Abstract
Fms-like tyrosine kinase 3 (FLT3) is the most frequently mutated gene in acute myeloid leukemia (AML). Modern targeting of FLT3 with inhibitors has improved clinical outcomes and FLT3 inhibitors have been incorporated into the treatment of AML in all phases of the disease, including the upfront, relapsed/refractory and maintenance settings. This review will discuss the current understanding of FLT3 biology, the clinical use of FLT3 inhibitors, resistance mechanisms and emerging combination treatment strategies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute Myeloid Leukemia (AML) is an aggressive hematologic malignancy with a heterogenous genetic landscape [1]. Fms-like tyrosine kinase 3 (FLT3) is the most frequently mutated gene in AML, with approximately 30% of newly diagnosed AML patients harboring an FLT3 mutation, and FLT3 mutations are associated with aggressive disease biology [2]. Since FLT3 mutations in AML were first described over 25 years ago, our understanding of the biology, clonal dynamics, targetability, and prognostic significance of FLT3 has evolved. Worldwide, there are now 3 approved FLT3 inhibitors, which have been studied as single agents, in combination with cytotoxic chemotherapy, and more recently in combination with other targeted agents. Paralleling progress in FLT3 targeting, the prognosis for patients with FLT3-mutated AML has improved in tandem, and as of 2022, high FLT3 allele burden is no longer considered high-risk in AML [3]. Despite these advances, heterogenous resistance mechanisms to FLT3 inhibitors represent an ongoing challenge and multiple questions regarding FLT3 in AML remain. In this review, we highlight several recent advances in the understanding of FLT3 biology, clonal selection, therapeutic targeting, and resistance.
FLT3 mutational landscape: where are we today?
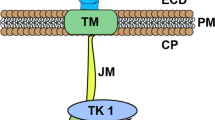
The FLT3 gene encodes FLT3, a membrane-bound protein and member of the receptor tyrosine kinase family [4]. Upon binding to the FLT3 ligand, FLT3 undergoes homodimerization and activation, leading to increased cellular signaling via multiple pathways, including RAS/MAPK, JAK/STAT, and PI3K/AKT [4]. Together, this increased signaling promotes cellular proliferation, inhibition of apoptosis, and inhibition of differentiation [5]. FLT3 is widely expressed on normal hematopoietic progenitor cells and is overexpressed on the majority of AML blasts [5]. In normal hematopoiesis, FLT3 activation is tightly regulated via phosphorylation of the protein’s juxtamembrane domain (JMD). When mutations develop in the JMD or adjacent tyrosine kinase domain (TKD), this tight regulation is disrupted, causing constitutive activation, ligand-independent signaling, and clonal proliferation [6].
FLT3-ITD and TKD
The FLT3 internal tandem duplication (ITD) is the most clinically significant FLT3 mutation, and patients with FLT3-ITD mutated AML have increased rates of disease relapse, and inferior overall survival [7]. Present in approximately 25% of newly diagnosed patients, FLT3-ITD mutations occur within the JMD and are variable in size, ranging from 3 to > 1,000 nucleotides [6]. The less common TKD mutations, present in approximately 5% of patients, are missense point mutations in the activation loop of FLT3, most commonly at D835. Like FLT3-ITD mutations, FLT3-TKD stabilizes the active kinase formation and result in constitutive receptor activation [5]. Unlike FLT3-ITD mutations, FLT3-TKD have an unclear impact on patient prognosis and are not currently included in consensus risk assessments [3, 8].
Non-canonical FLT3 mutations
In recent years, an increasing number of non-ITD and D835 mutations in FLT3 have been described [9,10,11]. The landscape and frequency of these non-canonical (NC) FLT3 mutations are challenging to assess, as they are not detected using standard-of-care PCR-based assays. Estimated prevalence from next-generation sequencing (NGS)-based studies is on the order of 5–8%. In a large whole genome sequencing study of 799 pediatric patients with AML, 7.6% of patients harbored NC FLT3 mutations, 9 of which were JMD mutations [12]. Similarly, in a recent study using high-throughput genomic sequencing of patients treated on the RATIFY trial, 26/275 (5.5%) of patients harbored NC FLT3 mutations concurrent with either FLT3-ITD or TKD mutations [13].
To date, most NC FLT3 mutations have been described in either the JMD or TKD [9, 10, 12], although NC FLT3 mutations have been described in the extracellular domain as well [14]. Of the JMD and KD mutations described in patients with functional correlates, most demonstrate increased signaling activation or autophosphorylation [10, 15,16,17]. In a recent study of NC JMD FLT3 deletion mutations, the Catalogue of Somatic Mutations in Cancer (COSMIC) database was queried for deletions observed in patients spanning FLT3 residues 572–575 [10]. Mutations at four residues were identified (Y572, E573, S574, Q575), and when these deletions were introduced into cell lines, all demonstrated FLT3 autophosphorylation and increased downstream signaling [10]. This suggests that at least some NC FLT3 mutations are driver mutations, actively promoting leukemic proliferation and survival. Consistent with this, another recent study of JMD deletion mutations reported sensitivity to FLT3 inhibition [18]. As sequencing technologies improve and broad NGS panels are increasingly used, the full landscape of NC FLT3 mutations observed in AML patients and their prognostic significance will become more apparent.
FLT3 clonal architecture, selection, and evolution
AML is characterized by a complex polyclonal architecture and evolution. While 30% of patients with newly diagnosed AML harbor a canonical FLT3 mutation, many patients will additionally develop a de novo FLT3 mutation after treatment. Pre-existing FLT3-mutated clones can also expand under therapeutic pressure.
Historically, fit patients with FLT3-mutated AML were treated with cytotoxic chemotherapy alone. Although patients with FLT3-ITD mutated disease frequently responded to chemotherapy at rates similar to patients with FLT3-WT disease, responses were short-lived, with rapid outgrowth of the FLT3 mutated clone [19]. Along with cytotoxic chemotherapy, selection for FLT3-mutated clones is a common resistance mechanism to venetoclax-based therapies which are the current standard-of-care for older or unfit patients with newly-diagnosed AML [20, 21]. In an analysis of 81 older or unfit patients treated with venetoclax doublets, expansion or acquisition of FLT3-mutant clones was the second-most common adaptive resistance mechanisms, second only to biallelic TP53 mutations [20].
