Abstract
Background
Reverse shoulder arthroplasty (RSA) is a valuable solution for patients with shoulder pain or injury primarily due to a rotator cuff tear or secondary to traumatic events. Nevertheless, several complications are known to appear, with the most frequent being scapular notching (SN) on the inferior and posterior scapular neck. Controversial data exist about the clinical relevance of SN. Since further consequences are still not clearly understood, we aimed to provide more clarity on which factors, especially external rotation (ER), contribute to the appearance and progress of notching.
Methods
Constant Score (CS), Subjective Shoulder Value (SSV), flexion, abduction, and ER were evaluated retrospectively in 153 shoulders of 147 patients (mean age 79±7.7 years; 62% women) who underwent RSA between 2005 and 2010. Anteroposterior radiographs were evaluated before and 1, 2, 3, and 5 years after RSA for SN according to the Sirveaux classification. The evaluation was performed by two independent surgeons. Spearman’s coefficient and t-test were used.
Results
CS, SSV, flexion, and abduction increased significantly 1 year after RSA compared to before (all p < 0.0001). No improvement was shown for ER between the same timepoints. Between 2 and 5 years of follow-up, only flexion decreased by 5°(p = 0.02) while CS, SSV, abduction, and ER remained constant. After RSA, notching increases over time. There was no association between SN and CS, SSV, flexion, abduction or ER at any of the measured timepoints. Higher flexion correlated with higher abduction after RSA at every follow-up (1 year r = 0.88, 2 years r = 0.89, 3 years r = 0.86, 5 years r = 0.86). The interrater correlation test showed a strong correlation (r = 0.7).
Conclusion
We verified the functional benefits of RSA for patients. Additionally, our findings show that despite radiographic progression of notching and unchanged limited ER, the postoperative improvements in CS, SSV, flexion, and abduction are preserved over 5 years.
Zusammenfassung
Hintergrund und Fragestellung
Die inverse Schulterendoprothetik (RSA) hat sich zu einer wertvollen Lösung entwickelt für Patienten mit Schmerzen sowie Verletzungen, die auf einer Rotatorenmanschettenruptur oder sekundär traumatischen Ereignissen basieren. Zu den häufigsten Komplikationen zählt das Skapula-Notching (SN) am inferoposterioren Skapulahals. Über die damit zusammenhängende klinische Relevanz gibt es kontroverse Aussagen. Da die Konsequenz noch nicht abschließend geklärt ist, möchten die Autoren mehr Klarheit schaffen, welche klinischen Faktoren, insbesondere die Außenrotation (AR), zum Auftreten und Fortschreiten des SN beitragen.
Methode
Constant Score (CS), subjektiver Schulterwert (SSV), Flexion, Abduktion und AR wurden retrospektiv bei 153 Schultern von 147 Patienten (Durchschnittsalter 79 Jahre ±7,7; 62 % Frauen) ausgewertet, die sich zwischen 2005 und 2010 einer RSA unterzogen. Anteroposteriore Röntgenaufnahmen wurden vor und 1, 2, 3 und 5 Jahre nach der RSA gemäß der Sirveaux-Klassifikation ausgewertet. Die Auswertung wurde von 2 unabhängigen orthopädischen Chirurgen durchgeführt. Außerdem wurden der Spearman-Koeffizient und der t‑Test für die Vergleiche zwischen den Zeitpunkten verwendet.
Ergebnisse
CS, SSV, Flexion und Abduktion nahmen ein Jahr nach RSA im Vergleich zu vorher signifikant zu (alle p < 0,0001). Bei der AR wurde zwischen denselben Zeitpunkten keine Verbesserung festgestellt. Zwischen 2 und 5 Jahren nahm die Flexion um 5°ab (p = 0,02), während CS, SSV, Abduktion und AR konstant blieben. Nach RSA zeigt das SN einen zunehmenden Trend im Verlauf. Kein Zusammenhang konnte zwischen SN und CS, SSV, Flexion, Abduktion oder AR zu sämtlichen Zeitpunkten gefunden werden. Bessere Flexion korreliert mit besserer Abduktion nach RSA in jedem Follow-up (1 Jahr: r = 0,88; 2 Jahre: r = 0,89; 3 Jahre: r = 0,86; 5 Jahre: r = 0,86). Die Interrater-Reliabilität zeigte eine starke Korrelation (r = 0,7).
