Abstract
The influences of trait diversity (i.e., the niche complementarity effect) and functional composition (i.e., the mass ratio effect) on aboveground biomass (AGB) is a highly debated topic in forest ecology. Therefore, further studies are needed to explore these mechanisms in unstudied forest ecosystems to enhance our understanding, and to provide guidelines for specific forest management. Here, we hypothesized that functional composition would drive AGB better than trait diversity and stem size inequality in the (sub-) tropical forests of Nepal. Using data from 101 forest plots, we tested 25 structural equation models (SEMs) to link elevation, stem DBH inequality, trait diversity (i.e., trait richness, evenness, dispersion and divergence), functional composition [i.e., community-weighted of maximum height mean (CWM of Hmax), specific leaf area (CWM of SLA), leaf dry matter content (CWM of LDMC), and wood density (CWM of WD)] and AGB. The best-fitted SEMs indicated that CWM of Hmax promoted AGB while overruling the impacts of trait diversity indices on AGB. However, low trait diversity indices were linked with higher AGB while overruling the effects of CWM of SLA, LDMC and WD on AGB. In addition, AGB decreased with increasing elevation, whereas stem size inequality did not influence AGB. Our results suggest that divergent species’ functional strategies could shape AGB along an altitudinal gradient in tropical forests. We argue that forest management practices should include plant functional traits in the management plan for the co-benefits of biodiversity conservation and carbon sequestration that underpins human wellbeing.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Functional trait diversity and composition, stand structure inequality, topography, soil nutrients and climate conditions shape aboveground biomass (AGB) or productivity in forest ecosystems (Paquette et al. 2015; Prado-Junior et al. 2016; Wang and Ali 2021b; Yuan et al. 2020). As plant traits can affect plant growth, survival, fitness and capabilities (Violle et al. 2007), linking traits with ecosystem functions can represent a better picture of underlying ecological mechanisms as compared to species’ taxonomic identity (Díaz et al. 2007; van der Plas 2019). More importantly, this approach is crucial to identify key functional traits that can substantially enhance AGB, higher atmospheric carbon dioxide absorption and climate change mitigation that could underpin human wellbeing (Díaz et al. 2007). Nonetheless, much debate remains regarding the underlying causes for linking plant trait diversity and functional composition with AGB in forests as compared to species diversity and structural attributes (Ali 2019; van der Plas 2019; Wang and Ali 2021b). Therefore, further inquiries are needed to explore these mechanisms in unstudied forest ecosystems to further enhance our understanding, and to provide guidelines for specific forest management.
Two ecological hypotheses (i.e., the niche complementarity and the mass ratio hypothesis) have been used extensively to interpret the impacts of trait diversity and functional composition on AGB (van der Plas 2019; Wang and Ali 2021b). The niche complementarity hypothesis assumes that higher resource-use efficiency can boost ecosystem functions through the niche differentiation in functionally diverse plant communities (Loreau et al. 2001; Poorter et al. 2017). The mass ratio hypothesis suggests that the traits of the most dominant species play a majority role in regulating AGB that can be measured by the community-weighted mean of a trait (Conti et al. 2013; Finegan et al. 2015; Pyles et al. 2018). Although both hypotheses are well supported by previous studies, the mass ratio mechanism seems to explain AGB better than the niche complementarity effect in forest ecosystems (van der Plas 2019; Wang and Ali 2021b). For instance, acquisitive species may grow fast [i.e., high leaf nutrient content and specific leaf area (SLA)] and could enhance AGB via higher photosynthetic rates. In contrast, conservative species may grow slow [i.e., high wood density (WD) and leaf dry matter content (LDMC)] and yet be associated with hydraulic efficiency and higher volumetric construction that contribute to higher AGB (Chave et al. 2009; Reich 2014). Moreover, positive, negative and/or negligible impacts of functional composition on AGB may be attributable to divergent species’ resource-use strategies in forest communities (Chiang et al. 2016; Prado-Junior et al. 2016; Rosenfield and Muller 2020).
Topographic factors are crucial for shaping plant growth, species distribution, diversity pattern and ecosystem functioning (Jucker et al. 2018; Moeslund et al. 2013; Toledo et al. 2012). Furthermore, local-scale variation in topographic factors primarily leads to habitat differentiation that may affect the community structure of tropical forests (Rodrigues et al. 2020). The status of diversity could be site-specific and elevation-dependent. For example, taxonomic, phylogenetic and trait diversity typically decrease at higher altitudes (Xu et al. 2019a, 2019b) as a consequence of severe conditions with the effect of environmental filtering (Schöb et al. 2012; Stagnol et al. 2016). Thus, it is obvious that the lower altitude region is richer in diversity than mid to high elevation regions. In addition, it has been recognized that compact canopy packing with several vertical strata could facilitate light capture and resource-use among co-occurring individuals that underpin higher AGB through the niche complementarity effects (Ali 2019; Jucker et al. 2015; Yachi and Loreau 2007). Thus, the direct and indirect effects of stand structural complexity (i.e., stem size inequality) on AGB may modulate the impacts of trait diversity and functional composition on AGB in forest ecosystems (Poorter et al. 2017; Yuan et al. 2020). However, stem size inequality, trait diversity and functional composition are not only affecting AGB directly, but also, they are mediating the responses of AGB to topographic and climatic factors, and soil fertility (Chiang et al. 2016; Poorter et al. 2017; Wang and Ali 2021a; Yuan et al. 2020). For instance, microclimate conditions change along altitudinal gradients due to differences in some important above- and below-ground resource availabilities (Jucker et al. 2018; Rodrigues et al. 2020). Thus, elevation can influence species evolutionary history and their coexistence which in turn can shape the relationships among trait diversity indices, CWM values, and stem size inequality with AGB (Chun et al. 2020; Jucker et al. 2018).
We have recently reported that higher stand density of coexisting species rather than stem size inequality increases AGB along an altitudinal gradient (Kunwar et al. 2021b), whereas the divergent effects of phylogenetic diversity indices and species richness determine AGB better than stem size inequality across tropical forests in Nepal (Kunwar et al. 2021a). Here, we attempt to clarify the effects of stem DBH inequality, multi-trait diversity indices (i.e., trait richness, evenness, dispersion, and divergence), and functional composition values [i.e., CWM of leaf and wood traits such as CWM of Hmax (i.e., functional dominance), CWM of SLA, CWM of LDMC and CWM of WD] on AGB along altitudinal gradients in tropical forests. According to the specific contribution of this study, we asked the following main research questions: (1) Do the joint impacts of trait diversity indices and CWM values regulate AGB? (2) What is the most important predictor—trait diversity or functional composition or both—of AGB in tropical forests? (3) How do abiotic (i.e., elevation) and biotic (i.e., trait diversity and functional composition) factors explain AGB across local forest types? We hypothesize that functional composition will drive AGB better than trait diversity and stem size inequality in (sub-) tropical forests. In addition, as shown in the proposed conceptual model (Fig. 1a), we addressed a debated research question in tropical forests, i.e., what are the direct and indirect effects of stem DBH inequality, trait diversity indices and CWM values on AGB along altitudinal gradients in tropical forests?
