Abstract
Sparse information is available on pharmacokinetic changes of drugs over time after bariatric surgery. By reviewing the literature on the short- and long-term pharmacokinetic changes of drugs, several patterns were identified for 39 drugs. No relevant pharmacokinetic changes were identified for roughly a third of the drugs. Of the remaining drugs, levels were variable and partly unpredictable shortly after the surgery. In the long term, most of the drug levels remain altered, but in some cases they returned to preoperative values. Based on the changes and the efficacy-safety balance of each drug, clinicians may need to perform additional clinical monitoring for specific drugs, including measuring drug levels. This review provides suggestions for clinicians and pharmacists for specific time-dependent drug dosing advice.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
With the increasing number of patients undergoing bariatric surgery, there is a growing awareness of potential problems that can occur after bariatric surgery. Nutrient insufficiencies due to malabsorption are a common and known complication [1]. Less knowledge is available on the possibly altered pharmacokinetics of drugs due to malabsorption and other physiological changes. This may have challenging consequences for drug dosing after bariatric surgery [2].
To date, some reports suggest that pharmacokinetic alterations after bariatric surgery are time-dependent. In 2012, Hamad et al. [3] demonstrated that the plasma concentrations of sertraline and citalopram were initially decreased 1 month after a Roux-en-Y gastric bypass (RYGB). These levels returned to preoperative values in most patients at 6 months, and they sometimes even exceeded preoperative values at 1 year after the surgery. Steele et al. [4] described apixaban levels to increase at 1 month after surgery, to return to preoperative levels at 6 months and subsequently to decrease after 1 year. Thus far, changes in pharmacokinetics after bariatric surgery have mainly been described in studies with relatively limited numbers of patients. Moreover, pharmacokinetics was often assessed at a specific single time point or at a random time point regardless of the time after surgery (e.g., 1 to 9 years). Thus, the available data do not allow to distinguish the differences between the short- and long-term pharmacokinetics after bariatric surgery.
Dosing advice after bariatric surgery is greatly needed. Therefore, the European Association for the Study of Obesity has provided general dosing advice [5]. In the Netherlands, more specific dosing advice for patients after bariatric surgery has been incorporated in the Dutch national drug database. However, these current recommendations for dosing after surgery do not consider the influence of time after surgery. Such information could help clinicians and pharmacists in the treatment and follow-up of patients with bariatric surgery.
The aim of this review was to summarize the current knowledge regarding time-dependent changes in pharmacokinetics of drugs after bariatric surgery and to provide recommendations for dosing. Before presenting the methods and results, some background information on the 2 main types of bariatric surgery with associated physiological changes is provided.
Bariatric Surgical Procedures and Associated Physiological Changes
After bariatric surgery, several physiological changes take place in the short term, i.e., up to 1 month after surgery, and in the long term, i.e., more than 1 month after surgery. This review focuses on the 2 most performed bariatric surgical techniques, the Roux-en-Y gastric bypass (RYGB) and the sleeve gastrectomy (SG) or gastric sleeve. The latter is solely a restrictive procedure, in which around 75% of the stomach is excised [6]. By contrast, RYGB is a malabsorptive-restrictive procedure, in which a small gastric pouch of around 30 ml is anastomosed to the distal limb of a jejunotomy performed at the mid-jejunum conform a Roux-en-Y construction [6]. This review did not include studies on gastric banding or biliopancreatic diversion (BPD)/duodenal switch. These techniques have lost popularity due to ineffectiveness and band-related complications in the case of gastric banding and nutritional complications in the case of BPD/duodenal switch. In recent years, other bariatric surgery procedures have been developed, such as one-anastomosis gastric bypass (OAGB), single anastomosis stomach-ileal bypass (SASI) or single anastomosis duodeno ileal-bypass (SADI). While these newer techniques are beyond the scope of the present review, they are expected to show similar pharmacokinetic alterations as RYGB.
Gastrointestinal Tract
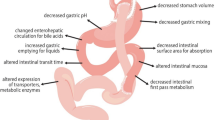
After both RYGB and SG, the scintigraphy data showed a reduction of gastric emptying time, both with and without food [7]. This reduction was observed for solids, semi-solids and liquids, both in the short term and in the long term up to 2 years [7].
In SG patients, the pH in the gastric pouch depends on the time after surgery [8]. Within a day after SG, the gastric pH rapidly increased from 1.7 to 5.0 [9]. One year after SG, the acid exposure returned to preoperative values [8]. In contrast to SG patients, the pH in RYGB patients remains high both in the short and long term after surgery [10]. In these RYGB patients, the median pH of the entire oro-cecal segment is increased from 5.5 to 7.0 [11].
Compared to the preoperative period, total bile acid concentrations in patients have been reported to increase after RYGB in a time-dependent manner over 5 years follow-up [12]. This is reflected in the increased risk of gallstone formation (cholelithiasis) in this group [13]. The inlet of bile and pancreatic fluid is delayed in RYGB patients [14].
