Abstract
The developments in HIV treatments have increased the life expectancy of people living with HIV (PLWH), a situation that makes cardiovascular disease (CVD) in that population as relevant as ever. PLWH are at increased risk of CVD, and our understanding of the underlying mechanisms is continually increasing. HIV infection is associated with elevated levels of multiple proinflammatory molecules, including IL-6, IL-1β, VCAM-1, ICAM-1, TNF-α, TGF-β, osteopontin, sCD14, hs-CRP, and D-dimer. Other currently examined mechanisms include CD4 + lymphocyte depletion, increased intestinal permeability, microbial translocation, and altered cholesterol metabolism. Antiretroviral therapy (ART) leads to decreases in the concentrations of the majority of proinflammatory molecules, although most remain higher than in the general population. Moreover, adverse effects of ART also play an important role in increased CVD risk, especially in the era of rapid advancement of new therapeutical options. Nevertheless, it is currently believed that HIV plays a more significant role in the development of metabolic syndromes than treatment-associated factors. PLWH being more prone to develop CVD is also due to the higher prevalence of smoking and chronic coinfections with viruses such as HCV and HBV. For these reasons, it is crucial to consider HIV a possible causal factor in CVD occurrence, especially among young patients or individuals without common CVD risk factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Human Immunodeficiency Virus (HIV) infection is a chronic disease that is a known risk factor for CVD, a leading cause of mortality worldwide [1]. It is estimated that rates of morbidity and mortality from CVD are 50–100% higher in those with HIV than in a well-matched population without HIV infection [2]. Among the most prevalent cardiovascular conditions in people living with HIV (PLWH) are hypertension, hypercholesterolemia, low HDL-cholesterol, hypertriglyceridemia, and high serum glucose. Moreover, PLWH are more prone to experience ischemic stroke, arrhythmias, heart failure, myocardial infarction, and sudden cardiac death [2, 3]. The mechanisms leading to increased cardiovascular risk in PLWH include viral stimulation of pro-inflammatory molecules, CD4 + lymphocyte depletion, increased intestinal permeability, microbial translocation, and altered cholesterol metabolism [4]. Moreover, the higher prevalence of smoking and other chronic viral coinfections also play important roles in the altered pro-inflammatory status of PLWH [5, 6].
Antiretroviral therapy (ART), the treatment of choice, has significantly contributed to the management of HIV infection and therefore to the increase in life expectancy for PLWH [7]. However, it has been reported that ART can not only suppress the virus and restore immune system function but also may be harmful in terms of cardiovascular risk [8, 9]. Such unfavorable effects depend on the form of ART and the specific drugs applied; however, it is currently believed that antiretroviral therapy plays only a minor role in cardiovascular risk in comparison to HIV itself [9].
Since cardiovascular disease is more prevalent among PLWH than in the general population of the same age, this study aims to explore the finding that early CVD development may be a symptom of HIV infection [10]. In PLWH, similarly to the general population, there are both unmodifiable and modifiable risk factors for CVD. Therefore, HIV infection should be considered in patients with early occurrence of dyslipidemia, hypertension, and high serum glucose levels, with a focus on individuals without the usual risk factors for CVD.
Unmodifiable CVD Risk Factors in PLWH
Age
In the general population, age ≥ 45 years for men and ≥ 55 years for women is considered one of the main unmodifiable risk factors of CVD [8]. Studies have reported that multiple chronic comorbidities, including CVD, occur in PLWH approximately a decade earlier than in the general population [10]. There are also reports of increased prevalence of early atherosclerosis and heart failure among PLWH and the beginning of excess heart age in early adulthood [11, 12]. Moreover, studies have suggested that cardiovascular manifestations of HIV infection, especially low HDL cholesterol and hypertriglyceridemia, may already occur in childhood [13]. As a result, the risk of death from CVD appears to be significantly higher among PLWH from 25 to 64 years for every 10-year age group, ranging from a 31% elevated risk among those aged 55–64 years to 202% among those aged 25–34 years in comparison to the general population [14].
Gender
It has been well documented that in the general population, the risk of CVD is higher in men than in premenopausal women [15]. Interestingly, studies have shown that women living with HIV have 1.5 to 2-fold higher cardiovascular disease risk than men living with HIV [16]. That difference is pronounced in premenopausal women and seems to diminish in old age [17]. The reasons for the increased CVD risk in women seem to be multivariate, and the traditional risk factors may occur more often among women than among men living with HIV. The prevalence of cigarette smoking is higher in women than in men among those living with HIV [18]. Moreover, women living with HIV are more likely to be overweight or obese and to gain weight following antiretroviral therapy compared to men [19, 20]. From a pathophysiological point of view, 59 differentially expressed genes were found in intermediate monocytes in women living with HIV, and these included known atherosclerosis genes such as the liver X receptor gene nuclear receptor subfamily 1 group H member 2 (NR1H2), Nexilin (NEXN), TNF Receptor Associated Factor 1 (TRAF1), Toll-like Receptor 7 (TLR7), and Galectin 3 Binding Protein (LGALS3BP) [21]. Women living with HIV experience menopause earlier than women in the general population, a phenomenon that is associated with an increased risk of visceral fat, reduced muscle mass, and changes in bone density, all of which are HIV-independent but are well-known risk factors for CVD [22].
Studies have shown that transgender women receiving gender-affirming hormonal therapy (GAHT) experience a gain in fat, a decline in lean body mass, and an increase in insulin resistance, all of which are risk factors for cardiovascular disease [23]. In contrast, these effects are not seen in transgender men, possibly because testosterone used as GAHT decreases body fat and increases lean body mass, effects that usually lead to unchanged body mass index (BMI) [24]. Cyproterone acetate, a widely used GAHT in transgender women, leads to an increase in body fat, especially in the android region [24]. Transgender PLWH may be additionally prone to CVD due to other complex factors such as drug-drug interactions with ART, social stressors, and stigma [25,26,27].
Race
In the general population, the prevalence of CVD is highest in Black individuals [28]. That association seems to occur also in PLWH. Studies have shown that CVD-related hospitalization rates in PLWH were 45% higher for African Americans than Whites [29]. One of the probable mechanisms explaining this epidemiology is that Black PLWH may be more amenable to inflammation since it has been observed that they experience over 50% higher D-dimer levels while having a detectable HIV viral load in comparison to Whites [30]. Data concerning the mechanism explaining this phenomenon are limited; however, in a study of the general population, several fibrinogen gene polymorphisms, including the Thr312Ala alpha chain variant and the FGG-10,034 C/T variant seem to be associated with 20% higher D-dimer concentrations and may partially explain the racial differences in D-dimer concentration [31].
The lowest hospitalization rates due to CVD have been observed in Asian men living with HIV. Compared to Black men living with HIV, Asians had a three-fold lower rate of hospitalization due to cardiovascular reasons [32]. Those inequalities may result from a lesser number of CVD risk factors among Asians. The cardiovascular health score defined by the American Heart Association comprises seven health factors and behaviors: dietary quality, smoking, physical activity, body mass index, blood pressure, cholesterol, and blood glucose. The average is 0.24 points lower in Asians than Whites and 0.47 points lower than in Blacks [33].
Modifiable CVD Risk Factors in PLWH
CD4 + lymphocyte Depletion and Recovery
Untreated HIV infection is associated with a gradual depletion of memory CD4 + lymphocyte count, resulting in higher IL-2 levels and thus increasing the incidence of atherosclerosis and other related inflammatory diseases, including CVD [34]. A lower CD4 + lymphocyte count may be related to the impairment of endothelial function, since it has been reported that circulating microparticles, mostly platelets and endothelial particles, are strongly associated with arterial stiffness in PLWH with advanced immune suppression [35]. A low CD4 + level is also associated with elevated blood pressure, blood glucose, and triglycerides, and decreased HDL cholesterol [36]. There are also reports that PLWH having a CD4 + lymphocyte count < 350 cells/μl have a 30% higher likelihood of having a low HDL cholesterol concentration compared to those with CD4 cell counts > 350 cells/μl [37].
Studies have shown that PLWH with a low CD4 + lymphocyte count have higher proportions of T helper type 17 cells (Th17) and senescent cells, which are associated with higher cardiovascular risk [38]. Senescent cells are known to be connected to atherosclerosis and cardiac fibrosis, and sustained production of Th17 may be a pro-inflammatory factor [39, 40]. The prevalence of clonal hematopoiesis, which has been associated with higher cardiovascular mortality, is higher in PLWH with lower CD4 + lymphocyte counts and residual HIV transcriptional activity [41].
PLWH with lower CD4 + lymphocyte counts has been reported to have low cholesterol efflux and higher sensitivity to C-reactive protein (hs-CRP), both of which are CVD risk factors [42]. Cholesterol efflux capacity is a proinflammatory factor associated with atherosclerosis: a lower cholesterol efflux is negatively associated with the elevation of many proinflammatory molecules, including CRP, fibrinogen, interleukin-6 (IL-6), and serum amyloid A, and positively associated with cardiovascular mortality [43].
Concerning CD4 + lymphocyte count and cardiovascular risk, a special population of patients is those PLWH with poor immunological reconstruction (immunological non-responders; INRs), the patients with persistently lower CD4 + counts and CD4:CD8 ratios despite receiving effective antiretroviral therapy. INRs reportedly have higher rates of mortality due to cardiovascular disease [44]. However, after ART initiation, increases in HDL and LDL-cholesterol levels were observed in INRs, a result that makes it difficult to explicitly assess cardiovascular risk [45].
Microbial Translocation
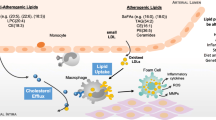
Microbial translocation is a hallmark of HIV disease progression. It is defined as the movement of microorganisms or microbial products from the gastrointestinal mucosa into the systemic circulation [46]. The malfunctioning of the barrier leads to an enhanced microbial translocation that further leads to immune activation and inflammation, thereby increasing the risk of cardiovascular disease via pro-inflammatory mechanisms [47].
During infection, the depletion of CD4 + lymphocytes involves the Th17 CD4 + lymphocyte population, the role of which is to defend against various pathogens at mucosal barriers such as the gastrointestinal tract [48]. The depletion of these cells leads to an imbalance of the Th17/Treg ratio and enhanced production of cytokines, including IL-6, IL-17, IL-1β, IL-12, and IL-4, which disrupts epithelial junctions in the gastrointestinal mucosal barrier and therefore leads to increased microbial translocation [49, 50]. There are known markers of microbial translocation, including plasma levels of lipopolysaccharide and soluble CD14, which are bacterial products, and (1➔3)-β-D-Glucan, a fungal product, that are elevated in untreated PLWH [51, 52]. In addition to Th17 CD4 + lymphocytes, the decrease of mucosal-associated invariant T cells (MAIT) induced by chronic inflammation may contribute to increased susceptibility to microbial translocation. It has been suggested that HIV triggers highly activated MAITs to migrate to the colorectal mucosa where they are later subjected to bacteria-induced apoptosis [53]. This phenomenon is followed by increased levels of the proinflammatory cytokines IL-12 and IL-18 and thus an elevated risk of CVD [54].
