Abstract
Extreme climate has increasingly led to negative impacts on forest ecosystems globally, especially in semiarid areas where forest ecosystems are more vulnerable. However, it is poorly understood how tree growth is affected by different drought events. In 2006–2009, the larch plantations in the semiarid areas of Northwest China were negatively affected by four consecutive dry years, which was a very rare phenomenon that may occur frequently under future climate warming. In this study, we analyzed the effect of these consecutive dry years on tree growth based on the data of the tree rings in the dominant layer of the forest canopy on a larch plantation. We found that the tree-ring width index (RWI) in dry years was lower than that in normal years, and it experienced a rapidly decreasing trend from 2006 to 2009 (slope = − 0.139 year−1, r = − 0.94) due to water supply deficits in those dry years. Drought induced legacy effects of tree growth reduction, and consecutive dry years corresponded with greater growth reductions and legacy effects. Growth reductions and legacy effects were significantly stronger in the third and fourth consecutive dry years than that of single dry year (p < 0.05), which might have been due to the cumulative stress caused by consecutive dry years. Our results showed that larch trees experienced greater tree growth reduction due to consecutive dry years and their legacy effect, and the trees had lower recovery rates after consecutive dry years. Our results highlight that consecutive dry years pose a new threat to plantations under climate warming, and thus, the effect of climate extremes on tree growth should be considered in growth models in semiarid areas.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change involved increases in temperature and changes in precipitation patterns, which affected the frequency and intensity of droughts (IPCC 2021). These changes had a great negative impact on the growth of trees in the years they occur, and these impacts included stem shrinkage (Butz et al. 2017; Salomón et al. 2022) and tree-ring increment reduction (D’Orangeville et al. 2018; Schnabel et al. 2022). Studies had shown that legacy effects were widespread in forest ecosystems following severe drought events (Anderegg et al. 2013b, 2015b; Kannenberg et al. 2019a, b), and even widespread tree and forest mortality occurred (Allen et al. 2010; Anderegg et al. 2015b; Choat et al. 2018), which might lead to changes in forest dynamics and ecosystem functioning (Anderegg et al. 2013a; Zhou et al. 2013). Therefore, a better understanding of the effects of drought on tree growth was urgently needed to establish reliable models of forest dynamics in semiarid areas under future climate warming.
Tree rings were archives of past growing conditions and can reflect past climatic and moisture conditions (Schnabel et al. 2022). Several studies had analyzed the relationship between tree growth and climate by correlating ring width with seasonal and monthly climate factors (Wang et al. 2019a, 2020a, 2021; Yan et al. 2020; Yu and Liu 2020), and they had found that tree growth was sensitive to climate change (Peng et al. 2022; Wang et al. 2019a, 2020a, 2021; Zhang et al. 2019). Moreover, an increasing number of studies had analyzed how drought affects tree growth through analyses of tree rings (Kannenberg et al. 2019a, b; Schnabel et al. 2022). The effects of drought on tree growth could be quantified using growth reduction and legacy effects (Bohner and Diez 2021; Kannenberg et al. 2019a, b; Schnabel et al. 2022). The studies had found that legacy effects resulted in a 10% decrease in ring width increments following extreme drought (Kannenberg et al. 2019b), that the timing of drought affected legacy effects and tree recovery at the global scale (Bohner and Diez 2021; Huang et al. 2018) and that trees could not recover immediately and completely after severe drought (Anderegg et al. 2015b; Wang et al. 2021). However, it was poorly understood how different drought events, such as single and consecutive dry years, affected tree growth on plantations in semiarid areas.
Larch (Larix principis-rupprechtii) plantations, i.e., one of the main afforestation tree species used in North and Northwest China, had been established since the 1980s in the Liupan Mountains, an important headwater area of the Loess Plateau in Northwest China (Tian et al. 2021, 2023). These plantations were particularly vulnerable to drought, and thus, they were ideal ecosystems to study the effect of drought on tree growth. In these regions, the intra-annual stem radial growth of larch trees was largely governed by temperature and water availability (Liu et al. 2017; Ma et al. 2020). However, we did not yet have a clear understanding of the inter-annual trend of tree growth and how different drought events affected cumulative growth on semiarid larch plantations.
Our study hypothesized that (1) tree growth was sensitive to drought and (2) consecutive dry years would cause greater growth reduction and legacy effects relative to those of single dry year. To test the above hypothesis, we used tree-ring sampling to investigate tree growth in a semiarid larch plantation in Northwest China. The objectives of our study were to (1) quantify inter-annual trends in tree growth, (2) detect the main climatic factors that limit tree growth, and (3) quantify drought effects and legacy effects in single and consecutive dry years. Our results would contribute to improving the understanding of the impact of climate extremes on plantations and to predicting the dynamics of plantations in semiarid areas under future climate warming.