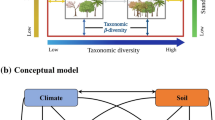
A conceptual model (a) with hypothesized pathways and their explanation (b) for testing the proposed questions and hypothesis in the tropical forests of Nepal. Images for forest diversity, structure and aboveground biomass are provided for illustration purposes only. FRic trait richness; FEve trait evenness; FDis trait dispersion; FDiv trait divergence; and CWMHmax, CWMSLA, CWMLDMC, and CWMWD stand for the community-weighted plant maximum height mean, specific leaf area, leaf dry matter content, and wood density, respectively
Materials and methods
Study area and forest inventory data assembly
This study was conducted in (sub-) tropical forests (28.7708° to 29.2621° N and 80.8975° to 80.6424° E) that covered five sites, and the altitude ranged from 218 to 1850 m a.s.l. in Sudurpashchim Province of Nepal (see map in Supplementary Fig. S1) (Kunwar et al. 2021a, 2021b). The mean annual temperature and precipitation were 16 °C to 23 °C and 1280 to 1470 mm, respectively. The forest soil contained 27.5 to 33 cmol kg−1 of cation exchange capacity, and soil pH values ranged in 5.05–5.15. We performed a standard forest protocol to measure the total height and diameter at breast height (DBH) of all stems having DBH ≥ 10 cm in 101 (each 0.05 ha in size) circular plots that were established randomly on five different sites in 2019. Specifically, tropical forests’ sites located at lower elevations were characterized by Shorea robusta as a dominant species, whereas sub-tropical coniferous forests’ sites with the dominance of Pinus roxburghii were located at higher elevations (Jackson et al. 1994).
Quantification of predictors used in the analyses
In this study, trait diversity indices (trait richness, evenness, dispersion and divergence), functional composition values (CWM of Hmax, WD, SLA and LDMC), elevation, and stem DBH inequality were quantified to explain patterns in AGB (see Fig. 1; Table S1).
Stem size inequality was computed through the coefficient of variation (CV) of DBH within each plot (Ali 2019). CWM values and trait diversity indices were quantified by available measurements of some species’ leaf traits in the TRY Plant Trait Database (Kattge et al. 2020). Leaf traits of other species were measured during the plant growing period following the standard field and laboratory protocols in 2020 (Cornelissen et al. 2003). More specifically, three to five young and mature individual trees per species were selected, and then at least 10 mature healthy leaves were collected per individual from different positions (i.e., fully and/or mostly sunlit sides) (Ali et al. 2017; Cornelissen et al. 2003). Leaf samples were stored in closed plastic bags and kept cool up to further analysis in the laboratory. The SLA was obtained through a one-sided fresh leaf area divided by its oven-dried mass (dried at 80 °C for 48 h), and LDMC as the oven-dried mass of a leaf divided by its water-saturated fresh mass. In each plot, Hmax of each species was used to determine its maximum height (Prado-Junior et al. 2016). The WD values (i.e., species-level) species were acquired from the global databases (Kattge et al. 2020; Reyes 1992; Zanne et al. 2009). In this study, trait diversity indices and CWM values explicitly relied on a total of four traits, i.e., SLA, LDMC, Hmax and WD, because these traits are recognized as the main indicators of AGB and productivity in (sub-) tropical forests (Ali et al. 2017; Finegan et al. 2015; Poorter and Bongers 2006; Prado-Junior et al. 2016). The CWM of a trait value was computed as the average trait value in the community (i.e., plot) (Garnier et al. 2004), whereas trait diversity indices were determined according to multidimensional trait space, i.e., trait richness, evenness, divergence and dispersion (Mason et al. 2005; Villéger et al. 2008). Both trait diversity indices and CWM values were weighted by species relative basal area as it best predicts AGB (Prado-Junior et al. 2016). Trait values were standardized before calculating the trait diversity indices.
AGB for each tree was estimated through widely used best-fit allometric equation for tropical trees species (Chave et al. 2014):
where, WD, DBH and H stand for species’ wood density, tree diameter at breast height and total tree height, respectively.
Conceptual model development and statistical analyses
Here, we proposed a conceptual model according to the following supposed direct paths: (1) elevation influenced stem DBH inequality, trait diversity, and functional composition; (2) stem DBH inequality affected trait diversity, and functional composition; (3) elevation, stem DBH inequality, trait diversity, and functional composition affected AGB; and (4) relationship between trait diversity and functional composition. Consequently, we also evaluated the indirect impacts of (1) elevation on AGB via stem DBH inequality, trait diversity, and functional composition; and (2) stem DBH inequality on AGB through trait diversity and functional composition. The known theoretical relationships were used to develop a conceptual model and to test the research questions by applying the structural equation models (SEMs) (Grace et al. 2016; Wang and Ali 2021b). However, we had four trait diversity (i.e., trait richness, evenness, divergence and dispersion) indices and four functional composition values (CWM of SLA, LDMC, Hmax and WD) values, and hence we constructed several SEMs according to the following four approaches. First of all, one comprehensive SEM was constructed based on the combination of latent variables, i.e., latent trait diversity indices (hereafter refers to Latent-FTD; i.e., incorporating trait richness, evenness, divergence and dispersion) and latent CWM values (hereafter refers to Latent-CWMt; i.e., incorporating CWM of SLA, LDMC, Hmax and WD). By doing so, we found that tested SEM was converged, but model-fit statistics were less than suggested cut-off values (see Tables S2 and S3). In the second step, we constructed four SEMs based on the combinations of Latent-FTD and each of CWM values (i.e., either CWM of SLA, LDMC, Hmax and WD) that indicated the suitable model-fit (Table S2). Then, four extra SEMs were constructed based on the combinations of Latent-CWMt and each of FTD index (i.e., either trait richness, evenness, divergence or dispersion) that showed poor fit to the data (Table S2). The effects of predictors (i.e., direct and indirect) on AGB in these four SEMs showed in Tables S4–S7. Finally, we constructed 16 SEMs based on the possible combinations of trait diversity indices (i.e., either trait richness, evenness, divergence or dispersion) and CWM values (i.e., either CWM of SLA, LDMC, Hmax and WD) that all were saturated and over-fitted (Table S2).
Bivariate relationships of all predicted paths according to SEMs were assessed using regressions analysis and Pearson’s correlation coefficients. To simplify and to complement the complex SEMs outcomes, we performed principal component analysis (PCA) including all predictors and AGB while highlighting local forest types i.e., sub-tropical coniferous forests, tropical evergreen and deciduous forests, and tropical evergreen and mixed hardwood forests. PCA was carried out to show the correlation circles for predictors and AGB across main local forest types, and to clarify the abiotic (i.e., elevation) and biotic (i.e., trait diversity, functional composition and stem size inequality) gradients to explain AGB along two main axes of PCA.