Other indicators of intestinal damage are intestinal fatty acid-binding protein (I-FABP), zonulin, and regenerating islet-derived protein-3α (REG3α), all of which are considered intestinal permeability markers [55]. Both I-FABP and REG3α plasma levels are significantly elevated in PLWH not receiving ART, and they remain higher even after the introduction of ART compared to healthy controls [56, 57]. REG3α can be used to assess the degree of gut damage and systemic immune activation, and its plasma levels are positively correlated with other proinflammatory biomarkers such as IL-6, IL-8, CXCL13, and IDO-1, the fungal translocation product (1➔3)-β-D-Glucan, and the HIV viral load [58]. Zonulin levels are also elevated in PLWH, causing unclenching of the tight junctions between gut epithelial cells, leading to increases in permeability and macromolecule absorption [59]. I-FABP is involved in the uptake and transport of long-chain fatty acids from the intestinal lumen and may be a marker for mucosal compromise or injury [60]. There are reports that both I-FABP and zonulin can be used to predict mortality in ART-treated PLWH [61].
HIV-related microbial translocation may also be the result of microbial dysbiosis, primarily expressed as decreased diversity or the outgrowth of potentially pathogenic bacteria [62]. A higher proportion of opportunistic pathogens may promote AIDS-related infections, and lower abundances of butyrate-producing bacteria may induce inflammatory bowel disease [63–65]. Likewise, microbial dysbiosis can lead to activation of the gut and peripheral T cells and increases in plasma pro-inflammatory factors such as TNF-α and soluble CD14 [44].
Dyslipidemia in PLWH
Since the beginning of the HIV epidemic, it has been reported that metabolic syndrome is twice as frequent in PLWH than in the general population [66]. This may be due to altered lipid metabolism causing low HDL cholesterol and hypertriglyceridemia, factors that are considered high-risk lipid profiles for atherosclerosis and cardiovascular disease [67]. Moreover, the prevalence of hypertriglyceridemia, lower HDL cholesterol, and glucose abnormalities are much more common in younger PLWH than in older healthy controls [68]. It has been reported that low HDL cholesterol and hypertriglyceridemia may already occur in children living with HIV [13].
Studies have shown that untreated HIV infection is associated with cardiovascular abnormalities, especially endothelial dysfunction and carotid intima-media thickening [69]. A possible reason for this is the synthesis of Tat by infected cells. The Tat protein elevates the expression levels of IFN-γ, TNF-α, IL-6, and IL-17 and therefore induces apoptosis of endothelial cells. This enables low-density lipoproteins to permeate the sub-endothelial space, thereby causing atherosclerotic lesions [70]. Thus, there is an association between HIV viral load and the risk of dyslipidemia, since the larger number of HIV copies promotes the expression of adhesive proteins and cytokines such as IFN-γ, Il-1β, IL-8, IL-15, and IL-17 [37]. Another possible explanation for the altered lipid metabolism in untreated HIV infection could be the impact of TNF-α decreasing the activity of adipose tissue lipoprotein lipase, an enzyme whose role is to hydrolyze the triacylglycerol component of chylomicrons and VLDL into non-esterified fatty acids and monoacylglycerols [71].
A relationship between lipid metabolism and CD4 + count has also been suggested: PLWH with lower CD4 + lymphocyte counts were reported to have lower concentrations of HDL cholesterol and higher levels of triglycerides than PLWH with higher CD4 + lymphocyte counts [37]. Apart from a low CD4 + lymphocyte count, a history of AIDS-defining events was also reported to be associated with higher total cholesterol and triglyceride concentrations; however, improvement over time has been observed, generally due to the use of lipid-lowering agents [72].
Hypertension in PLWH
The estimated prevalence of hypertension among PLWH varies from 4 to 50% depending on the country and on the quality of the available data [73]. Despite effective antihypertensive drugs, the achievement of blood pressure control in PLWH remains a challenge [74].
Pathophysiologic mechanisms of hypertension in PLWH are a combination of typical, well-known factors occurring in the general population and the chronic inflammation resulting from HIV infection. It has been reported that higher levels of IL-17 A, IFN-γ, IL-6, and CRP were significantly associated with hypertension in ART-treated PLWH [75]. Moreover, the levels of intermediate monocytes CD14 + 16 + were increased with higher HIV viral load, and this may lead to microbial translocation that drives systemic inflammation [76]. All of these factors enhance the activation of the renin-angiotensin-aldosterone system, a key factor in the development of hypertension [77]. Older age, high BMI, obesity, previous cardiovascular events, chronic kidney disease, a family history of hypertension, and dyslipidemia are traditional risk factors common in PLWH, all of which contribute to the development of hypertension [78].
Besides HIV infection, ART is another risk factor for hypertension in PLWH, since during ART the risk of hypertension is over 1.5-fold higher compared with ART-naïve patients [79]. The negative role of ART in arterial blood pressure involves protease inhibitors (PI) and integrase inhibitors (InSTI) [80]. The use of PIs is associated with carotid artery intima-media thickness and arterial stiffness progression, and InSTIs may promote weight gain and therefore increase the risk of hypertension [81, 82].
Glucose Metabolism in PLWH
It has been reported that PLWH have higher leptin concentrations, and this may increase central fat mass, worsen insulin sensitivity, and lead to higher glucose levels [83]. Another mechanism of altered glucose metabolism in PLWH may involve lower adiponectin levels that are associated with an increased risk of coronary stenosis [84]. ART may also negatively impact glucose metabolism, as a higher prevalence of insulin resistance was shown in PLWH receiving nucleoside reverse transcriptase inhibitors (NRTI) and PI treatment [85]. PLWH with diabetes mellitus have higher cardiovascular risk according to the Framingham equation and the RAMA-EGAT score, and they more often develop cerebrovascular complications or chronic kidney disease than non-diabetic PLWH [86].
Type 2 diabetes mellitus poses a burden for PLWH, especially for women. It is estimated that the prevalence of diabetes mellitus among women living with HIV is 23% compared with 16% among men living with HIV [87]. Moreover, women living with HIV have a 1.31 greater odds of acquiring diabetes mellitus in comparison to women without HIV infection [87]. Type 2 diabetes, as it involves prolonged hyperglycemia and insulin resistance, impacts the formation of advanced glycation end products and overproduction of reactive oxygen species and activation of protein kinase C, further leading to chronic vascular inflammation resulting in the development of atherosclerotic cardiovascular disease [88].
Obesity in PLWH and Physical Activity
Obesity as a major metabolic syndrome is one of the traditional risk factors for CVD [89]. A study based on United States registries revealed that among PLWH, 19% of men and 42% of women are obese, while in the general United States population one in three adults is obese [90]. Currently, obesity is less prevalent in PLWH than in the general population; however, the BMI of PLWH has been increasing at a rate more than three times that of the HIV-negative population, and it is estimated that it can soon exceed that of the general population [91].
ART also has a crucial role in increasing the rate of obesity in PLWH [92]. Among widely used antiretroviral agents, weight gain is largely associated with InSTIs, especially bictegravir and dolutegravir [93]. The non-nucleoside reverse transcriptase inhibitors (NNRTIs) such as rilpivirine and NRTIs such as tenofovir alafenamide (TAF) also have a higher potential to cause weight gain than other drugs from these classes of ART [93]. A study analyzing mitochondrial DNA haplogroups in PLWH gaining weight on ART observed that the European haplogroup clade UK and the African haplogroup L3 were associated with significantly greater weight gain after switching to InSTI-based ART [94]. Studies have also suggested the role of direct ART interference with the melanocortin 4 receptor, since modulation of the melanocortin system can influence food intake and body weight. However, these results are currently debatable [95].
Physical activity plays a beneficial role in the reduction of CVD risk in both PLWH and the general population [96]. However, previous observational studies have demonstrated that the level of physical activity of PLWH is lower compared with the general population, a factor that may also have an impact on the increased prevalence of cardiovascular disease [97]. It has been reported that physical activity decreases the risk of CVD and increases the quality of life in PLWH [98].
Smoking
Smokers are more prone to develop heart failure, atrial fibrillation, venous thromboembolism, and ischemic episodes [6]. The mechanisms by which smoking increases CVD risk generally involve endothelial function, as smoking leads to the impairment of the endothelial cells’ ability to perform repair mechanisms. This in turn results in increased levels of total and apoptotic circulating endothelial microparticles and progenitor cells in smokers [99]. Smoking also impairs endothelium-independent vasodilatation; nitrate-mediated and flow-mediated arterial dilation were lower in smokers than in the non-smoking population [100]. Furthermore, smoking leads to an increased expression of adhesion molecules and proinflammatory cytokines, including IL6, TNF-α, and IL1β [101]. These effects seem to concern not only traditional tobacco smoke; the use of alternative smoking products (e.g., e-cigarettes) was also associated with increased adhesion of monocytes to endothelial cells and increased ICAM-1 and VCAM-1 expression, although with a smaller effect size [102].
Smoking also plays a crucial role in CVD development among PLWH, since PLWH smoke two to three times more than the general population [103]. Smoking is a factor that shortens life expectancy, and in PLWH who smoke, mortality rates of three times those of nonsmokers without HIV infection have been observed. Moreover, tobacco use dramatically increases the mortality risk among PLWH [104]. Additionally, PLWH who smoke tobacco are less likely to quit. One of the possible reasons for the difficulty in quitting is the relatively higher nicotine metabolism in PLWH as measured by the nicotine metabolite ratio (NMR, 3-hydroxycotinine/cotinine). High nicotine metabolism is also responsible for a lower response to transdermal nicotine therapy [105].
Coinfections
Another important risk factor for CVD development and progression is the presence of coinfections of HIV with other viruses [106]. It has been estimated that approximately 10–15% of the mortalities in PLWH are due to liver diseases, primarily viral hepatitis [107]. Chronic HBV and HCV infections are prevalent among PLWH since the diseases are transmitted through similar routes as HIV [108]. Several reports have suggested that people with HIV/HCV coinfection have elevated levels of plasma inflammation and microbial translocation biomarkers, especially sCD14 and IL-6, compared to PLWH [109]. Patients with HIV/HBV coinfection are more likely to have increased serum TNF-α, IL-6, IL-8, and IL-12p70 concentrations [110]. Additionally, it has been reported that mucosal-associated invariant T cells (MAIT) are depleted in chronic viral infections, a factor that may contribute to increased susceptibility to microbial translocation and therefore to elevated CVD risk [111].
