Abstract
Purpose
Acute ischemic stroke (AIS) due to tandem lesions (TLs) of extracranial Internal Carotid Artery and Anterior Cerebral Circulation are challenging for endovascular treatment (EVT). This study aims to evaluate feasibility, safety and efficacy of EVT for TLs’ AIS, with or without emergent carotid artery stenting (eCAS), in a real-life scenario.
Methods
Retrospective review of prospectively collected non-randomized thrombectomy databases from five stroke centers between 2015 and 2019. Consecutive patients with TLs’ AIS were selected. Clinical, neuroimage and procedure features, as well as antiplatelet therapy regimen, were evaluated. Primary outcome was 90-day mRS ≤ 2. Secondary outcomes included: mTICI score 2b-3, extracranial recanalization, procedural complications, symptomatic intracerebral hemorrhage (SICH) and 90-day mortality.
Results
Two hundred twenty-seven patients were enrolled (67.8% males; mean age 65.9 ± 12.9 years). We obtained mTICI 2b-3 in 77.1%, extracranial recanalization in 86.8%, 90-day mRS (available in 201/227 cases) ≤ 2 in 49.8%. Procedural complications occurred in 16.7%, SICH in 9.7%; 90-day mortality rate (available in 201/227 cases) was 14.4%. The strongest predictors of good clinical outcome were young age (p < 0.0001), low baseline NIHSS (p = 0.008), high baseline ASPECTS (p < 0.0001), good collateral flow (p = 0.013) and extracranial recanalization (p = 0.001). The most significant predictors of SICH were low baseline ASPECTS (p < 0.0001), occurrence of complications (p < 0.0001) and eCAS (p = 0.002).
Conclusion
In our real-life series, the EVT for TLs’ AIS was feasible, safe and effective in improving 90-day functional outcome with acceptable morbi-mortality rates. ECAS increased the risk of SICH, independently from the antiplatelet therapy regimen.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Since 2015, many trials [1,2,3,4,5] assessed the superiority of mechanical thrombectomy in the treatment of acute ischemic stroke (AIS) in comparison with standard intravenous fibrinolysis (IV-FL). The same cannot be said for tandem lesions (TLs) of extracranial Internal Carotid Artery (ICA) and Anterior Cerebral Circulation [6], representing about 15% of AIS [7].
TLs consist in the presence of occlusion, sub-occlusion or stenosis of extracranial ICA, due to dissection or atheromatous plaque, together with simultaneous intracranial large vessel occlusion [7,8,9,10,11].
They show both poor responsiveness to IV-FL [12] and technical difficulties for endovascular treatment (EVT), resulting in low recanalization rates and more unfavorable outcomes [10]. Moreover, there is a lack of current guidelines about their management [13].
The aim of this study is to assess feasibility, safety and efficacy of EVT for AIS due to TLs. A comprehensive overview of these complex endovascular situations will also be provided, particularly focusing on the causal link between extracranial and intracranial lesions which constitute TLs.
Methods
Patient selection, variables and outcomes
This retrospective study was approved by the institutional review board at our hospital. The requirement for written informed consent was waived because of the retrospective study design. Prospectively collected non-randomized thrombectomy databases from five major Italian stroke centers were retrospectively reviewed. All the consecutive patients with anterior circulation AIS due to TLs who underwent EVT between 01 January 2015 and 31 August 2019 were enrolled.
Angiography inclusion criteria were:
-
(1)
Extracranial ICA occlusion or sub-occlusion, due to dissection or atheromatous plaque, and simultaneous intracranial vessel occlusion (IVO);
-
(2)
Extracranial ICA stenosis, due to unstable plaque, ≥ 80% NASCET (North American Symptomatic Carotid Endarterectomy Trial) [14] and simultaneous IVO;
-
(3)
Extracranial ICA occlusion or sub-occlusion without IVO but with insufficient Willis compensation (defined as “hemodynamic TLs” by the Authors).
Exclusion criteria were:
-
(1)
Absence of TLs;
-
(2)
Extracranial ICA occlusion or sub-occlusion and simultaneous IVO by another cause, e.g., cardiogenic embolism;
-
(3)
Extracranial ICA stenosis, due to stable plaque, ≤ 80% NASCET and simultaneous IVO, being the two lesions not apparently linked;
-
(4)
Pre-stroke modified Rankin Scale (mRS) score > 2;
-
(5)
Age < 18 years.