All statistical analyses were conducted in R 3.6.0 (R Development Core Team 2019). For comparison of the effect size of multiple numerical predictors in a model, all those variables were ln-transformed and standardized prior to SEM construction (Zuur et al. 2009). The SEM was performed using the lavaan package (Rosseel 2012). We performed PCA using the factoextra and FactoMineR packages (Husson et al. 2016).
Results
The tested SEMs based on the four possible combinations of Latent-FTD and four CWM values fit the data well, while the relationships of four trait diversity indices to Latent-FTD varied from positive (i.e., trait richness), nonsignificant (i.e., trait divergence) to negative (i.e., trait evenness and dispersion), and indicated overall low FTD (Fig. 2; Tables S8–S11). Specifically, elevation had negative direct effects on CWM of SLA, CWM of LDMC, CWM of WD and AGB (Fig. 2; Tables S8–S11). CWM of Hmax exhibited a positive direct effect on AGB, whereas the direct effects of other CWM values were nonsignificant and weak in effect size (Fig. 2; Tables S8–S11). We noted that trait diversity overruled the effects of CWM of SLA, CWM of LDMC and CWM of WD on AGB, whereas CWM of Hmax overruled the effects of trait diversity on AGB (Fig. 2; Tables S8–S11). As such, trait diversity interacted positively with CWM of Hmax, but showed negative interactions with CWM of SLA (Fig. 2a and b).
Best-fitted structural equation models for evaluating the direct and indirect effects of elevation, stem DBH inequality, latent-FTD, functional (trait) composition [either a CWM of Hmax, b CWM of SLA, c CWM of LDMC, or d CWM of WD] on aboveground biomass in tropical forests. Significant (P < 0.05) positive and negative effects are shown by solid arrows with green and red colors, respectively, whereas black dashed arrows show nonsignificant effects (P > 0.05). R2 is the coefficient of determination. Natural-log transformed and standardized data were used in this analysis. See Tables S4–S7 for the model summary. Abbreviations are provided in Fig. 1 and Table S1
The extra 16 tested SEMs based on all possible combinations of CWM values and trait diversity indices showed that only CWM of Hmax was the main driver of AGB (Figs. S2–S5). Moreover, the negative direct impacts of elevation on CWM of SLA, CWM of LDMC, CWM of WD and AGB did not change in effect size and were similar to the four best-fitted SEMs (Figs. 2; S2–S5). We found various interactions between each CWM value and trait diversity indices that strongly changed among tested 16 SEMs (Figs. S2–S5). The positive direct effect of stem DBH inequality on CWM of SLA was similar in effect size to that of the main SEMs (Figs. 2b and S3c). Except for divergent (i.e., positive and negative) direct effects of trait diversity indices on AGB that strongly associated with CWM values included in tested SEMs, the other observed relationships in four main SEMs were almost similar to16 extra SEMs (Figs. 2; S2–S5). In addition, we found that indirect effects of elevation via mediators (i.e., trait diversity indices, CWM values, and stem DBH inequality) were relatively weak in effect size in comparison to its direct effects on AGB (Tables S8–S11).
Moreover, the bivariate relationships showed that AGB increased significantly with all CWM values, but decreased with trait dispersion and elevation (Fig. 3; Table S12). The bivariate relationships were mostly similar to the hypothesized paths in tested SEMs. A small difference between each assumed path used in the SEMs and bivariate relationships may be attributable to the interdependency of multiple tested variables that might outperform the significant effects of others (Figs. 2 and 3; S2–S5). Pearson’s correlation matrix that indicates the correlation coefficient of each pair of tested predictors is presented in Fig. S6.
Bivariate relationships according to hypothesized paths in structural equation models. Only significant relationships (P < 0.05) are shown here, whereas a summary of all relationships is provided in Table S12. Natural-log transformed and standardized data were used in this analysis. Abbreviations are provided in Fig. 1 and Table S1
The first axis of the PCA (i.e., PC1) explained 37.3% of the variation in studied plots (Fig. 4a). To the PC1 axis, elevation contributed positively whereas CWM of WD, CWM of SLA and CWM of LDMC contributed negatively, indicating that AGB was negatively controlled by elevation (Fig. 4b). The second axis of PCA (i.e., PC2) showed gradients for biotic factors, i.e., species’ functional strategies and trait diversity that explained an additional 17.8% of the variation in studied plots (Fig. 4a). To the PC2 axis, AGB itself contributed positively and was highly positively related to CWM of Hmax and trait richness whereas negatively related to trait dispersion and evenness (Fig. 4c). We also noted that sub-tropical coniferous forest plots were strongly controlled by an abiotic factor (i.e., elevation), whereas tropical evergreen and deciduous forests, and tropical evergreen and mixed hardwood forests plots were strongly controlled by biotic factors (i.e., trait diversity and CWM values), indicating that the observed SEMs relationships could be highly dependent on local forest types (Fig. 4a).
Principal component analysis (PCA) shows a the abiotic and biotic gradients for explaining patterns in aboveground biomass across three forest types that denoted by different colors ellipses, b and c bar plots indicate the loadings of the abiotic and biotic (with the contribution values in % over the bars) on PC1 and PC2 in the study area. Abbreviations are provided in Fig. 1
Discussion
Our study examines the joint importance of the niche complementarity and mass ratio effects on AGB along an altitudinal gradient in tropical forests of Nepal. Our findings showed the positive effects of CWM of Hmax on AGB along higher trait richness gradient, but lower trait evenness and dispersion along decreasing altitudinal gradient. Hence, our findings support the mass ratio effect whereas the negative impacts of high trait diversity or positive impacts of low trait diversity on AGB suggest a negligible effect of niche complementarity effect to explain AGB in studied tropical forests (Chiang et al. 2016; Finegan et al. 2015; van der Plas 2019; Wang and Ali 2021b). However, the PCA suggested that these two ecological mechanisms are not mutually exclusive but work jointly for shaping community assembly processes and functions (Ali et al. 2017; Loreau et al. 2001).
