Abstract
Purpose
Medical follow-up after bariatric surgery is recommended. However, the compliance was poor. This study aimed to evaluate the feasibility of a smartphone-based fully remote follow-up (FU) program for patients after bariatric surgery.
Methods
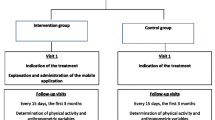
In the interventional group, patients were followed up using a smartphone application (app), through which questionnaires were sent regularly. Participants in the control group underwent standard FU at the outpatient clinic every three months. After 12 months, all the participants were evaluated at an outpatient clinic.
Results
Between August 2020 and March 2021, 44 and 43 patients in the interventional and control groups, respectively, were included in the analysis after three patients were lost to FU, and three withdrew their informed consent because they wished for more personal contact with medical caregivers. After 12 months, total weight loss (TWL), %TWL, and percentage of excess weight loss (%EWL) did not differ between groups. There were no significant differences in the complication rates, including surgical complications, malnutrition, and micronutrition deficiency. The parameters of bioelectrical impedance analysis and quality of life did not differ between the groups. Vitamins and minerals in serum were similar in both groups except for calcium, which was significantly higher in the interventional group (2.52 mmol/L vs. 2.35 mmol/L, p = 0.038).
Conclusion
Fully remote FU with a smartphone application is at least as effective as traditional in-person FU in an outpatient clinic after bariatric surgery. Through remote FU, patients can save time and medical professionals may have more resources for patients with more severe problems.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Over the past decades, bariatric surgery has been proven to be the most effective and durable therapy to reduce weight and comorbidities in patients with clinically severe obesity over the past decades [1,2,3]. Since bariatric surgeries are associated with long-term complications such as malnutrition and micronutrient deficiency [4], follow-up (FU) care after bariatric care is recommended in national and international guidelines [5,6,7,8]. Besides the early recognition of complications, loss to short-term FU (up to 3 years after surgery) is associated with insufficient weight loss (WL) [9,10,11,12,13], while the data for long-term FU (> three years) are controversial [14,15,16]. However, some remarkable problems concerning FU remain unsolved. The compliance to FU was poor: 62.1% of the patients adhered to FU after 12 months [17], and the adherence rate dropped to 44.6% during a 3-year postoperative period [18]. With the increasing number of bariatric surgeries owing to an increase in the incidence of extreme obesity, the number of patients who need postoperative FU is also rising, which results in logistical and financial challenges for bariatric centers [19].
Mobile health (mHealth), including smartphone applications (apps), has become a popular platform for delivering health services since the COVID-19 pandemic [20,21,22,23,24]. mHealth interventions as tools for the education and engagement of patients before bariatric surgery seem promising [25, 26]. Nevertheless, there is limited evidence regarding its effectiveness in the postoperative FU phase. In our usability study, a smartphone-based FU program was proven to be well accepted after six months [27].
This study aimed to evaluate smartphone-based health interventions for patients after bariatric surgery. We hypothesized that the mHealth intervention was at least as effective in weight loss and preventing vitamin and mineral deficiencies as a standard in-person follow-up in a bariatric center.
Methods
Study Design
This study was designed as a prospective, single-center analysis comparing the effects of in-person and smartphone app-based postoperative follow-up after bariatric surgery. The study was approved by the University Faculty Ethics Committee and Institutional Review Board(#2018-643N-MA) and was conducted at the university hospital. The trial is registered in the German Clinical Trials Register (DRKS00016143).
Inclusion
Participants were patients with obesity (body mass index [BMI] ≥ 35 kg/m2 with one or more comorbidities [e.g., diabetes, arterial hypertension, sleep apnea] or BMI ≥ 40 kg/m2) who underwent primary bariatric surgery.
Participants in the interventional group were included in the BELLA trial when they presented to the outpatient clinic for their first postoperative follow-up two weeks after the surgery. Patients with re-do procedures, impaired mental state, inability to use a smartphone, language barriers, or severe postoperative complications (grade II, III, and IV according to the Clavien-Dindo classification [28]) up to the first follow-up visit were excluded.