FLT3 clonal selection also drives resistance to other targeted therapies [22,23,24]. In a study of 174 patients treated with the isocitrate dehydrogenase 1 (IDH1) inhibitor ivosidenib, pre-treatment presence of a FLT3 mutation was associated with a significantly lower likelihood of response, and both FLT3-ITD and TKD mutations commonly emerged on relapse [22]. In a longitudinal analysis of 60 patients treated with IDH1 or 2 inhibitors, no patients with concurrent FLT3 mutations responded to IDH inhibition, although this was not statistically significant due to the small sample size [23].
Targeting FLT3 today
Despite the therapeutic resistance of FLT3-mutated AML, AML blasts harboring FLT3-ITD and TKD mutations are sensitive to small molecule inhibitors; as such, these targeted FLT3 inhibitors are an important part of the therapeutic approach to treating patients with FLT3-mutated AML.
Newly diagnosed disease
Fit patients
For newly diagnosed, fit patients with FLT3-mutated AML, the standard of care is defined by the phase III RATIFY trial, in which patients with FLT3-ITD or -TKD mutated AML were randomized to standard induction and consolidation chemotherapy ± midostaurin [25]. The addition of midostaurin resulted in superior overall survival (OS) compared to chemotherapy alone (median OS 74.7 vs 25.6 months, p = 0.0009) [25]. Based on these results, in 2019, midostaurin became the first approved agent for FLT3-mutated AML. Although RATIFY only included patients through age 60, the recent AMLSG 16–10 trial compared midostaurin plus chemotherapy against historical outcomes in fit adults aged 61–70 and found midostaurin-containing therapy to be superior as well [26]. Midostaurin has also been evaluated in combination with liposomal daunorubicin and cytarabine (CPX-351) in newly diagnosed adults with FLT3-mutated AML up to age 75; preliminary results of 23 patients indicate this regimen is safe and efficacious, with a complete response (CR) rate of 82% [27].
More recently, the phase III QuANTUM-First trial randomized 539 patients ages 18–75 with FLT3-ITD mutated AML to induction and consolidation chemotherapy plus quizartinib vs placebo; median overall survival was superior in the quizartinib arm (31.9 vs 15.1 months, p = 0.032) [28]. Unlike RATIFY, QuANTUM-FIRST included an assessment of the depth of response measured by measurable residual disease (MRD). In this assessment, the proportion of patients with composite complete remission with FLT3-ITD MRD less than 10−4 was similar across groups (25% for quizartinib versus 21% for placebo) but the proportion of patients with composite complete remission with undetectable MRD was higher for quizartinib (14% versus 7%), suggesting increased depth of response. Based on these results, quizartinib recently received approval in Japan for newly diagnosed FLT3-ITD mutant AML and is currently undergoing similar regulatory consideration by the U.S. Food and Drug Administration (FDA). As quizartinib and midostaurin-containing regimens have not been directly compared, it is unclear how quizartinib will factor into current treatment paradigms for FLT3-ITD + patients, though midostaurin should remain the standard of care for patients with FLT3 TKD mutations. Additional phase III frontline trials of cytotoxic chemotherapy with crenolanib (NCT03258931) and gilteritinib (NCT03936209; HOVON 156 AML/AMLSG 28–18) vs midostaurin are ongoing. Importantly, similar to QuANTUM-First, these ongoing trials incorporate MRD monitoring, providing key information as to how well these regimens suppress the FLT3-mutant allele and whether depth of response correlates with outcome.
Unfit patients
For older and/or unfit patients, venetoclax plus hypomethylating agents (HMA) are the standard of care. While the first-generation FLT3 inhibitor sorafenib plus azacitidine is an approved regimen per the National Comprehensive Cancer Network (NCCN) guidelines [28], it does not appear to outperform approved venetoclax-based regimens in this population. In a phase II trial of sorafenib plus azacitidine in newly diagnosed older adults with AML, the overall response rate (ORR) was 78% with an OS was 8.3 months [29]. By contrast, in older/unfit patients with newly diagnosed, FLT3-mutated AML treated with HMA plus venetoclax, the ORR was 70% with a median OS of 15 months in subgroup analysis [30].
Relapsed or refractory disease
For both fit and unfit patients, the current standard of care for relapsed or refractory (R/R) FLT3-mutated AML in the U.S. and Europe is single-agent gilteritinib; in Japan, single-agent quizartinib is also an option. The phase III ADMIRAL trial compared gilteritinib vs salvage chemotherapy; gilteritinib demonstrated a greater CR rate (34 vs 15%, p = 0.0001) and superior median OS (9.3 vs 5.6 months, p < 0.0001) [30]. Similarly, in the phase III QUANTUM-R trial, single-agent quizartinib was associated with superior OS compared to salvage chemotherapy (6.2 vs 4.7 months, p = 0.02) [31]. Quizartinib is now approved in Japan, although concerns about study design led to both FDA and EMA rejection [31].
Maintenance
Post-transplant
Consolidation with allogeneic hematopoietic cell transplantation (HCT) is currently recommended in eligible patients [32]. Following HCT, maintenance therapy with various FLT3 inhibitors has been studied. In the phase II RADIUS trial, FLT3-ITD + patients in the first CR were randomized to midostaurin vs placebo maintenance for 12 months; there was no difference in relapse-free survival [33]. In the phase II SORMAIN trial, patients with FLT3-ITD-mutated AML in remission after HCT were randomized to sorafenib vs placebo. Sorafenib maintenance resulted in superior 2-year estimated probability of survival (90.5% vs 66.2%, p = 0.007), although the study was terminated early after not reaching the targeted accrual [34]. In a subsequent phase III study, 202 patients were randomized to sorafenib maintenance vs standard care post-HCT, and sorafenib resulted in fewer relapses at 1-year post-HCT (7% vs 24.5%, p = 0.001) [35]. Of note, in both trials, only 21% and 24% of patients had received an FLT3 inhibitor prior to HCT and both included patients in first and second CR. It is unknown whether the benefit of sorafenib applies to patients transplanted in the first CR after standard-of-care frontline therapy [34, 35]. More recently, the phase III MORPHO trial randomized patients to gilteritinib vs placebo following HCT for patients transplanted in the first CR, and indicated no overall difference in RFS for patients treated with gilteritinib [36]. However, in the 50.6% of patients with MRD positivity (detected using PCR NGS at sensitivity of 10–6 or greater), the effect of gilteritinib on RFS was pronounced (HR = 0.515, 95% CI: 0.316, 0.838, p = 0.0065) compared to patients without detectable MRD (HR = 1.213, 95% CI: 0.616, 2.387, p = 0.575) [37]. These data indicate a benefit of gilteritinib maintenance for patients with MRD transplanted in CR1.
