Abstract
Understanding the spatiotemporal dynamics of trophic structures, or trophodynamics, is important for assessing the overall condition and function of estuarine ecosystems. We examined spatial and seasonal trophodynamics of nekton assemblages across the seascape of a subtropical estuary (Charlotte Harbor, FL, USA) using historical nekton data from an ongoing monitoring program. We also examined relationships between trophodynamics and the spatial distribution of vegetated habitats, such as salt marshes, mangroves, and seagrass beds, as well as their absence along hardened shorelines (seawalls or riprap) using seascape metrics (area and edge density of habitats within several search radii of sample locations). We found that nekton assemblages in shallow waters (from the shoreline to a depth of ≤ 1.5 m) transitioned from mainly planktivores in the lower Peace and Myakka Rivers to mainly benthivores (those feeding on benthic infauna, epifauna, or seagrass) in the middle and lower portions of Charlotte Harbor. Overall nekton assemblages throughout the estuary appeared to be largely sustained through benthic pathways in areas with abundant seagrass and nearby mangrove habitat, particularly during the cool-dry winter when planktivore densities were seasonally low. The dramatic seasonal decline in planktivore densities we found in the lower rivers may be related to low freshwater inflow and increased predation resulting from seasonal recruitment pulses of several omnivorous species. Trophodynamics linked with seascape metrics of estuarine habitat structure described here may be used as a spatially explicit baseline of the estuarine seascape to monitor ecosystem health as well as parameterize spatial ecosystem models used in fisheries management in Charlotte Harbor and similar subtropical estuaries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Estuarine nekton assemblages, consisting of numerous species of small fish and invertebrates, function as a significant trophic link between primary production and higher trophic levels (Beck et al. 2001; Nelson et al. 2015; Whitfield 2017). Trophic composition of nekton assemblages has been identified as an important indicator of ecosystem health (Elliott and Dewailly 1995; Deegan et al. 1997; Mathieson et al. 2000; Whitfield and Elliot 2002; Elliot et al. 2007; Sheaves et al. 2012), and ecological indices based on trophic composition of nekton assemblages have been incorporated into many ecosystem management directives across the world (Water Framework Directive in Europe, Oceans Act in the USA, Australia, or Canada, National Water Act in South Africa) to gauge health of coastal and estuarine ecosystems (Harrison and Whitfield 2006; Borja et al. 2008). In addition, spatial data on trophic functioning and relative importance of trophic pathways are needed to develop spatial food web models (e.g., Ecopath with Ecosim with EcoSpace, Walters et al. 2010; Atlantis, Fulton et al. 2011) used in ecosystem-based fishery management applications (Heymans et al. 2016; Grüss et al. 2018). Examining trophodynamics of estuarine nekton assemblages is therefore an important step in better understanding energy flows (Campos et al. 2015), as well as gauging and managing the human impact on these important ecosystem processes (Whitfield and Elliott 2002; McLusky and Elliott 2004; Sheaves et al. 2017).
Mangroves, salt marshes, and seagrass beds function as important primary producers in subtropical estuaries by providing food as well as habitat for many nekton species (see Whitfield 2017 for review). Relationships between habitat structure, defined as the amount and spatial arrangement of habitats (e.g., salt marshes, mangroves, and seagrass beds), and taxonomic composition of nekton assemblages have been found in estuaries worldwide (e.g., Pittman et al. 2004, Whaley et al. 2007, Franco et al. 2008, Olds et al. 2012, Santos et al. 2018, Henderson et al. 2021). Gilby et al. (2018) recently found that area and proximity to seagrass beds and mangroves, as well as proximity to the ocean, were related to taxonomic changes in fish assemblages in 13 estuaries along the southeastern coastline of Queensland, Australia. Similar relationships between nekton trophic composition and estuarine habitat structure have rarely been examined. However, a positive relationship was recently found between biomass of benthic and piscivorous nekton and seagrass area measured at a broad scale (800-m radius) in Venice Lagoon, Italy (Scapin et al. 2018). Given the trophic connection between nekton and vegetated habitats, trophic composition of nekton may be similarly related to broad-scale patterns of vegetated habitats in other estuaries as well. The extent that vegetated habitats have been replaced with hardened shorelines may also influence trophic functioning of nekton assemblages (Yabsley et al. 2020). Understanding spatial relationships between estuarine habitat structure and ecosystem processes, such as trophic pathways, is important for conservation and management of these estuarine ecosystems (Bostrom et al. 2011, Pittman et al. 2021).