Schlussfolgerung
Die Autoren konnten den funktionellen Nutzen der RSA für die Patienten nachweisen. Darüber hinaus zeigen die vorliegenden Ergebnisse, dass trotz des fortschreitenden radiologischen Trends des SN und unverändert eingeschränkter AR postoperativ Verbesserungen in CS, SSV, Flexion und Abduktion über 5 Jahre erhalten bleiben.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With the Grammont type of reverse prosthesis that was introduced in the late 1980s, a new biomechanical concept was established by lowering and medializing the center of rotation [6, 17]. Therefore, a greater lever arm results in the deltoid muscle leading to an improved shoulder function, especially its efficacy for abduction and to a lesser extent also forward elevation [10, 24]. Further biomechanical advantages of the Grammont type reverse shoulder arthroplasty (RSA) are a larger ball that offers more stability and a greater range of motion (ROM) and the neckless placement of the center of rotation on the glenoid surface, thereby reducing the shear forces at the point of fixation [6]. Shoulder function and autonomy of daily activities are improved after RSA [7]. Some of these adaptations, compared to the previous constrained implant models (TSA), are able to partially compensate the deficiency in the superior rotator cuff muscles. Nevertheless, several authors have reported that clinically internal and external rotation (ER) do not necessarily improve after RSA despite preservation of the infraspinatus and subscapularis muscle tendon unit [6, 9]. Explanations for this are that the ROM is restrained as a consequence of the limited lateral offset of the prosthesis and that the lever arm of the remaining rotator cuff is reduced by medializing and lowering the center of rotation. Additionally, the potential damage of the suprascapular nerve with the posterior screw during surgery may affect active external rotation following RSA [6].
A specific radiographic sign associated with RSA implantation is called “scapular notching.” Scapular notching describes a glenoid erosion that usually appears on the inferior scapular pillar and was classified by Sirveaux–Nérot et al. [33, 38] into four different stages:
-
Grade 0 shows no defect
-
Grade 1 defect corresponds to an erosion within the inferior scapular pillar
-
Grade 2 defect erosion reaches the inferior border of the lower screw
-
Grade 3 defect extends further than the lower screw
-
Grade 4 defects are considered when the notching progresses to contact with the bottom of the baseplate
Several factors seem to be relevant in predicting inferior notching, the most common of which are: the anatomy of the scapular neck, the base plate orientation, the surgical approach, the patient’s rotator cuff condition before and after surgery and degenerative disorders. One of the most important risk factors for this impingement of the humeral polyethylene cup seems to be the position of the base plate [18, 30, 37]. Lower positioning shows less notching incidence [42] while the absence of the neck from the glenosphere [6] in combination with superior tilt should be avoided causing more likely an underlap of the humeral cup [25, 29]. An additional risk factor is a large scapular neck angle which was shown by Simovitch ([37]: 9° in shoulders without and 31° for shoulders with inferior notching). The same author reported that a large peg-glenoid rim distance also influences inferior notching negatively (20.1 mm without and 24.7 mm with notching). An anterosuperior surgical approach showed higher notching incidence compared to a deltopectoral approach [28].
The goal of the present study was to analyze the parameters of ROM, especially ER, before and after RSA along with the incidence and progression of scapular notching. We hypothesize that a poor shoulder function, especially a decreased ER, is more likely to correlate with a higher degree of notching.
Patients and methods
Inclusion and exclusion criteria
For this retrospective study, patients with implantation of an Anatomical Shoulder Inverse System (Zimmer Inc., Warsaw, IN, USA) and ages between 18 and 99 years were included between 2005 and 2010 from our institutional database. The time period was selected on the basis of the implant that was mostly used at that time. All patients who had surgery before the RSA (except soft tissue intervention such as subacromial debridement or repair of rotator cuff tear), primary or secondary tendon transfer (e.g., latissimus dorsi transfer), incomplete clinical or radiological data, and rejection of informed consent were excluded.
Surgical procedure
All surgeries were performed or supervised by two experienced shoulder surgeons. The deltopectoral approach was mainly used, in rare cases the superolateral approach was chosen. An Anatomical Shoulder Inverse (Zimmer Biomet, Warsaw, IN, USA) was implanted in all patients with a neck angle of 155°. Glenospheres with 36 and 42 mm were used. The surgical procedure is described in detail elsewhere [16]. Repair of the subscapularis muscle and tendon was performed routinely unless anatomical circumstances (e.g., muscle atrophy) did not allow it.
