Abstract
Agricultural intensification has caused a simplification of agricultural landscapes, accompanied by increasing field sizes and a reduction of non-crop habitats. To mitigate negative impacts of intensification, it is necessary to understand to what extent different non-crop habitats contribute to the maintenance of biodiversity in agroecosystems. Here, we compared the taxonomic diversity of three beetle families among four habitat types—wheat fields, grassy field margins, wildflower-sown areas under power poles, and permanent grassland fallows, in an agricultural landscape in western Germany. Carabidae were caught by pitfall trapping, Chrysomelidae and Curculionidae by suction sampling. We found surprisingly little variation among habitat types, though the rarefied species number tended to be higher in grassland fallows and field margins than under power poles and in wheat fields. Nevertheless, species assemblages differed substantially among habitat types. In Carabidae, grassland fallows were dominated by hygrophilous species with poor dispersal ability as opposed to all other habitat types being dominated by open landscape species with high dispersal ability. In Chrysomelidae and Curculionidae, power pole islands differed from the other habitat types with predominantly open landscape species, whereas wheat fields and grassland fallows were clearly dominated by eurytopic species. Our results thus highlight the need for a combination of different conservation measures for enhancing the functional diversity of beetle assemblages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The intensification of agricultural land use in recent decades has resulted in a simplification of agricultural landscapes worldwide, a concomitantly reduced crop diversity, and a substantial loss of non-crop habitats important for wildlife (Robinson and Sutherland 2002; Tscharntke et al. 2005). The resulting lack of breeding and foraging habitats in agroecosystems has led to a decline in species diversity observed for several taxa (Wilson et al. 1999; Weibull et al. 2000; Benton et al. 2003; Sánchez-Bayo and Wyckhuys 2019). Other reasons for the decline of biodiversity on farmlands include detrimental effects of agrochemicals (Batáry et al. 2008) and conventional tillage (Hatten et al. 2007). High landscape heterogeneity, in contrast, more specifically the diversity of habitats and their configuration, was found to promote biodiversity in agricultural landscapes (Fahrig et al. 2011). Thus, non-crop habitats such as grassland fallows, field margins or wildflower-sown fields are increasingly important reservoirs of arthropod diversity in agroecosystems (Gayer et al. 2019; Plath et al. 2021). For instance, non-crop habitats can serve as source habitats for species contributing to pest control by spilling over into croplands (Tschumi et al. 2016). Such habitats are especially important for predatory species with limited dispersal ability, such as many though not all ground beetles (Coleoptera: Carabidae), e.g. by providing sites for reproduction and overwintering (Boetzl et al. 2019). Even species thriving in agricultural landscapes may benefit from non-crop habitats, e.g. during temporal disturbances (tillage, harvest; Schneider et al. 2016) or for overwintering (Schmidt-Entling and Döbeli 2009). Thus, non-crop habitats are of essential importance for biodiversity conservation in agricultural landscapes.
Generalist predators such as Carabidae are of particular importance in agricultural landscapes, because they contribute to pest control (Lövei and Sunderland 1996; Kosewska et al. 2014). For example, they may reduce aphid densities in wheat fields (Collins et al. 2002; Symondson et al. 2002). Larger Carabidae are more sensitive to temporal variability in the availability of resources compared to smaller species (Blake et al. 1994; Ribera et al. 2001). In addition, larger carabids are more negatively affected by tillage or pesticide use (Rusch et al. 2013). Thus, they tend to prefer more undisturbed habitats, which is also due to their limited dispersal ability (Cole et al. 2002). Consequently, differences in morphological characteristics, i.e. body size in turn reflecting mobility, may affect habitat use in Carabidae (Rainio and Niemela 2003; Kotze and O’Hara 2003; Hanson et al. 2016). While, therefore, Carabidae have been often used as indicator group in agricultural landscapes, smaller phytophagous families, such as Chrysomelidae and Curculionidae, residing in the herb and shrub layer, have been less frequently studied in agroecosystems (Woodcock et al. 2005). The species within these families are often intimately associated with specific host plants, and thus respond rapidly to changes in land use (Marvaldi et al. 2002; Linzmeier and Ribeiro-Costa 2012). Accordingly, a high diversity of Chrysomelidae and Curculionidae can be found in non-crop habitats (Rischen et al. 2021), which also applies to other beetle families (e.g. Haaland et al. 2011; Frank et al. 2012).
Maintaining and creating non-crop habitats is undoubtedly one of the most effective conservation measure in intensively managed agricultural landscapes (Tscharntke et al. 2002; Knapp and Řezáč 2015). Thus, agri-environmental schemes under the Common Agricultural Policy (CAP) in European agricultural landscapes promote such habitats (Haaland et al. 2011; Gallé et al. 2020). Permanent grassland fallows are particularly important non-crop habitats for the conservation of arthropods in agricultural landscapes, due to their complex vegetation structure and often higher levels of soil moisture (Hendrickx et al. 2007; Plath et al. 2021). However, the effectiveness of other non-crop habitats such as grassy field margins or set-aside fields sown with wildflowers is more controversial. Still, set-aside wildflower fields (Plath et al. 2021) or wildflower strips along field margins (Haaland et al. 2011) are increasingly established to increase habitat heterogeneity. Furthermore, grassy field margins may contribute to the conservation of farmland species by providing less disturbed habitats that are compatible with agricultural practices (Marshall and Moonen 2002).