Post-chemotherapy
For patients who are either ineligible for or do not proceed directly to transplant, maintenance FLT3 inhibitor therapy can be considered. While RATIFY did allow for midostaurin monotherapy after consolidation and is approved as such in Europe, midostaurin maintenance was not efficacious in post hoc analysis and approval was not extended by the FDA [38]. Similarly, in the SORAML trial of sorafenib vs placebo plus chemotherapy in newly diagnosed FLT3 mutated AML, patients randomized to the sorafenib arm received post-chemotherapy sorafenib maintenance. While the RFS curves did separate throughout the maintenance phase, the trial was not powered to detect whether maintenance sorafenib contributed to improved outcomes [39].
Multiple studies of maintenance FLT3 inhibitors are ongoing. In the recently completed QuANTUM-First trial, patients randomized to the quizartinib arm received both post-chemotherapy and/or post-HCT quizartinib maintenance; report of these outcomes is anticipated in future publications [28]. Similarly, ongoing frontline trials of crenolanib vs midostaurin and gilteritinib in combination with chemotherapy will include crenolanib and gilteritinib maintenance, respectively (NCT03258931, NCT03936209, HOVON 156 AML/AMLSG 28-18).
Resistance to FLT3 inhibitors
Although FLT3 inhibitors have significantly improved the survival of patients with FLT3-mutated AML, resistance remains an ongoing challenge. Resistance mechanisms to FLT3 inhibitors are heterogenous and comprise both cell intrinsic and extrinsic processes (Fig. 1) as well as complex clonal selection and evolution (Fig. 2).
Described mechanisms of cell-intrinsic and extrinsic FLT3 inhibitor resistance mechanisms, including on-target secondary mutations within FLT3, off-target mutations in parallel and/or downstream pathways, upregulation of antiapoptotic proteins, and factors upregulated in the bone marrow microenvironment
Fish plot highlighting complex clonal selection with FLT3 inhibitor resistance in an imagined patient. The green, blue, and purple portions of the plot represent clones with FLT3-ITD + IDH2, FLT3-ITD + FLT3 F691L, and NRAS genotypes, respectively. Over time and with FLT3 inhibitor resistance, all three of these clones expand
Genetic resistance: on-target secondary mutations
One key resistance mechanism to FLT3 inhibition is the development of on-target mutations within the FLT3 gene. In many cases, these secondary mutations are not detected prior to FLT3 inhibitor treatment and are instead presumed to evolve de novo, or at least below the detection limit of current sequencing technologies [40, 41]. In type II FLT3 inhibitor therapy, on target resistance is frequently medicated by FLT3-TKD mutations, most commonly at D835 although mutations in kinase domain residues I836, D839, and Y842 have been described as well [41]. Less commonly, on target resistance can occur via development of the “gatekeeper” mutation F691L, which confers resistance to both type I and type II FLT3 inhibitors [42]. Practically, the development of F691L mutations likely represents a minority of gilteritinib-resistant leukemias. In studies profiling mutations in patients who relapsed following gilteritinib monotherapy on the ADMIRAL trial, only 5/4 (12.5%) of evaluable patients developed F691L mutations at relapse [40, 43].
As sequencing technologies improve, an increasing number of NC FLT3 mutations are described in on-target resistance. Multiple in vitro studies have now demonstrated variable FLT3 inhibitor resistance mediated by NC FLT3 mutations [44,45,46]. The resistance caused by these NC mutations may be unique to individual inhibitors. For example, one in vitro study of co-mutated FLT3 N701K plus FLT3-ITD cells demonstrated resistance to type I inhibitor to gilteritinib and sensitivity to type II inhibitor quizartinib, a pattern opposite to that seen in FLT3-TKD on-target resistance mutations [44]. Data describing whether and to what extent NC FLT3 mutations convey resistance in patients is more limited. In the same analysis of 40 patients with FLT3-mutated AML treated with gilteritinib monotherapy on the ADMIRAL trial, 2 patients developed de novo NC JMD mutations at E598D [40]. Similarly, the development of JMD mutation at Q575 has been described in a patient resistant to cytotoxic chemotherapy plus gilteritinib [10], and the development of KD mutation at N676 has been described in several patients with resistance to midostaurin [44, 47]. While these case studies are intriguing, full sequencing of the FLT3 gene is needed in correlative analyses of future trials to fully understand the impact of the NC FLT3 mutations on disease resistance.
Genetic resistance: off-target mutations
A second mechanism of cell-intrinsic FLT3 inhibitor resistance is the emergence or expansion of clones with mutations outside of FLT3. These off-target mutations are heterogenous, including genes involved in Ras/MAPK signaling (NRAS, KRAS, PTPN11) as well as ASXL1, TP53, TET2, IDH1/2 [14, 40, 43, 48]. Development of BCR-ABL1 fusion genes with gilteritinib resistance has been described as well [43, 49]. In a study of paired pre-treatment and relapse samples from patients treated on the RATIFY trial, at relapse, almost half of patients (46%) became FLT3-negative but acquired mutations in other signaling pathways, indicating a strong selective advantage for these clones [48].