As reported by previous studies around the globe, we also found that CWM of Hmax is a potentially powerful driver of AGB in forests (Ali et al. 2017; Conti et al. 2013; Finegan et al. 2015; Ruiz-Jaen and Potvin 2011). Tall-statured trees are usually linked with higher AGB due to higher stem volume, and their potential capability to capture and use available resources (Stephenson et al. 2014). The higher ability of light use efficiency and root system expansion of taller trees could lead to higher photosynthesis rates and more soil–water and nutrient exploitation, which in turn could enhance plant growth and productivity (Baker et al. 2009; Fauset et al. 2015). However, elevation affected AGB negatively but did not significantly affect the CWM of Hmax which might be attributable to the selection effect (Jucker et al. 2018; Villa et al. 2020). Consequently, we found that the functional dominance of tall-statured trees at low-land tropical forests was associated with higher AGB in comparison with high-altitude sub-tropical forests (Cavanaugh et al. 2014). Moreover, the nonsignificant effects of CWM of WD, LDMC and SLA on AGB indicate that expectations derived from the direct contribution of acquisitive and conservative species to stand growth and productivity might be overruled by the superior role of tall-statured trees with fast resources acquisition and turnover rates (Ali et al. 2017; Rüger et al. 2012). According to the leaf and wood economics spectra and even whole-plant economics spectrum, acquisitive and conservative species may react differently in a community due to ecological trade-offs such as fast versus slow growth and longevity versus mortality rates (Chave et al. 2009; Reich 2014).
Our results illustrate the negative impacts of functional evenness and dispersion, but the positive effect of functional richness, and the nonsignificant effect of functional divergence on AGB, indicating that low trait diversity promotes AGB in studied forests. However, the relationships between trait diversity indices and AGB seem to be related to elevation-dependent resource availability that affects plant growth due to favorable climatic conditions across local forest types (Jucker et al. 2018; Wang and Ali 2021a). For example, we detected that low-land tropical forests were occupied by different functional trait diversity indices as compared to high-altitude subtropical forests, indicating that specific species’ functional strategy can determine AGB (Wang and Ali 2021b). Nevertheless, the negative or nonsignificant impacts of trait diversity on AGB indicates the strong role of niche overlap or functionally-similar species (Chiang et al. 2016; Prado-Junior et al. 2016). As such, we observe the negligible effect of stem DBH inequality on AGB which might be attributable to the structural homogeneity of studied forests (see Kunwar et al. 2021b, for a more detailed explanation) due to the dominance of tall-statured tree species, again indicating the niche overlap or asymmetric competition (Ali 2019; Bourdier et al. 2016). This mechanism might be reasonable because we note that CWM of Hmax overruled the effects of trait diversity on AGB, indicating that niche space might be covered by tall-statured tree species within a community (Chiang et al. 2016; Finegan et al. 2015; Wang and Ali 2021b). Furthermore, the positive interaction between CWM of Hmax and low trait diversity supports the general notion that AGB is strongly controlled by mass ratio effect compared to the niche complementarity effect in tropical forests (Finegan et al. 2015; Prado-Junior et al. 2016; Wang and Ali 2021b).
The CWM of Hmax, LDMC, SLA and WD have usually linked with species’ functional strategies along leaf and wood economics spectra which could greatly determine the species distribution specimens along an altitudinal gradient (Reich 2014; Toledo et al. 2012). As such, we detect that elevation negatively affected the CWM of LDMC, SLA and WD as compared to Hmax which might be due to the community assembly processes in relation to competition for the resource-use across local forest types (Carroll et al. 2011; Reich 2014). PC1 separated forest plots based primarily on elevation and trait values (i.e., the CWM of LDMC, SLA and WD), whereas PC2 separated plots based on AGB, the CWM of Hmax, and measurements of functional trait diversity (i.e., FEve, FRic, and FDis). Along the first axis of PCA, we found that elevation was negatively correlated whereas acquisitive and conservative species’ functional strategies were positively correlated with AGB, indicating the strong role of abiotic or environmental filtering (Fortunel et al. 2014). However, along the second axis of PCA, we found that AGB was positively correlated with CWM of Hmax and negatively correlated with trait diversity metrics (particularly functional evenness and dispersion), indicating the strong role of biotic filtering (Hillebrand et al. 2008; Wang and Ali 2021b). Thus, we argue that studies forests are controlled by both local abiotic and biotic filtering for shaping AGB. We argue that our results will facilitate further studies to tease apart the confounding impacts of species’ functional strategies on AGB within each local forest type by using an extended required number of plots for each forest type (but see Kunwar et al. 2021a for some evidence based on evolutionary diversity indices).
Conclusions
This study shows that the mass ratio effect determines AGB due to the strongest role of tall-statured trees, and hence, the niche complementarity effect seems unimportant in the studied forests. Therefore, it is important to manage tropical forests based on the specific plant strategies that can enhance forest functions. However, we argue that local forest types according to the species composition and diversity along altitudinal gradient should be considered during the plans to manage forest ecosystems well.
