Abstract
In this study, we investigated the ability of commonly used neuropsychological tests to detect cognitive and functional decline across the Alzheimer’s disease (AD) continuum. Moreover, as preclinical AD is a key area of investigation, we focused on the ability of neuropsychological tests to distinguish the early stages of the disease, such as individuals with Subjective Memory Complaints (SMC). This study included 595 participants from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) dataset who were cognitively normal (CN), SMC, mild cognitive impairment (MCI; early or late stage), or AD. Our cognitive measures included the Rey Auditory Verbal Learning Test (RAVLT), the Everyday Cognition Questionnaire (ECog), the Functional Abilities Questionnaire (FAQ), the Alzheimer’s Disease Assessment Scale–Cognitive Subscale (ADAS-Cog), the Montreal Cognitive Assessment scale (MoCA), and the Trail Making test (TMT-B). Overall, our results indicated that the ADAS-13, RAVLT (learning), FAQ, ECog, and MoCA were all predictive of the AD progression continuum. However, TMT-B and the RAVLT (immediate and forgetting) were not significant predictors of the AD continuum. Indeed, contrary to our expectations ECog self-report (partner and patient) were the two strongest predictors in the model to detect the progression from CN to AD. Accordingly, we suggest using the ECog (both versions), RAVLT (learning), ADAS-13, and the MoCA to screen all stages of the AD continuum. In conclusion, we infer that these tests could help clinicians effectively detect the early stages of the disease (e.g., SMC) and distinguish the different stages of AD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is estimated that 50 million people are living with dementia and that dementia costs approximately 818 billion USD worldwide (Alzheimer’s Disease International, 2015). Alzheimer’s disease (AD) is the leading cause of dementia and accounts for approximately 70-80% of diagnoses (Mendez, 2021; Moustafa, 2021). AD is defined as a progressive neurodegenerative disorder that impacts factors such as declarative memory, episodic memory, executive functioning, spatial and visuoperceptual processing, and language (McKhann et al., 2011; Mendez, 2021). The initial symptoms of AD generally appear in older adults (over 65 years); however, early-onset AD can begin between 30 and 60 years of age (National Institute on Aging, 2017). Despite the overwhelming burden of the disease, the causes of AD are unknown and current treatments are largely unsuccessful in preventing or slowing disease progression (Ang et al., 2020; Cutsuridis & Moustafa, 2017; Cutsuridis & Moustafa, 2016; Rasmussen & Langerman, 2019; Warren & Moustafa, 2022). Accordingly, there is a critical need to understand the progression of AD.
AD diagnoses are generally received in the later stages of the disease when the neurological damage is deemed irreversible (Hill et al., 2020; Rasmussen & Langerman, 2019). In turn, it is difficult to understand the progression of the disease. However, it is now recognized that AD may begin decades before the manifestation of clinical symptoms (Dubois et al., 2016a). In 2011, The National Institute on Aging and Alzheimer’s Association workgroup (NIA-AA) outlined a three-stage clinical diagnostic criterion for AD to understand how the disease progresses from normal cognition to dementia (Jack et al., 2011; Sperling et al., 2011). The workgroup defined these three stages as preclinical AD, mild cognitive impairment (MCI), and AD dementia (Jack et al., 2011). Since 2011, a conceptual shift has occurred that considers the three AD stages existing on a continuum spanning from asymptomatic to significantly impaired (Dubois et al., 2016b; Jack et al., 2018). In turn, medical professionals and researchers increasingly acknowledge that targeting preclinical symptoms and early-stage cognitive decline could delay AD and prevent an impending public health crisis (Eppig et al., 2020). Developments in early treatment and diagnosis can also be beneficial to individuals via improving their quality of life, healthcare systems via decreasing the intensity and cost of care, and caregivers via preserving individuals cognitive and functional ability (for more information see, Dubois et al., 2016a, b; Leifer, 2003; Weimer & Sager, 2009).
Preclinical AD is an early stage of the disease where symptoms are clinically asymptomatic but precede longitudinal cognitive decline (Aisen et al., 2017; Budson & Solomon, 2012a). Studying preclinical AD is difficult as it requires highly detailed measures and because individuals often present as clinically ‘normal’ (Aisen et al., 2017; Budson & Solomon, 2012b; Sperling et al., 2011). In the literature, neuropsychological (e.g., cognitive tests), neuroimaging, and biomarker measures are frequently combined to detect early-stage AD (i.e.., preclinical AD) and predict MCI to AD conversion (Brown, 2015). In clinical practice, neuropsychological tests are the predominant methods used to identify AD. However, these cognitive and behavioral tests do have some inherent limitations. For example, commonly used multi-domain cognitive tests are often over-generalized and lack the specificity and sensitivity to detect subtle cognitive changes in early-stage AD (Arevalo-Rodriguez et al., 2021). Moreover, many functional and cognitive tests can only assess the later-stage symptoms of AD. However, research suggests that some cognitive measures—such as progressive episodic-memory decline—can be effectively captured by domain-specific tests and can be used to detect early-stage AD (El Haj et al., 2015; Warren et al., 2021). Nonetheless, the early stages of AD, such as individuals with a significant memory concern (SMC), are under-researched.
Prior research
Prior research has focused on three general types of neuropsychological tests for AD assessment. Specifically, the literature often studies specific cognitive tests of one domain (e.g., episodic memory), tests of functional impairment, and cognitive batteries. Regarding specific tests, the Rey Auditory Verbal Learning Test (RAVLT) is the most common measure used in the literature. The RAVLT is a psychometrically rigorous measure of episodic memory, widely used in dementia and pre-dementia assessment. Previous research has shown that the RAVLT can differentiate between individuals categorized as cognitively normal (CN), SMC, MCI, and AD (Estevez-Gonzalez et al., 2003). More recently, Moradi et al. (2017) found a strong association between RAVLT scores and early-stage AD-related brain atrophy. Moreover, Warren et al. (2021) found that the RAVLT could predict conversion from MCI to AD. In turn, prior research suggests that the RAVLT may be an effective tool in highlighting subtle episodic-memory decline in individuals with SMC at risk of progressing to MCI (Moradi et al., 2017).
