Abstract
Purpose
We aimed to determine the effects of a classmate-supported, supervised, in-hospital physical activity program during treatment primarily on cardiorespiratory fitness and secondarily on physical function.
Methods
A multicenter non-randomized controlled intervention study including children diagnosed with cancer, 6–18 years at diagnosis treated with chemo-/radiotherapy. The intervention comprised (i) an educational session on cancer in the child’s school class; (ii) selection of two “ambassadors”—classmates who were co-admitted, supporting the child’s everyday hospital life; and (iii) supervised in-hospital physical activity from diagnosis and throughout intensive treatment. One-year post-treatment, physical testing included cardiorespiratory fitness (primary outcome), Sit-to-Stand test, Timed-Up-and-Go, and Handgrip Strength.
Results
The intervention group included 75 of 120 children (61% boys, 13.4 ± 3.1 years); the control groups included 33 of 58 children with cancer (58% boys, 13.5 ± 2.5 years), and 94 age- and sex-matched children without a cancer history. One-year post-treatment, cardiorespiratory fitness tended to be higher in the intervention group (37.0 ± 6.0 mL/kg/min) than in the patient control group with cancer (32.3 ± 9.7 mL/kg/min) (mean difference 4.7 [0.4 to 9.1], p = 0.034). The intervention group performed better in the secondary outcomes. Compared with community controls, both patient groups had lower cardiorespiratory fitness. The patient control group had lower Sit-to-Stand, Timed Up and Go, and Handgrip Strength, while the intervention group had strength comparable to that of the community controls.
Conclusions
Peer-supported, supervised, in-hospital physical activity during treatment may improve cardiorespiratory fitness and muscle strength 1-year post-treatment in children with cancer; however, survivors continue to have lower cardiorespiratory fitness than community controls.
Implications for Cancer Survivors
Children with cancer may benefit from in-hospital physical activity in improving long-term cardiorespiratory fitness and muscle strength.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
The improvements in childhood cancer survival rates create a need to lessen long-term treatment-related late effects to promote the best possible return to everyday life, including social, academic, and physical activities [1, 2]. Childhood survivors of cancer (CCS) experience prolonged absence from school, sports, and leisure activities during treatment, reducing their peer interaction and disrupting their development of social skills [3,4,5]. Impaired cardiorespiratory fitness [6,7,8,9,10], muscle strength [11, 12], and physical performance [11, 13] are common long-term physiological consequences of anti-cancer treatment, affecting CCS’ ability to perform activities of daily living and their self-perception [14, 15] and reducing their ability to fully participate in social activities and education [16, 17]. Consequently, the CCS are vulnerable to social exclusion [3], which further diminishes their incentive to be physically active [18, 19]. Taken together, the impairments in physical and social functioning impact their health-related quality of life [20, 21]. Accordingly, there is an urgent call for interventions to address these aspects to ensure the children’s optimal return to everyday life after treatment.
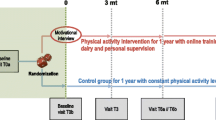
At diagnosis, we initiated a multimodal intervention entitled “Rehabilitation including Social and Physical Activity and Education in Children and Teenagers with Cancer” (RESPECT), which included hospital “co-admission” of healthy classmates as ambassadors to support the children with cancer and to promote the social network between hospital, school, and peers. This was combined with a supervised in-hospital physical activity program [22, 23]. The intervention was initiated at diagnosis to maintain both social relationships and physical functioning because ambassadors can increase the motivation of the child with cancer to engage in physical activity [24,25,26]. The overall aim of the RESPECT study is to facilitate children with cancer’s reentry into everyday life after treatment, including adequate physical performance. We have previously shown that this intervention can maintain the children’s cardiorespiratory fitness and physical function during the first six months of treatment, whereas children receiving usual care experienced a decline in cardiorespiratory fitness and physical function [27]. Therefore, the primary objective of the present study was to investigate the effects of a multimodal social and physical activity intervention on cardiorespiratory fitness, muscle strength, and physical function 1-year post-treatment when compared with both CCS controls and children not previously diagnosed with cancer.
Methods
Design and setting
This study is a multicenter, prospective, non-randomized controlled multicomponent study entitled “Rehabilitation including Social and Physical Activity and Education in Children and Teenagers with Cancer” (RESPECT) (Clinical Trial registration NCT01772849 and NCT01772862) and is part of the work of the Center for Integrated Rehabilitation (CIRE) [28].