Postoperative care
Postoperative rehabilitation started on the day after surgery with certain limitations as follows: active assisted elevation in the case of physical complaints, no internal rotation against resistance, ER maximally up to 0°, and activation of the deltoid muscle for 6 weeks. After 6 weeks, patients were allowed to increase ROM gradually according to their symptoms. Physiotherapy was prescribed for a duration of 6 weeks, followed by strengthening. An arm sling was given facultatively.
Clinical and radiological assessment
The Constant Score (CS), Subjective Shoulder Value (SSV), flexion, abduction, and ER were clinically assessed before surgery and at every follow-up 1, 2, 3, and 5 years postoperatively after RSA and analyzed retrospectively.
Radiographs in anteroposterior and axillary view were taken according to a standardized internal protocol before surgery and at every follow-up. The Sirveaux–Nérot classification was used to grade SN (grade 0: signs of notching absent—grade 4: notching up to the inferior screw and glenoid peg; [33, 38]) using the anteroposterior and axillary view. Preoperatively, on the anteroposterior radiograph we measured the acromiohumeral distance ([AHD], in millimeters; [21]) and assessed Hamada stage (stage 1, AHD > 6 mm—stage 5, AHD < 7 mm with osteonecrosis of humeral head; [19]). Retrospectively, anteversion of the glenoid scapular angle was analyzed from CT scans according to Friedmann (measured in degrees; [14]) in addition to the glenoid type classification after Walch (grade A1/A2: centered humeral head, concentric wear, no subluxation of the humeral head with minor/major erosion—grade C: > 25° retroversion, dysplastic or biconcave glenoid, posterior translation of the humeral head; [41]). Radiologic readout was performed by two independent orthopedic surgeons over a period of 3 months. The PACS-Software Merlin (Phönix-PACS GmbH, Freiburg in Breisgau, Germany) was used to visualize radiographs.
Statistical analysis
Pearson’s correlation coefficient was applied to test for correlation between the radiological readouts, and Spearman’s correlation coefficient was used to determine correlations between parameters and notching grading. Spearman’s correlation coefficients range from −1.0 to 1.0, indicating perfect inverse and direct correlation, respectively, while values from 0.70 to 0.89 were interpreted as strong and 0.90–1.00 as very strong correlation [35]. The two-tailed Wilcoxon signed rank test was used for the comparisons between the follow-up time points. To rule out measurement error, minimal detectable change (MDC) was assessed based on the 95% confidence interval [40]. The level of significance was set at p < 0.05. Analysis was conducted in R (R Core Team, 2017, R Foundation for Statistical Computing, Vienna, Austria).
Results
In total, 153 shoulders of 147 patients were included in the study (Table 1). There were 38% men (58 shoulders) and 62% women (95 shoulders) with an average age of 79 years (SD ± 7.7). Overall, 110 patients were diagnosed with cuff tear arthropathy, four with osteoarthritis and 39 with cuff deficiency or posttraumatic injury. Three patients had surgery due to an emergency, all others were planned in advance for RSA. The surgical approach chosen was deltopectoral in 90.3 and superolateral in 7.3% of cases. The remaining 2.4% approaches were anterosuperior and made using the already existing scar. A total of 70.3% of patients received a glenosphere of 36 mm in diameter, while 29.1% received one of 42 mm (for one case there were no data). The RSA was on the dominant side in 114 cases, on the contralateral side in 38, and for one case there was no information about dominance.
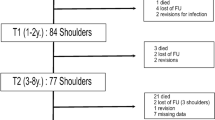
For the 1‑year follow-up we registered 130 patients, for the 2‑year follow-up 116 patients showed up, for the 3‑year follow-up 103 patients, and for the 5‑year follow-up there were 85 patients. Reasons for absence were surgical revisions (e.g., fracture), emigration, bad general condition, missed consultation (reason not documented), or death (Table 2). Interrater correlation for the radiological reading of SN showed a strong correlation with r = 0.70 (p < 0.001).
Preoperatively, AHD was measured and calculated with a mean of 5.4 mm (SD ± 3.6). According to the Hamada classification there were 50 shoulders with grade 1, 26 with grade 2, 21 with grade 3, 29 with grade 4a/b, and 27 with grade 5. In total, 53% of the scapulae showed a retroversion according to Friedman [14] with a mean of 5.4° (SD ± 5.0) and 47% an anteversion with a mean of −6.7° (SD ± 6.0). Our cohort mostly presented with Walch A1 and A2 configuration (93 and 46 shoulders respectively), whereas only nine shoulders corresponded to B1, two to B2, and three to C. A significant difference was found in the posttraumatic group compared with the others, with a lower mean Hamada grade (2.6 and 3.4, respectively; p = 0.026) and a higher AHD (6.9 and 4.8, respectively; p < 0.001).
