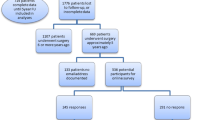
Abstract
Introduction
Definitions and prevalence of weight regain (WR) after bariatric surgery remains inconsistent and their clinical significance unclear.
Objectives
To assess WR five years after sleeve gastrectomy (LSG), employing six definitions; and appraise their association with patient characteristics/clinical outcomes.
Methods
Consecutive patients (N = 589) who underwent LSG were followed up for 5 years. WR prevalence was calculated yearly employing six definitions. Regression analysis assessed associations between WR at 5 years, and patient characteristics (age, sex, preop BMI, number of follow-up visits, number of comorbidities) and remission of comorbidities (type 2 diabetes, hypertension, and dyslipidemia).
Results
Sample’s mean age and BMI were 34 ± 11.6 years and 43.13 ± 5.77 kg/m2, and 64% were females. Percentage of patients with WR at 2, 3, 4, and 5 years fluctuated between 2.53% and 94.18%, subject to definition, and time point. The definition “Any WR” generated the highest prevalence of WR (86–94%) across all time points. At 5 years, for patient characteristics, preoperative BMI was associated with three definitions (P 0.49 to < 0.001), sex was associated with two (P < 0.026–0.032), and number of comorbidities was associated with one definition (P = 0.01). In terms of comorbidities, only hypertension was associated with WR (one definition, P = 0.025). No other definitions of WR were associated with any of the variables under examination.
Conclusion
Weight regain is reasonably expected after BMS. WR definitions were of minor clinical significance due to weak associations with limited comorbidities. Dichotomous definitions might offer some guidance while managing individual patients. However, its utility as a comparator metric across patients/procedures requires refinements.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Due to the excellent short- and long-term weight loss (WL) and durable improvements in obesity-associated comorbidities, bariatric and metabolic surgery (BMS) is considered the best treatment for extreme obesity [1,2,3,4,5,6]. However, not all patients maintain their achieved WL after BMS, and some experience weight regain (WR). For instance, the prevalence of WR ranged between 5.7 and 76% at 2–6 years after laparoscopic sleeve gastrectomy (LSG) [7, 8]; and a meta-analysis of patients with ≥ 7 years follow-up demonstrated a long-term WR rate of 27.8% (range 14–37%) [9].
Such wide variability in the proportion of patients who regain weight after BMS probably reflects the inconsistencies in the way WR is assessed. While standard definitions for WL after BMS surgery exist [10], the lack of a standardized WR definition results in variations and therefore imprecise comparisons across studies, procedures, settings, patient groups, and countries. This contributes to our poor understanding of WR and its significance [11].
Indeed, the definitions of WR after BMS are numerous and employ a wide range of different parameters, e.g., BMI or excess weight loss percentage (EWL%) as well as different cutoffs [12,13,14,15,16,17,18,19,20]. There have been calls to rectify this deficiency but identifying a single definition of clinically significant WR is challenging [21, 22].
Despite such calls, sparse research has assessed the prevalence of WR using the different definitions, and the literature remains limited in number and scope. For instance, in terms of the BMS procedure, most studies assessed the effects of WR definitions after Roux en Y gastric bypass (RYGB) only or after other BMS procedures, but not after laparoscopic sleeve gastrectomy (LSG), despite its popularity and different outcomes [12, 14, 21, 23,24,25]. Hence, the current study focused on LSG to bridge this gap. In addition, some studies appraised the effects of various definitions on WR rates at single time point, e.g., at five years [12, 21] without providing the prevalence of WR at multiple successive time points. While such an approach is useful, it provides a limited “single snapshot” view rather than the ongoing mechanics of the WR definitions across time. Equally important is that very few studies have evaluated how various WR definitions are associated with different comorbidities. Such understanding is critical to appraise the clinical implications of each of these definitions [14, 23]. WR has important health consequences including relapse of obesity-associated comorbidities including type 2 diabetes (T2DM), hypertension, and dyslipidemia [12, 24,25,26,27,28]
Given the above knowledge gaps, the aim of the present study was to assess the prevalence of WR after primary LSG using the different definitions. The specific objectives were to evaluate the extent of variability of WR rates based on six definitions measured at four time points (years 2, 3, 4, and 5 after LSG); and, appraise the associations between the different WR definitions and a range of patient characteristics as well as the remission of three comorbidities at 5 years after LSG. Clearer definitions will help to understand the clinical significance of such definitions, and hence provide guidance when intervention is required [11, 29]. We selected LSG as it is the most common procedure performed at our institution, comprising almost 95% of our surgeries. Patients with revisional BMS were excluded as nadir weight, which is essential for four of the six WR definitions we examined, would be difficult to determine precisely [14].