Likewise, the herpesviruses CMV and EBV also appear to play important roles in the risk of CVD in PLWH. CMV can disrupt epithelial junctions in the gastrointestinal tract, thereby enhancing microbial translocation [112]. Chronic CMV infection is also associated with higher serum IL-6 levels and higher proportions of CMV pp65 (NLV)-specific CD8 + T cells [113]. HIV/EBV coinfection was found to be associated with higher IFN-γ, TGF-β1, and IL-2 expression levels [114].
Metabolic Effects of Antiretroviral Therapy
The most frequently used antiretroviral medication groups are integrase inhibitors, nucleoside reverse transcriptase inhibitors, non-nucleoside reverse transcriptase inhibitors, and protease inhibitors. According to the European AIDS Clinical Society, the majority of currently recommended regimens are based on InSTI. Another first-line treatment is based on doravirine (NNRTI) instead of InSTI. Common alternative regimens allow the usage of darunavir, a protease inhibitor. All recommended schemes of ART contain either one or two NRTIs [115]. Novel ARTs are marked by their high potency, low toxicity, and high effectiveness [116].
Integrase inhibitors are a relatively new class of ART that are supposed to have better efficacy, reduced treatment discontinuation, and higher genetic barrier to drug resistance than older classes of ART [117]. In terms of cardiovascular risk, InSTIs are generally associated with weight gain, obesity, and weight-related comorbidities [118]. Among InSTIs, PLWH taking bictegravir and dolutegravir are at greater risk of weight gain compared to elvitegravir [93]. The demographic factors associated with an increase in BMI are female gender and Black race [93]. Regardless of weight gain, the cardiovascular risk as assessed by the incidence of major adverse cardiac events such as myocardial infarction, ischemic stroke, coronary artery bypass grafting, and percutaneous coronary intervention appears to be decreased among patients receiving InSTI-based regimens in comparison to other classes of ART [119].
The potentially adverse cardiovascular effects of NRTIs are mitophagy-associated endothelial toxicity and mitochondrial oxidative stress [120]. The decrease in mitochondrial DNA copy number in late-passage human aortic endothelial cells and the elevation of senescence-associated β-galactosidase accumulation have been observed in PLWH receiving NRTIs [120]. Moreover, NRTI administration seems to induce increases in the production of reactive oxygen species, accumulation of β-galactosidase, and diminished ATP-linked respiration [121]. The safety profiles of TAF and tenofovir disoproxil (TDF), two widely used forms of tenofovir, show that even a change in the form of the same drug may result in a huge difference in adverse effects. TAF is generally associated with a better safety profile but a possible increase in cardiovascular risk after the switch from ART regimens containing TDF to TAF, especially via increases in total and LDL cholesterol and BMI [93, 122].
Doravirine has beneficial metabolic profiles and can reduce the risk of CVD. Studies have demonstrated decreases in total cholesterol, LDL cholesterol, and triglycerides after switching to doravirine from different regimens [123]. In contrast, PIs are generally considered to have unfavorable effects in terms of cardiovascular risk. The mechanisms responsible include the triggering of reactive oxygen species production, impaired mitochondrial function, and ubiquitin-proteasome system dysregulation, factors that can in turn initiate transcriptional changes that contribute to the perturbation of lipid metabolism [124].
The impact of HIV infection and antiretroviral therapy on chronic inflammation.
Proinflammatory Molecules
The molecules associated with HIV that promote inflammation and may lead to immune dysfunction are considered below.
High-sensitivity CRP, one of the most common markers of inflammation, is a well-known risk factor for CVD and a predictor of all-cause mortality [125]. Higher concentrations of hs-CRP in PLWH in comparison to the general population have been demonstrated. Increased levels of D-dimer, a marker of deterioration of CV condition and endothelial dysfunction, are also associated with increased HIV viral load, microbial translocation, immune activation, and mortality risk [126].
Interleukin-6 belongs to the interleukin-6 family, a group of cytokines that includes IL-6, IL-11, IL-27, ciliary neurotrophic factor, leukemia inhibitory factor, oncostatin M, cardiotrophin 1, and cardiotrophin-like cytokine [127]. IL-6 is a pro-inflammatory cytokine in which higher circulating levels are associated with HIV replication [128]. Increased levels of IL-6 are related to the development of CVD and can predict mortality due to CVD or CV events [71]. In HIV infection, IL-1β induces TNF-α and IL-6 expression, leading to sustained proinflammatory responses. HIV is also a factor in the production of IL‐1β via transforming pro-IL-1β into bioactive IL-1β, a cytokine that is associated both with the progression to AIDS and higher CVD risk [129]. A detectable HIV viral load induces a higher TNF-α serum concentration that can initiate and accelerate apoptosis, atherogenesis, thrombosis, vascular remodeling, and oxidative stress and therefore increase cardiovascular risk [130, 131]. TGF-β is related to atherosclerosis-associated vascular inflammation, and the overexpression of TGF-β in PLWH promotes viral replication and plays an important role in the progression of HIV infection and associated diseases [132]. Chronic increase in osteopontin level, reported in PLWH, is another risk factor for CVD, since osteopontin plays a role in the secretion of multiple proinflammatory molecules, including IL-10, IL-12, IL-3, IFN-γ, and can also be used to predict major adverse cardiovascular events [133]. Elevated levels of sCD14 observed in PLWH have been associated with microbial translocation, increased immune activation, and a greater risk of mortality and morbidity due to CVD [134].
The expression of the adhesion molecules VCAM-1 and ICAM-1, which mediate inflammation and promote leukocyte migration, is stimulated by HIV-Tat-1 protein and pro-inflammatory cytokines such as TNF-α and IL-1β [135]. Toll-like receptors activate the expression of VCAM-1 and ICAM-1 in the endothelium, a response that is strongly associated with increased intimal leukocyte accumulation, an important factor in the pathogenesis of human atherosclerosis [136]. VCAM-1 is a diagnostic biomarker of endothelial dysfunction and vascular injury; together with ICAM-1, it has been used in many clinical studies to estimate the risk of CVD [137]. It has been reported that the expression of adhesion molecules in PLWH is significantly higher than in the general population [138].
The Impact of Antiretroviral Therapy on Pro-Inflammatory Biomarkers
ART is beneficial in terms of the decrease of the proinflammatory effect induced by HIV infection: studies evaluating the levels of IL-6, IL-1β, D-dimer ICAM-1, VCAM-1, and TNF-α showed a significant decrease in the concentration of those biomarkers in PLWH after receiving antiretroviral therapy. However, the levels of those biomarkers were still elevated in comparison to healthy controls [139,140,141]. Residual immune activation may continue in compartments such as the central nervous system, the gastrointestinal tract, or the lymph nodes, where ART penetrates insufficiently to completely suppress viral replication, resulting in residual systemic inflammation [142].
The summary of the mechanisms contributing to the elevated risk for cardiovascular disease among PLWH was presented in Fig. 1.
Conclusions
PLWH experience increased risk of cardiovascular disease, and the reasons for this are multivariate, including the impact of HIV infection itself, the adverse effects of antiretroviral therapy, the ambiguous effect of CD4 + cell count depletion and recovery, and other independent risk factors such as e smoking and chronic viral infections. Although HIV infection is an uncommon disease, clinicians should bear it in mind for people with early occurrence of cardiovascular disease. Dyslipidemia, hypertension, or high serum glucose levels, especially in young patients, should be considered in terms of HIV infection. The cooperation of specialists is crucial for providing the best medical care for people living with HIV and cardiovascular disease.
References
Benjamin, E. J., Blaha, M. J., Chiuve, S. E. (2017). Heart Disease and Stroke Statistics-2017 Update: A Report From the American Heart Association [published correction appears in Circulation 2017;135(10):e646] [published correction appears in Circulation 136(10:e196]. Circulation 135(10), e146-e603. https://doi.org/10.1161/CIR.0000000000000485
Freiberg, M. S., Chang, C. H., Skanderson, M., et al. (2017). Association between HIV Infection and the risk of Heart Failure with reduced ejection fraction and preserved ejection fraction in the antiretroviral therapy era: Results from the veterans Aging Cohort Study. JAMA Cardiology, 2(5), 536–546. https://doi.org/10.1001/jamacardio.2017.0264
Perkins, M. V., Joseph, S. B., Dittmer, D. P., & Mackman, N. (2023). Cardiovascular Disease and Thrombosis in HIV Infection. Arteriosclerosis, thrombosis, and vascular biology 43(2), 175–191. https://doi.org/10.1161/ATVBAHA.122.318232
So-Armah, K., Benjamin, L. A., Bloomfield, G. S., et al. (2020). HIV and Cardiovascular Disease. The Lancet HIV, 7(4), e279–e293. https://doi.org/10.1016/S2352-3018(20)30036-9
Jabeen, S., Rasheed, A., Jabeen, N., et al. (2020). Prevalence and association of HBV and HCV Infection with Cardiovascular Disease risk factors in a peri-urban population. Journal of Pakistan Medical Association, 70(1), 58–63. https://doi.org/10.5455/JPMA.1542
Jeong, S. M., Yoo, J. E., Park, J., Jung, W., Lee, K. N., Han, K., Lee, C. M., Nam, K. W., Lee, S. P., & Shin, D. W. (2023). Smoking behavior change and risk of Cardiovascular Disease incidence and mortality in patients with type 2 Diabetes Mellitus. Cardiovascular Diabetology, 22(1), 193. https://doi.org/10.1186/s12933-023-01930-4
Marcus, J. L., Leyden, W. A., Alexeeff, S. E., Anderson, A. N., Hechter, R. C., Hu, H., Lam, J. O., Towner, W. J., Yuan, Q., Horberg, M. A., & Silverberg, M. J. (2020). Comparison of overall and comorbidity-free life expectancy between insured adults with and without HIV Infection, 2000–2016. JAMA Network open, 3(6), e207954. https://doi.org/10.1001/jamanetworkopen.2020.7954
Bishop, J. D., DeShields, S., Cunningham, T., & Troy, S. B. (2016). CD4 Count Recovery after initiation of antiretroviral therapy in patients infected with Human Immunodeficiency Virus. The American Journal of the Medical Sciences, 352(3), 239–244. https://doi.org/10.1016/j.amjms.2016.05.032
Pambou, H. O. T., Gagneux-Brunon, A., Fossi, B. T., Roche, F., Guyot, J., Botelho-Nevers, E., Dupre, C., Bongue, B., & Nkenfou, C. N. (2022). Assessment of cardiovascular risk factors among HIV-infected patients aged 50 years and older in Cameroon. AIMS public health 9(3), 490–505. https://doi.org/10.3934/publichealth.2022034
Hsue, P. Y., Lo, J. C., Franklin, A., Bolger, A. F., Martin, J. N., Deeks, S. G., & Waters, D. D. (2004). Progression of Atherosclerosis as assessed by carotid intima-media thickness in patients with HIV Infection. Circulation, 109(13), 1603–1608. https://doi.org/10.1161/01.CIR.0000124480.32233.8A
Erqou, S., Jiang, L., Choudhary, G., et al. (2021). Age at diagnosis of Heart Failure in United States Veterans with and without HIV Infection. Journal of the American Heart Association, 10(11), e018983. https://doi.org/10.1161/JAHA.120.018983
Knudsen, A. D., Fuchs, A., Benfield, T., Gerstoft, J., Køber, L., Trøseid, M., Kofoed, K. F., & Nielsen, S. D. (2023). Coronary Artery Disease in Persons With Human Immunodeficiency Virus Without Detectable Viral Replication. Open forum infectious diseases 10(7), ofad298. https://doi.org/10.1093/ofid/ofad298
Tadesse, B. T., Foster, B. A., Chala, A., Chaka, T. E., Bizuayehu, T., Ayalew, F., Meskel, H., Tadesse, G., Jerene, S., Makonnen, D., & Aklillu, E., E (2019). HIV and cART-Associated Dyslipidemia among HIV-Infected Children. Journal of Clinical Medicine, 8(4), 430. https://doi.org/10.3390/jcm8040430
Hanna, D. B., Ramaswamy, C., Kaplan, R. C. (2018). Trends in Cardiovascular Disease Mortality Among Persons With HIV in New York City, 2001–2012 [published correction appears in Clin Infect Dis. 2018;66(6):985]. Clinical Infectious Diseases 63(8), 1122–1129. https://doi.org/10.1093/cid/ciw470
Cybulska, B., Kłosiewicz-Latoszek, L., Szostak, W. (2022). Profilaktyka chorób serowo-naczyniowych. 2022. Available online: https://www.mp.pl/interna/chapter/B16.II.2.3 (accessed on 02 February 2023).