Liver
Drug-metabolizing cytochrome P450 (CYP) enzymes have been quantified in liver and intestinal biopsies of patients with RYGB on the day of surgery [15, 16]. However, no systematic information is available on CYP enzymes from biopsies after bariatric surgery.
Based on MRI data, the liver volume was decreased by 20% 3 months after bariatric surgery [17]. Subsequently, the liver volume remained relatively constant at 6, 12 and 24 months postoperatively at a decrease of 17 to 27% compared to the preoperative status. At 3 months after bariatric surgery, cardiac output was reduced by 20% and remained stable at this value up to the end of follow-up, which was 1 year after surgery [18]. The liver blood flow remains a constant percentage relative to cardiac output across BMI values in patients with obesity before surgery [19]. Therefore, the liver flow can be assumed to be approximately 20% lower after bariatric surgery.
Kidney
After bariatric surgery, renal function may change over time. The extent of change seems to be dependent on the initial renal function. In patients with preoperative hyperfiltration, the glomerular filtration rate (GFR), as determined by iohexol clearance, normalizes at 1 year after surgery [20]. However, in patients with pre-existent renal impairment, GFR seems to remain unchanged [21]. The GFR in patients with a normal kidney function remain similar on the short term and is reported to decrease at 1 year after RYGB [22].
The assessment of the renal function before and after bariatric surgery can be challenging when using a serum creatinine-based estimate of the renal function [20]. The reliability of renal function estimates is affected directly by weight loss because of the decrease in muscle mass. When using an indexed estimated GFR, i.e., normalized estimated GFR to a body surface area of 1.73 m2, this can lead to underestimation in individuals with obesity [20]. It has been suggested that it might be better to monitor the renal function after bariatric surgery with a method based on an exogenous marker such as iohexol [20, 23]. Alternatively, the estimation of renal function can be improved by de-indexing the estimated GFR in patients with class III obesity [24]. However, de-indexed estimated GFR may slightly overestimate the renal function in patients after bariatric surgery regardless of whether they are still obese [25].
In clinical practice, changes over time in renal function in bariatric patients could be blurred if renal function is estimated based on serum creatinine. The values after surgery may not be directly comparable with preoperative values. However, the debate on how to assess and interpret renal function after bariatric surgery is still ongoing.
Methods
Systematic searches in PubMed and Embase were performed using the following MeSH terms: “bariatrics”, “gastric bypass”, “sleeve gastrectomy”, “pharmacokinetics” and “time”. Furthermore, studies were also included if they contained the non-MeSH terms “drug concentration” or “drug level” and one of the following terms: “time after surgery”, “change*”, “long-term” or “short-term”. The search strategy can be found in the appendix. Studies were excluded if they were written in other languages than English, Dutch, German or French or if they included pediatric patients.
We included results from studies on drugs of which the pharmacokinetics were assessed on at least 2 different occasions after bariatric surgery. Pertinent citations from retrieved articles were also included. If available, the reported postoperative drug levels were compared to preoperative levels. Generally, if the area under the concentration-time curve (AUC) or trough level was between 80 and 120% of the preoperative values, the difference was considered to be non-relevant. If no preoperative values were available, the reported postoperative drug levels were compared to those in patients with obesity. Results on drugs for which data of less than 5 patients in total were available were described separately.
Results
Of the 2533 retrieved results, 276 articles were selected for screening after removing duplicates and reading title and abstract. After exclusion of study proposals, cases of gastric banding, BPD and total gastrectomy only, and studies discussing vitamin and mineral suppletion and ethanol, 224 results were reviewed (Fig. 1). In total, 56 articles described the pharmacokinetics of drugs at 2 or more time points after surgery. Of these 56 included articles, 22 investigated the effects of bariatric surgery on pharmacokinetics of drugs up to 6 months after bariatric surgery (Fig. 2a). The numbers of drugs studied beyond 1 year after bariatric surgery are shown in Fig. 2b.
Numbers of studied drugs of which the pharmacokinetics were assessed at multiple time points up to 1 year after bariatric surgery (A) and from 1 to 5 years after surgery (B). The Y-axis depicts the number of studied drugs. The size of the dots indicates the total number of patients that were studied
Time-dependent effects on pharmacokinetic changes in patients with bariatric surgery were reported for 39 different drugs. The findings of 32 drugs for which data of ≥ 5 patients in total were available, are summarized in Table 1. The drugs are sorted by therapeutic area. A practical advice for clinicians is provided. Table 1 shows results of drugs for which altered pharmacokinetic was reported, while Table 2 shows drugs for which no pharmacokinetic changes were found. Since no relevant differences in pharmacokinetics between RYGB and SG were identified, no separate practical advice was provided for either type of surgery.