Of these heterogenous off-target resistance mechanisms, mutations activating the downstream Ras/MAPK pathways are particularly common, especially in resistance to type I inhibitors gilteritinib or crenolanib [14, 40, 43]. In patients who relapsed following gilteritinib monotherapy, new mutations in Ras/MAPK pathway genes occurred in 45% of patients, nearly triple the observed number of new on-target mutations [40]. In a similar study of 41 patients treated with single-agent gilteritinib, treatment-emergent Ras/MAPK mutations were identified in 37%; of these, over half had multiple RAS mutations [43]. Finally, in a recent analysis of 12 patients treated with the combination of gilteritinib and venetoclax, 4/12 patients developed dominate N/KRAS mutations at relapse; notably, no patients developed on-target mutations [50]. While best described in resistance to type II inhibitors, Ras/MAPK pathway mutations may be an important resistance mechanism to type II inhibitors as well. In an analysis of 8 patients with relapsed disease after quizartinib monotherapy, 2/8 patients demonstrated outgrowth of N/KRAS mutations, although on-target mutations were the dominate resistance mechanism in this cohort, present in 7/8 patients [41].
Recently, single-cell sequencing studies have shed light on the clonal architecture of these off-target mutations. While off-target mutations can occur in FLT3-mutant blasts, they frequently occur in cells without co-occurring FLT3 mutations and are often present prior to FLT3 inhibitor therapy [14, 41, 43, 50]. In a study of 12 patients treated with gilteritinib and venetoclax and analyzed by single-cell sequencing, all 4 patients with dominate N/KRAS mutations at relapse had mutations detected prior to therapy [50]. This suggests that resistance via off-target mutations frequently arises via selection of pre-existing subclones which harbor a survival advantage with FLT3 inhibition, a mechanism distinct from that seen in the development of de novo on-target secondary mutations.
Non-genetic resistance
Non-genetic mechanisms are also potential key mediators of FLT3 inhibitor resistance. In a study of 40 patients relapsed after gilteritinib with paired pre- and post-treatment samples, 13/40 patients had no new detectable mutations detected at relapse, suggesting that non-genetic mechanisms may be driving resistance in a significant proportion of patients [40].
Multiple components of the bone marrow microenvironment may facilitate FLT3 inhibitor resistance. The bone marrow microenviroment harbors FLT3 ligand, and levels may be particularly increased during induction or consolidation chemotherapy [51, 52]. Although FLT3 inhibitors effectively target FLT3 mutations, they are less effective at disrupting wild-type FLT3 signaling, and high FLT3 ligand concentrations are protective against FLT3 inhibitors [51, 53]. Bone marrow stromal cells also have high levels of CYP34A expression, leading to increased FLT3 inhibitor metabolism [54]. Paralleling the observation that off-target mutations in downstream pathways are key in cell-intrinsic resistance, bone marrow stromal cells can also directly upregulate Ras/MAPK signaling via FGF2, activate STAT5 signaling, and inhibit blasts apoptosis via activation of the ATM/mTOR pathway [55,56,57]. Together, these alterations in the bone marrow microenviroment may be protective of residual leukemia and contribute to early disease resistance relative to genetic resistance mechanisms [58].
Finally, emerging evidence suggests leukemic differentiation state may also contribute to FLT3 inhibitor resistance. In a large study of ex vivo drug sensitivity testing of samples from 279 patients with newly diagnosed AML, monocytic cell state was associated with increased resistance to sorafenib, independent of other genetic alterations [59]. In a multi-omic single-cell sequencing analysis of 12 patients with resistance to gilteritinib and venetoclax, multiple subclones of heterogenous genotypes demonstrated increased expression of monocytic markers with therapy resistance [50]. Whether monocytic cell state is truly an independent resistance mechanism to FLT3 inhibitors or a bystander to other cell-intrinsic or extrinsic resistance mechanisms is unknown.
Targeting FLT3 today: FLT3 combination therapies
Identifying strategies to overcome resistance and extend disease response is an area of active research. Given the broad array of resistance mechanisms to FLT3 inhibition, as well as the polyclonal nature of FLT3-mutated AML, one of the most promising current strategies is to combine FLT3 inhibitors with other antileukemic agents (Tables 1, 2).
Hypomethylating agents
Aside from conventional cytotoxic chemotherapy, one of the earliest FLT3 inhibitor combinations was with hypomethylating agents (HMA) azacitidine or decitabine. In a phase II trial of sorafenib plus azacitidine in newly diagnosed older adults with FLT3-ITD mutated AML, the ORR rate was 78% with an OS of 8.3 months [29]. In a similar trial of patients with R/R FLT3-ITD mutated disease, sorafenib plus azacitidine demonstrated an ORR 46% [60].
It is unclear whether combinations with the more potent second-generation FLT3i are more promising. In a phase I/II trial of patients with FLT3-ITD mutated AML treated with quizartinib plus azacitidine, the CR rate and median OS were promising at 87% and 19.2 months and 64% and 12.8 months in the frontline and R/R settings, respectively [61]. By contrast, in the recent randomized phase III LACEWING trial of older/unfit adults with newly diagnosed FLT3-mutated AML, there was no difference in OS for gilteritinib plus azacitidine vs azacitidine alone (9.8 vs 8.9 months), although these results were clouded by the fact that many patients on the azacitidine arm terminated the study early and received subsequent FLT3 inhibitor therapy [62]. Finally, the multi-kinase inhibitor ponatinib in combination with azacitidine demonstrated a durable ORR of 52% in unfit newly diagnosed patients with FLT3-mutated AML [63, 64].
Venetoclax
Venetoclax, an oral inhibitor of the anti-apoptotic protein BCL-2, is particularly promising in combination with FLT3 inhibitors. Upregulation of antiapoptotic proteins is a mechanism of FLT3 inhibitor resistance [65], and the emergence or outgrowth of FLT3 mutations is associated with venetoclax resistance [20, 66]. In vitro studies have demonstrated synthetic lethality with venetoclax combined with multiple FLT3 inhibitors [67,68,69]. In a phase Ib trial of 61 patients with R/R FLT-mutated AML treated with gilteritinib plus venetoclax, the modified CR rate was 75% with a median OS of 10 months [70]. This is substantially higher than the 54% modified CR rate observed in the ADMIRAL trial of single-agent gilteritinib using identical response criteria [71]. A trial of quizartinib plus venetoclax is ongoing (NCT03735875).