After recruiting participants in the interventional group, the control cohort was selected based on comparable patient characteristics including median age, sex, preoperative weight, preoperative BMI, and type of surgery. All medical records were extracted from electronic patient files by the coauthors.
Participants in the control group were recruited 12 months after the bariatric surgery during the follow-up visit.
Participation for all patients was predicated on written informed consent.
Intervention
The detailed procedures and questionnaire have been described in our previous publication [27]. Patients in the interventional group were followed up exclusively using a smartphone app instead of attending in-person visits to the outpatient clinic. The smartphone application MYONCARE™ (OnCare GmbH, Munich, Germany) was installed on their personal smartphones after obtaining informed consent. As a replacement for the in-person follow-up visits, a standardized questionnaire (see Appendix) based on the database of the German register for obesity and metabolic surgery [29] was sent to the participants via the app on scheduled appointments: every six weeks during the first nine months and then one year after the surgery. A warning message was delivered to the account of the authorized healthcare professionals if the patients' responses surpassed a predetermined threshold and suggested a potential concern (i.e., patients reported experiencing severe pain with a numerical rating scale (NRS) score > 5). Minor health problems could usually be handled by bariatric or study nurses. If patients reported major health issues, they were forwarded to physicians or the emergency department (ED) on time. The in-app communication feature of the app allows users to communicate with medical professionals. However, patients were informed not to use the in-app chat function in emergencies but to go to the ED. The app enabled the transmission of test results by general practitioners or other medical specialists. Additionally, the participants received weekly push notifications to remind them of taking vitamins or exercising. Laboratory tests were performed by primary physicians or endocrinologists six months after the surgery and then in the bariatric center 12 months after the surgery during the final visit.
Standard Care
The control group received the usual care at the bariatric center. After the first postoperative follow-up (two weeks after surgery) with a bariatric surgeon, they were required to be present in the bariatric center for follow-up every three months in the first year after surgery. According to the German guideline [8], a postoperative follow-up visit included monitoring weight and comorbidity, assessment of eating behavior and physical activity, monitoring of vitamin and mineral supplementation, screening for mental problems and complications, such as malnutrition or malabsorption, and initiation of interventions. Laboratory tests were performed by primary physicians or in the bariatric center every six months in the first year.
Measures
Participants in the interventional and control groups were required to attend an in-person visit to the bariatric center 12 months after surgery. Sociodemographic (sex and age) and clinical parameters (weight, height, metabolic diseases or other comorbidities) were assessed immediately before and 12 months after surgery.
Weight Loss Outcomes
Percentage of excess weight loss (%EWL) and percentage of excess BMI loss (%EBMIL) are the two most widely used outcome measures in bariatric surgery [30]. %EWL is less accurate as an outcome measure than absolute weight [31] and the results show significant variation by initial BMI. The percentage of total weight loss (%TWL) has been reported to be less influenced by confounding factors [32]. However, a 50%EWL is often used as a milestone for bariatric goals [32]. These weight loss parameters were calculated as follows:
Bariatric Quality of Life Index
The Bariatric Quality of Life Index (BQL) is a validated instrument that assesses patients’ life (QoL) before and after bariatric surgery [33, 34]. The original BQL consists of 30 questions divided into two parts: non-QoL and QoL. In the German Nationwide Register, only the QoL part, consisting of 13 items with a five-point Likert scale ranging from 1 to 5 points, was included. The final score was calculated by adding all item scores, with a higher score representing a better QoL.
Lab Tests
The following blood tests were performed 12 months after bariatric surgery: complete blood count (CBC), liver function tests, lipid profile, iron studies, calcium, vitamin D3, vitamin B12, vitamin B1, parathyroid hormone, serum folate, and serum zinc.
