Abstract
Purpose
Oral endocrine therapy (OET) is recommended in prevention and treatment of hormone receptor-positive breast cancer (HR+ BC). Despite the reduced incidence, recurrence, and mortality, OET adherence is poor in this patient population. The aim of this study was to review the latest literature to identify effective interventions to improve medication adherence in patients taking OET for prevention or treatment of HR+ BC.
Methods
The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) framework was used to perform this review. We utilized PubMed, SCOPUS, EMBASE, Cochrane, and Web of Science to acquire articles using search terms including breast cancer, adherence, persistence, and acceptability. Inclusion criteria included publication in peer-reviewed journal, primary data source, longitudinal, patients on OET such as aromatase inhibitors (AIs) or selective estrogen receptor modulators (SERMs), measuring adherence, persistence, or acceptability.
Results
Out of 895 articles identified, 10 articles were included. Majority of patients had early-stage HR+ BC. Two out of two studies incorporating technological intervention, two out of three studies with text communication-based intervention, and three out of five studies with verbal communication-based intervention reported significant improvement in OET adherence and/or persistence.
Conclusions
While the interventions tested so far have shown to improve OET adherence in HR+ BC patients in some studies, there is a need to design combination interventions addressing multiple barriers in this population.
Implications for Cancer Survivors
This study showcases effectiveness of novel interventions to improve OET adherence and the need to further develop patient-centered strategies to benefit all patients with HR+ BC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Worldwide, 2.26 million women were newly diagnosed with breast cancer (BC) in 2020 making BC the most common type of cancer in women globally [1, 2]. It is also the second most common type of cancer in the United States (U.S.). Compared to lung and colorectal cancers, BC has a higher overall survival rate due to the availability of screening programs for early detection and effective prevention and treatment options [3]. Oral endocrine therapy (OET) is the standard mode of treatment in patients with hormone receptor-positive (HR+) BC, comprising of 70–80% of all BCs [4]. Common OETs include selective estrogen receptor modulators (SERMs) such as tamoxifen, or aromatase inhibitors (AIs) such as anastrozole, letrozole, and exemestane [5]. Five years of therapy with tamoxifen can reduce 15-year risks of BC recurrence by as much as 40% and mortality by 30% [6]. In general, AIs have demonstrated better recurrence and survival benefits than tamoxifen [7]. Ten years of adjuvant OET has been linked to superior outcomes than 5 years of therapy and is encouraged by clinicians in at least some patients [8,9,10]. Even in the prevention settings, OETs have indicated a consistent reduction in the risk of developing HR+ BC in high-risk women [11,12,13,14].
Despite the clinical benefit of OET, numerous studies have continuously reported less-than-ideal adherence rates (typically set at 80%) in patients in the real-world setting. In 2010, a cohort study reported that only 49% of U.S. patients were adherent to OET during the 5-year treatment period [15]. In 2012, a systematic review of 29 studies found prevalence of OET adherence ranged from 41 to 72% with discontinuation rates anywhere from 31 to 73% [16]. In 2021, a retrospective cross-sectional Surveillance, Epidemiology, and End Results (SEER) database study indicated an average 1-year adherence rate of 87% with a 5-year adherence rate dropping to 65.2% [17]. The practice of prescribing preventive therapy is also still far from ideal with utilization in 2013 only at 14.7% [18].
Non-adherence to OET is affected by many patient-, treatment-, or healthcare system–related factors [4, 19]. While there is an abundance of evidence identifying barriers to OET adherence, effective interventions to improve OET adherence are lacking. Our earlier systematic review looking at interventions to improve adherence in BC patients on OET published in 2018 found no effective intervention strategy [20]. As the number of BC patients continues to rise worldwide, there is a dire need for successful interventions to improve OET adherence. The aim of this review is to evaluate the latest studies looking at interventions to improve OET adherence, which is linked to improved clinical outcomes in HR+ BC patients.
Methods
The structure of this systematic review was founded on the PRISMA 2020 Systematic Review Checklist [21]. The primary research question was created based on the PICO method (population, intervention, comparator, and outcomes). The population of interest included patients on OET for BC prevention or treatment. The intervention was defined as any intervention not limited to education, counseling, technology, communication, etc. The comparator was standard of care or no active intervention. The outcomes evaluated were any measure of adherence, persistence, and acceptability to OET between the intervention and comparator.