Alonso, A., Barnes, A. E., Guest, J. L., Shah, A., Shao, I. Y., & Marconi, V. (2019). HIV Infection and incidence of Cardiovascular Diseases: An analysis of a large Healthcare Database. Journal of the American Heart Association, 8(14), e012241. https://doi.org/10.1161/JAHA.119.012241
Kentoffio, K., Temu, T. M., Shakil, S. S., Zanni, M. V., & Longenecker, C. T. (2022). Cardiovascular Disease risk in women living with HIV. Current Opinion in HIV and AIDS, 17(5), 270–278. https://doi.org/10.1097/COH.0000000000000756
Johnston, P. I., Wright, S. W., Orr, M., Pearce, F. A., Stevens, J. W., Hubbard, R. B., & Collini, P. J. (2021). Worldwide relative Smoking prevalence among people living with and without HIV. AIDS (London England), 35(6), 957–970. https://doi.org/10.1097/QAD.0000000000002815
Thompson-Paul, A. M., Wei, S. C., Mattson, C. L., Robertson, M., Hernandez-Romieu, A. C., Bell, T. K., & Skarbinski, J. (2015). Obesity among HIV-Infected adults receiving Medical Care in the United States: Data from the Cross-sectional Medical Monitoring Project and National Health and Nutrition Examination Survey. Medicin, 94(27), e1081. https://doi.org/10.1097/MD.0000000000001081
Bares, S. H., Smeaton, L. M., Xu, A., Godfrey, C., & McComsey, G. A. (2018). HIV-Infected women Gain more weight than HIV-Infected men following the initiation of antiretroviral therapy. Journal of Women’s Health (2002), 27(9), 1162–1169. https://doi.org/10.1089/jwh.2017.6717
Lin, J., Ehinger, E., Hanna, D. B., Qi, Q., Wang, T., Ghosheh, Y., Mueller, K., Anastos, K., Lazar, J. M., Mack, W. J., Tien, P. C., Berman, J. W., Cohen, M. H., Ofotokun, I., Gange, S., Liu, C., Heath, S. L., Tracy, R. P., Hodis, H. N., Landay, A. L., et al. (2023). HIV Infection and Cardiovascular Disease have both shared and distinct monocyte gene expression features: Women’s interagency HIV study. PloS One, 18(5), e0285926. https://doi.org/10.1371/journal.pone.0285926
Van Ommen, C. E., King, E. M., & Murray, M. C. M. (2021). Age at menopause in women living with HIV: A systematic review. Menopause (New York N Y), 28(12), 1428–1436. https://doi.org/10.1097/GME.0000000000001871
Klaver, M., de Blok, C. J. M., Wiepjes, C. M., Nota, N. M., Dekker, M. J. H. J., de Mutsert, R., Schreiner, T., Fisher, A. D., T’Sjoen, G., & den Heijer, M. (2018). Changes in regional body fat, lean body mass and body shape in trans persons using cross-sex hormonal therapy: Results from a multicenter prospective study. European Journal of Endocrinology, 178(2), 163–171. https://doi.org/10.1530/EJE-17-0496
Suppakitjanusant, P., Ji, Y., Stevenson, M. O., Chantrapanichkul, P., Sineath, R. C., Goodman, M., Alvarez, J. A., & Tangpricha, V. (2020). Effects of gender-affirming hormone therapy on body mass index in transgender individuals: A longitudinal cohort study. Journal of Clinical & Translational Endocrinology, 21, 100230. https://doi.org/10.1016/j.jcte.2020.100230
Lake, J. E., Wang, R., Barrett, B. W., Bowman, E., Hyatt, A. N., Debroy, P., Candelario, J., Teplin, L., Bodnar, K., McKay, H., Plankey, M., Brown, T. T., Funderburg, N., & Currier, J. S. (2022). Trans women have worse cardiovascular biomarker profiles than cisgender men Independent of hormone use and HIV serostatus. AIDS (London England), 36(13), 1801–1809. https://doi.org/10.1097/QAD.0000000000003346
Hiransuthikul, A., Himmad, L., Kerr, S. J., Janamnuaysook, R., Dalodom, T., Phanjaroen, K., Pankam, T., Kongkapan, J., Mills, S., Vannakit, R., Phanuphak, P., & Phanuphak, N. (2021). Drug-drug interactions among Thai Transgender Women living with human immunodeficiency undergoing feminizing hormone therapy and antiretroviral therapy: The iFACT Study. Clinical Infectious Diseases, 72(3), 396–402. https://doi.org/10.1093/cid/ciaa038
Tun, W., Pulerwitz, J., Shoyemi, E., Fernandez, A., Adeniran, A., Ejiogu, F., Sangowawa, O., Granger, K., Dirisu, O., & Adedimeji, A. A. (2022). A qualitative study of how stigma influences HIV services for transgender men and women in Nigeria. Journal of the International AIDS Society, 25(Suppl 1), e25933. https://doi.org/10.1002/jia2.25933
Vyas, A., Desai, R., Went, T. R., Wiltshire, D., Priyadarshni, S., Shalaby, M., & Khalife, W. (2023). Cardiovascular Disease Burden and Major Adverse Cardiac Events in Young Black Patients: A National Analysis of 2 cohorts 10 years apart (2017 Versus 2007). Journal of the American Heart Association, 12(15), e029895. https://doi.org/10.1161/JAHA.123.029895
Oramasionwu, C. U., Morse, G. D., Lawson, K. A., Brown, C. M., Koeller, J. M., & Frei, C. R. (2013). Hospitalizations for Cardiovascular Disease in African americans and whites with HIV/AIDS. Population Health Management, 16(3), 201–207. https://doi.org/10.1089/pop.2012.0043
OʼBryan, T. A., Agan, B. K., Tracy, R. P., Freiberg, M. S., Okulicz, J. F., So-Armah, K., Ganesan, A., Rimland, D., Lalani, T., Deiss, R. G., & Tramont, E. C. (2018). Brief report: Racial comparison of D-Dimer levels in US Male Military Personnel before and after HIV Infection and viral suppression. Journal of Acquired Immune Deficiency Syndromes (1999), 77(5), 502–506. https://doi.org/10.1097/QAI.0000000000001626
Lange, L. A., Reiner, A. P., Carty, C. L., Jenny, N. S., Cushman, M., & Lange, E. M. (2008). Common genetic variants associated with plasma fibrin D-dimer concentration in older European- and African-American adults. Journal of thrombosis and haemostasis. JTH 6(4), 654–659. https://doi.org/10.1111/j.1538-7836.2008.02906.x
Davy-Mendez, T., Napravnik, S., Eron, J. J., Cole, S. R., Van Duin, D., Wohl, D. A., Gebo, K. A., Moore, R. D., Althoff, K. N., Poteat, T., Gill, M. J., Horberg, M. A., Silverberg, M. J., Nanditha, N. G. A., Thorne, J. E., & Berry, S. A., North American AIDS cohort collaboration on Research and Design (NA-ACCORD) of IeDEA (2021) racial, ethnic, and gender disparities in hospitalizations among persons with HIV in the United States and Canada, 2005–2015. AIDS (London England) 35(8), 1229–1239. https://doi.org/10.1097/QAD.0000000000002876
Shah, N. S., Huang, X., Petito, L. C., Bancks, M. P., Ning, H., Cameron, N. A., Kershaw, K. N., Kandula, N. R., Carnethon, M. R., Lloyd-Jones, D. M., & Khan, S. S. (2023). Social and Psychosocial Determinants of Racial and Ethnic Differences in Cardiovascular Health in the United States Population. Circulation 147(3), 190–200. https://doi.org/10.1161/CIRCULATIONAHA.122.061991
Wang, Y., Lifshitz, L., Silverstein, N. J., Mintzer, E., Luk, K., StLouis, P., Brehm, M. A., Wolfe, S. A., Deeks, S. G., & Luban, J. (2023). Transcriptional and chromatin profiling of human blood innate lymphoid cell subsets sheds light on HIV-1 pathogenesis. The EMBO journal e114153. Advance online publication. https://doi.org/10.15252/embj.2023114153
Kelly, C., Gurung, R., Kamng’ona, R., Sheha, I., Chammudzi, M., Jambo, K., Mallewa, J., Rapala, A., Heyderman, R., Mallon, P., Mwandumba, H., Khoo, S., & Klein, N. (2022). Circulating microparticles are increased amongst people presenting with HIV and advanced immune suppression in Malawi and correlate closely with arterial stiffness: A nested case control study. Wellcome open Research, 6, 264. https://doi.org/10.12688/wellcomeopenres.17044.2
