Abstract
Summary
In an analysis of claims data from a city in Japan, male patients and patients with dementia were less likely to receive osteoporosis pharmacotherapy after hip fracture. Treatment initiation rate has improved between 2014 and 2017.
Purpose
Older adults with recent hip fractures are at a high risk of recurrent fractures. However, the post-fracture care gap has been reported globally. This study examines factors associated with pharmacotherapy non-initiation within 1 year after hip surgery.
Methods
Using medical and long-term care (LTC) claims, and LTC needs certification data in Tsukuba City, Japan, we identified individuals aged 65 years or older who had hip fractures with subsequent surgical procedures between October 1, 2014, and December 31, 2017. Patient (age, sex, dementia, and comorbidities) and health service–related characteristics (fiscal year, type of hospital, number of hospital beds, and admission to recovery phase rehabilitation wards) were examined. The association of these factors with non-pharmacotherapy for osteoporosis within 1 year after hip fracture using multivariable logistic models was analyzed.
Results
We identified 275 patients with hip fractures who did not receive pharmacotherapy pre-fracture. Forty percent of them received pharmacotherapy within 1 year of post-fracture. Male sex (odds ratio (OR) = 4.49 [2.14–9.44]) and dementia (OR = 1.90 [1.03–3.52]) were associated with no pharmacotherapy, whereas later fiscal year (OR = 0.64 [0.48–0.87]) and admission to rehabilitation wards (OR = 0.25 [0.14–0.46]) were associated with pharmacotherapy initiation within 1 year of post-fracture. Comorbidities were not associated with the initiation of pharmacotherapy.
Conclusion
Pharmacotherapy for osteoporosis was less likely to be initiated after a hip fracture in male patients and patients with dementia. These patients should be considered for pharmacotherapy because they are at high risk of recurrent fractures.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fragility hip fractures pose a substantial burden on both patients and society. Hip fracture results in disability, reduced quality of life (QOL) [1, 2], and increased mortality [3, 4]. There is a heavy economic burden associated with fragility hip fractures, and the total medical cost for hip fractures was estimated to be USD $2.99 billion per year in Japan [5]. In addition, the risk of subsequent hip fracture increases after the initial hip fracture [6, 7]. Therefore, pharmacotherapy for osteoporosis is crucial for preventing recurrent fractures and reducing the burden on patients and society.
Evidence for the efficacy of osteoporosis pharmacotherapy has been established. Reports indicate that bisphosphonates can prevent secondary hip fractures and reduce mortality [8]. However, the osteoporosis care gap has been reported globally, with reported treatment rates after hip fractures of < 40% [9,10,11,12]. In particular, the treatment initiation rate after hip fracture is low [12,13,14]. Some patient and healthcare provider factors for non-treatment for osteoporosis after fragility fracture include men, chronic kidney disease, patient perception, and clinical judgment [15,16,17].
To fill the osteoporosis care gap, the fracture liaison service (FLS) was introduced, which provides routine assessment and necessary treatment to patients with a recent fracture. Improvements in outcomes such as bone mineral density (BMD) testing, treatment initiation, treatment adherence, subsequent fracture, and mortality have been reported [18,19,20]. In Japan, certification of the Osteoporosis Liaison Service (OLS) Coordinator by the Japan Osteoporosis Society was started in 2015 to disseminate the liaison service for osteoporosis treatment [21]. However, patient- and health service–related factors affecting treatment initiation for osteoporosis after hip fracture and improvement in treatment initiation rate in Japan are not fully known.
The purpose of this study was to exploratorily examine the factors associated with the initiation of pharmacotherapy for osteoporosis in patients with hip fractures and to assess whether the initiation rate has changed since 2015, using medical and long-term care (LTC) claims data as well as LTC needs certification data in Japan.
Methods
Data sources
The present study utilized medical and pharmacy claims data, LTC claims data, and LTC needs certification data between April 1, 2014, and March 31, 2019, in Tsukuba City, Japan. The city is located in the suburbs of Tokyo, Japan. It had a population of approximately 220,000, and 18% of them were aged 65 years or older in 2014 [22]. The medical and pharmacy claims data included data from the Late Elder’s Health Insurance (public medical insurance for all individuals aged ≥ 75 years and those aged 65–74 years with certified disability) and National Health Insurance (public medical insurance for unemployed or self-employed people and retirees aged < 75 years). The database comprised two health insurance datasets covering 89% of citizens aged ≥ 65 years as of October 2018 [23]. In Japan, most of the population has some form of medical insurance, and a small percentage of adults aged 65 years or older are covered by Employees’ Health Insurance or public assistance. Our database did not include these older adults. All data were anonymized with unique identification numbers, which helped merge the medical claims and LTC data.