Data availability
Datasets and R codes used in the analyses are provided upon reasonable request to the corresponding author.
References
Ali A (2019) Forest stand structure and functioning: current knowledge and future challenges. Ecol Ind 98:665–677. https://doi.org/10.1016/j.ecolind.2018.11.017
Ali A, Yan E-R, Chang SX, Cheng J-Y, Liu X-Y (2017) Community-weighted mean of leaf traits and divergence of wood traits predict aboveground biomass in secondary subtropical forests. Sci Total Environ 574:654–662. https://doi.org/10.1016/j.scitotenv.2016.09.022
Baker TR, Phillips OL, Laurance WF, Pitman NCA, Almeida S, Arroyo L, DiFiore A, Erwin T, Higuchi N, Killeen TJ, Laurance SG, Nascimento H, Monteagudo A, Neill DA, Silva JNM, Malhi Y, López Gonzalez G, Peacock J, Quesada CA, Lewis SL, Lloyd J (2009) Do species traits determine patterns of wood production in Amazonian forests? Biogeosciences 6:297–307. https://doi.org/10.5194/bg-6-297-2009
Bourdier T, Cordonnier T, Kunstler G, Piedallu C, Lagarrigues G, Courbaud B (2016) Tree size inequality reduces forest productivity: an analysis combining inventory data for ten European species and a light competition model. PLoS ONE 11:e0151852. https://doi.org/10.1371/journal.pone.0151852
Carroll IT, Cardinale BJ, Nisbet RM (2011) Niche and fitness differences relate the maintenance of diversity to ecosystem function. Ecology 92:1157–1165. https://doi.org/10.1890/10-0302.1
Cavanaugh KC, Gosnell JS, Davis SL, Ahumada J, Boundja P, Clark DB, Mugerwa B, Jansen PA, O’Brien TG, Rovero F, Sheil D, Vasquez R, Andelman S (2014) Carbon storage in tropical forests correlates with taxonomic diversity and functional dominance on a global scale. Glob Ecol Biogeogr 23:563–573. https://doi.org/10.1111/geb.12143
Chave J, Coomes D, Jansen S, Lewis S, Swenson N, Zanne A (2009) Towards a worldwide wood economics, spectrum. Ecol Lett 12:351–366. https://doi.org/10.1111/j.1461-0248.2009.01285.x
Chave J, Rejou-Mechain M, Burquez A, Chidumayo E, Colgan MS, Delitti WB, Duque A, Eid T, Fearnside PM, Goodman RC, Henry M, Martinez-Yrizar A, Mugasha WA, Muller-Landau HC, Mencuccini M, Nelson BW, Ngomanda A, Nogueira EM, Ortiz-Malavassi E, Pelissier R, Ploton P, Ryan CM, Saldarriaga JG, Vieilledent G (2014) Improved allometric models to estimate the aboveground biomass of tropical trees. Glob Change Biol 20:3177–3190. https://doi.org/10.1111/gcb.12629
Chiang JM, Spasojevic MJ, Muller-Landau HC, Sun IF, Lin Y, Su SH, Chen ZS, Chen CT, Swenson NG, McEwan RW (2016) Functional composition drives ecosystem function through multiple mechanisms in a broadleaved subtropical forest. Oecologia 182:829–840. https://doi.org/10.1007/s00442-016-3717-z
Chun J-H, Ali A, Lee C-B (2020) Topography and forest diversity facets regulate overstory and understory aboveground biomass in a temperate forest of South Korea. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2020.140783
Conti G, Díaz S, Lavorel S (2013) Plant functional diversity and carbon storage—an empirical test in semi-arid forest ecosystems. J Ecol 101:18–28. https://doi.org/10.1111/1365-2745.12012
Cornelissen JHC, Lavorel S, Garnier E, Díaz S, Buchmann N, Gurvich DE, Reich PB, Steege HT, Morgan HD, Heijden MGAVD, Pausas JG, Poorter H (2003) A handbook of protocols for standardised and easy measurement of plant functional traits worldwide. Aust J Bot 51:335–380. https://doi.org/10.1071/BT02124
Díaz S, Lavorel S, de Bello F, Quetier F, Grigulis K, Robson M (2007) Incorporating plant functional diversity effects in ecosystem service assessments. Proc Natl Acad Sci USA 104:20684–20689. https://doi.org/10.1073/pnas.0704716104
Fauset S, Johnson MO, Gloor M, Baker TR, Monteagudo A, Brienen RJ, Feldpausch TR, Lopez-Gonzalez G, Malhi Y, Ter Steege H (2015) Hyperdominance in Amazonian forest carbon cycling. Nat Commun 6:1–9
Finegan B, Pena-Claros M, de Oliveira A, Ascarrunz N, Bret-Harte MS, Carreno-Rocabado G, Casanoves F, Diaz S, Eguiguren Velepucha P, Fernandez F, Carlos Licona J, Lorenzo L, Salgado Negret B, Vaz M, Poorter L (2015) Does functional trait diversity predict above-ground biomass and productivity of tropical forests? Testing three alternative hypotheses. J Ecol 103:191–201. https://doi.org/10.1111/1365-2745.12346
Fortunel C, Paine CET, Fine PVA, Kraft NJB, Baraloto C (2014) Environmental factors predict community functional composition in Amazonian forests. J Ecol 102:145–155. https://doi.org/10.1111/1365-2745.12160
Garnier E, Cortez J, Billès G, Navas M-L, Roumet C, Debussche M, Laurent G, Blanchard A, Aubry D, Bellmann A, Neill C, Toussaint J-P (2004) Plant functional markers capture ecosystem properties during secondary succession. Ecology 85:2630–2637. https://doi.org/10.1890/03-0799
Grace JB, Anderson TM, Seabloom EW, Borer ET, Adler PB, Harpole WS, Hautier Y, Hillebrand H, Lind EM, Partel M, Bakker JD, Buckley YM, Crawley MJ, Damschen EI, Davies KF, Fay PA, Firn J, Gruner DS, Hector A, Knops JM, MacDougall AS, Melbourne BA, Morgan JW, Orrock JL, Prober SM, Smith MD (2016) Integrative modelling reveals mechanisms linking productivity and plant species richness. Nature 529:390–393. https://doi.org/10.1038/nature16524
Hillebrand H, Bennett DM, Cadotte MW (2008) Consequences of dominance: a review of evenness effects on local and regional ecosystem processes. Ecology 89:1510–1520. https://doi.org/10.1890/07-1053.1
Husson F, Josse J, Le S, Mazet J, Husson MF (2016) Package ‘FactoMineR.’ R Package 96:698
Jackson JK, Forest R, Survey C (1994) Manual of afforestation in Nepal. Forest Research and Survey Centre, Ministry of Forests and Soil Conservation, Kathmandu
Jucker T, Bouriaud O, Coomes DA, Baltzer J (2015) Crown plasticity enables trees to optimize canopy packing in mixed-species forests. Funct Ecol 29:1078–1086. https://doi.org/10.1111/1365-2435.12428