Saloner, R., Marquine, M. J., Sundermann, E. E., Hong, S., McCutchan, J. A., Ellis, R. J., Heaton, R. K., Grant, I., & Cherner, M. (2019). COMT Val158Met Polymorphism, Cardiometabolic Risk, and Nadir CD4 synergistically increase risk of neurocognitive impairment in men living with HIV. Journal of Acquired Immune Deficiency Syndromes (1999), 81(5), e148–e157. https://doi.org/10.1097/QAI.0000000000002083
Nosik, M., Belikova, M. G., Ryzhov, K., Avdoshina, D., Sobkin, A., Zverev, V., & Svitich, O. (2023). Unique Profile of Proinflammatory Cytokines in Plasma of Drug-Naïve Individuals with Advanced HIV/TB Co-Infection. Viruses 15(6), 1330. https://doi.org/10.3390/v15061330
Kundu, S., Freiberg, M. S., Tracy, R. P., So-Armah, K. A., Koethe, J. R., Duncan, M. S., Tindle, H. A., Beckman, J. A., Feinstein, M. J., McDonnell, W. J., Justice, A., Doyle, M. F., & Veterans Aging Cohort Study. (2022). Circulating T cells and Cardiovascular Risk in People with and without HIV Infection. Journal of the American College of Cardiology, 80(17), 1633–1644. https://doi.org/10.1016/j.jacc.2022.08.756
Pham, L. M., Kim, E. C., Ou, W., Phung, C. D., Nguyen, T. T., Pham, T. T., Poudel, K., Gautam, M., Nguyen, H. T., Jeong, J. H., Yong, C. S., Park, S. Y., Kim, J. R., & Kim, J. O. (2021). Targeting and clearance of senescent foamy macrophages and senescent endothelial cells by antibody-functionalized mesoporous silica nanoparticles for alleviating aorta Atherosclerosis. Biomaterials, 269, 120677. https://doi.org/10.1016/j.biomaterials.2021.120677
Lopez Krol, A., Nehring, H. P., Krause, F. F., Wempe, A., Raifer, H., Nist, A., Stiewe, T., Bertrams, W., Schmeck, B., Luu, M., Leister, H., Chung, H. R., Bauer, U. M., Adhikary, T., & Visekruna, A. (2022). Lactate induces metabolic and epigenetic reprogramming of pro-inflammatory Th17 cells. EMBO Reports, 23(12), e54685. https://doi.org/10.15252/embr.202254685
van der Heijden, W. A., van Deuren, R. C., van de Wijer, L., van den Munckhof, I. C. L., Steehouwer, M., Riksen, N. P., Netea, M. G., de Mast, Q., Vandekerckhove, L., de Voer, R. M., van der Ven, A. J., & Hoischen, A. (2022). Clonal hematopoiesis is Associated with low CD4 Nadir and increased residual HIV Transcriptional Activity in Virally suppressed individuals with HIV. The Journal of Infectious Diseases, 225(8), 1339–1347. https://doi.org/10.1093/infdis/jiab419
Tort, O., Escribà, T., Egaña-Gorroño, L., de Lazzari, E., Cofan, M., Fernandez, E., Gatell, J. M., Martinez, E., Garcia, F., & Arnedo, M. (2018). Cholesterol efflux responds to viral load and CD4 counts in HIV + patients and is dampened in HIV exposed. Journal of Lipid Research, 59(11), 2108–2115. https://doi.org/10.1194/jlr.M088153
Ritsch, A., Duerr, A., Kahler, P., Hunjadi, M., Stojakovic, T., Silbernagel, G., Scharnagl, H., Kleber, M. E., & März, W. (2020). Cholesterol Efflux Capacity and Cardiovascular Disease: The Ludwigshafen Risk and Cardiovascular Health (LURIC) Study. Biomedicines 8(11), 524. https://doi.org/10.3390/biomedicines8110524
Pacheco, Y. M., Jarrin, I., Rosado, I., Campins, A. A., Berenguer, J., Iribarren, J. A., Rivero, M., Muñoz-Medina, L., Bernal-Morell, E., Gutiérrez, F., & Leal, M., CoRIS (2015). Increased risk of non-AIDS-related events in HIV subjects with persistent low CD4 counts despite cART in the CoRIS cohort. Antiviral Research, 117, 69–74. https://doi.org/10.1016/j.antiviral.2015.03.002
Masip, J., Jorba, R., López-Dupla, M., et al. (2022). Lipoprotein Profile in Immunological Non-responders PLHIV after antiretroviral therapy initiation. International Journal of Molecular Sciences, 23(15), 8071. https://doi.org/10.3390/ijms23158071
Noguera-Julian, M., Rocafort, M., Guillén, Y., Rivera, J., Casadellà, M., Nowak, P., Hildebrand, F., Zeller, G., Parera, M., Bellido, R., Rodríguez, C., Carrillo, J., Mothe, B., Coll, J., Bravo, I., Estany, C., Herrero, C., Saz, J., Sirera, G., Torrela, A. (2016). Gut Microbiota Linked to Sexual Preference and HIV Infection. EBioMedicine 5, 135–146. https://doi.org/10.1016/j.ebiom.2016.01.032
Renault, C., Veyrenche, N., Mennechet, F., et al. (2022). Th17 CD4 + T-Cell as a preferential target for HIV reservoirs. Frontiers in Immunology, 13, 822576. https://doi.org/10.3389/fimmu.2022.822576
Fert, A., Raymond Marchand, L., Salinas, W., & Ancuta, T. R., P (2022). Targeting Th17 cells in HIV-1 remission/cure interventions. Trends in Immunology, 43, 580–594. https://doi.org/10.1016/j.it.2022.04.013
Singh, A., Vajpayee, M., Ali, S. A., & Chauhan, N. K. (2014). Cellular interplay among Th17, Th1, and Treg cells in HIV-1 subtype C Infection. Journal of Medical Virology, 86(3), 372–384. https://doi.org/10.1002/jmv.23810
Bi, X., Ishizaki, A., Nguyen, L. V., Matsuda, K., Pham, H. V., Phan, C. T., Ogata, K., Giang, T. T., Phung, T. T., Nguyen, T. T., Tokoro, M., Pham, A. N., Khu, D. T., & Ichimura, H. (2016). Impact of HIV Infection and anti-retroviral therapy on the Immune Profile of and Microbial Translocation in HIV-Infected Children in Vietnam. International Journal of Molecular Sciences, 17(8), 1245. https://doi.org/10.3390/ijms17081245
Tian, X., Xie, Y., Chen, J., Yin, W., Zhao, Y. L., Yao, P., Dong, M., Jin, C., & Wu, N. (2023). Increased Microbial translocation is a prognostic biomarker of different Immune responses to ART in people living with HIV. Infection and Drug Resistance, 16, 3871–3878. https://doi.org/10.2147/IDR.S404384
Isnard, S., Royston, L., Scott, S. C., Mabanga, T., Lin, J., Fombuena, B., Bu, S., Berini, C. A., Goldberg, M. S., Finkelman, M., Brouillette, M. J., Fellows, L. K., Mayo, N. E., & Routy, J. P. (2023). Translocation of bacterial LPS is associated with self-reported cognitive abilities in men living with HIV receiving antiretroviral therapy. AIDS Research and Therapy, 20(1), 30. https://doi.org/10.1186/s12981-023-00525-z
Xia, P., Xing, X. D., Yang, C. X., Liao, X. J., Liu, F. H., Huang, H. H., Zhang, C., Song, J. W., Jiao, Y. M., Shi, M., Jiang, T. J., Zhou, C. B., Wang, X. C., He, Q., Zeng, Q. L., Wang, F. S., & Zhang, J. Y. (2022). Activation-induced pyroptosis contributes to the loss of MAIT cells in chronic HIV-1 infected patients. Military Medical Research, 9(1), 24. https://doi.org/10.1186/s40779-022-00384-1
Gaardbo, J. C., Trøsied, M., Stiksrud, B., Midttun, Ø., Ueland, P. M., Ullum, H., & Nielsen, S. D. (2015). Increased Tryptophan Catabolism Is Associated With Increased Frequency of CD161 + Tc17/MAIT Cells and Lower CD4 + T-Cell Count in HIV-1 Infected Patients on cART After 2 Years of Follow-Up. Journal of acquired immune deficiency syndromes (1999) 70(3), 228–235. https://doi.org/10.1097/QAI.0000000000000758
Seethaler, B., Basrai, M., Neyrinck, A. M., Nazare, J. A., Walter, J., Delzenne, N. M., & Bischoff, S. C. (2021). Biomarkers for assessment of intestinal permeability in clinical practice. American journal of physiology. Gastrointestinal and liver physiology 321(1), G11–G17. https://doi.org/10.1152/ajpgi.00113.2021
Isnard, S., Ramendra, R., Dupuy, F. P., Lin, J., Fombuena, B., Kokinov, N., et al. (2020). Plasma levels of c-type lectin REG3alpha and gut damage in people with human immunodeficiency virus. Journal of Infectious Diseases, 221(1), 110–121. https://doi.org/10.1093/infdis/jiz423
Kentoffio, K., Temu, T. M., Shakil, S. S., Zanni, M. V., & Longenecker, C. T. (2022). Cardiovascular disease risk in women living with HIV. Current Opinion in HIV and AIDS 17(5), 270–278. doi: 0.1097/COH.0000000000000756.