Patients’ relevant data were collected. Clinical features included: gender, age, risk factors (hypertension, diabetes mellitus, dyslipidemia and smoking habit), baseline National Institutes of Health Stroke Scale (NIHSS) and administration of IV-FL. Neuroimaging features were: baseline Alberta Stroke Program Early CT Score (ASPECTS), intracranial lesion site (ICA not T-type, ICA T-type, M1-Middle Cerebral Artery [MCA], M2 or distal-MCA, “hemodynamic TLs”), etiology (plaque or dissection) and type (occlusion, sub-occlusion or stenosis) of extracranial lesion and collateral flow status, assessed with the ASITN/SIR (American Society of Interventional and Therapeutic Neuroradiology/Society of Interventional Radiology) grading system [15] on Digital Subtraction Angiography (DSA) and subsequently classified as "good" (ASITN/SIR 3–4), "moderate" (ASITN/SIR 2) or "poor" (ASITN/SIR 0–1).
Procedural features included: type of anesthesia (general anesthesia or conscious sedation, according to local guidelines), intracranial thrombectomy technique (Thromboaspiration Alone [TA] or Stent-retriever Combined Thrombectomy [SCoT]), emergent carotid artery stenting (eCAS) performed in anterograde (i.e., treatment of the proximal lesion first) or retrograde (i.e., treatment of the distal lesion first) fashion, super selective administration of adjunct antiplatelet therapy during the procedure (Acetylsalicylic Acid [ASA], P2Y12 inhibitors or Glycoprotein IIb/IIIa [GpIIb/IIIa] inhibitors), time from Onset To Groin puncture (OTG) and from Groin puncture To Reperfusion (GTR).
After the procedure, all the patients were admitted to a neuro-dedicated Intensive Care Unit or to a Stroke Unit, under a strict blood pressure control.
The primary outcome was functional independence (mRS ≤ 2) at 90-day follow-up. Secondary outcomes were good intracranial reperfusion (modified Thrombolysis In Cerebral Infarction [mTICI] score 2b-3) and extracranial ICA recanalization.
Data regarding complications, such as embolization of new territories and iatrogenic dissection or perforation (recognized at the time of treatment) and symptomatic intracerebral hemorrhage (SICH), were also collected. SICH was defined, according to the SWIFT PRIME trial [4], as any parenchymal hematoma, intracerebral, subarachnoid or intraventricular hemorrhage causing a decline of four or more points in the NIHSS within 24 h from the end of the revascularization procedure.
Neuroimaging data (CT and DSA) were consensually reviewed by an experienced interventional neuroradiologist and a senior radiology resident with experience in interventional neuroradiology.
Statistical analysis
Patients data were collected in a single database. All data were validated and then submitted to usual descriptive techniques. Continuous variables were described either as mean and standard deviation (SD) or as median and interquartile range (IQR), depending on their distribution, which was checked using the Shapiro–Wilk test. Categorical variables (nominal and ordinal) were provided as relative and absolute frequency tables.
Comparisons between continuous variables were performed using either the Student’s t test or the Mann–Whitney U test, depending on their distribution and on the variance homogeneity (previously evaluated through the Levene’s test). The Fisher’s exact test was used for comparison between categorical variables.
The strongest predictors of 90-day functional independence and of SICH were identified using the multivariate logistic regression with the likelihood ratio test for the model as a whole and through the Wald’s test for each regressor. At the beginning, the multivariable regression included the regressors having p < 0.2 at univariate analysis, which were subsequently excluded in a stepwise fashion, in order to get the best BIC and McFadden R2 values.
Statistical analyses were performed using Stata/SE 12.0 (The Statacorp, College Station, USA). All calculated p-values were two-sided, and statistical significance was assumed for p < 0.05.
Results
During the study period, 1482 patients with anterior circulation AIS underwent EVT, with 227 of them having a TL. Over the 227 patients, 26 were lost at 90-day follow-up and therefore 90-day mRS and mortality were available for 201/227 (88.6%) patients, while intra-procedural and post-procedural data were available for all 227 patients.