Bioelectrical Impedance Analysis
Bioelectrical impedance analysis (BIA) is commonly performed for the evaluation of pre- and postoperative body composition, delivering the parameters of body cell mass (BCM), fat mass (FM), FM in %, and phase angle, which can be regarded as a marker of training and nutritional status [35].
Statistical Analysis
All statistical calculations were performed with the SAS statistical program, release 9.4 (SAS Institute Inc., Cary, North Carolina, USA) and R version 3.6.3. For quantitative variables, mean and standard deviation were assessed. For qualitative factors, absolute and relative frequencies were calculated. To compare the two treatment groups with respect to baseline values, Fisher’s exact test or two-sample t-test was applied, as appropriate. In general, the results of a statistical test were considered statistically significant at a p-value of < 0.05.
Results
Baseline Characteristics of Participants
Between August 2020 and March 2021, of the 75 patients assessed for eligibility in the interventional group, 52 agreed to participate in the study. Eight patients dropped out of the study: one was excluded because of limited reading ability, one lost his smartphone and could not afford placement, three were lost to follow-up, and three withdrew their informed consent. Therefore, 44 participants (84.6%) were included in the final analysis. In the control group, 43 of the 74 patients assessed for eligibility were included. Medical care was provided to patients within a wide geographical area (up to 355 km). The average distance from their residence to the clinic was greater in the interventional group than the control group (42.5 km vs. 23.0 km, p = 0.06), but the dispersion of the data was very high (standard deviation = 62.5 in the interventional group and 25.0 in the control group).
The demographic information of the participants is presented in Table 1. The basic patient characteristics did not differ significantly between the groups.
Weight Loss Outcomes
Weight was assessed during the mandatory in-person visit to the bariatric center 12 months after surgery. As shown in Table 2, none of the weight loss parameters differed significantly between the intervention and control groups.
Complications and Readmission
One patient (2%) from each group visited the ED within 30 days, but none were readmitted. During the entire FU period, eight (18.6%) patients visited the ED in the control group: four (9.3%) patients presented with abdominal pain, one(2%) patient with nausea, one (2%) with superficial surgical site infection and one (2%) with anastomotic ulceration; only one (2%) patient was diagnosed with acute cholecystitis or symptomatic cholelithiasis and was readmitted for surgery. In the interventional group, 14 (31.8%) patients presented to the ED: seven (15.9%) with abdominal pain, one (2%) with diarrhea; six (13.6%) required readmission for surgery (five (11.4%) for acute cholecystitis or symptomatic cholelithiasis, one (2%) for bile reflux). These differences were not statistically significant (Table 3).
Bariatric Quality of Life Index
The BQL score was slightly higher in the intervention group, but the difference was not significant. (52.6 versus 51.9, p = 0.7602).
Malnutrition and Micronutrient Deficiency
Albumin
The albumin level in blood did not differ significantly between the interventional and control group (39.9 mg/dL vs. 39.2 mg/dL, p = 0.644). In the interventional group, one of 29 patients (3.4%) had hypoalbuminemia (< 35 mg/dL), and in the control group three of 23 (13.0%) had hypoalbuminemia. The difference in hypoalbuminemia was not significant (p = 0.310).
Calcium
The serum calcium of the interventional group was significantly higher than the control group (2.52 mmol/L vs. 2.35 mmol/L, p = 0.038). Hypocalcemia (< 2.18 mmol/l) was diagnosed in two out of 22 patients in the control group and none in the interventional group (p = 0.181).
Lipid Profile
No significant difference in total cholesterol and triglyceride levels was found between the interventional and control group (166.4 vs. 161.9, p = 0.710; 94.5 vs. 105.7, p = 0.359, respectively). For LDL and HDL, statistical analysis was not possible owing to missing values.