Materials and methods
Study area
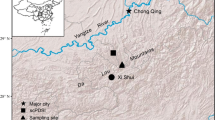
The study area was located in the northern part of the Liupan Mountains, in the middle of the Loess Plateau in Northwest China (35°50′ − 36°20′ N, 105°48′ − 106°24′ E).The climate of the study area was semiarid continental monsoon. Based on the meteorological data of Guyuan station (36°02′03″ N, 106°14′07″ E, 1835.5 m above sea level (a.s.l.)), from 1989 to 2019, the mean annual air temperature (Ta) was 7.4 °C, and it continued to rise at a rate of 0.64 °C per decade (Fig. 1). The mean annual precipitation (P) was 455.4 mm, varying between 310 and 710 mm with a nonsignificant decreasing trend (p > 0.05). Approximately 85% of rainfall occurred between April and September. In our study area, the standardized precipitation evapotranspiration index (SPEI) at 6- and 12-month scales, obtained by precipitation minus potential evapotranspiration (Schnabel et al. 2022; Vicente-Serrano et al. 2010), did not significantly increase from 1989 to 2019 (p > 0.05, Fig. 1). In addition, based on the meteorological data of the nearby study plot from 2000 to 2018 (35°58′25″N, 106°8′55″E, 2020 m a.s.l.), the mean annual Ta was 6.8 °C, and the mean annual P was 596.0 mm, varying between 434.0 and 808.1 mm.
Variations in annual precipitation (P, (a)), mean annual air temperature (Ta, (b)), SPEI at a 12-month scale from January-December (SPEI12, (c)) and SPEI at a 6-month scale from April-September (SPEI6, (d)) from 1989 to 2019. The horizontal line in (a) was the mean annual precipitation from 1989 to 2019. The horizontal lines in (c) and (d) were the SPEI values of − 1 below which it was viewed as dry years
Afforestation had been encouraged since the 1980s, montane forests were mainly distributed on shady and semi-shady slopes at elevations of 1400–2700 m a.s.l. The dominant forest in the study area was the Larix principis-rupprechtii plantation (Li et al. 2016; Wang et al. 2020b). The soil type in the study area was mainly gray forest soil (Wang et al. 2020b). The soil thickness varied from 40 to 300 cm.
Study plot
A pure larch plantation plot (35°58′10″ N, 106°8′54″ E) with an area of 30 m × 30 m was established at the bottom of the shady slope in the Diediegou watershed in the northern part of the Liupan Mountains. The slope gradient of this plot was 11 °, and the elevation was 2055 m a.s.l. Based on field measurements in July 2020, this plot had a tree age of 35 years, a tree density of 1367 stem·ha−1, an mean diameter at breast height (DBH) of 12.4 ± 5.3 cm, an mean tree height of 11.2 ± 3.6 m, and an mean canopy diameter of 2.9 ± 1.1 m. The understory vegetation was mainly Carex spp. and Fragaria vesca. The coverage and biomass of the herbaceous layer were 76% and 2.5 t ha− 1, respectively.
Tree-ring sampling and laboratory treatment
A total of 24 trees in the dominant layer of the forest canopy (i.e., health and mean shape) were selected as sample trees to examine climate sensitivity. Two incremental cores of each sample tree were collected using a 5.15 mm increment borer. Tree cores were acquired at a distance of 1.3 m aboveground. The collected cores were then air-dried, mounted on slotted wooden strips, and polished from coarse to fine sandpaper in the laboratory.
The tree-ring width (TRW) of each tree core was measured with a LINTAB™ 6 tree-ring measurement station with an accuracy of 0.001 mm. All series of TRW were quality checked and interactively cross-dated using COFECHA software (Holmes 1983). Because the larch trees in this plot were younger, we detrended each growth series to tree-ring width indices (RWIs) with a negative exponential function (Mood et al. 2021; Schnabel et al. 2022) using the R package ‘dplR’ (Bunn 2008; Bunn et al. 2020) to eliminate the effects of age trends.
Climate data
The daily climate data were taken from a nearby weather station (Guyuan Station, 36°02′03″ N, 106°14′07″ E, 1835.5 m a.s.l.) approximately 10 km from the plot. To evaluate the availability of the climate data, we compared the precipitation and air temperature from the Guyuan weather station with 9 years of these data (from 2010 to 2018) from the Diediegou weather station in our study area (35°58′10″ N, 106°8′54″ E, 2050 m a.s.l.). The correlation between the two weather stations was significant for mean annual air temperature (r = 0.74, p < 0.05) and annual precipitation (r = 0.84, p < 0.05), as shown via Eqs. 1 and 2.
where Ta1 and Ta2 are the mean annual air temperature at the Diediegou and Guyuan weather stations, respectively. P1 and P2 are the mean annual precipitation at the Diediegou and Guyuan Weather Stations, respectively.