Clinical, neuroimage and procedure data are shown in Table 1, while the results of statistical analysis for functional independence (mRS ≤ 2), technical outcomes (mTICI 2b-3 and extracranial ICA recanalization) and SICH are reported in Table 2, 3 and 4, respectively.
The functional independence was achieved in 100/201 (49.8%) cases. Patients who performed EVT under general anesthesia showed lower functional independence rates (58.8% vs 41.4%; p = 0.017) and higher median baseline NIHSS (median, 18.0; IQR, 13.8–21.3 vs. median, 15.0; IQR, 10.0–18.0; p = 0.0002) compared to conscious sedations.
A 2b-3 mTICI score was observed in 175/227 (77.1%) cases. Mechanical thrombectomy was not performed in 50/227 (22.0%) patients: IVO was more distal than M3-MCA division in three cases, extracranial stenosis was too tight to be crossed in 15 and a “hemodynamic TL” was present in 32.
Among the 146 patients receiving both IV-FL and EVT, no statistical differences in terms of good cerebral reperfusion (78.1% vs. 75.3%; p = 0.625) and of functional independence (51.5% vs. 47.8%; p = 0.656) were found compared to the 81 cases not undergoing IV-FL.
A carotid stent was positioned in 132/227 (58.1%) cases. Extracranial ICA recanalization was obtained in 197/227 (86.8%) patients, with 126 of them undergoing eCAS (therefore, in 6/132 eCAS patients extracranial ICA was not recanalized). For the other patients, recanalization was achieved through percutaneous transluminal angioplasty alone. Furthermore, extracranial recanalization was achieved in 27/32 “hemodynamic TLs” (in 23 patients through eCAS while via angioplasty in 4).
Among the 132/227 (58.1%) eCAS patients (105/172 atheromasic plaques, 61.0% vs. 27/55 dissections, 49.1%; p = 0.157), in 67 (50.8%) cases the stent was positioned with an anterograde approach (23 “hemodynamic TLs”), while in 65 (49.2%) via a retrograde one.
With regard to the etiopathogenesis of the extracranial lesion, atheromasic patients were older compared to those with dissection (mean, 70.6 years; SD, 9.7 vs. mean, 51.3 years; SD, 10.7; p < 0.00001). However, independently from the etiology, both plaque and dissection patients achieved similar extracranial recanalization rates (88.4% vs. 81.8%, p = 0.252).
ECAS was significantly associated with extracranial recanalization (95.5% vs. 74.7%; p < 0.00001), good cerebral reperfusion (86.4% vs. 64.2%; p = 0.0001) and higher SICH rates (15.2% vs 2.1%; p = 0.001) but not with 90-day functional improvement (54.3% vs. 43.5%; p = 0.154).
However, considering only patients without SICH, those who underwent eCAS achieved 90-day functional independence more frequently (65.6% vs. 44.6%; p = 0.007).
Furthermore, among the 132 patients undergoing eCAS, the median baseline ASPECTS was significantly lower in the 20 ones who developed SICH (median, 8.0; IQR, 7.8–9.0 vs. median, 9.0; IQR, 8.0–10.0; p = 0.009).
Super selective antiplatelet therapy was administered in 130/227 (57.3%) procedures, in particular ASA in 63 (27.8%), P2Y12 inhibitor in 8 (3.5%) and GpIIb/IIIa inhibitor in 59 (26.0%): 55, 8 and 56 of these patients, respectively, underwent eCAS.
Procedural complications occurred in 38/227 (16.7%, 10.1% embolization of new territories and 6.6% iatrogenic perforation or dissection), more frequently when using a stent-retriever (15.7% vs. 30.4%; p = 0.025). SICH was observed in 22/227 (9.7%) patients, none of them having a “hemodynamic TL.” Rate of 90-day mortality was 29/201 (14.4%), with 15 cases of stroke-related death AIS (14 of them from SICH).
With reference to the timings recorded, no statistical association was found between OTG and the outcomes considered. On the contrary, patients achieving 90-day functional independence showed shorter GTR (mean, 70.3; SD, 43.3 vs. mean, 84.7; SD, 42.8; p = 0.022).