Participants
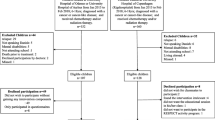
We included participants during January 2013–February 2018. Inclusion criteria were age 6–18 years; any cancer diagnosis or myelodysplastic syndrome (MDS) or Langerhans cell histiocytosis (LCH); treatment with chemotherapy and/or radiation therapy; enrolled in school at diagnosis; and able to communicate in Danish. Exclusion criteria were mental disability (e.g., Down syndrome) and severe co-morbidity. We included children treated at the University Hospital of Copenhagen, Rigshospitalet, in the intervention group, and children treated at Odense University Hospital and Aarhus University Hospital in the patient control group. The patient control group received standard institutional, guided care. We excluded participants if they had experienced a recurrence of their primary diagnosis or were diagnosed with a secondary cancer. Further, we included age- and sex-matched children without a cancer history and/or chemotherapy/radiation as a community control group. The community control group consisted of a subgroup of ambassadors (n = 64) and a subgroup of participants without a history of cancer from the Acute Lymphoblastic Leukemia Survivor Toxicity and Rehabilitation (ALL-STAR) study (n = 30) assessed at the University Hospital of Copenhagen [29]. Figure 1 shows the enrollment process.
Intervention components
The intervention consisted of three components. (1) We conducted a 90-min educational session for the child with cancer’s school class on cancer treatment and its side effects, everyday life at the hospital, supportive care, the benefits of physical activity, and the RESPECT study. (2) We selected two classmates as “ambassadors” in collaboration with the teachers, the classmates’ parents, and the child with cancer [30]. The ambassadors were invited to be co-admitted every 14th in- and outpatient day throughout the entire treatment period. The ambassadors were co-admitted to the hospital for the day (i.e., 9 a.m. to 3 p.m.) and were present during the daily routines at the department and participated in school, social, and physical activities. The primary role of the ambassadors was to provide peer support, maintain social inclusion, and increase the motivation of the child with cancer to engage in school and physical activities. The planning of an ambassador co-admission has been presented previously [27]. (3) We conducted an in-hospital supervised physical activity intervention (the RESPECT physical activity program) carried out during admissions to the Department of Pediatric Oncology. The RESPECT physical activity program consisted of individually designed activities (duration 5–30 min) offered three times per week (Monday, Wednesday, and Friday) and group sessions (duration 30–120 min) including all eligible children with cancer and their ambassadors on Tuesdays and Thursdays, as shown in Table 1. Daily, we designed each physical activity session to accommodate the wellbeing (e.g., presence of nausea, pain, and dizziness), training category (able to walk/not in isolation, able to walk/ in isolation, and bedbound), and physical capacity of the child with cancer (Table 1). We had not pre-defined a targeted intensity of the physical activity program before study initiation. The aim of the physical activity sessions was to mobilize the children and accomplish as high an intensity as possible on a given day. Each session started with cardiorespiratory fitness exercises spanning simple mobilization to targeted aerobic exercises (provided the child’s wellbeing permitted) followed by activities and/or games designed to improve muscle strength and balance [31]. Key equipment consisted of stationary cycle-ergometers, treadmills, dumbbells, balls, and various other items to facilitate games. We previously reported the intensity during group sessions elsewhere [6]. We measured the intensity of the individual and group physical activity program in a subgroup of CCS (n = 50) from September 2013 to September 2015. The mean heart rate was 145 beats/min [95% CI 142 to 149] or 69.3% [68.1 to 70.4%] of age-specific predicted maximal heart rate. The maximal heart rate was 185 beats/min [95% CI 174 to 184] or 89% [95% CI 87.7% to 90.4%] of age-specific predicted maximal heart rate [6]. Training frequency was calculated by dividing the number of days with physical activity by the number of weekdays admitted to the pediatric oncology department (excluding weekends and holidays).
Anthropometry, body composition, and medical characteristics
We weighed the participants to the nearest 0.1 kg and measured height to the nearest 0.1 cm. Body mass index (BMI) was calculated by dividing weight by height2.
Physical outcome evaluation
The primary outcome was VO2peak measured with the cardiopulmonary exercise test (CPET). The secondary outcomes were Sit-to-Stand, Timed Up and Go, and Handgrip Strength. We carried out the tests 1-year post-treatment ± 180 days. The treating physician permitted the tests providing the child’s thrombocyte count was > 10 billion/L, hemoglobin count was > 5 mmol/L, and the temperature was < 38°. Exclusion criteria (for testing) included active diarrhea, cough or a cold, and side effects preventing testing. We held annual meetings with all centers to ensure comparability, and we distributed instruction videos to all members of the test teams. The tests are described in detail elsewhere [22]. All children in the age- and sex-matched control group were tested at Copenhagen University Hospital, Rigshospitalet, using the same equipment as the intervention group.
Following a modified Godfrey protocol, we performed the CPET on an electronically braked cycle ergometer (Lode Corival Pediatric or Monark Ergomedic 839 E) [22, 32]. We determined breath-by-breath ventilation and gas exchange data (INNOCOR ergo-spirometry-system, INNO00010, Innovision, DK-5260 Odense, Denmark, or Jaeger Master Screen® CPX System (MS-CPX) and JLAB Software Package™). VO2peak was defined as the highest mean over 60 s and expressed in mL/kg/min. The maximal watt of the test was recorded. Heart rate and oxygen saturation were measured every 30 s (Polar FT2 sport tester Polar Electro, Kemple, Finland). Following consultation with experts on CPET testing in healthy children [33], we considered the CPET to be valid if one subjective criterion and two objective criteria were fulfilled. The subjective criteria were signs of intense effort. The objective criteria were heart rate > 180 beats/min and respiratory exchange ratio > 1.05 [33]. We stopped the test if oxygen saturation was under 90 or the child could not maintain the minimum required tempo (70 rpm).