Triplet combinations
Given the promising outcomes of both HMA and venetoclax doublet combinations, as well as the efficacy of HMA and venetoclax combinatory therapy, it is not surprising that triplet combinations have received particular attention. In a retrospective analysis of 87 newly diagnosed patients with FLT3-mutated AML treated with either triplet (HMA + Venetoclax + FLT3i) or doublet (HMA + FLT3i) therapy, patients receiving triplet therapy had significantly longer OS without increased cytopenias [72]. Intriguingly, while patients receiving doublet therapy still demonstrated a benefit from allogeneic transplant, survival for those receiving triplet therapy was similar irrespective of transplant status, suggesting a benefit of triplet therapy in the upfront setting may lay in a potential to forgo transplant.
In phase I/II trial of 28 patients with R/R FLT3-ITD mutated AML with prior FLT3 inhibitor treatment, the triplet quizartinib, decitabine, and venetoclax showed promising interim results, with a composite CR (cCR) rate of 82% [73]. Furthermore, in a smaller cohort of 7 newly-diagnosed patients treated with the same regimen, all patients achieved a cCR [73]. A trial of an all-oral version of the same regimen, using oral decitabine plus ceduazurdine (ASTX727), is ongoing [74]. In a phase I/II study of gilteritinib, azacitidine, and venetoclax in a similar patient population, 100% of newly diagnosed patients and 74% of patients with R/R disease achieved composite CR [75]. While these combinations have encouraging outcomes, both triplets had high incidence of myelosuppression, with a median time of both neutrophil and platelet recovery of over a month [73, 75]. Further data is needed to understand how to maximize the safety of these regimens, particularly in older and/or frailer patients, as well as how these regimens compare to HMA plus venetoclax or FLT3 inhibitor plus venetoclax doublet therapies, especially in R/R patients where response rates to FLT3i/venetoclax doublets and triplets are similar. [76, 77]
Will these combinatory therapies be able to fully overcome established resistance mechanisms? Thus far, it appears unlikely. For example, in patients who relapsed after single-agent gilteritinib in the ADMIRAL trial, the most common mutations associated with treatment resistance were in genes associated with the Ras/MAPK pathway [40]. Similarly, mutations in NRAS and KRAS appear to be the dominant genetic resistance mechanism to gilteritinib plus venetoclax [50], and patients with Ras/MAPK mutations had the lowest response rate to the triplet quizartinib/decitabine/venetoclax [73]. Although inhibitors to Ras/MAPK signaling, including BRAF V600E and KRAS G12C inhibitors, are approved for other cancers, the Ras/MAPK pathway mutations observed in resistance to FLT3 inhibitors are not sensitive to these agents [78, 79]. New targeted therapies, either in combination with or sequential to FLT3 inhibitors will be needed to overcome these dominant resistance mechanisms.
Novel FLT3 targeting approaches
In addition to established combination partners like HMA or venetoclax, FLT3 inhibitors have also been combined with novel small molecules, including the spleen tyrosine kinase inhibitor lanraplenib, lysine-specific demethylase-1 inhibitor iadademstat, and approved IDH1/2 inhibitors ivosidenib and enasidenib [80, 81]. Studies involving multikinase inhibitors, such as the FLT3/SYK/JAK/KIT kinase inhibitor tuspetinib [82], the FLT3/BTK inhibitor luxeptinib [83], and the FLT3/FGFR inhibitor MAX-40279 [84] are ongoing as well. In addition to these agents, novel covalent FLT3 inhibitors and FLT3-targeted biologic agents are in active pre-clinical and clinical development as well [85, 86] (Table 3).
Prognosis of FLT3-mutated AML in the modern era
Historically, the presence of a high FLT3-ITD allelic ratio (AR), defined as a FLT3-ITD to FLT3-WT ratio of > = 0.5, was associated with highest disease risk, FLT3-ITD AR < 0.5 with a co-occurring NPM1 mutation was associated with favorable disease risks, and FLT3-ITD AR < 0.5 AR without a co-occurring NPM1 mutation was associated with intermediate disease risk. This risk stratification was described in the 2017 European LeukemiaNet (ELN) schema [8].
When these 2017 guidelines were developed, no FLT3 inhibitors were approved. Today, the paradigm has shifted considerably. Given this new treatment landscape, the prognostic significance of FLT3 mutations has evolved. In an analysis of 513 patients with newly diagnosed AML, 96 patients with FLT3-ITD mutations experienced survival comparable with other patients with intermediate-risk features, and neither co-mutations in NPM1 nor FLT3-ITD AR influenced outcomes [87]. Notably, in this cohort, only 41% of patients received an FLT3 inhibitor, so it is possible outcomes would be even better if all patients received current standard-of-care FLT3 inhibition [87]. In a retrospective analysis of the RATIFY trial, midostaurin plus chemotherapy significantly improved overall survival for all 2017 ELN risk groups, with similar OS probabilities for the midostaurin arm in both intermediate- and adverse-risk disease [88]. Based on these and other studies, as well as challenges in standardizing AR measurements across laboratories, FLT3-ITD AR and co-mutational status is no longer taken into consideration in the most recent 2022 ELN risk stratification schema [3]. Patients who are FLT3-ITD positive are classified as intermediate risk, irrespective of allelic ratio or concurrent mutations in NPM1 [3].
This evolving risk stratification and treatment landscape raises many questions. Now that all FLT3-ITD mutated AML is classified as intermediate risk, should all eligible patients with FLT3-ITD mutated AML receive an allogeneic hematopoietic stem cell transplant (HCT) in the first CR, a strategy most beneficial in patients at the highest risk of relapse, or can transplant be reserved for first relapse or MRD positive disease? In addition to flow cytometry-based MRD measurements, there is emerging evidence that pre-transplant FLT3 DNA-NGS-based MRD can predict post-transplant outcomes [28, 37]. How will new therapies, such as quizartinib in newly diagnosed fit patients and FLT3 inhibitor/venetoclax-based combinations alter the current treatment paradigm and associated prognosis? Fig. 3 outlines possible directions for the treatment of FLT3-mutated AML in the future.