Search strategy
The literature search and evidence extraction were performed by three researchers. The database used to identify articles was PubMed, SCOPUS, EMBASE, Cochrane, and Web of Science. Following the PICO format, the final search strategy used search terms based on the primary research question which is provided in Supplementary Table 1. To update our findings published earlier [20], the search was restricted from July 1, 2017, to July 31, 2023.
Data collection and selection process
Once the search strategy yielded the final list of articles, a review of titles and abstracts was conducted followed by a full-text review to obtain a final compilation of articles to synthesize the resulting evidence. An electronic review tool, Rayyan.ai software [22], was used to curate evidence. A brief orientation to the electronic tool was carried out to ensure all researchers could use the electronic review tool by all those involved.
Study inclusion and exclusion criteria
The curation of evidence at the title and abstract level along with the full-text review was carried out in accordance with the following inclusion and exclusion criteria. Inclusion criteria were (1) articles in English; (2) publication in a peer-reviewed journal; (3) utilization of primary data; (4) prospective and longitudinal study design; (5) inclusion of patients prescribed or initiated on OET including SERMs such as tamoxifen, raloxifene, or AIs such as anastrozole, letrozole, or exemestane for BC prevention or treatment; (6) the outcome measures including adherence, persistence, or acceptability; and (7) active interventions. Exclusion criteria were (1) non-randomized clinical trials, (2) studies without a comparator, and (3) publication types including reviews, protocols, editorials, or commentaries.
Quality and risk of bias assessment of selected studies
Qualitative analysis of the study design was performed using the modified Downs and Black 27-item methodological scale with a maximum possible score of 28 points for randomized and non-randomized health interventions as done in other studies [23,24,25]. Each article was scored based on “poor” (≤ 14) or “appropriate” (15–28 points). Two investigators ranked the studies, and those scoring below 15, or as “poor,” were excluded from the final analysis.
Outcome measures
Characteristics of included studies and patients were summarized descriptively including sample size, age, race/ethnicity, OET medications prescribed, tumor stage of study subjects, and medication/surgical history. Data collected from studies included types of interventions, specific interventions, and study results. Primary outcome measures included adherence, persistence, and acceptability to OET. Adherence was evaluated as the degree to which patients adhere to their recommended OET treatment plans. Various assessment methods included self-reported measures, electronic monitoring devices, pill counts, and pharmacy refill records. Persistence was measured as the duration for which patients continuously adhered to their OET treatment plans over time. Acceptability was measured as patients’ willingness or openness to adopt and engage with the intervention. Secondary outcomes included any other measure related to OET adherence, persistence, or acceptability. Each of the above outcome measures was grouped and reported by technological interventions, text communications, or verbal communications.
Results
Study selection
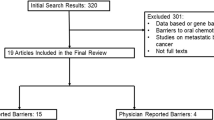
Figure 1 shows the systematic process of retrieving, screening, and the final selection of the articles based on the search strategy. Of the original 895 articles collected, 282 articles were removed as being duplicates resulting in an overall 613 articles, which underwent title and abstract screening. A total of 587 articles did not meet the inclusion criteria and were excluded. Of the 26 full-text articles reviewed, 16 were excluded because they either were non-randomized controlled trials (RCTs) (n = 12), had no comparator (n = 2), or were an abstract or protocol for a potential study (n = 1). Finally, 10 articles were included in the final synthesis of this review, all of which also met the appropriate Downs and Black scoring criteria.
Risk of bias in studies
Regarding the Downs and Black’s scoring for potential risk of bias, the studies varied widely in methodological quality; all the results meeting inclusion and exclusion criteria had an overall score ranging from 18 to 25 with a median score of 21. The median score for reporting bias was 9 with a range from 7 to 10. External validity had a median of 3 with no range. External validity had a median of 4 with a range of 3 to 5. Selection bias had a median of 4 with a range from 3 to 6. Power was only captured in four studies. All 10 studies met the appropriate Downs and Black scoring criteria (Table 1).