All climate variables from 1989 to 2019 were summarized at seasonal level (spring: March–May, summer: June–August, autumn: September–November, and winter: December–February) and other periods, such as the previous October to the current September and the current May to August.
Data analysis
Drought year identification
Drought identification was central to analyzing the effects of drought on tree growth. The SPEI was a commonly used drought index that was based on standardized monthly water balance of precipitation minus potential evapotranspiration (Hari et al. 2020; Schnabel et al. 2022; Schwarz et al. 2020; Skiadaresis et al. 2019). The SPEI quantified drought severity and could be calculated on different time scales to represent the climatic water balance (Vicente-Serrano et al. 2010). Here, we used the SPEI at 6- and 12-month scales, i.e., two SPEI scales for the main growing season (6-month scale, April-September) and full year (12-month scale, January-December). The SPEI was calculated by using the SPEI package in R (Schnabel et al. 2022; Vicente-Serrano et al. 2010) from monthly precipitation (mm) and potential evapotranspiration (mm) data derived from the Guyuan Weather Station near the plot.
According to the SPEI at 6- and 12-month scales, we divided the years into dry years (with SPEI values below − 1), wet years (values greater than 1), and normal years (values between − 1 and 1) (Mckee et al. 1993). Therefore, we selected 2000 and 2006 as single dry years and 2006–2009 as consecutive dry years (Fig. 1c and d).
Tree growth-climate relationship analysis
The growth trends of larch trees were simulated using simple linear models. The growth trend was the slope of the regression line of the mean measured and detrended tree-ring width index. A positive slope indicated an increase in tree growth, while a negative slope indicated a decrease in growth (Wang et al. 2020a).
Pearson’s correlations between the mean detrended tree-ring width index (RWI) and climate factors (using seasonal and monthly data that included mean, minimum and maximum daily temperature (Ta, Ta-min and Ta-max), precipitation (P) and SPEI) were calculated for 1989–2019 using the R package treeclim (Zang and Biondi 2015).
Growth reduction and legacy effect quantification
Growth reduction and legacy effects could be used to analyze the effects of drought on tree growth (Bohner and Diez 2021; Kannenberg et al. 2019a, b). For each tree-ring series, we estimated growth reduction in every dry year as the difference between the mean RWI in normal years (\({\overline{RWI} }_{non-drought}\)) and the RWI in every dry year (RWIdrought) using Eq. 3 (Bohner and Diez 2021). We estimated the mean \({\overline{RWI} }_{non-drought}\), which did not include a year after the drought, and thus, the legacy year was not considered for the \({\overline{RWI} }_{non-drought}\) estimate (Griffin-Nolan et al. 2018; Kannenberg et al. 2019a). Growth reduction in dry years was normalized to zero, with positive values indicating lower growth and negative values indicating higher growth than normal years.
For each tree-ring series, we qualified legacy effects based on the climatic water balance in the year after the drought event (Anderegg, et al. 2015b; Kannenberg et al. 2019a). Legacy effects were calculated as the difference between the predicted and observed RWI using Eq. 4 (Anderegg, et al. 2015b; Kannenberg et al. 2019a). We predicted the RWI by creating a linear model between the observed RWI and SPEI at the 12-month scale in normal years (not including a year after the drought). Considering the high rainfall in autumn in the study area, which had a great impact on the growth, we used the SPEI at a 12-month scale to capture the climatic water balance throughout the year (January-December) to compare the legacy effects of the dry years (Schnabel et al. 2022). In this study, the predicted RWI was obtained by a linear model between the RWI and SPEI at a 12-month scale in 1989–2019, with a mean correlation coefficient of 0.39. A zero legacy effect corresponded to the expected growth based on climatic conditions, while positive values indicated legacy effects in the year following the drought.
Results
Tree growth trends
The mean tree-ring width (TRW) of the larch trees decreased slowly from 1989 to 2005 (slope = − 0.032 mm year−1, r = − 0.29) and from 2010 to 2019 (slope = − 0.050 mm year− 1, r = − 0.49), while those in the 2006–2009 consecutive dry years showed a rapid decline (slope = − 0.468 mm year−1, r = − 0.95) (Fig. 2a).
The mean detrended tree-ring width index (RWI) increased from 1989 to 2005 (slope = 0.027 year−1, r = 0.66) and from 2010 to 2019 (slope = 0.039 year−1, r = 0.55), while those in the 2006 − 2009 consecutive dry years showed a rapid decline (slope = − 0.139 year−1, r = − 0.94) (Fig. 2b). The RWI in 2000 and 2007 − 2009 dry years was lower than that in normal years (mean value of 1.014).
