Abstract
Planting crops to push or pull pests from a main crop and floral enhancements to attract natural enemies are emerging as pivotal agroecological strategies to shift away from synthetic pesticides. The brassica, Lobularia maritima, has great potential to act as a floral enhancement as it is attractive to many insects and improves the fitness of and biocontrol services provided by multiple natural enemies. It has been mainly deployed as an insectary plant; few studies have been conducted on its use as a trap plant. We explored the potential of L. maritima as a targeted flower enhancement in sheltered cropping systems through three case studies: (1) as flower strips alongside a cucumber crop, (2) as flower strips alongside a strawberry crop in a replicated on-farm experiment, and (3) as hanging pots under strawberry cultivation gutters. We monitored beneficial and pest insects in flowers and crops and assessed crop damage. Lobularia maritima stands out as an excellent floral enhancement plant due to its ease of cultivation and its ability to flower consistently over an extended period. Generalist predatory bugs, such as Orius laevigatus and Macrolophus pygmaeus, had higher densities on L. maritima than on other flowers grown next to strawberries. We found more thrips on L. maritima and less thrips on crops in two out of three experiments, compared with controls. Further research is needed to confirm if L. maritima is an effective trap crop for thrips and other pests and to detect possible dis-services, such as the attraction of phytophagous bugs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Trap cropping and floral enhancements emerge as a pivotal agroecological strategy facilitating the shift away from synthetic pesticides, underscoring its significance in sustainable agricultural practices (Albrecht et al. 2020). This approach offers a multifaceted solution, targeting various ecosystem services and dis-services simultaneously, thereby addressing complex ecological dynamics (Sutter et al. 2018). Importantly, it is crucial to recognize that these ecosystem services are interconnected rather than independent entities, emphasizing the need for holistic approaches in agroecological management (Sutter 2016). Selecting appropriate plant species for floral enhancement programs becomes paramount, especially considering the specific requirements of target insect species and the desired outcomes, whether conservation or biological control (Sutter et al. 2017). Thus, understanding the intricate relationships between plant species, insect communities, and ecosystem services is fundamental in effectively harnessing the potential of floral enhancement strategies for sustainable agriculture.
The same plant species can have different effects on biological pest control depending in which cropping system it is deployed. “A flowering plant which attracts and possibly maintains, with its nectar and pollen resources, a population of natural enemies which contribute to biological pest management on crops” is an insectary plant (Parolin et al. 2012a). By providing suitable conditions for their establishment and reproduction, it acts as a nursery plant for beneficials. An insectary plant has a direct positive effect on natural enemies, which in turn reduce pest density. As a result, an insectary plant has an indirect positive effect on the main crop (Parolin et al. 2012b). A plant species that serves as an insectary plant for a natural enemy can also be a trap plant for an arthropod pest. Trap plants “attract insects or other organisms like nematodes to protect target crops from pest attack, preventing the pests from reaching the crop or concentrating them in a certain part of the field where they can be economically destroyed” (Shelton and Badenes-Perez 2006). Target pests have a direct negative effect on the trap plant, which results in an indirect positive effect of the latter on the main crop (Parolin et al. 2012b). Finding floral species that encompass several categories of secondary plants in the same growing system could boost biological control while reducing the workload and complexity.
Lobularia maritima L. Desv. (Brassicaceae), also known as sweet alyssum, is an annual or short-lived perennial herbaceous plant native to the Mediterranean region (Picó and Retana 2003). This low, spreading plant grows up to 30 cm high and has narrow, lanceolate leaves arranged in rosettes. Its inflorescences are initially umbelliform, then elongate into multiflora clusters composed of white or purple-petalled flowers (Lauber et al. 2009). Lobularia maritima grows best in sandy, well-drained soil and needs moderate watering (Henson et al. 2006; Landolt and Bäumler 2010). It has been widely cultivated as an ornamental plant and features multiple properties for flower enhancement in agriculture.
Being an excellent source of pollen and nectar, L. maritima is very attractive to many natural enemies (Landis et al. 2000; González-Chang et al. 2019). It has an extended, uninterrupted flowering period of around 10 months (Picó and Retana 2003) and establishes rapidly, making it competitive against weeds (Begum et al. 2006; Grasswitz 2013). Lobularia maritima was found to enhance the presence of beneficial insects including pollinators (Barbir et al. 2015; Scarlato et al. 2023), aphidophagous hoverflies (Pineda and Marcos-García 2008; Gillespie et al. 2011; Amorós-Jiménez et al. 2014; Harris-Cypher et al. 2023), various parasitoid wasps (Gámez-Virués et al. 2009; Aparicio et al. 2018; Arnó et al. 2018; Buchanan et al. 2018; Madeira et al. 2022) and predatory bugs (Haseeb et al. 2018).
Positive effects on the fitness of natural enemies have also been observed in multiple studies. Supplying L. maritima boosted the longevity (Johanowicz and Mitchell 2000; Munir et al. 2018), fecundity (Hogg et al. 2011) or both (Berndt and Wratten 2005; Pumariño and Alomar 2012; Balzan and Wäckers 2013; Araj et al. 2019; Herz et al. 2021), as well as the sex ratio (Berndt and Wratten 2005) and body weight (Nilsson et al. 2011) of various parasitoids and predators. In the presence of L. maritima, improved biocontrol services have been recorded against several hemipteran (Hogg et al. 2011; Irvin et al. 2021; Lopez and Liburd 2022; Zuma et al. 2023) and lepidopteran pests (Gámez-Virués et al. 2009; Shrestha et al. 2019). In several cases, this flowering species promoted natural enemies without benefiting pests (Begum et al. 2006; Munir et al. 2018; Scarlato et al. 2023). Its ability to attract beneficial insects and improve biological control makes L. maritima as a potential trap plant a useful tool for integrated pest management strategies.