Vitamins and Minerals
Serum levels of vitamin D, vitamin B12, and vitamin B1 were not significantly different between the groups (29.7 vs. 32.9, p = 0.534; 719.0 vs. 746.2, p = 0.854; 81.4 vs. 80.3, p = 0.880, respectively). There was no significant difference in the serum zinc or folate significant (14.6 vs. 16.0, p = 0.837; 18.0 vs. 19.5, p = 0.634).
Vitamin D deficiency (< 20 ng/mL) was diagnosed in eight of 27 patients (29.6%) in the interventional group and five of 17 (29.4%) in the control group (p = 1). No patient in either group was diagnosed with vitamin B1 or B12 deficiency.
Compliance with Supplements of Vitamins and Minerals
Compliance with supplements was higher in the control group:69.2% of the participants in the interventional group and 88.4% in the control group stated that they had been taking vitamin and mineral supplements daily. However, this difference was not statistically significant (p = 0.054).
BIA
BCM
BCM did not differ significantly between groups (29.4 vs. 27.8, p = 0.341).
Fat Mass and FM in %
Both FM and %FM did not significantly differ (32.8 vs. 33.2, p = 0.902; 34.2 vs. 35.2, p = 0.568).
Phase Angle
The phase angle was slightly higher in the intervention group (5.4 vs. 5.1°), but the difference did not reach significance (p = 0.086).
Discussion
This study reports the first published German experience of follow-up after bariatric surgery using a mobile application. In our interventional cohort of patients, 84.6% were successfully followed-up with mHealth within the first 12 months. Weight loss outcomes, quality of life, malnutrition, micronutrient deficiency, and body impedance analysis results were similar to those of a cohort of patients receiving standard in-person follow-up care. These data suggest that postoperative follow-up using a mobile application is feasible in patients undergoing bariatric surgery, and a fully remote follow-up program represents a conceivable alternative to the standard in-person follow-up in an outpatient clinic.
Compliance with post-bariatric FU remains challenging and a global problem. Several patient-related risk factors for loss to FU have been identified, including younger age, persistent comorbidities, and financial challenges [9, 15]. Younger patients without comorbidities may not understand the importance of FU and may be less aware of their personal health. Therefore, they may feel it unnecessary to “waste time” visiting the hospital for a simple check-up. It is conceivable to involve this cohort in a remote FU using smartphones since 95% of adults aged 18 to 49 own a smartphone in 2021 in the USA [36], and they spend a remarkable amount of time on it. As of April 2021, the average daily time spent on a phone, excluding speaking on the phone, has climbed in recent years, totaling 4 h and 23 min [37]. Additionally, the geographical distance to the clinic/hospital may impact the readiness for follow-up.
The two most important reasons for adherence to FU after bariatric surgery are sufficient weight loss and the early detection of complications. Although the importance of FU for mid-to-long-term WL remains controversial [15, 38], a significant association between WL and FU within one year after bariatric surgery has been pointed out by several authors [9,10,11, 39]. In the current study, both groups had comparable BMI before surgery. After 12 months of FU using a mobile app instead of in-person visits to the bariatric center, participants in the interventional group achieved similar %EWL and %TWL compared to those with in-person visits. Some predictive factors of WL are no longer influenceable after surgery, such as sex, age, type and quality of surgery, and previous comorbid conditions. However, other factors, such as behavioral variables, can still be influenced, including physical activity and eating behaviors [40]. mHealth has been proven to be a valuable tool for managing nutrition and exercise programs in patients with overweight or obesity in diverse life situations [41,42,43,44] and plays an important role in WL in our patients. Changes in body composition after bariatric surgery have been reported in previous studies [45, 46], and the changes were similar in both groups. Moreover, the scores of bariatric quality of life were similar, which is a valid instrument for patients after bariatric surgery with better responsiveness than generic questionnaires [33].