Schematic describing the current standard of care (top) and potential future treatment paradigm (bottom) of patients with FLT3-mutated AML. SOC standard of care, OS overall survival, RFS relapse-free survival, MRD measurable residual disease, HCT hematopoietic cell transplantation, ELN European LeukemiaNet
Conclusion
Increasingly, treating FLT3-mutated AML represents the forefront of personalized medicine and targeted therapy in AML. As targeted FLT3 inhibitors and combinatory therapies become increasingly adopted, future risk stratification and treatment schema will evolve in tandem.
References
Dohner H, Weisdorf DJ, Bloomfield CD. Acute myeloid leukemia. N Engl J Med. 2015;373:1136–52.
Papaemmanuil E, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374:2209–21.
Dohner H, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140:1345–77.
Berenstein R. Class III receptor tyrosine kinases in acute leukemia - biological functions and modern laboratory analysis. Biomark Insights. 2015;10:1–14.
Gilliland DG, Griffin JD. The roles of FLT3 in hematopoiesis and leukemia. Blood. 2002;100:1532–42.
Schnittger S, et al. Diversity of the juxtamembrane and TKD1 mutations (exons 13–15) in the FLT3 gene with regards to mutant load, sequence, length, localization, and correlation with biological data. Genes Chromosom Cancer. 2012;51:910–24.
Kottaridis PD, et al. The presence of a FLT3 internal tandem duplication in patients with acute myeloid leukemia (AML) adds important prognostic information to cytogenetic risk group and response to the first cycle of chemotherapy: analysis of 854 patients from the United Kingdom Medical Research Council AML 10 and 12 trials. Blood. 2001;98:1752–9.
Dohner H, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129:424–47.
Kennedy VE, Smith CC. FLT3 mutations in acute myeloid leukemia: key concepts and emerging controversies. Front Oncol. 2020;10: 612880.
Young DJ, et al. Deletions in FLT-3 juxtamembrane domain define a new class of pathogenic mutations: case report and systematic analysis. Blood Adv. 2021;5:2285–93.
Knight TE, Edwards H, Meshinchi S, Taub JW, Ge Y. FLipping" the story: FLT3-mutated acute myeloid leukemia and the evolving role of FLT3 Inhibitors. Cancers (Basel). 2022;14:3398.
Tarlock K, et al. Discovery and functional validation of novel pediatric specific FLT3 activating mutations in acute myeloid leukemia: results from the COG/NCI target initiative. Blood. 2015;126:87.
Jahn N, et al. Genomic landscape of patients with FLT3-mutated acute myeloid leukemia (AML) treated within the CALGB 10603/RATIFY trial. Leukemia. 2022;36:2218–27.
Zhang H, et al. Clinical resistance to crenolanib in acute myeloid leukemia due to diverse molecular mechanisms. Nat Commun. 2019;10:244.
Chatain N, et al. Rare FLT3 deletion mutants may provide additional treatment options to patients with AML: an approach to individualized medicine. Leukemia. 2015;29:2434–8.
Nguyen B, et al. FLT3 activating mutations display differential sensitivity to multiple tyrosine kinase inhibitors. Oncotarget. 2017;8:10931–44.
Frohling S, et al. Identification of driver and passenger mutations of FLT3 by high-throughput DNA sequence analysis and functional assessment of candidate alleles. Cancer Cell. 2007;12:501–13.
Ge SS, et al. Mutation spectrum of FLT3 and significance of non-canonical FLT3 mutations in haematological malignancy. Br J Haematol. 2023;202(3):539–49.
Levis M. FLT3/ITD AML and the law of unintended consequences. Blood. 2011;117:6987–90.
DiNardo CD, et al. Molecular patterns of response and treatment failure after frontline venetoclax combinations in older patients with AML. Blood. 2020;135:791–803.
Chyla BJ, et al. Response to venetoclax in combination with low intensity therapy (LDAC or HMA) in untreated patients with acute myeloid leukemia patients with IDH, FLT3 and other mutations and correlations with BCL2 family expression. Blood. 2019;134:546–546.
Choe S, et al. Molecular mechanisms mediating relapse following ivosidenib monotherapy in IDH1-mutant relapsed or refractory AML. Blood Adv. 2020;4:1894–905.
Wang F, et al. Leukemia stemness and co-occurring mutations drive resistance to IDH inhibitors in acute myeloid leukemia. Nat Commun. 2021;12:2607.
Quek L, et al. Clonal heterogeneity of acute myeloid leukemia treated with the IDH2 inhibitor enasidenib. Nat Med. 2018;24:1167–77.
Stone RM, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377:454–64.
Dohner H, et al. Midostaurin plus intensive chemotherapy for younger and older patients with AML and FLT3 internal tandem duplications. Blood Adv. 2022;6:5345–55.
McCloskey J, et al. V-FAST master trial: subgroup analysis of outcomes with CPX-351 plus midostaurin in adults with newly diagnosed acute myeloid leukemia by FLT3 mutation type. Blood. 2022;140:3312–4.
Erba HP, et al. Quizartinib plus chemotherapy in newly diagnosed patients with FLT3-internal-tandem-duplication-positive acute myeloid leukaemia (QuANTUM-First): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2023;401:1571–83.
Ohanian M, et al. Sorafenib combined with 5-azacytidine in older patients with untreated FLT3-ITD mutated acute myeloid leukemia. Am J Hematol. 2018;93:1136–41.
Ghorab A, et al. Venetoclax plus hypomethylating agent (HMA) based induction for treatment-naive older/unfit patients with FLT3-mutated acute myeloid leukemia (AML). Blood. 2022;140:9019–20.
Cortes JE, et al. Quizartinib versus salvage chemotherapy in relapsed or refractory FLT3-ITD acute myeloid leukaemia (QuANTUM-R): a multicentre, randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2019;20:984–97.
Bazarbachi A, et al. Clinical practice recommendation on hematopoietic stem cell transplantation for acute myeloid leukemia patients with FLT3-internal tandem duplication: a position statement from the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Haematologica. 2020;105:1507–16.