Based on the available data, various patterns were identified in altered pharmacokinetics in the short and long term. First, no pharmacokinetic changes after bariatric surgery were reported for 6 drugs: venlafaxine, ciprofloxacin, rivaroxaban (only prophylactic dose reported), desogestrel, ethinylestradiol and dextromethorphan. In addition, the pharmacokinetic changes over time were considered not to be clinically relevant for digoxin, omeprazole and levonorgestrel.
As a second group, we identified drugs of which the pharmacokinetics are significantly altered in both the short and the long term: lithium, tacrolimus, carbamazepine, duloxetine, midazolam, morphine, paracetamol, hydrocortisone, levothyroxine, rosuvastatin and metformin. Lithium levels can increase shortly after surgery [26,27,28,29, 79], which is attributed either to improved absorption of lithium or to a lower fluid intake resulting in a lower drug clearance. Consequently, severe lithium intoxications have been reported [26,27,28]. Most patients could be stabilized and discharged with a lower dose [26, 28, 79]. An a priori reduction of 30–70% in lithium dose is recommended in the first week after bariatric surgery, combined with monitoring of lithium levels (Therapeutic Drug Monitoring, TDM).
For other drugs in this group, such as tacrolimus and carbamazepine, intensive monitoring of drug levels shortly after surgery seems necessary. For duloxetine, reduced efficacy may be anticipated due to lower levels in both the short and the long term after bariatric surgery. In case of reduced efficacy, measuring duloxetine levels and increasing the dosage may be required. Studies on midazolam, morphine and paracetamol showed higher and earlier peak concentrations after surgery [16, 32, 64, 65]. This could potentially cause more adverse events at the time of maximal concentration (Tmax), while the clinical effects could fade off earlier. Hydrocortisone, levothyroxine, rosuvastatin, metformin showed various effects, as studies reported both increased and decreased levels over time. For these drugs, it is important to periodically monitor the effects through laboratory values and adjust the dose accordingly.
As a third class, we identified drugs that showed increased levels shortly after surgery and decreased values in the long term post-surgery. Among these drugs are pregabalin, lamotrigine, atorvastatin, simvastatin, warfarin, apixaban and amoxicillin. Additional clinical monitoring of effects and adverse events is recommended for pregabalin and lamotrigine. In case of epilepsy patients, measuring drug concentrations and adjusting doses accordingly can be helpful to prevent the occurrence of seizures. Atorvastatin, simvastatin and warfarin can be dosed according to their effects. Frequent LDL and INR measuring in the first few months after surgery can help to titrate the correct dose. Apixaban is a high-risk drug. Overexposure increases the risk of bleeding, while underexposure increases the risk of thromboembolic events. Hence, prescribers are advised to switch to drugs that can be monitored more easily, such as vitamin K antagonists.
Amoxicillin was shown to have initially increased exposure, followed by decreased exposure later on [39,40,41]. However, the results of these studies may have been influenced by differences in the formulation and dosages used, together with the known non-linear absorption of amoxicillin in higher doses [80]. Since the amoxicillin levels in these pharmacokinetics studies were all above the target minimum inhibitory value of 4 mg/L, no dose adjustments are advised for amoxicillin after bariatric surgery.
As a last category, the following drugs first showed lower drug levels, followed by higher drug concentrations that approached preoperative levels: sertraline, mirtazapine, phenytoin and probably citalopram/escitalopram. For these drugs, it seems important to monitor clinical effects in the short term. If altered effects are suspected, it is advisable to measure drug levels, i.e., to perform TDM, in addition to clinical monitoring. For epilepsy patients, it may be advisable to consider measuring phenytoin concentrations and adjusting doses accordingly.
For 7 drugs, data from less than 5 patients per drug was available (Table 3). Early after surgery, buprenorphine was absorbed faster, which normalized over time with the exposure decreasing by 40%. The plasma concentrations of tricyclic antidepressants seem to decrease over time. In contrast, the pharmacokinetics of gabapentin, topiramate and efavirenz did not alter over time.
Discussion
In this review, we describe results regarding pharmacokinetic changes over time for 39 drugs. Based on the findings of 56 included studies, we recommend that clinicians and pharmacists be aware of the pharmacokinetic changes that may occur over time. To support optimal therapy after bariatric surgery, more frequent clinical monitoring (i.e., drug counselling and outpatient follow-up) may be considered. Measuring specific drug levels in the first year after surgery could be helpful. Since some pharmacokinetic changes can still occur within the first year, further clinical monitoring may be required to ensure efficacy and safety in the longer term. For some drugs, no alterations in pharmacokinetics were identified. As such, in contrast to earlier reports in literature, there is no evidence to discourage the use of extended-release preparations (e.g., venlafaxine and morphine) after bariatric surgery [67, 74].