Complication rates, including surgical complications, malnutrition, and micronutrient deficiency, were comparable between the groups. The most frequent reasons for visiting the ED were abdominal pain of unknown origin and acute cholecystitis/symptomatic cholelithiasis, which is in line with previous data in the literature [47]. The number of readmissions was slightly higher in the interventional group owing to a higher incidence of acute cholecystitis/symptomatic cholecystolithiasis; however, the difference was not statistically significant. This tendency might be explained by the fact that patients in the intervention group had easier access to medical professionals due to the chat function of the app and were thus more aware of the symptoms.
It has been well established that bariatric surgery is associated with malnutrition and micronutrient deficiency, mainly attributed to lower absorption [48]. Serum albumin level has been identified as an indicator of malnutrition, and hypoalbuminemia is usually defined as an albumin concentration of < 35 mg/dL. The serum albumin level and prevalence of hypoalbuminemia did not differ between the groups. The incidence of vitamin D deficiency after bariatric surgery has been reported to be between 10 and 73% after bariatric surgery [49]. After bariatric surgery, 3.6% of the patients were diagnosed with hypocalcemia, and the prevalence depended on the surgical type [50]. In addition to vitamin D, micronutrient deficiency was rare in both groups, and the prevalence was comparable in both groups, which might be explained by two reasons. First, all patients took part in a minimally 6-month-long multimodal concept including intensive consultation by a nutritional therapist before the surgery. Thus, they were aware of the consequences of malnutrition and micronutritional deficiencies. Second, the compliance with FU and supplement intake has proven to be the best in the first year [51]. If the patients were followed up for a more extended period with longer intervals, a dramatic drop in compliance with FU would be expected in the control group according to previous experience in the literature. Patients could benefit from a remote FU using a mobile app, since regular reminders of vitamin supplementation could be sent even years after surgery. The first indication is a significantly higher calcium level in the interventional group.
Patients could benefit from a remote FU after bariatric surgery because they do not need to travel a long distance to reach the hospital, spend time parking and waiting in a busy outpatient clinic. Furthermore, they were free to answer FU questionnaires and contact medical professionals in non-emergency anywhere and any when [27]. Medical professionals could also benefit from a remote FU: instead of organizing outpatient appointments and seeing post-bariatric patients four times per year, a standard FU could be performed, and minor problems could be solved remotely. It can take pressure away from medical professionals so that they can focus on caring for patients with more severe problems.
In our current trials, eight participants in the intervention could not be followed up using the mobile app; one patient was excluded because of limited reading ability, which had not been conscious for us during the recruitment. One patient lost his smartphone and could not afford a placement. Three patients were lost to follow-up for unknown reasons, and three withdrew their informed consent because they experienced technical problems with the app and wished for more personal contact with medical caregivers.
Limitations
Our study had some limitations. First, selection bias could not be ruled out because it was not a randomized trial, and the control group was recruited during their 12-month follow-up appointment in the outpatient clinic. Most patients in the control group underwent bariatric surgery before the COVID-19 pandemic, while some patients in the interventional group underwent surgery during the pandemic. This might have had some impact on the willingness to participate in a remote follow-up program. Second, we only assessed data up to 12 months after surgery, which might not reflect problems in mid-to long-term follow-up. Third, owing to the characteristics of a feasibility study and missing previous data, a sample size calculation was not possible, and the number of included patients might be too low to detect differences between the groups. These conclusions need to be verified in further randomized trials. Based on the data obtained in the pilot study, we were able to perform a sample size calculation and conduct a further multicenter, randomized clinical trial to remove the ambiguities described above. In this pilot trial, we focused on the feasibility of remote follow-up and did not explicitly assess its financial aspect. To address the financial questions, the workload of healthcare providers and the travel costs of patients for in-person clinic visits will be documented and analyzed in the main trial.
Conclusions
To the best of our knowledge, this is the first study to investigate the feasibility of a smartphone-based follow-up program for patients after bariatric surgery. Our data indicate that a fully remote follow-up program is at least as effective as a conventional in-person follow-up after bariatric surgery. Thus, a fully remote follow-up might help save resources in the outpatient clinic so that medical professionals can focus on patients with more severe problems.