Study population
We identified patients aged 65 years or older who were newly diagnosed with hip fractures and subsequently underwent a surgical procedure between October 1, 2014, and December 31, 2017, using disease codes for hip fractures (femoral neck and trochanteric fractures) and procedure codes (open surgery for femur fracture, bipolar hip arthroplasty, or total hip replacement) (n = 561). The index date was defined as the date of the first recorded surgical procedure for hip fractures. In Japan, 95% of patients with hip fractures receive surgical treatment, and the median wait time before surgery is 3 days [24]. In the case of multiple hip fractures during the study period, the first hip fracture of the patient was treated as an index hip fracture. We only included patients who were continuously enrolled for medical insurance in this city for at least 6 months prior to and at least 15 months after a month of the index date (n = 428). Withdrawal due to death was noted for 114 patients. If the patients were admitted to hospital beds or facilities under the comprehensive payment system for medical expenses (recovery phase rehabilitation ward, community comprehensive care ward, long-term care bed, and geriatric health services facilities), drug prescription data may not have been available. Therefore, we excluded patients with hip fractures who were in these hospital beds or facilities during the 3 months before (n = 22) or 12 months consecutively (n = 39) after the index month using medical and LTC claims data. Older patients remain admitted if they need rehabilitation or need to prepare for returning home or transferring to care facilities after their condition has stabilized. In addition, these patients remain admitted if they require long-term medical care after the acute medical care, nursing care, and support (due to disabilities) under a physician’s supervision until they return home. Additionally, we excluded patients who had disease codes for bone metastasis during the 15 months following the index month (n = 3) to focus on fragility hip fracture cases. None of the patients had disease codes for Paget’s disease.
Osteoporosis medications
The study outcome was pharmacotherapy for osteoporosis, defined as the occurrence of at least one medical or pharmacy claims for osteoporosis pharmacotherapy within 12 months post-fracture (Fig. 1). We determined the prescription of drugs (oral and parenteral) with approved indications for osteoporosis treatment in Japan. These included bisphosphonates (alendronate, risedronate, minodronate, ibandronate, etidronic acid, and zoledronic acid), selective estrogen receptor modulators (raloxifene and bazedoxifene acetate), receptor activator of nuclear factor kappa-B ligand antibodies (denosumab), parathyroid hormone (both daily and weekly), active vitamin D3 single-agent (eldecalcitol), and others (alfacalcidol, calcitriol, menatetrenone, ipriflabon, calcium aspartate, calcium phosphate, and elcatonin).
Osteoporosis pharmacotherapy within 90 days prior to the index date was determined based on 6-month prescription data before the index date. The last drug-available day was estimated based on the date of the prescription and the number of days of supply according to the types of osteoporosis drugs in each prescription.
Assessment of co-variables
The types of hospital (national or public, university, and other hospitals) where the patients underwent hip fracture surgery, the number of beds in each hospital, and admission to the recovery phase rehabilitation ward within three months after the index month were identified from medical claims data. Other fractures within 1 year of the index hip fracture were identified using diagnostic codes. As a proxy for general comorbidity, the Charlson comorbidity index (CCI) score was calculated using the International Statistical Classification of Diseases and Related Health Problems (ICD-10) codes in the three months preceding the index months with the algorithms by Quan et al., which has been previously used in medical and long-term care claims data in Japan [25,26,27].
Anti-dementia drugs (donepezil, rivastigmine, galantamine, and memantine), proton pump inhibitors (PPI), non-steroidal anti-inflammatory drugs (NSAIDs), and steroids prescribed within 90 days before the index date were ascertained using medication codes. Polypharmacy was defined as the median number of prescribed drug components within 3 months prior to the index month being ≥ 5.
LTC needs certification determined the eligibility and care need level (7 levels: support 1, support 2, care 1, care 2, care 3, care 4, and care 5) of individuals aged ≥ 65 years or those aged 40–64 years with disability due to specific diseases. The certification is based on an assessment of physical and cognitive functions as well as the opinion of the patients’ primary physicians. LTC certification data includes the graded activities of daily living related to dementia noted by a primary physician with eight grades and one missing category: Grade 0, I (almost independent daily living is possible despite having some type of dementia), IIa, IIb, IIIa, IIIb, IV, and M (marked psychiatric symptoms requiring expert management). In Japan, most people aged ≥ 65 years with dementia applied for LTC needs certification [28]. For LTC service recipients, a cutoff of grade I or greater had 83% sensitivity and 92% specificity with the gold standard diagnosis made by neuropsychiatrists [28]. Hence, we defined patients with probable dementia as those who had ≥ grade I in the latest LTC needs certification before the index date, had diagnosis codes of dementia within 3 months prior to the index months, or were prescribed anti-dementia drugs 90 days preceding the index date [29].