After bariatric surgery, patients may be more prone to adverse effects due to shorter gastric emptying time. Higher absorption rates and, consequently, higher and earlier peak concentrations (Cmax) could cause a quicker drug onset, which may be of relevance for sedatives or opioids such as midazolam and morphine. Thus, a change in the drug formulation, for example converting tablets into liquid, can have dramatic consequences, as was shown by a reported lithium intoxication [28]. Over time, the Tmax of most drugs becomes shorter, while the Cmax may vary over time: the concentrations either remain higher or return to preoperative values after several months.
Besides absorption-related differences due to intestinal adaptation, time-dependent pharmacokinetic changes can also be associated with reversal of obesity. Therefore, significant changes attributed to weight loss occur more gradually after bariatric surgery. One of these changes involves CYP3A activity, which is reduced in patients with obesity [83]. Assuming all other factors to remain similar after bariatric surgery, CYP3A activity normalizes over time, until it resembles the activity in patients without obesity [32]. However, due to changes in other physiological parameters, such as liver flow, this normalization may not necessarily translate into higher clearance of drugs that are hepatically metabolized by CYP3A [32].
Recent studies have shown that the pharmacokinetics of various contraceptives (i.e., desogestrel, ethinylestradiol and levonorgestrel) seem unaltered after RYGB [69, 78]. This finding is not in line with current advice that strongly discourages the use of certain drugs after bariatric surgery, such as oral contraceptives. This advice was based on a case series of 2 women who became pregnant after BPD despite the use of oral contraceptives [84]. A relevant culprit might have been the occurrence of chronic diarrhea in these women. Based on the recent studies, the absolute contraindication of any bariatric surgery for several oral contraceptives cannot be supported by pharmacokinetic data.
It should be noted that we did not a priori exclude case series, which can be considered a strength of our narrative review. In a systematic review, the extensive and very informative case reports of lithium intoxications would have been missed due to a low evidence level. Based on the scarce available evidence for most of the included drugs, we nevertheless tried to propose practical advice for clinicians. As for any review, the strength of this review is related to the strength of the reviewed papers. We realize that in the present review, the evidence was not graded and bias was not assessed in a systematic way, as was done by McLachlan [85]. Nor did we analyze the pooled data based on drug properties (e.g., lipophilicity, renal excretion, or involvement of CYP metabolism).
Clinicians may question how to dose patients who regain weight after bariatric surgery. A regain of weight of 10% or more occurs in 17.6% of the patients after bariatric surgery [86]. However, patients with weight regain were not specifically described in the included studies. That is why we could not assess whether patients with weight regain after bariatric surgery need different dosing advice over time.
Another question that remains to be answered is whether pharmacokinetics can be affected by differences in the practice of bariatric surgery. RYGB procedures differ between surgeons and clinics throughout the world in terms of the lengths of both the alimentary limb and the biliary limb, and therefore also in terms of the common channel. As far as we know, the effect of these differences on the pharmacokinetics of drugs have not been studied. However, as the current medication advice does not differ between RYGB and SG, we hypothesize that the differences in surgical practice may be of little clinical relevance.
Lastly, there is growing interest in microbiota changes after bariatric surgery [87, 88]. Obesity and weight loss seem to be associated with compositional changes in gut microbiota. More interestingly, the effect of weight loss on microbiota diversity seems to be time-dependent [87]. As microbiota can modulate both the absorption and the metabolism of drugs, they may constitute a missing link in the assessment of pharmacokinetic changes after bariatric surgery. To date, however, this is a highly understudied area.
This review highlights the importance of time-dependent dosing of drugs after bariatric surgery. In this complex matter, clinicians and pharmacists can collaborate in multidisciplinary teams to deliver the best care. This collaboration may involve additional or more intensive monitoring of clinical effects, performing TDM and adjusting doses. It should be acknowledged that information on the effects of bariatric surgery on the pharmacokinetics from 1 year and further on are very scarce. Thus, more research is needed to determine the optimal dosing at the time post-bariatric surgery, especially for drugs with a narrow therapeutic window.
Conclusions
Pharmacokinetic changes after bariatric surgery can be time-dependent, leading to different changes in the short and long term after surgery. Due to the variability in the magnitude and directions of the changes in plasma concentrations over time, no general dosing advice can be provided. Besides, considering that the pharmacokinetic profiles of drugs may differ over time, it is recommended to monitor the clinical effects of certain drugs and, if needed, to measure drug levels and adjust drug doses accordingly. Consequently, close cooperation between clinicians and pharmacists is required to optimize drug doses for the intended clinical effect in post-bariatric patients.
References
Bal BS, Finelli FC, Shope TR, et al. Nutritional deficiencies after bariatric surgery. Nat Rev Endocrinol. 2012;8(9):544–56.
Kingma JS, Burgers DMT, Monpellier VM, et al. Oral drug dosing following bariatric surgery: General concepts and specific dosing advice. Br J Clin Pharmacol. 2021;87(12):4560–76.
Hamad GG, Helsel JC, Perel JM, et al. The effect of gastric bypass on the pharmacokinetics of serotonin reuptake inhibitors. Am J Psychiatry. 2012;169(3):256–63.