Jucker T, Bongalov B, Burslem DFRP, Nilus R, Dalponte M, Lewis SL, Phillips OL, Qie L, Coomes DA (2018) Topography shapes the structure, composition and function of tropical forest landscapes. Ecol Lett 21:989–1000. https://doi.org/10.1111/ele.12964
Kattge J, Bönisch G, Díaz S, Lavorel S, Prentice IC, Leadley P, Tautenhahn S, Werner GDA, Aakala T, Abedi M, Acosta ATR, Adamidis GC, Adamson K, Aiba M, Albert CH, Alcántara JM, Alcázar C C, Aleixo I, Ali H, Amiaud B, Ammer C, Amoroso MM, Anand M, Anderson C, Anten N, Antos J, Apgaua DMG, Ashman T-L, Asmara DH, Asner GP, Aspinwall M, Atkin O, Aubin I, Baastrup-Spohr L, Bahalkeh K, Bahn M, Baker T, Baker WJ, Bakker JP, Baldocchi D, Baltzer J, Banerjee A, Baranger A, Barlow J, Barneche DR, Baruch Z, Bastianelli D, Battles J, Bauerle W, Bauters M, Bazzato E, Beckmann M, Beeckman H, Beierkuhnlein C, Bekker R, Belfry G, Belluau M, Beloiu M, Benavides R, Benomar L, Berdugo-Lattke ML, Berenguer E, Bergamin R, Bergmann J, Bergmann Carlucci M, Berner L, Bernhardt-Römermann M, Bigler C, Bjorkman AD, Blackman C, Blanco C, Blonder B, Blumenthal D, Bocanegra-González KT, Boeckx P, Bohlman S, Böhning-Gaese K, Boisvert-Marsh L, Bond W, Bond-Lamberty B, Boom A, Boonman CCF, Bordin K, Boughton EH, Boukili V, Bowman DMJS, Bravo S, Brendel MR, Broadley MR, Brown KA, Bruelheide H, Brumnich F, Bruun HH, Bruy D, Buchanan SW, Bucher SF, Buchmann N, Buitenwerf R, Bunker DE, Bürger J, Burrascano S, Burslem DFRP, Butterfield BJ, Byun C, Marques M, Scalon MC, Caccianiga M, Cadotte M, Cailleret M, Camac J, Camarero JJ, Campany C, Campetella G, Campos JA, Cano-Arboleda L, Canullo R, Carbognani M, Carvalho F, Casanoves F, Castagneyrol B, Catford JA, Cavender-Bares J, Cerabolini BEL, Cervellini M, Chacón-Madrigal E, Chapin K, Chapin FS, Chelli S, Chen S-C, Chen A, Cherubini P, Chianucci F, Choat B, Chung K-S, Chytrý M, Ciccarelli D, Coll L, Collins CG, Conti L, Coomes D, Cornelissen JHC, Cornwell WK, Corona P, Coyea M, Craine J, Craven D, Cromsigt JPGM, Csecserits A, Cufar K, Cuntz M, da Silva AC, Dahlin KM, Dainese M, Dalke I, Dalle Fratte M, Dang-Le AT, Danihelka J, Dannoura M, Dawson S, de Beer AJ, De Frutos A, De Long JR, Dechant B, Delagrange S, Delpierre N, Derroire G, Dias AS, Diaz-Toribio MH, Dimitrakopoulos PG, Dobrowolski M, Doktor D, Dřevojan P, Dong N, Dransfield J, Dressler S, Duarte L, Ducouret E, Dullinger S, Durka W, Duursma R, Dymova O, E-Vojtkó A, Eckstein RL, Ejtehadi H, Elser J, Emilio T, Engemann K, Erfanian MB, Erfmeier A, Esquivel-Muelbert A, Esser G, Estiarte M, Domingues TF, Fagan WF, Fagúndez J, Falster DS, Fan Y, Fang J, Farris E, Fazlioglu F, Feng Y, Fernandez-Mendez F, Ferrara C, Ferreira J, Fidelis A, Finegan B, Firn J, Flowers TJ, Flynn DFB, Fontana V, Forey E, Forgiarini C, François L, Frangipani M, Frank D, Frenette-Dussault C, Freschet GT, Fry EL, Fyllas NM, Mazzochini GG, Gachet S, Gallagher R, Ganade G, Ganga F, García-Palacios P, Gargaglione V, Garnier E, Garrido JL, de Gasper AL, Gea-Izquierdo G, Gibson D, Gillison AN, Giroldo A, Glasenhardt M-C, Gleason S, Gliesch M, Goldberg E, Göldel B, Gonzalez-Akre E, Gonzalez-Andujar JL, González-Melo A, González-Robles A, Graae BJ, Granda E, Graves S, Green WA, Gregor T, Gross N, Guerin GR, Günther A, Gutiérrez AG, Haddock L, Haines A, Hall J, Hambuckers A, Han W, Harrison SP, Hattingh W, Hawes JE, He T, He P, Heberling JM, Helm A, Hempel S, Hentschel J, Hérault B, Hereş A-M, Herz K, Heuertz M, Hickler T, Hietz P, Higuchi P, Hipp AL, Hirons A, Hock M, Hogan JA, Holl K, Honnay O, Hornstein D, Hou E, Hough-Snee N, Hovstad KA, Ichie T, Igić B, Illa E, Isaac M, Ishihara M, Ivanov L, Ivanova L, Iversen CM, Izquierdo J, Jackson RB, Jackson B, Jactel H, Jagodzinski AM, Jandt U, Jansen S, Jenkins T, Jentsch A, Jespersen JRP, Jiang G-F, Johansen JL, Johnson D, Jokela EJ, Joly CA, Jordan GJ, Joseph GS, Junaedi D, Junker RR, Justes E, Kabzems R, Kane J, Kaplan Z, Kattenborn T, Kavelenova L, Kearsley E, Kempel A, Kenzo T, Kerkhoff A, Khalil MI, Kinlock NL, Kissling WD, Kitajima K, Kitzberger T, Kjøller R, Klein T, Kleyer M, Klimešová J, Klipel J, Kloeppel B, Klotz S, Knops JMH, Kohyama T, Koike F, Kollmann J, Komac B, Komatsu K, König C, Kraft NJB, Kramer K, Kreft H, Kühn I, Kumarathunge D, Kuppler J, Kurokawa H, Kurosawa Y, Kuyah S, Laclau J-P, Lafleur B, Lallai E, Lamb E, Lamprecht A, Larkin DJ, Laughlin D, Le Bagousse-Pinguet Y, le Maire G, le Roux PC, le Roux E, Lee T, Lens F, Lewis SL, Lhotsky B, Li Y, Li X, Lichstein JW, Liebergesell M, Lim JY, Lin Y-S, Linares JC, Liu C, Liu D, Liu U, Livingstone S, Llusià J, Lohbeck M, López-García Á, Lopez-Gonzalez G, Lososová Z, Louault F, Lukács BA, Lukeš P, Luo Y, Lussu M, Ma S, Maciel Rabelo Pereira C, Mack M, Maire V, Mäkelä A, Mäkinen H, Malhado ACM, Mallik A, Manning P, Manzoni S, Marchetti Z, Marchino L, Marcilio-Silva V, Marcon E, Marignani M, Markesteijn L, Martin A, Martínez-Garza C, Martínez-Vilalta J, Mašková T, Mason K, Mason N, Massad TJ, Masse J, Mayrose I, McCarthy J, McCormack ML, McCulloh K, McFadden IR, McGill BJ, McPartland MY, Medeiros JS, Medlyn B, Meerts P, Mehrabi Z, Meir P, Melo FPL, Mencuccini M, Meredieu C, Messier J, Mészáros I, Metsaranta J, Michaletz ST, Michelaki C, Migalina S, Milla R, Miller JED, Minden V, Ming R, Mokany K, Moles AT, Molnár V A, Molofsky J, Molz M, Montgomery RA, Monty A, Moravcová L, Moreno-Martínez A, Moretti M, Mori AS, Mori S, Morris D, Morrison J, Mucina L, Mueller S, Muir CD, Müller SC, Munoz F, Myers-Smith IH, Myster RW, Nagano M, Naidu S, Narayanan A, Natesan B, Negoita L, Nelson AS, Neuschulz EL, Ni J, Niedrist G, Nieto J, Niinemets Ü, Nolan R, Nottebrock H, Nouvellon Y, Novakovskiy A, Network TN, Nystuen KO, O’Grady A, O’Hara K, O’Reilly-Nugent A, Oakley S, Oberhuber W, Ohtsuka T, Oliveira R, Öllerer K, Olson ME, Onipchenko V, Onoda Y, Onstein RE, Ordonez JC, Osada N, Ostonen I, Ottaviani G, Otto