Isnard, S., Ramendra, R., Dupuy, F. P., Lin, J., Fombuena, B., Kokinov, N., Kema, I., Jenabian, M. A., Lebouché, B., Costiniuk, C. T., Ancuta, P., Bernard, N. F., Silverman, M. S., Lakatos, P. L., Durand, M., Tremblay, C., Routy, J. P., & Aging Cohort Groups. (2020). Montreal Primary HIV Infection Study, the Canadian Cohort of HIV + Slow Progressors, and the Canadian HIV and Plasma Levels of C-Type Lectin REG3α and Gut Damage in People With Human Immunodeficiency Virus. The Journal of infectious diseases 221(1), 110–121. https://doi.org/10.1093/infdis/jiz423
Fasano, A. (2011). Zonulin and its regulation of intestinal barrier function: The biological door to inflammation, autoimmunity, and cancer. Physiological Reviews, 91(1), 151–175. https://doi.org/10.1152/physrev.00003.2008
Ockner, R. K., & Manning, J. A. (1974). Fatty acid-binding protein in small intestine. Identification, isolation, and evidence for its role in cellular fatty acid transport. The Journal of Clinical Investigation, 54(2), 326–338. https://doi.org/10.1172/JCI107768
Hunt, P. W., Sinclair, E., Rodriguez, B., Shive, C., Clagett, B., Funderburg, N., et al. (2014). Gut epithelial barrier dysfunction and innate immune activation predict mortality in treated HIV Infection. Journal of Infectious Diseases, 210(8), 1228–1238. https://doi.org/10.1093/infdis/jiu238
Jabłonowska, E., Strzelczyk, J., Piekarska, A., & Wójcik-Cichy, K. (2021). Gut microbiota diversity in HIV-Infected patients on successful antiretroviral treatment is linked to sexual preferences but not CD4 Nadir. Archivum Immunologiae et therapiae experimentalis, 69(1), 14. https://doi.org/10.1007/s00005-021-00616-7
Zhang, Y., Xie, Z., Zhou, J., Li, Y., Ning, C., Su, Q., Ye, L., Ai, S., Lai, J., Pan, P., Liu, N., Liao, Y., Su, Q., Li, Z., Liang, H., Cui, P., & Huang, J. (2023). The altered metabolites contributed by dysbiosis of gut microbiota are associated with microbial translocation and immune activation during HIV Infection. Frontiers in Immunology, 13, 1020822. https://doi.org/10.3389/fimmu.2022.1020822
Facchin, S., Vitulo, N., Calgaro, M., Buda, A., Romualdi, C., Pohl, D., Perini, B., Lorenzon, G., Marinelli, C., D’Incà, R., Sturniolo, G. C., & Savarino, E. V. (2020). Microbiota changes induced by microencapsulated sodium butyrate in patients with inflammatory bowel Disease. Neurogastroenterology and Motility, 32(10), e13914. https://doi.org/10.1111/nmo.13914
Holle, J., Bartolomaeus, H., Löber, U., Behrens, F., Bartolomaeus, T. U. P., Anandakumar, H., Wimmer, M. I., Vu, D. L., Kuhring, M., Brüning, U., Maifeld, A., Geisberger, S., Kempa, S., Schumacher, F., Kleuser, B., Bufler, P., Querfeld, U., Kitschke, S., Engler, D., Kuhrt, L. D., et al. (2022). Inflammation in children with CKD linked to Gut Dysbiosis and Metabolite Imbalance. Journal of the American Society of Nephrology: JASN, 33(12), 2259–2275. https://doi.org/10.1681/ASN.2022030378
Vargas-Pacherrez, D., Cotrim, H. P., Pires, L., Cunha, V., Coelho, V., Brites, C., & Daltro, C. (2020). Metabolic syndrome in HIV-patients in antiretroviral therapy. Current HIV Research, 18(6), 388–395. https://doi.org/10.2174/1570162X18666200609115615
Padmapriyadarsini, C., Ramesh, K., Sekar, L., Ramachandran, G., Reddy, D., Narendran, G., Sekar, S., Chandrasekar, C., Anbarasu, D., Wanke, C., & Swaminathan, S. (2017). Factors affecting high-density lipoprotein cholesterol in HIV-infected patients on nevirapine-based antiretroviral therapy. The Indian Journal of Medical Research, 145(5), 641–650. https://doi.org/10.4103/ijmr.IJMR_1611_15
Jumare, J., Dakum, P., Sam-Agudu, N., Memiah, P., Nowak, R., Bada, F., Oguama, U., Odonye, G., Adebiyi, R., Cairo, C., Kwaghe, V., Adebamowo, C., Abimiku, A., & Charurat, M. (2023). Prevalence and characteristics of metabolic syndrome and its components among adults living with and without HIV in Nigeria: A single-center study. BMC Endocrine Disorders, 23(1), 160. https://doi.org/10.1186/s12902-023-01419-x
Low, H., Hoang, A., Pushkarsky, T., Dubrovsky, L., Dewar, E., Di Yacovo, M. S., Mukhamedova, N., Cheng, L., Downs, C., Simon, G., Saumoy, M., Hill, A. F., Fitzgerald, M. L., Nestel, P., Dart, A., Hoy, J., Bukrinsky, M., & Sviridov, D. (2019). HIV Disease, metabolic dysfunction and Atherosclerosis: A three year prospective study. PloS One, 14(4), e0215620. https://doi.org/10.1371/journal.pone.0215620
Feeney, E. R., McAuley, N., O’Halloran, J. A., et al. (2013). The expression of cholesterol metabolism genes in monocytes from HIV-infected subjects suggests intracellular cholesterol accumulation. The Journal of Infectious Diseases, 207(4), 628–637. https://doi.org/10.1093/infdis/jis723
Gori, E., Mduluza, T., Nyagura, M., Stray-Pedersen, B., & Gomo, Z. A. (2016). Inflammation-modulating cytokine profile and lipid interaction in HIV-related risk factors for Cardiovascular Diseases. Therapeutics and Clinical risk Management, 12, 1659–1666. https://doi.org/10.2147/TCRM.S117980
Williams, P., Wu, J., Cohn, S., Koletar, S., McCutchan, J., Murphy, R., Currier, J., & AIDS Clinical Trials Group 362 Study Team. (2009). Improvement in lipid profiles over 6 years of follow-up in adults with AIDS and immune reconstitution. HIV Medicine, 10(5), 290–301. https://doi.org/10.1111/j.1468-1293.2008.00685.x
Xu, Y., Chen, X., & Wang, K. (2017). Global prevalence of Hypertension among people living with HIV: A systematic review and meta-analysis. Journal of the American Society of Hypertension, 11(8), 530–540. https://doi.org/10.1016/j.jash.2017.06.004
Jackson, I. L., Lawrence, S. M., Igwe, C. N., et al. (2022). Prevalence and control of Hypertension among people living with HIV receiving care at a Nigerian hospital. The Pan African Medical Journal, 41, 153. https://doi.org/10.11604/pamj.2022.41.153.21809
Chepchirchir, A., Nyagol, J., & Jaoko, W. (2018). Cytokine expression and Hypertension Comorbidity in HIV/AIDS patients at Kenyatta National Hospital HIV Care Centre, Nairobi, Kenya. International Journal of Cardiovascular Research, 07, 2. https://doi.org/10.4172/2324-8602.1000343
Siedner, M. J., Kim, J. H., Nakku, R. S., Bibangambah, P., Hemphill, L., Triant, V. A., Haberer, J. E., Martin, J. N., Mocello, A. R., Boum, Y. 2nd, Kwon, D. S., Tracy, R. P., Burdo, T., Huang, Y., Cao, H., Okello, S., Bangsberg, D. R., & Hunt, P. W. (2016). Persistent Immune activation and carotid Atherosclerosis in HIV-Infected ugandans receiving antiretroviral therapy. The Journal of Infectious Diseases, 213(3), 370–378. https://doi.org/10.1093/infdis/jiv450
Shen, G., Thomas, T. S., Walpert, A. R., McClure, C. M., Fitch, K. V., deFilippi, C., Torriani, M., Buckless, C. G., Adler, G. K., Grinspoon, S. K., & Srinivasa, S. (2022). Role of renin-angiotensin-aldosterone system activation and other metabolic variables in relation to arterial inflammation in HIV. Clinical Endocrinology, 97(5), 581–587. https://doi.org/10.1111/cen.14784
Peck, R. N., Shedafa, R., Kalluvya, S., Downs, J. A., Todd, J., Suthanthiran, M., Fitzgerald, D. W., & Kataraihya, J. B. (2014). Hypertension, Kidney Disease, HIV and antiretroviral therapy among Tanzanian adults: A cross-sectional study. BMC Medicine, 12, 125. https://doi.org/10.1186/s12916-014-0125-2
Xu, Y., Chen, X., & Wang, K. (2017). Global prevalence of Hypertension among people living with HIV: A systematic review and meta-analysis. Journal of the American Society of Hypertension: JASH, 11(8), 530–540. https://doi.org/10.1016/j.jash.2017.06.004
Byonanebye, D. M., Polizzotto, M. N., Neesgaard, B., Sarcletti, M., Matulionyte, R., Braun, D. L., Castagna, A., de Wit, S., Wit, F., Fontas, E., Vehreschild, J. J., Vesterbacka, J., Greenberg, L., Hatleberg, C., Garges, H., Gallant, J., Volny Anne, A., Öllinger, A., Mozer-Lisewska, I., Surial, B., et al. (2022). Incidence of Hypertension in people with HIV who are treated with integrase inhibitors versus other antiretroviral regimens in the RESPOND cohort consortium. HIV Medicine, 23(8), 895–910. https://doi.org/10.1111/hiv.13273
González-Cordón, A., Doménech, M., Camafort, M., Martínez-Rebollar, M., Torres, B., Laguno, M., Rojas, J., Loncà, M., Blanco, J. L., Mallolas, J., Gatell, J. M., de Lazzari, E., & Martínez, E. (2018). Subclinical Cardiovascular Disease in patients starting contemporary protease inhibitors. HIV Medicine. https://doi.org/10.1111/hiv.12619
Brennan, A. T., Nattey, C., Kileel, E. M., Rosen, S., Maskew, M., Stokes, A. C., Fox, M. P., & Venter, W. D. F. (2023). Change in body weight and risk of hypertension after switching from efavirenz to dolutegravir in adults living with HIV: evidence from routine care in Johannesburg, South Africa. eClinicalMedicine 57, 101836. https://doi.org/10.1016/j.eclinm.2023.101836
Santiprabhob, J., Chokephaibulkit, K., Khantee, P., Maleesatharn, A., Phonrat, B., Phongsamart, W., Lapphra, K., Wittawatmongkol, O., Rungmaitree, S., Tanchaweng, S., Maturapat, S., Lermankul, W., & Tungtrongchitr, R. (2020). Adipocytokine dysregulation, abnormal glucose metabolism, and lipodystrophy in HIV-infected adolescents receiving protease inhibitors. Cytokine, 136, 155145. https://doi.org/10.1016/j.cyto.2020.155145
Ketlogetswe, K. S., Post, W. S., Li, X., Palella, F. J., Jr, Jacobson, L. P., Margolick, J. B., Kingsley, L. A., Witt, M. D., Dobs, A. S., Budoff, M. J., & Brown, T. T. (2014). Lower adiponectin is associated with subclinical Cardiovascular Disease among HIV-infected men. AIDS (London England), 28(6), 901–909. https://doi.org/10.1097/QAD.0000000000000186
Blázquez, D., Ramos-Amador, J. T., Saínz, T., Mellado, M. J., García-Ascaso, M., De José, M. I., Rojo, P., Navarro, M. L., Muñoz-Fernández, M., Saavedra, J., Roa, M. A., Jiménez, S., Beceiro, J., Prieto, L., Hortelano, M. G., & González-Tomé, M. I. (2015). Lipid and glucose alterations in perinatally-acquired HIV-infected adolescents and young adults. BMC infectious diseases 15, 119. https://doi.org/10.1186/s12879-015-0853-8
Putcharoen, O., Wattanachanya, L., Sophonphan, J., Siwamogsatham, S., Sapsirisavat, V., Gatechompol, S., Phonphithak, S., Kerr, S. J., Chattranukulchai, P., Avihingsanon, Y., Ruxrungtham, K., Avihingsanon, A., & HIV-NAT 006 team. (2017). New-onset Diabetes in HIV-treated adults: Predictors, long-term renal and cardiovascular outcomes. AIDS (London England), 31(11), 1535–1543. https://doi.org/10.1097/QAD.0000000000001496
Birabaharan, M., Strunk, A., Kaelber, D. C., Smith, D. M., & Martin, T. C. S. (2022). Sex differences in type 2 Diabetes Mellitus prevalence among persons with HIV. AIDS (London England), 36(3), 383–389. https://doi.org/10.1097/QAD.0000000000003127
Poznyak, A., Grechko, A. V., Poggio, P., Myasoedova, V. A., Alfieri, V., & Orekhov, A. N. (2020). The Diabetes Mellitus-Atherosclerosis Connection: The Role of Lipid and Glucose Metabolism and Chronic Inflammation. International journal of molecular sciences 21(5), 1835. https://doi.org/10.3390/ijms21051835
Dikaiou, P., Björck, L., Adiels, M., Lundberg, C. E., Mandalenakis, Z., Manhem, K., & Rosengren, A. (2021). Obesity, overweight and risk for Cardiovascular Disease and mortality in young women. European Journal of Preventive Cardiology, 28(12), 1351–1359. https://doi.org/10.1177/2047487320908983
Thompson-Paul, A. M., Wei, S. C., Mattson, C. L. (2015). Obesity Among HIV-Infected Adults Receiving Medical Care in the United States: Data From the Cross-Sectional Medical Monitoring Project and National Health and Nutrition Examination Survey. Medicine 94(27), e1081. https://doi.org/10.1097/MD.0000000000001081
Silverberg, M., Leyden, W., Alexeeff, S. (2020). Changes in body mass index over time in persons with and without HIV. AIDS 2020: 23rd International AIDS Conference Virtual. July 6–10, 2020.