Steele KE, Prokopowicz GP, Canner JP, et al. The APB study: apixaban pharmacokinetics in bariatric patients before to 1 year after vertical sleeve gastrectomy or Roux-en-Y gastric bypass. Surg Obes Relat Dis. 2022;18(5):594–603.
Busetto L, Dicker D, Azran C, et al. Obesity management task force of the European association for the study of obesity released “practical recommendations for the post-bariatric surgery medical management.” Obes Surg. 2018;28(7):2117–21.
Al-Najim W, Docherty NG, le Roux CW. Food intake and eating behavior after bariatric surgery. Physiol Rev. 2018;98(3):1113–41.
Steenackers N, Vanuytsel T, Augustijns P, et al. Adaptations in gastrointestinal physiology after sleeve gastrectomy and Roux-en-Y gastric bypass. Lancet Gastroenterol Hepatol. 2021;6(3):225–37.
Porat D, Vaynshtein J, Gibori R, et al. Stomach pH before vs. after different bariatric surgery procedures: Clinical implications for drug delivery. Eur J Pharm Biopharm. 2021;160:152–7.
Tolone S, Savarino E, de Bortoli N, et al. Esophageal high-resolution manometry can unravel the mechanisms by which different bariatric techniques produce different reflux exposures. J Gastrointest Surg. 2020;24(1):1–7.
Jaruvongvanich V, Matar R, Ravi K, et al. Esophageal pathophysiologic changes and adenocarcinoma after bariatric surgery: A systematic review and meta-analysis. Clin Transl Gastroenterol. 2020;11(8):e00225.
Ladebo L, Pedersen PV, Pacyk GJ, et al. Gastrointestinal pH, motility patterns, and transit times after Roux-en-Y gastric bypass. Obes Surg. 2021;31(6):2632–40.
Risstad H, Kristinsson JA, Fagerland MW, et al. Bile acid profiles over 5 years after gastric bypass and duodenal switch: results from a randomized clinical trial. Surg Obes Relat Dis. 2017;13(9):1544–53.
Wan Q, Zhao R, Chen Y, et al. Comparison of the incidence of cholelithiasis after sleeve gastrectomy and Roux-en-Y gastric bypass: a meta-analysis. Surg Obes Relat Dis. 2021;17(6):1198–205.
Hachon L, Declèves X, Faucher P, et al. RYGB and drug disposition: How to do better? analysis of pharmacokinetic studies and recommendations for clinical practice. Obes Surg. 2017;27(4):1076–90.
Lloret-Linares C, Daali Y, Abbara C, et al. CYP450 activities before and after Roux-en-Y gastric bypass: correlation with their intestinal and liver content. Surg Obes Relat Dis. 2019;15(8):1299–310.
Kvitne KE, Robertsen I, Skovlund E, et al. Short- and long-term effects of body weight loss following calorie restriction and gastric bypass on CYP3A-activity - a non-randomized three-armed controlled trial. Clin Transl Sci. 2022;15(1):221–33.
Meyer-Gerspach AC, Peterli R, Moor M, et al. Quantification of liver, subcutaneous, and visceral adipose tissues by mri before and after bariatric surgery. Obes Surg. 2019;29(9):2795–805.
de Witte D, Wijngaarden LH, van Houten VAA, et al. Improvement of cardiac function after Roux-en-Y gastric bypass in morbidly obese patients without cardiac history measured by cardiac MRI. Obes Surg. 2020;30(7):2475–81.
Berton M, Bettonte S, Stader F, et al. Repository describing the anatomical, physiological, and biological changes in an obese population to inform physiologically based pharmacokinetic models. Clin Pharmacokinet. 2022;61(9):1251–70.
Favre G, Schiavo L, Lemoine S, et al. Longitudinal assessment of renal function in native kidney after bariatric surgery. Surg Obes Relat Dis. 2018;14(9):1411–8.
Navaneethan SD, Malin SK, Arrigain S, et al. Bariatric surgery, kidney function, insulin resistance, and adipokines in patients with decreased GFR: a cohort study. Am J Kidney Dis. 2015;65(2):345–7.
Friedman AN, Considine RV, Quinney SK. Inquiry into the short- and long-term effects of Roux-en-Y gastric bypass on the glomerular filtration rate. Ren Fail. 2020;42(1):624–8.
Grangeon-Chapon C, Laurain A, Esnault VLM, et al. Extracellular fluid volume: A suitable indexation variable to assess impact of bariatric surgery on glomerular filtration rate in patients with chronic kidney disease. PLoS ONE. 2021;16(8):e0256234.
Donker E, Bet P, Nurmohamed A, et al. Estimation of glomerular filtration rate for drug dosing in patients with very high or low body mass index (BMI). Clin Transl Sci. 2022;15(9):2206–17.