Study characteristics
The study and patient characteristics are summarized in Table 2. Of the 10 RCTs included, six studies included patients on both AIs and SERMs, one study included patients on only SERMs, and three studies included patients on only AIs. Five studies were single-centered [27, 29, 32, 34, 35], while five were multi-centered studies [28, 30, 31, 33, 34]. Five studies were conducted in the U.S. [27, 30,31,32, 34], and five were conducted internationally: Australia [29], Denmark [35], Ethiopia [33], Singapore [28], and South Korea [26]. Patient enrollment ranged from the smallest study enrolling 44 subjects [27] to the largest study enrolling 702 subjects [30]. The duration of intervention ranged from 4 weeks to 8 years among the studies measuring adherence, persistence, and acceptability. Most patients included in these studies were Non-Hispanic White and were diagnosed with stage I or II BC. As summarized in Table 3, two studies used technology-based interventions [26, 27], three used text-based communication [28,29,30], and five used verbal communication [31,32,33,34,35]. While all studies reported adherence as one of the outcomes, one study reported persistence [33], and only four studies reported acceptability of the intervention(s) [28, 31, 34, 35].
Interventions
Technological interventions
In the two studies utilizing technological interventions, one included smart pill bottles with the Pillsy mobile application [26] while the other utilized provision of information such as weekly adherence reminders through a mobile application [27]. In both studies, technological interventions significantly improved adherence in comparison to no intervention in the control group (Table 3). However, self-efficacy was not significantly higher with the Smart pill bottle used for 4 weeks in 61 patients. Mobile app weekly reminders for 8 weeks had significantly higher usage among patients in the intervention compared to the control group without significant improvement in symptom burden in 44 patients.
Text communications
In the studies utilizing text communication, the studies included personalized or standardized unidirectional text reminders to the patient’s designated mobile device with information regarding adherence sent by a nurse or through an automated system at scheduled intervals [28,29,30]. Two out of three studies reported significant improvement (Table 3). SMS reminder texts with notifications to take OET significantly improved adherence at 1 year in 244 patients in one study [28] with the majority of patients also thought that the intervention was easy to understand (99%), useful (79%), and provided enough information (97%). Additionally, 86% recommended it as a part of routine care [28]. The use of text reminders providing health education also significantly reduced the probability of missing more than one dose of OET as scheduled after 6 months in 160 patients in another study [29]. Self-efficacy and quality of life were largely indifferent between control and intervention arms. However, in a larger study with 702 patients, text messages sent twice a week focusing on barriers to adherence, cues to action, efficacy, and reminders were not able to significantly reduce adherence failure rates at 3 years [30].
Verbal communications
Studies employing verbal communication as the intervention included any in-person discussion over various topics regarding the patient medication regimen between a healthcare provider and patient in various settings of which three studies reported significant improvement (Table 3) [31,32,33,34,35]. The Resources and Education for Adherence to Cancer Hormonal therapy (REACH) intervention combined with education significantly improved adherence compared to education alone after 1 month of follow-up in 88 patients [31]. While the REACH + education cohort continued to demonstrate better adherence until month 4 of follow-up, the difference was not statistically significant at the later time point. In addition, lower negative attitudes were observed in the REACH + education arm for the first 3 months. In another study, relaxation training significantly reduced the likelihood of forgetfulness and intentional nonadherence compared to cognitive behavioral therapy (CBT) or health education in 59 patients [32]. Nurse-led counseling sessions in addition with phone call reminders and medication monitoring significantly improved adherence and persistence in 87 patients in another study [33]. The symptom-targeted randomized intervention for distress and adherence to adjuvant endocrine therapy (STRIDE) intervention using 6 weekly video conferencing sessions with 2 individual calls significantly improved symptom distress without any effect on OET adherence or satisfaction among 100 patients [34]. However, patient-initiated follow-up care was also ineffective in improving adherence, satisfaction, or QoL in 134 patients [35].
Discussion
This review highlights current studies that have aimed to evaluate various types of interventions to improve OET adherence, persistence, and acceptability in HR+ BC patients. Our current review found seven of the 10 studies provided a significant improvement in the primary outcome of adherence, while the remaining 3 either indicated a trend towards improvement or improvement in other outcomes. Both studies evaluating technological interventions involving mobile application-based reminders significantly improved OET adherence. Two studies with text communication-based interventions showed significant improvement in OET adherence at 6 or 12 months, whereas one study measuring adherence at 3 years did not. Three of five verbal communication-based intervention significantly improved OET adherence or persistence.