Functional impairments such as difficulty performing activities of daily living (ADL) are also used to study the stages of AD (McKhann et al., 2011). However, these functional tests are often developed for late-stage dementia and can lack the sensitivity required to detect the subtle functional impairments in SMC and MCI (Knopman & Caselli, 2012). To overcome this late-stage bias, researchers suggest using self-report and informant-report measures more sensitive to the early stages of AD (Knopman & Caselli, 2012). For example, self-report may be useful in capturing early functional decline in SMC, and informant-report measures may be beneficial for capturing decline during MCI due to the overt anosognosia accompanying the disorder (Knopman & Caselli, 2012; Ryan et al., 2019). Instrumental ADLs (IADL) are functional activities such as cooking, transportation, and managing finances that allow an individual to live independently (Marshall et al., 2012). As IADL relies heavily on cognitive functioning, they are generally detected during the MCI stage (Marshall et al., 2012). However, IADL measures sensitive to subtle functional decline may also help detect SMC (Marshall et al., 2012). The Functional Activities Questionnaire (FAQ) (Pfeffer et al., 1982) is a subjective, informant-rated test of IADL. Research has shown the total FAQ score can discriminate between CN, MCI, and AD dementia (Marshall et al., 2012). However, due to floor effects, the FAQ has previously failed to capture early functional changes in CN individuals at-risk of AD (Marshall et al., 2015). Subsequently, further research is required to understand the ability of functional tests to detect the early stages of AD, such as SMC.
Cognitive batteries or general cognitive tests are also commonly used to assess AD. These general tests are the most common clinical assessment tool and usually contain various cognitive measures. Multiple general tests have been used in the literature, such as the Everyday Cognition Questionnaire (ECog), Alzheimer’s Disease Assessment Scale – Cognitive Scales (ADAS-Cog), Montreal Cognitive Assessment Scale (MoCA), and the Trail Making Test, Part B (TMT-B). The ECog (Farias et al., 2008) is a self and informant-rated questionnaire that assesses subjective perception of functional performance across memory, language, semantic knowledge, visuospatial abilities, and executive functioning. These domains have been found to correlate with objective cognitive measures, AD biomarkers, and neuroimaging indicators of AD (Farias et al., 2008). Longitudinal studies have further demonstrated that baseline ECog performance in CN individuals can predict subsequent progression to MCI (Farias et al., 2013). Similarly, the ADAS-Cog is a neuropsychological assessment that can determine the severity of the cognitive dysfunction or decline in individuals with AD or MCI (Rosen et al., 1984). The two versions—the ADAS-Cog-11 and ADAS-Cog-13—assess memory, language, praxis, and orientation to determine an individual’s level of cognitive functioning (Skinner et al., 2012). The MoCA has also been used to assess cognition in MCI and dementia. It is a short screening tool that evaluates an individual’s attention, working memory, episodic memory, executive function, language, and visuospatial abilities. It has been shown to have high sensitivity and specificity for determining whether individuals have MCI (Nasreddine et al., 2005). Lastly, the TMT-B measures executive function, including the cognitive processes of working memory, inhibition, and attention. A decline in executive function usually occurs in the early stages of AD and contributes to cognitive impairment (Baudic et al., 2006). To our knowledge, the TMT-B has not been applied to SMC and evidence suggests that it cannot significantly distinguish CN from MCI (Papp et al., 2014). In conclusion, multiple general tests are widely used to assess MCI and AD dementia. However, further research is required to understand the ability of general tests to detect the early stages of AD, such as SMC.
The current study
Prior research has shown that neuropsychological tests can detect the middle-to-late stages of the AD continuum, such as MCI and AD. However, there is less research on the early stages of the disease, such as SMC and early-stage MCI (EMCI). Accordingly, we plan to evaluate the use of neuropsychological tests to identify the various stages of the AD continuum. Accordingly, in this study, we will investigate whether commonly used cognitive and functional assessments can predict CN, SMC, EMCI, late-stage MCI (LMCI), and AD dementia (for more information on the characteristics and differences between EMCI and LMCI see, Alzheimer’s Disease Neuroimaging Initiative, 2008, p. 27). We have specifically chosen to use the RAVLT, ECog, FAQ, ADAS-Cog, MoCA, and TMT-B as these measures are widely used but lack application to early-stage AD. Specifically, we examine whether these measures can predict the progression from CN, SMC, EMCI, LMCI, and AD. In turn, we hypothesize that the RAVLT (i.e., immediate, learning, forgetting, or forgetting percentage), ADAS-cog (i.e., ADAS-11 and ADAS-13), FAQ, ECog, and MoCA will predict progression towards AD. That is, for every unit increase in these test scores, there will be a significant increase in the odds of an individual progressing toward AD.
Methods
Data source and acquisition
We obtained the data for this study from the Alzheimer’s Disease Neuroimaging Initiative (ADNI) dataset. ADNI recruits its participants through its website, media advertisement (newspaper, radio, and social media), and third-party health care providers. ADNI collects and deidentifies data at various ADNI sites across North America using trained clinicians. As in prior studies (Alashwal et al., 2020; Moustafa et al., 2021; Warren et al., 2021), The ADNI data can be categorized into four different collection periods, ADNI-1 (2004 to 2009), ADNI-GO (2009 to 2011), ADNI-2 (2011 to 2016), and ADNI-3 (2016 to 2021). Each of these stages involves the addition of new measures and participants as well as the continued testing of some prior participants. Access to the ADNI dataset was obtained via a prior application, and institutional ethics approval was obtained from our university’s Human Research Ethics Committee in April 2021. Therefore, this study was in compliance with the 1964 Declaration of Helsinki and its later addenda. For security, we stored the data on a password-protected computer that was only accessible to the research team.