Analysis
Initially, the percentages of participants with and without osteoporosis pharmacotherapy before hip fracture but who received pharmacotherapy within 1 year after hip surgery were calculated.
To focus on pharmacotherapy initiation, only patients with hip fracture without osteoporosis pharmacotherapy within 90 days before the index date were included. Patient- and health service–related characteristics at baseline, admission to the recovery phase rehabilitation ward within 3 months, and other fractures within a year of initial hip fracture were compared between patients with and without osteoporosis pharmacotherapy within 1 year of post-fracture. This was done using t tests for continuous variables and chi-square tests or Fisher’s exact tests for categorical variables. The four types of hospitals (university hospitals, national hospitals, public hospitals, and other hospitals) were categorized into two types: university, national or public hospitals, and other hospitals. The number of hospital beds was categorized into one of three ranges: 100–199, 200–499, and ≥ 500. The CCI was categorized into one of three groups: 0, 1–2, and ≥ 3. The LTC care needs levels were categorized into one of four groups: without certification, support 1 and 2, care 1–3, and care 4 and 5. We also considered two kinds of dementia variables: the use of anti-dementia medication, and probable dementia based on LTC needs certification, disease codes, or anti-dementia medication use before hip fracture. Pharmacotherapy initiation rates post-fracture were calculated by fiscal year.
The association between the characteristics and pharmacotherapy for osteoporosis within 1-year post-fracture was examined exploratorily using multiple logistic regression models. The outcome was no pharmacotherapy within 1 year after the index date. Age at surgery and sex were included in the models, regardless of statistical significance. Owing to the limited number of outcomes and avoiding saturated models, the independent variables were chosen based on their clinical relevance, the results of the chi-square tests, and better model fit based on Akaike’s information criterion (AIC). For example, hospital type and number of hospital beds were related; however, only the number of hospital beds was included in the model because of the better model fit than hospital type. The fiscal year of hip surgery was treated as a linear variable after verifying that the overall results did not change if it was treated as a categorical variable. In the sensitivity analysis, instead of dementia, the LTC care needs level was included in the logistic regression model. Adjusted odds ratios (ORs) and corresponding 95% confidence intervals (CIs) were estimated. All analyses were conducted using SAS 9.3 (SAS Institute Inc. Carny, NC), and an alpha level of 0.05 was defined as statistically significant.
Ethical approval
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required. This study was approved by the Medical Ethics Committee of the University of Tsukuba (approval number 1445-10).
Results
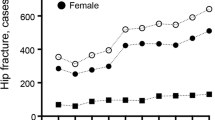
We identified 364 patients with hip fractures (males: 21.4%). Among these, 89 (24.5%) had osteoporosis pharmacotherapy prior to hip fracture, and 275 (75.6%) did not. In patients with pharmacotherapy pre-fracture, almost all patients (96.6%) received osteoporosis pharmacotherapy within 1 year of post-fracture. However, in patients without pharmacotherapy pre-fracture, only 110 (40.0%) patients received osteoporosis pharmacotherapy within 1 year of post-fracture. The rates of pharmacotherapy initiation by fiscal year are shown in Fig. 2. The initiation rate was higher in later years.
The mean age of the 275 patients without pharmacotherapy for osteoporosis pre-fracture was 83.9 (standard deviation = 7.6), and 25.8% (n = 71) were males. The characteristics of patients are listed in Table 1. Among these 275 participants, those who did not receive pharmacotherapy post-fracture were more likely to be males, underwent hip surgery in hospitals other than university, national, or public hospitals, or were treated in hospitals with < 500 beds. They were also more likely to have probable dementia or be prescribed anti-dementia drugs before the hip fracture, require a high care need level, not be admitted to the recovery phase rehabilitation ward within 3 months, and have no other fractures within 1 year of the hip fracture. No association was found between polypharmacy and osteoporosis pharmacotherapy after hip fracture.