In our previous review in 2018, we reviewed 5 studies, none of which reported a significant improvement in OET adherence with educational material as an intervention in HR+ BC patients [20]. Since then, recent literature has considered advances in technology and incorporated other methodologies that also include bidirectional communication with health care providers to improve adherence barriers. Bidirectional social support has reported higher levels of acceptability and educational, physical, and emotional benefits in a qualitative semi-structured interview of BC patients on OET for 12 months across three states in the USA [36]. In accordance, future research could investigate exploring interventions that combine the aspects of technology, text, or verbal interventions to address multi-factorial barriers. For example, technology and text messaging can help address forgetfulness, whereas both text and verbal communications can improve patient-clinician relationship and provide education on importance of OET. Frequent check-ins may also be required to ensure the effectiveness of these intervention over time. The customized intervention should also have the feature of flexibility to address the changing needs of patients. Hence, a structured plan that incorporates various methods, check-ins, and adaptability to modify the interventions is necessary to improve OET adherence, persistence, and acceptance in HR+ BC patients.
While most studies have focused on improving adherence to OET in treatment of HR+ BC patients, an emphasis on OET adherence in the prevention setting for patients who are at higher risk of developing BC is lacking. Literature has shown preventative measures are able to reduce the burden on the patient and healthcare system clinically and economically when compared between patient cases diagnosed at early and advanced stages [37]. However, utilization of OET as prevention strategy is largely underutilized to its full capacity for a variety of reasons including, but not limited to, a lack of physicians’ and patients’ knowledge of available cancer risk evaluation tools or prevention measures, and underestimation of the benefits complemented with the overestimation of risk of taking the medications [37, 38]. Even among the patients taking OET for BC prevention, the adherence is poor and need to be improved to help reduce the overall incidence and severity of BC in the chronic setting [37, 39].
Potential limitations of the studies included here are as follows: first, most studies had a relatively short follow-up period of 1 year or less. OET is typically given to patients anywhere from 5 to 10 years [9]. While longer follow-up periods should provide more information regarding the efficacy of the intervention, it is also likely after time a single intervention alone is no longer efficacious. For example, one study in our review with a follow-up of 3 years did not significantly improve OET adherence [30]. Potentially multiple interventions are needed to maintain adherence superiority at longer follow-up periods. In addition, apart from one study in this review measuring true adherence with devices such as MEMS Caps, all studies measured adherence indirectly. This can be potentially problematic given that adherence may be over-estimated with the use of patient-reported adherence, rating scales and surveys, or pill counts by as much as 17% [40]. Future studies should look to evaluate past and future interventions using true adherence measured by devices at the time of administration to truly capture the effect of the intervention. Lastly, none of the studies reported any information on the fidelity of the interventions, which is a critical aspect when considering if the outcome of an intervention is due to the clinical aspect of the lack of proper implementation and delivery of the intervention.
An assessment of why an intervention succeeds or fails would better enable clinicians in deciding whether to utilize it in clinical practice. Using theoretical models such as the Anderson Behavior Model (ABM) when developing adherence interventions and methods to measure adherence would increase the overall validity of the studies and help qualitatively assess the potential pitfalls of any study that did not indicate an improvement. ABM explains how an overall outcome is directly a result of patient health behaviors which consists of personal health practices and use the of health services [41]. A start would be using modern technology such as pill tracking smart bottles linked to mobile services providing information to both providers and patients [42] or by combining various complementary interventions together capitalizing on the strengths by resolving any weaknesses of the respective interventions in the structure and process portions to achieve the best outcomes of the patient [43]. Few studies go beyond adherence, persistence, or acceptability to capture personal health practices. Measures that could provide additional vital information include feasibility or utilization of the health interventions. However, further research is still needed to provide high-quality, effective, and cost-efficient interventions.
Conclusion
In conclusion, this systematic review showcases the scope of the current literature’s effectiveness of technological, text, and verbal communications in improving OET adherence in HR+ BC patients. While these interventions have shown effectiveness in a small number of patients followed for a short period of time, more work is needed before they can be incorporated in routine practice. Future research should design and test customizable interventions that may incorporate some of the interventions discussed here, standardize adherence measurement, assess acceptability and fidelity outcomes, conduct more RCTs with larger sample sizes, and explore long-term effects of the interventions. In addition, an effort should be made to constantly improve the interventions by teams of healthcare professionals, insurance companies, hospital representatives, and private sector to design, test, and implement novel strategies seamlessly for all involved parties. By addressing these gaps, we can advance the field of OET adherence interventions and improve outcomes in HR+ BC patients.