In an intensively used agricultural landscape in Western Germany, the Maifeld, quadratic areas (12 × 12 m) under poles of a power line were set aside and sown with wildflower seed mixtures in 2018 to promote farmland biodiversity. We here set out to compare the effectiveness of these ‘power pole islands’ as compared with grassy field margins, permanent grassland fallows, and wheat fields for insect conservation. We investigated ground- (Carabidae) as well as vegetation-dwelling species (Chrysomelidae, Curculionidae) to test the following predictions: (1) Beetle diversity and activity density are higher in grassy field margins and power pole islands compared to wheat fields, due to a lower management intensity and more complex vegetation structure, but lower than in much larger permanent grassland fallows. (2) Wheat fields harbour a higher proportion of open landscape and eurytopic beetle species compared to non-crop habitats, especially grassland fallows. (3) Grassland fallows, comprising relatively stable habitats, harbour more poor dispersers (i.e. larger, wingless carabids) than other habitat types and especially wheat fields.
Material and methods
Study area
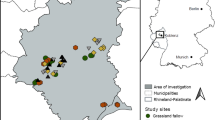
This study was performed in an intensively used agricultural landscape dominated by crop fields within the Eifel mountain range in western Germany (50°14’ N, 7°21’ E). The climate of the study area, which belongs to the ‘Maifeld’ region, is oceanic with a precipitation of ~ 598 mm/year and a mean annual temperature of ~ 10 °C (Agrarmeteorologie Rheinland-Pfalz 2020). In 2018, crop fields comprised 72.2%, forests 11.8%, settlement and traffic areas 13.9%, and other biotopes 2.1% of the Maifeld (Statistisches Landesamt Rheinland-Pfalz 2018). The study area is thus characterised by a mosaic of intensively used crop fields and small patches of non-crop habitats such as grassland fallows, field margins, and set-aside areas sown with wildflower mixtures. We compared four habitat types, each represented by nine replicate plots, to investigate the influence of land use on beetle assemblages (Online resource: Table S1): (1) wheat fields sown with Triticum aestivum (conventional management with fertilizing, fungicide and herbicide spraying; but all next to grassland fallows), (2) grassy field margins bordering wheat fields (mown once during sampling, several decades old), (3) set-aside habitat islands under power poles (two years old fallows located within wheat fields, sown with a commercially available mixture of wildflower seeds (including Calendula sp., Centaurea sp., Echium sp., Phacelia sp. etc.) as ‘greening’ measure; 12 × 12 m), and (4) grassland fallows (long-term grassland fallows with a diverse natural vegetation cover, formerly used as meadows but abandoned decades ago).
Beetle sampling
Beetles were sampled using two methods, pitfall trapping (to capture Carabidae) and suction sampling (to capture Chrysomelidae and Curculionidae). Pitfall trapping was performed between 28th of April and the 7th of July 2020. Two PET cups (∅ = 5.6 cm, volume 125 ml, filled with 70% water and 30% monopropylene glycol) were used per replicate plot. Both traps were buried into the ground at a distance of five meters and covered by a plastic roof for protection. In wheat fields and grassland fallows, pitfall traps were placed at a distance of 30 m from the respective edge, while in field margins and power pole islands they were set up in the centre of each plot. While on power pole islands the distance to the edges was about five meters, this varied in the field margins due to different widths of this habitat type, being typically less than 3 m. Traps were emptied every two weeks (i.e. a total of six times), with the number of trapping days ranging between 82–83 days per plot. Samples were stored in 70% ethanol and ground beetles identified to species level using taxonomic keys (Freude et al. 1964–1983).
Suction sampling was carried out on the same plots as above using a modified leaf blower (Stihl SH 56, Dieburg, Germany) with a polyamide stocking inserted into the nozzle. Each plot was sampled four times on dry and warm days in May and June 2020, with sampling conducted two weeks apart. Per sampling, the vegetation was sucked along a five meter transect (i.e. between both pitfall traps), with the nozzle being inserted into the vegetation 10 times for 7 s each (Brook et al. 2008). Please note the substantial difference in sampling effort between both methods employed. Thus, while our focus was clearly on pitfall trapping, we still believe that suction sampling, even though with reduced effort, provides interesting additional insights into the responses of vegetation-dwelling beetles. Samples were afterwards transferred to plastic bags, frozen at -18 °C, and then stored in 70% ethanol. Chrysomelidae and Curculionidae were identified to species level using taxonomic keys (Freude et al. 1964–1983).
Data analyses
Data from pitfall trapping (Carabidae) and suction sampling (Chrysomelidae and Curculionidae) were analysed separately, pooling all respective data per plot. We calculated the numbers of species and individuals, and the effective number of species for each plot. To estimate the latter, the Shannon–Wiener entropy index was converted to true diversity using the formula ‘exp^H’ (Jost 2006). To account for differences in detection probability between habitat types, rarefied species richness was calculated for the pitfall trapping data for a sample coverage of 90% using the iNEXT package (Hsieh et al. 2020) in R 4.0.5 (R Development Core Team 2021). For the suction sampling data, this analysis was not possible due to a high number of 0 values. Data on body size of each species were taken from Freude et al. (1964–1983) and Homburg et al. (2014). Community weighted mean (CWM) values for body size were calculated by weighting the respective body size by each species' abundance (Ricotta and Moretti 2011). Freude et al. (1964–1983) was also used to assign habitat preferences (open landscape, eurytopic or hygrophilous species) to each beetle species. In addition, Carabidae were classified according to their flight ability as good (winged) or poor (wingless and dimorphic species), using data available in Homburg et al. (2014). For subsequent analyses, we standardized the above data by giving the respective proportion per plot. Finally, we obtained the length, width and thereby size of each plot using the distance tool in GoogleMaps. To assess the shape of plots, the perimeter-to-area ratio was calculated.