Maziarz RT, et al. Midostaurin after allogeneic stem cell transplant in patients with FLT3-internal tandem duplication-positive acute myeloid leukemia. Bone Marrow Transplant. 2021;56:1180–9.
Burchert A, et al. Sorafenib maintenance after allogeneic hematopoietic stem cell transplantation for acute myeloid leukemia with FLT3-internal tandem duplication mutation (SORMAIN). J Clin Oncol. 2020;38:2993–3002.
Xuan L, et al. Sorafenib maintenance in patients with FLT3-ITD acute myeloid leukaemia undergoing allogeneic haematopoietic stem-cell transplantation: an open-label, multicentre, randomised phase 3 trial. Lancet Oncol. 2020;21:1201–12.
Levis, M. BMT-CTN 1506 (MORPHO): A Randomized Trial of the FLT3 Inhibitor Gilteritinib as Post-Transplant Maintenance for FLT3-ITD AML. European Hematology Society Annual Meeting, 2023.
Dillon LW, et al. DNA sequencing to detect residual disease in adults with acute myeloid leukemia prior to hematopoietic cell transplant. JAMA. 2023;329:745–55.
Larson RA, et al. An analysis of maintenance therapy and post-midostaurin outcomes in the international prospective randomized, placebo-controlled, double-blind trial (CALGB 10603/RATIFY [Alliance]) for newly diagnosed acute myeloid leukemia (AML) patients with FLT3 mutations. Blood. 2017;130:145–145.
Rollig C, et al. Addition of sorafenib versus placebo to standard therapy in patients aged 60 years or younger with newly diagnosed acute myeloid leukaemia (SORAML): a multicentre, phase 2, randomised controlled trial. Lancet Oncol. 2015;16:1691–9.
Smith CC, et al. Molecular profile of FLT3-mutated relapsed/refractory patients with AML in the phase 3 ADMIRAL study of gilteritinib. Blood Adv. 2022;6:2144–55.
Peretz CAC, et al. Single-cell DNA sequencing reveals complex mechanisms of resistance to quizartinib. Blood Adv. 2021;5:1437–41.
Smith CC, et al. Characterizing and overriding the structural mechanism of the quizartinib-resistant FLT3 “gatekeeper” F691L mutation with PLX3397. Cancer Discov. 2015;5:668–79.
McMahon CM, et al. Clonal selection with RAS pathway activation mediates secondary clinical resistance to selective FLT3 inhibition in acute myeloid leukemia. Cancer Discov. 2019;9:1050–63.
Heidel F, et al. Clinical resistance to the kinase inhibitor PKC412 in acute myeloid leukemia by mutation of Asn-676 in the FLT3 tyrosine kinase domain. Blood. 2006;107:293–300.
Pauwels D, Sweron B, Cools J. The N676D and G697R mutations in the kinase domain of FLT3 confer resistance to the inhibitor AC220. Haematologica. 2012;97:1773–4.
Tarver TC, et al. Gilteritinib is a clinically active FLT3 inhibitor with broad activity against FLT3 kinase domain mutations. Blood Adv. 2020;4:514–24.
Roloff GW, et al. Clinical and molecular response of acute myeloid leukemia harboring non-canonical FLT3 N676K driver mutations to contemporary FLT3 inhibitors. Haematologica. 2023;36(9):2218.
Schmalbrock LK, et al. Clonal evolution of acute myeloid leukemia with FLT3-ITD mutation under treatment with midostaurin. Blood. 2021;137:3093–104.
Kasi PM, Litzow MR, Patnaik MM, Hashmi SK, Gangat N. Clonal evolution of AML on novel FMS-like tyrosine kinase-3 (FLT3) inhibitor therapy with evolving actionable targets. Leuk Res Rep. 2016;5:7–10.
Kennedy VE, et al. Multi-omic single-cell sequencing reveals genetic and immunophenotypic clonal selection in patients with FLT3-mutated AML treated with gilteritinib/venetoclax. Blood. 2022;140:2244–6.
Sato T, et al. FLT3 ligand impedes the efficacy of FLT3 inhibitors in vitro and in vivo. Blood. 2011;117:3286–93.
Yang X, Sexauer A, Levis M. Bone marrow stroma-mediated resistance to FLT3 inhibitors in FLT3-ITD AML is mediated by persistent activation of extracellular regulated kinase. Br J Haematol. 2014;164:61–72.
Chen F, Ishikawa Y, Akashi A, Naoe T, Kiyoi H. Co-expression of wild-type FLT3 attenuates the inhibitory effect of FLT3 inhibitor on FLT3 mutated leukemia cells. Oncotarget. 2016;7:47018–32.
Chang YT, et al. Role of CYP3A4 in bone marrow microenvironment-mediated protection of FLT3/ITD AML from tyrosine kinase inhibitors. Blood Adv. 2019;3:908–16.
Traer E, et al. FGF2 from marrow microenvironment promotes resistance to FLT3 inhibitors in acute myeloid leukemia. Cancer Res. 2016;76:6471–82.
Sung PJ, Sugita M, Koblish H, Perl AE, Carroll M. Hematopoietic cytokines mediate resistance to targeted therapy in FLT3-ITD acute myeloid leukemia. Blood Adv. 2019;3:1061–72.
Park HJ, et al. Therapeutic resistance in acute myeloid leukemia cells is mediated by a novel ATM/mTOR pathway regulating oxidative phosphorylation. Elife. 2022. https://doi.org/10.7554/eLife.79940.
Joshi SK, et al. The AML microenvironment catalyzes a stepwise evolution to gilteritinib resistance. Cancer Cell. 2021;39:999-1014 e1018.
Bottomly D, et al. Integrative analysis of drug response and clinical outcome in acute myeloid leukemia. Cancer Cell. 2022;40:850-864 e859.
Ravandi F, et al. Phase 2 study of azacytidine plus sorafenib in patients with acute myeloid leukemia and FLT-3 internal tandem duplication mutation. Blood. 2013;121:4655–62.