References
Wilkinson L, Gathani T. Understanding breast cancer as a global health concern. Br J Radiol. 2022;95(1130):20211033. https://doi.org/10.1259/bjr.20211033.
Alkabban FM, Ferguson T. Breast cancer. [Updated 2022 Sep 26]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023. Available from: https://www-ncbi-nlm-nihgov.ezproxy.lib.uh.edu/books/NBK482286/.
Loomans-Kropp HA, Umar A. Cancer prevention and screening: the next step in the era of precision medicine. NPJ Precis Oncol. 2019;3:3. https://doi.org/10.1038/s41698-018-0075-9.
Paranjpe R, John G, Trivedi M, Abughosh S. Identifying adherence barriers to oral endocrine therapy among breast cancer survivors. Breast Cancer Res Treat. 2019;174(2):297–305. https://doi.org/10.1007/s10549-018-05073-z.
Chumsri S, Howes T, Bao T, Sabnis G, Brodie A. Aromatase, aromatase inhibitors, and breast cancer. J Steroid Biochem Mol Biol. 2011;125(1-2):13–22. https://doi.org/10.1016/j.jsbmb.2011.02.001.
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG), Davies C, Godwin J, et al. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet Lond Engl. 2011;378(9793):771–84. https://doi.org/10.1016/S0140-6736(11)60993-8.
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials. Lancet Lond Engl. 2015;386(10001):1341–52. https://doi.org/10.1016/S0140-6736(15)61074-1.
Davies C, Pan H, Godwin J, et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet Lond Engl. 2013;381(9869):805–16. https://doi.org/10.1016/S0140-6736(12)61963-1.
Goss PE, Ingle JN, Pritchard KI, et al. Extending aromatase-inhibitor adjuvant therapy to 10 years. N Engl J Med. 2016;375(3):209–19. https://doi.org/10.1056/NEJMoa1604700.
Bartlett JMS, Sgroi DC, Treuner K, et al. Breast Cancer Index and prediction of benefit from extended endocrine therapy in breast cancer patients treated in the Adjuvant Tamoxifen-To Offer More? (aTTom) trial. Ann Oncol Off J Eur Soc Med Oncol. 2019;30(11):1776–83. https://doi.org/10.1093/annonc/mdz289.
Fisher B, Costantino JP, Wickerham DL, et al. Tamoxifen for the prevention of breast cancer: current status of the National Surgical Adjuvant Breast and Bowel Project P-1 study. J Natl Cancer Inst. 2005;97(22):1652–62. https://doi.org/10.1093/jnci/dji372.
Vogel VG, Costantino JP, Wickerham DL, et al. Update of the National Surgical Adjuvant Breast and Bowel Project Study of Tamoxifen and Raloxifene (STAR) P-2 Trial: preventing breast cancer. Cancer Prev Res (Phila). 2010;3(6):696–706. https://doi.org/10.1158/1940-6207.CAPR-10-0076.
Goss PE, Ingle JN, Alés-Martínez JE, et al. Exemestane for breast-cancer prevention in postmenopausal women. N Engl J Med. 2011;364(25):2381–91. https://doi.org/10.1056/NEJMoa1103507.
Cuzick J, Sestak I, Forbes JF, et al. Anastrozole for prevention of breast cancer in high-risk postmenopausal women (IBIS-II): an international, double-blind, randomised placebo-controlled trial. Lancet Lond Engl. 2014;383(9922):1041–8. https://doi.org/10.1016/S0140-6736(13)62292-8.
Hershman DL, Kushi LH, Shao T, et al. Early discontinuation and nonadherence to adjuvant hormonal therapy in a cohort of 8,769 early-stage breast cancer patients. J Clin Oncol Off J Am Soc Clin Oncol. 2010;28(27):4120–8. https://doi.org/10.1200/JCO.2009.25.9655.
