Abstract
Background
The EU-wide, cross-sectional observational study of lipid-lowering therapy (LLT) use in secondary and primary care (DA VINCI) assessed the proportion of patients achieving low-density lipoprotein cholesterol (LDL-C) goals recommended by the European Society of Cardiology (ESC)/European Atherosclerosis Society (EAS) guidelines and provided an insight into regional use of LLT in Europe, including Ireland.
Aims
This analysis focuses on data from patients in Ireland who participated in the DA VINCI study.
Methods
The DA VINCI study enrolled patients receiving LLT at primary and secondary care sites across 18 European countries between June 2017 and November 2018. The study assessed the achievement of risk-based 2016 and 2019 ESC/EAS LDL-C goals. This subgroup analysis aimed to evaluate LDL-C goal attainment in an Irish cohort of primary and secondary care patients.
Results
In total, 198 patients from Ireland were enrolled from three primary care and three secondary care centres. Most patients were White and male, and were receiving moderate- or high-intensity statin therapy (most frequently atorvastatin or rosuvastatin). Few patients (< 10%) were receiving combination therapy of statin and ezetimibe. Approximately 60% of patients achieved their 2016 ESC/EAC LDL-C goals while less than half the patients achieved their 2019 ESC/EAS goals. Approximately half of secondary prevention patients achieved their 2016 ESC/EAS goals and only 20% of secondary prevention patients achieved their 2019 ESC/EAS goals.
Conclusions
These results highlight the disparity between dyslipidaemia management in clinical practice in Ireland and guideline recommendations.
Trial registration
ENCePP; EU PAS 22,075; date registered 06 February 2018.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiovascular (CV) disease (CVD) is a major cause of mortality and morbidity, accounting for 45% of all deaths in Europe [1]. In Ireland, an estimated 10,000 people die each year from CVD [2]. It is also associated with a burden on healthcare costs [3]. Overall, CVD-related healthcare costs, including hospitalisations and loss of productivity because of illness, are estimated to cost the EU economy €210 billion per year [1].
Elevated low-density lipoprotein cholesterol (LDL-C) level is a well-established causal risk factor for atherosclerotic cardiovascular disease (ASCVD) [4]. In addition, elevated LDL-C level is directly correlated with an increased risk of CV events, including myocardial infarction and ischaemic stroke [4, 5]. LDL-C reduction with lipid-lowering therapy (LLT) has been proven to lower the incidence of fatal and non-fatal CV events [5, 6]. CV primary and secondary prevention aims to improve patients’ health-related quality of life by reducing the risk of disease progression and hospitalisation. Several landmark trials investigating the efficacy of proprotein convertase subtilisin/kexin 9 inhibitors (PCSK9is) have demonstrated an additional mortality benefit associated with further reductions in LDL-C levels [7, 8]. This has been reflected in the European Society of Cardiology (ESC)/European Atherosclerosis Society (EAS) guidelines on dyslipidaemia management [9, 10]. The 2016 ESC/EAS guidelines recommended LDL-C goals of less than 2.6 mmol/L (100 mg/dL) for individuals at high risk and less than 1.8 mmol/L (70 mg/dL) for those at very high risk [9]. The updated 2019 ESC/EAS guidelines, based on high-quality clinical trial data, recommend more stringent LDL-C goals than those from 2016 for patients with established ASCVD and patients in the moderate-, high-, and very-high-risk primary prevention categories [10]. The updated 2019 ESC/EAS guidelines now recommend at least a 50% reduction in LDL-C levels from the untreated state in addition to LDL-C goals of less than 1.8 mmol/L (70 mg/dL) and less than 1.4 mmol/L (55 mg/dL) for those at high and very high risk, respectively [10]. Furthermore, a new, more aggressive goal of LDL-C less than 1.0 mmol/L has been established for individuals presenting with recurrent CV events [10].
Although it is well recognised that elevated LDL-C level is a causal risk factor for CV events, there remains a disparity between guideline-directed LDL-C goals and observational data on LDL-C levels achieved in real-world settings [4]. Several observational studies assessing the impact of the 2016 ESC/EAS dyslipidaemia guidelines, such as EUROASPIRE, revealed that a high percentage of patients failed to achieve their LDL-C goals [11, 12]. These findings have also been observed in more recent studies assessing the impact and attainment of the 2019 ESC/EAS dyslipidaemia guidelines [12, 13]. In these studies, the proportion of patients attaining their LDL-C goals was found to be even lower than that achieved with the 2016 goals [12, 13]. The EU-wide cross-sectional observational study of lipid-modifying therapy use in secondary and primary care (DA VINCI [ENCePP; EU PAS 22075]) set out to determine the proportion of patients across Europe achieving 2016 ESC/EAS risk-based LDL-C goals and provided an insight into how LLT was being employed in different regions [13]. One hundred and ninety-eight Irish patients were included in the original study. We performed a subgroup analysis of the Irish cohort to characterise the findings of this specific group and gain an understanding of whether CV disease is managed optimally in Ireland, as per guidelines.
