Abstract
The pine wood nematode (PWN), Bursaphelenchus xylophilus (Steiner & Buhrer) Nickle, is the pathogen of pine wilt disease (PWD) which can devastate forests. PWN can be of high or low severity and the mechanisms underlying the differences in virulence are unclear. Therefore, it is necessary to study the relationship between differentiation of PWN severity and its resistance to the main defensive substances of pine species (i.e., α-pinene and H2O2). The feeding rate and fecundity of PWN was examined at different levels of virulence under conditions of α-pinene and H2O2 stress. Moreover, the expression patterns of the main resistance genes of PWN with different virulence were determined under conditions of α-pinene and H2O2 stress. The feeding rate and fecundity of the high virulence strain AMA3 were higher than those of the low virulence strain YW4. The expression levels of the autophagy gene BxATG5, cytochrome P450 gene BxCYP33D3, and glutathione S-transferase genes BxGST1 and BxGST3 in AMA3 increased significantly upon exposure to α-pinene for 2 h, while these genes showed smaller degrees of upregulation in YW4. Under conditions of H2O2 stress, the expression levels of BxATG5, catalase genes Bxy-ctl-1and Bxy-ctl-2, and the 2-cysteine peroxiredoxin gene BxPrx in AMA3 were higher than those in YW4. These findings suggest that high virulence PWN has greater resistance to pine defensive substances α-pinene and H2O2 than low virulence PWN, and resistance genes mediate the differential resistance of PWN strains. This study will contribute to the clarification of the mechanism underlying virulence differentiation of PWN and will advance understanding of the pathogenic mechanism of PWD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pine wilt disease (PWD) is caused by the well-known quarantine pest, the pine wood nematode (PWN), Bursaphelenchus xylophilus (Steiner & Buhrer) Nickle, which belongs to the family Aphelenchoididae, and has a devastating effect on Pinus species (Zhou et al. 2017). PWN infects 58 species of Pinus and 13 other coniferous species (He et al. 2014). The disease originated in North America and is found in Canada, the United States, and Mexico. It also occurs in Spain and Portugal in Europe, and in China, Japan, South and North Korea in Asia (He et al. 2014; Ye 2019). PWD was first discovered in 1982 at the Sun Yat-sen Mausoleum in Nanjing, China, and has since spread rapidly (Cheng et al. 1983). China is currently the country most threatened by pine wilt disease (Huang et al. 2019; Ye 2019). However, the pathogenic mechanism of PWD is not well understood and it remains difficult to prevent and control (Li et al. 2018).
The life cycle of the disease involves the dispersal phase and the propagative phase (Futai 2013). When encountering adverse environments such as food scarcity or low temperatures, the second stage propagative larvae molt and develop into third-stage dispersal larvae, and then grow into a specific dispersal stage or alternate developmental stage, of dauer larvae (Ogura and Nakajima 2002; Kikuchi et al. 2011). Following dispersal from dead to healthy pines via Monochamus vector beetles (Zhao et al. 2016; Wu et al. 2019), the dauer larvae infect the pine host to feed (Vicente et al. 2013). In the infection process, the pines secrete various defensive substances to attack the nematodes. Pine resin consisting of turpentine (monoterpenes and sesquiterpenes) and rosin (diterpenes) is the first line of defense (Zulak and Bohlmann 2010). Monoterpenes, the most essential defensive volatile substances, can help plants resist relevant pathogen and herbivore invasions (Lewinsohn et al. 1993). The monoterpenes secreted by conifers are mainly α-Pinene (Tiao et al. 2006). Li et al. (2019) reported that the reproduction of PWN was dramatically inhibited by 56.3 mg mL‒1 of α-pinene. In addition, plants can also produce reactive oxygen species (ROS) to damage organelles and inhibit cell functions in pathogens (Baker and Orlandi 1995). The most abundant and stable ROS in pines is hydrogen peroxide, H2O2 (Vellosillo et al. 2010). The resin and ROS levels in pine trees increase markedly during the early stages of the disease in response to invasion by the nematodes (Yang et al. 2003; He et al. 2010; Yu et al. 2013). On the other hand, resistance genes protect the nematodes from damage by pine defensive substances. Autophagy is a subcellular degradation pathway which helps to degrade and cycle aging and damaged organelles and proteins in eukaryotes (Ktistakis et al. 2016). In addition, the autophagy gene BxATG5 shows strong affiliations with the growth and development, stress resistance, and virulence of PWN (Liu et al. 2019a; 2020). The degradation of pine defensive substances by the pine wood nematodes is vital for successful infection. The cytochrome P450 gene BxCYP33D3 is responsible for the detoxification of α-pinene (Xu et al. 2015). PWN responds to the accumulation of α-pinene by upregulating cytochrome P450 gene expression (Wang et al. 2017). Glutathione S-transferase (GST) is an important detoxifying enzyme in eukaryotes. The GST genes, BxGST1 and BxGST3, are closely associated with feeding rate, reproduction, and pathogenicity of the nematodes (Nian 2017). The catalase (CAT) genes, Bxy-ctl-1 and Bxy-ctl-2, and the 2-cysteine peroxiredoxin gene BxPrx were reported to be the main antioxidant genes in PWN, which can convert H2O2 to water by regulating the activities of CAT and peroxiredoxin (Li et al. 2011; Vicente et al. 2015; Xu et al. 2019).