Physical function tests
The children performed the Sit-to-Stand test [34] using a chair that allowed the child to flex the legs at a 90° angle. The child was instructed to fold his/her arms across the chest or to let them hang to the side, stand straight, and then touch the chair with their bottom while returning to a seated position. Strong verbal encouragement was given during the test. The test score equated the number of repetitions during a 30 second period.
The children performed the Timed Up and Go 3-m test [35] using a chair that allowed the child to flex the legs at a 90° angle. From the start position, with the back resting against the chair and arms on knees, we instructed the child to stand up, walk 3 m as fast as possible, turn around, and return to the start position. Completion time was recorded in seconds to the nearest two decimals. Strong verbal encouragement was given during the test. The last score of three tries was used in the analysis.
Handgrip Strength was measured using a Saehan hand dynamometer (Glanford Electronics, Scunthorpe, UK) and measured in kilograms. Two attempts per arm were performed either standing or sitting and without use of the elbow or the dynamometer touching anything. Strong verbal encouragement was given during the test and the highest score was used in the analysis [36].
Ethics approval and consent to participate
All participants and their parents gave written informed consent to participate in the educational sessions, to the inclusion of ambassadors, and to participation in the RESPECT activity program. The study was approved by the Regional Ethics Committee for the Capital Region (file. H 3-2012-105) and the Danish Data Protection Agency (file. 2007-58-0015/nr.30-0734) and complies with the Helsinki II Declaration.
Statistical method
The power calculation is based on the primary endpoint 1-year post-treatment being VO2peak, and the power calculation is based on an anticipated 10% higher VO2peak in the intervention group compared with the control group. We based the power calculation on a pilot that found a baseline VO2peak of 24.3 (SD 5.9) [37]. The significance level 1 year after treatment end was 0.025, and the power was 0.90, resulting in 120 children in each group of children with cancer [22].
We analyzed VO2peak (mL/kg/min), VO2peak (L/min), max watt, Sit-to-Stand, Timed Up and Go, and Handgrip Strength using analysis of covariance (ANCOVA) models with the residual variance depending on the group (intervention group, patient control group, and community control group). VO2peak (L/min), Sit-to-Stand, Timed Up and Go, and Handgrip Strength were log-transformed before analyses, and the back-transformed relative effects were presented as percentage difference to the reference level. To investigate whether the impact of adjusting for the differences between the three groups could result from differences in sex, age, cancer diagnosis, and time since diagnosis, the groups were compared in three different models: (1) a raw model without any adjustments, (2) a model adjusted for the sex-dependent effects of relative age differences (10% increase in age), and (3) a model further adjusted for cancer-type-dependent effects of time since diagnosis. We categorized the types of cancers in three groups: (1) hematological cancers receiving maintenance therapy (i.e., acute lymphoblastic leukemia (ALL), acute promyelocytic leukemia, t-cell non-Hodgkin’s lymphoma); (2) other hematological cancers (i.e., Hodgkin’s lymphoma, Burkitt non-Hodgkin lymphoma, acute myeloblastic leukemia, myelodysplastic syndrome, Langerhans cell histiocytosis and children with acute lymphoblastic leukemia who were treated with hematopoietic stem cell transplantation); and (3) other oncological diseases (extracranial solid tumors and tumors located in the central nervous system).
We categorized the types of cancers in these three groups based on two previous observations. Firstly, we previously showed that children with acute lymphoblastic leukemia responded differently to physical activity than other oncological diseases (extracranial solid tumors and tumors located in the central nervous system) but not between children with extracranial solid tumors and children with tumors located in the central nervous system [38]. Secondly, we decided to account for the time since the diagnosis, as length of treatment could affect the results. In addition, we evaluated whether the difference between the three groups (intervention group, patient control group, and community control group) depended on sex or age by adding two-factor interactions as well as three-factor interactions between group, sex, and relative age difference to Model 3. The three-factor interaction was insignificant for all outcomes (all p > 0.18), and the two-factor interaction between group and sex was insignificant for all outcomes (all p > 0.20). However, for some outcomes, the differences between the groups appeared to depend on age. Therefore, estimated group differences for age 8 years and age 18 years are presented for all outcomes. We performed all analyses in R (version 3.6.0) and R-studio.