At the multivariate analysis, the strongest predictors of good clinical outcome were: young age (mean, 62.5; SD, 12.8 vs. mean, 69.5; SD, 12.3; OR, 0.94; 95% CI, 0.91–0.97; p > 0.0001), low median baseline NIHSS (median, 15.0; IQR, 10.0–18.0 vs. median, 18.0; IQR, 15.0–22.0; OR, 0.93; 95% CI, 0.88–0.98; p = 0.008), high baseline ASPECTS (median, 9.0; IQR, 8.0–10.0 vs. median, 8.0; IQR, 7.0–9.0; OR, 1.65; 95% CI, 1.33–2.06; p < 0.0001), extracranial ICA recanalization (53.5% vs. 46.5%; OR, 6.05; 95% CI, 2.03–18.06; p = 0.001), good collateral flow (62.9% vs. 37.1%; OR, 0.57; 95% CI, 0.37–0.87; p = 0.013) and absence of procedural complications (32.3% vs. 67.7%; OR, 0.27; 95% CI, 0.10–0.72; p = 0.009).
Conversely, male gender was strongly associated with poor functional outcome (54% vs. 46%; OR, 2.46; 95% CI, 1.15–5.26; p = 0.020).
SICH predictors were: low baseline ASPECTS (median, 9.0; IQR, 8.0–10.0 vs. median, 8.0; IQR, 7.3–9.0; OR, 0.60; 95% CI, 0.51–0.70; p < 0.0001), occurrence of complications (73.7% vs. 26.3%; OR, 6.27; 95% CI, 2.24–17.54; p < 0.0001) and carotid stent placement (84.8% vs. 15.2%; OR, 8.18; 95% CI, 2.15–31.12; p = 0.002).
Discussion
In our experience, TLs prevalence was 15.3% of all the consecutive stroke cases treated endovascularly in the time frame considered (n = 227/1482). This data is consistent with those of the ESCAPE and REVASCAT trials [2, 5], which reported TLs rates of 12.7% and 15.8%, respectively.
Currently, no recommendations or guidelines regarding TLs management are available [13] and therefore the choice about whether and how to treat is left to the single-center experience.
This paper provides a real-life view on these challenging situations and identifies predictors of good technical and clinical outcome.
Furthermore, it is our opinion that current definition of TL as simultaneous occlusion of the extracranial ICA together with an anterior intracranial large vessel [7,8,9,10,11] presents several limitations. First, it does not stress the causal link between extracranial and intracranial lesions; second, it does not consider small vessels occlusions, although they could cause serious clinical consequences [16]; third, it excludes other injurious mechanisms, different from occlusion, which could cause severe neurological symptoms, as hemodynamic impairment.
For these reasons, according to the authors, a TL should be defined as the “simultaneous presence of two lesions on the same vascular axis, being the extracranial lesion, either an ICA plaque or dissection, the actual cause of the intracranial one, either for thromboembolism or for hemodynamic impairment.”
In the following paragraphs, we will discuss the variables that correlated the most with the outcomes considered.
Clinical features
At 90-day follow-up, 49.8% of the patients achieved functional independence, similar to what observed by Gory et Al. [17]. The strongest predictors of good clinical outcome were young age, low baseline NIHSS, high baseline ASPECTS, good collateral flow and extracranial ICA recanalization (Table 2), as reported by other studies [18,19,20].
Extracranial lesion etiology
The 90-day functional independence rate was significantly lower in patients with carotid plaques compared to those with dissections. Although atherosclerosis might be associated with better clinical outcome, being a slow-growing disease that allows the development of collateral circulation [17], our result could be interpreted by considering the older age of plaque patients, who therefore had a lower functional reserve compared to those with dissection.
Nevertheless, both plaques and dissections achieved similar rates of extracranial recanalization, suggesting that extracranial lesion etiology should not condition the decision of performing EVT, as already suggested by Gory et al. [17]
In addition, no differences were found regarding stent placement between plaque and dissection patients, meaning that the necessity of performing eCAS did not correlate with extracranial lesion etiology.
Procedural, pharmacological and technical features
Today, no consensus exists about optimal anesthetic strategy during EVT for AIS [21, 22].
In our TLs series, conscious sedation was significantly associated with higher 90-day functional independence rates at univariate analysis: this result is out of line with SIESTA, AnStroke and GOLIATH trials [23,24,25]. However, patients receiving general anesthesia had higher median baseline NIHSS scores and thus more severe AIS, compared to those receiving conscious sedation.