Materials and Methods
Study Design, Ethics, and Participants
This retrospective study was approved by the Medical Research Centre at Hamad Medical Corporation in Doha, Qatar (IRB Protocol #MRC-01–20-658). The inclusion criteria were all adults aged ≥ 18 years with BMI ≥ 40 or BMI ≥ 35 kg/m2 with comorbidities who underwent primary LSG at the BMS Centre in our institution and had ≥ 5 years follow-up data. Hence, patients operated upon from February 2016 to September of 2017 were eligible. A total of 598 patients underwent LSG during this time period. Eleven of these subsequently underwent revisional surgery and were excluded. The remaining 587 patients were included in the current analysis. The follow-up rate was 88.5, 85.8, 81, 74.9, and 61.1% at years 1, 2, 3, 4, and 5 respectively. The BMS service at our institution includes pre- and postoperative care provided in line with standard international guidelines [30]. The surgical technique of LSG at our institution has been described elsewhere and has not changed much since the establishment of the BMS department [5, 31].
Procedures and Data Collection
We searched patients’ electronic records and retrieved pre- as well postoperative data across the five years. Information retrieved included demographics (age, sex) anthropometric measures (weight, height) data, and other patient characteristics [(number of follow-up visits, number of comorbidities, medication use (based on pharmacy dispense records and physician documentation)]. We also retrieved clinical data [systolic and diastolic blood pressure (SBP, DBP)], as well as cardiometabolic/laboratory values [total cholesterol (TC), triglycerides (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL)].
Weight Loss and Regain Measures
We computed the BMI, BMI change, excess weight (EW), excess weight loss percentage (EWL%), weight loss (WL), and total weight loss percentage (TWL%) at 1, 2, 3, 4, and 5 years after surgery using formulas recommended in previous studies [10, 31, 32]. Nadir weight was the lowest value achieved of all postoperative weight measures available prior to year five. WR was calculated at 4 time points (starting from the second year) based on the six definitions. Definitions, cutoffs, and calculations of WR are shown in Table 1.
Definitions of Comorbidities and their Remission
The presence of the three comorbidities prior to surgery were retrieved and ascertained from the patients’ electronic records including type 2 diabetes (T2DM), hypertension (HTN), and dyslipidemia. Such patients were either already diagnosed before presenting to our bariatric clinic or diagnosed when screened prior to surgery according to agreed international standards [10, 33, 34]. At 5-year follow-up, the status of these comorbidities was determined using the ASMBS guidelines [10].
The Bariatric and Metabolic Service: Standard Postoperative Care
The evolution and components of the Bariatric and Metabolic Surgery Department at our institution have been detailed elsewhere [35]. After surgery, patients are routinely followed by a multidisciplinary team of bariatric surgeons, physicians, dietitians and physiotherapists. Follow-up visits are scheduled at 2 weeks and then at 1, 3, 6, and 12 months, and yearly thereafter. Dieticians and physical therapists individually counsel patients on the routine post-surgery dietary intake and physical activity in accordance with international guidelines.
Statistical Analysis
All values for weight loss and regain were calculated in Microsoft Excel using basic descriptive statistics and customized formulae. Categorical variables were expressed as frequencies and percentages, and continuous variables as means and standard deviations. Logic tests were used to determine patients who met individual definitions at each time point according to the definitions outlined in Table 1 and by others [21].
Binary logistic regression was undertaken to assess the associations between baseline patient characteristics (age, sex, preop BMI, number of follow-up visits, number of comorbidities) and WR at five years according to each of the six definitions. Logistic regression was also used to test the associations between WR by each definition with the remission of the three comorbidities under examination (hypertension, type 2 diabetes mellitus, and dyslipidemia). Assumptions regarding regression were tested and met. IBM SPSS Statistics version 20 (UK) was used for the analysis, and P value < 0.05 was considered statistically significant.