Statistical analyses
Kruskal–Wallis ANOVAs were used to test for significant differences among habitat types in the numbers of species and individuals, the number of individuals for the most common Carabidae, effective number of species, rarefied species richness, CWM body size, plot size and shape (dependent variables). Because normal distribution and homogeneity of variance were not met, we used a non-parametric test. Multiple comparisons were used to locate significant differences. Using Pearson correlations, we tested for significant effects of plot size and shape on dependent variables. Only for the suction sampling data, we found significant correlations between plot shape and the number of species (r = -0.347, p = 0.038) and the effective number of species (r = -0.362, p = 0.030) as well as between plot size and CWM body size (r = -0.347, p = 0.038). For the above cases, we additionally performed ANCOVAs by including plot shape or size as covariates in addition to habitat type. Effects of covariates were non-significant throughout (Online resource: Table S2) such that within the article only analyses excluding covariates are presented. We also tested for spatial autocorrelation using Moran’s I tests (ape-package; Dormann et al. 2007; Paradis and Schliep 2019), but found no evidence of spatial autocorrelation in any of the dependent variables (Online resource: Table S3).
We used non-metric multidimensional scaling (NMDS) analyses, based on the Bray–Curtis index of dissimilarity, to visualise differences in the species assemblages of Carabidae among the four habitat types. Suction sampling data were insufficient for NMDS ordination, because of 0 values. For calculating Bray–Curtis dissimilarities, proportional data were used to standardise for differences in the total abundance of beetles. We tested for differences in carabid assemblages using permutational multivariate analyses of variance (PERMANOVA, with 999 permutations). PERMANOVAs (999 permutations) were also used to test for significant differences between habitat types in the proportion of species with specific habitat preferences (all families) or flight ability (Carabidae only). For standardization, we used the relative abundance of species throughout. Pairwise PERMANOVAs were performed to test for significant differences between habitat types. Statistical analyses were performed with Statistica 12.0 (Tulsa, StatSoft) or using the vegan package in R 4.0.5 (R Development Core Team 2021; Oksanen et al. 2020).
Results
A total of 5491 Carabidae of 70 species were captured with pitfall trapping. Suction sampling yielded 94 Chrysomelidae belonging to 23 species and 125 Curculionidae belonging to 26 species (Online resource: Table S1). Considering the pitfall data, five carabids accounted for 63% of all individuals: Nebria salina (Fairmaire and Laboulbène, 1854; 1034 individuals in total), Pterostichus melanarius (Illiger, 1798; 776), Anchomenus dorsalis (Pontoppidan, 1763; 620), Brachinus explodens (Duftschmid, 1812; 616), and Poecilus cupreus (Linnaeus, 1758; 425). Six Carabidae found are included in the red list of Germany (Geiser 1998) as near threatened (3 species; Chlaenius nigricornis Fabricius, 1787; Harpalus dimidiatus Rossi, 1790; Harpalus serripes Quensel, 1806) or vulnerable (3 species; Carabus convexus Fabricius, 1775; Harpalus hirtipes Panzer, 1796; Harpalus melancholicus Dejean, 1829). No threatened species were recorded for Chrysomelidae and Curculionidae (cf. Freude et al. 1964–1983; Geiser 1998).
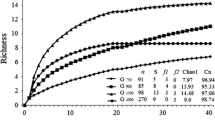
Habitat type only significantly affected the rarefied species number and the effective number of species of Carabidae, and no parameter of the Chrysomelidae and Curculionidae (Table 1; Fig. 1; Online resource: Fig. S1, Fig. S2). For Carabidae, the rarefied species number was significantly higher in field margins and grassland fallows than in power pole islands, and the effective number of species tended to be higher in field margins than in power pole islands. Furthermore, there were significant differences in plot size and shape between habitat types (size: H3,32 = 26.99, p < 0.0001; shape: H3,32 = 30.05, p < 0.0001; Online resource: Fig. S3). Wheat fields and grassland fallows were larger than power pole islands and field margins, the latter being much more elongated than the other habitat types.
Numbers of species, individuals, and rarefied species and effective number of species (ENS) of Carabidae captured by pitfall trapping across four habitat types. Boxplots display the interquartile range (25–75%; box) and the median (line in the box). Whiskers represent 1.5 times the lower or upper interquartile range. Different letters above bars indicate significant differences among habitat types (multiple comparisons after Kruskal–Wallis ANOVA; n = 9 plots each)
Kruskal–Wallis tests also showed that habitat type significantly influenced the number of individuals in N. salina (H3 = 22.32, p < 0.001), A. dorsalis (H3 = 18.79, p < 0.001), B. explodens (H3 = 17.38, p < 0.001), and P. cupreus (H3 = 11.42, p < 0.01), while there were no significant differences for P. melanarius (H3 = 3.94, p = 0.268). For all five former species, the number of individuals were lowest in grassland fallows (Fig. 2). This is also reflected by the respective percentages of individuals across habitat types. Wheat fields, field margins and power pole islands were clearly dominated by the five dominant carabids (55–85% of all individuals), while they were poorly represented in grassland fallows (< 10% of all individuals; Fig. 3).
Number of individuals for the five most common Carabidae captured by pitfall trapping across four habitat types. Boxplots display the interquartile range (25–75%; the box) and the median (line in the box). Whiskers represent 1.5 times the lower or upper interquartile range. Different letters above bars indicate significant differences among habitat types (multiple comparisons after Kruskal–Wallis ANOVA; n = 9 plots each)
The NMDS ordination of Carabidae showed significant variation in species composition between habitat types (PERMANOVA: F3,32 = 3.35, p = 0.001, R2 = 0.24; Fig. 4). There were significant differences between all habitat types (PERMANOVA, pairwise comparisons: p < 0.05), except for field margins and power pole islands (F1,15 = 1.92, p = 0.103, R2 = 0.11), with strongest differences between the assemblages of wheat fields and grassland fallows as well as power pole islands and grassland fallows. Variation among grassland fallows was much higher compared with all other habitat types.