The results of the multivariable logistic regression analysis are presented in Table 2. The final logistic model included age at surgery, sex, probable dementia, CCI, fiscal year of hip surgery, number of hospital beds, and admission to the recovery-phase rehabilitation ward within 3 months (model 1). Sex, probable dementia, fiscal year, and no admission to the recovery phase rehabilitation wards were significantly associated with no osteoporosis pharmacotherapy within 1 year after the hip fracture. Male patients were four times more likely to have no therapy (OR = 4.49 [2.14–9.44]). Patients with probable dementia were almost twice as likely to not receive osteoporosis pharmacotherapy after hip fracture (OR = 1.90 [1.03–3.52]). Later fiscal years were negatively associated with no therapy (OR = 0.64 [0.48–0.87]). Patients admitted to recovery phase rehabilitation wards post-fracture were more likely to receive osteoporosis pharmacotherapy (OR = 0.25 [0.14–0.46]). CCI was not associated with pharmacotherapy after hip fractures. When anti-dementia medication usage before hip fracture instead of probable dementia was included in the model, the overall results did not change (model 2). Anti-dementia drug usage was significantly associated with no pharmacotherapy (OR = 2.66 [1.12–6.30]), and patients treated in hospitals with ≥ 500 beds were more likely to receive pharmacotherapy for osteoporosis than those treated in hospitals with 200–499 beds (OR = 0.21 [0.05–0.95]). In the analysis using the LTC care needs level, instead of dementia, a high LTC care needs level (level 4 or 5) was significantly associated with non-therapy (OR = 12.54 [2.54–61.79], Supplementary table).
Discussion
Among hip fracture patients without pharmacotherapy for osteoporosis pre-fracture, only 40% received osteoporosis pharmacotherapy within 1 year of hip fracture, whereas 96.6% of patients with pharmacotherapy pre-fracture received osteoporosis pharmacotherapy post-fracture. Among older adults without pharmacotherapy pre-fracture, male patients, those with dementia, and those who were not admitted to the recovery phase rehabilitation ward after hip fracture were associated with no osteoporosis pharmacotherapy initiation. Patients who had hip fractures in the later fiscal years were more likely to receive pharmacotherapy after a hip fracture.
In our study, the overall rate of pharmacotherapy within 1 year after hip fracture was 53.9% in older adults aged ≥ 65 years and 40.0% in those without pharmacotherapy pre-fracture. The overall rate was higher than the recent report of 31.6% in Japan, which included men and women who were ≥ 50 years and diagnosed with hip fractures between 2013 and 2018 [12]. In that study, the rate of pharmacotherapy in those without pharmacotherapy before fracture was 19% based on their table, which was lower than that in the current study. However, a direct comparison was difficult to interpret because their study included younger patients and did not include some drugs (calcitriol, ipriflabon, calcium aspartate, calcium phosphate, and elcatonin) which were included in our study, using the National Database of Health Insurance Claims. In addition, we excluded patients receiving continuous care in hospital beds or care facilities after hip fracture where drug prescription may not appear in claims. Nonetheless, the initiation rate for pharmacotherapy is still suboptimal considering that fragility hip fracture is a criterion for osteoporosis pharmacotherapy initiation regardless of BMD in the current Japanese guidelines [30].
Dementia was associated with non-pharmacotherapy initiation after hip fracture, whereas the burden of comorbid diseases represented by the CCI was not. Oyamada et al. reported the reasons for non-treatment during hospitalization among hip fracture patients receiving initial pharmacotherapy from the perspective of physicians. The reasons include physicians’ clinical judgment and dementia [17]. Although the reasons for the non-treatment of patients with initial therapy were examined, dementia may influence physicians’ decision to prescribe pharmacotherapy for older adults with hip fracture. Based on administrative insurance claims data in the USA, Solomon et al. also reported that dementia was associated with a lack of osteoporosis medication use within 12 months after discharge in patients with hip fractures [9]. The possible factors affecting physicians’ prescription decisions may include patients’ understanding or perception of osteoporosis therapy, as well as the issue of medication adherence in older adults with dementia. Beaton et al. reported that the perceived need of patients was associated with the initiation of osteoporosis pharmacotherapy in patients aged ≥ 50 years who sustained a fragility fracture in Canada [16]. Misconceptions about the disease and lack of perceived benefit of therapy were factors associated with lower medication adherence [31]. Psychiatric conditions, such as depression are associated with poor medication adherence among patients with osteoporosis [31]. García-Sempere et al. reported that dementia was associated with non-adherence following hip fracture in patients aged ≥ 65 years [32]. Physicians may believe that older adults with dementia face difficulty adhering to pharmacotherapy, or that the assessment of adherence by caregivers in older adults may be difficult. However, cognitive impairment is a risk factor for falls which result in hip fractures [33]. Thus, pharmacotherapy for recurrent fracture prevention is beneficial for older adults with dementia.
Other characteristics were associated with pharmacotherapy for osteoporosis after hip fracture. Male patients were over four times less likely to receive pharmacotherapy after a hip fracture than female patients. Lower treatment rates for men were also reported in the recent studies [12, 15]. The risk of subsequent hip fracture is also high in men as in older women [7, 34]. In addition, excess mortality after hip fracture is high in men [4, 35]. Therefore, older men with hip fractures should receive pharmacotherapy to prevent recurrent fractures.