Kumar, S., & Samaras, K. (2018). The Impact of Weight Gain During HIV Treatment on Risk of Pre-diabetes, Diabetes Mellitus, Cardiovascular Disease, and Mortality. Frontiers in Endocrinology 9, 705. Published 2018 Nov 27. https://doi.org/10.3389/fendo.2018.00705
Sax, P. E., Erlandson, K. M., Lake, J. E., Mccomsey, G. A., Orkin, C., Esser, S., Brown, T. T., Rockstroh, J. K., Wei, X., Carter, C. C., Zhong, L., Brainard, D. M., Melbourne, K., Das, M., Stellbrink, H. J., Post, F. A., Waters, L., & Koethe, J. R. (2020). Weight Gain Following Initiation of Antiretroviral Therapy: Risk Factors in Randomized Comparative Clinical Trials. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America, 71(6), 1379–1389. https://doi.org/10.1093/cid/ciz999
Erlandson, K. M., Wu, K., Lake, J. E., Samuels, D. C., Bares, S. H., Tassiopoulos, K., Koethe, J. R., Brown, T. T., Leonard, M., Benson, C. A., Haas, D. W., & Hulgan, T., A5001, A5322 protocol teams (2021) mitochondrial DNA haplogroups and weight gain following switch to integrase strand transfer inhibitor-based antiretroviral therapy. AIDS (London England) 35(3), 439–445. https://doi.org/10.1097/QAD.0000000000002771
McMahon, C., Trevaskis, J. L., Carter, C., Holsapple, K., White, K., Das, M., Collins, S., Martin, H., & Burns-Naas, L. A. (2020). Lack of an association between clinical INSTI-related body weight gain and direct interference with MC4 receptor (MC4R), a key central regulator of body weight. PloS One, 15(2), e0229617. https://doi.org/10.1371/journal.pone.0229617
Jeong, S. W., Kim, S. H., Kang, S. H., Kim, H. J., Yoon, C. H., Youn, T. J., & Chae, I. H. (2019). Mortality reduction with physical activity in patients with and without Cardiovascular Disease. European Heart Journal, 40(43), 3547–3555. https://doi.org/10.1093/eurheartj/ehz564
Sahel-Gozin, N., Loutfy, M., & O’Brien, K. K. (2023). Exploring experiences engaging in exercise from the perspectives of women living with HIV: A qualitative study. PloS One, 18(6), e0286542. https://doi.org/10.1371/journal.pone.0286542
Martin, K., Naclerio, F., Karsten, B., et al. (2019). Physical activity and quality of life in people living with HIV. Aids Care, 31(5), 589–598. https://doi.org/10.1080/09540121.2019.1576848
García-Lucio, J., Peinado, V. I., de Jover, L., Del Pozo, R., Blanco, I., Bonjoch, C., Coll-Bonfill, N., Paul, T., Tura-Ceide, O., & Barberà, J. A. (2018). Imbalance between endothelial damage and repair capacity in Chronic Obstructive Pulmonary Disease. PloS One, 13(4), e0195724. https://doi.org/10.1371/journal.pone.0195724
Lanza, G. A., Spera, F. R., Villano, A., Russo, G., Di Franco, A., Lamendola, P., & Crea, F. (2015). Effect of smoking on endothelium-independent vasodilatation. Atherosclerosis 240(2), 330–332. https://doi.org/10.1016/j.atherosclerosis.2015.03.041
Wu, M., Lai, T., Jing, D., Yang, S., Wu, Y., Li, Z., Wu, Y., Zhao, Y., Zhou, L., Chen, H., Shen, J., Li, W., Ying, S., Chen, Z., Wu, X., & Shen, H. (2021). Epithelium-derived IL17A Promotes Cigarette Smoke-induced Inflammation and Mucus Hyperproduction. American journal of respiratory cell and molecular biology 65(6), 581–592. https://doi.org/10.1165/rcmb.2020-0424OC
Giebe, S., Hofmann, A., Brux, M., et al. (2021). Comparative study of the effects of cigarette smoke versus next generation Tobacco and nicotine product extracts on endothelial function. Redox Biology, 47, 102150. https://doi.org/10.1016/j.redox.2021.102150
Bui, T. C., Hoogland, C. E., Chhea, C., Sopheab, H., Ouk, V., Samreth, S., Hor, B., Vidrine, J. I., Businelle, M. S., Shih, Y. C. T., Sutton, S. K., Jones, S. R., Shorey Fennell, B., Cottrell-Daniels, C., Frank-Pearce, S. G., Ngor, C., Kulkarni, S., & Vidrine, D. J. (2023). Ending Tobacco Use Through Interactive Tailored Messaging for Cambodian People With HIV (Project EndIT): Protocol for a Randomized Controlled Trial. JMIR research protocols 12, e48923. https://doi.org/10.2196/48923
Bui, T. C., Hoogland, C. E., Chhea, C., Sopheab, H., Ouk, V., Samreth, S., Hor, B., Vidrine, J. I., Businelle, M. S., Shih, Y. C. T., Sutton, S. K., Jones, S. R., Shorey Fennell, B., Cottrell-Daniels, C., Frank-Pearce, S. G., Ngor, C., Kulkarni, S., & Vidrine, D. J. (2023). Ending Tobacco Use through interactive tailored messaging for Cambodian people with HIV (Project EndIT): Protocol for a Randomized Controlled Trial. JMIR Research Protocols, 12, e48923.
Ashare, R. L., Thompson, M., Leone, F. (2019). Differences in the rate of nicotine metabolism among smokers with and without HIV. AIDS 33(6), 1083–1088. https://doi.org/10.1097/QAD.0000000000002127
Gillis, J., Smieja, M., Cescon, A., Rourke, S. B., Burchell, A. N., Cooper, C., Raboud, J. M., & OHTN Cohort Study Group. (2014). Risk of Cardiovascular Disease associated with HCV and HBV coinfection among antiretroviral-treated HIV-infected individuals. Antiviral Therapy, 19(3), 309–317. https://doi.org/10.3851/IMP2724
National Center for HIV/AIDS, Hepatitis, V., STD, and, Prevention, T. B., & Mortality, H. I. V. (2021). https://www.cdc.gov/hiv/pdf/library/slidesets/cdc-hiv-surveillance-slideset-mortality-2021.pdf (accessed on 05 October 2023).
Zicari, S., Sessa, L., Cotugno, N. (2019). Immune Activation, Inflammation, and Non-AIDS Co-Morbidities in HIV-Infected Patients under Long-Term ART. Viruses 11(3), 200. https://doi.org/10.3390/v11030200
Juno, J. A., Phetsouphanh, C., Klenerman, P., et al. (2019). Perturbation of mucosal-associated invariant T cells and iNKT cells in HIV Infection. Current Opinion in HIV and AIDS, 14(2), 77–84. https://doi.org/10.1097/COH.0000000000000526
Chen, X., Liu, X., Duan, S., et al. (2020). Plasma inflammatory biomarkers Associated with Advanced Liver Fibrosis in HIV-HCV-Coinfected individuals. International Journal of Environmental Research and Public Health, 17(24), 9474. https://doi.org/10.3390/ijerph17249474
Shata, M. T. M., Abdel-Hameed, E. A., Rouster, S. D., et al. (2019). HBV and HIV/HBV infected patients have distinct Immune exhaustion and apoptotic serum biomarker profiles. Pathogens and Immunity, 4(1), 39–65. https://doi.org/10.20411/pai.v4i1.267
Maidji, E., Somsouk, M., Rivera, J. M., Hunt, P. W., & Stoddart, C. A. (2017). Replication of CMV in the gut of HIV-infected individuals and epithelial barrier dysfunction. PLoS Pathogens, 13(2), e1006202. https://doi.org/10.1371/journal.ppat.1006202
Li, H., Weng, P., Najarro, K., et al. (2014). Chronic CMV Infection in older women: Longitudinal comparisons of CMV DNA in peripheral monocytes, anti-CMV IgG titers, serum IL-6 levels, and CMV pp65 (NLV)-specific CD8(+) T-cell frequencies with twelve year follow-up. Experimental Gerontology, 54, 84–89. https://doi.org/10.1016/j.exger.2014.01.010
Nazim, F., Kayani, H. A., Nathwani, A. (2022). A.,. CMV and EBV Co-Infection in HIV-Infected Children: Infection Rates and Analysis of Differential Expression of Cytokines in HIV Mono- and HIV-CMV-EBV Co-Infected Groups. Viruses 14(8), 1823. https://doi.org/10.3390/v14081823
EACS European AIDS Clinical Society (2022). Guidelines Version 11.1 October 2022. Available online: https://www.eacsociety.org/media/guidelines-11.1_final_09-10.pdf (accessed on 01 August 2023).