Methods
Study design
The DA VINCI study was a cross-sectional observational investigation of 5888 adults receiving LLT in European clinical practice [13]. The study took place across 18 European countries between June 2017 and November 2018. Approximately equal numbers of primary and secondary prevention patients were recruited. Secondary prevention patients included those with coronary artery, cerebrovascular, or peripheral arterial disease. The study protocol was approved by all relevant ethics committees. Amgen sponsored both the original study and this subgroup analysis. The subgroup analysis of the Irish cohort of the DA VINCI study was performed with data collected from 198 patients as part of the original study.
Study aims and outcomes
The DA VINCI study explored LDL-C goal attainment, based on individual CV risk, recommended by the 2016 and 2019 (post hoc analysis) ESC/EAS guidelines across Europe. The study aimed to provide an insight into variability in lipid management among diverse healthcare systems. LDL-C goal attainment was evaluated in patients at LDL-C measurement while receiving stabilised LLT, which was defined as no change in dose or regimen for at least 28 days before LDL-C measurement. The estimated 10-year CV risk at LDL-C measurement was established using the Systematic COronary Risk Evaluation (SCORE) for primary prevention patients and the REACH (REduction of Atherothrombosis for Continued Health) score for secondary prevention patients [14, 15].
Ireland was one of the countries included in the overall DA VINCI study. The current subgroup analysis aimed to assess LDL-C goal attainment in an Irish cohort of primary and secondary care patients.
Data collection and analysis
Full eligibility criteria and methodology are available in the primary DA VINCI study report [13]. Briefly, patients were recruited at routine clinic visits. Only patients being prescribed LLT at enrolment or within 12 months before enrolment and having an LDL-C measurement recorded up to 14 months before enrolment were included. Data were collected using a standardised electronic case report form, and included baseline characteristics, background medical history, previous vascular history, LLT, and most recent lipid profile. Continuous variables were reported as mean, standard deviation, median, interquartile range, and minimum/maximum. Categorical data were reported as numbers and percentages.
Results
Patient characteristics, CV risk profile, and management
The DA VINCI study enrolled 5888 patients from 18 countries and 128 sites across Europe. The Irish cohort comprised 198 patients, from three primary care and three secondary care centres. The age of the patients enrolled ranged from 35 to 87 years, with a mean age of 64 years. Most patients were White and male. Similar baseline characteristics were observed in the Irish cohort and the overall European cohort (Table 1), but there tended to be more White men in the Irish study population (68%) as compared to the overall study (58%).
There was a similar proportion of non-smokers and ex-smokers in the Irish and European cohorts, with proportionally less active smokers in both groups (Table 1). Hypertension (59.6% and 70.3%, in the Irish and European cohorts, respectively) and diabetes mellitus (27.3% and 38.9%, in the Irish and European cohorts, respectively) were the two most prevalent medical comorbidities in both groups, with a tendency for a lower prevalence of hypertension, type 2 diabetes mellitus, chronic kidney disease, and left ventricular hypertrophy in the Irish cohort than in the European cohort (Table 1).
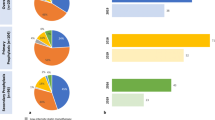
A total of 101 Irish primary prevention patients participated in the study, of whom, 74 were evaluable for LDL-C goal attainment. The majority of primary prevention patients were in the moderate-risk category, measured by SCORE, in both the Irish and overall European cohorts (Fig. 1). Overall, 101 Irish secondary prevention patients were enrolled in the study, and 70 were evaluable for goal attainment. Table 2 describes the manifestations of established ASCVD documented for the patients, as well as previous vascular interventions. By definition, all of these patients were categorised as being at very high risk and were therefore assigned a more stringent LDL-C goal than low-, moderate-, or high-risk patients in both the 2016 and 2019 guidelines.