Results
Participant characteristics
We included 120 of 128 (94%) eligible children in the intervention group and 58 of 107 (54%) eligible children in the control group. In the intervention group, two children did not wish classmates to participate, four children found the intervention irrelevant, one child did not wish the educational session in his/her class, and one child did not wish to participate in the RESPECTS activity program. In the patient control group, the children declined participation because they did not gain any intervention components. One-year post-treatment, 45 children had been excluded or had dropped out of the intervention group, and 17 had been excluded or had dropped out of the patient control group (Fig. 1). Thus, the intervention group consists of 75 CCS and the patient control group consisted of 33 CCS at 1-year post-treatment. We observed no difference between groups in age, sex, height, weight, BMI, or diagnosis distribution. Anthropometric and clinical characteristics are shown in Table 2. The treatment protocols of the included children are presented in Supplementary 1.
Training frequency, harms, and feasibility
The median number of physical activity sessions attended per child was 34 [interquartile range: 19 to 50], corresponding to a participation rate of median 64% [interquartile range 50 to 82%] or three sessions per 5 days of in-hospital admission (excluding weekends and holidays). Overall, the children’s participation was spread over a total of 3364 individual and 726 group physical activity sessions. No additional adverse events occurred during the physical activity sessions apart from the six minor events reported in earlier publications: four children experienced minor bruising, one child had a nosebleed during warm-up, and one child fainted shortly after exercise but had no further complications [22, 23].
Effect of the RESPECT activity program
One-year post-treatment, VO2peak tended to be higher in the intervention group compared with the patient control group with a mean difference of 4.7 mL/kg/min [95% CI 0.4 to 9.1 mL/kg/min]. This mean difference remained similar when we adjusted sex-dependent age and for diagnosis-dependent time since diagnosis (Table 3). Watt max and VO2 (L/min) during the CPET test were similar between the intervention- and the patient control group. The intervention group had a higher Sit-to-Stand score than that of the patient control group, with a mean difference of 7 repetitions [95% CI 4 to 10]. Moreover, the intervention group completed the Timed Up and Go test faster than the patient control group with a mean difference of − 20% [95% CI − 26 to − 13]. The intervention group was stronger in Handgrip Strength compared with the patient control group in both hands (see Table 3). In Sit-to-Stand, Timed Up and Go, and Handgrip Strength, the mean difference remained similar when we adjusted for sex-dependent age and for diagnosis-dependent time since diagnosis (Table 3). The mean and standard deviations and median and 10th to 90th percentile of cardiorespiratory fitness, muscle strength and physical function are presented in supplementary 2.
Cardiorespiratory fitness, muscle strength, and physical function compared with age- and sex-matched community control group
One-year post-treatment, both the intervention- and patient control group had lower cardiorespiratory fitness than the community control group (mean difference − 4.7 [95% CI − 7.1 to − 4.7]) (Table 4). The intervention group and community control group performed similarly in Sit-to-Stand (mean difference 0 repetitions [95% CI − 2 to 2]), Timed Up and Go (mean difference − 3% [95% CI − 7 to 1]), and Handgrip Strength (right hand: mean difference − 4% [95% CI − 17 to 10], left hand: mean difference − 5% [95% CI − 18 to 10]). However, the patient control group had a lower Sit-to-Stand score (mean difference − 6.29 [− 9.29 to -3.27] repetitions), was slower to complete the Timed Up and Go test (mean 21 [12 to 32] %) and had lower Handgrip Strength (right hand: mean difference − 23 [− 41 to − 1] %, left hand: − 24 [− 40 to − 4] %) than did the community control group (Table 4). The mean differences remained similar when we adjust for sex-dependent age (Table 4).
Discussion
In this multicenter, prospective, non-randomized, controlled, multimodal study, we showed that children who received a peer-supported, supervised in-hospital physical activity program during treatment had higher cardiorespiratory fitness, muscle strength, and physical function than children who received usual care 1-year after ended treatment. Moreover, we showed that children with cancer in the intervention group had similar muscle strength and physical function to children with no history of cancer (i.e., community controls); however, the children with cancer still displayed lower cardiorespiratory fitness than community controls. Our previous study showed that the intervention and patient control groups were comparable in anthropometric and diagnosis distribution prior to inclusion [27]. Our study suggested that children in the intervention group could maintain their cardiorespiratory fitness, muscle strength, and physical function during treatment. In contrast, the control group experienced a further decline in cardiorespiratory fitness [27]. Furthermore, the intervention group and patient control group were comparable in cardiorespiratory fitness and handgrip strength but not in Sit-To-Stand and Timed-Up-and-Go at baseline (Table 2). Collectively, this suggests that a peer-supported, supervised in-hospital physical activity program during treatment may have long-lasting benefits for CCS regarding cardiorespiratory fitness, muscle strength, and physical function. However, the observed differences between the intervention- and patient control groups in physical function may be due to baseline differences. This indicate that children with cancer could benefit from early in-hospital physical activity programs, also in their everyday life after treatment.