Murphy CC, Bartholomew LK, Carpentier MY, Bluethmann SM, Vernon SW. Adherence to adjuvant hormonal therapy among breast cancer survivors in clinical practice: a systematic review. Breast Cancer Res Treat. 2012;134(2):459–78. https://doi.org/10.1007/s10549-012-2114-5.
Zhao H, Lei X, Niu J, et al. Prescription patterns, initiation, and 5-year adherence to adjuvant hormonal therapy among commercially insured patients with breast cancer. JCO Oncol Pract. 2021;17(6):e794–808. https://doi.org/10.1200/OP.20.00248.
Law R, Krupa K, Rusby J. Preventative therapy for breast cancer: a clinical experience. Breast Cancer Res Treat. 2023;201(2):205–13. https://doi.org/10.1007/s10549-023-06985-1.
Spencer JC, Reeve BB, Troester MA, Wheeler SB. Factors associated with endocrine therapy non-adherence in breast cancer survivors. Psychooncology. 2020;29(4):647–54. https://doi.org/10.1002/pon.5289.
Ekinci E, Nathoo S, Korattyil T, et al. Interventions to improve endocrine therapy adherence in breast cancer survivors: what is the evidence? J Cancer Surviv Res Pract. 2018;12(3):348–56. https://doi.org/10.1007/s11764-017-0674-4.
Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. J Clin Epidemiol. 2009;62(10):e1–34. https://doi.org/10.1016/j.jclinepi.2009.06.006.
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev. 2016;5(1):210. https://doi.org/10.1186/s13643-016-0384-4.
Downs SH, Black N. The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non-randomised studies of health care interventions. J Epidemiol Community Health. 1998;52(6):377–84. https://doi.org/10.1136/jech.52.6.377.
Van der Worp H, de Poel HJ, Diercks RL, van den Akker-Scheek I, Zwerver J. Jumper’s knee or lander’s knee? A systematic review of the relation between jump biomechanics and patellar tendinopathy. Int J Sports Med. 2014;35(8):714–22. https://doi.org/10.1055/s-0033-1358674.
Vetrano M, Castorina A, Vulpiani MC, Baldini R, Pavan A, Ferretti A. Platelet-rich plasma versus focused shock waves in the treatment of jumper’s knee in athletes. Am J Sports Med. 2013;41(4):795–803. https://doi.org/10.1177/0363546513475345.
Park HR, Kang HS, Kim SH, Singh-Carlson S. Effect of a smart pill bottle reminder intervention on medication adherence, self-efficacy, and depression in breast cancer survivors. Cancer Nurs. 2022;45(6):E874–82. https://doi.org/10.1097/NCC.0000000000001030.
Graetz I, McKillop CN, Stepanski E, Vidal GA, Anderson JN, Schwartzberg LS. Use of a web-based app to improve breast cancer symptom management and adherence for aromatase inhibitors: a randomized controlled feasibility trial. J Cancer Surviv Res Pract. 2018;12(4):431–40. https://doi.org/10.1007/s11764-018-0682-z.
Tan EH, Wong ALA, Tan CC, et al. Improving medication adherence with adjuvant aromatase inhibitor in women with breast cancer: a randomised controlled trial to evaluate the effect of short message service (SMS) reminder. Breast Edinb Scotl. 2020;53:77–84. https://doi.org/10.1016/j.breast.2020.06.012.
Singleton AC, Raeside R, Partridge SR, et al. Supporting women’s health outcomes after breast cancer treatment comparing a text message intervention to usual care: the EMPOWER-SMS randomised clinical trial. J Cancer Surviv Res Pract. Published online April 23. 2022:1–13. https://doi.org/10.1007/s11764-022-01209-9.
Hershman DL, Unger JM, Hillyer GC, et al. Randomized trial of text messaging to reduce early discontinuation of adjuvant aromatase inhibitor therapy in women with early-stage breast cancer: SWOG S1105. J Clin Oncol Off J Am Soc Clin Oncol. 2020;38(19):2122–9. https://doi.org/10.1200/JCO.19.02699.
Arch JJ, Crespi CM, Levin ME, et al. Randomized controlled pilot trial of a low-touch remotely-delivered values intervention to promote adherence to adjuvant endocrine therapy among breast cancer survivors. Ann Behav Med Publ Soc Behav Med. 2022;56(8):856–71. https://doi.org/10.1093/abm/kaab118.