Relationship between tree growth and climatic factors
The mean detrended tree-ring width index (RWI) showed a high sensitivity to the SPEI (Fig. 3), which was especially obvious from the previous autumn to the current summer. The RWI was significantly positively correlated with the SPEI from the previous August to the current June (p < 0.05) and precipitation (P) in the previous August (p < 0.05). Moreover, high spring temperatures and summer precipitation positively affected growth. The RWI showed a positive correlation with the mean, minimum and maximum daily temperatures (Ta, Ta-min and Ta-max, respectively) from March to April and precipitation from April to September.
Pearson’s correlation coefficients between the mean detrended tree-ring width index (RWI) and climatic factors (mean, minimum and maximum daily temperatures (Ta, Ta-min and Ta-max, respectively), total precipitation (P) and SPEI under different seasonal and monthly conditions. Asterisks indicate a significant correlation (* indicates p < 0.05). p and c represent the climatic factors of the previous and current years, respectively
Drought effect on tree growth
Tree growth showed pronounced responses to drought stress, with larger growth reductions during the four consecutive dry years (Fig. 4a and b). The growth reduction gradually increased in the consecutive dry years of 2006–2009 (Fig. 4b). In 2008–2009, i.e., the third and fourth consecutive dry years, the growth reduction was significantly stronger than that in a single dry year (p < 0.05). The mean growth reduction ranged close to zero in the single dry years of 2000 and 2006 (Fig. 4a), while the drought effect in 2000 and 2006 differed significantly, i.e., growth in 2000 tended to decrease, while growth in 2006 tended to increase.
Growth reduction (a, b) and legacy effect (c, d) in the single dry year of 2000 and 2006 and the consecutive dry years of 2006–2009. The significant differences in growth reduction and legacy effect in the consecutive dry years of 2006–2009 compared to those in single dry year are indicated by asterisks (*** and * indicate p < 0.001 and p < 0.05, respectively)
Large growth reduction in dry years were often accompanied by large legacy effects. There was a legacy effect after single and consecutive dry years (Fig. 4c and d). The legacy effect in the consecutive dry years of 2006–2009 increased significantly. Moreover, the legacy effect in the third and fourth consecutive dry years was significantly higher than that in a single dry year (p < 0.05).
Discussion
Drought effect on cumulative tree growth
The tree growth of the larch trees showed a downward trend from 2006 to 2009. This downward trend was not the result of tree aging, as the larch trees in this plot were planted in 1986 as a young-aged plantation, and they remained in the rapid growth phase at this stage (Xu et al. 2023). Moreover, no thinning measures were carried out in this plot (Wang et al. 2020b). Related studies had shown that tree growth for healthy trees should exhibit a continued upward trend for decades after reaching a stable phase (Choi et al. 2001; Piovesan et al. 2010; Wang et al. 2019a). Therefore, the declining tree ring-width index (RWI) trend in 2006–2009 should not have been due to tree age and other disturbance measures, and it was mainly due to climate drought. Related studies had found that tree growth was sensitive to drought in arid and semiarid areas (Liang et al. 2016; Wang et al. 2019a) and that annual growth and the rate of radial growth were determined by water availability and temperature, which were especially obvious during periods of drought (Liu et al. 2019; Ma et al. 2020; Wan et al. 2020, 2022).
A downward trend in tree growth and a low growth rate during drought had been found in many species in semiarid areas, such as Qinghai spruce (Wang et al. 2019a, 2021; Wan et al. 2020, 2022), Korean pine (Wang et al. 2019b), and Norway spruce (Sidor et al. 2015). Our findings also found that the RWI of larch trees showed a downward trend and low cumulative growth from 2006 to 2009 due to climate drought. The climate drought during this period was caused by high temperatures and low precipitation, which led to increased soil moisture deficits. After 2010, however, the RWI increased. This scenario occurred because the precipitation and SPEI trend obviously increased, resulting in a relatively wetter climate that alleviated drought stress, and thus, warm temperatures and high-water availability could prolong the growth days and increase annual growth.
In this study, the RWI showed a high sensitivity to the SPEI, which was especially obvious from the previous autumn to the current summer (Fig. 3), verifying the first hypothesis that tree growth is highly sensitive to drought. This result showed that the SPEI played a vital role in tree growth. The SPEI was an indicator of drought intensity that considered the effects of precipitation and potential evapotranspiration (Vicente-Serrano et al. 2010). Similarly, many researchers had also found that tree growth was affected by the SPEI in arid and semiarid areas (Wang et al. 2019a, 2020a, 2021; Zhang et al. 2019). In addition, high spring temperatures and summer precipitation positively affected growth, further confirming that drought stress had a great effect on the growth of larch trees in semiarid areas.