Furthermore, patients who received IV-FL did not achieve higher rates of good cerebral reperfusion and functional independence, confirming that TLs are poorly responsive to IV-FL, independently from the etiology [12]. This result is in favor with ongoing randomized controlled trials which hypothesize the non-inferiority of mechanical thrombectomy compared to bridging therapy (NCT03192332; ISRCTN80619088; NCT03469206; NCT 03,494,920).
Mechanical thrombectomy
Regarding intracranial thrombectomy technique, no significant differences in terms of good cerebral reperfusion (Table 3) and of good clinical outcome (Table 2) were found between TA and SCoT, even if a stent-retriever was used less frequently. These results are in line with recent studies [26, 27]. However, in the present study, the use of a stent-retriever resulted in significantly higher rates of procedural complications.
Emergent carotid artery stenting
Today, there are no standardized guidelines regarding eCAS in literature [8].
In our series, eCAS was significantly associated with extracranial recanalization and good cerebral reperfusion. Therefore, by restoring an adequate anterograde flow, eCAS could be a key factor in achieving a good intracranial reperfusion, especially when hemodynamic impairment occurs (Fig. 1).
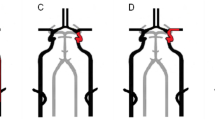
Patient with aphasia, right upper limb hyposthenia and paresthesia (NIHSS 9). Computed tomography angiography, performed at the Spoke Hospital, revealed left M1-MCA sub-occlusion (a, arrow). Patient was centralized for thrombectomy to the Hub Hospital immediately after administration of IV-FL. Digital subtraction angiography revealed a complete occlusion of the left extracranial ICA (b), with normal opacification of the previously sub-occluded vessel via retrograde anastomotic filling from the ipsilateral ophthalmic artery (c). For these reasons, intracranial thrombectomy was not carried out. Since hemodynamic impairment was observed, as poor activation of Willis’ circle (d), it was decided to perform eCAS (e). Post-procedure angiography showed good cerebral reperfusion (f, g), mTICI 2b. 90-day mRS 0
Nevertheless, stenting was not significantly associated with 90-day functional improvement, as also reported by Zhu et Al. in their analysis adjusted for confounders [7]. This could depend on the close association between eCAS and SICH: if only patients without SICH are considered, those who underwent eCAS achieved higher 90-day functional independence rates.
Symptomatic intracerebral hemorrhage (SICH)
In our series, SICH occurred in 9.7% of the total cases, and this is in line with the 10% cutoff value of SICH following EVT, established as a standard of practice by the 2018 international multi-society guidelines [28].
Unfortunately, no patient with SICH achieved 90-day functional independence (Table 2). As expected, SICH was strongly associated with low baseline ASPECTS, procedural complications and carotid stent placement, independently from the antiplatelet drug regimen and from IV-FL administration (Table 4).
ECAS and SICH are generally linked by the so-called “hyperperfusion syndrome,” mostly in atheromasic patients [29, 30]. Interestingly, we found no statistical differences in terms of SICH between plaques and dissections.
Furthermore, considering only patients who underwent eCAS, SICH was more frequently observed in cases of low baseline ASPECTS, pointing out that a careful patient preselection based on the extent of the ischemic core, under strict blood pressure control, might reduce the risk of SICH [31, 32].
Antiplatelet therapy, as well as IV-FL, is usually thought to be associated with higher SICH risk [33, 34]. Although in our series the choice of how, how much and which drug to administer to each patient was left to the single-center operator expertise, both the use of antiplatelet therapy or of IV-FL did not increase SICH risk, as also reported by Zhu et Al. [34]. Therefore, our result is in favor of using antiplatelet drugs and, in accordance with the current literature, preferably with greater, faster and shorter antiplatelet activity [35].
Limitations and strengths
We are aware that this study has several limitations: first of all, its retrospective, uncontrolled and observational design. Its multicenter design allowed a broader patients recruitment, but it also resulted in higher heterogeneity regarding techniques, devices and patient selection for EVT in each participating center. Furthermore, uniform imaging assessment of stent patency was not evaluated at late follow-up, although pre/post and procedural neuroimaging data were consensually reviewed by two expert readers. Additionally, in our analysis we also included the “hemodynamic TLs,” a group that is often not considered by other authors, despite the same hemodynamic consequences. Further studies considering this particular type of TLs separately and involving a greater number of cases are needed to confirm our results.