Results
Study Population: Preoperative and Selected Postoperative Characteristics
Table 2 depicts that the mean age of patients was 34 years and females were slightly more represented. More than half the sample were married and about three quarters were current smokers. Mean BMI was 43.13 kg/m2. The most common comorbidities were dyslipidemia, T2DM, and hypertension (about one fifth of the sample each). These were followed by prediabetes, asthma, hypothyroidism and back pain (10–11% each). Gastroesophageal reflux disease, obstructive sleep apnea, and coronary artery disease were less common (2–9% each). Other comorbidities were rare (< 1%). Mean blood pressure of the sample was normal. Mean number of follow-up visits with the bariatric clinics were 3.1 visits.
Anthropometric Characteristics at 5 Time Points
Across the total population, both weight and BMI showed a steady decrease until year 3 followed by a slight increase to year 5, echoed by the weight loss that displayed a steady decrease until year 3 followed by a slight increase to year 5 (Table 3). BMI change fluctuated to reach its maximum in year 2 (13.96 kg/m2) and its minimum in year 5 (11.67 kg/m2). Both %EWL and %TWL increased until year 2 and then started slightly decreasing up to year 5.
The number and percentage of patients reaching their nadir weight at each of the 4 time points were: at year 1 (189, 36.4%), year 2 (218, 43.3%), year 3 (89, 18.7%), year 4 (73, 16.82%); and for the whole sample, mean nadir weight was 75.40 ± 13.71 kg. A total of 529 (92.97%) patients achieved successful weight loss (defined as %EWL at nadir > 50), with a mean time to achieve minimum weight of 2.08 ± 1.00 years.
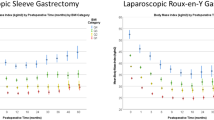
%TWL at 5 Time Points by WR Definition
The %TWL is presented in Fig. 1 for all patients and for every weight regain group separately. The %TWL for all groups was calculated relative to preoperative weight. As WR was calculated relative to nadir and the earliest time point where patients could have measured nadir was at year one, WR definitions were only applied from year 2 onwards. Definition 5 (any WR) consistently yielded the highest %TWL for any given year across all the examined years. All other definitions generated relatively lower %TWL for any given year across all the years.
Weight Regain Rates
Table 4 depicts that during years 2, 3, 4, and 5, the percentage of patients with WR fluctuated between 2.53% and 94.18%, subject to the definition and time point. At the 5-year follow-up, the highest prevalence of WR was that of definition 5 (any WR), where about 86% of patients had experienced WR. For the remaining definitions, the percentages of patients classified as experiencing weight regain ranged between 18 and 32%.
The definition yielding the least prevalence of WR differed by year but generally, definitions 3 and 4 were responsible for the lowest WR rates across the five time points. The remaining three definitions generated WR rates that fell in between the maximum and minimum rates (Fig. 2).
Associations of Weight Regain Definitions with Patient Characteristics at 5 Years
Table 5 shows that at 5 years, male sex was less likely to be associated with two definitions (defs 1 and 6; P range < 0.026 to 0.032). Patients with higher preoperative BMI had a higher likelihood of experiencing weight regain in three definitions (defs. 1, 3, and 4; P < 0.001–0.049). The number of comorbidities was positively associated with one definition (def. 2; P = 0.01). The remaining parameters under examination were not associated with any WR definitions.
On the other hand, definition 1 was significantly associated with two characteristics (sex, preoperative BMI), while definitions 2, 3, 4, and 6 were each significantly associated with only one parameter (def. 2 with number of comorbidities; definitions 3 and 4 with preoperative BMI; def. 6 with sex).
Associations of Weight Regain Definitions with Remission of Comorbidities at 5 Years
At five years, remission of hypertension was the only one associated with any of the WR definitions (def. 5, any WR, P = 0.025) (Table 6). No other WR definition was significantly associated with the remission of the comorbidities under examination.
Discussion
To date, definitions of WR after BMS are premised on a variety of different parameters, and within each parameter, employ a wide range of different cutoffs. Despite the variability in the way WR is defined and measured, it is frequently used as a key outcome and employed in comparisons of mid- and long-term effectiveness of bariatric procedures vis-a-vis each other [7, 9, 27, 36]. Such a situation inevitably raises the question: When definitions differ, are comparisons meaningful and do differences matter? In response, the current study appraised the extent of WR among a large sample of patients and explored its dynamics across five years using 6 different definitions. We also assessed the associations between different WR definitions and patient's characteristics as well as the remission of comorbidities at five years.