PWN can have high or low virulence (Shinya et al. 2012; Ding et al. 2016), and its severity and adaptability to unfavorable environments are strongly correlated with the spread and development of pine wilt disease (Liu et al. 2019b). The degree of virulence of PWN is positively correlated with its reproductive ability in pines and on Botrytis cinerea Pers. or grey mould (Wang et al. 2005; Aikawa and Kikuchi 2007). Eo et al. (2011) reported that the virulence of pine wood nematodes was not correlated with its migration rate. However, the mechanism underlying differences in PWN virulence are unknown. Therefore, in this study, we evaluated the effectiveness of the pine defensive substances α-pinene and H2O2 on the feeding, reproduction, and resistance gene expression patterns of PWN at different levels of virulence to explore the relationship between nematode resistance and its virulence differentiation. The results of this study should further our understanding of the mechanism underlying the virulence differentiation of pine wood nematodes and the sophisticated pathogenesis of pine wilt disease.
Materials and methods
Culturing of PWN
The nematode strain AMA3 that exhibited high virulence was isolated from diseased Pinus thunbergii Parl. from Anhui, China; the strain YW4 that showed low virulence was isolated from diseased Pinus kesiya var. langbianensis (A.Chev.) Gaussen ex Bui from Yunnan, China. Both strains were supplied by the Forest Protection Laboratory, Nanjing Forestry University. Experimental nematodes were cultivated on mats of B. cinerea grown on potato dextrose agar (PDA) plates of 7 g of fresh potatoes, 0.7 g of dextrose, 0.7 g of agar, and 30 mL of sterile water at 25 °C. When the hypha was totally consumed, nematodes at various stages were extracted by a Baermann funnel (Viglierchio and Schmitt 1983) and washed three times with M9 buffer (30 g L‒1 KH2PO4, 60 g L‒1 K2HPO4, and 50 g L‒1 NaCl).
Assay of feeding rate and reproduction of PWN under H2O2 and α-pinene stress
Five hundred samples of AMA3 and YW4 treated with double distilled water (ddH2O), 0.5% Triton X-100, 56.3 mg mL‒1 α-pinene, or 15 mM H2O2, were inoculated on fungal mats of B. cinerea and incubated at 25 °C in three replicates and photographed periodically. Subsequently, the nematodes were isolated from the plates by the Baermann funnel method and counted under a stereomicroscope (Leica DM500; Leica Microsystems, Heerbrugg, Switzerland) when at least one of the treatments had completely consumed the hypha.
Collection of PWN treated with α-pinene and H2O2
Organisms usually respond quickly by regulating defense genes after being stressed (Tsyglakova et al. 2019). Therefore hourly treatments time up to five hours were selected. Samples of 7000 AMA3 and 7000 YW4 were soaked in 1 mL of 56.33 mg mL‒1 α-pinene or 15 mM H2O2 hourly up to five hours in three replicates. The nematodes were washed and stored at ‒80 °C after rapid freezing by liquid nitrogen.
RNA extraction and cDNA synthesis
Samples were ground into powder by a high-speed tissue grinder and TRIzol reagent (Invitrogen, Carlsbad, CA, USA) added. After shaking and mixing, the samples stood for 5 min, then centrifuged (12,000 rpm), and the supernatants transferred to new tubes. After extraction with chloroform and isopropanol, the samples were washed with 75% ethanol and dried. Total RNA was extracted and dissolved in a volume of 20 μL diethyl pyrocarbonate-treated water at 50 °C for 2 min. The purity of total RNA was electrophoresed and measured on a Nanodrop 2000C spectrophotometer (Thermo Fisher Scientific, Waltham, MA, USA). cDNA was synthesized from 1 μg of total RNA according to HiScript® II Q RT SuperMix for qPCR (+ gDNA wiper; Vazyme, Nanjing, China).
Quantitative reverse transcription-polymerase chain reaction
The qRT-PCR primers for BxATG5 (MK387710) and BxCYP33D3 (KM973212) were designed according to their coding sequences (https://www.oligoarchitect.com); the products were 80–150 bp. Primers for BxGST1, BxGST3, Bxy-ctl-1, Bxy-ctl-2, and BxPrx were from previous studies (Vicente et al. 2015; Li et al. 2016; Nian 2017). The Actin gene (EU100952) of the pine woody nematode served as an internal control, with the primers listed in Table 1 (Hu et al. 2020).
SYBR Green Master Mix (Vazyme) and a two-step method were used for qRT-PCR; the conditions were as follows: 95 °C for 5 min for initial denaturation, followed by 40 cycles of 95 °C for 10 s and 60 °C for 34 s. Melting curve analysis was performed using the default program: 95 °C for 15 s, 60 °C for 1 min, and 95 °C for 15 s. Relative expression levels were calculated using the 2−ΔΔCt method of ABI Prism 7500 software (Applied Biosystems, Foster City, CA, USA) (Livak and Schmittgen 2001). qRT-PCR was conducted using three biological and technical replicates.