The CS, SSV, flexion and abduction increased significantly 1 year after RSA compared to before (all p < 0.0001; Tables 3 and 4, Fig. 1). No improvement was shown for ER after 1 year (p = 0.79) compared to the preoperative values. Between the 2- and 5‑year follow-up, only flexion decreased by 3.5° (p = 0.02; MDC of 7° was not reached; Table 4) while CS, SSV, abduction, and ER remained constant. The posttraumatic group had a higher CS before surgery compared to the nontraumatic group (32.4 and 28.8; p = 0.006, data not shown). After surgery, the former tended to have better ER in every follow-up than patients with arthropathy (mean +5.3°) with the most distinct difference of +8.0° (p = 0.006) 2y after RSA. Patients suffering from trauma showed no difference in SN compared to patients with no trauma.
a-c Means for measurement of clinical parameters as flexion, abduction, and external rotation preoperatively (pre) and for all follow-ups (lines indicate range of standard deviation). d,e Means for measurement of the Constant Score (CS) and percentage of Subjective Shoulder Values (SSV) preoperatively (pre) and for all follow-ups (lines indicate range of standard deviation). * p < 0.05
After RSA, notching showed an increasing tendency with higher grades over time in the mean, without statistical significance (mean Sirveaux 1 year = 2.20, mean 2 years = 2.45, mean 3 years = 2.70, mean 5 years = 2.74, Table 5 and Fig. 2). While absent notching (grade 0) showed a clear decreasing tendency from 33 to 11 cases during the follow-ups over 5 years, the incidence of the most severe notching grade 4 increased from one to 10 cases (Table 5). Despite this radiographic tendency of notching toward higher grades, no correlation was found at any follow-up time point with any of the clinical parameters of flexion, abduction, or ER (Fig. 3).
Overview of distribution for the severity of radiological Sirveaux gradings from Sirveaux grade 0 (absent) to grade 4 (most severe, notching up to inferior screw and glenoid peg) over all follow-ups from 1 year to 5 years after implantation of reverse shoulder arthroplasty. Vertical scale depicts frequency number
Spearman correlation coefficients for scapular notching depicted on the vertical scale for correlation with the clinical parameters Constant Score (CS), Subjective Shoulder Values (SSV), flexion, abduction, and external rotation (ER) over all follow-ups from 1 year to 5 years after implantation of reverse shoulder arthroplasty. Correlation from 0 to ±0.3 was considered as “weak”
Higher flexion correlated strongly with higher abduction after RSA in every follow-up (1 year r = 0.88, 2 years r = 0.89, 3 years r = 0.86, 5 years r = 0.86). Equivalently, CS showed strong a correlation with abduction and flexion over 5 years (all r > 0.72, data not shown).
Discussion
In this retrospective study of 147 patients 72% suffered from a cuff tear arthropathy along with pain and impaired function resulting in therapeutic RSA. We showed that 1 year after RSA the parameters SSV, CS, flexion, and abduction all increased significantly and remained constantly high over all follow-ups, whereas ER did not improve nor decrease. Improved CS and SSV was summarized in the review by Petrillo et al. with very similar pre- and postoperative values and a total increase for CS (∆ + 31 points) and SSV (∆ + 54%; [32]).
Our results confirm findings in the literature with the congruent reported average postoperative flexion of 124° after RSA from studies [26]. The slight decrease of −3.5° that we measured at 5 years compared to 2 years after surgery was interpreted as measuring error since an MDC of 7° was not reached [40]. Improvement in flexion from before surgery ranged, depending on the study, from 43 to 106° [26] encompassing our increase in motion after 5 years of 57°. The reason for this wider variation can be explained by the different follow-up time points and number of patients as well as by interrater reliability in measurement methods [12]. In active flexion the glenohumeral and scapulothoracic joint are involved which makes it difficult to separate a potential compensatory mechanism by the latter when measuring the flexion during physical examination [26], suggesting the lower values are more strictly related to the improved glenohumeral function. The results reported by Maier et al. are also in agreement with our findings, without a change in ER after RSA. However, other groups have found significant improvement in ER. Compared to our results, Petrillo et al. showed an improvement in ER after RSA of up to 27.7° in their review (408 shoulders in total) with the underlying diagnosis of either cuff tear arthropathy or irreparable massive rotator cuff tears. This might be explained by the much lower preoperative mean of 17.1° (compared to our pre- and postoperative mean of 25.6 and 27.9°, respectively; [32]). This highlights the fact that our study cohort already had a relatively acceptable extent in ER before surgery. Knowing the established method of a latissimus dorsi transfer to improve ER after RSA due to an atrophied teres minor muscle [2, 5, 15, 37], Berglund et al. compared a group with transfer against one without transfer finding no change in ER after surgery [1]. Another group examined 608 patients before and after RSA dividing patients into three groups according to their preoperative range of ER (stiff with a mean of −4°, weak with a mean of 16°, and normal with a mean of 44°), showing a significantly higher improvement for patients with a stiff range compared to those with a weak range (comparable to our cohort) and normal range after surgery [8]. This finding raises the issue that patients with a stiff range might suffer from an additional restriction (postoperative stiffness) that causes only temporary impairment.
