leukemia. 2018;32(9):1883–98.
Iula R, De Novellis D, Trastulli F, Della Pepa R, Fontana R, Carobene A, et al. Efficacy and safety of belantamab-mafodotin in triple-refractory multiple myeloma patients: a multicentric real-life experience. Front Oncol. 2022;12:1026251.
De Novellis D, Fontana R, Carobene A, Serio B, Ferrara I, Martorelli MC, et al. Serum Free light-chain ratio at diagnosis is associated with early renal damage in multiple myeloma: a case series real-world study. Biomedicines. 2022;10(7):1657.
Deaglio S, Mehta K, Malavasi F. Human CD38: a (r)evolutionary story of enzymes and receptors. Leuk Res. 2001;25(1):1–12.
De Novellis D, Fontana R, Giudice V, Serio B, Selleri C. Innovative anti-CD38 and anti-BCMA targeted therapies in multiple myeloma: mechanisms of action and resistance. Int J Mol Sci. 2022;24(1):645.
Usmani SZ, Weiss BM, Plesner T, Bahlis NJ, Belch A, Lonial S, et al. Clinical efficacy of daratumumab monotherapy in patients with heavily pretreated relapsed or refractory multiple myeloma. Blood. 2016;128(1):37–44.
Facon T, Kumar S, Plesner T, Orlowski RZ, Moreau P, Bahlis N, et al. Daratumumab plus lenalidomide and dexamethasone for untreated myeloma. N Engl J Med. 2019;380(22):2104–15.
Dimopoulos MA, Oriol A, Nahi H, San-Miguel J, Bahlis NJ, Usmani SZ, et al. Daratumumab, lenalidomide, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375(14):1319–31.
Dimopoulos MA, San-Miguel J, Belch A, White D, Benboubker L, Cook G, et al. Daratumumab plus lenalidomide and dexamethasone versus lenalidomide and dexamethasone in relapsed or refractory multiple myeloma: updated analysis of POLLUX. Haematologica. 2018;103(12):2088–96.
Moreau P, Attal M, Hulin C, Arnulf B, Belhadj K, Benboubker L, et al. Bortezomib, thalidomide, and dexamethasone with or without daratumumab before and after autologous stem-cell transplantation for newly diagnosed multiple myeloma (CASSIOPEIA): a randomised, open-label, phase 3 study. Lancet. 2019;394(10192):29–38.
King T, Jagger J, Wood J, Woodrow C, Snowden A, Haines S, et al. Best practice for the administration of daratumumab in multiple myeloma: Australian myeloma nurse expert opinion. Asia Pac J Oncol Nurs. 2018;5(3):270–84.
Palumbo A, Chanan-Khan A, Weisel K, Nooka AK, Masszi T, Beksac M, et al. Daratumumab, bortezomib, and dexamethasone for multiple myeloma. N Engl J Med. 2016;375(8):754–66.
Nooka AK, Gleason C, Sargeant MO, Walker M, Watson M, Panjic EH, Lonial S. Managing infusion reactions to new monoclonal antibodies in multiple myeloma: daratumumab and elotuzumab. J Oncol Pract. 2018;14(7):414–22.
Davis JA, Youngberg H, Gaffney K, Duco M, Hashmi H. ‘Fast but not so Furious’: short observation time after subcutaneous Daratumumab administration is both a safe and cost-effective strategy. Clin Lymphoma Myeloma Leuk. 2022;22(8):e680–4.
Bookbinder LH, Hofer A, Haller MF, Zepeda ML, Keller GA, Lim JE, et al. A recombinant human enzyme for enhanced interstitial transport of therapeutics. J Control Rel. 2006;114(2):230–41.
Usmani SZ, Nahi H, Mateos MV, van de Donk NWCJ, Chari A, Kaufman JL, et al. Subcutaneous delivery of daratumumab in relapsed or refractory multiple myeloma. Blood. 2019;134(8):668–77.
Chari A, Rodriguez-Otero P, McCarthy H, Suzuki K, Hungria V, Sureda Balari A, et al. Subcutaneous daratumumab plus standard treatment regimens in patients with multiple myeloma across lines of therapy (PLEIADES): an open-label Phase II study. Br J Haematol. 2021;192(5):869–78.