The main findings were that in terms of the time points, during years 2, 3, 4, and 5, the percentage of patients with WR fluctuated between 2.53 and 94.18%, subject to the definition and time point. At the 5-year follow-up, definition 5 (any WR) generated the highest prevalence of WR. The regression analysis at 5 years showed that sex, preoperative BMI and number of comorbidities were related to experiencing WR using various definitions. On the other hand, for remission of comorbidities, only hypertension remission was related to any of the WR definitions under examination. As expected, the definition of WR certainly influenced the WR prevalence, yielding considerable variations in the WR rates.
In terms of the breadth of the WR, at five years, 17.7–86% patients in the present study experienced WR subject to the definition used. Such a wide range of WR is very similar to the 16–87% WR prevalence five years after RYGB/LSG reported by Voorwinde et al. [21]. However, Lauti and colleagues [12] observed a higher WR (40–91%) using similar definitions as in the current study. Likewise, others observed WR ranging between 43.6–86.5% at five years after RYGB [14]. Two reasons might account for the lower WR rates observed by the current study and by Voorwinde et al. [21] compared to that of Lauti [12]. The first could be the lower mean preoperative BMI in our and Voorwinde’s studies [21] (43.12 and 44.8 kg/m2 respectively), compared to the higher baseline BMI of patients (50.7 kg/m2) in Lauti’s study [12]. Higher preoperative BMI is associated with a greater likelihood of WR [11, 37]. A second reason that cannot be ruled out could be the variations between centers and countries regarding the extent of multidisciplinary program before and after surgery and extent of follow-up on the recommended dietary, physical activity, and behavior modifications that might influence the sustainability of positive changes accomplished after BMS, hence playing a role in preventing WR or otherwise [11, 30, 38,39,40,41]. It is also not straight forward to rule out how patient characteristics might play a role if any, as 64.23% of our patients were females and mean age was 34 years; while these studies [12, 14, 21] had slightly higher percentage of females (79–81%) and an older population (46–49 years).
As for the magnitude of WR, based on the definition used, our highest proportion of patients with WR was with definition 6 (any WR, 86%), concurring with the findings of previous studies where this same definition yielded the highest WR rate (87–91%) [12, 21]. Notwithstanding, others found that the WR definition of “ ≥ 10% WR of maximum weight lost” was the one associated with highest proportion of patients with WR at five years (86.5%) [14]. On the other hand, in the current study, definition 4 yielded the lowest WR rate (17.7%), concurring with the findings of previous studies where this same definition resulted in the lowest WR rate (16%) [21].
Regarding the dynamics of WR across time, the proportion of patients with WR in the present study increased throughout the study period, regardless of the definition. This agrees with a study among RYGB patients and although the rate increased throughout the five years, the authors observed that the largest WR occurred during the first year after reaching nadir weight [14].
Collectively, the above findings highlight two points. The first is that some degree of WR occurs (and perhaps should be reasonably unsurprising) among most patients after BMS [11, 12, 21, 42] The second is that our findings as well as the literature illustrate a situation that requires overdue attention. They highlight the importance of debating and where possible standardizing the reporting of WR across the bariatric literature. The use of different WR definitions applied to the same population significantly alters the proportion of patients categorized as regaining weight. Such inconsistencies result in significant and clinically important findings.
As for the regression analysis, we observed that male sex was less likely to be associated with two definitions (definitions 1 and 6; P range < 0.026 to 0.032). Findings are inconsistent across studies, with some reporting no relation between sex and any of the 6 WR definitions [21]; or male gender being a predictor of WR [43]; or no association between gender and WR [23]. In connection with age, the current study noted no associations between age and WR across any of the definitions, where others found that age was inversely related to WR [21, 43].
As for preoperative BMI, it was positively associated with three WR definitions that incorporated weight and/or BMI (defs. 1, 3 and 4; P range < 0.001 to 0.049). This concurs with other studies where preoperative BMI was positively related to WR when definitions were based on changes in BMI, %EWL, and kilograms [21]. Likewise, one study showed that pre-surgery BMI was sometimes a positive significant predictor and at other times a negative predictor of WR subject to the definition used and its components (WR of “10 kg nadir,” “an increase of > 20% of maximum weight loss,” or “increase of > 25% EWL from nadir”) [23]. A recent study found that a preoperative BMI of > 45 kg/m2 was associated with WR after LSG, regardless of the definition of WR [44].