Statistical analysis
All assays had three biological replicates and three technical replicates. Microsoft Excel software (Microsoft Corp., Redmond, WA, USA) was used for the calculation of mean and standard deviation of the three biological replicates. SPSS software (ver. 17.0; IBM China Company Ltd., Beijing, China) calculated statistical significance by conducting the paired Student’s t-test or Tukey’s HSD test. In all analyses, P < 0.05 indicated statistical significance.
Results
Feeding rate of High and low virulence PWN under conditions of α-pinene and H2O2 stress
The feeding rates of high virulence AMA3 and low virulence YW4 were first evaluated in the absence of stress. The dark area of the culture medium indicated that the hypha had been consumed. And the results show that both AMA3 and YW4 cultured on B. cinerea consumed all hyphae by day 5 (Fig. 1a). AMA3 and YW4 were next treated with Triton X-100, α-pinene, and H2O2 to evaluate their feeding rates. The results show that feeding rates did not differ between AMA3 and YW4 treated with Triton X-100 as a negative control for α-pinene. Both AMA3 and YW4 treated with α-pinene fed more slowly than those treated with Triton X-100, and α-pinene had a stronger inhibitory effect on YW4 than on AMA3. Under conditions of α-pinene stress, AMA3 began to feed on day 6, while YW4 had not begun. On day 7, AMA3 had almost finished feeding whereas YW4 had just begun (Fig. 1b). Under conditions of H2O2 stress, AMA3 had consumed a small portion of hyphae on day 5, while YW4 had not begun to feed. By day 6, AMA3 had finished feeding, while YW4 had consumed one quarter of the hyphae (Fig. 1c). Therefore, high virulence pine woody nematodes fed more rapidly than low virulence PWN under conditions of α-pinene and H2O2 stress.
Fecundity of the high virulence PWN compared with the low virulence PWN under conditions of α-pinene and H2O2 stress
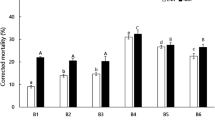
Levels of reproduction were similar for high virulence AMA3 and low virulence YW4 in the absence of stress (Fig. 2a). AMA3 and YW4 were then treated with Triton X-100, α-pinene, or H2O2. The results show that there was no difference in reproduction between AMA3 and YW4 treated with Triton X-100 as a negative control for α-pinene. The α-Pinene had no effect on the reproduction of AMA3 but markedly reduced the reproduction of YW4. The reproduction of AMA3 was more than three times higher than YW4 under conditions of α-pinene stress (Fig. 2b). Under H2O2 stress, the reproduction of AMA3 was normal while that of YW4 was inhibited. The reproduction of AMA3 was almost two-fold that of YW4 under conditions of H2O2 stress (Fig. 2c). Therefore, the fecundity of high virulence PWN was greater than that of low virulence nematodes under conditions of α-pinene and H2O2 stress.
Reproduction of pine wood nematodes with different levels of virulence; AMA3 and YW4 treated with ddH2O, α-pinene, Triton X-100, and H2O2. a ddH2O. b α-pinene. Triton X-100 served as negative control. c H2O2. Error bars indicate SE (n = 3); letters indicate significant differences at the 5% level by paired Student’s t-test or Tukey’s HSD test
Expression levels of BxATG5 in PWN with different virulence levels under conditions of α-pinene and H2O2 stress
The autophagy gene BxATG5 was reported to be related to the stress resistance of pine wood nematodes (Liu et al. 2019a). The expression of BxATG5 was examined in high virulence AMA3 and low virulence YW4 exposed to α-pinene and H2O2 hourly up to 5 h. The Actin gene (EU100952) of PWN served as an internal control. Under conditions of α-pinene stress, the expression level of BxATG5 in AMA3 showed two peaks at 2 and 4 h, with the first peak being much higher than the second. The expression level of BxATG5 in YW4 peaked at 4 h and was lower than the first peak of AMA3. The expression of BxATG5 in AMA3 was significantly upregulated more than five-fold by exposure to α-pinene for 2 h, but in YW4 it was downregulated almost 0.7-fold (Fig. 3a). These results indicate that the expression variation response to α-pinene of BxATG5 in AMA3 was greater than in YW4. Under conditions of H2O2 stress, the expression of BxATG5 showed significant upregulation at 2 h and remained on high level in AMA3. However, the expression of BxATG5 in YW4 increased slightly at 2 h, decreased immediately at 3 hand slowly returned to normal. AMA3 showed upregulated expression of BxATG5 by almost four-fold upon exposure to H2O2 for 2 h, while the expression of BxATG5 in YW4 showed less than two-fold increase at the same time point. Moreover, AMA3 showed a higher level of expression of BxATG5 at all time periods compared to YW4 (Fig. 3b). Therefore, the response of BxATG5 to H2O2 stress was stronger and of longer duration in high virulence PWN.