When comparing traumatic versus nontraumatic cases, it is not surprising that the latter have a higher Hamada score, lower AHD and lower SSV since we know that these patients already suffer from chronic osteoarthritis and cuff tear arthropathy [19, 21]. We believe that patients who have a clear trauma history have better ER postoperatively since they usually have not suffered from chronic degenerative changes of muscles and the joint, presenting with a more robust preoperative anatomical condition with higher potential for compensation.
We found a trend of notching showing an increasing tendency over 5 years after RSA from 2.20 up to 2.74. Overall, 33 patients did not show any radiological sign after 1 year whereas after 5 years only 11 patients had no evidence of notching (with a decreased follow-up cohort due to the drop-outs). The reported incidence of notching varies widely from 4 to 96% [13]. The debate of whether SN is clinically relevant and related to poorer outcome such as impaired ROM or higher incidence of complications [27, 36] or whether it is simply a radiological finding without a harmful effect [6, 25, 39, 42] is still ongoing. In 2019 Simovitch et al. summarized in a retrospective study of 324 patients with a 5-year follow-up that patients without notching have a significantly increased function in abduction, flexion, ER, and the CS compared to those with notching [36]. It should be mentioned here that notching grades 1–4 were pooled for a binary analysis and therefore higher power. Further, the authors discovered that patients with notching had a higher complication rate. Several factors have been discussed to influence the progress, e.g., implant type, surgical technique with inferior tilting [11], or bony increased offset [4], BMI, glenosphere size, or daily activity [11, 22, 30, 36]. We found no correlation at any follow-up time point between notching trend and any of the functional parameters of flexion, abduction, or ER (Fig. 3). Based on biomechanical and clinical studies, several groups stated that primary ER in adduction contributes to the progress of inferior notching [22, 23, 31]. This leads to the conclusion that a lack of sufficient improvement in ER might be protective for SN. However, our preoperative values of ER were higher than in other patient groups before RSA [32].
Overall, 75% of our older patient cohort were affected on their dominant side, revealing how important improvement in function for basic daily activity and therefore life quality can be. For future studies, the essential requirements for everyday living should be taken into account.
In summary, RSA is an established and important therapy option in older patients with cuff tear arthropathy, deficiency, and osteoarthritis suffering from pain and constrained ROM [3, 6, 34]; although after 5 years we did not find any negative correlation between the occurrence of SN and impaired function, care should be taken when the implant and surgical technique are chosen to minimize the risk, since some groups have found an association of an increased complication rate in the long-term period [27, 36].
A limitation of this study is the high drop-out rate during the follow-up, which can be explained by the older population. It was not uncommon to miss follow-ups when they were content. Furthermore, the patients’ physical activity was not evaluated, which is why no statement can be made about a potential impact on the notching process from the postulated risk of ER motion. We did not perform a standardized postoperative quality control for implant positioning on the radiographs to exclude other potential influencing factors.
A strength of this study is the double reviewing process of Sirveaux grading from the radiographs. Further, the follow-up period of 5 years let us draw a firm conclusion about the outcome data regarding the patient demands in this population. Since two senior shoulder surgeons mainly performed the operations with the same implant, variations of different angle techniques in the base plate can be ruled out.
Practical conclusion
-
Reverse shoulder arthroplasty (RSA) is an established therapy option in older patients with cuff tear arthropathy, deficiency, and osteoarthritis suffering from pain and constrained range of motion.
-
Significant functional benefits can be achieved with RSA but several complications can occur, the most frequent being scapular notching of the inferior and posterior scapular neck.