In terms of the number of comorbidities, we found it was positively associated with one definition (def. 2; P = 0.01). While there is no literature to directly compare this finding with, inconsistencies again exist. Some authors found that WR was associated with several comorbidities including obstructive sleep apnea and fatty liver disease [44], while others noted that the presence of T2DM or HTN were both not associated with WR definition [23].
Regarding to the remission of comorbidities, in the present study, WR was only significantly associated with remission of hypertension (def. 5 “any WR,” P = 0.025), albeit in a direction opposite to what would be expected. This suggests that comorbidity remission might not be entirely weight dependent. Such view is further supported by our observation that the remission of T2D and dyslipidemia were also not associated with any of the WR definitions. Our findings concur with other studies suggesting that WR might not largely be related to comorbidity remission. For instance, Voorwinde found that WR was associated with only one of the five comorbidities they examined (obstructive sleep apnea) [21]. Other mechanisms frequently play a role in resolution. For instance, several factors contribute to hypertension remission, including gut hormones (peptide YY and glucagon-like peptide-1), resolution of other obesity-related comorbidities that share pathophysiologic mechanisms with HTN (e.g., obstructive sleep apnea), decreases in systemic inflammation leading to reduced arterial stiffness, as well as decreased sodium reabsorption and diminished sympathetic activation [45,46,47,48].
As to the question of which WR definition is most appropriate to assess patient with WR, Table 7 provides a summary of design, patient populations, type of surgery, time points of assessment, and findings of the current and another four published studies that attempted to push the boundaries in order to answer this critical question. The table clearly illustrates the extent of the range of the variables that were assessed, when and how many times, as well as the inconsistencies in the findings and the wide variability in the reported range of values of WR and many dissimilarities in the associations/non-associations of different WR definitions with patient characteristics, resolution/remission of comorbidities, and patient perspectives (e.g., health-related QoL or satisfaction after surgery). The table undoubtedly shows that identifying one single categorical definition of clinically significant WR is difficult. Certainly, answering the question of which WR definition is the most appropriate to assess patient with WR requires further efforts and research from the bariatric community in order to reach a consensus.
This study has limitations. Nadir weight could have been reached between yearly assessments time points. Appraisal of remissions of obstructive sleep apnea and osteoarthritis would have been beneficial, although their prevalence was low among our sample. We had some missing data due to loss of follow-up and we are unable to confirm whether such missing values could be related to WR, as some studies found that patients that attend their postoperative appointments have better %EWL compared to those who failed to attend [49]. Data on patient perspectives would have been useful to collect, e.g., health-related quality of life or patient satisfaction with surgery. Future research should address these limitations. Despite this, the present study has many strengths. It is one of the very few studies to test different WR definitions, and one of the fewer to appraise such definitions across patients who had undergone LSG. Unlike others [21] where subjective patient-reported medication use and blood pressure were employed, the status of comorbidities and their remission in the current study was premised on objective findings (blood tests and medication used ascertained from pharmacy dispense records).
Final Thoughts—is Weight Regain a Useful Clinical Concept?
‘Exploring the unknown requires tolerating uncertainty’.
Brian Greene
Realistically, some WR is expected after BMS, thus multidisciplinary bariatric teams need to encourage long-term healthy lifestyle after surgery. Notwithstanding, the study findings uncover two critically challenging approaches.
On the one hand, using the findings of the current study pragmatically, awareness that higher preoperative BMI may be associated with longer term WR after LSG could prompt patients, when feasible, to seek earlier treatment, although this might not be entirely under their control. For the bariatric team, preoperative knowledge of patient groups at risk of WR, namely males with higher BMI and multiple comorbidities will help clinicians, where indicated, to offer earlier surgical intervention. Postoperatively, awareness of at-risk groups will also aid in providing early prevention or intervention when patients are not on their expected/optimal weight loss trajectory.
Conversely, on the other hand, the study findings raise concern about whether asking the question “Which WR definition is the most appropriate?” represents a suitable approach, and whether identifying a single categorical definition of clinically significant WR is a useful exercise. Perhaps the validity of WR as an outcome employed in comparisons and gauging of effectiveness of BMS procedures deserves a serious revisit. In particular, our findings as well as those of more recent studies [21] highly suggest that the current dichotomous WR definitions represent minor clinical significance as: (a) they showed mostly no associations or odd weak association with clinical outcomes; and (b) such associations were significant with only one of the comorbidities examined. Deliberations are required by expert groups, regional and international BMS agencies as to whether using metric definitions of WR after BMS represent an advantageous concept.