Mainly used as an insectary plant, L. maritima also has potential as a trap plant, attracting and retaining target pests, thereby reducing crop damage and yield losses (Tiwari et al. 2020; Arnó et al. 2021; Silva et al. 2022). However, this potential use has been poorly researched and further investigation is needed to fully understand and optimize it in various cropping systems.
The aim of this study is to explore the potential of L. maritima as a targeted flower enhancement for sheltered crops in different settings and, more specifically, to show its attractiveness to pests and beneficials in relation to the main crop. We conducted three case studies: (1) to assess the attractiveness of several flowering species, including L. maritima, to cucumber pests and their natural enemies, as well as associated effects on pest control and crop damage when sown alongside a greenhouse cucumber crop; (2) to evaluate these same parameters focusing on thrips and phytophagous bugs in a tunnel strawberry production system; and (3) to explore the influence of L. maritima in hanging pots under an off-ground strawberry crop on the control of thrips and phytophagous bugs.
Material and methods
Flower strip next to greenhouse cucumbers
The first case study was conducted from March to October 2022 in the facilities of Agroscope in Conthey (Valais, Switzerland). Two crop cycles of ungrafted single-stemmed cucumber plants cv. Pradera F1 (Rijk Zwann, Netherlands) were cultivated in two identical open-ground greenhouses (350 m2, no insect-proof netting on the openings). The first and the second crop cycles were planted in weeks 14 and 28, respectively, and grown for 12 weeks each using a high-wire system. Seven 23-m-long rows were set up in the reference greenhouse (0.45 m between plants, 1.7 m between rows). In the test greenhouse, the cucumber row furthest away from the reference greenhouse was replaced by a flower strip which was sown during week 12.
The flower strip was divided into three parts from north to south. A mix of six flowering species was sown in the northern and southern plots (1 m × 7.5 m), and single-species plots were sown in the mid-Sect. (6 × 1 m2). The six flowering species were Calendula officinalis L. (Asteraceae), Centaurea cyanus L. (Asteraceae), Coriandrum sativum L. (Apiaceae), Fagopyrum esculentum Moench (Polygonaceae), L. maritima (Brassicaceae) and Vicia sativa L. (Fabaceae). The mixed plots remained in place for the duration of the two crop cycles. Single-species plots were removed in week 28 and replaced by a mix of six species sown in week 29 (Fig. 1). The same mix of species was used, except for V. sativa, which was replaced by Medicago sativa L. (Fabaceae). The proportion of each species in the new mix was adjusted based on observations made in the first mix (Supplementary Information (SI) Table S1).
Design of the flower strip sown next to the first (A) and second (B) cucumber cycle. The main crop was divided into 18 plots separated by buffer plants (stripped). In the control greenhouse, the flower strip was replaced by a row of cucumber plants resulting in 21 cucumber plots. Single-species plots (1 × 1m2): FE Fagopyrum esculentum, LM Lobularia maritima, CO Calendula officinalis, CC Centaurea cyanus, CS Coriandrum sativum, VS Vicia sativa. Mixed species plots (7.5 × 1m2): MIX VS = FE + LM + CO + CC + CS + VS, MIX MS = FE + LM + CO + CC + CS + Medicago sativa. All flower plots were sown during week 12 except MIX MS which was sown during week 29
Two separate drip irrigation systems were installed in the cucumber crop (UNIRAM CNL 16010, 2.3 l/h, 0.3 m between drip holes, two pipes per plant row) and in the flower strip (T-tape 150 TSX, 5 l/h, 0.2 m between drip holes, four rows of pipes spaced 0.2 m apart). Only the cucumber crop was fertilized, and the drip system was disinfected between the two cucumber crop cycles. No insecticides were applied during the two cucumber cycles. Three fungicide treatments were applied in the first crop cycle: Armicarb® (Stähler, 0.3%) against powdery mildew and Airone® WG (Andermatt Biocontrol, 0.3%) against leaf mould during week 16 and Amistar® (Stähler, 0.1%) against cucurbit scab during week 17. In the second crop cycle, five fungicide treatments were applied: Airone® WG (Andermatt Biocontrol, 0.3%) against downy mildew once during week 32, Amistar® (Stähler, 0.1%) against downy mildew twice during week 33 and Armicarb® (Stähler, 0.3%) against powdery mildew once during weeks 35 and 36.
Augmentative releases of natural enemies were carried out in both crop cycles (SI Table S2). Aphidius colemani Viereck (Hymenoptera: Braconidae), Eupeodes corollae Fabricius (Diptera: Syrphidae) and Chrysoperla carnea Stephens (Neuroptera: Chrysopidae) were released against aphids. Amblyseius swirskii Athias-Henriot, A. cucumeris Oudemans (Acari: Phytoseiidae) and Orius laevigatus Fieber (Heteroptera: Anthocoridae) were released against thrips. Phytoseiulus persimilis Athias-Henriot (Acari: Phytoseiidae) and Feltiella acarisuga Vallot (Diptera: Cecidomyiidae) were released against pest mites. Encarsia formosa Gahan (Hymenoptera: Aphelinidae) was released against whiteflies.
Pests, natural enemies and inflorescences were monitored weekly in the flower strip. During the first cucumber crop cycle, data were collected in each single-species plot and in three predefined 1 m2 patches per mixed flower plot. During the second cucumber crop cycle, a single predefined 1 m2 patch per mixed plot was sampled. Insects were counted visually on ten leaves per flowering species in each 1 m2 patch, and each plant species was shaken ten times over a white tray to count thrips. In case of doubt about the identification of a species, a sample was taken back to the laboratory for examination under binoculars.