-
Despite radiographic progression of notching over time, however, no negative clinical association of external rotation and other parameters including abduction, flexion, Constant Score, and Subjective Shoulder Value was observed after 5 years of follow-up.
References
Berglund DD, Rosas S, Triplet JJ, Kurowicki J, Horn B, Levy JC (2018) Restoration of external rotation following reverse shoulder arthroplasty without latissimus dorsi transfer. JB JS Open Access 3:e54. https://doi.org/10.2106/JBJS.OA.17.00054
Boileau P, Chuinard C, Roussanne Y, Neyton L, Trojani C (2007) Modified latissimus dorsi and teres major transfer through a single delto-pectoral approach for external rotation deficit of the shoulder: as an isolated procedure or with a reverse arthroplasty. J Shoulder Elbow Surg 16:671–682. https://doi.org/10.1016/j.jse.2007.02.127
Boileau P, Gonzalez JF, Chuinard C, Bicknell R, Walch G (2009) Reverse total shoulder arthroplasty after failed rotator cuff surgery. J Shoulder Elbow Surg 18:600–606. https://doi.org/10.1016/j.jse.2009.03.011
Boileau P, Moineau G, Roussanne Y, O’Shea K (2011) Bony increased-offset reversed shoulder arthroplasty: minimizing scapular impingement while maximizing glenoid fixation. Clin Orthop Relat Res 469:2558–2567. https://doi.org/10.1007/s11999-011-1775-4
Boileau P, Rumian AP, Zumstein MA (2010) Reversed shoulder arthroplasty with modified L’Episcopo for combined loss of active elevation and external rotation. J Shoulder Elbow Surg 19:20–30. https://doi.org/10.1016/j.jse.2009.12.011
Boileau P, Watkinson DJ, Hatzidakis AM, Balg F (2005) Grammont reverse prosthesis: design, rationale, and biomechanics. J Shoulder Elbow Surg 14:147S–161S. https://doi.org/10.1016/j.jse.2004.10.006
Cappellari A, Trovarelli G, Andriolo M, Berizzi A, Ruggieri P (2020) Reverse shoulder arthroplasty for treatment of proximal humerus complex fractures in elderly: a single institution experience. Injury. https://doi.org/10.1016/j.injury.2020.07.056
Carofino B, Routman H, Roche C (2020) The influence of preoperative external rotation weakness or stiffness on reverse total shoulder arthroplasty. JSES Int 4:382–387. https://doi.org/10.1016/j.jseint.2020.02.006
Clark JC, Ritchie J, Song FS, Kissenberth MJ, Tolan SJ, Hart ND, Hawkins RJ (2012) Complication rates, dislocation, pain, and postoperative range of motion after reverse shoulder arthroplasty in patients with and without repair of the subscapularis. J Shoulder Elbow Surg 21:36–41. https://doi.org/10.1016/j.jse.2011.04.009
De Wilde L, Audenaert E, Barbaix E, Audenaert A, Soudan K (2002) Consequences of deltoid muscle elongation on deltoid muscle performance: a computerised study. Clin Biomech (Bristol, Avon) 17:499–505. https://doi.org/10.1016/s0268-0033(02)00065-7
Falaise V, Levigne C, Favard L, SOFEC (2011) Scapular notching in reverse shoulder arthroplasties: the influence of glenometaphyseal angle. Orthop Traumatol Surg Res 97:S131–137. https://doi.org/10.1016/j.otsr.2011.06.007
Feijen S, Tate A, Kuppens K, Struyf T, Claes A, Struyf F (2020) Intrarater and interrater reliability of a passive shoulder flexion range of motion measurement for latissimus dorsi flexibility in young competitive swimmers. J Sport Rehabil 29:855–858. https://doi.org/10.1123/jsr.2019-0294
Friedman RJ, Flurin PH, Wright TW, Zuckerman JD, Roche CP (2017) Comparison of reverse total shoulder arthroplasty outcomes with and without subscapularis repair. J Shoulder Elbow Surg 26:662–668. https://doi.org/10.1016/j.jse.2016.09.027
Friedman RJ, Hawthorne KB, Genez BM (1992) The use of computerized tomography in the measurement of glenoid version. J Bone Joint Surg Am 74:1032–1037
Gerber C, Pennington SD, Lingenfelter EJ, Sukthankar A (2007) Reverse Delta-III total shoulder replacement combined with latissimus dorsi transfer. A preliminary report. J Bone Joint Surg Am 89:940–947. https://doi.org/10.2106/JBJS.F.00955