Given the above contrasting views, it might be argued that dichotomous WR definitions could offer some guidance in the assessment and management of individual patients. However, as a comparator metric across patients and procedures, its utility might need to be refined. Some authors have suggested a potential alternative concept that is increasingly being employed across the bariatric literature [50], where health benefits are assessed according to weight loss (e.g., sustained 5%, 10%, 15%, and 20% weight loss) [10, 51].
Conclusion
The prevalence WR after BMS remains inconsistent and its clinical significance uncertain. Our findings indicate that some WR could be reasonably expected after BMS. If preventing WR per se is viewed as an important dimension of success of BMS due to amelioration of comorbidities, then our findings indicate that the present dichotomous WR definitions are of minor clinical significance due to the weak associations with limited comorbidities. A deeper debate is required, as identifying a single categorical definition of clinically significant WR is difficult, and its full potential remains to be determined. Future research should address this challenge.
References
Gu L, Huang X, Li S, et al. A meta-analysis of the medium- and long-term effects of laparoscopic sleeve gastrectomy and laparoscopic Roux-en-Y gastric bypass. BMC Surg. 2020;20:30.
Neagoe R, Muresan M, Timofte D, et al. Long-term outcomes of laparoscopic sleeve gastrectomy – a single-center prospective observational study. Videosurgery Miniinvasive Tech. 2019;14:242–8.
Guraya SY, Strate T. Surgical outcome of laparoscopic sleeve gastrectomy and Roux-en-Y gastric bypass for resolution of type 2 diabetes mellitus: a systematic review and meta-analysis. World J Gastroenterol. 2020;26:865–76.
Peterli R, Wölnerhanssen BK, Peters T, et al. Effect of laparoscopic sleeve gastrectomy vs laparoscopic Roux-en-Y gastric bypass on weight loss in patients with morbid obesity: the SM-BOSS randomized clinical trial. JAMA. 2018;319:255–65.
Elgenaied I, El Ansari W, Elsherif MA et al. Factors associated with complete and partial remission, improvement, or unchanged diabetes status of obese adults 1 year after sleeve gastrectomy. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2020;
Salminen P, Helmiö M, Ovaska J, et al. Effect of laparoscopic sleeve gastrectomy vs laparoscopic Roux-en-Y gastric bypass on weight loss at 5 years among patients with morbid obesity. JAMA. 2018;319:241–54.
Lauti M, Kularatna M, Hill AG, et al. Weight regain following sleeve gastrectomy-a systematic review. Obes Surg. 2016;26:1326–34.
Braghetto I, Csendes A, Lanzarini E, et al. Is laparoscopic sleeve gastrectomy an acceptable primary bariatric procedure in obese patients? Early and 5-year postoperative results. Surg Laparosc Endosc Percutan Tech. 2012;22:479–86.
Clapp B, Wynn M, Martyn C, et al. Long term (7 or more years) outcomes of the sleeve gastrectomy: a meta-analysis. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2018;14:741–7.
Brethauer SA, Kim J, el Chaar M, et al. Standardized outcomes reporting in metabolic and bariatric surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2015;11:489–506.
El Ansari W, Elhag W. Weight regain and insufficient weight loss after bariatric surgery: definitions, prevalence, mechanisms, predictors, prevention and management strategies, and knowledge gaps-a scoping review. Obes Surg. 2021;31:1755–66.
Lauti M, Lemanu D, Zeng ISL, et al. Definition determines weight regain outcomes after sleeve gastrectomy. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2017;13:1123–9.
Grover BT, Morell MC, Kothari SN, et al. Defining weight loss after bariatric surgery: a call for standardization. Obes Surg. 2019;29:3493–9.
King WC, Hinerman AS, Belle SH, et al. Comparison of the performance of common measures of weight regain after bariatric surgery for association with clinical outcomes. JAMA. 2018;320:1560–9.
Carmeli I, Golomb I, Sadot E, et al. Laparoscopic conversion of sleeve gastrectomy to a biliopancreatic diversion with duodenal switch or a Roux-en-Y gastric bypass due to weight loss failure: our algorithm. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2015;11:79–85.
Brethauer SA, Aminian A, Romero-Talamás H, et al. Can diabetes be surgically cured? Long-term metabolic effects of bariatric surgery in obese patients with type 2 diabetes mellitus. Ann Surg. 2013;258:628–36 (discussion 636-637).