For both sampling methods, PERMANOVAs showed significant differences among habitat types in habitat preferences (Carabidae: F3,32 = 4.88, p = 0.003, R2 = 0.31; Chrysomelidae and Curculionidae: F3,32 = 3.19, p = 0.014, R2 = 0.26). For Carabidae, all habitat types differed significantly from each other except for field margins and power pole islands. Open landscape species dominated except for grassland fallows, in which hygrophilous species were most abundant (Fig. 5). In Chrysomelidae and Curculionidae, power pole islands differed significantly from all other habitat types. Here, eurytopic species dominated in wheat fields and grassland fallows, while open landscape species clearly dominated in power pole islands. Finally, carabid assemblages differed significantly in flight ability (F3,32 = 6.61, p = 0.001, R2 = 0.38), with significant differences between grassland fallows and all other habitat types. In contrast to all other habitat types, grassland fallows were dominated by species with poor flight ability (Fig. 5c).
Distribution of habitat preferences (open landscape; eurytopic; hygrophilous) of Chrysomelidae and Curculionidae (a suction sampling) and Carabidae (b pitfall trapping) and flight ability (c good; poor) of ground beetles (pitfall trapping) across four habitat types. W: wheat field, M: field margin, I: power pole island, F: grassland fallow. Data for habitat preferences and flight ability according to Freude et al. (1964–1983) and Homburg et al. (2014)
Discussion
Non-crop habitats, such as grassy field margins, wildflower-sown areas, and grassland fallows, were expected to harbour more beetle species than wheat fields because beetles are in general highly sensitive to changes in habitat structures and agricultural practices (e.g. use of agrochemicals for Carabidae; Lövei and Sunderland 1996). Contrary to our expectations, though, we found no significant differences in species richness, activity-density, and effective number of species of three beetle families between wheat fields and non-crop habitats. Similarly, carabid species richness did not differ between newly established grasslands and adjoining cereal fields in a recent study (Hussain et al. 2021), and assemblages may be generally impoverished close to crop fields (see Rand et al. 2006). Note that non-significant results in Chrysomelidae and Curculionidae may also stem from the reduced sampling effort and concomitantly low sample sizes.
For Carabidae, rarefied species number was higher in field margins and fallows than in wheat fields and power pole islands. Also, the effective species number of Carabidae was higher in field margins than in power pole islands. The low values for the latter may well be explained by the young age of these habitats. The relatively high values found for wheat fields, in contrast, may at least partly result from methodological artefacts. In particular, the higher and denser vegetation of non-crop habitats compared to wheat fields may hamper beetle activity (i.e. speed) and also reduce the efficiency of suction sampling (Honek 1988; Thomas et al. 2006; Vician et al. 2015). Moreover, all wheat fields studied here were located close to grassland fallows, such that spillover effects may also contribute to their relatively high species richness. Carabids are known to often colonize crop fields from nearby non-crop habitats, which can lead to higher densities in adjacent fields (Weibull and Östman 2003; Purtauf et al. 2005; Fusser et al. 2018).
However, beetle assemblages differed clearly between the four habitat types. In Chrysomelidae and Curculionidae, small-scale power pole islands differed from the other habitat types with the dominance of open landscape species. In Carabidae, all habitat types differed from each other except for field margins and power pole islands, which were dominated by open landscape species, such as wheat fields, but grassland fallows by hygrophilous species. The dominance of hygrophilous Carabidae in grassland fallows is probably due to the high soil moisture of fallows (Marasas et al. 2010). The much lower proportion of hygrophilous species in the Chrysomelidae and Curculionidae even in fallows as compared to Carabidae is likely due to their vegetation-dwelling life style. Thus, we assume that ground-dwelling Carabidae are more dependent on and sensitive to soil moisture compared to both other taxa. This would also explain the high abundance of Agonum viduum (Panzer, 1796) in grassland fallows, which is a hygrophilous species with poor dispersal ability (Online resource: Table S4a). Our results on habitat preferences are overall consistent with our second hypothesis that wheat fields mainly harbour open landscape and eurytopic beetle species. Furthermore, the Carabidae found in grassland fallows were mainly poor dispersers, as opposed to wheat fields, field margins, and power pole islands, largely consistent with hypothesis 3. Arable fields are often dominated by highly dispersive pioneer species that are adapted to frequently changing conditions and different kinds of open habitats (Anjum-Zubair et al. 2015; Ribera et al. 2001). Accordingly, permanent grassland fallows in particular may support beetle species with specific habitat requirements that are most affected by the homogenisation of agricultural landscapes (Ulrich et al. 2004).
Differences among habitat types in species composition are also reflected by the distribution patterns of specific species. For example, the occurrence of many Curculionidae species is related to the presence of their food plant. Accordingly, Malvapion malvae (Fabricius, 1775) reached high densities in power pole islands being rich in mallows (Malva sp.), and Tychius breviusculus (Desbrochers, 1873) in grassy field margins with a high abundance of melilot (Melilotus sp.; Online resource: Table S4c). The establishment of additional plant species by sowing seed mixtures is especially relevant for such phytophagous beetle species (Anjum-Zubair et al. 2010). While phytophagous beetles (e.g. Chrysomelidae and Curculionidae) are thus often attracted to areas with high plant diversity (Frank et al. 2012), other factors (e.g. agronomic activities, edge habitats) appear to be more important for predatory carabids (Duflot et al. 2017; Gailis et al. 2017).