Drought effect and legacy effect for different extreme dry years
Our results supported the second hypothesis, stems experienced greater growth reductions in the consecutive dry years of 2006–2009 compared to those in the single dry year. The dry years of 2006–2009 were meteorologically similar (Fig. 1), but the growth reduction amount was greater than that in the previous dry year (Fig. 4). This finding indicated that the cumulative drought effects imposed by four consecutive dry years might have been the main driver of increased stress in 2007, 2008 and 2009. Similarly, the growth reduction for oak, maple and ash in Central Europe was significantly stronger in the second dry year than in a single dry year (Schnabel et al. 2022). Overall, our findings suggested that larch trees experienced consistent growth reduction under consecutive dry years, thus affecting forest dynamics and productivity.
Legacy effects in dry forest ecosystems were widespread after drought events (Anderegg et al. 2013b, 2015b; Kannenberg et al. 2019a, b; Schuldt et al. 2020). In this study, we also found legacy effects for larch trees in single and consecutive dry years, with more obvious legacy effects after three or four consecutive dry years, when trees had no time to recover. Similarly, in mesic forest ecosystems, significant legacies in radial growth lasting 1–3 years were found after severe drought (Kannenberg et al. 2019a), and the legacy effects varied with the degree and timing of drought across biomes (Anderegg et al. 2015b; Schwalm et al. 2017); in floodplain forest ecosystems, the extreme drought of 2018 led to the legacy effects of tree growth. These results suggested that the timing and intensity of drought events were the main factors influencing the magnitude of the legacy effects and that consecutive hotter droughts posed a great threat to forest ecosystems in arid and semiarid areas.
During consecutive dry years, growth damage and cavitation continued to increase (Anderegg et al. 2013a, b), and carbon allocation shifted to the canopy and root system (Kannenberg et al. 2019a, b), which resulted in a decrease in tree-ring increments and thereby amplified legacy effects caused by drought events. Moreover, the loss of leaf area and/or stored nonstructural carbohydrates during drought could impair xylem repair and growth, and thus a tree’s defense system weakened in subsequent years, which increased the harm caused by pests and pathogens to a forest (Anderegg et al. 2013a, b; Boyes et al. 2019; Hartmann et al. 2013; Schuldt et al. 2020). In the long term, consecutive drought inhibited tree growth and could lead to tree death, as observed in dry forest ecosystems (Allen et al. 2010; Anderegg et al. 2015a, b; Bohner and Diez 2021).
Larch plantation dynamics under future climate change
Climate warming-induced drought stress will have important consequences for larch plantations in semiarid areas in the future. Drier conditions not only lead to earlier cessation of stem growth (Liu et al. 2019; Wan et al. 2022) but also result in a reduction in cumulative stem growth (Ma et al. 2020; Salomón et al. 2022). In addition, soil water has fallen rapidly in recent decades, reaching a historic low level in semiarid areas, indicating an unprecedented trend toward soil aridification (Zhang et al. 2020). If climate and soil drought conditions increase in semiarid areas, then the drought effect and legacy effect increase, thus reducing forest functioning and tree vitality over time by inhibiting the accumulation of wood biomass. If more frequent and severe legacy effects cumulatively affect forest functioning, then future climate changes may stimulate tree adaptation, transfer ranges, or death in our study area. However, these impacts are still speculative, and further multi-scale studies are needed to determine how drought conditions affect the dynamics of stem growth and wood formation in the study area. Our results will help produce more accurate predictions of forest dynamics under climate warming in semiarid areas by improving vegetation dynamic models, especially extreme drought events.
Conclusion
Our study compared the stress responses of cumulative stem growth in consecutive dry years of 2006–2009 with the responses in a single dry year. Drought effects and legacy effects in the third and fourth consecutive dry years were obviously higher than those in a single dry year, indicating that consecutive drought posed a new stress. Moreover, drought stress was alleviated after 2010, and the tree-ring width index increased slowly, indicating that larch trees started to recover after drought. Therefore, larch trees are highly tolerant to drought and recover to some extent after drought, which is of great significance to plantations in semiarid areas. Consecutive drought events are expected to become more frequent with future climate changes, and thus, our findings will provide important insights for predicting the effects of climatic extremes on forest ecosystems in semiarid areas.