Chang AR, George J, Levey AS, et al. Performance of glomerular filtration rate estimating equations before and after bariatric surgery. Kidney Med. 2020;2(6):699-706.e1.
Dahan A, Porat D, Azran C, et al. Lithium toxicity with severe bradycardia post sleeve gastrectomy: A case report and review of the literature. Obes Surg. 2019;29(2):735–8.
Niessen R, Sottiaux T, Schillaci A, et al. Lithium toxicity after bariatric surgery. Rev Med Liege. 2018;73(2):82–7.
Musfeldt D, Levinson A, Nykiel J, et al. Lithium toxicity after Roux-en-Y bariatric surgery. BMJ Case Rep. 2016;2016:bcr2015214056.
Altieri M, Pryor A, Vosswinkel J, et al. Lithium induced thyroid storm after sleeve gastrectomy. Crit Care Med. 2015;43(12):316.
Wallerstedt SM, Nylén K, Axelsson MAB. Serum concentrations of antidepressants, antipsychotics, and antiepileptics over the bariatric surgery procedure. Eur J Clin Pharmacol. 2021;77(12):1875–85.
Marzinke MA, Petrides AK, Steele K, et al. Decreased escitalopram concentrations post-roux-en-Y gastric bypass surgery. Ther Drug Monit. 2015;37(3):408–12.
Brill MJ, van Rongen A, Dongen EP, et al. The pharmacokinetics of the CYP3A substrate midazolam in morbidly obese patients before and one year after bariatric surgery. Pharm Res. 2015;32(12):3927–36.
Tandra S, Chalasani N, Jones DR, et al. Pharmacokinetic and pharmacodynamic alterations in the Roux-en-Y gastric bypass recipients. Ann Surg. 2013;258(2):262–9.
Chan LN, Lin YS, Tay-Sontheimer JC, et al. Proximal Roux-en-Y gastric bypass alters drug absorption pattern but not systemic exposure of CYP3A4 and P-glycoprotein substrates. Pharmacotherapy. 2015;35(4):361–9.
Triplett JD, Simpson HD, Clemmons RS, et al. The effect of weight reduction surgery on the efficacy and tolerability of epilepsy pharmacotherapy. Epilepsy Behav. 2021;124:108307.
Koutsavlis I, Lasebai M. Dose-dependent carbamazepine-induced agranulocytosis following bariatric surgery (sleeve gastrectomy): A possible mechanism. Bariatr Surg Pract Patient Care. 2015;10(3):130–4.
Brown CS, Rabinstein AA, Nystrom EM, et al. Antiseizure medication use in gastric bypass patients and other post-surgical malabsorptive states. Epilepsy Behav Rep. 2021;16:100439.
Pournaras DJ, Footitt D, Mahon D, et al. Reduced Phenytoin levels in an epileptic patient following Roux-En-Y gastric bypass for obesity. Obes Surg. 2011;21(5):684–5.
Rocha MBS, De Nucci G, Lemos FN, et al. Impact of bariatric surgery on the pharmacokinetics parameters of amoxicillin. Obes Surg. 2019;29(3):917–27.
Montanha MC, Diniz A, Silva NMEN, et al. Physiologically-based pharmacokinetic model on the oral drug absorption in Roux-en-Y gastric bypass bariatric patients: amoxicillin tablet and suspension. Mol Pharm. 2019;16(12):5025–34.
Montanha MC, dos Santos Magon TF, de Souza AC, et al. Reduced bioavailability of oral amoxicillin tablets compared to suspensions in Roux-en-Y gastric bypass bariatric subjects. Br J Clin Pharmacol. 2019;85(9):2118–25.
Jakobsen GS, Skottheim IB, Sandbu R, et al. Long-term effects of gastric bypass and duodenal switch on systemic exposure of atorvastatin. Surg Endosc. 2013;27(6):2094–101.
El-Zailik A, Cheung LK, Wang Y, et al. Longitudinal impacts of gastric bypass surgery on pharmacodynamics and pharmacokinetics of statins. Obes Surg. 2019;29(8):2571–83.
Irwin AN, McCool KH, Delate T, et al. Assessment of warfarin dosing requirements after bariatric surgery in patients requiring long-term warfarin therapy. Pharmacotherapy. 2013;33(11):1175–83.
Strong A, Sharma G, Hanipah ZN, et al. Adjustments to warfarin dosing following gastric bypass and sleeve gastrectomy. Surg Obes Relat Dis. 2017;13(10):S136-S.
Mallappa A, Nella AA, Kumar P, et al. Alterations in hydrocortisone pharmacokinetics in a patient with congenital adrenal hyperplasia following bariatric surgery. J Endocr Soc. 2017;1(7):994–1001.
de Heide LJM, de Boer HHR, van Borren M, et al. Pharmacokinetics of glucocorticoid replacement before and after bariatric surgery in patients with adrenal insufficiency. J Endocr Soc. 2018;2(12):1338–44.