In addition, the total number of inflorescences per species was recorded in each patch. Pests and natural enemies were monitored weekly in the crop. Insects were counted on six plants per row in every other row. One leaf at the top, middle and bottom of each plant was sampled. Insect damage on the crop was assessed twice a week at harvest in 18 plots per greenhouse. The crop line replacing the flower strip in the control greenhouse was left out of this assessment. All cucumbers weighing between 350 and 500g were harvested and sorted. They were then visually inspected and sorted into three categories: marketable cucumbers, cucumbers with insect damage and others (e.g. natural curvature greater than 20mm per 10cm length).
Flower strip next to covered strawberries in a replicated on-farm experiment
The second case study was conducted from March to October 2021 in four commercial off-ground strawberry crops located between Conthey and Martigny (Valais, Switzerland). Everbearing strawberries cv. Vivara (Salvi Vivai, Italy) were planted in substrate bags (Pro-Mix GBX, Premier Tech, Canada) placed under plastic tunnels. Irrigation and fertilization were carried out using a drip system. A flower strip was sown next to each of the four tunnel groups. Crop and flower strip features varied between plots (Table 1).
Pests and natural enemies were monitored weekly in the flower strips from week 23 to 32 (in all plots). Samples were collected by aspiration (ecoVac – Insektensauger, ecoTech Umwelt-Messsysteme GmbH) of ten linear metres per flowering species with the nozzle at vegetation level. Each sample was cooled, transferred into a labelled bag and frozen. Insects were then identified and counted under a binocular. Pest and natural enemies were monitored weekly in the crop from week 24 to 35 (in plots 1 to 3; no data collection in weeks 26, 33 and 34). Insects were collected by shaking one flower spike on each of 40 strawberry plants next to the flower strip and 40 others located 20 m from it. The number of flowers per spike was counted at each sampling round and used to calculate the number of insects per flower. In addition, the percentage of fruit with visible thrips or bug damage was assessed weekly from week 23 to 36 (in plots 1 to 3).
Potted L. maritima in covered strawberries
The third case study was conducted from March to August 2022 in a commercial off-ground strawberry crop in Saxon (Valais, Switzerland). In March, everbearing strawberries cv. Vivara (Salvi Vivai) were planted in substrate bags (Pro-Mix GBX, Premier Tech) placed under two identical plastic tunnels (400 m2). Four 50-m-long rows (8 plants per linear metre) were set up in each tunnel. Irrigation and fertilization were carried out using a drip system.
The two following treatments were compared: (1) strawberry plants above hanging pots of L. maritima; and (2) strawberry plants only (control). Each tunnel was divided into three parts (separated by buffer zones) where three replicates of each treatment were set up alternately. Lobularia maritima was sown in 60 × 17 × 14 cm3 white pots (50 seeds/pot) in week 9. The pots were fastened with two wires to the gutters supporting the strawberry crop (1 pot for two linear metres) in week 14 (Fig. 2). Irrigation was carried out using a drip system.
Pest and natural enemies were monitored weekly in L. maritima and in the crop from week 17 to 33. Insects were collected by shaking one flower spike on each of 28 L. maritima plants and/or 28 strawberry plants in each repetition of both treatments. In addition, the number of flowers per spike was counted at each sampling round, and the percentage of fruit with visible thrips or bug damage was assessed at each harvest.
Data analysis
The experiment conducted in 2021 in sheltered cucumbers consisted in the comparison of two greenhouses managed differently (i.e. flower strip treatment in one greenhouse vs. no intervention in the other). Similarly, the experiment conducted in 2022 in sheltered strawberries consisted in the comparison between six plots with different management methods (i.e. three plots with potted L. maritima vs. three plots with no intervention). There is no true statistical replication in these experiments. Further, although we conducted a replicated on-farm trial in sheltered strawberries in 2021, the limited number of replicates results in small statistical power. For these reasons, we preferred presenting the mean differences (with standard error where possible), instead of presenting non-parametric mean comparison with p-values, leading to potentially misleading conclusions.
Results
Flowering period of L. maritima compared with six other floral species
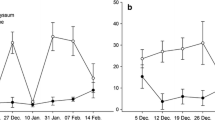
Lobularia maritima started to flower approximately one month after sowing and produced inflorescences continuously throughout both cucumber crop cycles (Fig. 3). It represented 81.2% of all inflorescences recorded in the flower strip. Fagopyrum esculentum flowered earliest, three weeks after sowing. Flowering peaked for three weeks and then rapidly decreased with no flowering during the second cucumber crop cycle. Fagopyrum esculentum produced 15.2% of all inflorescences in the flower strip. Calendula officinalis started to flower eight weeks after sowing and continued to do so for the rest of the cucumber crop cycles. It represented 3% of the total number of inflorescences counted in the flower strip. Coriandrum sativum produced a small number of flowers in weeks 9 to 11, corresponding to just 0.6% of all inflorescences. Centaurea cyanus and V. sativa developed poorly with negligible flowering.
In the new mixed plot sown in week 29 at the beginning of the second cucumber crop cycle, L. maritima began to flower 5 weeks after sowing (Fig. 4). As in the first mixed plots, flowering was continuous throughout the crop but L. maritima produced only 8.7% of the total number of inflorescences. Again, F. esculentum flowered earliest at four weeks after sowing. Flowering peaked for four weeks and then rapidly declined. No flowers were recorded on any of the other plant species during the second cucumber crop cycle.
Attractiveness of L. maritima to pests and natural enemies compared with six other floral species
In the flower strip next to the greenhouse cucumbers, L. maritima attracted the most insects during both crop cycles, i.e. 25.4% and 31.4% of all insects recorded, respectively (Fig. 5). Twenty to 350 times more thrips were recorded on L. maritima than on other plant species. In contrast, it attracted up to 22 times less aphids than other plant species and a negligible number of pest mites. In the second cucumber crop cycle, the phytophagous bug, Lygus rugulipennis Poppius (Heteroptera: Miridae), was observed on both L. maritima and C. officinalis, with a stronger presence on the first flowering species.