The relatively high activity-densities of carabids in wheat fields were at least partly caused by the presence of very common predatory species that thrived in wheat fields, grassy field margins and power pole islands, but which were largely lacking in grassland fallows (Figs. 2 and 3). For instance, A. dorsalis, B. explodens, P. cupreus, and P. melanarius are characteristic species of cereal fields over a wide area (Kromp 1999; Hussain et al. 2021). In contrast, the abundant occurrence of N. salina in our study area is more unusual, as this species is typically recorded in lower numbers in agroecosystems. However, the abundant occurrence of the five most common carabids found in crop fields has also been shown in other studies (e.g. Pfiffner and Luka 2003; Purtauf et al. 2005).
Morpho-ecological traits of beetle communities such as diet preference, wing morphology, or body size may be more suitable indicators of anthropogenic impacts on habitats than species richness or abundance (Gobbi and Fontaneto 2008). In our case, wheat fields, field margins and power pole islands were dominated by carabids with good dispersal ability, which can readily colonise highly disturbed sites. In general, ground beetles living in unstable environments have good dispersal abilities that allow them to move to more stable habitats when less favourable conditions appear (Ribera et al. 2001). With the exception of P. melanarius, which is often recorded in high densities in field interiors (e.g. Gayer et al. 2019; Bennewicz and Barczak 2020), the remaining most common carabids in our study have good flight ability and almost exclusively colonised wheat fields, grassy field margins, and power pole islands. In contrast, grassland fallows provided suitable habitats for flightless carabids, which are particularly at risk of local extinction in intensively used agricultural landscapes (Griffiths et al. 2007), possibly due to the high age of this habitat type.
In summary, the habitat types investigated harboured different carabid assemblages, except from field margins and power pole islands, thus enhancing overall beetle diversity. Thus, assemblages of grassland fallows, non-crop habitats (power pole islands and fields margins), and wheat fields were different. This highlights the importance of landscape composition and configuration for maintaining biodiversity in agroecosystems (Martin et al. 2019). This is in line with other studies on beetle communities in agricultural landscapes, having documented positive effects of heterogeneous habitat structures (Benton et al. 2003; Diekötter et al. 2010; Fahrig et al. 2011; Knapp and Řezáč 2015). Increased habitat diversity may provide access to additional food including ephemeral resources and overwintering sites (Coombes and Sothertons 1986; Pfiffner and Luka 2000; Macfadyen and Muller 2013). In particular, many carabid species (e.g. P. cupreus) are known to use wheat fields and adjacent non-crop habitats as complementary habitats (Duflot et al. 2015). Such structures may also represent important corridors for the dispersal of Carabidae (Šustek 1994). Notably, the within-group variation of the carabid assemblages in grassland fallows was much higher compared to other habitat types, which is likely related to differences in ecological factors and stresses the high importance of permanent non-crop habitats for conservation in agricultural landscapes. Feng et al. (2021) also showed that older fallows, such as the grassland follows studied here, support more diverse communities and are particularly important for the conservation of habitat specialists.
Conclusions
Our results did not support our initial expectation of a higher species richness in non-crop habitats in an agricultural landscape. However, different habitat types harboured different species assemblages, with grassland fallows showing the largest within-group variation. Permanent grassland fallows are thus of particular importance for nature conservation, also as they provided refuge areas for hygrophilous and flightless beetle species, whereas wheat fields were mainly colonised by open landscape species with high dispersal ability. In addition, species assemblages of non-crop habitats differed from wheat fields, showing their potential to increase overall insect diversity in agricultural landscapes. Therefore, functional traits (e.g. flight ability, body size) rather than species richness may represent more robust indicators for assessing habitat quality in relation to anthropogenic disturbance. Overall, our results show that different types of non-crop habitats harbour different beetle assemblages and thus contribute to promoting diversity in agroecosystems. Thus, both small- and large-scale non-crop habitats should be taken into account to improve the functional diversity of beetle assemblages in agricultural landscapes.
Data availability
All data are provided as online resource.
References
Agrarmeteorologie Rheinland-Pfalz (2020) Langjährige Mittelwerte der Wetterstation Münstermaifeld. https://www.am.rlp.de. Accessed 10 Sep 2020
Anjum-Zubair M, Entling MH, Bruckner A et al (2015) Differentiation of spring carabid beetle assemblages between semi-natural habitats and adjoining winter wheat. Agric For Entomol 17:355–365. https://doi.org/10.1111/afe.12115
Anjum-Zubair M, Schmidt-Entling MH, Querner P, Frank T (2010) Influence of within-field position and adjoining habitat on carabid beetle assemblages in winter wheat. Agric For Entomol 12:301–306. https://doi.org/10.1111/j.1461-9563.2010.00479.x