S, Overbeck GE, Ozinga WA, Pahl AT, Paine CET, Pakeman RJ, Papageorgiou AC, Parfionova E, Pärtel M, Patacca M, Paula S, Paule J, Pauli H, Pausas JG, Peco B, Penuelas J, Perea A, Peri PL, Petisco-Souza AC, Petraglia A, Petritan AM, Phillips OL, Pierce S, Pillar VD, Pisek J, Pomogaybin A, Poorter H, Portsmuth A, Poschlod P, Potvin C, Pounds D, Powell AS, Power SA, Prinzing A, Puglielli G, Pyšek P, Raevel V, Rammig A, Ransijn J, Ray CA, Reich PB, Reichstein M, Reid DEB, Réjou-Méchain M, de Dios VR, Ribeiro S, Richardson S, Riibak K, Rillig MC, Riviera F, Robert EMR, Roberts S, Robroek B, Roddy A, Rodrigues AV, Rogers A, Rollinson E, Rolo V, Römermann C, Ronzhina D, Roscher C, Rosell JA, Rosenfield MF, Rossi C, Roy DB, Royer-Tardif S, Rüger N, Ruiz-Peinado R, Rumpf SB, Rusch GM, Ryo M, Sack L, Saldaña A, Salgado-Negret B, Salguero-Gomez R, Santa-Regina I, Santacruz-García AC, Santos J, Sardans J, Schamp B, Scherer-Lorenzen M, Schleuning M, Schmid B, Schmidt M, Schmitt S, Schneider JV, Schowanek SD, Schrader J, Schrodt F, Schuldt B, Schurr F, Selaya Garvizu G, Semchenko M, Seymour C, Sfair JC, Sharpe JM, Sheppard CS, Sheremetiev S, Shiodera S, Shipley B, Shovon TA, Siebenkäs A, Sierra C, Silva V, Silva M, Sitzia T, Sjöman H, Slot M, Smith NG, Sodhi D, Soltis P, Soltis D, Somers B, Sonnier G, Sørensen MV, Sosinski Jr EE, Soudzilovskaia NA, Souza AF, Spasojevic M, Sperandii MG, Stan AB, Stegen J, Steinbauer K, Stephan JG, Sterck F, Stojanovic DB, Strydom T, Suarez ML, Svenning J-C, Svitková I, Svitok M, Svoboda M, Swaine E, Swenson N, Tabarelli M, Takagi K, Tappeiner U, Tarifa R, Tauugourdeau S, Tavsanoglu C, te Beest M, Tedersoo L, Thiffault N, Thom D, Thomas E, Thompson K, Thornton PE, Thuiller W, Tichý L, Tissue D, Tjoelker MG, Tng DYP, Tobias J, Török P, Tarin T, Torres-Ruiz JM, Tóthmérész B, Treurnicht M, Trivellone V, Trolliet F, Trotsiuk V, Tsakalos JL, Tsiripidis I, Tysklind N, Umehara T, Usoltsev V, Vadeboncoeur M, Vaezi J, Valladares F, Vamosi J, van Bodegom PM, van Breugel M, Van Cleemput E, van de Weg M, van der Merwe S, van der Plas F, van der Sande MT, van Kleunen M, Van Meerbeek K, Vanderwel M, Vanselow KA, Vårhammar A, Varone L, Vasquez Valderrama MY, Vassilev K, Vellend M, Veneklaas EJ, Verbeeck H, Verheyen K, Vibrans A, Vieira I, Villacís J, Violle C, Vivek P, Wagner K, Waldram M, Waldron A, Walker AP, Waller M, Walther G, Wang H, Wang F, Wang W, Watkins H, Watkins J, Weber U, Weedon JT, Wei L, Weigelt P, Weiher E, Wells AW, Wellstein C, Wenk E, Westoby M, Westwood A, White PJ, Whitten M, Williams M, Winkler DE, Winter K, Womack C, Wright IJ, Wright SJ, Wright J, Pinho BX, Ximenes F, Yamada T, Yamaji K, Yanai R, Yankov N, Yguel B, Zanini KJ, Zanne AE, Zelený D, Zhao Y-P, Zheng J, Zheng J, Ziemińska K, Zirbel CR, Zizka G, Zo-Bi IC, Zotz G, Wirth C (2020) TRY plant trait database-enhanced coverage and open access. Glob Change Biol 26:119–188. https://doi.org/10.1111/gcb.14904
Kunwar S, Wang L-Q, Chaudhary R, Joshi PR, Ali A (2021a) Evolutionary diversity and species richness predict aboveground biomass better than tree size variation in local-scale tropical forest types of Nepal. For Ecol Manag 490:119146. https://doi.org/10.1016/j.foreco.2021.119146
Kunwar S, Wang L-Q, Chaudhary R, Joshi PR, Ali A (2021b) Stand density of co-existing species regulates above-ground biomass along a local-scale elevational gradient in tropical forests. Appl Veg Sci 24:e12577. https://doi.org/10.1111/avsc.12577
Loreau M, Naeem S, Inchausti P, Bengtsson J, Grime J, Hector A, Hooper D, Huston M, Raffaelli D, Schmid B, Tilman D, Wardle D (2001) Biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294:804–808. https://doi.org/10.1126/science.1064088
Mason NWH, Mouillot D, Lee WG, Wilson JB (2005) Functional richness, functional evenness and functional divergence: the primary components of functional diversity. Oikos 111:112–118. https://doi.org/10.1111/j.0030-1299.2005.13886.x
Moeslund JE, Arge L, Bøcher PK, Dalgaard T, Svenning JC (2013) Topography as a driver of local terrestrial vascular plant diversity patterns. Nord J Bot 31:129–144
Paquette A, Joly S, Messier C (2015) Explaining forest productivity using tree functional traits and phylogenetic information: two sides of the same coin over evolutionary scale? Ecol Evol 5:1774–1783. https://doi.org/10.1002/ece3.1456
Poorter L, Bongers F (2006) Leaf traits are good predictors of plant performance across 53 rain forest species. Ecology 87:1733–1743. https://doi.org/10.1890/0012-9658(2006)87[1733:LTAGPO]2.0.CO;2
Poorter L, van der Sande MT, Arets EJMM, Ascarrunz N, Enquist B, Finegan B, Licona JC, Martínez-Ramos M, Mazzei L, Meave JA, Muñoz R, Nytch CJ, de Oliveira AA, Pérez-García EA, Prado-Junior J, Rodríguez-Velázques J, Ruschel AR, Salgado-Negret B, Schiavini I, Swenson NG, Tenorio EA, Thompson J, Toledo M, Uriarte M, Pvd H, Zimmerman JK, Peña-Claros M (2017) Biodiversity and climate determine the functioning of Neotropical forests. Glob Ecol Biogeogr 26:1423–1434. https://doi.org/10.1111/geb.12668
Prado-Junior JA, Schiavini I, Vale VS, Arantes CS, van der Sande MT, Lohbeck M, Poorter L (2016) Conservative species drive biomass productivity in tropical dry forests. J Ecol 104:817–827. https://doi.org/10.1111/1365-2745.12543
Pyles MV, Prado-Junior JA, Magnago LFS, de Paula A, Meira-Neto JAA (2018) Loss of biodiversity and shifts in aboveground biomass drivers in tropical rainforests with different disturbance histories. Biodivers Conserv 27:3215–3231. https://doi.org/10.1007/s10531-018-1598-7
R Development Core Team (2019) R version 3.6.0. R Foundation for Statistical Computing, Vienna
Reich BP (2014) The world-wide ‘fast-slow’plant economics spectrum: a traits manifesto. J Ecol 102:275–301. https://doi.org/10.1111/1365-2745.12211
Reyes G (1992) Wood densities of tropical tree species. US Department of Agriculture, Forest Service, Southern Forest Experiment Station, New Orleans, USA