Gerber C, Pennington SD, Nyffeler RW (2009) Reverse total shoulder arthroplasty. J Am Acad Orthop Surg 17:284–295. https://doi.org/10.5435/00124635-200905000-00003
Grammont PM, Baulot E (1993) Delta shoulder prosthesis for rotator cuff rupture. Orthopedics 16:65–68
Gutiérrez S, Comiskey CA, Luo ZP, Pupello DR, Frankle MA (2008) Range of impingement-free abduction and adduction deficit after reverse shoulder arthroplasty. Hierarchy of surgical and implant-design-related factors. J Bone Joint Surg Am 90:2606–2615. https://doi.org/10.2106/JBJS.H.00012
Hamada K, Fukuda H, Mikasa M, Kobayashi Y (1990) Roentgenographic findings in massive rotator cuff tears. A long-term observation. Clin Orthop Relat Res 254:92–96
Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG (2009) Research electronic data capture (REDCap)—a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42:377–381. https://doi.org/10.1016/j.jbi.2008.08.010
Kolk A, Overbeek CL, de Groot JH, Nelissen RGHH, Nagels J (2020) Reliability and discriminative accuracy of 5 measures for craniocaudal humeral position: an assessment on conventional radiographs. JSES Int 4:189–196. https://doi.org/10.1016/j.jseint.2019.11.005
Kolmodin J, Davidson IU, Jun BJ, Sodhi N, Subhas N, Patterson TE, Li ZM, Iannotti JP, Ricchetti ET (2018) Scapular notching after reverse total shoulder arthroplasty: prediction using patient-specific osseous anatomy, implant location, and shoulder motion. J Bone Joint Surg Am 100:1095–1103. https://doi.org/10.2106/JBJS.17.00242
Lädermann A, Gueorguiev B, Charbonnier C, Stimec BV, Fasel JH, Zderic I, Hagen J, Walch G (2015) Scapular notching on kinematic simulated range of motion after reverse shoulder arthroplasty is not the result of impingement in adduction. Medicine 94:e1615. https://doi.org/10.1097/MD.0000000000001615
Lädermann A, Walch G, Lubbeke A, Drake GN, Melis B, Bacle G, Collin P, Edwards TB, Sirveaux F (2012) Influence of arm lengthening in reverse shoulder arthroplasty. J Shoulder Elbow Surg 21:336–341. https://doi.org/10.1016/j.jse.2011.04.020
Lévigne C, Boileau P, Favard L, Garaud P, Molé D, Sirveaux F, Walch G (2008) Scapular notching in reverse shoulder arthroplasty. J Shoulder Elbow Surg 17:925–935. https://doi.org/10.1016/j.jse.2008.02.010
Maier MW, Caspers M, Zeifang F, Dreher T, Klotz MC, Wolf SI, Kasten P (2014) How does reverse shoulder replacement change the range of motion in activities of daily living in patients with cuff tear arthropathy? A prospective optical 3D motion analysis study. Arch Orthop Trauma Surg 134:1065–1071. https://doi.org/10.1007/s00402-014-2015-7
Mollon B, Mahure SA, Roche CP, Zuckerman JD (2017) Impact of scapular notching on clinical outcomes after reverse total shoulder arthroplasty: an analysis of 476 shoulders. J Shoulder Elbow Surg 26:1253–1261. https://doi.org/10.1016/j.jse.2016.11.043
Nicholson GP, Strauss EJ, Sherman SL (2011) Scapular notching: recognition and strategies to minimize clinical impact. Clin Orthop Relat Res 469:2521–2530. https://doi.org/10.1007/s11999-010-1720-y
Nyffeler RW, Werner CM, Gerber C (2005) Biomechanical relevance of glenoid component positioning in the reverse Delta III total shoulder prosthesis. J Shoulder Elbow Surg 14:524–528. https://doi.org/10.1016/j.jse.2004.09.010
Nyffeler RW, Werner CM, Simmen BR, Gerber C (2004) Analysis of a retrieved delta III total shoulder prosthesis. J Bone Joint Surg Br 86:1187–1191. https://doi.org/10.1302/0301-620x.86b8.15228
Permeswaran VN, Caceres A, Goetz JE, Anderson DD, Hettrich CM (2017) The effect of glenoid component version and humeral polyethylene liner rotation on subluxation and impingement in reverse shoulder arthroplasty. J Shoulder Elbow Surg 26:1718–1725. https://doi.org/10.1016/j.jse.2017.03.027
Petrillo S, Longo UG, Papalia R, Denaro V (2017) Reverse shoulder arthroplasty for massive irreparable rotator cuff tears and cuff tear arthropathy: a systematic review. Musculoskelet Surg 101:105–112. https://doi.org/10.1007/s12306-017-0474-z
