Summary
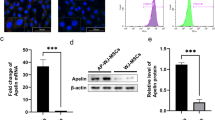
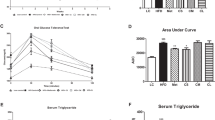
Insulin resistance is an essential characteristic of type 2 diabetes mellitus (T2DM), which can be induced by glucotoxicity and adipose chronic inflammation. Mesenchymal stem cells (MSCs) and their exosomes were reported to ameliorate T2DM and its complications by their immunoregulatory and healing abilities. Exosomes derived from MSCs contain abundant molecules to mediate crosstalk between cells and mimic biological function of MSCs. But the role of exosomes derived from human umbilical cord mesenchymal stem cells (hUC-MSCs) in insulin resistance of human adipocytes is unclear. In this study, exosomes were harvested from the conditioned medium of hUC-MSCs and added to insulin-resistant adipocytes. Insulin-stimulated glucose uptake was measured by glucose oxidase/peroxidase assay. The signal pathway involved in exosome-treated adipocytes was detected by RT-PCR and Western blotting. The biological characteristics and function were compared between hUC-MSCs and human adipose-derived mesenchymal stem cells (hAMSCs). The results showed that hAMSCs had better adipogenic ability than hUC-MSCs. After induction of mature adipocytes by adipogenesis of hAMSC, the model of insulin-resistant adipocytes was successfully established by TNF-α and high glucose intervention. After exosome treatment, the insulin-stimulated glucose uptake was significantly increased. In addition, the effect of exosomes could be stabilized for at least 48 h. Furthermore, the level of leptin was significantly decreased, and the mRNA expression of sirtuin-1 and insulin receptor substrate-1 was significantly upregulated after exosome treatment. In conclusion, exosomes significantly improve insulin sensitivity in insulin-resistant human adipocytes, and the mechanism involves the regulation of adipokines.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Reusch JE, Manson JE. Management of Type 2 Diabetes in 2017: Getting to Goal. JAMA, 2017,317(10):1015–1016
Zhang Y, Mei HL, Chang XA, et al. Adipocyte-derived microvesicles from obese mice induce M1 macrophage phenotype through secreted miR-155. J Mol Cell Biol, 2016,8(6):505–517
Ronningen T, Shah A, Reiner A, et al. Epigenetic priming of inflammatory response genes by high glucose in adipose progenitor cells. Biochem Biophys Res Commun, 2015,467(4): 979–986
Lo KA, Kennedy N, Han M, et al. Analysis of in vitro insulin-resistance models and their physiological relevance to in vivo diet-induced adipose insulin resistance. Cell Reports, 2013,5(1):259–270
Kupsal K, Mudigonda S, Gundapaneni K, et al. Glucotoxicity and lipotoxicity induced beta-cell apoptosis in type 2 diabetes mellitus. Int J Anal Bio-sci, 2015,3(4):84–89
An X, Li L, Chen Y, et al. Mesenchymal Stem Cells Ameliorated Glucolipotoxicity in HUVECs through TSG-6. Int J Mol Sci, 2016,17(4):483
Rebelatto C, Aguiar A, Moretão M, et al. Dissimilar differentiation of mesenchymal stem cells from bone marrow, umbilical cord blood, and adipose tissue. Exp Biol Med (Maywood), 2008,233(7):901–913
Xie ZY, Hao HJ, Tong C, et al. Human umbilical cord-derived mesenchymal stem cells elicit macrophages into an anti-inflammatory phenotype to alleviate insulin resistance in type 2 diabetic rats. Stem Cells, 2016,34(3): 627–639
Xie M, Hao HJ, Cheng Y, et al. Adipose-derived mesenchymal stem cells ameliorate hyperglycemia through regulating hepatic glucose metabolism in type 2 diabetic rats. Biochem Biophys Res Commun, 2017,483(1): 435–441
Phinney D, Pittenger M. Concise Review: MSC-Derived Exosomes for Cell-Free Therapy. Stem Cells, 2017,35(4):851–858
Lawson C, Vicencio J, Yellon D, et al. Microvesicles and exosomes: new players in metabolic and cardiovascular disease. J Endocrinol, 2016,228(2):R57–R71