Usmani SZ, Nahi H, Legiec W, Grosicki S, Vorobyev V, Spicka I, et al. Final analysis of the phase III non-inferiority COLUMBA study of subcutaneous versus intravenous daratumumab in patients with relapsed or refractory multiple myeloma. Haematologica. 2022;107(10):2408–17.
Mateos MV, Nahi H, Legiec W, Grosicki S, Vorobyev V, Spicka I, et al. Subcutaneous versus intravenous daratumumab in patients with relapsed or refractory multiple myeloma (COLUMBA): a multicentre, open-label, non-inferiority, randomised, phase 3 trial. Lancet Haematol. 2020;7(5):e370–80.
Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014;15(12):e538–48.
Koh Y. AL amyloidosis: advances in diagnosis and management. Blood Res. 2020;55(S1):S54–7.
Delforge M, Ludwig H. How I manage the toxicities of myeloma drugs. Blood. 2017;129(17):2359–67.
Voorhees PM, Kaufman JL, Laubach J, Sborov DW, Reeves B, Rodriguez C, et al. Daratumumab, lenalidomide, bortezomib, and dexamethasone for transplant-eligible newly diagnosed multiple myeloma: the GRIFFIN trial. Blood. 2020;136(8):936–45.
Moore DC, Arnall JR, Thompson DL, Martin AL, Robinson J, Ndiaye A, et al. Evaluation of montelukast for the prevention of infusion-related reactions with daratumumab. Clin Lymphoma Myeloma Leuk. 2020;20(10):e777–81.
Acknowledgements
This research was supported by the Intramural Program of the Department of Medicine, Surgery and Dentistry, University of Salerno, Italy.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
Open access funding provided by Università degli Studi di Salerno within the CRUI-CARE Agreement. No external funding was used in the preparation of this manuscript.
Conflicts of interest
Danilo De Novellis, Raffaele Fontana, Salvatore Palmieri, Roberta Della Pepa, Maria Di Perna, Giusy Cetani, Daniela Esposito, Angela Amendola, Giuseppe Delle Cave, Bianca Serio, Denise Morini, Michela Rizzo, Laura Mettivier, Fabio Trastulli, Stefano Rocco, Anastasia Pagano, Serafina Barbato, Aldo Leone, Martina La Magna, Rosario Bianco, Gabriella Rascato, Angela Carobene, Bianca Cuffa, Marialuigia Iannalfo, Valentina Giudice, Gino Svanera, Mario Annunziata, Michele Pizzuti, Ferdinando Frigeri, Catello Califano, Felicetto Ferrara, Fabrizio Pane, and Carmine Selleri declare that they have no conflicts of interest that might be relevant to the contents of this manuscript.
Ethical approval
The study protocol was approved by the local ethic committee (Ethics Committee “Campania Sud,” Brusciano, Naples, Italy; prot./SCCE n. 24988).
Consent to participate
Informed consent was obtained in accordance with the Declaration of Helsinki (World Medical Association 2013) and protocols were approved by the local ethic committee (Ethics Committee “Campania Sud,” Brusciano, Naples, Italy; prot./SCCE n. 24988).
Conenst to publish
Not applicable.
Data availability statement
Data are available upon request of the authors.
Statements
The authors declare that the material is original, has not been published before, nor is under consideration in any other journal.
Author Contributions
Conceptualization: D.D.N., B.S., and C.S.; clinical data: D.D.N., R.F., S.P., R.D.P., M.D.P., G.C., D.E., A.A., G.D.C., B.S., D.M., M.R., L.M., F.T., S.R., A.P., S.B., A.L., M.L.M., R.B., G.R., A.C., B.C., M.I., V.G., G.V., M.A., M.P., F.Fr., C.C., F.P., F.Fe., and C.S.; methodology: D.D.N. and V.G.; writing—original draft preparation: D.D.N. and V.G.; writing—review and editing: C.S. All authors have read and agreed to the published version of the manuscript.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
De Novellis, D., Fontana, R., Palmieri, S. et al. Safety of Subcutaneous Daratumumab in Anti-CD38 Monoclonal Antibody-Naïve Patients with Plasma Cell Disorders: A Multicenter Real-Life Experience. Targ Oncol 18, 885–892 (2023). https://doi.org/10.1007/s11523-023-01001-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11523-023-01001-4