Detoxification gene response to α-pinene in high virulence and low virulence PWN
We examined the expression patterns of the cytochrome P450 gene BxCYP33D3 and the GST genes BxGST1 and BxGST3 in high virulence AMA3 and low virulence YW4 exposed to α-pinene by qRT-PCR. Under conditions of α-pinene stress, the expression level of BxCYP33D3 in AMA3 peaked at 2 h and then decreased to the normal level by 5 h. In YW4, a smaller peak was observed at 2 h and then returned to normal by 3 h. AMA3 showed rapid upregulation of BxGST1 expression to a peak at 2 h followed by a decrease to a low level, while the expression of BxGST1 in YW4 remained normal throughout. There was a surge in the expression level of BxGST3 in AMA3 at 2 h, and the expression remained normal at other time points. The expression of BxGST3 in YW4 increased slightly up to 4 h. In AMA3, the expression levels of the detoxification genes BxCYP33D3, BxGST1, and BxGST3 peaked upon exposure to α-pinene for 2 h, showing 1.5-, 2.8-, and 12.7-fold increases, respectively, whereas no such upregulation was detected in YW4 (Fig. 4). These results indicate that AMA3 is more efficient at upregulating detoxification genes to reduce damage by α-pinene in comparison with YW4. Therefore, the virulence of PWN was dependent on its ability to upregulate genes related to the detoxification of α-pinene.
Expression patterns of the cytochrome P450 gene BxCYP33D3 and glutathione S-transferases genes BxGST1 and BxGST3 in pine wood nematodes with different levels of virulence under conditions of α-pinene stress; a BxCYP33D3, b BxGST1, c BxGST3; error bars indicate SE (n = 3), letters indicate significant differences at the 5% level by Tukey’s HSD test
Effects of H2O2 on the expression of antioxidant genes in high virulence and low virulence PWN
The expression of the CAT genes Bxy-ctl-1 and Bxy-ctl-2 and the peroxidase gene BxPrx in high virulence AMA3 and low virulence YW4 exposed to H2O2 by qRT-PCR were examined. Bxy-ctl-1 expression patterns were similar in AMA3 and YW4, increasing slowly to peak with H2O2 treatment for 4 h followed by a decrease by 5 h under conditions of H2O2 stress. However, the overall expression level of Bxy-ctl-1 in AMA3 was higher than in YW4; the peak of Bxy-ctl-1 in AMA3 was 1.4-fold higher than in YW4 (Fig. 5a). The expression of Bxy-ctl-2 in AMA3 increased significantly after exposure to H2O2 for 1 h, returned to normal by 4 h, and increased again at 5 h. YW4 showed downregulation of Bxy-ctl-2 expression at 4 h and upregulation at 5 h under conditions of H2O2 stress. Importantly, AMA3 showed a higher expression level of Bxy-ctl-2 than YW4 throughout the period of hydrogen peroxide treatment (Fig. 5b). The trend of expression by BxPrx in AMA3 was consistent with that in YW4 under conditions of H2O2 stress, showing a gradual increased to a peak, followed by a decrease, and then increasing again. Importantly, AMA3 also showed a higher expression level of BxPrx than YW4 during the period of H2O2 treatment. (Fig. 5c), according to the expression of antioxidant genes, suggesting that the antioxidant capacity of high virulence PWN is stronger than that of low virulence PWN.
Expression patterns of the catalase genes Bxy-ctl-1 and Bxy-ctl-2 and peroxidase gene BxPrx of pine wood nematodes with different levels of virulence under conditions of H2O2 stress; a Bxy-ctl-1, b Bxy-ctl-2, c BxPrx; error bars indicate SE (n = 3); letters indicate significant differences at the 5% level by Tukey’s HSD test
Discussion
Hydrogen peroxide and α-Pinene are closely related to the resistance of pine species to pine wood nematodes. Therefore, the differences in virulence of PWN are likely related to the responses to these defense molecules. Tan et al. (2009) reported that α-pinene had no effect on the fertility of PWN. However, Li et al. (2019) noted that the reproduction of the nematodes was severely inhibited by 56.3 mg mL‒1 of α-pinene. In this study, we examined the impact of α-pinene and H2O2 on the feeding rate and fecundity of PWN at different levels of virulence and found that both compounds markedly reduced feeding rates and reproduction of YW4, but not of AMA3. These results show that the differential resistance of the nematodes to pine defensive substances is a key factor in their pathogenic differentiation.
The autophagy gene ATG5 is a central factor involved in autophagosome assembly in Saccharomyces cerevisiae Desm., and its protein product has an important role in ubiquitin-like modifications in the process of autophagy (Matsushita et al. 2007). Miller et al. (2008) noted that ATG5 has major roles in B cell survival during development. Previous studies showed that α-pinene and ROS production in pine species were induced after infection with PWN (Liu et al. 2008). H2O2 and α-Pinene induced significant upregulation of BxATG5 expression in PWN (Liu et al. 2019a). We treated the nematodes with different virulence levels with α-pinene and H2O2 in vitro. The expression of BxATG5 in high virulence PWN peaked at 2 h, while in low virulence PWN decreased under conditions of α-pinene stress. High virulence PWN retained a higher expression level of BxATG5 than low virulence PWN when stressed with H2O2. Moreover, autophagy assists eukaryotes to degrade aberrant cytoplasmic components for cellular homeostasis (Boya et al. 2013; Liu et al. 2019c). High virulence pine wood nematodes showed significant upregulation of BxATG5 expression earlier than low virulence PWN under conditions of α-pinene and H2O2 stress. Therefore, high virulence PWN may have a greater ability to remove the deleterious effects and recycle α-pinene and H2O2 by activating autophagy genes earlier.