Swaminathan M, et al. A phase I/II study of the combination of quizartinib with azacitidine or low-dose cytarabine for the treatment of patients with acute myeloid leukemia and myelodysplastic syndrome. Haematologica. 2021;106:2121–30.
Wang ES, et al. Phase 3 trial of gilteritinib plus azacitidine vs azacitidine for newly diagnosed FLT3mut+ AML ineligible for intensive chemotherapy. Blood. 2022;140:1845–57.
Shah NP, et al. Ponatinib in patients with refractory acute myeloid leukaemia: findings from a phase 1 study. Br J Haematol. 2013;162:548–52.
Kipp D, et al. A Phase-Ib/II clinical evaluation of ponatinib in combination with azacitidine in FLT3-ITD and CBL-mutant acute myeloid leukemia (PON-AZA study). Blood. 2021;138:2350–2350.
Kohl TM, et al. BH3 mimetic ABT-737 neutralizes resistance to FLT3 inhibitor treatment mediated by FLT3-independent expression of BCL2 in primary AML blasts. Leukemia. 2007;21:1763–72.
Chyla B, et al. Genetic biomarkers of sensitivity and resistance to venetoclax monotherapy in patients with relapsed acute myeloid leukemia. Am J Hematol. 2018;93:E202-205.
Singh Mali R, et al. Venetoclax combines synergistically with FLT3 inhibition to effectively target leukemic cells in FLT3-ITD+ acute myeloid leukemia models. Haematologica. 2021;106:1034–46.
Brinton LT, et al. Synergistic effect of BCL2 and FLT3 co-inhibition in acute myeloid leukemia. J Hematol Oncol. 2020;13:139.
Zhu R, et al. FLT3 tyrosine kinase inhibitors synergize with BCL-2 inhibition to eliminate FLT3/ITD acute leukemia cells through BIM activation. Signal Transduct Target Ther. 2021;6:186.
Daver N, et al. Venetoclax plus gilteritinib for FLT3-mutated relapsed/refractory acute myeloid leukemia. J Clin Oncol. 2022;40:4048–59.
Perl AE, et al. Gilteritinib or chemotherapy for relapsed or refractory FLT3-mutated AML. N Engl J Med. 2019;381:1728–40.
Yilmaz M, et al. Hypomethylating agent and venetoclax with FLT3 inhibitor “triplet” therapy in older/unfit patients with FLT3 mutated AML. Blood Cancer J. 2022;12:77.
Yilmaz M, et al. Quizartinib (QUIZ) with decitabine (DAC) and venetoclax (VEN) is active in patients (pts) with FLT3-ITD mutated acute myeloid leukemia (AML): A phase I/II clinical trial. J Clin Oncol. 2022;40:7036–7036.
Ong F, et al. A phase I/II study of combination of ASTX727, gilteritinib and venetoclax in patients with relapsed/refractory FLT3 mutated acute myeloid leukemia (AML) and frontline FLT3 mutated AML patients unfit for chemotherapy. Blood. 2022;140:9029–31.
Short NJ, et al. A triplet combination of azacitidine, venetoclax and gilteritinib for patients with FLT3-mutated acute myeloid leukemia: results from a phase I/II study. Blood. 2021;138:696.
Chua CC, et al. High deliverability of a midostaurin triplet regimen incorporating venetoclax and low dose cytarabine in non-adverse cytogenetic risk acute myeloid leukaemia: a sub-analysis of the Australasian leukaemia lymphoma group (ALLG) intervene study. Blood. 2022;140:3362–4.
Maiti A, et al. Triplet therapy with venetoclax, FLT3 inhibitor and decitabine for FLT3-mutated acute myeloid leukemia. Blood Cancer J. 2021;11:25.
Croce L, Coperchini F, Magri F, Chiovato L, Rotondi M. The multifaceted anti-cancer effects of BRAF-inhibitors. Oncotarget. 2019;10:6623–40.
O’Bryan JP. Pharmacological targeting of RAS: recent success with direct inhibitors. Pharmacol Res. 2019;139:503–11.
Carvajal LA, et al. SYK inhibitors, entospletinib and lanraplenib, show potent anti-leukemic activity in combination with targeted agents. Blood. 2022;140:5932–3.
Fathi AT, et al. The frida study: an option for mutated FLT3 relapsed/refractory acute myeloid leukemia patients with a novel iadademstat and gilteritinib combination therapy. Blood. 2022;140:11755–6.
Daver N, et al. A Phase 1/2 dose escalation study of the myeloid kinase inhibitor HM43239 in patients with relapsed or refractory acute myeloid leukemia. Blood. 2022;140:6197–9.
Goldberg AD, et al. A phase 1a/b dose escalation study of the FLT3/BTK inhibitor luxeptinib (CG-806) in patients with relapsed or refractory acute myeloid leukemia. Blood. 2022;140:6220–1.
Wang Y, et al. Preclinical evaluation of MAX-40279, a FLT3/FGFR dual kinase inhibitor for treatment of acute myeloid leukemia. Blood. 2018;132:3997–3997.
Law B, et al. Bmf-500: an orally bioavailable covalent inhibitor of FLT3 with high selectivity and potent antileukemic activity in FLT3-mutated AML. Blood. 2022;140:6191–2.
Heitmann JS, et al. First-in-human phase I dose escalation and expansion study evaluating the Fc optimized FLT3 antibody flysyn in acute myeloid leukemia patients with minimal residual disease. Blood. 2020;136:8–9.
Lachowiez CA, et al. Comparison and validation of the 2022 European LeukemiaNet guidelines in acute myeloid leukemia. Blood Adv. 2023;7:1899–909.
Dohner K, et al. Impact of NPM1/FLT3-ITD genotypes defined by the 2017 European LeukemiaNet in patients with acute myeloid leukemia. Blood. 2020;135:371–80.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
C. C. Smith reports research funding from Abbvie, Inc. and Revolution Medicines, and has served as an advisory board member of Abbvie, Inc. and Genentech.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Kennedy, V.E., Smith, C.C. FLT3 targeting in the modern era: from clonal selection to combination therapies. Int J Hematol (2023). https://doi.org/10.1007/s12185-023-03681-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12185-023-03681-0