Participants
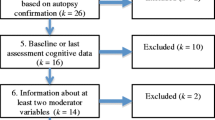
This study includes 596 participants aged 55 to 90 years old who identified as Male (n = 287) or Female (n = 309). Of these participants, 140 were diagnosed as CN, 191 as SMC, 162 as EMCI, 68 as LMCI, and 35 as AD. These diagnoses are confirmed by ADNI clinicians using the Cognitive Change Index (CCI), Clinical Dementia Rating Scale (CDR), the Logical Memory subtest (LMT) of the Wechsler Memory Scale-Revised, the Mini-mental State Exam (MMSE), and criteria from the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer’s Disease and Related Disorders Association Alzheimer’s Criteria (NINCDS-ADRDA). It is important to note that our SMC participants could be operationalized as individuals with Subjective Cognitive impairments because of the tests used in this study. However, we maintain the use of the term SMC as it is the diagnostic term used by ADNI in our data. For more information on the diagnostic criteria, see the ADNI procedures manual (Alzheimer’s Disease Neuroimaging Initiative, 2017). Before the main analysis, we ran a sample size analysis using the software G*Power (Faul et al., 2009; Faul et al., 2007). We found that a sample size of 178 would be sufficient to detect if the neuropsychological assessments could predict AD progression (f2 = .15, power = 0.95; n-predictors =11, α = .05).
Scales
Our analysis used six neuropsychological scales to detect cognitive and functional decline across the AD spectrum. These scales are outlined below.
Rey Auditory Verbal Learning Test (RAVLT)
The RAVLT (Rey, 1964) is an episodic-memory test that assesses participants’ ability to learn 15 nouns across five learning trials, to recall the words immediately following an interference word list, and to recognize and recall words after a 30-minute time delay. Four summary scores are derived from RAVLT raw scores, including the following: Immediate (participants’ recall immediately following the first list learning trial), Forgetting (number of words forgotten across all trials), Learning (the number of correct words recalled across all trials), and Percent Forgetting (percentage quantification of the RAVLT forgetting score). The RAVLT is widely used in AD research because it targets episodic-memory decline, a fundamental aspect of early-stage AD progression (Warren et al., 2021).
Everyday Cognition (ECog)
The ECog (Farias et al., 2008) is a 39-item questionnaire that assesses the subjective perception of everyday cognitive functioning and performance across six domains: memory, language, semantic knowledge, visuospatial abilities, and executive functioning. Disfunctions across these six domains are markers of the neuropsychological decline (e.g., cognitive and functional decline) observed in AD. The ECog includes identical self-report and study-partner versions. Participants rate perceived ability on tasks compared to their ability ten years prior. The questionnaire is rated on a four-point scale where one is “better or no change compared with ten years earlier,” and four is “consistently much worse.” A total self-report score, and total study-partner score, are calculated by averaging the ratings across the 39 items.
Functional Activities Questionnaire (FAQ)
The FAQ (Pfeffer et al., 1982) is a 10-item, informant-rated measure of IADL. Decline in IADL is a significant marker of AD decline as discussed in the introduction above (p. 4). In the FAQ, each item is scored on a three-point scale where higher scores indicate greater impairment (0 = normal; 1 = has difficulty but does by self; 2 = requires assistance; 3 = dependent). No established cut-off score signifies functional impairment; however, research suggests a score of ≥6 out of a possible 30 (Nitrini et al., 2004).
Alzheimer’s Disease Assessment Scale – Cognitive Scales (ADAS-Cog)
The ADAS-Cog-11 (Rosen et al., 1984) measure consists of 11 subtests that assess numerous cognitive abilities whose impairment is associated with AD (e.g., executive function). The ADAS-Cog-13 includes these 11 subtests and two additional tests to assess executive and daily functioning associated with MCI. The measures take approximately 30-45 minutes to administer using a test kit. ADAS-Cog-11 scores range from 0 to 70, and ADAS-Cog-13 scores are from 0 to 85.
Montreal Cognitive Assessment (MoCA)
The MoCA (Nasreddine et al., 2005) is a cognitive screening instrument developed to detect mild cognitive impairment through assessing multiple cognitive domains with 11 tasks. The scores of all subtests are summed, and an additional point is given if an individual has completed 12 years or less of formal education. The maximum score possible is 30 points, and scores of 26 and above are considered normal. It takes ten minutes to administer using paper and a pencil. Unlike other scales, the MoCA was created specifically for MCI and is sensitive to mid-to-early stages symptoms of AD, such as short-term memory recall and executive functioning.
Trail Making Test, Part B (TMT-B)
The TMT-B (Reitan, 1955) task requires an individual to create an ascending pattern of alternating numbers and letters quickly and accurately. Mistakes are pointed out to participants who should correct them immediately, and the final score is based on the task completion time. It is completed in under 300 seconds, and the average score is 75 seconds, while a deficient score is greater than 273 seconds. The TMT-B assesses neuropsychological mechanism such as task switching and visual attention that decline with AD progression.
Statistical analysis
All statistical analyses were performed using SPSS for Windows (version 28.0). Frequencies and percentages are used for categorical variables and means, SDs, and 95% confidence intervals are reported for continuous variables. Pearson correlation coefficients are reported between all variables. The generalized linear model in SPSS was used to conduct an ordinal logistic regression to determine if the neuropsychological assessments predicted cognitive and functional decline across the AD spectrum.
Results
The descriptive statistics and correlations for the variables included in the ordinal regression are presented in Table 1. Notably, these findings indicate that the ADAS-11 and ADAS-13 are strongly correlated. Therefore, only the ADAS-13 was used in the following ordinal regression model.
Multi-collinearity
The correlation coefficients in Table 1 indicate the presence multi-collinearity amongst some of the variables (e.g., ADNI 11 & ADNI 13; forgetting and forgetting %). To further investigate, VIF and tolerance values were obtained for each of the IVs in the hypothesised model. The analysis showed that the assumption of multicollinearity was violated (i.e., VIF > 5 and TOL < 0.10) for ADAS-11 (VIF = 18.22, Tol = .06), ADAS-13 (VIF = 24.35, Tol = .04), Forgetting (VIF = 6.34, Tol = .16), Forgetting % (VIF = 0.01, Tol = .10). After removing ADAS-11 and forgetting percentage the assumptions for multi-collinearity were met (i.e., all variables had a TOL > .10 and VIF <5).