References
Wadden TA, et al. Eight-year weight losses with an intensive lifestyle intervention: the look AHEAD study. Obesity. 2014;22:5–13.
Sheng B, et al. The long-term effects of bariatric surgery on type 2 diabetes remission, microvascular and macrovascular complications, and mortality: a systematic review and meta-analysis. Obes Surg. 2017;27:2724–32.
Docherty NG, le Roux CW. Bariatric surgery for the treatment of chronic kidney disease in obesity and type 2 diabetes mellitus. Nat Rev Nephrol. 2020;16:709–20.
Bal BS, Finelli FC, Shope TR, Koch TR. Nutritional deficiencies after bariatric surgery. Nat Rev Endocrinol. 2012;8:544–56.
Mechanick JI, et al. Clinical Practice Guidelines for the Perioperative Nutrition, Metabolic, and Nonsurgical Support of Patients Undergoing Bariatric Procedures - 2019 Update: Cosponsored by American Association Of Clinical Endocrinologists/American College of Endocrinology, The Obesity Society, American Society for Metabolic & Bariatric Surgery, Obesity Medicine Association, and American Society Of Anesthesiologists - Executive Summary. Endocr Pract Off J Am Coll Endocrinol Am Assoc Clin Endocrinol. 2019;25:1346–59.
Shiau J, Biertho L. Canadian adult obesity clinical practice guidelines: bariatric surgery: postoperative management. Downloaded from: https://obesitycanada.ca/guidelines/postop. Accessed 4 Apr 2023.
The National Institute for Health and Care Excellence. Obesity: identification, assessment and management. Downloaded from https://www.nice.org.uk/guidance/cg189/resources/obesity-identification-assessment-and-management-pdf-35109821097925. Accessed 4 Apr 2023.
Deutsche Gesellschaft für Allgemein- und Viszeralchirurgie e.V. (DGAV)S3-Leitlinie: Chirurgie der Adipositas und metabolischer Erkrankungen. Downloaded from https://register.awmf.org/assets/guidelines/088-001l_S3_Chirurgie-Adipositas-metabolische-Erkrankugen_2018-02.pdf. Accessed 4 Apr 2023.
McVay MA, Friedman KE, Applegate KL, Portenier DD. Patient predictors of follow-up care attendance in Roux-en-Y gastric bypass patients. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2013;9:956–62.
Kim HJ, Madan A, Fenton-Lee D. Does patient compliance with follow-up influence weight loss after gastric bypass surgery? A systematic review and meta-analysis. Obes Surg. 2014;24:647–51.
Shen S-C, Lin H-Y, Huang C-K, Huang C-K, Yen Y-C. Adherence to psychiatric follow-up predicts 1-year BMI loss in gastric bypass surgery patients. Obes Surg. 2016;26:810–5.
Fried M, et al. Interdisciplinary European guidelines on metabolic and bariatric surgery. Obes Surg. 2014;24:42–55.
Gould JC, Garren MJ, Boll V, Starling JR. Laparoscopic gastric bypass: risks vs. benefits up to two years following surgery in super-super obese patients. Surgery. 2006;140:524–9.
Morgan DJR, Ho KM, Armstrong J, Litton E. Long-term clinical outcomes and health care utilization after bariatric surgery: a population-based study. Ann Surg. 2015;262:86–92.
Reiber BMM, Barendregt R, de Vries R, Bruin SC, van der Peet DL. Is adherence to follow-up after bariatric surgery necessary? A systematic review and meta-analysis. Obes Surg. 2022;32:904–11.
Reiber BMM, Leemeyer A-MR, Bremer MJM, de Brauw M, Bruin SC. Weight loss results and compliance with follow-up after bariatric surgery. Obes Surg. 2021;31:3606–14.
Larjani S, et al. Preoperative predictors of adherence to multidisciplinary follow-up care postbariatric surgery. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2016;12:350–6.