Liu SY-W, Wong SK-H, Lam CC-H, et al. Long-term results on weight loss and diabetes remission after laparoscopic sleeve gastrectomy for a morbidly obese Chinese population. Obes Surg. 2015;25:1901–8.
Homan J, Betzel B, Aarts EO, et al. Secondary surgery after sleeve gastrectomy: Roux-en-Y gastric bypass or biliopancreatic diversion with duodenal switch. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2015;11:771–7.
Abdallah E, El Nakeeb A, Youssef T, et al. Impact of extent of antral resection on surgical outcomes of sleeve gastrectomy for morbid obesity (a prospective randomized study). Obes Surg. 2014;24:1587–94.
Bohdjalian A, Langer FB, Shakeri-Leidenmühler S, et al. Sleeve gastrectomy as sole and definitive bariatric procedure: 5-year results for weight loss and ghrelin. Obes Surg. 2010;20:535–40.
Voorwinde V, Steenhuis IHM, Janssen IMC, et al. Definitions of long-term weight regain and their associations with clinical outcomes. Obes Surg. 2020;30:527–36.
Elhag W, Ansari WE. Weight regain and insufficient weight loss after bariatric surgery: a call for action. In: Bariatric surgery - from the non-surgical approach to the post-surgery individual care, Nieves Saiz-Sapena and Juan Miguel Oviedo (Eds) [Internet]. IntechOpen; 2020 [cited 2021 Oct 24]. Available from: https://www.intechopen.com/online-first/74559
Torrego-Ellacuría M, Barabash A, Larrad-Sainz A, et al. Weight regain outcomes after bariatric surgery in the long-term follow-up: role of preoperative factors. Obes Surg. 2021;31:3947–55.
Uittenbogaart M, de Witte E, Romeijn MM, et al. Primary and secondary nonresponse following bariatric surgery: a survey study in current bariatric practice in the Netherlands and Belgium. Obes Surg. 2020;30:3394–401.
Grönroos S, Helmiö M, Juuti A, et al. Effect of laparoscopic sleeve gastrectomy vs Roux-en-Y gastric bypass on weight loss and quality of life at 7 years in patients with morbid obesity. JAMA Surg. 2021;156:137–46.
Karmali S, Brar B, Shi X, et al. Weight recidivism post-bariatric surgery: a systematic review. Obes Surg. 2013;23:1922–33.
Aminian A, Vidal J, Salminen P, et al. Late relapse of diabetes after bariatric surgery: not rare, but not a failure. Diabetes Care. 2020;43:534–40.
Abdulrazzaq S, Elhag W, El Ansari W, et al. Is Revisional gastric bypass as effective as primary gastric bypass for weight loss and improvement of comorbidities? Obes Surg. 2020;30:1219–29.
Nedelcu M, Khwaja HA, Rogula TG. Weight regain after bariatric surgery-how should it be defined? Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2016;12:1129–30.
Mechanick JI, Apovian C, Brethauer S, et al. Clinical practice guidelines for the perioperative nutrition, metabolic, and nonsurgical support of patients undergoing bariatric procedures - 2019 update: cosponsored by American Association of Clinical Endocrinologists/American College of Endocrinology, The Obesity Society, American Society for Metabolic & Bariatric Surgery, Obesity Medicine Association, and American Society of Anesthesiologists. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2020;16:175–247.
Hatoum IJ, Kaplan LM. Advantages of percent weight loss as a method of reporting weight loss after Roux-en-Y gastric bypass. Obes Silver Spring Md. 2013;21:1519–25.
Lemmens HJM, Brodsky JB, Bernstein DP. Estimating ideal body weight–a new formula. Obes Surg. 2005;15:1082–3.
2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2022 | Diabetes Care | American Diabetes Association [Internet]. [cited 2022 Nov 18]. Available from: https://diabetesjournals.org/care/article/45/Supplement_1/S17/138925/2-Classification-and-Diagnosis-of-Diabetes
Unger T, Borghi C, Charchar F, et al. International Society of Hypertension Global Hypertension Practice Guidelines. Hypertens Am Heart Assoc. 2020;75:1334–57.
Elhag W, El Ansari W. Durability of cardiometabolic outcomes among adolescents after sleeve gastrectomy: first study with 9-year follow-up. Obes Surg. 2021;
Baig SJ, Priya P, Mahawar KK, et al. Weight Regain after bariatric surgery-a multicentre study of 9617 patients from Indian Bariatric Surgery Outcome Reporting Group. Obes Surg. 2019;29:1583–92.