Cumulative number of pests and natural enemies (Nat. en.) recorded in 2021 in the different species in the flower strip during the first (A) and second (B) cucumber crop cycles. LM Lobularia maritima, FE Fagopyrum esculentum, CO Calendula officinalis, CC Centaurea cyanus, CS Coriandrum sativum, VS Vicia sativa (in the first crop cycle only) and MS Medicago sativa (in the second crop cycle only)
Few natural enemies were found in the flower strip during the first cucumber crop cycle. Calendula officinalis attracted the most individuals, in particular A. colemani. Larger populations were observed in the flower strip during the second crop cycle. Lobularia maritima and C. officinalis were the most attractive species to beneficial insects, accounting for 60.5% and 25.8% of all individuals, respectively. Macrolophus pygmaeus Rambur (Hemiptera: Miridae) was found on both species. However, more than twice as many individuals were found on L. maritima as on C. officinalis. Lobularia maritima also attracted O. laevigatus, which was not found on any other flowering species.
As in the cucumber experiment, L. maritima was the most attractive species for insects in the flower strip next to covered strawberries (Fig. 6). It accounted for 39.7% of all the individuals recorded in the flower strip, exceeding the 12 species mix on which 25.9% of insects were observed. Lobularia maritima attracted 1.7 to 3.1 times more thrips and 1.2–3.3 times more phytophagous bugs than other flowering species. Lygus rugulipennis was the most prevalent species of phytophagous bugs.
Cumulative number of pests and natural enemies (Nat. en.) recorded in 2021 in the different species in the flower strip next to the sheltered strawberry crop. Samples were collected by aspiration of ten linear metres per flowering species with the nozzle at vegetation level. LM Lobularia maritima, MIX 12 species mix, MS Medicago sativa, TR Trifolium repens
Regarding natural enemies, L. maritima attracted 1.9–7.4 times more O. laevigatus and 2.1–5.7 times more parasitoids than other flowering species. It accounted for 46.5% of all recorded natural enemies, followed by M. sativa (22.9%), the 12 species mix (17.3%), and T. repens (13.3%).
Effect of floral enhancement measures on population densities of pests and natural enemies in cucumber and strawberry crops
In the first cucumber crop cycle, equally low numbers of pests and natural enemies were observed in the crop grown next to the flower strip as in the reference crop (Fig. 7). The main pests present in the crop were whiteflies, thrips, pest mites and aphids. Thrips represented the greatest risk of direct harvest losses, while aphids and whiteflies were more likely to transmit viruses and indirectly affect yield via plant vigour. Among natural enemies, Amblyseius spp. were the most prevalent.
In the second cucumber crop cycle, pest and natural enemy populations recorded in the reference crop were 3.4 and 4.3 times larger, respectively, than in the crop next to the flower strip. The main differences between the two treatments were observed in the numbers of aphids and A. colemani. Populations of aphids and parasitoid wasps were reduced by 93% and 99%, respectively, in the cucumbers grown next to the flower strip compared to the reference crop. Thrips populations decreased by 32% in the cucumbers next to the flower strip compared to the reference crop.
Similarly, the number of thrips per strawberry flower decreased by 31% in the strawberry rows next to the flower strip compared with rows 20 m away from it (Fig. 8). Similar numbers of L. rugulipennis and O. laevigatus were found at both distances.
Cumulative number of thrips (A), Lygus rugulipennis (B) and Orius laevigatus (C) per strawberry flower recorded in 2021 in the sheltered strawberry crop. CO strawberry plants 20 m away from the flower strip (control), FS strawberry plants next to the flower strip. Error bars represent the standard error of the mean
In the experiment carried out in 2022 with potted L. maritima under off-ground strawberries, no difference in thrips and phytophagous bug populations was found between those plots with and those without flower enhancement.
Effect of floral enhancement measures on crop damage in cucumbers and strawberries
Similar percentages of crop damage were found in both the greenhouses with and without flower strips during the first cucumber crop cycle (Fig. 9A, B). In contrast, twice as much damage was measured in the greenhouse with the flower strip as in the reference greenhouse during the second crop cycle.
Average percentage of crop damage recorded in 2021 in the first (A) and second (B) cucumber crop cycles. Average percentage of strawberries with damage caused by thrips (C) and tarnished plant bugs (D) recorded in 2021 in the covered strawberry crops. In A and B: CO cucumber greenhouse without flower strip (control), FS cucumber greenhouse with flower strip. In C and D: CO strawberry plants 20 m from the flower strip (control), FS strawberry plants next to the flower strip. Error bars represent the standard error of the mean
Comparable percentages of strawberries damaged by thrips were observed next to and 20 m from the flower strip in covered strawberries (Fig. 9C, D). Damage caused by tarnished plant bugs tended to be lower in strawberry plants grown next to the flower strip than in those 20 m away.
In the 2022 case study, lower thrips damage was recorded in strawberry plants above suspended pots of L. maritima than in the control plots. On average over the season, the number of strawberries damaged by thrips was reduced by 50% in plots with L. maritima (Fig. 10). Damage reduction varied from 3 to 84% in the different sampling rounds. In contrast, 44% more strawberries with damage caused by tarnished plant bugs were found in the plots with L. maritima than in the control.