Barka I, et al. Sociodemographic factors associated with loss to follow-up after bariatric surgery. Obes Surg. 2021;31:2701–8.
Marjanovic G, Fink J, Seifert G, Rupp E. Adipositas-Chirurgie: Die Nachsorge wird zum Tsunami. Dtsch Arztebl. 2018;115:45.
Lee B, Ibrahim SA, Zhang T. Mobile apps leveraged in the COVID-19 pandemic in East and South-East Asia: review and content analysis. JMIR MHealth UHealth. 2021;9:e32093.
Smith RB, et al. Mindfulness effects in obstetric and gynecology patients during the Coronavirus Disease 2019 (COVID-19) pandemic: a randomized controlled trial. Obstet Gynecol. 2021;137:1032–40.
Özkan Şat S, YamanSözbir Ş. Use of mobile applications by pregnant women and levels of pregnancy distress during the COVID-19 (Coronavirus) pandemic. Matern Child Health J. 2021;25:1057–68.
Doraiswamy S, Abraham A, Mamtani R, Cheema S. Use of Telehealth during the COVID-19 pandemic: scoping review. J Med Internet Res. 2020;22:e24087.
Hincapié MA, et al. Implementation and usefulness of Telemedicine during the COVID-19 pandemic: a scoping review. J Prim Care Community Health. 2020;11:2150132720980612.
Mundi MS, Lorentz PA, Grothe K, Kellogg TA, Collazo-Clavell ML. Feasibility of smartphone-based education modules and ecological momentary assessment/intervention in pre-bariatric surgery patients. Obes Surg. 2015;25:1875–81.
Sysko R, Michaelides A, Costello K, Herron DM, Hildebrandt T. An initial test of the efficacy of a digital health intervention for bariatric surgery candidates. Obes Surg. 2022. https://doi.org/10.1007/s11695-022-06258-8.
Yang C, et al. Smartphone application-based follow-up care of patients after bariatric surgery: a mixed-method study of usability. Digit Health. 2022;8:20552076221129070.
Clavien PA, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250:187–96.
van de Laar A. Bariatric Outcomes Longitudinal Database (BOLD) suggests excess weight loss and excess BMI loss to be inappropriate outcome measures, demonstrating better alternatives. Obes Surg. 2012;22:1843–7.
Dallal RM, Quebbemann BB, Hunt LH, Braitman LE. Analysis of weight loss after bariatric surgery using mixed-effects linear modeling. Obes Surg. 2009;19:732–7.
van de Laar A, de Caluwé L, Dillemans B. Relative outcome measures for bariatric surgery. Evidence against excess weight loss and excess body mass index loss from a series of laparoscopic Roux-en-Y gastric bypass patients. Obes Surg. 2011;21:763–7.
Weiner S, et al. Validation of the adapted Bariatric Quality of Life Index (BQL) in a prospective study in 446 bariatric patients as one-factor model. Obes Facts. 2009;2(Suppl 1):63–6.
Weiner S, et al. The Bariatric Quality of Life index: a measure of well-being in obesity surgery patients. Obes Surg. 2005;15:538–45.
Walter-Kroker A, Kroker A, Mattiucci-Guehlke M, Glaab T. A practical guide to bioelectrical impedance analysis using the example of chronic obstructive pulmonary disease. Nutr J. 2011;10:35.
Statista. Smartphone ownership in the US by age 2021. Downloaded from: https://www.statista.com/statistics/489255/percentage-of-us-smartphone-owners-by-age-group/. Accessed 4 Apr 2023.
Statista. Mobile phone daily usage time in U.S.2023. Downloaded from: https://www.statista.com/statistics/1045353/mobile-device-daily-usage-time-in-the-us/. Accessed 4 Apr 2023.
Lujan J, et al. Impact of routine and long-term follow-up on weight loss after bariatric surgery. Obes Surg. 2020;30:4293–9.