Rodrigues AC, Villa PM, Ali A, Ferreira-Júnior W, Neri AV (2020) Fine-scale habitat differentiation shapes the composition, structure and aboveground biomass but not species richness of a tropical Atlantic forest. J for Res 31:1599–1611. https://doi.org/10.1007/s11676-019-00994-x
Rosenfield MF, Muller SC (2020) Plant traits rather than species richness explain ecological processes in subtropical forests. Ecosystems 23:52–66. https://doi.org/10.1007/s10021-019-00386-6
Rosseel Y (2012) lavaan: An R package for structural equation modeling. J Stat Softw 48:1–36
Rüger N, Wirth C, Wright SJ, Condit R (2012) Functional traits explain light and size response of growth rates in tropical tree species. Ecology 93:2626–2636
Ruiz-Jaen MC, Potvin C (2011) Can we predict carbon stocks in tropical ecosystems from tree diversity? Comparing species and functional diversity in a plantation and a natural forest. New Phytol 189:978–987. https://doi.org/10.1111/j.1469-8137.2010.03501.x
Schöb C, Butterfield BJ, Pugnaire FI (2012) Foundation species influence trait-based community assembly. New Phytol 196:824–834
Stagnol D, Bacouillard L, Davoult D (2016) Relationships between taxonomic and functional diversity: insights into assembly processes. PeerJ PrePrints. https://doi.org/10.7287/peerj.preprints.1646v1
Stephenson NL, Das AJ, Condit R, Russo SE, Baker PJ, Beckman NG, Coomes DA, Lines ER, Morris WK, Ruger N, Alvarez E, Blundo C, Bunyavejchewin S, Chuyong G, Davies SJ, Duque A, Ewango CN, Flores O, Franklin JF, Grau HR, Hao Z, Harmon ME, Hubbell SP, Kenfack D, Lin Y, Makana JR, Malizia A, Malizia LR, Pabst RJ, Pongpattananurak N, Su SH, Sun IF, Tan S, Thomas D, van Mantgem PJ, Wang X, Wiser SK, Zavala MA (2014) Rate of tree carbon accumulation increases continuously with tree size. Nature 507:90–93. https://doi.org/10.1038/nature12914
Toledo M, Peña-Claros M, Bongers F, Alarcón A, Balcázar J, Chuviña J, Leaño C, Licona JC, Poorter L (2012) Distribution patterns of tropical woody species in response to climatic and edaphic gradients. J Ecol 100:253–263. https://doi.org/10.1111/j.1365-2745.2011.01890.x
van der Plas F (2019) Biodiversity and ecosystem functioning in naturally assembled communities. Biol Rev 94:1220–1245. https://doi.org/10.1111/brv.12499
Villa PM, Ali A, Martins SV, de Oliveira Neto SN, Rodrigues AC, Teshome M, Carvalho FA, Heringer G, Gastauer M (2020) Stand structural attributes and functional trait composition overrule the effects of functional divergence on aboveground biomass during Amazon forest succession. For Ecol Manag 477:118481
Villéger S, Mason NW, Mouillot D (2008) New multidimensional functional diversity indices for a multifaceted framework in functional ecology. Ecology 89:2290–2301
Violle C, Navas M-L, Vile D, Kazakou E, Fortunel C, Hummel I, Garnier E (2007) Let the concept of trait be functional! Oikos 116:882–892. https://doi.org/10.1111/j.0030-1299.2007.15559.x
Wang L-Q, Ali A (2021a) Climate regulates the functional traits—aboveground biomass relationships at a community-level in forests: a global meta-analysis. Sci Total Environ 761:143238. https://doi.org/10.1016/j.scitotenv.2020.143238
Wang L-Q, Ali A (2021b) Functional identity regulates aboveground biomass better than trait diversity along abiotic conditions in global forest metacommunities. Ecography. https://doi.org/10.1111/ecog.05854
Xu J, Dang H, Wang M, Chai Y, Guo Y, Chen Y, Zhang C, Yue M (2019a) Is phylogeny more useful than functional traits for assessing diversity patterns under community assembly processes? Forests 10:1159
Xu W, Luo W, Zhang C, Zhao X, von Gadow K, Zhang Z (2019b) Biodiversity-ecosystem functioning relationships of overstorey versus understorey trees in an old-growth temperate forest. Ann for Sci 76:64. https://doi.org/10.1007/s13595-019-0845-8
Yachi S, Loreau M (2007) Does complementary resource use enhance ecosystem functioning? A model of light competition in plant communities. Ecol Lett 10:54–62. https://doi.org/10.1111/j.1461-0248.2006.00994.x
Yuan Z, Ali A, Ruiz-Benito P, Jucker T, Mori AS, Wang S, Zhang X, Li H, Hao Z, Wang X, Loreau M (2020) Above- and below-ground biodiversity jointly regulate temperate forest multifunctionality along a local-scale environmental gradient. J Ecol 108:2012–2024. https://doi.org/10.1111/1365-2745.13378
Zanne A, Lopez-Gonzalez G, Coomes D, Ilic J, Jansen S, Lewis S, Miller R, Swenson N, Wiemann M, Chave J (2009) Data from: towards a worldwide wood economics spectrum. Dryad Digit Repos
Zuur A, Ieno EN, Walker N, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgements
We thank the staff of Division Forest Offices, Dadeldhura and Pahalmanpur, of Sudurpashchim Province, Nepal for their help during fieldwork.
Author information
Authors and Affiliations
Contributions
SK, RC, and PRJ collected data. AA and SK designed the study and idea for analysis. SK, L-QW, and MKL analyzed data through support from AA. SK, MKL and AA wrote the paper whereas all coauthors contributed to the draft.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding
This study was supported by the Jiangsu Science and Technology Special Project (Grant No. BX2019084), Research Startup Funding at Nanjing Forestry University (Grant No. 163010230), and the Faculty Startup Funding (to Arshad Ali) for establishing Forest Ecology Research Group at Hebei University (Special Project No. 521100221033).
The online version is available at http://www.springerlink.com.
Corresponding editor: Yu Lei.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kunwar, S., Wang, LQ., Kazempour Larsary, M. et al. Functional composition of tall-statured trees underpins aboveground biomass in tropical forests. J. For. Res. 34, 333–344 (2023). https://doi.org/10.1007/s11676-022-01460-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-022-01460-x