Average percentage of strawberries with damage caused by thrips (A) and tarnished plant bugs (B) recorded in 2022 in the covered strawberry crops. Solid line strawberry plots with potted Lobularia maritima, dashed line strawberry plots without potted L. maritima (control). Error bars represent the standard error of the mean
Discussion
This research illustrates the potential of L. maritima as a targeted flower enhancement in sheltered crops. This flowering species was found to be well adapted and practical for greenhouse and tunnel cultivation. It was suitable for soil and pot growing and performed well in both conditions. Lobularia maritima attracted many insects and has a high potential in integrated management of thrips in strawberries. However, a potential increase in phytophagous bug populations and associated crop damage needs to be considered in future research.
In line with previous results (Picó and Retana 2003; Irvin et al. 2021), L. maritima grown next to the greenhouse cucumber crop had an early, extended and uninterrupted flowering period. It had the longest and most abundant flowering of all the tested plant species, comprising more than 80% of all the recorded inflorescences. Blooming started four weeks after sowing and continued for more than five months thereafter. At the last monitoring, L. maritima was still producing more than 300 inflorescences per m2. Fagopyrum esculentum, another common insectary plant, started flowering a week earlier than L. maritima, but only for a short period of about five weeks. This species had to be reseeded to obtain new flowers during the season. However, it rapidly developed after the summer sowing and started flowering within four weeks. In contrast, C. officinalis began to flower one month after L. maritima but bloomed continuously thereafter. In a previous study, seed germination of L. maritima peaked at 10°C and progressively decreased at both higher and lower temperatures (Picó and Retana 2003). Consistent with this, L. maritima established and flowered more rapidly after spring sowing when temperatures were lower than after summer sowing. Therefore, we recommend sowing it under greenhouse early in spring or possibly in autumn (provided the infrastructure is kept frost-free during the winter) to ensure an optimal development.
Due to its long flowering period, L. maritima provided pollen and nectar throughout the growing season. It attracted the highest number of insects amongst all the flowering species tested alongside cucumbers and strawberries. However, natural enemies were slow to colonize it in our cucumber case study. Only a few predatory mites were recorded in L. maritima during the first crop cycle and the flower strip had no effect on the presence of pests and natural enemies in the crop. It took until the second crop cycle to observe larger populations of generalist predatory bugs and a strongly reduced presence of pests in the crop next to the flower strip compared to the reference greenhouse. This reduction was mainly due to a decreased number of aphids, which either migrated to the flower strip or were controlled by natural enemies. As a result, populations of A. colemani also strongly decreased in the crop.
Interestingly, the flower strip boosted the presence of M. pygmaeus in the crop although this mirid predator was previously found to be unable to maintain itself on cucumber plants (Perdikis and Lykouressis 2003). Calendula officinalis is known as a natural host of this zoophytophagous species (Ingegno et al. 2011) and used as a banker plant to foster its establishment and biocontrol services in tomato greenhouses (Ardanuy et al. 2022). Consistent with this, M. pygmaeus moved from the adjacent tomato greenhouse and naturally colonized C. officinalis in our cucumber case study. However, we recorded twice as many individuals of this species on L. maritima, highlighting the latter’s potential as a banker plant for M. pygmaeus. Further research should be conducted to assess if growing L. maritima alongside various crops proves beneficial to M. pygmaeus and increases its biocontrol services. This would be useful as this generalist predator feeds on multiple pests such as whiteflies, spider mites, thrips and moths.
Another positive trait of L. maritima recorded in both case studies carried out in 2021 is its high attractiveness for O. laevigatus. Higher densities of this predatory bug were recorded on L. maritima than on all the other floral species tested. Bennison et al. (2011) mentioned L. maritima’s potential use as a combined trap plant for the Western flower thrips, Frankliniella occidentalis Pergande (Thysanoptera: Thripidae) and banker plant to promote the predatory bug O. laevigatus. No such dual effect of L. maritima could be demonstrated in the present case studies, however this should be further researched as it would be ideal for optimizing thrips control. In another experiment, the presence of L. maritima in a strawberry cropping system positively influenced the population growth of the predatory bug and the biocontrol services it provided against aphids (Zuma et al. 2023). In our cucumber case study, the higher density of O. laevigatus may have contributed to reducing aphid pressure during the second crop cycle.
Despite a strong reduction in pest populations in the second cucumber crop cycle, insect crop damage was twice as high next to the flower strip as in the reference greenhouse. Although L. rugulipennis was not directly recorded in the crop (probably due to its high mobility), most of the additional damage was caused by this pest. In the flower strip, the phytophagous bug was mainly found on L. maritima, highlighting the importance of considering possible dis-services in future assessments of this flowering species. For example, twice as many tarnished plant bugs were found in flowered strips composed of L. maritima as in mown border strips set up along an asparagus crop (Buchanan et al. 2018). Miridae were reported to be attracted to L. maritima and larger populations of homopteran pests were recorded in the inter-rows of an organic vineyard when this floral species was sown (Burgio et al. 2016). Further, Köneke et al. (2023) reported L. maritima to cause an increase of flea beetle infestation when intercropped with cabbage. Contrarily to the results of the cucumber case study, damage caused by tarnished plant bugs tended to be lower in strawberries next to the flower strip as in those 20 m from it. Regarding thrips, approximately one third fewer individuals were found on cucumber plants next to the flower strip as in the reference greenhouse. A similar reduction was observed between strawberries grown next to the flower strip and those 20 m from it. However, there was no effect of the flower strips on thrips damage in the crops grown in 2021. As both flower strips were composed of several species, the contribution of L. maritima to this result remains to be determined.