Spaniolas K, Kasten KR, Celio A, Burruss MB, Pories WJ. Postoperative follow-up after bariatric surgery: effect on weight loss. Obes Surg. 2016;26:900–3.
Mitchell JE, et al. Psychopathology before surgery in the longitudinal assessment of bariatric surgery-3 (LABS-3) psychosocial study. Surg Obes Relat Dis Off J Am Soc Bariatr Surg. 2012;8:533–41.
Calcaterra V, et al. Telehealth: a useful tool for the management of nutrition and exercise programs in pediatric obesity in the COVID-19 era. Nutrients. 2021;13:3689.
Gonzalez-Plaza E, et al. Effectiveness of a step counter smartband and midwife counseling intervention on gestational weight gain and physical activity in pregnant women with obesity (Pas and Pes Study): randomized controlled trial. JMIR MHealth UHealth. 2022;10:e28886.
Bennell KL, et al. Comparing video-based, telehealth-delivered exercise and weight loss programs with online education on outcomes of knee osteoarthritis : A Randomized Trial. Ann Intern Med. 2022;175:198–209.
Falkenhain K, et al. Use of an mHealth ketogenic diet app intervention and user behaviors associated with weight loss in adults with overweight or obesity: secondary analysis of a randomized clinical trial. JMIR MHealth UHealth. 2022;10:e33940.
Martínez MC, et al. The impact of bariatric surgery on the muscle mass in patients with obesity: 2-year follow-up. Obes Surg. 2022;32:625–33.
de Oliveira PAP, Montenegro ACP, Bezerra LRA, da Conceição Chaves de Lemos M, Bandeira F. Body composition, serum sclerostin and physical function after bariatric surgery: performance of dual-energy X-ray absorptiometry and multifrequency bioelectrical impedance analysis. Obes Surg. 2020;30:2957–62.
Gero D, et al. Defining global benchmarks in bariatric surgery: a retrospective multicenter analysis of minimally invasive Roux-en-Y gastric bypass and sleeve gastrectomy. Ann Surg. 2019;270:859–67.
Gasmi A, et al. Micronutrients deficiences in patients after bariatric surgery. Eur J Nutr. 2022;61:55–67.
Chakhtoura MT, Nakhoul NN, Shawwa K, Mantzoros C, El Hajj Fuleihan GA. Hypovitaminosis D in bariatric surgery: a systematic review of observational studies. Metabolism. 2016;65:574–85.
Shah M, et al. Hypocalcemia after bariatric surgery: prevalence and associated risk factors. Obes Surg. 2017;27:2905–11.
Thereaux J, et al. Long-term follow-up after bariatric surgery in a national cohort. Br J Surg. 2017;104:1362–71.
Acknowledgements
We would like to thank Paperpal (https://paperpal.com) for the English language review.
Funding
Open Access funding enabled and organized by Projekt DEAL. This project was supported by the Center for Innovative Care grant by the Baden-Wuerttemberg Ministry of Science, Research and Art (42-04HV.MED(18)/25/1). The app was provided free of charge by Oncare GmbH (Munich, Germany).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval
All procedures of the study are ethically compliant and were approved (EK #2018-643N-MA) by the Institutional Review Board of the University Medicine Mannheim.
Informed Consent
Written informed consent was obtained from all study participants.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key Points
1. A fully remote follow-up with a smartphone app is at least as effective as the traditional follow-up in an outpatient clinic concerning weight loss and the prevalence of complications after 12 months.
2. A fully remote follow-up program can help patients save time and caregivers save resources for patients with more severe problems.
3. Some patients need more in-person contact, especially when technical problems with the app occur.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yang, C., Kessler, M., Taebi, N. et al. Remote Follow-up with a Mobile Application Is Equal to Traditional Outpatient Follow-up After Bariatric Surgery: the BELLA Pilot Trial. OBES SURG 33, 1702–1709 (2023). https://doi.org/10.1007/s11695-023-06587-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-023-06587-2