It is possible that the improved physical function supported the children’s educational and social rehabilitation as they may have fewer difficulties in matching the physical functioning of peers [39]. These results suggest that physically active children during treatment require less rehabilitation post-treatment to regain age-matched physical function but still require targeted interventions to improve their cardiorespiratory fitness. Conflicting evidence on the effects of physical activity during treatment exists. Several studies show benefits for cardiorespiratory fitness [23, 40,41,42], muscle strength [43,44,45], and physical function [46, 47], whereas others show no effect [48, 49]. Collectively, data have been synthesized in two meta-analyses, showing that physical activity during treatment can improve muscle strength [50] and physical function [51]. In agreement with the present study, CCS have lower cardiorespiratory fitness and muscle strength several years post-treatment [8, 9, 11, 52]. Factors that can contribute to lower cardiorespiratory fitness and muscle strength include cardiac, pulmonary, and vascular limitations, as well as peripheral neuropathy and altered body composition [9, 52]. The present study showed that physical activity during treatment has both an immediate effect and a long-term effect manifesting a year after intervention end. The effects of the intervention might reduce the children’s risk of developing cardiorespiratory fitness-related medical conditions for years after their treatment has ended. In adults, studies have shown that a change in VO2peak of 1 mL/kg/min corresponds to a 9–10% reduction in the incidence of cardiac mortality [53, 54] and a 5% cardiovascular disease risk reduction [55]. This is further supported by a recent study showing that exercise during childhood cancer treatment maintained left ventricular function post-treatment, whereas this was not the case in a control group with no exercise [56].
The RESPECT project is the first to include healthy classmates as ambassadors during cancer treatment and in a physical activity program [22]. Through semi-structured interviews, we previously explored children with cancer’s motivation to engage in physical activity while admitted to the hospital [57, 58]. The children with cancer described how their motivation to be physically active increased during treatment because their ambassadors participated in the physical activity sessions [57, 58]: the ambassadors’ presence provided distractions from common side effects (i.e., nausea, pain) and everyday hospital life, motivating them to get out of bed. Qualitatively, parents and children have described how the intervention supported the children in re-entering everyday life post-treatment, including physical activities, social interactions, and school attendance [57, 58]. The ambassadors provided an opportunity to receive support from peers when performing physical activities [57]. Moreover, they represented a unique opportunity to incorporate the child’s everyday life into the hospital setting and increase the child’s willingness to engage in rehabilitation offers [57]. However, involving healthy classmates as ambassadors may be more difficult in other settings. Thus, exploring alternative approaches to including healthy children in physical activity programs for children with cancer is critical. Through semi-structured interviews, we previously investigated the experience of being part of the RESPECT study [58]. The parents described how participating in the RESPECT intervention increased their understanding of how anti-cancer treatment and sedentary behavior affected their child’s physical capacity [58]. They expressed that they learnt the importance of physical activity both during and after treatment and that this enabled them to support their child’s physical activity post-treatment [58]. Throughout the study, one exercise professional or physical therapist conducted the physical activity program at a given time. Thus, achieving the physical activity program in other settings requires few additional human resources.
Taken together, the findings show that children with cancer need physical rehabilitation. Without physical rehabilitation the children risk long-term impairments in cardiorespiratory fitness, muscle strength, and physical function. Further, this study indicates that physical activity is beneficial for children with cancer, thus supporting the recommendations from the international Pediatric Oncology Exercise Guidelines (iPOEG) stating that children with cancer should be physically active and do what they can, when they can [59]. Building on the iPOEG guidelines, we recommend that clinicians emphasize physical activity during treatment, when side effects (i.e., nausea, pain) are most common. Targeted exercise interventions including cardiorespiratory fitness may be more suitable later in the treatment trajectory when treatment is less intense (e.g., maintenance phase of ALL treatment) or after treatment end. This remains to be investigated.
Strengths and limitations
The strength of this study is the high inclusion rate in the intervention group, with 94% of eligible children completing the intervention. Prior to the initiation of the RESPECT study, we expected some selection bias, given the study design. However, the limited participation rate in the control group (47%) introduced the possibility of further selection bias. The number of dropouts and excluded patients at 1-year post-treatment is a limitation of the study. We, therefore, suspected attrition bias because of poor retention one-year post-treatment. Thus, we performed post hoc analyses and tested for systematic dropouts or excluded patients concerning diagnosis distribution, sex, age, socioeconomic status, and ethnicity. Further, we tested whether these variables were comparable at baseline between the intervention and patient control groups. None of these variables were associated with dropout or exclusion in this study. No differences were observed in any of these variables at baseline or one-year post treatment. We observed more relapse cases in the intervention group compared to the patient control group. We expected this as the treatment of most rare and high-risk cancers is centralized at The University Hospital of Copenhagen. This discrepancy in eligible patients between the centers, combined with the high number of non-responders in the control group, limits the generalizability and certainty of the study results.