However, our analysis has a fundamental strength: as opposite to randomized controlled trials, which consider patients selected under ideal conditions, in our series patients are representative of those normally observed in a real-life setting, not enrolled according to severe inclusive criteria.
Conclusion
This is the first retrospective real-life case series of TLs patients undergoing EVT which also includes the “hemodynamic TL” (here defined as extracranial ICA occlusion or sub-occlusion being the actual cause of AIS for insufficient Willis compensation). In our series, this group showed no significant differences, in terms of 90-day functional independence and technical success rates, compared to “thromboembolic TLs.” However, no patient with “hemodynamic TLs” developed SICH.
High rates of good cerebral reperfusion (77.1%) and of extracranial recanalization (86.8%) were achieved, with consequent significant improvement in 90-day functional outcome, demonstrating that in these complex situations EVT is technically feasible, effective and safe. ECAS was found to correlate with the risk of SICH, unlike antiplatelet therapy and IV-FL. Anyhow, in our series morbidity and mortality rates did not result higher compared to the current literature.
AIS due to TLs still remain challenging for stroke physicians: more data and new trials are needed to better understand how to optimize the reperfusion strategies in this multifactorial, multidistrict, often dramatic vascular pathology.
References
Berkhemer OA, Fransen PSS, Beumer D, van den Berg LA, Lingsma HF, Yoo AJ et al (2015) A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med 372(1):11–20. https://doi.org/10.1056/NEJMoa1411587
Goyal M, Demchuk AM, Menon BK, Eesa M, Rempel JL, Thornton J et al (2015) Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med 372(11):1019–1030. https://doi.org/10.1056/NEJMoa1414905
Campbell BCV, Mitchell PJ, Kleinig TJ, Dewey HM, Churilov L, Yassi N et al (2015) (2015) Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 372(11):1009–1018. https://doi.org/10.1056/NEJMoa1414792
Saver JL, Goyal M, Bonafe A, Diener H-C, Levy EI, Pereira VM, et al. (2015) Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med 372(24):2285–95. https://doi.org/https://doi.org/10.1056/NEJMoa1415061
Jovin TG, Chamorro A, Cobo E, de Miquel MA, Molina CA, Rovira A et al (2015) Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med 372(24):2296–2306. https://doi.org/10.1056/NEJMoa1503780
Sivan-Hoffmann R, Gory B, Armoiry X, Goyal M, Riva R, Labeyrie PE et al (2017) Stent-retriever thrombectomy for acute anterior ischemic stroke with tandem occlusion: a systematic review and meta-analysis. Eur Radiol 27(1):247–254. https://doi.org/10.1007/s00330-016-4338-y
Zhu F, Bracard S, Anxionnat R, Derelle A-L, Tonnelet R, Liao L et al (2019) Impact of emergent cervical carotid stenting in tandem occlusion strokes treated by thrombectomy: a review of the TITAN Collaboration. Front Neurol. https://doi.org/10.3389/fneur.2019.00206
Jadhav AP, Zaidat OO, Liebeskind DS, Yavagal DR, Haussen DC, Hellinger FR et al (2019) Emergent management of tandem lesions in acute ischemic stroke. Stroke 50(2):428–433. https://doi.org/10.1161/STROKEAHA.118.021893
Wilson MP, Murad MH, Krings T, Pereira VM, O’Kelly C, Rempel J et al (2018) Management of tandem occlusions in acute ischemic stroke - intracranial versus extracranial first and extracranial stenting versus angioplasty alone: a systematic review and meta-analysis. J Neurointerv Surg 10(8):721–728. https://doi.org/10.1136/neurintsurg-2017-013707
Rubiera M, Ribo M, Delgado-Mederos R, Santamarina E, Delgado P, Montaner J et al (2006) Tandem internal carotid artery/middle cerebral artery occlusion. Stroke 37(9):2301–2305. https://doi.org/10.1161/01.STR.0000237070.80133.1d