Finally, the results of the case study carried out in 2022 provide some insights into its use for the management of thrips, possibly as a trap crop. While similar thrips populations were recorded on strawberry plants grown above hanging pots of L. maritima as in control plots, thrips damage was halved in flowered plots. We assume that L. maritima is a trap plant for thrips, as previously reported by Bennison et al. (2011), and that its position in the crop plays a key role in reducing crop damage. In our case study, L. maritima was grown in hanging pots below the cultivation gutters. We postulate that winged adult thrips preferred potted L. maritima to strawberry plants for oviposition and that the young non-winged larvae, which cause the most damage, were then unable or unwilling to climb back onto the crop. These results should be considered carefully, as they are based on observations only and thrips pressure was relatively low in 2022. However, this is the first time to our knowledge that such a phenomenon has been reported.
We believe that L. maritima has great potential as a multifunctional companion plant and that further investigations are necessary to reveal it. These should include considerations on the optimal position (i.e. potted vs. in-ground plants, connected vs. disconnected from the main crop) and density of this floral species in the main crop. Various combinations of L. maritima with other flowering plants could also be tested in order to mitigate potential dis-services, such as the attraction of tarnished plant bugs to the main crop. Further experiments should also aim to prove the attractiveness of L. maritima to predatory bugs and the usefulness of this property in different cropping systems (e.g. biocontrol services of M. pygmaeus on crops other than tomatoes).
Conclusions
We illustrated the potential of L. maritima for flower enhancement in sheltered crops and postulated its possible applications for biological control of thrips. Lobularia maritima is a flowering species with many advantages. It is easy to grow, performs well in pots and in open ground, and is suitable for open-field and covered cultivation. Lobularia maritima has a long and abundant flowering period compared to other floral species, making it attractive to insects, in general. Generalist predatory bugs, such as Orius laevigatus and Macrolophus pygmaeus, had higher densities on L. maritima than on other flowers grown next to the main crop. We found more thrips on L. maritima and less thrips on crops in two out of three experiments, compared with controls. Further research is needed to confirm if L. maritima is an effective trap crop for thrips and other pests and to detect possible dis-services, such as the promotion of phytophagous bugs. We assume that a targeted management of L. maritima in the main crop would make it possible to take advantage of both its attractiveness to natural enemies (parasitoids, predatory bugs) and its properties as a trap plant for thrips.
Data availability
All data underlying the analyses will be available upon acceptance of this manuscript.
References
Albrecht M, Kleijn D, Williams NM, Tschumi M, Blaauw BR, Bommarco R, Campbell AJ, Dainese M, Drummond FA, Entling MH, Ganser D, Arjen de Groot G, Goulson D, Grab H, Hamilton H, Herzog F, Isaacs R, Jacot K, Jeanneret P, Jonsson M, Knop E, Kremen C, Landis DA, Loeb GM, Marini L, McKerchar M, Morandin L, Pfister SC, Potts SG, Rundlöf M, Sardiñas H, Sciligo A, Thies C, Tscharntke T, Venturini E, Veromann E, Vollhardt IMG, Wäckers F, Ward K, Westbury DB, Wilby A, Woltz M, Wratten S, Sutter L (2020) The effectiveness of flower strips and hedgerows on pest control, pollination services and crop yield: a quantitative synthesis. Ecol Lett 23(10):1488–1498. https://doi.org/10.1111/ele.13576
Amorós-Jiménez R, Pineda A, Fereres A, Marcos-García MÁ (2014) Feeding preferences of the aphidophagous hoverfly Sphaerophoria rueppellii affect the performance of its offspring. Biocontrol 59(4):427–435. https://doi.org/10.1007/s10526-014-9577-8
Aparicio Y, Gabarra R, Arnó J (2018) Attraction of Aphidius ervi (Hymenoptera: Braconidae) and Aphidoletes aphidimyza (Diptera: Cecidomyiidae) to sweet alyssum and assessment of plant resources effects on their fitness. J Econ Entomol 111(2):533–541. https://doi.org/10.1093/jee/tox365
Araj S-E, Shields MW, Wratten SD (2019) Weed floral resources and commonly used insectary plants to increase the efficacy of a whitefly parasitoid. Biocontrol 64(5):553–561. https://doi.org/10.1007/s10526-019-09957-x
Ardanuy A, Figueras M, Matas M, Arnó J, Agustí N, Alomar Ò, Albajes R, Gabarra R (2022) Banker plants and landscape composition influence colonisation precocity of tomato greenhouses by mirid predators. J Pest Sci 95(1):447–459. https://doi.org/10.1007/s10340-021-01387-y
Arnó J, Oveja MF, Gabarra R (2018) Selection of flowering plants to enhance the biological control of Tuta absoluta using parasitoids. Biol Control 122:41–50. https://doi.org/10.1016/j.biocontrol.2018.03.016
Arnó J, Molina P, Aparicio Y, Denis C, Gabarra R, Riudavets J (2021) Natural enemies associated with Tuta absoluta and functional biodiversity in vegetable crops. Biocontrol 66(5):613–623. https://doi.org/10.1007/s10526-021-10097-4