There is a possible geographical difference between The University Hospital of Copenhagen and the rest of the country concerning the testing personnel and differences in standard institutional guided care unrelated to treatment protocols. Nevertheless, all institutions are subject to the same regulations and have the same financial resources available to treat children. We included all children who received chemotherapy and/or radiation therapy. Consequently, the study consists of a heterogeneous group; therefore, we cannot conclude on the effects of the intervention for children with a specific diagnosis. Parents declining participation in the patient control group explained that the requirements to their children were excessive, as there were no benefits from participating in the study (i.e., no physical activity or ambassador visits). It can be speculated that the children in the patient control group consisted of children with an interest in exercise, consequently resulting in an underestimation of the effects.
Moreover, it can be speculated that children with the most severe long-term adverse effects declined participation or dropped out of the study, limiting the generalizability and certainty of the study results. Further, the study is limited by the few completed CPET. The missing data indicate that the children with the best physical capacity completed the CPET, thus limiting the generalizability and certainty of the effects of the intervention.
Conclusion
This study indicates that a peer-supported and supervised in-hospital physical activity intervention initiated from diagnosis may be beneficial on cardiorespiratory fitness and muscle strength in children with cancer post-treatment. The study also indicates that physical activity during treatment may improve muscle strength and physical function to a level similar to that of children without a history of cancer, although cardiorespiratory fitness requires a more targeted approach. However, the results should be interpreted with caution because of the limitations present in the study. Overall, improved physical function might not only improve the children’s long-term physical performance but may also be a core element in their social and educational rehabilitation.
Data availability
The datasets used during the current study are available from the corresponding author on reasonable request after the last follow-up data have been collected and published.
Abbreviations
- RESPECT:
-
Rehabilitation including Social and Physical Activity and Education in Children and Teenagers with Cancer
- LCH:
-
Langerhans cell histiocytosis
- MDS:
-
Myelodysplastic syndrome
- CPET:
-
Cardiopulmonary exercise test
- CI:
-
Confidence interval
- SD:
-
Standard deviation
References
Armstrong GT, et al. Aging and risk of severe, disabling, life-threatening, and fatal events in the childhood cancer survivor study. J Clin Oncol. 2014;32(12):1218–27.
Hudson MM, et al. Clinical ascertainment of health outcomes among adults treated for childhood cancer. JAMA. 2013;309(22):2371–81.
Collins DE, et al. A systematic review summarizing the state of evidence on bullying in childhood cancer patients/survivors [formula: see text]. J Pediatr Oncol Nurs. 2019;36(1):55–68.
Oeffinger KC, et al. Chronic health conditions in adult survivors of childhood cancer. N Engl J Med. 2006;355(15):1572–82.
Vannatta K, et al. Intensity of CNS treatment for pediatric cancer: prediction of social outcomes in survivors. Pediatr Blood Cancer. 2007;49(5):716–22.
Thorsteinsson T, et al. Cardiorespiratory fitness and physical function in children with cancer from diagnosis throughout treatment. BMJ Open Sport Exerc Med. 2017;3(1):e000179.
Yildiz Kabak V, et al. Short and long-term impairments of cardiopulmonary fitness level in previous childhood cancer cases: a systematic review. Support Care Cancer. 2019;27(1):69–86.
Fridh MK, et al. Cardiorespiratory fitness and physical performance after childhood hematopoietic stem cell transplantation: a systematic review and meta-analysis. Bone Marrow Transplant. 2021;56(9):2063–78.
Berkman AM, Lakoski SG. A review of cardiorespiratory fitness in adolescent and young adult survivors of childhood cancer: factors that affect its decline and opportunities for intervention. J Adolesc Young Adult Oncol. 2016;5(1):8–15.
Ness KK, et al. Exercise intolerance, mortality, and organ system impairment in adult survivors of childhood cancer. J Clin Oncol. 2020;38(1):29–42.
Sontgerath R, Eckert K. Impairments of lower extremity muscle strength and balance in childhood cancer patients and survivors: a Systematic review. Pediatr Hematol Oncol. 2015;32(8):585–612.
Ness KK, et al. Body composition, muscle strength deficits and mobility limitations in adult survivors of childhood acute lymphoblastic leukemia. Pediatr Blood Cancer. 2007;49(7):975–81.
Ness KK, et al. Physical performance limitations in the childhood cancer survivor study cohort. J Clin Oncol. 2009;27(14):2382–9.
Gilchrist L, Tanner L. Gait patterns in children with cancer and vincristine neuropathy. Pediatr Phys Ther. 2016;28(1):16–22.
Ness KK, et al. Limitations on physical performance and daily activities among long-term survivors of childhood cancer. Ann Intern Med. 2005;143(9):639–47.
McLoone JK, Wakefield CE, Cohn RJ. Childhood cancer survivors’ school (re)entry: Australian parents’ perceptions. Eur J Cancer Care (Engl). 2013;22(4):484–92.
Gurney JG, et al. Social outcomes in the childhood cancer survivor study cohort. J Clin Oncol. 2009;27(14):2390–5.