Patients admitted to recovery phase rehabilitation wards within three months after hip fracture were more likely to receive pharmacotherapy within 1 year. It is possible that physicians were more likely to prescribe osteoporosis drugs to patients who were at higher risk of falls and needed rehabilitation after surgery compared to those who did not. In a recent Japanese report, a low Barthel index, which is a common score for activities of daily living, was associated with non-pharmacotherapy [15]. In our sensitivity analysis using LTC care needs levels instead of dementia, admission to the recovery phase rehabilitation wards was still significantly associated with higher pharmacotherapy initiation, adjusting for LTC care needs levels. Over 60% of the patients not admitted to a recovery phase rehabilitation ward were either in their own homes or some form of housing for elderly persons 3 months after the index months. In addition, among the patients not admitted to a recovery phase rehabilitation ward, there were more patients in intensive care homes run by local government or non-profit organizations, which provided long-term care for older adults who required consistent care before the hip fracture and returned after the hip fracture. Fourteen percent were in hospital beds other than those in recovery phase rehabilitation wards. It is possible that patients who were admitted to the rehabilitation wards were more likely to have access to the OLS team or BMD testing during hospitalization than those who left the initial hospital for their houses or other care facilities after surgery; however, we could not ascertain, based on claims data, whether the FLS had adequate access to the patient and their health-care provider in each hospital’s recovery phase rehabilitation wards.
In our study, a later fiscal year was associated with a higher rate of initiation of osteoporosis pharmacotherapy after hip fracture. Similar results were reported in Hokkaido in Japan [36]. The fracture liaison service is a secondary prevention program designed to provide routine assessment and necessary treatment to patients with recent fractures. The improvement of BMD testing, treatment initiation, treatment adherence, and cost-effectiveness has been reported [18,19,20, 37, 38]. In Japan, OLS includes FLS, and certification of the OLS Coordinator began in 2015 [21]. Patients in our study underwent hip surgery between October 2014 and December 2017. Improvement in the treatment initiation rate is multifactorial. For example, yearly zoledronic acid hydrate was marketed in November 2016 in Japan, which may have influenced physicians’ prescription behavior. In addition, among the overall patients included in the present study, in 2014 only patients who had hip fracture surgery between October 1 and December 31 were included. Seasonal differences regarding patients’ characteristics and medical facilities’ circumstances might have influenced the osteoporosis pharmacotherapy initiation rate in 2014. Therefore, this result should be interpreted with caution. FLS was not covered by medical insurance and did not appear in medical claims during that period. Therefore, direct causal relationship cannot be known. Nonetheless, the educational certification courses for OLS coordinators may have positively impacted the initiation of osteoporosis treatment in Japan.
Older adults who underwent hip fracture surgery in larger hospitals were more likely to receive pharmacotherapy after hip fracture. Possible reasons may include a lack of BMD testing and liaison teams in smaller hospitals; however, based on claims data, we could not determine whether the FLS were active in each hospital. Upon examining claims data, Sugiyama et al. found that process quality measures of diabetes care, such as proportions of patients receiving retinopathy examination, were better in medical institutions with a higher number of beds and institutions with certification as an educational institution by the Japan Diabetes Society [39]. Martin et al. reported in their meta-analysis that interventions targeting health systems as well as healthcare professionals were effective in improving the prescription of osteoporosis therapy [40]. The introduction of a multidisciplinary OLS team in small hospitals or building an OLS network with neighboring hospitals might be useful in improving pharmacotherapy initiation for osteoporosis. However, we did not explore the medical facilities in which these older adults received care after discharge from the initial hospital. Further studies are needed to assess the influence of the characteristics of medical facilities on treatment initiation.
The strength of this study lies in the combined medical claims data, LTC claims data, and LTC needs certification data for this analysis. Hence, we considered the disability levels of hip fracture patients, but excluded patients who continuously stayed in hospitals or care facilities where drug prescription data may not be reported, which has not been highlighted in the previous studies using medical claims data. Our findings of dementia and admission to recovery phase rehabilitation wards as factors associated with pharmacotherapy initiation build on the previous literature. We identified hip fracture patients using both diagnostic and procedure codes, which improved the specificity of diagnosis, although some hip fracture patients without a surgical procedure were not included in our study.