Assis Z, Menon BK, Goyal M, Demchuk AM, Shankar J, Rempel JL et al (2018) Acute ischemic stroke with tandem lesions: technical endovascular management and clinical outcomes from the ESCAPE trial. J Neurointerv Surg 10(5):429–433. https://doi.org/10.1136/neurintsurg-2017-013316
Kim YS, Garami Z, Mikulik R, Molina CA, Alexandrov AV (2005) Early recanalization rates and clinical outcomes in patients with tandem internal carotid artery/middle cerebral artery occlusion and isolated middle cerebral artery occlusion. Stroke 36(4):869–871. https://doi.org/10.1161/01.STR.0000160007.57787.4c
Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, et al. (2018) 2018 Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the american heart association/American stroke association. stroke 49(3):e46–110. https://doi.org/https://doi.org/10.1161/STR.0000000000000158
Ferguson GG, Eliasziw M, Barr HWK, Clagett GP, Barnes RW, Wallace MC et al (1999) The North American symptomatic carotid endarterectomy trial. Stroke 30(9):1751–1758. https://doi.org/10.1161/01.STR.30.9.1751
Higashida RT, Furlan AJ, Roberts H, Tomsick T, Connors B, Barr J et al (2003) Trial design and reporting standards for intra-arterial cerebral thrombolysis for acute ischemic stroke. Stroke 34(8):e109–e137. https://doi.org/10.1161/01.STR.0000082721.62796.09
Grossberg JA, Rebello LC, Haussen DC, Bouslama M, Bowen M, Barreira CM et al (2018) Beyond large vessel occlusion strokes. Stroke 49(7):1662–1668. https://doi.org/10.1161/STROKEAHA.118.020567
Gory B, Piotin M, Haussen DC, Steglich-Arnholm H, Holtmannspötter M, Labreuche J et al (2017) Thrombectomy in acute stroke with tandem occlusions from dissection versus atherosclerotic cause. Stroke 48(11):3145–3148. https://doi.org/10.1161/STROKEAHA.117.018264
Grigoryan M, Haussen DC, Hassan AE, Lima A, Grossberg J, Rebello LC et al (2016) Endovascular treatment of acute ischemic stroke due to tandem occlusions: large multicenter series and systematic review. Cerebrovasc Dis 41(5–6):306–312. https://doi.org/10.1159/000444069
Gory B, Haussen DC, Piotin M, Steglich-Arnholm H, Holtmannspötter M, Labreuche J et al (2018) Impact of intravenous thrombolysis and emergent carotid stenting on reperfusion and clinical outcomes in patients with acute stroke with tandem lesion treated with thrombectomy: a collaborative pooled analysis. Eur J Neurol 25(9):1115–1120. https://doi.org/10.1111/ene.13633
Papanagiotou P, Haussen DC, Turjman F, Labreuche J, Piotin M, Kastrup A et al (2018) Carotid stenting with antithrombotic agents and intracranial thrombectomy leads to the highest recanalization rate in patients with acute stroke with tandem lesions. JACC Cardiovasc Interv 11(13):1290–1299. https://doi.org/10.1016/j.jcin.2018.05.036
Cappellari M, Pracucci G, Forlivesi S, Saia V, Nappini S, Nencini P et al (2020) General anesthesia versus conscious sedation and local anesthesia during thrombectomy for acute ischemic stroke. Stroke 51:2036–2044. https://doi.org/10.1161/STROKEAHA.120.028963
Campbell D, Diprose WK, Deng C, Barber PA (2019) General Anesthesia Versus Conscious Sedation in Endovascular Thrombectomy for Stroke. J Neurosurg Anesthesiol . https://doi.org/10.1097/ANA.0000000000000646
Schönenberger S, Uhlmann L, Hacke W, Schieber S, Mundiyanapurath S, Purrucker JC et al (2016) Effect of conscious sedation vs general anesthesia on early neurological improvement among patients with ischemic stroke undergoing endovascular thrombectomy: A randomized clinical trial. JAMA - J Am Med Assoc 316(19):1986–1996. https://doi.org/10.1001/jama.2016.16623