Batáry P, Kovács A, Báldi A (2008) Management effects on carabid beetles and spiders in Central Hungarian grasslands and cereal fields. Community Ecol 9:247–254. https://doi.org/10.1556/ComEc.9.2008.2.14
Bennewicz J, Barczak T (2020) Ground beetles (Carabidae) of field margin habitats. Biologia 75:1631–1641. https://doi.org/10.2478/s11756-020-00424-y
Benton TG, Vickery JA, Wilson JD (2003) Farmland biodiversity: is habitat heterogeneity the key? Trends Ecol Evol 18:182–188. https://doi.org/10.1016/S0169-5347(03)00011-9
Blake S, Foster GN, Eyre MD, Luff ML (1994) Effects of habitat type and grassland management practices on the body size distribution of carabid beetles. Pedobiologia 38:502–512
Boetzl FA, Krimmer E, Krauss J, Steffan-Dewenter I (2019) Agri-environmental schemes promote ground-dwelling predators in adjacent oilseed rape fields: diversity, species traits and distance-decay functions. J Appl Ecol 56:10–20. https://doi.org/10.1111/1365-2664.13162
Brook AJ, Woodcock BA, Sinka M, Vanbergen AJ (2008) Experimental verification of suction sampler capture efficiency in grasslands of differing vegetation height and structure. J Appl Ecol 45:1357–1363. https://doi.org/10.1111/j.1365-2664.2008.01530.x
Cole LJ, Mccracken DI, Dennis P et al (2002) Relationships between agricultural management and ecological groups of ground beetles (Coleoptera: Carabidae) on Scottish farmland. Agric For Entomol 93:323–336. https://doi.org/10.1016/S0167-8809(01)00333-4
Collins KL, Boatman ND, Wilcox A et al (2002) Influence of beetle banks on cereal aphid predation in winter wheat. Agric Ecosyst Environ 93:337–350. https://doi.org/10.1016/S0167-8809(01)00340-1
Coombes DS, Sothertons NW (1986) The dispersal and distribution of polyphagous predatory Coleoptera in cereals. Ann Appl Biol 108:461–474. https://doi.org/10.1111/j.1744-7348.1986.tb01985.x
Diekötter T, Wamser S, Wolters V, Birkhofer K (2010) Landscape and management effects on structure and function of soil arthropod communities in winter wheat. Agric Ecosyst Environ 137:108–112. https://doi.org/10.1016/j.agee.2010.01.008
Dormann CF, McPherson JM, Araújo MB et al (2007) Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography 30:609–628. https://doi.org/10.1111/j.2007.0906-7590.05171.x
Duflot R, Aviron S, Ernoult A et al (2015) Reconsidering the role of ‘semi-natural habitat’ in agricultural landscape biodiversity: a case study. Ecol Res 30:75–83. https://doi.org/10.1007/s11284-014-1211-9
Duflot R, Ernoult A, Aviron S et al (2017) Relative effects of landscape composition and configuration on multi-habitat gamma diversity in agricultural landscapes. Agric Ecosyst Environ 241:62–69. https://doi.org/10.1016/j.agee.2017.02.035
Fahrig L, Baudry J, Brotons L et al (2011) Functional landscape heterogeneity and animal biodiversity in agricultural landscapes. Ecol Lett 14:101–112. https://doi.org/10.1111/j.1461-0248.2010.01559.x
Feng L, Arvidsson F, Smith HG, Birkhofer K (2021) Fallows and permanent grasslands conserve the species composition and functional diversity of carabid beetles and linyphiid spiders in agricultural landscapes. Insect Conserv Divers 14:825-836. https://doi.org/10.1111/icad.12520
Frank T, Aeschbacher S, Zaller JG (2012) Habitat age affects beetle diversity in wildflower areas. Agric Ecosyst Environ 152:21–26. https://doi.org/10.1016/j.agee.2012.01.027
Freude H, Harde KW, Lohse GA (1964-1983) Die Käfer Mitteleuropas. Bände 1–11. Goecke & Evers, Krefeld
Fusser MS, Holland JM, Jeanneret P et al (2018) Interactive effects of local and landscape factors on farmland carabids. Agric For Entomol 20:549–557. https://doi.org/10.1111/afe.12288
Gailis J, Turka I, Ausmane M (2017) The most frequent ground beetles (Coleoptera, Carabidae) are differently affected by main soil treatment and crop rotation in winter wheat fields. Acta Biol Univ Daugavp 17:29–52
Gallé R, Geppert C, Földesi R et al (2020) Arthropod functional traits shaped by landscape-scale field size, local agri-environment schemes and edge effects. Basic Appl Ecol 48:1–10. https://doi.org/10.1016/j.baae.2020.09.006
Gayer C, Lövei GL, Magura T et al (2019) Carabid functional diversity is enhanced by conventional flowering fields, organic winter cereals and edge habitats. Agric Ecosyst Environ 284:106579. https://doi.org/10.1016/j.agee.2019.106579
Geiser R (1998) Rote Liste der Käfer (Coleoptera). In: Binot M, Bless R, Boye P et al (eds) Rote Liste gefährdeter Tiere Deutschlands. - Schriftenreihe für Landschaftspflege und Naturschutz. Bonn-Bad Godesberg, pp 178–179
Gobbi M, Fontaneto D (2008) Biodiversity of ground beetles (Coleoptera: Carabidae) in different habitats of the Italian Po lowland. Agric Ecosyst Environ 127:273–276. https://doi.org/10.1016/j.agee.2008.04.011
Griffiths GJK, Winder L, Holland JM, Thomas CFG (2007) The representation and functional composition of carabid and staphylinid beetles in different field boundary types at a farm-scale. Biol Conserv 135:145–152. https://doi.org/10.1016/j.biocon.2006.09.016
Haaland C, Naisbit RE, Bersier LF (2011) Sown wildflower strips for insect conservation: a review. Insect Conserv Divers 4:60–80. https://doi.org/10.1111/j.1752-4598.2010.00098.x
Hanson HI, Palmu E, Birkhofer K et al (2016) Agricultural land use determines the trait composition of ground beetle communities. PLoS One 11:1–13. https://doi.org/10.1371/journal.pone.0146329