Ordinal regression
An ordinal regression was conducted to test if gender, age, and neuropsychological assessments can predict the cognitive and functional decline across the AD spectrum (i.e., cognitive normal, Subjective Memory Complaints, early mild cognitive impairment, late cognitive impairment, and AD). Note that from the complete ADNI data set (N = 2129), only 596 participants had data for all the Neuropsychological assessments at baseline. Table 2 presents the descriptive statistics for each variable across all AD levels (i.e., CN, SMC, EMCI, LMCI, AD).
The omnibus model for the ordinal regression was statistically significant χ2 (11, 596) = 455.23, p < .001; and the results confirmed the model was a good fit for the data, Pearson χ2 (2369) = 2053.60, p = .867. The results of the ordinal regression are presented in Table 3.
As presented in Table 3, the model’s predictive power was improved by age, ADAS-13, RAVLT learning, FAQ, MOCA, Ecog-patient, and Ecog-partner. However, gender, RAVLT immediate, RAVLT forgetting, and TMT-B scores did not contribute significantly to the model. The odds ratio for age indicated that for every single unit increase in age, there was a small 2.00% significant decrease in the probability that an individual would progress towards AD. For RAVLT learning, there was a significant 8.30% decrease in the odds that a person would progress towards AD. There was a significant 13.69% increase in the odds that a person would progress towards AD for every unit increase in FAQ. A single unit increase in MOCA resulted in a significant 8.91% decrease in the odds that an individual would progress towards AD. For every single unit increase in a participant’s self-reported cognitive decline, there was a significant 152.37% increase in the odds of progressing towards AD. Finally, there was a significant 274.13% increase in the odds that a person would progress towards AD for every single unit increase in partner reported cognitive decline.
Discussion
In this study, we aimed to distinguish CN, SMC, EMCI, LMCI, and AD groups using cognitive and functional tests (the RAVLT, ECog, FAQ, ADAS-Cog, MoCA, and TMT-B). We focused on these measures because identifying differences in cognition and function could have potential real-world implications for detecting early-stage AD. We hypothesized that—out of the tests studied—the RAVLT (i.e., immediate, learning, forgetting, or forgetting percentage), ADAS-cog (i.e., ADAS-11 and ADAS-13), FAQ, ECog, and MoCA would predict progression towards AD. Our results provide evidence that some cognitive and functional tests can successfully predict the stages of the AD continuum (i.e., ADAS-13, RAVLT learning, FAQ, MOCA, and ECog). However, other neuropsychological assessments were not predictive of the stages of AD (i.e., RAVLT immediate and forgetting, and TMT-B).
Cognitive and functional measures in different dementia stages
We investigated the ability of the RAVLT to predict the different stages of the AD continuum (i.e., CN, SMC, EMCI, LMCI, and AD groups). As episodic-memory decline is an early symptom of AD, it was hypothesized that RAVLT scores would predict the progression towards AD. Surprisingly, this hypothesis was only confirmed for the RAVLT learning but the forgetting sub-scales were not predictive of the progression towards AD. Our findings support previous research that found that the RAVLT can differentiate between CN individuals and individuals with MCI and AD, suggesting that RAVLT scores accurately reflect underlying dementia pathology (Estevez-Gonzalez et al., 2003; Moradi et al., 2017; Warren et al., 2021). However, our findings did not support previous research suggesting that individuals with SMC have accelerated episodic-memory decline compared to healthy individuals without SMC (Samieri et al., 2014). Building on previous research, the current study found that the RAVLT is also sensitive to subtle episodic-memory differences between SMC and EMCI as well as EMCI and LMCI and may be a useful measure to pinpoint where an individual is on the AD spectrum. Therefore, future research should aim to develop measures of episodic memory sensitive to differentiating between healthy controls and SMC to capture at-risk individuals earlier in the disease progression.
We also investigated the ability of the ECog to predict the different stages of the AD continuum (i.e., CN, SMC, EMCI, LMCI, and AD groups). As predicted, results showed that the ECog Total self-report strongly predicted AD categories. Indeed, for every single unit increase in a participant’s self-reported cognitive decline, there was a 166.60% increase in the odds of progressing towards AD. The ECog partner reported cognitive decline was the strongest predictor within our model. For example, for every additional single unit increase in partner reported cognitive decline, there was a significant 307.40% increase in the odds that a person would progress towards AD. Contrary to our hypothesis, our results showed that the ECog Total study-partner was a significant predictor and the strongest predictor of the AD categories. These findings oppose previous research that suggests self-report measures are less viable the further individuals’ decline (Marshall et al., 2012). These findings suggest that subjective self-report measures may be important in detecting SMC or early-stage AD and that informant-report measures may be more viable for middle to late-stage AD. Accordingly, both measures should be used to effectively capture the transitions between the various stages of the AD continuum (Marshall et al., 2012).
We assessed the ability of the FAQ to predict differences in IADL across the AD continuum. Results confirmed our hypothesis and showed that the FAQ significantly predicted the progression toward AD. This finding supports previous research that found the FAQ could discriminate between IADL scores for CN, MCI, and AD dementia (Marshall et al., 2012). However, due to floor effects, the FAQ is not sensitive enough to capture early functional changes between healthy controls and individuals with SMC (Marshall et al., 2015). This result may be because the FAQ is an entirely informant-based measure and may not be appropriate for use in CN individuals for whom self-report may be more accurate. There is a critical need for IADL measures that can detect subtle changes in otherwise healthy individuals who report SMC. Accordingly, future research should consider administering a self-report version of the FAQ (Ryan et al., 2019), investigate whether individual items of the FAQ can distinguish CN and SMC, and develop scales that focus exclusively on complex ADL tasks in early-stage AD (Marshall et al., 2012).
Regarding the ADAS-Cog, we hypothesized that the ADAS-13 would predict the different stages of the AD Continuum. This hypothesis was based on research that demonstrated that the ADAS-Cog-11 can effectively differentiate between CN and those with MCI or early AD (Zainal et al., 2016). Similarly, research conducted by Skinner et al. (Skinner et al., 2012) using the ADNI dataset demonstrated that the ADAS-Cog-13 was a valid measure with high responsiveness for detecting MCI and AD. The current study’s results supported the hypothesis that the ADAS-Cog-13 scores significantly predict the five stages of AD included in our analysis. This finding also supports the notion that all five groups have been shown to have differing levels of cognitive impairment (Roberts et al., 2009). From these findings, we recommend that the ADAS-Cog-13 be used in clinical practice where individuals present with minor memory issues, as it can differentiate between the levels of cognitive functioning expected throughout the AD continuum.