Hendén PL, Rentzos A, Karlsson J-E, Rosengren L, Leiram B, Sundeman H et al (2017) General Anesthesia Versus Conscious Sedation for Endovascular Treatment of Acute Ischemic Stroke. Stroke 48(6):1601–1607. https://doi.org/10.1161/STROKEAHA.117.016554
Simonsen CZ, Yoo AJ, Sørensen LH, Juul N, Johnsen SP, Andersen G et al (2018) Effect of general anesthesia and conscious sedation during endovascular therapy on infarct growth and clinical outcomes in acute ischemic stroke a randomized clinical trial. JAMA Neurol 75(4):470–477. https://doi.org/10.1001/jamaneurol.2017.4474
Lapergue B, Blanc R, Gory B, Labreuche J, Duhamel A, Marnat G et al (2017) Effect of Endovascular Contact Aspiration vs Stent Retriever on Revascularization in Patients With Acute Ischemic Stroke and Large Vessel Occlusion. JAMA 318(5):443. https://doi.org/10.1001/jama.2017.9644
Romano DG, Frauenfelder G, Casseri T, Piano M, Vinci S, Comai A et al (2019) Efficacy of ADAPT with large-bore reperfusion catheter in anterior circulation acute ischemic stroke: a multicentric Italian experience. Radiol Med 125(1):57–65. https://doi.org/10.1007/s11547-019-01069-x
Sacks D, Baxter B, Campbell BC, Carpenter JS, Cognard C, Dippel D et al (2018) STANDARDS OF PRACTICE Multisociety Consensus Quality Improvement Revised Consensus Statement for Endovascular Therapy of Acute Ischemic Stroke. J Vasc Interv Radiol 29:441–453. https://doi.org/10.1016/j.jvir.2017.11.026
Kirchoff-Torres KF, Bakradze E (2018) Cerebral Hyperperfusion Syndrome After Carotid Revascularization and Acute Ischemic Stroke. Curr Pain Headache Rep 22(4):24. https://doi.org/10.1007/s11916-018-0678-4
Yan Z, Yang M, Niu G, Zou Y (2017) Analysis of Hemodynamic Changes in Early Stage after Carotid Stenting by Transcranial Doppler-A Preliminary Study. Ann Vasc Surg 45:85–91. https://doi.org/10.1016/j.avsg.2017.06.124
Yen-Heng L, Hon-Man L (2020) Update on Cerebral Hyperperfusion Syndrome. J Neurointerv Surg 12(8):788–793. https://doi.org/10.1136/NEURINTSURG-2019-015621
Cappellari M, Mangiafico S, Saia V, Pracucci G, Nappini S, Nencini P et al (2019) IER-SICH Nomogram to Predict Symptomatic Intracerebral Hemorrhage After Thrombectomy for Stroke. Stroke 50(4):909–916. https://doi.org/10.1161/STROKEAHA.118.023316
Pieraccini M, Guerrini S, Laiolo E, Puliti A, Roviello G, Misuraca L et al (2018) Acute Massive and Submassive Pulmonary Embolism: Preliminary Validation of Aspiration Mechanical Thrombectomy in Patients with Contraindications to Thrombolysis. Cardiovasc Intervent Radiol 41:1840–1848. https://doi.org/10.1007/s00270-018-2011-3
Zhu F, Anadani M, Labreuche J, Spiotta A, Turjman F, Piotin M et al (2020) Impact of Antiplatelet Therapy during Endovascular Therapy for Tandem Occlusions: A Collaborative Pooled Analysis. Stroke 51:1522–1529. https://doi.org/10.1161/STROKEAHA.119.028231
Kang DH, Yoon W, Kim SK, Baek BH, Lee YY, Kim YW et al (2019) Endovascular treatment for emergent large vessel occlusion due to severe intracranial atherosclerotic stenosis. J Neurosurg 1306:1949–1956. https://doi.org/10.3171/2018.1.JNS172350
Funding
Open Access funding provided by Università degli Studi di Siena. The authors received no funding for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This retrospective study was approved by the institutional board at our hospital. The requirement for written informed consent was waived because of the retrospective study design.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bracco, S., Zanoni, M., Casseri, T. et al. Endovascular treatment of acute ischemic stroke due to tandem lesions of the anterior cerebral circulation: a multicentric Italian observational study. Radiol med 126, 804–817 (2021). https://doi.org/10.1007/s11547-020-01331-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-020-01331-7