References
Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg EHT (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For Ecol Manage 259(4):660–684. https://doi.org/10.1016/J.FORECO.2009.09.001
Anderegg WRL, Kane JM, Anderegg LD (2013a) Consequences of widespread tree mortality triggered by drought and temperature stress. Nat Clim Chang 3(1):30–36. https://doi.org/10.1038/nclimate1635
Anderegg WRL, Plavcová L, Anderegg LD, Hacke UG, Berry JA, Field CB (2013b) Drought’s legacy: multiyear hydraulic deterioration underlies widespread aspen forest die-off and portends increased future risk. Glob Change Biol 19(4):1188–1196. https://doi.org/10.1111/gcb.12100
Anderegg WRL, Hicke JA, Fisher RA, Allen CD, Aukema J, Bentz B, Hood S, Lichstein JW, Macalady AK, McDowell N (2015a) Tree mortality from drought, insects, and their interactions in a changing climate. New Phytol 208(3):674–683. https://doi.org/10.1111/nph.13477
Anderegg WRL, Schwalm C, Biondi F, Camarero JJ, Koch G, Litvak M, Ogle K, Shaw JD, Shevliakova E, Williams AP (2015b) Pervasive drought legacies in forest ecosystems and their implications for carbon cycle models. Science 349:528–532. https://doi.org/10.1126/science.aab1833
Bohner T, Diez J (2021) Tree resistance and recovery from drought mediated by multiple abiotic and biotic processes across a large geographic gradient. Sci Total Environ 789:147744. https://doi.org/10.1016/j.scitotenv.2021.147744
Boyes KN, Hietala-Henschell KG, Barton AP, Storer AJ, Marshall JM (2019) Linking tree growth rate, damage repair, and susceptibility to a genus-specific pest infestation. J for Res 30(5):1935–1941. https://doi.org/10.1007/s11676-019-00896-y
Bunn AG (2008) A dendrochronology program library in R (dplR). Dendrochronologia 26(2):115–124. https://doi.org/10.1016/j.dendro.2008.01.002
Bunn AG, Korpela M, Biondi F, Campelo F, Mérian P, Qeadan F, Zang C (2020) dplR: Dendrochronology program library in R. Accessed from https://CRAN.R-project.org/package=dplR
Butz P, Raffelsbauer V, Graefe S, Peters T, Cueva E, Hölscher D, Bräuning A (2017) Tree responses to moisture fluctuations in a neotropical dry forest as potential climate change indicators. Ecol Ind 83:559–571. https://doi.org/10.1016/j.ecolind.2016.11.021
Choat B, Brodribb TJ, Brodersen CR, Duursma RA, López R, Medlyn BE (2018) Triggers of tree mortality under drought. Nature 558(7711):531–539. https://doi.org/10.1038/s41586-018-0240-x
Choi J, Lorimer CG, Vanderwerker J, Cole WG, Martin GL (2001) A crown model for simulating long-term stand and gap dynamics in northern hardwood forests. For Ecol Manage 152(1–3):235–258. https://doi.org/10.1016/S0378-1127(00)00606-X
D’Orangeville L, Maxwell J, Kneeshaw D, Pederson N, Duchesne L, Logan T, Houle D, Arseneault D, Beier CM, Bishop DA et al (2018) Drought timing and local climate determine the sensitivity of eastern temperate forests to drought. Glob Change Biol 24(6):2339–2351. https://doi.org/10.1111/gcb.14096
Griffin-Nolan RJ, Carroll CJW, Denton EM, Johnston MK, Collins SL, Smith MD, Knapp AK (2018) Legacy effects of a regional drought on aboveground net primary production in six central US grasslands. Plant Ecol 219(5):505–515. https://doi.org/10.1007/s11258-018-0813-7
Hari V, Rakovec O, Markonis Y, Hanel M, Kumar R (2020) Increased future occurrences of the exceptional 2018–2019 central European drought under global warming. Sci Rep 10(1):12207. https://doi.org/10.1038/s41598-020-68872-9
Hartmann H, Ziegler W, Trumbore S, Knapp A (2013) Lethal drought leads to reduction in nonstructural carbohydrates in Norway spruce tree roots but not in the canopy. Funct Ecol 27(2):413–427. https://doi.org/10.1111/1365-2435.12046
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull 43:69–75
Huang M, Wang X, Keenan TF, Piao S (2018) Drought timing influences the legacy of tree growth recovery. Glob Change Biol 24(8):3546–3559. https://doi.org/10.1111/gcb.14294
IPCC (2021) Climate change 2021: The physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA
Kannenberg SA, Maxwell JT, Pederson N, D’Orangeville L, Ficklin DL, Phillips RP (2019a) Drought legacies are dependent on water table depth, wood anatomy and drought timing across the eastern US. Ecol Lett 22(1):119–127. https://doi.org/10.1111/ele.13173
Kannenberg SA, Novick KA, Alexander MR, Maxwell JT, Moore DJP, Phillips RP, Anderegg WRL (2019b) Linking drought legacy effects across scales: from leaves to tree rings to ecosystems. Glob Change Biol 25(9):2978–2992. https://doi.org/10.1111/gcb.14710