We predicted that the MoCA would be useful in predicting the stages of AD, as some researchers have used it as a diagnostic measure to separate those with SMC, EMCI, LMCI, or AD (Larner, 2019). A meta-analysis (Breton et al., 2019) demonstrated that the MoCA has good diagnostic accuracy for identifying individuals with MCI. Our research showed that the MoCA could predict the AD continuum in line with the existing literature. For example, our results support previous findings that a decline in cognitive functioning—as detected by the MOCA—is a significant predictor of the progression toward AD. (Montejo Carrasco et al., 2017). Therefore, the findings supported our prediction, as the tool can correctly identify the progression of cognitive decline between all five AD continuum groups. In contrast, we expected that the TMT-B would be unable to classify individuals with SMC correctly. Specifically, research has suggested that the TMT-B cannot significantly differentiate between individuals who are CN, have MCI (Papp et al., 2014), or those with MCI or AD (Ashendorf et al., 2008). Our findings supported the previous literature as we observed that the TMT-B scores were not a significant predictor of AD progression. Accordingly, the results from our study call into question the use of the TMT-B as diagnostic measures in research and as screening tools for SMC in clinical practice.
Strengths
While extensive research has been carried out to investigate AD and MCI, there is limited evidence on SMC overall. A particular strength of the current study is that we identified four existing tools that are significant predictors of the progression from SMC to AD (i.e., ADAS-13, RAVLT – learning, FAQ, MoCA, and ECog). Our study also determined measures that cannot predict disease progression accurately (i.e., RAVLT immediate and forgetting, and TMT-B). This finding is important as the measures investigated are used in routine assessments of cognitive impairment; if clinicians know which tools can accurately detect SMC, they could be used when less severe memory issues are suspected. This change in practice would likely reduce the burden on the individual and the healthcare system, as fewer individuals would require a full AD or MCI assessment. We also suggest that cognitive measures which assess recall of instructions or visual words be further investigated to define their usefulness in SMC assessments. Research must continue to be conducted in this area so a clearer link between cognition and SMC can be established, and the definition of SMC can be streamlined to ensure consistency in assessment and diagnosis. This research is important in caring for the hundreds of thousands of dementia patients living with memory decline, as early SMC interventions may help prevent further deterioration into the later stages of dementia, for which there currently are no treatments or cures.
Limitations
Despite the beneficial clinical implications of this research, there are some limitations. Firstly, while the measures studied are used to assess function and cognition, they are only part of the diagnostic process; these diagnoses also require reports from the individual and their carers, together with neuroimaging and neuropsychological assessments. This scope means that cognitive tests alone, while useful, cannot be relied upon for complete diagnoses of any level of impaired cognitive functioning. Similarly, most measures do not consider a person’s ability before cognitive impairment or other issues such as vision or English language proficiency. These are important factors that can impact performance and indicate whether there is a true decline in the cognitive domain of memory or whether the person’s poor performance can be attributed to a different cause. Regarding the data, the ADNI sample is a highly characterized Northern American sample and does not represent the general population. All groups showed a considerably high mean level of education (15–16 years), which may affect the generalisability of the results. Some measures that rely on subjective-report (such as the ECog and the FAQ) may also be inherently biased and have been criticized as they do not assess an individual’s objective abilities (Marshall et al., 2012). Similarly, the information provided by study partners may be influenced by confounding factors, including; whether the informant lives with the participant, the relationship status between participant and informant, and the number of hours a week they spend together and in what circumstances (Ryan et al., 2019). These limitations should be considered when generalizing our results.
Conclusions and future directions
It is predicted that the prevalence of dementia will increase across the globe. Research is therefore focused on earlier diagnosis and intervention at the preclinical stage. This research requires using sensitive cognitive measures that are fast and inexpensive to administer by clinicians in a primary health care setting (Marshall et al., 2012). Such tests can be used as initial AD screeners to identify at-risk individuals for follow-up assessments using biomarker tests. As the current study highlights, commonly used cognitive scales in AD clinical trials (such as some RAVLT measures, the ECog, and FAQ) reliably detect decline as individuals transition from SMC, to MCI, to AD; however, these scales are less sensitive to capturing subtle decline between healthy individuals and those with SMC in preclinical AD. Therefore, developing highly sensitive measures that emphasize episodic-memory decline, complex cognitive functioning, and IADL tasks in preclinical AD will help identify those at risk of AD and allow for earlier treatment. We hope this research can help clinicians identify the early stages of AD, such as SMC, and encourage further research into neuropsychological tests for the AD continuum.
Data availability
“This data is available via application to the Alzheimer’s Disease Neuroimaging Initiative (ADNI). Data collection and sharing for this project was funded by ADNI (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defence award number W81XWH-12-2-0012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: AbbVie, Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; Araclon Biotech; BioClinica, Inc.; Biogen; Bristol-Myers Squibb Company; CereSpir, Inc.; Cogstate; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; EuroImmun; F. Hoffmann-La Roche Ltd. and its affiliated company Genentech, Inc.; Fujirebio; GE Healthcare; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Lumosity; Lundbeck; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Neurotrack Technologies; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Takeda Pharmaceutical Company; and Transition Therapeutics. The Canadian Institutes of Health Research is providing funds to support ADNI clinical sites in Canada. Private sector contributions are facilitated by the Foundation for the National Institutes of Health (www.fnih.org). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s Therapeutic Research Institute at the University of Southern California. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.” Note that this excerpt is from the ADNI data use agreement and is required for publication.
Code availability
Not applicable.
Change history
03 March 2023
Missing Open Access funding information has been added in the Funding Note.
References
Aisen, P. S., Cummings, J., Jack, C. R., Jr, Morris, J. C., Sperling, R., Frolich, L., Jones, R. W., Dowsett, S. A., Matthews, B. R., Raskin, J., Scheltens, P., & Dubois, B. (2017). On the path to 2025: Understanding the Alzheimer's disease continuum. Alzheimer's Research & Therapy, 9(1), 60. https://doi.org/10.1186/s13195-017-0283-5.