Nonetheless, this study has some limitations. Since we only considered osteoporosis pharmacotherapy for 3 months before hip fracture based on 6-month medical claims data due to data availability, some patients might have received pharmacotherapy earlier. Thus, some misclassifications regarding osteoporosis pharmacotherapy before hip fractures are possible. Additionally, we could not determine whether these older patients used the prescribed medication because we used medical claims data.
In conclusion, pharmacotherapy for osteoporosis was less likely to be initiated after hip fracture in male patients and patients with dementia, although the treatment initiation rate was better in later years. These patients should be considered for pharmacotherapy for osteoporosis because they are at a high risk of falls and recurrent fractures.
References
Boonen S, Autier P, Barette M, Vanderschueren D, Lips P, Haentjens P (2004) Functional outcome and quality of life following hip fracture in elderly women: a prospective controlled study. Osteoporos Int 15:87–94. https://doi.org/10.1007/s00198-003-1515-z
Adachi JD, Loannidis G, Berger C et al (2001) The influence of osteoporotic fractures on health-related quality of life in community-dwelling men and women across Canada. Osteoporos Int 12:903–908. https://doi.org/10.1007/s001980170017
Brauer CA, Coca-Perraillon M, Cutler DM, Rosen AB (2009) Incidence and mortality of hip fractures in the United States. JAMA 302:1573–1579. https://doi.org/10.1001/jama.2009.1462
Katsoulis M, Benetou V, Karapetyan T et al (2017) Excess mortality after hip fracture in elderly persons from Europe and the USA: the CHANCES project. J Intern Med 281:300–310. https://doi.org/10.1111/joim.12586
Mori T, Komiyama J, Fujii T et al (2022) Medical expenditures for fragility hip fracture in Japan: a study using the nationwide health insurance claims database. Arch Osteoporos 17:61. https://doi.org/10.1007/s11657-022-01096-8
Hagino H, Sawaguchi T, Endo N, Ito Y, Nakano T, Watanabe Y (2012) The risk of a second hip fracture in patients after their first hip fracture. Calcif Tissue Int 90:14–21. https://doi.org/10.1007/s00223-011-9545-6
Center JR, Bliuc D, Nguyen TV, Eisman JA (2007) Risk of subsequent fracture after low-trauma fracture in men and women. JAMA 297:387–394. https://doi.org/10.1001/jama.297.4.387
Peng J, Liu Y, Chen L, Peng K, Xu Z, Zhang D, Xiang Z (2016) Bisphosphonates can prevent recurrent hip fracture and reduce the mortality in osteoporotic patient with hip fracture: a meta-analysis. Pak J Med Sci 32:499–504. https://doi.org/10.12669/pjms.322.9435
Solomon DH, Johnston SS, Boytsov NN, McMorrow D, Lane JM, Krohn KD (2014) Osteoporosis medication use after hip fracture in U.S. patients between 2002 and 2011. J Bone Miner Res 29:1929–1937. https://doi.org/10.1002/jbmr.2202
Kim SC, Kim MS, Sanfélix-Gimeno G et al (2015) Use of osteoporosis medications after hospitalization for hip fracture: a cross-national study. Am J Med 128:519–26.e1. https://doi.org/10.1016/j.amjmed.2015.01.014
Wilk A, Sajjan S, Modi A, Fan CP, Mavros P (2014) Post-fracture pharmacotherapy for women with osteoporotic fracture: analysis of a managed care population in the USA. Osteoporos Int 25:2777–2786. https://doi.org/10.1007/s00198-014-2827-x
Nakatoh S, Fujimori K, Ishii S, Tamaki J, Okimoto N, Ogawa S, Iki M (2021) Insufficient increase in bone mineral density testing rates and pharmacotherapy after hip and vertebral fracture: analysis of the National Database of Health Insurance Claims and Specific Health Checkups of Japan. Arch Osteoporos 16:130. https://doi.org/10.1007/s11657-021-00992-9
Cheung MY, Ho AW, Wong SH (2018) Postfracture care gap: a retrospective population-based analysis of Hong Kong from 2009 to 2012. Hong Kong Med J 24:579–583. https://doi.org/10.12809/hkmj187227
Desai RJ, Mahesri M, Abdia Y, Barberio J, Tong A, Zhang D, Mavros P, Kim SC, Franklin JM (2018) Association of osteoporosis medication use after hip fracture with prevention of subsequent nonvertebral fractures: an instrumental variable analysis. JAMA Netw Open 1:e180826. https://doi.org/10.1001/jamanetworkopen.2018.0826
Kanahori M, Matsumoto Y, Fujiwara T et al (2021) Predictive factors of non-treatment and non-persistence to osteoporosis medication after fragility hip fractures at 3 years after discharge: a multicentre, prospective cohort study in the northern Kyushu district of Japan. Arch Osteoporos 16:132. https://doi.org/10.1007/s11657-021-00988-5