Sabry D, Marzouk S, Zakaria R, et al. The effect of exosomes derived from mesenchymal stem cells in the treatment of induced type 1 diabetes mellitus in rats. Biotechnol Lett, 2020,42(8):1597–1610
Zhu LL, Huang X, Yu W, et al. Transplantation of adipose tissue-derived stem cell-derived exosomes ameliorates erectile function in diabetic rats. Andrologia, 2018,50(2):e12871
Wang J, Mi Y, Wu S, et al. Exosomes from adipose-derived stem cells protect against high glucose-induced erectile dysfunction by delivery of corin in a streptozotocin-induced diabetic rat model. Regenerative therapy, 2020,14:227–233
Zhao H, Shang Q, Pan Z, et al. Exosomes from Adipose-Derived Stem Cells Attenuate Adipose Inflammation and Obesity through Polarizing M2 Macrophages and Beiging in White Adipose Tissues. Diabetes, 2018,67: 235–247
Moreira A, Kahlenberg S, Hornsby P, et al. Therapeutic potential of mesenchymal stem cells for diabetes. J Mol Endocrinol, 2017,59(3):R109–R120
Pan XH, Huang X, Ruan GP, et al. Umbilical cord mesenchymal stem cells are able to undergo differentiation into functional islet-like cells in type 2 diabetic tree shrews. Mol Cell Probes, 2017,34:1–12
Kobolak J, Dinnyes A, Memic A, et al. Mesenchymal stem cells: Identification, phenotypic characterization, biological properties and potential for regenerative medicine through biomaterial micro-engineering of their niche. Methods, 2016,99:62–68
Vishwanath D, Srinivasan H, Patil MS, et al. Novel method to differentiate 3T3 L1 cells in vitro to produce highly sensitive adipocytes for a GLUT4 mediated glucose uptake using fluorescent glucose analog. J Cell Commun Signal, 2013,7(2):129–140
Xiao Y, Zheng L, Zou X, et al. Extracellular vesicles in type 2 diabetes mellitus: key roles in pathogenesis, complications, and therapy. J Extracell Vesicles, 2019, 8(1):1625677
Noren H, Evans M. Extracellular vesicles as signaling mediators in type 2 diabetes mellitus. Am J Physiol Cell Physiol, 2020,318(6):C1189–C1199
Ying W, Riopel M, Bandyopadhyay G, et al. Adipose Tissue Macrophage-Derived Exosomal miRNAs Can Modulate In Vivo and In Vitro Insulin Sensitivity. Cell, 2017,171(2):372–384
Kita S, Maeda N, Shimomura I. Interorgan communication by exosomes, adipose tissue, and adiponectin in metabolic syndrome. J Clin Invest, 2019, 129(10):4041–4049
Obata Y, Kita S, Koyama Y, et al. Adiponectin/T-cadherin system enhances exosome biogenesis and decreases cellular ceramides by exosomal release. JCI Insight, 2018,3(8):e99680
Könner A, Brüning J. Selective insulin and leptin resistance in metabolic disorders. Cell Metab, 2012, 16(2):144–152
Liang F, Kume S, Koya D. SIRT1 and insulin resistance. Nat Revi Endocrinol, 2016,5(7):178–183
Yuan Y, Shi M, Li L, et al. Mesenchymal stem cell-conditioned media ameliorate diabetic endothelial dysfunction by improving mitochondrial bioenergetics via the Sirt1/AMPK/PGC-1 alpha pathway. Clin Sci (Lond), 2016,130(23):2181–2198
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was supported by the grants from National Key Research and Development Program of China (2016YFA0101002), National Key R&D Program of China (2017YFC1309603), and National Natural Science Foundation of China (Nos. 81170736, 81570715, 81870579).
Conflict of Interest Statement
The authors have declared that there is no conflict of interest.
Electronic Supplementary Material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License https://creativecommons.org/licenses/by/4.0/), which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chen, Mt., Zhao, Yt., Zhou, Ly. et al. Exosomes Derived from Human Umbilical Cord Mesenchymal Stem Cells Enhance Insulin Sensitivity in Insulin Resistant Human Adipocytes. CURR MED SCI 41, 87–93 (2021). https://doi.org/10.1007/s11596-021-2323-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11596-021-2323-4