Gadiraju S, Lee CJ, Cooper DS. Levothyroxine dosing following bariatric surgery. Obese Surg. 2016;26:2538–42.
Pirola I, Formenti AM, Gandossi E, et al. Oral liquid L-thyroxine (L-t4) may be better absorbed compared to L-T4 tablets following bariatric surgery. Obes Surg. 2013;23(9):1493–6.
Aggarwal S, Modi S, Jose T. Laparoscopic sleeve gastrectomy leads to reduction in thyroxine requirement in morbidly obese patients with hypothyroidism. World J Surg. 2014;38(10):2628–31.
Sundaram U, McBride C, Shostrom V, et al. Prevalence of preoperative hypothyroidism in bariatric surgery patients and postoperative change in thyroid hormone requirements. Bariatr Surg Pract Patient Care. 2013;8(4):147–51.
Fierabracci P, Martinelli S, Tamberi A, et al. Weight loss and variation of levothyroxine requirements in hypothyroid obese patients after bariatric surgery. Thyroid. 2016;26(4):499–503.
Raftopoulos Y, Gagné DJ, Papasavas P, et al. Improvement of hypothyroidism after laparoscopic Roux-en-Y gastric bypass for morbid obesity. Obes Surg. 2004;14(4):509–13.
Gkotsina M, Michalaki M, Mamali I, et al. Improved levothyroxine pharmacokinetics after bariatric surgery. Thyroid. 2013;23(4):414–9.
Rubio IG, Galrão AL, Santo MA, et al. Levothyroxine absorption in morbidly obese patients before and after Roux-En-Y gastric bypass (RYGB) surgery. Obes Surg. 2012;22(2):253–8.
Richou M, Gilly O, Taillard V, et al. Levothyroxine dose adjustment in hypothyroid patients following gastric sleeve surgery. Annales d’endocrinologie. 2020;81(5):500–6.
Yska JP, Arfman IJ, van Oijen MA, et al. Long-term effect of bariatric surgery on the use of levothyroxine and thyroid levels. Obes Surg. 2022;32(3):742–8.
Padwal RS, Gab RQ, Sharm AM, et al. Gastric bypass surgery increases the absorption and bioavailability of metformin. Can J Diabetes. 2011;35(2):150.
El-Zailik A, Cheung LK, Sherman V, et al. Population pharmacokinetic modeling of metformin in patients undergoing gastric bypass surgery. Clin Pharmacol Drug Dev. 2017;6:15–6.
Collares-Pelizaro RVA, Santos JS, Nonino CB, et al. Omeprazole absorption and fasting gastrinemia after Roux-en-Y gastric bypass. Obes Surg. 2017;27(9):2303–7.
Mitrov-Winkelmolen L, van Buul-Gast MW, Swank DJ, et al. The effect of Roux-en-Y gastric bypass surgery in morbidly obese patients on pharmacokinetics of (acetyl)salicylic acid and omeprazole: The ERY-PAO study. Obes Surg. 2016;26(9):2051–8.
Picado AL, Paterna ABR, Bruton RS, et al. Effect of Roux-en-Y gastric surgery on omeprazole pharmacokinetics. an obvious effect? Basic Clin Pharmacol Toxicol. 2018;123:32.
Goday Arno A, Farré M, Rodríguez-Morató J, et al. Pharmacokinetics in morbid obesity: Influence of two bariatric surgery techniques on paracetamol and caffeine metabolism. Obes Surg. 2017;27(12):3194–201.
Chen KF, Chan LN, Senn TD, et al. The impact of proximal Roux-en-Y gastric bypass surgery on acetaminophen absorption and metabolism. Pharmacotherapy. 2020;40(3):191–203.
Lloret-Linares C, Hirt D, Bardin C, et al. Effect of a Roux-en-Y gastric bypass on the pharmacokinetics of oral morphine using a population approach. Clin Pharmacokinet. 2014;53(10):919–30.
Lloret-Linares C, Luo H, Rouquette A, et al. The effect of morbid obesity on morphine glucuronidation. Pharmacol Res. 2017;118:64–70.
Hachon L, Reis R, Labat L, et al. A comparison of morphine and metabolites plasma levels following sustained release formulation of morphine administration in Roux-en-Y gastric bypass subjects versus matched nonsurgical control subjects: The OBEMO 2 study. Fundam Clin Pharmacol. 2017;31:46.
Ginstman C, Kopp Kallner H, Fagerberg-Silwer J, et al. Pharmacokinetics of oral Levonorgestrel in women after Roux-en-Y gastric bypass surgery and in BMI-matched controls. Obes Surg. 2020;30(6):2217–24.
Moreira de Brito C, de Melo ME, Mancini MC, et al. Pharmacokinetics of oral levonorgestrel and ethinylestradiol in women after Roux-en-Y gastric bypass surgery. Surg Obes Relat Dis. 2021;17(4):673–81.