Hatten TD, Bosque-Pérez NA, Labonte JR et al (2007) Effects of tillage on the activity density and biological diversity of carabid beetles in spring and winter crops. Environ Entomol 36:356–368. https://doi.org/10.1603/0046-225X(2007)36[356:EOTOTA]2.0.CO;2
Hendrickx F, Maelfait J, Van Wingerden W et al (2007) How landscape structure, land-use intensity and habitat diversity affect components of total arthropod diversity in agricultural landscapes. J Appl Ecol 44:340–351. https://doi.org/10.1111/j.1365-2664.2006.01270.x
Homburg K, Homburg N, Schäfer F et al (2014) Carabids. org - a dynamic online database of ground beetle species traits (Coleoptera, Carabidae). Insect Conserv Divers 7:195–205. https://doi.org/10.1111/icad.12045
Honek A (1988) The effect of crop density and microclimate on pitfall trap catches of Carabidae, Staphylinidae (Coleoptera), and Lycosidae (Araneae) in cereal fields. Pedobiologia 32:233–242
Hsieh TC, Ma KH, Chao A (2020) iNEXT: iNterpolation and EXTrapolation for species diversity. R package version 2.0.20
Hussain RI, Brandl M, Maas B et al (2021) Re-established grasslands on farmland promote pollinators more than predators. Agric Ecosyst Environ 319:107543. https://doi.org/10.1016/j.agee.2021.107543
Jost L (2006) Entropy and diversity. Oikos 113:363–375. https://doi.org/10.1111/j.2006.0030-1299.14714.x
Knapp M, Řezáč M (2015) Even the smallest son-crop habitat islands could be beneficial: distribution of carabid beetles and spiders in agricultural landscape. PLoS One 10(4):e0123052. https://doi.org/10.1371/journal.pone.0123052
Kosewska A, Skalski T, Nietupski M (2014) Effect of conventional and non-inversion tillage systems on the abundance and some life history traits of carabid beetles (Coleoptera: Carabidae) in winter triticale fields. Eur J Entomol 111:669–676. https://doi.org/10.14411/eje.2014.078
Kotze DJ, O’Hara RB (2003) Species decline - but why? Explanations of carabid beetle (Coleoptera, Carabidae) declines in Europe. Oecologia 135:138–148. https://doi.org/10.1007/s00442-002-1174-3
Kromp B (1999) Carabid beetles in sustainable agriculture: a review on pest control efficacy, cultivation impacts and enhancement. Agric Ecosyst Environ 74:187–228. https://doi.org/10.1016/b978-0-444-50019-9.50014-5
Linzmeier AM, Ribeiro-Costa CS (2012) Spatial-temporal composition of Chrysomelidae (Insecta: Coleoptera) communities in southern Brazil. J Nat Hist 46:1921–1938. https://doi.org/10.1080/00222933.2012.707237
Lövei GL, Sunderland KD (1996) Ecology and behavior of ground beetles (Coleoptera: Carabidae). Annu Rev Entomol 41:231–256. https://doi.org/10.1146/annurev.ento.41.1.231
Macfadyen S, Muller W (2013) Edges in agricultural landscapes: species interactions and movement of natural enemies. PLoS One 8(3):e59659. https://doi.org/10.1371/journal.pone.0059659
Marasas ME, Sarandón SJ, Cicchino A (2010) Semi-natural habitats and field margins in a typical agroecosystem of the Argentinean Pampas as a reservoir of carabid beetles. J Sustain Agric 34:153–168. https://doi.org/10.1080/10440040903482563
Marshall EJ, Moonen AC (2002) Field margins in northern Europe: their functions and interactions with agriculture. Agric Ecosyst Environ 89:5–21. https://doi.org/10.1016/S0167-8809(01)00315-2
Martin EA, Dainese M, Clough Y et al (2019) The interplay of landscape composition and configuration: new pathways to manage functional biodiversity and agroecosystem services across Europe. Ecol Lett 22:1083–1094. https://doi.org/10.1111/ele.13265
Marvaldi AE, Sequeira AS, O’Brien CW, Farrel BD (2002) Molecular and morphological phylogenetics of weevils (Coleoptera, Curculionoidea): do niche shifts accompany diversification? Syst Biol 51:761–785. https://doi.org/10.1080/10635150290102465
Oksanen J, Blanchet FG, Friendly M et al (2020) vegan: Community Ecology Package. R package version 2.5–7
Paradis E, Schliep K (2019) ape 5.0: an environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 35:526–528. https://doi.org/10.1093/bioinformatics/bty633
Pfiffner L, Luka H (2003) Effects of low-input farming systems on carabids and epigeal spiders - a paired farm approach. Basic Appl Ecol 4:117–127. https://doi.org/10.1078/1439-1791-00121
Pfiffner L, Luka H (2000) Overwintering of arthropods in soils of arable fields and adjacent semi-natural habitats. Agric Ecosyst Environ 78:215–222. https://doi.org/10.1016/S0167-8809(99)00130-9
Plath E, Rischen T, Mohr T, Fischer K (2021) Biodiversity in agricultural landscapes: Grassy field margins and semi-natural fragments both foster spider diversity and body size. Agric Ecosyst Environ 316:107457. https://doi.org/10.1016/j.agee.2021.107457
Purtauf T, Roschewitz I, Dauber J et al (2005) Landscape context of organic and conventional farms: influences on carabid beetle diversity. Agric Ecosyst Environ 108:165–174. https://doi.org/10.1016/j.agee.2005.01.005
R Core Development Core Team (2021): R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rainio J, Niemela J (2003) Ground beetles (Coleoptera: Carabidae) as bioindicators. Biodivers Conserv 12:487–506. https://doi.org/10.1023/A:1022412617568
Rand TA, Tylianakis JM, Tscharntke T (2006) Spillover edge effects: the dispersal of agriculturally subsidized insect natural enemies into adjacent natural habitats. Ecol Lett 9:603–614. https://doi.org/10.1111/j.1461-0248.2006.00911.x
Ribera I, Dolédec S, Downie IS, Foster GN (2001) Effect of land disturbance and stress on species traits of ground beetle assemblages. Ecology 82:1112–1129. https://doi.org/10.1890/0012-9658(2001)082[1112:EOLDAS]2.0.CO;2