Most Irish patients were treated with statins (94.1%; most frequently atorvastatin or rosuvastatin). Compared to the overall study population, Irish patients more frequently received high-intensity statins (43.1% and 31.8%, respectively) (Table 3). However, less than half of the Irish patients had had their medication titrated onto the high-intensity statin doses. Overall, 8.6% of patients had a history of statin intolerance and 8.6% had reports of statin side effects. A minority of patients received combination therapy or non-statin LLT (Table 3). In the Irish cohort, 13 patients were receiving a statin/ezetimibe combination, one patient was receiving a statin/PCSK9i combination, with eight patients receiving non-statin LLT (bile acid sequestrants, fibrates, and fish oils).
ESC/EAS guideline LDL-C goal attainment
Forty-five Irish patients were excluded from the goal attainment analyses as they were not receiving stabilised LLT at the time of LDL-C measurement. Of the 153 Irish patients with evaluable data for LDL-C goal attainment, 64.1% (n = 98 [95% confidence interval (CI): 56.2–71.2]) of patients had achieved their LDL-C goals as per the 2016 ESC/EAS dyslipidaemia guidelines (Table 4, Fig. 2). In the post hoc analysis of data for the 2019 ESC/EAS guidelines, only 40.5% (n = 62 [95% CI: 33.1–48.4]) of Irish patients achieved their LDL-C goals (Table 4, Fig. 2).
Achievement of LDL-C goals among the secondary prevention cohort was examined according to the patient’s main vascular bed involvement (coronary artery, cerebrovascular, or peripheral arterial disease). With the exception of patients with cerebrovascular disease, LDL-C goal attainment was low in all groups, with less than or half of the patients achieving their goals (Fig. 3).
According to additional analyses of the Irish cohort by gender and age, males were numerically more likely to achieve their LDL-C goals than females as per the 2016 ESC/EAS guidelines (67.9% versus 55.3%, respectively), but this trend was not seen for the attainment of the 2019 ESC/EAS guidelines (39.6% versus 42.6%). With respect to age, patients > 64 years were numerically more likely than those ≤ 64 years to achieve their LDL-C goals as per both the 2016 ESC/EAS guidelines (68.0% versus 60.3%, respectively) and the 2019 ESC/EAS guidelines (45.3% versus 35.9%).
Discussion
This subgroup analysis set out to assess LDL-C goal attainment in the Irish cohort of the DA VINCI study. Over half of the patients in Ireland who participated in DA VINCI did not achieve the 2019 ESC/EAS LDL-C goals. Similar findings were identified across Europe in the DA VINCI study, as previously reported [13]. These results highlight the disparity between dyslipidaemia management in clinical practice in Ireland compared with guideline recommendations.
The 2016 guidelines were in effect at the time of data collection, yet only half of the patients reached their LDL-C goal. These findings are concerning, given that every patient in this cohort had proven vascular disease and many had already experienced a CV event or required vascular intervention. Despite the guidelines pertaining to all forms of established ASCVD, particular attention is paid to outcomes in coronary artery disease in the literature. Our study highlights poor adherence to guidelines for patients with cerebrovascular disease and peripheral arterial disease. When the more stringent 2019 ESC/EAS LDL-C guidelines were applied, as expected, an even smaller proportion of patients (Irish and overall) reached their LDL-C goal. In the Irish secondary prevention cohort, this was particularly low, with nearly four in five patients failing to reach their LDL-C goal. Among patients with established ASCVD, those with coronary artery disease had the highest rate of goal attainment, with lower rates observed in the cerebrovascular and peripheral arterial disease cohorts. More large-scale clinical trials are required to call to attention the benefits of strict LDL-C control in these cohorts.
As was seen in the secondary prevention subgroup, a lower proportion of patients in the primary prevention cohort achieved the more stringent 2019 risk-based LDL-C goals compared with the 2016 goals. However, compared with the secondary prevention cohort, a higher proportion of primary prevention patients met their LDL-C goals with statin monotherapy. In this regard, it should be noted that the majority of these primary prevention patients were in the moderate-risk category, whereas all of the secondary prevention patients were categorised as very high risk. The ESC/EAS LDL-C goals for high-/very-high-risk patients are considerably more stringent than those at moderate risk. Therefore, the difference in proportions of goal attainment between the primary and secondary prevention cohorts was likely influenced by the distribution of lower patient risk in the primary prevention category (i.e. it is easier to attain LDL-C goals when at lower risk because the levels are not as stringent).