Balzan MV, Wäckers FL (2013) Flowers to selectively enhance the fitness of a host-feeding parasitoid: adult feeding by Tuta absoluta and its parasitoid Necremnus artynes. Biol Control 67(1):21–31. https://doi.org/10.1016/j.biocontrol.2013.06.006
Barbir J, Badenes-Pérez FR, Fernández-Quintanilla C, Dorado J (2015) The attractiveness of flowering herbaceous plants to bees (Hymenoptera: Apoidea) and hoverflies (Diptera: Syrphidae) in agro-ecosystems of Central Spain. Agr Forest Entomol 17(1):20–28. https://doi.org/10.1111/afe.12076
Begum M, Gurr GM, Wratten SD, Hedberg PR, Nicol HI (2006) Using selective food plants to maximize biological control of vineyard pests. J Appl Ecol 43(3):547–554. https://doi.org/10.1111/j.1365-2664.2006.01168.x
Bennison J, Pope T, Maulden K (2011) The potential use of flowering alyssum as a ‘banker’ plant to support the establishment of Orius laevigatus in everbearer strawberry for improved biological control of western flower thrips. In: Vänninen I (ed) Working Group “Integrated Control in Protected crops, Temperate Climate”. Proceedings of the Meeting at Sutton Scotney (United Kingdom), 18–22 September, 2011
Berndt LA, Wratten SD (2005) Effects of alyssum flowers on the longevity, fecundity, and sex ratio of the leafroller parasitoid Dolichogenidea tasmanica. Biol Control 32(1):65–69. https://doi.org/10.1016/j.biocontrol.2004.07.014
Buchanan A, Grieshop M, Szendrei Z (2018) Assessing annual and perennial flowering plants for biological control in asparagus. Biol Control 127:1–8. https://doi.org/10.1016/j.biocontrol.2018.08.013
Burgio G, Marchesini E, Reggiani N, Montepaone G, Schiatti P, Sommaggio D (2016) Habitat management of organic vineyard in Northern Italy: the role of cover plants management on arthropod functional biodiversity. Bull Entomol Res 106(6):759–768. https://doi.org/10.1017/S0007485316000493
Gámez-Virués S, Gurr G, Raman A, La Salle J, Nicol H (2009) Effects of flowering groundcover vegetation on diversity and activity of wasps in a farm shelterbelt in temperate Australia. Biocontrol 54(2):211–218. https://doi.org/10.1007/s10526-008-9182-9
Gillespie M, Wratten S, Sedcole R, Colfer R (2011) Manipulating floral resources dispersion for hoverflies (Diptera: Syrphidae) in a California lettuce agro-ecosystem. Biol Control 59(2):215–220. https://doi.org/10.1016/j.biocontrol.2011.07.010
González-Chang M, Tiwari S, Sharma S, Wratten SD (2019) Habitat management for pest management: Limitations and prospects. Ann Entomol Soc Am 112(4):302–317. https://doi.org/10.1093/aesa/saz020
Grasswitz TR (2013) Development of an insectary plant mixture for New Mexico and its effect on pests and beneficial insects associated with pumpkins. Southwest Entomol 38(3):417–436. https://doi.org/10.3958/059.038.0306
Harris-Cypher A, Roman C, Higgins G, Scheufele S, Legrand A, Wallingford A, Sideman RG (2023) A field survey of syrphid species and adult densities on annual flowering plants in the Northeastern United States. Environ Entomol 52(2):175–182. https://doi.org/10.1093/ee/nvad016
Haseeb M, Gordon TL, Kanga LHB, Legaspi JC (2018) Abundance of natural enemies of Nezara viridula (Hemiptera: Pentatomidae) on three cultivars of sweet alyssum. J Appl Entomol 142(9):847–853. https://doi.org/10.1111/jen.12552
Henson DY, Newman SE, Hartley DE (2006) Performance of selected herbaceous annual ornamentals grown at decreasing levels of irrigation. Hortic Sci 41(6):1481–1486. https://doi.org/10.21273/HORTSCI.41.6.1481
Herz A, Dingeldey E, Englert C (2021) More power with flower for the pupal parasitoid Trichopria drosophilae: a candidate for biological control of the spotted wing drosophila. InSects 12(7):628. https://doi.org/10.3390/insects12070628
Hogg BN, Nelson EH, Mills NJ, Daane KM (2011) Floral resources enhance aphid suppression by a hoverfly. Entomol Exp Appl 141(2):138–144. https://doi.org/10.1111/j.1570-7458.2011.01174.x
Ingegno BL, Pansa MG, Tavella L (2011) Plant preference in the zoophytophagous generalist predator Macrolophus pygmaeus (Heteroptera: Miridae). Biol Control 58(3):174–181. https://doi.org/10.1016/j.biocontrol.2011.06.003
Irvin NA, Pierce C, Hoddle MS (2021) Evaluating the potential of flowering plants for enhancing predatory hoverflies (Syrphidae) for biological control of Diaphorina citri (Liviidae) in California. Biol Control 157:104574. https://doi.org/10.1016/j.biocontrol.2021.104574
Johanowicz DL, Mitchell ER (2000) Effects of sweet alyssum flowers on the longevity of the parasitoid wasps Cotesia marginiventris (Hymenoptera: Braconidae) and Diadegma insulare (Hymenoptera: Ichneumonidae). Fla Entomol 83(1):41. https://doi.org/10.2307/3496226
Köneke A, Uesugi R, Herz A, Tabuchi K, Yoshimura H, Shimoda T, Nagasaka K, Böckmann E (2023) Effects of wheat undersowing and sweet alyssum intercropping on aphid and flea beetle infestation in white cabbage in Germany and Japan. J Plant Dis Prot 130(3):619–631. https://doi.org/10.1007/s41348-023-00730-y
Landis DA, Wratten SD, Gurr GM (2000) Habitat management to conserve natural enemies of arthropod pests in agriculture. Annu Rev Entomol 45:175–201. https://doi.org/10.1146/annurev.ento.45.1.175
Landolt E, Bäumler B (2010) Flora indicativa: Ökologische Zeigerwerte und biologische Kennzeichen zur Flora der Schweiz und der Alpen, 2nd edn. Editions des Conservateurs et Jardin botaniques de la Ville de Genève; Haupt Verlag, Genève, Bern.