Alashwal, H., Diallo, T. M. O., Tindle, R., & Moustafa, A. A. (2020). Latent class and transition analysis of Alzheimer’s disease data. Frontiers in Computer Science, 2, 551481.
Alzheimer’s Disease International. (2015). World Alzheimer report 2015. https://www.alzint.org/resource/worldalzheimer-report-2015/
Alzheimer’s Disease Neuroimaging Initiative (2008). ADNI2 procedures manuals. https://adni.loni.usc.edu/wp-content/uploads/2008/07/adni2-procedures-manual.pdf
Alzheimer’s Disease Neuroimaging Initiative (2017). ADNI3 procedures manuals. http://adni.loni.usc.edu/adni-3/procedures-manuals/
Ang, T. F., Joshi, P., & Au, R. (2020). Vascular risk factors and their relationship to brain aging: Findings from the Framingham heart study. In D. J. Libon, M. Lamar, R. A. Swenson, & K. M. Heilman (Eds.), Vascular disease, Alzheimer’s disease, and mild cognitive impairment: Advancing an integrated approach (pp. 3–29). Oxford University Press. https://doi.org/10.1093/oso/9780190634230.003.0002
Arevalo-Rodriguez, I., Smailagic, N., Roque-Figuls, M., Ciapponi, A., Sanchez-Perez, E., Giannakou, A., Pedraza, O. L., Bonfill Cosp, X., & Cullum, S. (2021). Mini-mental state examination (MMSE) for the early detection of dementia in people with mild cognitive impairment (MCI). Cochrane Database of Systematic Reviews, 7, CD010783. https://doi.org/10.1002/14651858.CD010783.pub3
Ashendorf, L., Jefferson, A. L., O'Connor, M. K., Chaisson, C., Green, R. C., & Stern, R. A. (2008). Trail making test errors in normal aging, mild cognitive impairment, and dementia. Archives of Clinical Neuropsychology, 23(2), 129–137. https://doi.org/10.1016/j.acn.2007.11.005
Baudic, S., Barba, G. D., Thibaudet, M. C., Smagghe, A., Remy, P., & Traykov, L. (2006). Executive function deficits in early Alzheimer's disease and their relations with episodic memory. Archives of Clinical Neuropsychology, 21(1), 15–21. https://doi.org/10.1016/j.acn.2005.07.002
Breton, A., Casey, D., & Arnaoutoglou, N. A. (2019). Cognitive tests for the detection of mild cognitive impairment (MCI), the prodromal stage of dementia: Meta-analysis of diagnostic accuracy studies. International Journal of Geriatric Psychiatry, 34(2), 233–242. https://doi.org/10.1002/gps.5016
Brown, J. (2015). The use and misuse of short cognitive tests in the diagnosis of dementia. Journal of Neurology, Neurosurgery & Psychiatry, 86(6), 680–685. https://doi.org/10.1136/jnnp-2014-309086
Budson, A. E., & Solomon, P. R. (2012a). New criteria for Alzheimer disease and mild cognitive impairment: Implications for the practicing clinician. The Neurologist, 18(6), 356–363. https://doi.org/10.1097/NRL.0b013e31826a998d
Budson, A. E., & Solomon, P. R. (2012b). New diagnostic criteria for Alzheimer's disease and mild cognitive impairment for the practical neurologist. Practical Neurology, 12(2), 88–96. https://doi.org/10.1136/practneurol-2011-000145
Cutsuridis, V., & Moustafa, A. A. (2016). Multiscale models of pharmacological, immunological and neurostimulation treatments in Alzheimer’s disease. Drug Discovery Today: Disease Models, 19, 85–91.
Cutsuridis, V., & Moustafa, A. A. (2017). Computational models of pharmacological and immunological treatment in Alzheimer’s disease. Computational Models of Brain and Behavior, 99–108.
Dubois, B., Hampel, H., Feldman, H. H., Scheltens, P., Aisen, P., Andrieu, S., Bakardjian, H., Benali, H., Bertram, L., Blennow, K., Broich, K., Cavedo, E., Crutch, S., Dartigues, J. F., Duyckaerts, C., Epelbaum, S., Frisoni, G. B., Gauthier, S., Genthon, R., et al. (2016a). Preclinical Alzheimer's disease: Definition, natural history, and diagnostic criteria. Alzheimers & Dementia, 12(3), 292–323. https://doi.org/10.1016/j.jalz.2016.02.002
Dubois, B., Padovani, A., Scheltens, P., Rossi, A., & Dell'Agnello, G. (2016b). Timely diagnosis for Alzheimer's disease: A literature review on benefits and challenges. Journal of Alzheimer's Disease, 49(3), 617–631. https://doi.org/10.3233/JAD-150692
El Haj, M., Antoine, P., Nandrino, J. L., & Kapogiannis, D. (2015). Autobiographical memory decline in Alzheimer's disease, a theoretical and clinical overview. Ageing Research Reviews, 23(Pt B), 183–192. https://doi.org/10.1016/j.arr.2015.07.001
Eppig, J., Werhane, M., Edmonds, E. C., Wood, L.-D., Bangen, K. J., Jak, A., Thomas, K. R., Wong, C., Weigand, A., & Bondi, M. W. (2020). Neuropsychological contributions to the diagnosis of mild cognitive impairment associated with Alzheimer’s disease. In M. L. D. J. Libon, R. A. Swenson, & K. M. Heilman (Eds.), Vascular disease, Alzheimer’s disease, and mild cognitive impairment (pp. 52–82). Oxford University Press. https://doi.org/10.1093/oso/9780190634230.003.0004
Estevez-Gonzalez, A., Kulisevsky, J., Boltes, A., Otermin, P., & Garcia-Sanchez, C. (2003). Rey verbal learning test is a useful tool for differential diagnosis in the preclinical phase of Alzheimer's disease: Comparison with mild cognitive impairment and normal aging. International Journal of Geriatric Psychiatry, 18(11), 1021–1028. https://doi.org/10.1002/gps.1010
Farias, S. T., Mungas, D., Reed, B. R., Cahn-Weiner, D., Jagust, W., Baynes, K., & Decarli, C. (2008). The measurement of everyday cognition (ECog): Scale development and psychometric properties. Neuropsychology, 22(4), 531–544. https://doi.org/10.1037/0894-4105.22.4.531
Farias, S. T., Chou, E., Harvey, D. J., Mungas, D., Reed, B., DeCarli, C., Park, L. Q., & Beckett, L. (2013). Longitudinal trajectories of everyday function by diagnostic status. Psychology and Aging, 28(4), 1070.