The cytochrome P450s of insects can help detoxify pesticides (Yue et al. 2007; Xu et al. 2009). The Ce-GST-p24 of the nematode, Caenorhabditis elegans Maupas was closely related to xenobiotic detoxification (Leiers et al. 2003). Tsai et al. (2016) reported transcriptomic differences in PWN from B. cinerea and pine species, and Li et al. (2019) conducted a comparative transcriptome analysis of PWN treated with sterile water and α-pinene. Both demonstrated upregulated expression of cytochrome P450 and GST family genes. These genes of PWN show significant roles in responding to α-pinene stress. Moreover, the expression of cytochrome P450 genes, related to the vitality, transmissibility, reproduction, pathogenicity, and metabolism of toxic substances of PWN, were shown to be involved in the response of PWN to the accumulation of α-pinene in pine species (Xu et al. 2015; Wang et al. 2017). Silencing of BxGST1 and BxGST3 inhibited the feeding, reproduction, and pathogenicity of PWN (Nian 2017). We further found that high virulence pine wood nematodes showed rapid upregulation of BxCYP33D3, BxGST1, and BxGST3 expression upon exposure to α-pinene, while low virulence PWN responded considerably more slowly, suggesting that differential responses to α-pinene in PWN with different virulence are mediated by detoxification genes.
CATs, which are widespread among Animalia, Plantae, and Fungi, are able to reduce H2O2 to water and to oxidize it to molecular oxygen (Zamocky et al. 2008). The regulation of CAT gene expression shows significant role in determining the levels of CAT proteins (Kodydkova et al. 2014). In pengze crucian carp (Carassius auratus L.), the content of active oxygen in the liver increases on stimulation with cadmium ions and the expression of CAT is also upregulated, indicating that CAT shows significant role in the antioxidant defense system (Li et al. 2013). In Arabidopsis thaliana (L.) Heynh., CAT1 helps to remove hydrogen peroxide from environmental stresses. CAT2 and CAT3 separately remove H2O2 under light and dark conditions to maintain the organism’s balance (Du et al. 2008). The CAT protein family of the fungal entomopathogen Beauveria bassiana (Bals.-Criv.) Vuill.consists of CAT-A (spore-specific), CAT-B (secreted), CAT-P (peroxisomal), CAT-C (cytoplasmic), and CAT-D (secreted peroxidase/catalase). These five CAT proteins function redundantly in regulating the tolerance of fungi to hyperosmotic or fungicil stress (Wang et al. 2013). Kim et al. (1989) discovered that oxidative stress can activate the expression of Peroxiredoxin in S. cerevisiae Desm.. In Haemonchus contortus Rudolphi, HcPrx2 can protect the nematode against oxidative damage (Hudson et al. 2011). Previous studies cloned the CAT genes Bxy-ctl-1 and Bxy-ctl-2 and the peroxidase gene BxPrx from PWN, which are related to the elimination of H2O2 produced by the host tree (Li et al. 2011; Vicente et al. 2015). We found upregulation of Bxy-ctl-1, Bxy-ctl-2, and BxPrx expression in PWN within 5 h of H2O2 stress, and the magnitude of upregulation was greater with high virulence than with low virulence PWN. Vicente et al. (2015) also found that the changes in expression levels of Bxy-ctl-1 and Bxy-ctl-2 in response to different concentrations of H2O2 were greater in high virulence than low virulence pine wood nematodes. Therefore, the antioxidant activity of high virulence PWN was stronger than that of low virulence PWN, which may have a significant role in the virulence differentiation mechanism of PWN. The resistance mechanisms of nematodes with different degrees of virulence should be investigated further to better understand virulence differentiation in PWN.
Conclusions
Feeding rate and fecundity of high virulence pine wood nematodes were significantly higher than for low virulence nematodes under conditions of α-pinene and H2O2 stress, suggesting that high virulence pine wood nematodes have greater resistance than low virulence PWN. Moreover, the expression levels of the main resistance genes, including autophagy genes, detoxification genes, and antioxidant genes, in high virulence pine wood nematodes were all greater than in low virulence PWN under conditions of α-pinene and H2O2 stress. These results suggest that the differential resistance of PWN with different degrees of virulence to α-pinene and H2O2 is mediated by nematode resistance genes. This study will increase our understanding of the mechanism of virulence differentiation in PWN, and provides insight into the pathogenic mechanism of pine wood nematodes.