Beaton DE, Dyer S, Jiang D, Sujic R, Slater M, Sale JE, Bogoch ER, Osteoporosis Fracture Clinic Screening Program Evaluation Team (2014) Factors influencing the pharmacological management of osteoporosis after fragility fracture: results from the Ontario Osteoporosis Strategy’s fracture clinic screening program. Osteoporos Int 25:289–296. https://doi.org/10.1007/s00198-013-2430-6
Oyamada A, Matsumoto Y, Wakata Y et al (2018) Characteristics of patients with fragility hip fractures in the northern Kyushu district in Japan: a multicenter prospective registry based on an electronic data capture system. J Bone Miner Metab 36:596–604. https://doi.org/10.1007/s00774-017-0869-9
McLellan AR, Gallacher SJ, Fraser M, McQuillian C (2003) The fracture liaison service: success of a program for the evaluation and management of patients with osteoporotic fracture. Osteoporos Int 14:1028–1034. https://doi.org/10.1007/s00198-003-1507-z
Huntjens KM, van Geel TA, van den Bergh JP, van Helden S, Willems P, Winkens B, Eisman JA, Geusens PP, Brink PR (2014) Fracture liaison service: impact on subsequent nonvertebral fracture incidence and mortality. J Bone Joint Surg Am 96:e29. https://doi.org/10.2106/JBJS.L.00223
Wu CH, Tu ST, Chang YF, Chan DC, Chien JT, Lin CH, Singh S, Dasari M, Chen JF, Tsai KS (2018) Fracture liaison services improve outcomes of patients with osteoporosis-related fractures: a systematic literature review and meta-analysis. Bone 111:92–100. https://doi.org/10.1016/j.bone.2018.03.018
Hagino H, Wada T (2019) Osteoporosis liaison service in Japan. Osteoporos Sarcopenia 5:65–68. https://doi.org/10.1016/j.afos.2019.09.003
City of Tsukuba (2014). Statistics. https://www.city.tsukuba.lg.jp/shisei/joho/toukei/1002336.html. Accessed 31 Jul 2022
Kuroda N, Iwagami M, Hamada S, Komiyama J, Mori T, Tamiya N (2022) Associations of polypharmacy and drugs with sedative or anticholinergic properties with the risk of long-term care needs certification among older adults in Japan: A population-based, nested case-control study. Geriatr Gerontol Int 22:497–504. https://doi.org/10.1111/ggi.14393
Hagino H, Endo N, Harada A, Iwamoto J, Mashiba T, Mori S, Ohtori S, Sakai A, Takada J, Yamamoto T (2017) Survey of hip fractures in Japan: recent trends in prevalence and treatment. J Orthop Sci 22:909–914. https://doi.org/10.1016/j.jos.2017.06.003
Quan H, Sundararajan V, Halfon P, Fong A, Burnand B, Luthi JC, Saunders LD, Beck CA, Feasby TE, Ghali WA (2005) Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care 43:1130–1139. https://doi.org/10.1097/01.mlr.0000182534.19832.83
Mori T, Tamiya N, Jin X, Jeon B, Yoshie S, Iijima K, Ishizaki T (2018) Estimated expenditures for hip fractures using merged healthcare insurance data for individuals aged ≥ 75 years and long-term care insurance claims data in Japan. Arch Osteoporos 13:37. https://doi.org/10.1007/s11657-018-0448-2
Mori T, Hamada S, Yoshie S, Jeon B, Jin X, Takahashi H, Iijima K, Ishizaki T, Tamiya N (2019) The associations of multimorbidity with the sum of annual medical and long-term care expenditures in Japan. BMC Geriatr 19:69. https://doi.org/10.1186/s12877-019-1057-7
Noda H, Yamagishi K, Ikeda A, Asada T, Iso H (2018) Identification of dementia using standard clinical assessments by primary care physicians in Japan. Geriatr Gerontol Int 18:738–744. https://doi.org/10.1111/ggi.13243
Kuroda N, Hamada S, Sakata N, Jeon B, Iijima K, Yoshie S, Ishizaki T, Jin X, Watanabe T, Tamiya N (2019) Antipsychotic use and related factors among people with dementia aged 75 years or older in Japan: a comprehensive population-based estimation using medical and long-term care data. Int J Geriatr Psychiatry 34:472–479. https://doi.org/10.1002/gps.5041
Japan Osteoporosis Society, the Japanese Society for Bone and Mineral Research, Japan Osteoporosis Foundation (2015) Japanese 2015 guidelines for prevention and treatment of osteoporosis (in Japanese). Life Science Publishing
Yeam CT, Chia S, Tan HCC, Kwan YH, Fong W, Seng JJB (2018) A systematic review of factors affecting medication adherence among patients with osteoporosis. Osteoporos Int 29:2623–2637. https://doi.org/10.1007/s00198-018-4759-3