Kindi FA, Chaaban A, Budruddin M, et al. Tacrolimus variability in renal transplant patient post gastric sleeve surgery over two years. Am J Kidney Dis. 2020;75(4):555.
Waldman G, Clark JE, Rogers CC, et al. Tacrolimus level variability in setting of gastric sleeve. J Heart Lung Transplant. 2021;40(4):S486–7.
Chan G, Hajjar R, Boutin L, et al. Prospective study of the changes in pharmacokinetics of immunosuppressive medications after laparoscopic sleeve gastrectomy. Am J Transplant. 2020;20(2):582–8.
Yemini R, Nesher E, Winkler J, et al. Bariatric surgery in solid organ transplant patients: Long-term follow-up results of outcome, safety, and effect on immunosuppression. Am J Transplant. 2018;18(11):2772–80.
Krieger CA, Cunningham JL, Reid JM, et al. Comparison of bioavailability of single-dose extended-release venlafaxine capsules in obese patients before and after gastric bypass surgery. Pharmacotherapy. 2017;37(11):1374–82.
Paterna ABR, Picado AL, Bruton RS, et al. Effect of Roux-En-y gastric surgery on ciprofloxacin pharmacokinetics: An obvious effect? Basic Clin Pharmacol Toxicol. 2018;123:19.
Kröll D, Stirnimann G, Vogt A, et al. Pharmacokinetics and pharmacodynamics of single doses of rivaroxaban in obese patients prior to and after bariatric surgery. Br J Clin Pharmacol. 2017;83(7):1466–75.
Kröll D, Stirnimann G, Borbely Y, et al. The effect of bariatric surgery on direct oral anticoagulant activity of rivaroxaban after bariatric surgery: The extension study. Obes Surg. 2018;28(1):S60–1.
Ginstman C, Frisk J, Carlsson B, et al. Plasma concentrations of etonogestrel in women using oral desogestrel before and after Roux-en-Y gastric bypass surgery: a pharmacokinetic study. BJOG: Int J Obstet Gynaecol. 2019;126(4):486–92.
Shah S, Bhattacharyya M, Addepalli R. Erratic lithium levels following vertical sleeve gastrectomy: A case report. Am J Geriatr Psychiatry. 2018;26(3):S130-S.
Paintaud G, Alván G, Dahl ML, et al. Nonlinearity of amoxicillin absorption kinetics in human. Eur J Clin Pharmacol. 1992;43(3):283–8.
Krabseth HM, Strømmen M, Spigset O, et al. Effect of sleeve gastrectomy on buprenorphine pharmacokinetics: A planned case observation. Clin Ther. 2020;42(11):2232–7.
Amouyal C, Buyse M, Lucas-Martini L, et al. Sleeve gastrectomy in morbidly obese hiv patients: Focus on anti-retroviral treatment absorption after surgery. Obes Surg. 2018;28(9):2886–93.
Smit C, De Hoogd S, Brüggemann RJM, et al. Obesity and drug pharmacology: a review of the influence of obesity on pharmacokinetic and pharmacodynamic parameters. Expert Opin Drug Metab Toxicol. 2018;14(3):275–85.
Gerrits EG, Ceulemans R, van Hee R, et al. Contraceptive treatment after biliopancreatic diversion needs consensus. Obes Surg. 2003;13(3):378–82.
McLachlan LA, Chaar BB, Um IS. Pharmacokinetic changes post–bariatric surgery: A scoping review. Obes Rev. 2020;21(5):e12988.
Athanasiadis DI, Martin A, Kapsampelis P, et al. Factors associated with weight regain post-bariatric surgery: a systematic review. Surg Endosc. 2021;35(8):4069–84.
Shen N, Caixàs A, Ahlers M, et al. Longitudinal changes of microbiome composition and microbial metabolomics after surgical weight loss in individuals with obesity. Surg Obes Relat Dis. 2019;15(8):1367–73.
Liu R, Hong J, Xu X, et al. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat Med. 2017;23(7):859–68.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical Approval Statement
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent Statement
Informed consent does not apply for this review.
Conflict of Interest
All the authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key points
• Of 39 drugs, differences in pharmacokinetics after bariatric surgery were absent for 9 drugs and considered not clinically relevant for 3 drugs.
• For 27 drugs, time after surgery altered the pharmacokinetics, which can be a result of both surgical alterations in the gastrointestinal tract and weight loss.
• There was a huge variability in the magnitude and directions of the changes in plasma concentrations over time.
• Medical doctors, bariatric surgeons and pharmacists should be aware of the time-dependent pharmacokinetic changes that can occur after bariatric surgery.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lau, C., van Kesteren, C., Smeenk, R. et al. Impact of Bariatric Surgery in the Short and Long Term: A Need for Time-Dependent Dosing of Drugs. OBES SURG 33, 3266–3302 (2023). https://doi.org/10.1007/s11695-023-06770-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-023-06770-5