References
Aikawa T, Kikuchi T (2007) Estimation of virulence of Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae) based on its reproductive ability. Nematology 9(3):371–377. https://doi.org/10.1163/156854107781352007
Baker CJ, Orlandi EW (1995) Active oxygen in plant pathogenesis. Annu Rev Phytopathol 33(1):299–321
Boya R, Reggiori R, Codogno P (2013) Emerging regulation and functions of autophagy. Nat Cell Biol 15(7):713–720. https://doi.org/10.1038/ncb2815
Cheng HR, Lin MS, Li WQ, Fang ZD (1983) Pine wilt disease on Pinus thunbergii in Nanjing. For Pest Dis 4:1–5 (in Chinese)
Ding XL, Ye JR, Lin SX, Wu XQ, Li DW, Nian B (2016) Deciphering the molecular variations of pine wood nematode Bursaphelenchus xylophilus with different virulence. PLoS ONE 11(5):e0156040. https://doi.org/10.1371/journal.pone.0156040
Du YY, Wang PC, Chen J, Song CP (2008) Comprehensive functional analysis of the catalase gene family in Arabidopsis thaliana. J Integr Plant Biol 50(10):1318–1326. https://doi.org/10.1111/j.1744-7909.2008.00741.x
Eo J, Takemoto S, Otobe K (2011) Is there a relationship between the intrinsic rate of propagation and in-vitro migration and virulence of the pinewood nematode, Bursaphelenchus xylophilus. Eur J Plant Pathol 130:231–237. https://doi.org/10.1007/s10658-011-9748-8
Futai K (2013) Pine wood nematode, Bursaphelenchus xylophilus. Annu Rev Phytopathol 51(51):61–83. https://doi.org/10.1146/annurev-phyto-081211-172910
He LX, Ji J, Qiu XW, Zhang LP (2014) Occurrence and control measures of pine wood nematode around the world. J For Eng 28(3):8–13 (in Chinese)
He LX, Wu XQ, Yu LZ, Ji J, Ye JR (2010) The difference of H2O2 and oxidative enzyme in the interaction of different resistance pines and Bursaphelenchus xylophilus. J Nanjing For Univ (Nat Sci Edn) 34(6):13–17. https://doi.org/10.1016/j.molbiopara.2010.11.006(inChinese)
Huang L, Wang P, Tian MQ, Zhu LH, Ye JR (2019) Major sperm protein BxMSP10 is required for reproduction and egg hatching in Bursaphelenchus xylophilus. Exp Parasitol 197:51–56. https://doi.org/10.1016/j.exppara.2019.01.004
Hudson AL, Sotirchos IM, Davey MW (2011) The activity and hydrogen peroxide sensitivity of the peroxiredoxins from the parasitic nematode Haemonchus contortus. Mol Biochem Parasitol 176:17–24
Hu LJ, Wu XQ, Li HY, Wang YC, Huang X, Wang Y, Li Y (2020) BxCDP1 from the pine wood nematode Bursaphelenchus xylophilus is recognized as a novel molecular pattern. Mol Plant Pathol. https://doi.org/10.1111/mpp.12939
Kikuchi T, Cotton JA, Dalzell JJ, Hasegawa K, Kanzaki N, McVeigh P, Takanashi T, Tsai IJ, Assefa SA, Cock PJA, Otto TD, Hunt M, Reid AJ, Sanchez-Flores A, Tsuchihara K, Yokoi T, Larsson MC, Miwa J, Maule AG, Sahashi N, Jones JT, Berriman M (2011) Genomic insights into the origin of parasitism in the emerging plant pathogen Bursaphelenchus xylophilus. PLoS Pathog 7(9):e1002219. https://doi.org/10.1371/journal.ppat.1002219
Kim IH, Kim K, Rhee SG (1989) Induction of an antixoidant protein of Saccharomyces cerevisiae by O2, Fe3+, or 2-mercaptoethanol. Proc Natl Acad Sci USA 86:6018–6022
Kodydkova J, Vavrova L, Kocik M, Zak A (2014) Human catalase, its polymorphisms, regulation and changes of its activity in different diseases. Folia Biol 60(4):153–167. https://doi.org/10.1158/2326-6066.CIR-13-0160
Ktistakis Walker SA, Karanasios E (2016) Assembly of early machinery for autophagy induction: novel insights from high resolution microscopy. Oncotarget 7(52): 85678–85679. https://doi.org/10.18632/oncotarget.13144
Leiers BR, Kampkotter A, Grevelding CG, Link CD, Johnson TE, Henkle-Duhrsen K (2003) A stress-responsive glutathione S-transferase confers resistance to oxidative stress in Caenorhabditis elegans. Free Radic Biol Med 34(11):1405–1415. https://doi.org/10.1016/S0891-5849(03)00102-3
Lewinsohn E, Savage TJ, Gijzen M, Croteau R (1993) Simultaneous analysis of monoterpenes and diterpenoids of conifer oleoresin. Phytochem Anal 4(5):220–225. https://doi.org/10.1002/pca.2800040506
Li LL, Tan JJ, Chen FM (2018) Bacillus pumilus strain LYMC-3 shows nematicidal activity against Bursaphelenchus xylophilus via the production of a guanidine compound. Biocontrol Sci Technol 28(12):1128–1139. https://doi.org/10.1080/09583157.2018.1514587
Li M, Zheng Y, Liang HW, Zou LH, Sun JJ, Zhang YY, Qin F, Liu SZ, Wang ZZ (2013) Molecular cloning and characterization of cat, gpx1 and Cu/Zn-sod genes in pengze crucian carp (Carassius auratus var. Pengze) and antioxidant enzyme modulation induced by hexavalent chromium in juveniles. Comput Biochem Physiol Part C Toxicol Pharmacol 157(3):310–321. https://doi.org/10.1016/j.cbpc.2013.02.003