Li ZH, Yu PT, Wang YH, Webb AA, He C, Wang YB, Yang LL (2016) A model coupling the effects of soil moisture and potential evaporation on the tree transpiration of a semi-arid larch plantation. Ecohydrology 10(1):1–11. https://doi.org/10.1002/eco.1764
Liang E, Leuschner C, Dulamsuren C, Wagner B, Hauck M (2016) Global warming-related tree growth decline and mortality on the north-eastern Tibetan plateau. Clim Change 134:163–176. https://doi.org/10.1007/s10584-015-1531-y
Liu ZB, Wang YH, Tian A, Yu PT, Xiong W, Xu LH, Wang YR (2017) Intra-annual variation of stem radius of Larix principis-rupprechtii and its response to environmental factors in liupan mountains of northwest China. Forests 8(10):382. https://doi.org/10.3390/f8100382
Liu XS, Wang CS, Zhao JK (2019) Seasonal drought effects on intra-annual stem growth of Taiwan Pine along an elevational gradient in subtropical China. Forests 10(12):1128. https://doi.org/10.3390/f10121128
Ma J, Guo JB, Wang YH, Liu ZB, Gao D, Hong L, Zhang ZY (2020) Variations in stem radii of Larix principis-rupprechtii to environmental factors at two slope locations in the Liupan Mountains, northwest China. J for Res 32(2):513–527. https://doi.org/10.1007/s11676-020-01114-w
Mckee TB, Doesken NJ, Kleist J (1993) The relationship of drought frequency and duration to time scales. In: proceedings of the 8th Conference on applied climatology 17(22): 179−183
Mood BJ, Bonsal B, Howat B, Laroque CP (2021) Multi-year white spruce drought legacies in southern Saskatchewan. For Ecol Manage 491:119114. https://doi.org/10.1016/j.foreco.2021.119144
Peng R, Liu H, Anenkhonov OA, Sandanov DV, Korolyuk AY, Shi L, Xu C, Dai J, Wang L (2022) Tree growth is connected with distribution and warming-induced degradation of permafrost in southern Siberia. Glob Change Biol 28(17):5243–5253. https://doi.org/10.1111/gcb.16284
Piovesan G, Biondi F, Filippo AD, Alessandrini A, Maugeri M (2010) Drought-driven growth reduction in old beech (Fagus sylvatica L.) forests of the central apennines. ItalyGlob Change Biol 14(6):1265–1281. https://doi.org/10.1111/j.1365-2486.2008.01570.x
Salomón RL, Peters RL, Zweifel R, Sass-Klaassen UGW, Stegehuis AI, Smiljanic M, Poyatos R, Babst F, Cienciala E, Fonti P et al (2022) The 2018 European heatwave led to stem dehydration but not to consistent growth reductions in forests. Nat Commun 13(1):28. https://doi.org/10.1038/s41467-021-27579-9
Schnabel F, Purrucker S, Schmitt L, Engelmann RA, Kahl A, Richter R, Seele-Dilbat C, Skiadaresis G, Wirth C (2022) Cumulative growth and stress responses to the 2018–2019 drought in a European floodplain forest. Glob Change Biol 28(5):1870–1883. https://doi.org/10.1111/gcb.16028
Schuldt B, Buras A, Arend M, Vitasse Y, Beierkuhnlein C, Damm A, Gharun M, Grams TEE, Hauck M, Hajek P et al (2020) A first assessment of the impact of the extreme 2018 summer drought on central European forests. Basic Appl Ecol 45:86–103. https://doi.org/10.1016/j.baae.2020.04.003
Schwalm CR, Anderegg WR, Michalak AM, Fisher JB, Biondi F, Koch G, Litvak M, Ogle K, Shaw JD, Wolf A (2017) Global patterns of drought recovery. Nature 548(7666):202–205. https://doi.org/10.1038/nature23021
Schwarz J, Skiadaresis G, Kohler M, Kunz J, Bauhus J (2020) Quantifying growth responses of trees to drought-a critique of commonly used resilience indices and recommendations for future studies. Curr for Rep 6:185–200. https://doi.org/10.1007/s40725-020-00119-2
Sidor CG, Popa I, Vlad R, Cherubini P (2015) Different tree-ring responses of Norway spruce to air temperature across an altitudinal gradient in the Eastern Carpathians (Romania). Trees 29(4):985–997. https://doi.org/10.1007/s00468-015-1178-3
Skiadaresis G, Schwarz JA, Bauhus J (2019) Groundwater extraction in floodplain forests reduces radial growth and increases summer drought sensitivity of pedunculate Oak trees (Quercus robur L.). Front for Glob Change 2:5. https://doi.org/10.3389/ffgc.2019.00005
Tian A, Wang YH, Webb AA, Liu ZB, Ma J, Yu PT, Wang X (2021) Water yield variation with elevation, tree age and density of larch plantation in the Liupan mountains of the Loess Plateau and its forest management implications. Sci Total Environ 752:141752. https://doi.org/10.1016/j.scitotenv.2020.141752