The findings described here are in line with those of previous studies of Irish patients with dyslipidaemia [16]. Irish data from the Dyslipidemia International Study (DYSIS) demonstrated that a significant number of patients had persistent dyslipidaemia despite statin therapy and that patients in Ireland failed to reach their LDL-C goals [16]. Our data from the Irish cohort in the DA VINCI study were also consistent with the results from previous observational studies examining LDL-C goal attainment among patients with ASCVD in Europe [17]. In EUROASPIRE, a large cross-sectional study of patients in Europe, most patients with coronary heart disease did not achieve their 2016 LDL-C goals [17]. Among patients on high-intensity statin therapy, only 27% achieved LDL-C < 1.8 mmol/L. Similarly, a study from the START registry in Italy examining goal attainment using the 2016 and 2019 ESC/EAS guidelines showed that 58% of patients achieved their 2016 LDL-C goals, while only 3% achieved their 2019 LDL-C goals [12]. As was also observed in our analysis, LDL-C goal attainment reduced considerably when 2019 goals were applied [10]. Findings from EUROASPIRE suggested that men and those at least 60 years of age may have been somewhat more likely to achieve their goals [17], and we observed similar trends in our analysis.
Given the hypothesis that ‘lower is better’, it is essential that we identify and address inadequacies in dyslipidaemia management. The 2019 ESC/EAS guidelines suggest up-titration of statins and subsequent addition of non-statin LLT to achieve LDL-C goals, with expeditious follow-up for a reassessment of LDL-C levels [10]. Despite these recommendations and the observed low level of LDL-C goal attainment, less than half the patients in the Irish cohort were treated with high-intensity statins. Furthermore, only 20 patients received ezetimibe (13 in combination) and nine patients were treated with other forms of non-statin LLT (PCSK9is, bile acid sequestrants, fish oils, or fibrates). Compared with other European countries, limited resources are available for dyslipidaemia management in Ireland, with only six designated lipid clinics and one specialist lipid nurse being available [18] and no structured programme in primary care for primary prevention and a limited secondary prevention programme implemented to date. Thus, the development of a structured care programme in primary care and education on guideline-directed lipid management in primary care and general secondary care clinics is of the utmost importance. Emphasis should be placed on achieving LDL-C goals according to EAS/ESC 2019 guidelines. In addition, choosing high-intensity statins for high-risk patients and close monitoring of patients with repeated LDL-C measurements should be encouraged to facilitate dose up-titration and use of combination therapy to achieve LDL goals.
Studies have shown that very low levels of LDL-C are not associated with detrimental health effects or concerns [4, 19]. Every 1 mmol/L reduction in LDL-C is associated with an approximately 21% reduction in major vascular events per annum [20]. As newer guidelines continue to lower the desired level of LDL-C, this raises the question of whether statins alone can allow us to meet the required goals. In this regard, non-statin LLT can be effective when prescribed in combination with statins, particularly when higher doses of statins cannot be tolerated owing to side effects. The addition of ezetimibe to statins has been shown to reduce LDL-C levels up to an additional 24%, resulting in improved CV outcomes [19]. With the emergence of PCSK9is, which reduce LDL-C levels by 50–60%, these benefits continue due to additional reductions in LDL-C concentration [7, 8]. In this study, only one Irish patient was treated with a PCSK9i, most likely because DA VINCI was conducted prior to reimbursement of PCSK9is in Ireland. One PCSK9i, evolocumab, was approved for reimbursement in July 2019; however, strict national reimbursement criteria (based partly on cost-effectiveness and budget impact analyses) limit its widespread use [21]. To date, other new cholesterol-lowering drugs such as bempedoic acid and inclisiran are not yet available in Ireland. Increasing the availability of novel, potent, non-statin LLTs may facilitate successful dyslipidaemia guideline adherence in Ireland, thereby improving patient outcomes. Future analyses from the overall DA VINCI population will be conducted to better understand how optimising treatment—including optimisation of statin monotherapy, as well as combination therapy (in accordance with ESC/EAS dyslipidaemia guidelines)—impacts on the risk of subsequent cardiovascular events.
Our study had limitations. It was a cross-sectional observational study; as such, LDL-C levels were only measured at a single point in time. Importantly, there are no data available on medication compliance, drug intolerance, or changes in LDL-C levels following dose titration. The data were also collected before the publication of the 2019 ESC/EAS guidelines. Thus, post hoc analysis against newer goals was performed with the knowledge that patients were being treated using the more lenient 2016 ESC/EAS guidelines. If the study was to be repeated now, at a time when the lower LDL-C goals are more routinely used, we are hopeful that goal attainment would be higher when 2019 goals were applied.