Li YX, Meng FL, Deng X, Wang X, Feng YQ, Zhang W, Pan L, Zhang XY (2019) Comparative transcriptome analysis of the pinewood nematode Bursaphelenchus xylophilus reveals the molecular mechanism underlying its defense response to host-derived α-pinene. Int J Mol Sci 20(4):911. https://doi.org/10.3390/ijms20040911
Li Z, Liu XX, Chu YN, Wang Y, Zhang QW, Zhou XG (2011) Cloning and characterization of a 2-Cys peroxiredoxin in the pine wood nematode, Bursaphelenchus xylophilus, a putative genetic factor facilitating the infestation. Int J Biol Sci 7(6):823–836. https://doi.org/10.7150/ijbs.7.823
Li Z, Zhang QW, Zhou XG (2016) A 2-Cys peroxiredoxin in response to oxidative stress in the pine wood nematode Bursaphelenchus xylophilus. Sci Rep 6(1):27438. https://doi.org/10.1038/srep27438
Liu HB, Rui L, Feng YQ, Wu XQ (2019) Molecular characterization and functional analysis of three autophagy genes, BxATG5, BxATG9, and BxATG16Bursaphelenchus xylophilus. Int J Mol Sci 20(18):3769. https://doi.org/10.3390/ijms20153769
Liu HB, Rui L, Feng YQ, Wu XQ (2020) Autophagy contributes to resistance to the oxidative stress induced by pine reactive oxygen species metabolism, promoting infection by Bursaphelenchus xylophilus. Pest Manage Sci. https://doi.org/10.1002/ps.5823
Liu HB, Wu F, Wu XQ, Ye JR (2019) Differential effects of rapamycin on Bursaphelenchus xylophilus with different virulence and differential expression of autophagy genes under stresses in nematodes. Acta Biochim Biophys Sin 51:254–262. https://doi.org/10.1093/abbs/gmy172
Liu HB, Wu XQ, Feng YQ, Rui L (2019) Autophagy contributes to the feeding, reproduction, and mobility of Bursaphelenchus xylophilus at low temperatures. Acta Biochim Biophys Sin 00(00):1–9. https://doi.org/10.1093/abbs/gmz068
Liu J, Wu XQ, Ying CX, He LX, Ye JR (2008) Study on superoxide anion of Pinus thunbergii infected by Bursaphelenchus xylophilus and B mucronatus in the earlier interaction. J Nanjing For Univ (Nat Sci Edn). 32(6):24–28 (in Chinese)
Livak K, Schmittgen D (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2–ΔΔCt method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Matsushita M, Suzuki NN, Obara K, Fujioka Y, Ohsumi Y, Inagaki F (2007) Structure of Atg5·Atg16, a complex essential for autophagy. J Biol Chem 282(9):6763–6772. https://doi.org/10.1074/jbc.M609876200
Miller BC, Zhao ZJ, Stephenson LM, Cadwell K, Pua HH, Lee HK, Mizushima N, Iwasaki A, He YW, Swat W, Virgin HW (2008) The autophagy gene ATG5 plays an essential role in B lymphocyte development. Autophagy 4(3):309–314. https://doi.org/10.4161/auto.5474
Nian B (2017) Cloning and Functional Analysis of Glutathione S-transferase Gene BxGST3 and BxGST1 in Bursaphelenchus xylophilus. Nanjing Forestry University, Nanjing
Ogura N, Nakajima Z (2002) In vitro occurrence of dispersal fourth stage juveniles in Bursaphelenchus xylophilus co-incubated with Monochamus alternatus. Jpn J Nematol 32(2):53–59
Shinya R, Takeuchi Y, Ichimura K, Takemoto S, Futai K (2012) Establishment of a set of inbred strains of the pine wood nematode, Bursaphelenchus xylophilus (Aphelenchida: Aphelenchoididae), and evidence of their varying levels of virulence. Appl Entomol Zool 47(4):341–350. https://doi.org/10.1007/s13355-012-0124-8
Tan JJ, Hao DJ, Pan YW, Zhai HY, Fan BQ, Ye JR, Du YB (2009) Effects of several pine volatiles on behavior of Bursaphelenchus xylophilus. J Northeast For Univ 12:60–61 (in Chinese)
Tiao N, Fan JT, Fang YL, Sun JH (2006) Changes in contents of host volatile terpenes under different damaged states and electroantennogram response of Monochamus alternatus Hope to these volatiles. Acta Entomol Sin 49:179–188. https://doi.org/10.16380/j.kcxb.2006.02.003 (in Chinese)
Tsai IJ, Tanaka R, Kanzaki N, Akiba M, Yokoi T, Espada M, Jones JT, Kikuchi T (2016) Transcriptional and morphological changes in the transition from mycetophagous to phytophagous phase in the plant-parasitic nematode Bursaphelenchus xylophilus. Mol Plant Pathol 17(1):77–83. https://doi.org/10.1111/mpp.12261
Tsyglakova M, McDaniel D, Hodes GE (2019) Immune mechanisms of stress susceptibility and resilience: Lessons from animal models. Front Neuroendocrinol 54:100771
Vellosillo T, Vicente J, Kulasekaran S, Hamberg M, Castresana C (2010) Emerging complexity in reactive oxygen species production and signaling during the response of plants to pathogens. Plant Physiol 154(2):444–448. https://doi.org/10.1104/pp.110.161273
Vicente CSL, Ikuyo Y, Mota M, Hasegawa K (2013) Pinewood nematode-associated bacteria contribute to oxidative stress resistance of Bursaphelenchus xylophilus. BMC Microbiol 13(1):299. https://doi.org/10.1186/1471-2180-13-299