Ricotta C, Moretti M (2011) CWM and Rao’s quadratic diversity: a unified framework for functional ecology. Oecologia 167:181–188. https://doi.org/10.1007/s00442-011-1965-5
Rischen T, Frenzel T, Fischer K (2021) Biodiversity in agricultural landscapes: different non-crop habitats increase diversity of ground-dwelling beetles (Coleoptera) but support different communities. Biodivers Conserv 30:3965–3981. https://doi.org/10.1007/s10531-021-02284-7
Robinson RA, Sutherland WJ (2002) Post-war changes in arable farming and biodiversity in Great Britain. J Appl Ecol 39:157–176. https://doi.org/10.1046/j.1365-2664.2002.00695.x
Rusch A, Bommarco R, Chiverton P et al (2013) Response of ground beetle (Coleoptera, Carabidae) communities to changes in agricultural policies in Sweden over two decades. Agric Ecosyst Environ 176:63–69. https://doi.org/10.1016/j.agee.2013.05.014
Sánchez-Bayo F, Wyckhuys KAG (2019) Worldwide decline of the entomofauna: a review of its drivers. Biol Conserv 232:8–27. https://doi.org/10.1016/j.biocon.2019.01.020
Schmidt-Entling MH, Döbeli J (2009) Sown wildflower areas to enhance spiders in arable fields. Agric Ecosyst Environ 133:19–22. https://doi.org/10.1016/j.agee.2009.04.015
Schneider G, Krauss J, Boetzl FA et al (2016) Spillover from adjacent crop and forest habitats shapes carabid beetle assemblages in fragmented semi-natural grasslands. Oecologia 182:1141–1150. https://doi.org/10.1007/s00442-016-3710-6
Statistisches Landesamt Rheinland-Pfalz (2018) Verbandsgemeinde Maifeld - Flächennutzung. http://infothek.statistik.rlp.de. Accessed 4 Jun 2020
Šustek Z (1994) Windbreaks as migration corridors for carabids in an agricultural landscape. In: Desender K, Dufrêne M, Loreau M et al (eds) Carabid beetles: ecology and evolution. Kluwer Academic Publishers, pp 377–382
Symondson WOC, Sunderland KD, Greenstone MH (2002) Can generalist predators be effective biocontrol agents? Annu Rev Entomol 47:561–594. https://doi.org/10.1146/annurev.ento.47.091201.145240
Thomas CFG, Brown NJ, Kendall DA (2006) Carabid movement and vegetation density: implications for interpreting pitfall trap data from split-field trials. Agric Ecosyst Environ 113:51–61. https://doi.org/10.1016/j.agee.2005.08.033
Tscharntke T, Klein AM, Kruess A et al (2005) Landscape perspectives on agricultural intensification and biodiversity - ecosystem service management. Ecol Lett 8:857–874. https://doi.org/10.1111/j.1461-0248.2005.00782.x
Tscharntke T, Steffan-Dewenter I, Kruess A, Thies C (2002) Contribution of small habitats to conservation of insect communities of grassland-cropland landscapes. Ecol Appl 12:354. https://doi.org/10.2307/3060947
Tschumi M, Albrecht M, Collatz J et al (2016) Tailored flower strips promote natural enemy biodiversity and pest control in potato crops. J Appl Ecol 53:1169–1176. https://doi.org/10.1111/1365-2664.12653
Ulrich W, Buszko J, Czarnecki A (2004) The contribution of poplar plantations to regional diversity of ground beetles (Coleoptea: Carabidae) in agricultural landscapes. Ann Zool Fennici 41:501–512
Vician V, Svitok M, Kočík K, Stašiov S (2015) The influence of agricultural management on the structure of ground beetle (Coleoptera : Carabidae) assemblages. Biologia 70:240–251. https://doi.org/10.1515/biolog-2015-0028
Weibull A-C, Östman Ö (2003) Species composition in agroecosystems: The effect of landscape, habitat, and farm management. Basic Appl Ecol 4:349–361. https://doi.org/10.1078/1439-1791-00173
Weibull AC, Bengtsson J, Nohlgren E (2000) Diversity of butterflies in the agricultural landscape: the role of farming system and landscape heterogeneity. Ecography 23:743–750. https://doi.org/10.1111/j.1600-0587.2000.tb00317.x
Wilson JD, Morris AJ, Arroyo BE et al (1999) A review of the abundance and diversity of invertebrate and plant foods of granivorous birds in northern Europe in relation to agricultural change. Agric Ecosyst Environ 75:13–30. https://doi.org/10.1016/S0167-8809(99)00064-X
Woodcock BA, Westbury DB, Potts SG et al (2005) Establishing field margins to promote beetle conservation in arable farms. Agric Ecosyst Environ 107:255–266. https://doi.org/10.1016/j.agee.2004.10.029
Acknowledgements
We are grateful to Roland Busch, Katharina Geisbüsch, and Daniel Ruppert for their help with the field work. We thank Thomas Wagner for help with species identifications. Thanks also to the local farmers and landowners for providing access to their land and for their support. We would like to thank two anonymous reviewers for their constructive criticism.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was financially supported by the Ministry for Environment, Energy, Nutrition and Forest Rheinland-Pfalz through the ‘Aktion Grün’.
Author information
Authors and Affiliations
Contributions
KF and TR designed the experiment, TR and KE collected field data and TR, KE and MH identified the beetles, KF and TR analysed the data with help from KE and MH, TR and KF wrote the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Conflicts of interest/Competing interests
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rischen, T., Ehringhausen, K., Heyer, M. et al. Responses of selected beetle families (Carabidae, Chrysomelidae, Curculionidae) to non-crop habitats in an agricultural landscape. Biologia 77, 2149–2159 (2022). https://doi.org/10.1007/s11756-022-01100-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01100-z