Valenti PH, Bouttens D, Nerot C (2001) Delta 3 reversed prothesis for osteoarthritis with massive rotator cuff tear, long term results (>5 years). Shoulder prosthesis…two to 10 years follow-up. Sauramps Medical, Montpellier, pp 253–259
Rittmeister M, Kerschbaumer F (2001) Grammont reverse total shoulder arthroplasty in patients with rheumatoid arthritis and nonreconstructible rotator cuff lesions. J Shoulder Elbow Surg 10:17–22. https://doi.org/10.1067/mse.2001.110515
Schober P, Boer C, Schwarte LA (2018) Correlation coefficients: appropriate use and interpretation. Anesth Analg 126:1763–1768. https://doi.org/10.1213/ANE.0000000000002864
Simovitch R, Flurin PH, Wright TW, Zuckerman JD, Roche C (2019) Impact of scapular notching on reverse total shoulder arthroplasty midterm outcomes: 5‑year minimum follow-up. J Shoulder Elbow Surg 28:2301–2307. https://doi.org/10.1016/j.jse.2019.04.042
Simovitch RW, Zumstein MA, Lohri E, Helmy N, Gerber C (2007) Predictors of scapular notching in patients managed with the Delta III reverse total shoulder replacement. J Bone Joint Surg Am 89:588–600. https://doi.org/10.2106/JBJS.F.00226
Sirveaux F, Favard L, Oudet D, Huquet D, Walch G, Molé D (2004) Grammont inverted total shoulder arthroplasty in the treatment of glenohumeral osteoarthritis with massive rupture of the cuff. Results of a multicentre study of 80 shoulders. J Bone Joint Surg Br 86:388–395. https://doi.org/10.1302/0301-620x.86b3.14024
Stechel A, Fuhrmann U, Irlenbusch L, Rott O, Irlenbusch U (2010) Reversed shoulder arthroplasty in cuff tear arthritis, fracture sequelae, and revision arthroplasty. Acta Orthop 81:367–372. https://doi.org/10.3109/17453674.2010.487242
Stratford PW, Binkley J, Solomon P, Finch E, Gill C, Moreland J (1996) Defining the minimum level of detectable change for the Roland-Morris questionnaire. Phys Ther 76:359–365. https://doi.org/10.1093/ptj/76.4.359 (discussion 366–358)
Walch G, Badet R, Boulahia A, Khoury A (1999) Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty 14:756–760. https://doi.org/10.1016/s0883-5403(99)90232-2
Werner CM, Steinmann PA, Gilbart M, Gerber C (2005) Treatment of painful pseudoparesis due to irreparable rotator cuff dysfunction with the Delta III reverse-ball-and-socket total shoulder prosthesis. J Bone Joint Surg Am 87:1476–1486. https://doi.org/10.2106/JBJS.D.02342
Funding
No external funding was provided. Funded by internal sources.
Funding
Open access funding provided by University of Zurich
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Grob, S. Bouaicha, M. Germann, S. Germann, C. Gerber and K. Wieser declare that they have no competing interests.
For this article no studies with animals were performed by any of the authors. All studies performed were in accordance with the ethical standards indicated in each case. Study was approved by the Ethics Committee from Canton of Zurich (KEK-ZH-Nr. 2015-0147).
Additional information
Availability of data and material
Study data were retrospectively analyzed after being captured and managed using REDCap electronic data capture tools hosted at the hospital [20]. REDCap (Research Electronic Data Capture) is a secure, web-based software platform designed to support data capture for research studies, providing: (1) an intuitive interface for validated data capture; (2) audit trails for tracking data manipulation and export procedures; (3) automated export procedures for seamless data downloads to common statistical packages; and (4) procedures for data integration and interoperability with external sources.

Scan QR code & read article online
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Grob, A., Bouaicha, S., Germann, M. et al. Is limited external rotation after reverse shoulder arthroplasty associated with glenoidal notching?. Obere Extremität 16, 272–280 (2021). https://doi.org/10.1007/s11678-021-00668-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11678-021-00668-3