Menéndez-Arias, L., & Delgado, R. (2022). Update and latest advances in antiretroviral therapy. Trends in Pharmacological Sciences, 43(1), 16–29. https://doi.org/10.1016/j.tips.2021.10.004
ang, L. L., Li, Q., Zhou, L. B., & Chen, S. Q. (2019). Meta-analysis and systematic review of the efficacy and resistance for human immunodeficiency virus type 1 integrase strand transfer inhibitors. International journal of antimicrobial agents 54(5), 547–555. https://doi.org/10.1016/j.ijantimicag.2019.08.008
Neesgaard, B., Greenberg, L., Miró, J. M., et al. (2022). Associations between integrase strand-transfer inhibitors and Cardiovascular Disease in people living with HIV: A multicentre prospective study from the RESPOND cohort consortium. The Lancet HIV, 9(7), e474–e485. https://doi.org/10.1016/S2352-3018(22)00094-7
O’Halloran, J. A., Sahrmann, J., Butler, A. M., Olsen, M. A., & Powderly, W. G. (2020). Brief Report: Integrase Strand Transfer Inhibitors Are Associated With Lower Risk of Incident Cardiovascular Disease in People Living With HIV. Journal of acquired immune deficiency syndromes (1999) 84(4), 396–399. https://doi.org/10.1097/QAI.0000000000002357
Chen, Y. F., Stampley, J. E., Irving, B. A., & Dugas, T. R. (2019). Chronic Nucleoside Reverse transcriptase inhibitors disrupt mitochondrial homeostasis and promote premature endothelial senescence. Toxicological Sciences: An Official Journal of the Society of Toxicology, 172(2), 445–456. https://doi.org/10.1093/toxsci/kfz203
Chen, Y. F., Hebert, V. Y., Stadler, K., Xue, S. Y., Slaybaugh, K., Luttrell-Williams, E., et al. (2019). Coenzyme Q10 alleviates chronic nucleoside reverse transcriptase inhibitor-induced premature endothelial senescence. Cardiovascular Toxicology, 19(6), 500–509. https://doi.org/10.1007/s12012-019-09520-1
Lacey, A., Savinelli, S., Barco, E. A. (2020). Investigating the effect of antiretroviral switch to tenofovir alafenamide on lipid profiles in people living with HIV. AIDS 34(8), 1161–1170. https://doi.org/10.1097/QAD.0000000000002541
Iannone, V., Farinacci, D., D’Angelillo, A., et al. (2022). Cardiovascular Disease Risk in a cohort of Virologically Suppressed people living with HIV switching to Doravirine: Preliminary Data from the Real Life. AIDS Research and Human Retroviruses, 38(11), 878–880. https://doi.org/10.1089/AID.2022.0050
Martini, S., Pisaturo, M., Russo, A., Palamone, M. G., Russo, M. T., Zollo, V., Maggi, P., & Coppola, N. (2023). Evaluation of lipid Profile and Intima Media Thickness in antiretroviral-experienced HIV-Infected patients treated with protease inhibitor-based regimens versus protease inhibitor-sparing regimens. Pathogens (Basel Switzerland), 12(7), 925. https://doi.org/10.3390/pathogens12070925
Sharif, S., Van der Graaf, Y., Cramer, M. J., Kapelle, L. J., de Borst, G. J., Visseren, F. L. J., Westerink, J., & SMART study group. (2021). Low-grade inflammation as a risk factor for cardiovascular events and all-cause mortality in patients with type 2 Diabetes. Cardiovascular Diabetology, 20(1), 220. https://doi.org/10.1186/s12933-021-01409-0
Martínez-Ayala, P., Alanis-Sánchez, G. A., Álvarez-Zavala, M., Sánchez-Reyes, K., Ruiz-Herrera, V. V., Cabrera-Silva, R. I., González-Hernández, L. A., Ramos-Becerra, C., Cardona-Muñoz, E., & Andrade-Villanueva, J. F. (2023). Effect of antiretroviral therapy on decreasing arterial stiffness, metabolic profile, vascular and systemic inflammatory cytokines in treatment-naïve HIV: A one-year prospective study. PloS One, 18(3), e0282728. https://doi.org/10.1371/journal.pone.0282728
Unver, N., & McAllister, F. (2018). IL-6 family cytokines: Key inflammatory mediators as biomarkers and potential therapeutic targets. Cytokine & Growth Factor Reviews, 41, 10–17. https://doi.org/10.1016/j.cytogfr.2018.04.004
Borges, Á. H., O’Connor, J. L., Phillips, A. N., Rönsholt, F. F., Pett, S., Vjecha, M. J., French, M. A., Lundgren, J. D., & INSIGHT SMART and ESPRIT Study Groups and the SILCAAT Scientific Committee. (2015). Factors Associated with plasma IL-6 levels during HIV Infection. The Journal of Infectious Diseases, 212(4), 585–595. https://doi.org/10.1093/infdis/jiv123
Guo, H., Gao, J., Taxman, D. J., et al. (2014). HIV-1 Infection induces interleukin-1β production via TLR8 protein-dependent and NLRP3 inflammasome mechanisms in human monocytes. Journal of Biological Chemistry, 289(31), 21716–21726. https://doi.org/10.1074/jbc.M114.566620
Collora, J. A., Liu, R., Pinto-Santini, D., Ravindra, N., Ganoza, C., Lama, J. R., Alfaro, R., Chiarella, J., Spudich, S., Mounzer, K., Tebas, P., Montaner, L. J., van Dijk, D., Duerr, A., & Ho, Y. C. (2022). Single-cell multiomics reveals persistence of HIV-1 in expanded cytotoxic T cell clones. Immunity, 55(6), 1013–1031e7. https://doi.org/10.1016/j.immuni.2022.03.004
Li, J., Das, J. R., Tang, P., Han, Z., Jaiswal, J. K., & Ray, P. E. (2017). Transmembrane TNF-α facilitates HIV-1 Infection of Podocytes cultured from children with HIV-Associated Nephropathy. Journal of the American Society of Nephrology: JASN, 28(3), 862–875. https://doi.org/10.1681/ASN.2016050564
Chinnapaiyan, S., Dutta, R. K., Nair, M., Chand, H. S., Rahman, I., & Unwalla, H. J. (2019). TGF-β1 increases viral burden and promotes HIV-1 latency in primary differentiated human bronchial epithelial cells. Scientific Reports, 9(1), 12552. https://doi.org/10.1038/s41598-019-49056-6
Mahmud, F. J., Boucher, T., Liang, S., et al. (2020). Osteopontin and integrin mediated modulation of Post-synapses in HIV Envelope Glycoprotein exposed hippocampal neurons. Brain Science, 10(6), 346. https://doi.org/10.3390/brainsci10060346
Affi, R., Gabillard, D., Dunyach-Remy, C., et al. (2021). Association of plasma Soluble Vascular Cell Adhesion Molecule-1 and sCD14 with mortality in HIV-1-Infected west African adults with high CD4 counts. Journal of Acquired Immune Deficiency Syndromes, 86(1), 138–145. https://doi.org/10.1097/QAI.0000000000002533
Pu, H., Tian, J., Flora, G., Lee, Y. W., Nath, A., Hennig, B., & Toborek, M. (2003). HIV-1 Tat protein upregulates inflammatory mediators and induces monocyte invasion into the brain. Molecular and cellular neurosciences 24(1), 224–237. https://doi.org/10.1016/s1044-7431(03)00171-4
Saud, A., Ali, N. A., Gali, F., & Hadi, N. (2022). The role of cytokines, adhesion molecules, and toll-like receptors in atherosclerosis progression: the effect of Atorvastatin. Journal of medicine and life 15(6), 751–756. https://doi.org/10.25122/jml-2021-0187
Steiner, O., Coisne, C., Cecchelli, R., Boscacci, R., Deutsch, U., Engelhardt, B., & Lyck, R. (2010). Differential roles for endothelial ICAM-1, ICAM-2, and VCAM-1 in shear-resistant T cell arrest, polarization, and directed crawling on blood-brain barrier endothelium. Journal of immunology (Baltimore, Md.: 1950) 185(8), 4846–4855. https://doi.org/10.4049/jimmunol.0903732
Guha, D., Misra, V., Yin, J., Horiguchi, M., Uno, H., & Gabuzda, D. (2023). Vascular injury markers associated with cognitive impairment in people with HIV on suppressive antiretroviral therapy. AIDS (London, England) Advance online publication. https://doi.org/10.1097/QAD.0000000000003675
Affi, R., Gabillard, D., Kouame, G. M., et al. (2022). Plasma sVCAM-1, antiretroviral therapy and mortality in HIV-1-infected west African adults. HIV Medicine, 23(7), 717–726. https://doi.org/10.1111/hiv.13230
Okay, G., Koc, M. M., Guler, E. M., et al. (2020). The effect of antiretroviral therapy on IL-6, IL-1β, TNF-α, IFN-γ levels and their relationship with HIV-RNA and CD4 + T cells in HIV patients. Current HIV Research, 18, 354–361. https://doi.org/10.2174/1570162X18666200712174642
Teasdale, C. A., Hernandez, C., Zerbe, A., et al. (2020). Changes in D-dimer after initiation of antiretroviral therapy in adults living with HIV in Kenya. BMC Infectious Diseases, 20(1), 508. https://doi.org/10.1186/s12879-020-05213-1
Mercurio, V., Fitzgerald, W., Vanpouille, C., Molodtsov, I., & Margolis, L. (2021). Mechanisms of residual immune activation in HIV-1-infected human lymphoid tissue ex vivo. AIDS (London England), 35(8), 1179–1190. https://doi.org/10.1097/QAD.000000000000288
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
Conceptualization: Agnieszka Lembas; Investigation: Agnieszka Lembas and Michał Peller; Writing - original draft preparation: Agnieszka Lembas and Michał Peller; Writing - review and editing: Andrzej Załęski, Tomasz Mikuła and Alicja Wiercińska-Drapało; Resources: Agnieszka Lembas; Supervision: Alicja Wiercińska-Drapało.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Ethics Approval
This is a review article. The Bioethics Committee of Medical University of Warsaw has confirmed that no ethical approval was required.
Additional information
Communicated by Daniel Conklin.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lembas, A., Załęski, A., Peller, M. et al. Human Immunodeficiency Virus as a Risk Factor for Cardiovascular Disease. Cardiovasc Toxicol 24, 1–14 (2024). https://doi.org/10.1007/s12012-023-09815-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-023-09815-4