Ochner CN, Jochner MCE, Caruso EA, et al. Effect of preoperative body mass index on weight loss after obesity surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2013;9:423–7.
Tettero OM, Aronson T, Wolf RJ, et al. Increase in physical activity after bariatric surgery demonstrates improvement in weight loss and cardiorespiratory fitness. Obes Surg. 2018;28:3950–7.
Bellicha A, van Baak MA, Battista F, et al. Effect of exercise training on weight loss, body composition changes, and weight maintenance in adults with overweight or obesity: an overview of 12 systematic reviews and 149 studies. Obes Rev Off J Int Assoc Study Obes. 2021;22(Suppl 4):e13256.
Sarwer DB, Moore RH, Spitzer JC, et al. A pilot study investigating the efficacy of postoperative dietary counseling to improve outcomes after bariatric surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2012;8:561–8.
Bradley LE, Forman EM, Kerrigan SG, et al. A pilot study of an acceptance-based behavioral intervention for weight regain after bariatric surgery. Obes Surg. 2016;26:2433–41.
Courcoulas AP, King WC, Belle SH, et al. Seven-year weight trajectories and health outcomes in the Longitudinal Assessment of Bariatric Surgery (LABS) study. JAMA Surg. 2018;153:427–34.
Shantavasinkul PC, Omotosho P, Corsino L, et al. Predictors of weight regain in patients who underwent Roux-en-Y gastric bypass surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2016;12:1640–5.
Yarigholi F, Bahardoust M, Mosavari H, et al. Predictors of weight regain and insufficient weight loss according to different definitions after sleeve gastrectomy: a retrospective analytical study. Obes Surg. 2022;
Climent E, Oliveras A, Pedro-Botet J, Goday A, Benaiges D. Bariatric surgery and hypertension. J Clin Med [Internet]. Multidisciplinary Digital Publishing Institute (MDPI); 2021 [cited 2022 Nov 19];10. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8467380/
Landsberg L, Aronne LJ, Beilin LJ, et al. Obesity-related hypertension: pathogenesis, cardiovascular risk, and treatment–a position paper of the The Obesity Society and The American Society of Hypertension. Obes Silver Spring Md. 2013;21:8–24.
le Roux CW, Welbourn R, Werling M, et al. Gut hormones as mediators of appetite and weight loss after Roux-en-Y gastric bypass. Ann Surg. 2007;246:780–5.
Pedrosa RP, Krieger EM, Lorenzi-Filho G, et al. Recent advances of the impact of obstructive sleep apnea on systemic hypertension. Arq Bras Cardiol. 2011;97:e40-47.
Shen R, Dugay G, Rajaram K, et al. Impact of patient follow-up on weight loss after bariatric surgery. Obes Surg. 2004;14:514–9.
Shahwan S, Oochit K, Campbell E, et al. Reporting of weight loss outcomes in bariatric surgery following introduction of 2015 ASMBS guidelines. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2022;18:1195–8.
Rubino DM, Greenway FL, Khalid U, et al. Effect of weekly subcutaneous semaglutide vs daily liraglutide on body weight in adults with overweight or obesity without diabetes: the STEP 8 randomized clinical trial. JAMA. 2022;327:138–50.
Acknowledgements
The authors thank the bariatric teams involved. We also thank the nursing team: Amira El Said, Hend Elsayed, Lobna Cherni, Habiba Ramadan, and Mr. Arnel Alviz for their assistance with data collection.
Funding
Open Access funding provided by the Qatar National Library.
Author information
Authors and Affiliations
Contributions
Walid El Ansari and Wahiba Elhag participated in the design of the study, acquisition of datasets, interpretation of findings, and writing and revising the manuscript. Merilyn Lock applied the weight regain definitions, undertook statistical analysis, and revised the manuscript. All authors critically reviewed and approved the final version of this paper.
Corresponding author
Ethics declarations
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Statement of Informed Consent
The informed consent was waived (IRB approved, HIPAA compliant retrospective study).
Statement of Human and Animal Rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Elhag, W., Lock, M. & El Ansari, W. When Definitions Differ, are Comparisons Meaningful? Definitions of Weight Regain After Bariatric Surgery and Their Associations with Patient Characteristics and Clinical Outcomes - A Need for a Revisit?. OBES SURG 33, 1390–1400 (2023). https://doi.org/10.1007/s11695-023-06528-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-023-06528-z