Faul, F., Erdfelder, E., Lang, A. G., & Buchner, A. (2007). G*power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behavior Research Methods, 39(2), 175–191. https://doi.org/10.3758/bf03193146
Faul, F., Erdfelder, E., Buchner, A., & Lang, A. G. (2009). Statistical power analyses using G*power 3.1: Tests for correlation and regression analyses. Behavior Research Methods, 41(4), 1149–1160. https://doi.org/10.3758/BRM.41.4.1149
Hill, N. L., Mogle, J., Bhargava, S., Bell, T. R., Bhang, I., Katz, M., & Sliwinski, M. J. (2020). Longitudinal relationships among depressive symptoms and three types of memory self-report in cognitively intact older adults. International Psychogeriatrics, 32(6), 719–732. https://doi.org/10.1017/S104161021900084X
Jack Jr., C. R., Albert, M. S., Knopman, D. S., McKhann, G. M., Sperling, R. A., Carrillo, M. C., Thies, B., & Phelps, C. H. (2011). Introduction to the recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers & Dementia, 7(3), 257–262. https://doi.org/10.1016/j.jalz.2011.03.004
Jack Jr., C. R., Bennett, D. A., Blennow, K., Carrillo, M. C., Dunn, B., Haeberlein, S. B., Holtzman, D. M., Jagust, W., Jessen, F., Karlawish, J., Liu, E., Molinuevo, J. L., Montine, T., Phelps, C., Rankin, K. P., Rowe, C. C., Scheltens, P., Siemers, E., Snyder, H. M., et al. (2018). NIA-AA research framework: Toward a biological definition of Alzheimer's disease. Alzheimers & Dementia, 14(4), 535–562. https://doi.org/10.1016/j.jalz.2018.02.018
Knopman, D. S., & Caselli, R. J. (2012). Appraisal of cognition in preclinical Alzheimer's disease: A conceptual review. Neurodegenerative Disease Management, 2(2), 183–195. https://doi.org/10.2217/NMT.12.5
Larner, A. J. (2019). Evaluating cognitive screening instruments with the "likelihood to be diagnosed or misdiagnosed" measure. International Journal of Clinical Practice, 73(2), e13265. https://doi.org/10.1111/ijcp.13265
Leifer B. P. (2003). Early diagnosis of Alzheimer's disease: clinical and economic benefits. Journal of the American Geriatrics Society, 51(5 Suppl Dementia), S281–S288. https://doi.org/10.1046/j.1532-5415.5153.x
Marshall, G. A., Amariglio, R. E., Sperling, R. A., & Rentz, D. M. (2012). Activities of daily living: Where do they fit in the diagnosis of Alzheimer's disease? Neurodegenerative Disease Management, 2(5), 483–491. https://doi.org/10.2217/nmt.12.55
Marshall, G. A., Zoller, A. S., Lorius, N., Amariglio, R. E., Locascio, J. J., Johnson, K. A., Sperling, R. A., & Rentz, D. M. (2015). Functional activities questionnaire items that best discriminate and predict progression from clinically normal to mild cognitive impairment. Current Alzheimer Research, 12(5), 493–502. https://doi.org/10.2174/156720501205150526115003
McKhann, G. M., Knopman, D. S., Chertkow, H., Hyman, B. T., Jack Jr., C. R., Kawas, C. H., Klunk, W. E., Koroshetz, W. J., Manly, J. J., Mayeux, R., Mohs, R. C., Morris, J. C., Rossor, M. N., Scheltens, P., Carrillo, M. C., Thies, B., Weintraub, S., & Phelps, C. H. (2011). The diagnosis of dementia due to Alzheimer's disease: Recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers & Dementia, 7(3), 263–269. https://doi.org/10.1016/j.jalz.2011.03.005
Mendez, M. F. (2021). The relationship between anxiety and Alzheimer’s disease. Journal of Alzheimer's Disease Reports, 5(1), 171–177. https://doi.org/10.3233/ADR-210294
Montejo Carrasco, P., Montenegro-Pena, M., Lopez-Higes, R., Estrada, E., Prada Crespo, D., Montejo Rubio, C., & Garcia Azorin, D. (2017). Subjective memory complaints in healthy older adults: Fewer complaints associated with depression and perceived health, more complaints also associated with lower memory performance. Archives of Gerontology and Geriatrics, 70, 28–37. https://doi.org/10.1016/j.archger.2016.12.007
Moradi, E., Hallikainen, I., Hanninen, T., Tohka, J., & Alzheimer’s Disease Neuroimaging, I. (2017). Rey’s auditory verbal learning test scores can be predicted from whole brain MRI in Alzheimer’s disease. Neuroimage Clinical, 13, 415–427. https://doi.org/10.1016/j.nicl.2016.12.011
Moustafa, A. A. (2021). Alzheimer’s disease: Understanding biomarkers, big data, and therapy. Elsevier.
Moustafa, A. A., Tindle, R., Alashwal, H., Diallo, T. M. O., & Alzheimer’s Disease Neuroimaging, I. (2021). A longitudinal study using latent curve models of groups with mild cognitive impairment and Alzheimer’s disease. Journal of Neuroscience Methods, 350, 109040. https://doi.org/10.1016/j.jneumeth.2020.109040
Nasreddine, Z. S., Phillips, N. A., Bedirian, V., Charbonneau, S., Whitehead, V., Collin, I., Cummings, J. L., & Chertkow, H. (2005). The Montreal cognitive assessment, MoCA: A brief screening tool for mild cognitive impairment. Journal of the American Geriatrics Society, 53(4), 695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x
National Institute on Aging (2017). What is dementia? Symptoms, types, and diagnosis. National Institute of Health. https://www.nia.nih.gov/health/what-dementia-symptoms-types-and-diagnosis.