Storch EA, et al. Peer victimization, psychosocial adjustment, and physical activity in overweight and at-risk-for-overweight youth. J Pediatr Psychol. 2007;32(1):80–9.
Gray WN, et al. The impact of peer victimization, parent distress and child depression on barrier formation and physical activity in overweight youth. J Dev Behav Pediatr. 2008;29(1):26–33.
Anthony SJ, et al. Quality of life of pediatric oncology patients: do patient-reported outcome instruments measure what matters to patients? Qual Life Res. 2017;26(2):273–81.
Deisenroth A, et al. Muscle strength and quality of life in patients with childhood cancer at early phase of primary treatment. Pediatr Hematol Oncol. 2016;33(6):393–407.
Thorsteinsson T, et al. Study protocol: Rehabilitation including Social and Physical activity and Education in Children and Teenagers with Cancer (RESPECT). BMC Cancer. 2013;13:544.
Nielsen MKF. Supervised in-hospital physical activity that includes healthy classmates as ambassadors in children and adolescent with cancer - a RESPECT study, in Graduate school of health and medical sciences. University of Copenhagen; 2020.
Salvy SJ, et al. Effect of peers and friends on youth physical activity and motivation to be physically active. J Pediatr Psychol. 2009;34(2):217–25.
Cox AE, Ullrich-French S. The motivational relevance of peer and teacher relationship profiles in physical education. Psychol Sport Exerc. 2010;11(5):337–44.
Fitzgerald A, Fitzgerald N, Aherne C. Do peers matter? A review of peer and/or friends’ influence on physical activity among American adolescents. J Adolesc. 2012;35(4):941–58.
Nielsen MKF, et al. Effects of a physical activity program from diagnosis on cardiorespiratory fitness in children with cancer: a national non-randomized controlled trial. BMC Med. 2020;18(1):175.
Adamsen L et al. Exercise in cancer survivors CIRE- Horizon 2020. https://www.cancer.dk/dyn/resources/File/file/5/6655/1505472246/exercise-in-cancer-survivors.pdf.
Andres-Jensen L, et al. National, clinical cohort study of late effects among survivors of acute lymphoblastic leukaemia: the ALL-STAR study protocol. BMJ Open. 2021;11(2):e045543.
Lindgren LH, et al. In sickness and in health: classmates are highly motivated to provide in-hospital support during childhood cancer therapy. Psychooncology. 2017;26(1):37–43.
Thorsteinsson T, et al. Cardiorespiratory fitness and physical function in children with cancer from diagnosis throughout treatment. BMJ Open Sport Exerc Med. 2017;3(1):e000179.
Shephard RJ, et al. The maximum oxygen intake. An international reference standard of cardiorespiratory fitness. Bull World Health Organ. 1968;38(5):757–64.
Steene-Johannessen J, et al. Cardiovascular disease risk factors in a population-based sample of Norwegian children and adolescents. Scand J Clin Lab Invest. 2009;69(3):380–6.
Bohannon RW. Sit-to-stand test for measuring performance of lower extremity muscles. Percept Mot Skills. 1995;80(1):163–6.
Podsiadlo D, Richardson S. The timed “Up & Go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39(2):142–8.
Abizanda P, et al. Validity and usefulness of hand-held dynamometry for measuring muscle strength in community-dwelling older persons. Arch Gerontol Geriatr. 2012;54(1):21–7.
San Juan AF, et al. Effects of an intrahospital exercise program intervention for children with leukemia. Med Sci Sports Exerc. 2007;39(1):13–21.
Nielsen MKF, et al. Testing physical function in children undergoing intense cancer treatment-a RESPECT feasibility study. Pediatr Blood Cancer. 2018;65(8):e27100.
Ingersgaard MV, et al. A qualitative study of adolescent cancer survivors perspectives on social support from healthy peers - a RESPECT study. J Adv Nurs. 2021;77(4):1911–20.
Saultier P, et al. A randomized trial of physical activity in children and adolescents with cancer. Cancers (Basel). 2021;13(1). https://doi.org/10.3390/cancers13010121.
Stossel S, et al. Benefits of exercise training for children and adolescents undergoing cancer treatment: results from the randomized controlled MUCKI trial. Front Pediatr. 2020;8:243.
Moyer-Mileur LJ, Ransdell L, Bruggers CS. Fitness of children with standard-risk acute lymphoblastic leukemia during maintenance therapy: response to a home-based exercise and nutrition program. J Pediatr Hematol Oncol. 2009;31(4):259–66.
Fiuza-Luces C, et al. Exercise intervention in pediatric patients with solid tumors: the physical activity in pediatric cancer trial. Med Sci Sports Exerc. 2017;49(2):223–30.
Braam KI, et al. Cost-effectiveness of a combined physical exercise and psychosocial training intervention for children with cancer: Results from the quality of life in motion stud. Eur J Cancer Care (Engl). 2017;26(6). https://doi.org/10.1111/ecc.12586.