Conclusion
Our study demonstrates that dyslipidaemia management is suboptimal in Ireland, even though well-established guidelines (based on high-quality evidence) are available to healthcare professionals.
In secondary prevention patients, nearly half failed to achieve the 2016 LDL-C goals. Emphasis should be placed on prescriber education with respect to the following: current dyslipidaemia management guidelines, the correct use of statins, appropriate use of combination therapy, and appropriate patient monitoring. Patient education regarding medication adherence is also important. In high-risk patients with inadequate LDL-C lowering on maximally tolerated conventional therapy, including patients with familial hypercholesterolaemia, and those with established CV disease, consideration should be given to the use of additional non-statin LLT to achieve LDL-C goals.
Availability of data and material
Qualified researchers may request data from Amgen clinical studies. Complete details are available at https://wwwext.amgen.com/science/clinical-trials/clinical-data-transparency-practices.
References
European cardiovascular disease statistics (2017). Available from: https://ehnheart.org/cvd-statistics/cvd-statistics-2017.html
CSO statistics yearly summary (2019). Available from: https://www.cso.ie/en/releasesandpublications/ep/p-vsys/vitalstatisticsyearlysummary2018/.
Leal J, Luengo-Fernández R, Gray A et al (2006) Economic burden of cardiovascular diseases in the enlarged European Union. Eur Heart J 27(13):1610–1619. https://doi.org/10.1093/eurheartj/ehi733
Packard C, Chapman MJ, Sibartie M et al (2021) Intensive low-density lipoprotein cholesterol lowering in cardiovascular disease prevention: opportunities and challenges. Heart 107(17):1369–1375. https://doi.org/10.1136/heartjnl-2020-318760
Schubert J, Lindahl B, Melhus H et al (2020) Low-density lipoprotein cholesterol reduction and statin intensity in myocardial infarction patients and major adverse outcomes: a Swedish nationwide cohort study. Eur Heart J 42(3):243–252. https://doi.org/10.1093/eurheartj/ehaa1011
Groenhof TKJ, Kofink D, Bots ML et al (2020) Low-density lipoprotein cholesterol target attainment in patients with established cardiovascular disease: analysis of routine care data. JMIR Med Inform 8(4):e16400. https://doi.org/10.2196/16400
Sabatine MS, Giugliano RP, Keech AC et al (2017) Evolocumab and clinical outcomes in patients with cardiovascular disease. N Engl J Med 376(18):1713–1722. https://doi.org/10.1056/NEJMoa1615664
Schwartz GG, Steg PG, Szarek M et al (2018) Alirocumab and cardiovascular outcomes after acute coronary syndrome. N Engl J Med 379(22):2097–2107. https://doi.org/10.1056/NEJMoa1801174
Catapano AL, Graham I, De Backer G et al (2016) 2016 ESC/EAS guidelines for the management of dyslipidaemias. Eur Heart J 37(39):2999–3058. https://doi.org/10.1093/eurheartj/ehw272
Mach F, Baigent C, Catapano AL et al (2020) 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J 41(1):111–188. https://doi.org/10.1093/eurheartj/ehz455
Kotseva K, De Backer G, De Bacquer D et al (2019) Lifestyle and impact on cardiovascular risk factor control in coronary patients across 27 countries: results from the European Society of Cardiology ESC-EORP EUROASPIRE V registry. Eur J Prev Cardiol 26(8):824–835. https://doi.org/10.1177/2047487318825350
De Luca L, Arca M, Temporelli PL et al (2020) Current lipid lowering treatment and attainment of LDL targets recommended by ESC/EAS guidelines in very high-risk patients with established atherosclerotic cardiovascular disease: insights from the START registry. Int J Cardiol 316:229–235. https://doi.org/10.1016/j.ijcard.2020.05.055
Ray KK, Molemans B, Schoonen WM et al (2020) EU-wide cross-sectional observational study of lipid-modifying therapy use in secondary and primary care: the DA VINCI study. Eur J Prev Cardiol. https://doi.org/10.1093/eurjpc/zwaa047
Conroy RM, Pyörälä K, Fitzgerald AP et al (2003) Estimation of ten-year risk of fatal cardiovascular disease in Europe: the SCORE project. Eur Heart J 24(11):987–1003. https://doi.org/10.1016/s0195-668x(03)00114-3
Wilson PW, D’Agostino R Sr, Bhatt DL et al (2012) An international model to predict recurrent cardiovascular disease. Am J Med 125(7):695-703.e691. https://doi.org/10.1016/j.amjmed.2012.01.014