García-Sempere A, Hurtado I, Sanfélix-Genovés J, Rodríguez-Bernal CL, Gil Orozco R, Peiró S, Sanfélix-Gimeno G (2017) Primary and secondary non-adherence to osteoporotic medications after hip fracture in Spain. The PREV2FO population-based retrospective cohort study. Sci Rep 7:11784. https://doi.org/10.1038/s41598-017-10899-6
Deandrea S, Lucenteforte E, Bravi F, Foschi R, La Vecchia C, Negri E (2010) Risk factors for falls in community-dwelling older people: a systematic review and meta-analysis. Epidemiol (Camb Mass) 21:658–668. https://doi.org/10.1097/EDE.0b013e3181e89905
Bliuc D, Alarkawi D, Nguyen TV, Eisman JA, Center JR (2015) Risk of subsequent fractures and mortality in elderly women and men with fragility fractures with and without osteoporotic bone density: the Dubbo Osteoporosis Epidemiology Study. J Bone Miner Res 30:637–646. https://doi.org/10.1002/jbmr.2393
Bliuc D, Nguyen ND, Milch VE, Nguyen TV, Eisman JA, Center JR (2009) Mortality risk associated with low-trauma osteoporotic fracture and subsequent fracture in men and women. JAMA 301:513–521. https://doi.org/10.1001/jama.2009.50
Nakatoh S, Fujimori K, Tamaki J, Okimoto N, Ogawa S, Iki M (2020) Insufficient increase in bone mineral density testing rates and pharmacotherapy after hip fracture in Japan. J Bone Miner Metab 38:589–596. https://doi.org/10.1007/s00774-020-01093-2
Yong JH, Masucci L, Hoch JS, Sujic R, Beaton D (2016) Cost-effectiveness of a fracture liaison service--a real-world evaluation after 6 years of service provision. Osteoporos Int 27:231–240. https://doi.org/10.1007/s00198-015-3280-1
Martin J, Viprey M, Castagne B, Barral M, Chapurlat R, Julien C, Serrier H, Schott AM (2020) Cost-effectiveness analyses of interventions to improve osteoporosis care in France. Arch Osteoporos 15:42. https://doi.org/10.1007/s11657-020-00720-9
Sugiyama T, Imai K, Ihana-Sugiyama N et al (2019) Variation in process quality measures of diabetes care by region and institution in Japan during 2015–2016: an observational study of nationwide claims data. Diabetes Res Clin Pract 155:107750. https://doi.org/10.1016/j.diabres.2019.05.029
Martin J, Viprey M, Castagne B, Merle B, Giroudon C, Chapurlat R, Schott AM (2020) Interventions to improve osteoporosis care: a systematic review and meta-analysis. Osteoporos Int 31:429–446. https://doi.org/10.1007/s00198-020-05308-0
Acknowledgements
We are grateful to the staff members of Tsukuba City Hall for their contributions to data acquisition. We would like to thank the members of the National Health Insurance Division, Medical Welfare and Pension Division, Nursing Care Insurance Division, Health Promotion Division, and Municipal Senior Services Support Division. We also thank the Tsukuba Healthcare and Long-term Care Claims Conference for their useful discussions. We thank Editage (http://www.editage.com] for the English language editing. This work was supported by JSPS KAKENHI (grant number JP20K10340).
Funding
This work was supported by JSPS KAKENHI (grant number JP20K10340).
Author information
Authors and Affiliations
Contributions
TF: conception and design, analysis and interpretation of data, drafting of the article, critical revision of the article for important intellectual content, and final approval. TM: conception and design, interpretation of data, critical revision of the article for important intellectual content, and final approval. JK: building the database, interpretation of data, critical revision of the article for important intellectual content, and final approval. NK: data acquisition, interpretation of data, critical revision of the article for important intellectual content, and final approval. NT: data acquisition, interpretation of data, critical revision of the article for important intellectual content, and final approval.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required. This study was approved by the Medical Ethics Committee of the University of Tsukuba (approval number 1445-10).
Conflicts of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 19 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fujii, T., Mori, T., Komiyama, J. et al. Factors associated with non-initiation of osteoporosis pharmacotherapy after hip fracture: analysis of claims data in Japan. Arch Osteoporos 18, 103 (2023). https://doi.org/10.1007/s11657-023-01314-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11657-023-01314-x