Marchese VG, Chiarello LA, Lange BJ. Effects of physical therapy intervention for children with acute lymphoblastic leukemia. Pediatr Blood Cancer. 2004;42(2):127–33.
Lanfranconi F, et al. Precision-based exercise as a new therapeutic option for children and adolescents with haematological malignancies. Sci Rep. 2020;10(1):12892.
Tanir MK, Kuguoglu S. Impact of exercise on lower activity levels in children with acute lymphoblastic leukemia: a randomized controlled trial from Turkey. Rehabil Nurs. 2013;38(1):48–59.
Cox CL, et al. Modifying bone mineral density, physical function, and quality of life in children with acute lymphoblastic leukemia. Pediatr Blood Cancer. 2018;65(4). https://doi.org/10.1002/pbc.26929.
Braam KI, et al. Effects of a combined physical and psychosocial training for children with cancer: a randomized controlled trial. BMC Cancer. 2018;18(1):1289.
Shi Q, Zheng J, Liu K. Supervised exercise interventions in childhood cancer survivors: A systematic review and meta-analysis of randomized controlled trials. Children (Basel). 2022;9(6):824. https://doi.org/10.3390/children9060824.
Morales JS, et al. Exercise training in childhood cancer: a systematic review and meta-analysis of randomized controlled trials. Cancer Treat Rev. 2018;70:154–67.
Goodenough CG, Partin RE, Ness KK. Skeletal muscle and childhood cancer: where are we now and where we go from here. Aging Cancer. 2021;2(1-2):13–35.
Kavanagh T, et al. Prediction of long-term prognosis in 12 169 men referred for cardiac rehabilitation. Circulation. 2002;106(6):666–71.
Kavanagh T, et al. Peak oxygen intake and cardiac mortality in women referred for cardiac rehabilitation. J Am Coll Cardiol. 2003;42(12):2139–43.
Kodama S, et al. Cardiorespiratory fitness as a quantitative predictor of all-cause mortality and cardiovascular events in healthy men and women: a meta-analysis. JAMA. 2009;301(19):2024–35.
Morales JS, et al. Inhospital exercise benefits in childhood cancer: A prospective cohort study. Scand J Med Sci Sports. 2020;30(1):126–34. https://doi.org/10.1111/sms.13545.
Thorsteinsson T, et al. Classmates motivate childhood cancer patients to participate in physical activity during treatment: a qualitative study. Eur J Cancer Care (Engl). 2019;28(5):e13121.
Petersen NN, et al. Childhood cancer survivors’ and their parents’ experiences with participation in a physical and social intervention during cancer treatment: a RESPECT study. J Adv Nurs. 2022;78(11):3806–16.
Wurz A, et al. The international pediatric oncology exercise guidelines (iPOEG). Transl Behav Med. 2021;11(10):1915–22.
Acknowledgements
We thank all children and their parents for participating in the study. We also thank the health personnel at the Copenhagen University Hospital, Rigshospitalet, Odense University Hospital, and Aarhus University Hospital for their help. This work is part of the Childhood Oncology Network Targeting Research, Organization & Life expectancy (CONTROL), supported by the Danish Cancer Society (R-257-A14720) and the Danish Childhood Cancer Foundation (2019-5934). Furthermore, we thank Lis Adamsen for her tremendous support and insightful feedback throughout the study.
Funding
Open access funding provided by Royal Library, Copenhagen University Library. This study received funding from the Novo Nordic Foundation, the Danish Child Cancer Foundation, Trygfonden, the Danish Cancer Society, Lundbeckfonden, Arvid Nilssons Foundation, Aase, and Ejnar Danielsen’s Foundation, Familien Hede Nielsens Foundations, ML Jørgensen & Gunnar Hansen Foundation, and the Toyota Foundation.
Author information
Authors and Affiliations
Contributions
HBL and KS designed the RESPECT study. MKF and HBL designed the current study. HBL, TT, PSA, and MKF were responsible for the data collection in the intervention group. HBL, HH, and PSW were responsible for the data collection in the control group. MKF, PSA, and LAJ were responsible for the data from the healthy age- and sex-matched control group. MKF, PSA, and TT carried out the intervention and performed all physical tests in the intervention group. MKF performed all statistical analyses. MKF wrote the first draft of the manuscript. All authors contributed to the final draft of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
All participants and their parents received oral and written information, and the parents gave written informed consent. The Regional Ethics Committee for the Capital Region (file. H 3-2012-105) and the Danish Data Protection Agency (file. 2007-58-0015/nr.30-0734) approved the study and the data protection structure.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fridh, M.K., Schmidt-Andersen, P., Andrés-Jensen, L. et al. Children with cancer and their cardiorespiratory fitness and physical function—the long-term effects of a physical activity program during treatment: a multicenter non-randomized controlled trial. J Cancer Surviv (2023). https://doi.org/10.1007/s11764-023-01499-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11764-023-01499-7