Lauber K, Wagner G, Gygax A (2009) Flora Helvetica: Illustrierte Flora der Schweiz. Haupt Verlag, Bern
Lopez L, Liburd OE (2022) Can the introduction of companion plants increase biological control services of key pests in organic squash? Entomol Exp Appl 170(5):402–418. https://doi.org/10.1111/eea.13147
Madeira F, Lumbierres B, Pons X (2022) Contribution of surrounding flowering plants to reduce abundance of Aphis nerii (Hemiptera: Aphididae) on Oleanders (Nerium oleander L.). Hortic 8(11):1038. https://doi.org/10.3390/horticulturae8111038
Munir S, Dosdall LM, Keddie A (2018) Selective effects of floral food sources and honey on life-history traits of a pest–parasitoid system. Entomol Exp Appl 166(6):500–507. https://doi.org/10.1111/eea.12695
Nilsson U, Rännbäck L-M, Anderson P, Eriksson A, Rämert B (2011) Comparison of nectar use and preference in the parasitoid Trybliographa rapae (Hymenoptera: Figitidae) and its host, the cabbage root fly, Delia radicum (Diptera: Anthomyiidae). Biocontrol Sci Techn 21(9):1117–1132. https://doi.org/10.1080/09583157.2011.605518
Parolin P, Bresch C, Desneux N, Brun R, Bout A, Boll R, Poncet C (2012a) Secondary plants used in biological control: a review. Int J Pest Manag 58(2):91–100. https://doi.org/10.1080/09670874.2012.659229
Parolin P, Bresch C, Poncet C, Desneux N (2012b) Functional characteristics of secondary plants for increased pest management. Int J Pest Manag 58(4):368–376. https://doi.org/10.1080/09670874.2012.734869
Perdikis DC, Lykouressis DP (2003) Aphis gossypii (Hemiptera: Aphididae) as a factor inhibiting the survival and population increase of the predator Macrolophus pygmaeus (Hemiptera: Miridae) on cucumber. Eur J Entomol 100(4):501–508. https://doi.org/10.14411/eje.2003.077
Picó FX, Retana J (2003) Seed ecology of a Mediterranean perennial herb with an exceptionally extended flowering and fruiting season. Bot J Linn Soc 142(3):273–280. https://doi.org/10.1046/j.1095-8339.2003.00172.x
Pineda A, Marcos-García MÁ (2008) Use of selected flowering plants in greenhouses to enhance aphidophagous hoverfly populations (Diptera: Syrphidae). Ann Soc Entomol Fr (n. s.) 44(4):487–492. https://doi.org/10.1080/00379271.2008.10697584
Pumariño L, Alomar O (2012) The role of omnivory in the conservation of predators: Orius majusculus (Heteroptera: Anthocoridae) on sweet alyssum. Biol Control 62(1):24–28. https://doi.org/10.1016/j.biocontrol.2012.03.007
Scarlato M, Bao L, Rossing W, Dogliotti S, Bertoni P, Bianchi F (2023) Flowering plants in open tomato greenhouses enhance pest suppression in conventional systems and reveal resource saturation for natural enemies in organic systems. Agric Ecosyst Environ 347:108389. https://doi.org/10.1016/j.agee.2023.108389
Shelton AM, Badenes-Perez E (2006) Concepts and applications of trap cropping in pest management. Annu Rev Entomol 51:285–309. https://doi.org/10.1146/annurev.ento.51.110104.150959
Shrestha B, Finke DL, Piñero JC (2019) The ‘Botanical Triad’: the presence of insectary plants enhances natural enemy abundance on trap crop plants in an organic cabbage agro-ecosystem. InSects 10(6):181. https://doi.org/10.3390/insects10060181
Silva JHC, Saldanha AV, Carvalho RMR, Machado CFM, Flausino BF, Antonio AC, Gontijo LM (2022) The interspecific variation of plant traits in brassicas engenders stronger aphid suppression than the intraspecific variation of single plant trait. J Pest Sci 95(2):723–734. https://doi.org/10.1007/s10340-021-01421-z
Sutter L, Albrecht M (2016) Synergistic interactions of ecosystem services: florivorous pest control boosts crop yield increase through insect pollination. Proceed Biol Sci. https://doi.org/10.1098/rspb.2015.2529
Sutter L, Jeanneret P, Bartual AM, Bocci G, Albrecht M (2017) Enhancing plant diversity in agricultural landscapes promotes both rare bees and dominant crop-pollinating bees through complementary increase in key floral resources. J Appl Ecol 54(6):1856–1864. https://doi.org/10.1111/1365-2664.12907
Sutter L, Albrecht M, Jeanneret P (2018) Landscape greening and local creation of wildflower strips and hedgerows promote multiple ecosystem services. J Appl Ecol 55(2):612–620. https://doi.org/10.1111/1365-2664.12977
Tiwari S, Saville DJ, Sharma S, Shields MW, Wratten SD (2020) Evaluation of potential trap plant species for the wheat bug Nysius huttoni (Hemiptera: Lygaeidae) in forage brassicas. Agr Forest Entomology 22(3):263–273. https://doi.org/10.1111/afe.12379
Zuma M, Njekete C, Konan KAJ, Bearez P, Amiens-Desneux E, Desneux N, Lavoir A-V (2023) Companion plants and alternative prey improve biological control by Orius laevigatus on strawberry. J Pest Sci 96(2):711–721. https://doi.org/10.1007/s10340-022-01570-9
Acknowledgements
We would like to thank all the growers and technicians who contributed to this work by making their land available, maintaining the crops and collecting essential data.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
JK, VD and LS conceived the study. DM, JK, VD and LS collected the data. JK analysed the data and led the writing of the manuscript. All authors contributed to the writing of the manuscript and gave final approval for its publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Handling Editor: Francisco Badenes-Pérez.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Koller, J., Norgrove, L., Dekumbis, V. et al. Pest trap and natural enemy nursery merged in Lobularia maritima?. Arthropod-Plant Interactions (2024). https://doi.org/10.1007/s11829-024-10092-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11829-024-10092-3