Horgan S, Crowley J, Feely J et al (2011) Prevalence of dyslipidaemia in statin-treated patients in Ireland: Irish results of the DYSlipidaemia International Study (DYSIS). Ir J Med Sci 180(2):343–349. https://doi.org/10.1007/s11845-011-0702-8
Reiner Ž, De Backer G, Fras Z et al (2016) Lipid lowering drug therapy in patients with coronary heart disease from 24 European countries–findings from the EUROASPIRE IV survey. Atherosclerosis 246:243–250. https://doi.org/10.1016/j.atherosclerosis.2016.01.018
Agar R, Prendergast M, Maher V (2020) Evaluation of lipid services in the Republic of Ireland. Ir J Med Sci 189(3):925–931. https://doi.org/10.1007/s11845-020-02187-6
Cannon CP, Blazing MA, Giugliano RP et al (2015) Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med 372(25):2387–2397. https://doi.org/10.1056/NEJMoa1410489
Mihaylova B, Emberson J, Blackwell L et al (2012) The effects of lowering LDL cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomised trials. Lancet 380(9841):581–590. https://doi.org/10.1016/s0140-6736(12)60367-5
Managed access protocol – Repatha® (evolocumab) (2020). Available from https://www.hse.ie/eng/about/who/cspd/ncps/medicines-management/pcsk9-inhibitors/hse-managed-access-protocol-repatha1.pdf
Acknowledgements
The authors would like to thank Ryan Woodrow (Aspire Scientific, Bollington, UK) and Sinéad Flannery (PharmaGenesis London, London, UK) for editorial support, which was funded by Amgen. K. K. R. acknowledges support from the NIHR Imperial Biomedical Research Centre and additional support from the NIHR ARC for Northwest London. The authors also acknowledge support from Catherine Markham (Tallaght University Hospital), Mary Hall (St James Hospital), and David Farrell (Beaumont Hospital).
Funding
Open Access funding provided by the IReL Consortium. This study was funded by Amgen (Europe) GmbH.
Author information
Authors and Affiliations
Contributions
G. O., C. O’C., and V. M. contributed substantially to the sub-study design and concept; P. O’C., G. O., C. O’C., M. S., and C. K. were involved in data acquisition; G.O., C. O’C., and M. S. conducted the data analyses; and all authors assisted with interpretation of the data. K. K. R. conceived, designed, secured funding, and supervised the overall DA VINCI study. All authors were involved in drafting the manuscript, provided critical revisions for important intellectual content, approved the final version submitted for publication, and agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Ethics approval
All relevant ethics approvals were obtained before the study started from the Ethics committee at Tallaght University Hospital and the Irish College of General Practice.
Consent to participate
All study participants provided individual informed consent.
Competing interests
G. O. reports no conflicts of interest; C. O’C. reports no conflicts of interest; C. K. reports no conflicts of interest; J. G. reports grant from Amgen and honoraria from Sanofi, Daiichi Sankyo, Novartis, AstraZeneca, and Boehringer Ingelheim; P. O’C. reports honoraria from Amgen; B. M. reports honoraria from Bayer, Boehringer Ingelheim, Bayer, and AstraZeneca and Novartis; K. K. R. reports grant from Amgen during the conduct of the study, and personal fees from AbbVie, Aegerion, Akcea, Algorithm, AstraZeneca, Bayer, Boehringer Ingelheim, Cerenis, Cipla, Dr Reddy’s, Esperion, Kowa, Lilly, The Medicines Company, Novartis, Silence Therapeutics, Takeda, and Zuellig Pharma, and grants and personal fees from Amgen, Daiichi Sankyo, MSD, Pfizer, and Sanofi/Regeneron, outside the submitted work. M. S. is an employee and shareholder of Amgen Ltd. V. M. reports Research Grant support from Sanofi and Amgen. He also has received honoraria for lecturing and advisory board participation for Servier, Nova Nordisk and Novartis.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Offiah, G., O’Connor, C., Kennedy, C. et al. The DA VINCI study: is Ireland achieving ESC/EAS guideline–directed LDL-C goals?. Ir J Med Sci 192, 1077–1084 (2023). https://doi.org/10.1007/s11845-022-03050-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11845-022-03050-6