Vicente CSL, Yoriko I, Ryoji S, Mota M, Hasegawa K (2015) Catalases induction in high virulence pinewood nematode Bursaphelenchus xylophilus under hydrogen peroxide-induced stress. PLoS One 10(4):e0123839. https://doi.org/10.1371/journal.pone.0123839
Viglierchio DR, Schmitt RV (1983) On the methodology of nematode extraction from field samples: Baermann funnel modifications. J Nematol 15:438–444
Wang X, Li YX, Liu ZY, Lv Q, Jia XZ, Zhang XY (2017) Relationship between the cytochrome P450 gene of pine wood nematode and the accumulation of pine pinene. Scientia Silvae Sinicae 53(6):105–110 (in Chinese)
Wang Y, Yamada T, Sakaue D, Suzuki K (2005) Variations in life history parameters and their influence on rate of population increase of different pathogenic isolates of the pine wood nematode, Bursaphelenchus xylophilus. Nematology 7:459–467. https://doi.org/10.1163/156854105774355545
Wang ZL, Zhang LB, Ying SH, Feng MG (2013) Catalases play differentiated roles in the adaptation of a fungal entomopathogen to environmental stresses. Environ Microbiol 15(2):409–418
Wu Y, Wickham JD, Zhao L, Sun JH (2019) CO2 drives the pine wood nematode off its insect vector. Curr Biol 29(13):R619–R620
Xu JY, Wang BW, Li DL (2019) Response pattern and functional validation of pine wood nematode (Bursaphelenchus xylophilus) BxPrx. J Hum Agric Univ (Nat Sci) 45(5):491–495 (in Chinese)
Xu XL, Wu XQ, Ye JR, Huang L (2015) Molecular characterization and functional analysis of three pathogenesis-related cytochrome P450 Genes from Bursaphelenchus xylophilus (Tylenchida: Aphelenchoidoidea). Int J Mol Sci 16(3):5216–5234. https://doi.org/10.3390/ijms16035216
Xu YQ, Wang JJ, Dou W, Tang PA, An FM (2009) Identification, characterization, and expression of P450 gene encoding CYP6BQ13v2 from the red flour beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Agric Sci China 8(10):1210–1218 (in Chinese)
Yang BJ, Pan HY, Tang J, Wang YY, Wang LF (2003) Pine wood nematode disease. Forestry Publishing House, Beijing, pp 45–48
Ye JR (2019) Epidemic status of pine wilt disease in China and its prevention and control techniques and counter measures. Scientia Silvae Sinicae 55(9):1–10 (in Chinese)
Yu LZ, Wu XQ, Ye JR, Zhang SN (2013) Relationships between nitric oxide response signal and external factors during the early interaction between Pinus thunbergii and Bursaphelenchus xylophilus. Chin J Appl Ecol 24:646–652 (in Chinese)
Yue LN, Yang YH, Wu SW, Wu YD (2007) Cloning and mRNA expression levels of cytochrome P450 genes CYP6AE12 and CYP9A18 in the cotton bollworm, Helicoverpa armigera (Hübner). Acta Entomol Sin 50(3):234–240 (in Chinese)
Zamocky M, Furtmuller PG, Obinger C (2008) Evolution of catalases from bacteria to humans. Antioxid Redox Signaling 10(9):1527–1548. https://doi.org/10.1089/ars.2008.2046
Zhao L, Zhang X, Wei Y, Zhou J, Zhang W, Qin PJ, Chinta S, Kong XB, Liu YP, Yu HY, Hu SN, Zou Z, Butcher RA, Sun JH (2016) Ascarosides coordinate the dispersal of a plant-parasitic nematode with the metamorphosis of its vector beetle. Nat Commun 7:12341. https://doi.org/10.1038/ncomms12341
Zhou LF, Chen FM, Xie LY, Pan HY, Ye JR (2017) Genetic diversity of pine-parasitic nematodes Bursaphelenchus xylophilus and Bursaphelenchus mucronatus in China. For Pathol 47(4):e12334. https://doi.org/10.1111/efp.12334
Zulak KG, Bohlmann J (2010) Terpenoid biosynthesis and specialized vascular cells of conifer defense. J Integr Plant Biol 52(1):86–97. https://doi.org/10.1111/j.1744-7909.2010.00910.x
Author information
Authors and Affiliations
Contributions
Lin Rui designed the study, conducted experiments, analyzed the data and prepared the manuscript; Hong-Bin Liu designed the study and revised the manuscript; Rui Liang conducted experiments; Xiao-Qin Wu designed the study, acquired the data, contributed reagents/materials/analysis tools and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: The work was funded partly by the National Key Research and Development Program of China (No. 2018YFD0600203), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), and Innovation and Entrepreneurship Training Program for Students of Jiangsu Higher Education Institutions (SPITP)/ Innovation and Entrepreneurship Training Program for Students of Nanjing Forestry University (No.201710298047Z).
The online version is available at http://www.springerlink.com.
Corresponding editor: Tao Xu.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rui, L., Liu, H., Liang, R. et al. Resistance genes mediate differential resistance to pine defensive substances α-Pinene and H2O2 in Bursaphelenchus xylophilus with different levels of virulence. J. For. Res. 32, 1753–1762 (2021). https://doi.org/10.1007/s11676-020-01182-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-020-01182-y