Nitrini, R., Caramelli, P., Herrera, E., Jr, Bahia, V. S., Caixeta, L. F., Radanovic, M., Anghinah, R., Charchat-Fichman, H., Porto, C. S., Carthery, M. T., Hartmann, A. P., Huang, N., Smid, J., Lima, E. P., Takada, L. T., & Takahashi, D. Y. (2004). Incidence of dementia in a community-dwelling Brazilian population. Alzheimer Disease and Associated Disorders, 18(4), 241–246. https://www.ncbi.nlm.nih.gov/pubmed/15592138
Papp, K. V., Amariglio, R. E., Dekhtyar, M., Roy, K., Wigman, S., Bamfo, R., Sherman, J., Sperling, R. A., & Rentz, D. M. (2014). Development of a psychometrically equivalent short form of the face-name associative memory exam for use along the early Alzheimer's disease trajectory. The Clinical Neuropsychologist, 28(5), 771–785. https://doi.org/10.1080/13854046.2014.911351
Pfeffer, R. I., Kurosaki, T. T., Harrah Jr., C. H., Chance, J. M., & Filos, S. (1982). Measurement of functional activities in older adults in the community. Journal of Gerontology, 37(3), 323–329. https://doi.org/10.1093/geronj/37.3.323
Rasmussen, J., & Langerman, H. (2019). Alzheimer's disease - why we need early diagnosis. Degenerative Neurological and Neuromuscular Disease, 9, 123–130. https://doi.org/10.2147/DNND.S228939
Reitan, R. M. (1955). The relation of the Trail Making Test to organic brain damage. Journal of Consulting Psychology, 19(5), 393–394. https://doi.org/10.1037/h0044509
Rey, A. (1964). L’examen clinique en psychologie. Presses Universitaires de France.
Roberts, J. L., Clare, L., & Woods, R. T. (2009). Subjective memory complaints and awareness of memory functioning in mild cognitive impairment: A systematic review. Dementia and Geriatric Cognitive Disorders, 28(2), 95–109. https://doi.org/10.1159/000234911
Rosen, W. G., Mohs, R. C., & Davis, K. L. (1984). A new rating scale for Alzheimer's disease. American Journal of Psychiatry, 141(11), 1356–1364. https://doi.org/10.1176/ajp.141.11.1356
Ryan, M. M., Grill, J. D., Gillen, D. L., & Alzheimer’s Disease Neuroimaging, I. (2019). Participant and study partner prediction and identification of cognitive impairment in preclinical Alzheimer's disease: Study partner vs. participant accuracy. Alzheimer's Research & Therapy, 11(1), 85. https://doi.org/10.1186/s13195-019-0539-3
Samieri, C., Proust-Lima, C., Glymour, M. M., Okereke, O. I., Amariglio, R. E., Sperling, R. A., Rentz, D. M., & Grodstein, F. (2014). Subjective cognitive concerns, episodic memory, and the APOE epsilon4 allele. Alzheimers & Dementia, 10(6), 752–759 e751. https://doi.org/10.1016/j.jalz.2014.06.012
Skinner, J., Carvalho, J. O., Potter, G. G., Thames, A., Zelinski, E., Crane, P. K., Gibbons, L. E., & Alzheimer’s Disease Neuroimaging, I. (2012). The Alzheimer's disease assessment scale-cognitive-plus (ADAS-cog-plus): An expansion of the ADAS-cog to improve responsiveness in MCI. Brain Imaging and Behavior, 6(4), 489–501. https://doi.org/10.1007/s11682-012-9166-3
Sperling, R. A., Aisen, P. S., Beckett, L. A., Bennett, D. A., Craft, S., Fagan, A. M., Iwatsubo, T., Jack Jr., C. R., Kaye, J., Montine, T. J., Park, D. C., Reiman, E. M., Rowe, C. C., Siemers, E., Stern, Y., Yaffe, K., Carrillo, M. C., Thies, B., Morrison-Bogorad, M., et al. (2011). Toward defining the preclinical stages of Alzheimer's disease: Recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers & Dementia, 7(3), 280–292. https://doi.org/10.1016/j.jalz.2011.03.003
Warren, S. L., & Moustafa, A. A. (2022). The efficacy of donepezil for the treatment of Alzheimer’s disease. In Alzheimer’s Disease: Understanding Biomarkers, Big Data, and Therapy (pp. 217–232). Academic Press. https://doi.org/10.1016/B978-0-12-821334-6.00001-6
Warren, S. L., Moustafa, A. A., Alashwal, H., & Alzheimer’s Disease Neuroimaging, I. (2021). Harnessing forgetfulness: Can episodic-memory tests predict early Alzheimer's disease? Experimental Brain Research. https://doi.org/10.1007/s00221-021-06182-w
Weimer, D. L., & Sager, M. A. (2009). Early identification and treatment of Alzheimer's disease: social and fiscal outcomes. Alzheimer's & dementia : The Journal of the Alzheimer's Association, 5(3), 215–226. https://doi.org/10.1016/j.jalz.2009.01.028
Zainal, N. H., Silva, E., Lim, L. L., & Kandiah, N. (2016). Psychometric properties of Alzheimer's disease assessment scale-cognitive subscale for mild cognitive impairment and mild Alzheimer's disease patients in an asian context. Annals of the Academy of Medicine Singapore, 45(7), 273–283. https://www.ncbi.nlm.nih.gov/pubmed/27523508
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. This study would like to acknowledge funding received as part of the Australian Governments Research Training Program Scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Our study gained ethics approval from Western Sydney Universities Human Research Ethics Committee in April 2021.
Consent to participate
Not applicable as this project uses secondary data and has no contact with participants. All consent is acquired by ADNI.
Consent for publication
Not applicable.
Conflict of interest
The authors of this study declare no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Warren, S.L., Reid, E., Whitfield, P. et al. Cognitive and behavioral abnormalities in individuals with Alzheimer’s disease, mild cognitive impairment, and subjective memory complaints. Curr Psychol 43, 800–810 (2024). https://doi.org/10.1007/s12144-023-04281-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12144-023-04281-1