Ream ME, Walsh EA, Jacobs JM, et al. Brief relaxation training is associated with long-term endocrine therapy adherence among women with breast cancer: post hoc analysis of a randomized controlled trial. Breast Cancer Res Treat. 2021;190(1):79–88. https://doi.org/10.1007/s10549-021-06361-x.
Getachew S, Addissie A, Seife E, et al. Breast nurse intervention to improve adherence to endocrine therapy among breast cancer patients in South Ethiopia. Oncologist. 2022;27(8):e650–60. https://doi.org/10.1093/oncolo/oyac081.
Jacobs JM, Post K, Massad K, et al. A telehealth intervention for symptom management, distress, and adherence to adjuvant endocrine therapy: a randomized controlled trial. Cancer. 2022;128(19):3541–51. https://doi.org/10.1002/cncr.34409.
Riis CL, Jensen PT, Bechmann T, Möller S, Coulter A, Steffensen KD. Satisfaction with care and adherence to treatment when using patient reported outcomes to individualize follow-up care for women with early breast cancer - a pilot randomized controlled trial. Acta Oncol Stockh Swed. 2020;59(4):444–52. https://doi.org/10.1080/0284186X.2020.1717604.
Toledo G, Ochoa CY, Farias AJ. Exploring the role of social support and adjuvant endocrine therapy use among breast cancer survivors. Support Care Cancer Off J Multinatl Assoc Support Care Cancer. 2020;28(1):271–8. https://doi.org/10.1007/s00520-019-04814-0.
Serrano D, Bonanni B, Brown K. Therapeutic cancer prevention: achievements and ongoing challenges - a focus on breast and colorectal cancer. Mol Oncol. 2019;13(3):579–90. https://doi.org/10.1002/1878-0261.12461.
DeCensi A, Thorat MA, Bonanni B, Smith SG, Cuzick J. Barriers to preventive therapy for breast and other major cancers and strategies to improve uptake. Ecancermedicalscience. 2015;9:595. https://doi.org/10.3332/ecancer.2015.595.
Rahimi S, Ononogbu O, Mohan A, Moussa D, Abughosh S, Trivedi M. Identifying the predictors of adherence to oral endocrine therapy in racial/ethnic minority patients with low socioeconomic status. Res Sq. Published online December 22. 2022; https://doi.org/10.21203/rs.3.rs-2379786/v1.
El Alili M, Vrijens B, Demonceau J, Evers SM, Hiligsmann M. A scoping review of studies comparing the medication event monitoring system (MEMS) with alternative methods for measuring medication adherence. Br J Clin Pharmacol. 2016;82(1):268–79. https://doi.org/10.1111/bcp.12942.
Lederle M, Tempes J, Bitzer EM. Application of Andersen’s behavioural model of health services use: a scoping review with a focus on qualitative health services research. BMJ Open. 2021;11(5):e045018. https://doi.org/10.1136/bmjopen-2020-045018.
McGrady ME, Ramsey RR. Using electronic monitoring devices to assess medication adherence: a research methods framework. J Gen Intern Med. 2020;35(9):2707–14. https://doi.org/10.1007/s11606-020-05905-z.
Tossaint-Schoenmakers R, Versluis A, Chavannes N, Talboom-Kamp E, Kasteleyn M. The challenge of integrating eHealth into health care: systematic literature review of the Donabedian model of structure, process, and outcome. J Med Internet Res. 2021;23(5):e27180. https://doi.org/10.2196/27180.
Funding
Dr. Trivedi is funded by the National Institute on Minority Health and Health Disparities (NIMHD) under Award Number U54MD015946.
Author information
Authors and Affiliations
Contributions
SG, SR, AL, and MVT contributed to the study conception and design. Literature search, data curation, validation, and/or analysis were performed by SG, SR, AL, and KL. MVT supervised the study and manuscript writing. The first draft of the manuscript was written by SG, SR, and AL. All authors reviewed, revised, and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Sourab Ganna, Sama Rahimi, and Meghana Trivedi are co-first authors.
Supplementary information
ESM 1
(DOCX 15 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ganna, S., Rahimi, S., Lu, A. et al. Interventions to improve oral endocrine therapy adherence in breast cancer patients. J Cancer Surviv (2024). https://doi.org/10.1007/s11764-023-01513-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11764-023-01513-y