Tian A, Wang YH, Webb AA, Yu PT, Wang X, Liu ZB (2023) Modelling the response of larch growth to age, density, and elevation and the implications for multifunctional management in northwest China. J for Res 34:1423–1436. https://doi.org/10.1007/s11676-022-01539-5
Vicente-Serrano SM, Beguería S, López-Moreno JI (2010) A multiscalar drought index sensitive to global warming: the standardized precipitation evapotranspiration index. J Clim 23(7):1696–1718. https://doi.org/10.1175/2009JCLI2909.1
Wan YF, Yu PT, Li XQ, Wang YH, Wang B, Yu YP, Zhang L, Liu XD, Wang SL (2020) Seasonal pattern of stem diameter growth of Qinghai spruce in the Qilian mountains. Northwest China for 11(5):494. https://doi.org/10.3390/f11050494
Wan YF, Yu PT, Li XQ, Wang YH, Wang B, Yu YP, Zhang L, Liu XD, Wang SL (2022) Divergent seasonal patterns of Qinghai spruce growth with elevation in Northwestern China. Forests 13(3):388. https://doi.org/10.3390/f13030388
Wang B, Yu PT, Zhang L, Wang YH, Yu YP, Wang SL (2019a) Differential trends of Qinghai spruce growth with elevation in Northwestern China during the recent warming hiatus. Forests 10(9):712. https://doi.org/10.3390/f10090712
Wang X, Pederson N, Chen Z, Lawton K, Zhu C, Han S (2019b) Recent rising temperatures drive younger and southern Korean pine growth decline. Sci Total Environ 649:1105–1116. https://doi.org/10.1016/j.scitotenv.2018.08.393
Wang B, Yu PT, Yu YP, Wan YF, Wang YH, Zhang L, Wang SL, Wang X, Liu ZB, Xu LH (2020a) Effects of canopy position on climate-growth relationships of Qinghai spruce in the central Qilian mountains. Northwest China Dendrochronol 64(8):125756. https://doi.org/10.1016/j.dendro.2020.125756
Wang YB, Wang YH, Li ZH, Yu PT, Han XS (2020b) Interannual variation of transpiration and its modeling of a larch plantation in semiarid Northwest China. Forests 11(12):1303. https://doi.org/10.3390/f11121303
Wang B, Yu PT, Yu YP, Wang YH, Zhang L, Wan YF, Wang SL, Liu XD (2021) Trees at a moderately arid site were more sensitive to long-term drought. Forests 12(5):579. https://doi.org/10.3390/f12050579
Xu XQ, Yu PT, Wang YH, Xu LH, Yu YP, Liu ZB (2023) The variation of stand structure with age and its hydrological effects of larch plantation in Liupan Mountains. For Res 36(1):109–116 ((in Chinese with English abstract))
Yan BQ, Yu J, Liu QJ, Wang LH, Hu LL (2020) Tree-ring response of Larix chinensis on regional climate and sea-surface temperature variations in alpine timberline in the Qinling Mountains. J for Res 31(1):204–213. https://doi.org/10.1007/s11676-018-0841-0
Yu J, Liu Q (2020) Larix olgensis growth-climate response between lower and upper elevation limits: an intensive study along the eastern slope of the Changbai mountains, Northeastern China. J for Res 31:231–244. https://doi.org/10.1007/s11676-018-0788-1
Zang C, Biondi F (2015) treeclim: an R package for the numerical calibration of proxy-climate relationships. Ecography 38(4):431–436. https://doi.org/10.1111/ecog.01335
Zhang L, Shi H, Yu PT, Wang YH, Pan SF, Wang B, Tian HQ (2019) Divergent growth responses to warming between stand-grown and open-grown trees in a dryland montane forest in Northwestern China. Forests 10(12):1133. https://doi.org/10.3390/f10121133
Zhang P, Jeong JH, Yoon JH, Kim H, Wang SYS, Linderholm HW, Fang K, Wu X, Chen D (2020) Abrupt shift to hotter and drier climate over inner East Asia beyond the tipping point. Science 370(6520):1095–1099. https://doi.org/10.1126/science.abb336
Zhou G, Peng C, Li Y, Liu S, Zhang Q, Tang X, Liu J, Yan J, Zhang D, Chu G (2013) A climate change-induced threat to the ecological resilience of a subtropical monsoon evergreen broad-leaved forest in Southern China. Glob Change Biol 19(4):1197–1210. https://doi.org/10.1111/gcb.12128
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: This study was funded by the National Natural Science Foundation of China (Nos. 42161144008; U21A2005; U20A2085) and the National Key Research and Development Program of China (2022YFF0801804; 2022YFF0801803; 2022YFF1300404).
The online version is available at https://springerlink.fh-diploma.de/.
Corresponding editor: Yu Lei.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wan, Y., Yu, P., Wang, Y. et al. More tree growth reduction due to consecutive drought and its legacy effect for a semiarid larch plantation in Northwest China. J. For. Res. 35, 39 (2024). https://doi.org/10.1007/s11676-023-01691-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11676-023-01691-6