We investigated spatial relationships between estuarine habitat structure (amount and arrangement of seagrass beds, mangroves, salt marshes, and hardened shorelines) and taxonomic and trophic composition of nekton assemblages during each of four seasons of the year within shallow waters of Charlotte Harbor, FL (USA), a subtropical estuary, as well as lower portions of the Peace and Myakka Rivers flowing into the Harbor. Our primary goals were to better understand the spatial and seasonal trophodynamics of nekton assemblages, examine potential spatial relationships between trophic composition and estuarine habitat structure, and create a historical baseline of trophodynamics across the shallow-water seascape of Charlotte Harbor. The specific objectives were to (1) describe relationships between estuarine habitat structure, environmental variables, and species composition of nekton assemblages, (2) characterize and map spatial distribution and seasonal dynamics of assemblage types based on dominant trophic guilds, and (3) describe spatial patterns of trophic guild dominance relative to habitat characteristics and seasonality.
Materials and Methods
Study Area
Charlotte Harbor (Fig. 1), located on Florida’s peninsular Gulf Coast, is one of the largest estuaries in the state, with an open-water area of approximately 489 km2. The Peace and Myakka Rivers are the primary freshwater drainages flowing into Charlotte Harbor. Charlotte Harbor’s climate is subtropical with a dry winter season and a pronounced rainy summer season. Approximately 60% of annual rainfall and peak streamflow occur from mid-June through early October (Flannery et al. 2010). Charlotte Harbor is considered microtidal with a semidiurnal tidal range of 0.6 m. A major inlet and several smaller passes through the barrier islands connect the Harbor to the Gulf of Mexico. Seawall and riprap shorelines have replaced natural shoreline vegetation in areas of the lower rivers, particularly along the Peace River adjacent to the municipalities of Punta Gorda and Port Charlotte (Electronic Supplemental Material, Fig. S1). Seagrass (principally shoal grass Halodule wrightii, turtle grass Thalassia testudinum, and manatee grass Syringodium filiforme) is common in shallow-water areas of Charlotte Harbor, with seagrass beds generally becoming more abundant with larger, more continuous patches in lower portions of the estuary. Shorelines in the estuary proper, defined as the area between the river mouths and inlets, are dominated by mangroves (red mangrove Rhizophora mangle, black mangrove Avicennia germinans, white mangrove Laguncularia racemosa). Salt marsh shorelines (primarily black needlerush Juncus roemerianus) generally occur in brackish portions of the rivers flowing into the estuary.
Estuarine Seascape Structure
We examined habitat structure across the estuary during the sampling period (1998–2003) by compiling GIS layers derived from aerial photography taken in 1999 and 2000 describing mangroves, salt marshes, seagrass beds, and human-altered shorelines (seawall and riprap). These subtidal and intertidal habitat types were selected because they represented the predominant habitats characterizing the “seascape” surrounding shallow-water (≤ 1.5 m) sample sites across the study area. We extracted mangrove and salt marsh habitat data from digital land use and land cover datasets from the South and Southwest Florida Water Management Districts for Charlotte Harbor (SFWMD and SWFWMD). These data were created by photointerpreting 1:12,000-scale United States Geological Survey color infrared digital orthophoto quarter quadrangle aerial images taken in 1999 and 2000. Photointerpretation was based on Florida Department of Transportation’s Florida Land Use and Cover Classification System (FDOT 1999). For seagrass habitat, we merged a map of seagrass habitat (SWFWMD) covering the northern part of Charlotte Harbor with a map covering the southern portion (SFWMD). Both data sets were derived by photointerpreting 1:24,000-scale natural color aerial photographs taken on December 26, 1999. We used maps of hardened shorelines (seawall or riprap) derived from aerial photography as spatial indicators of coastal development using Florida’s Environmental Sensitivity Index database (Jensen et al. 1998). More details regarding GIS habitat datasets can be found in Whaley et al. (2007).
We measured several types of habitat structure using these maps of estuarine habitats and altered shorelines. We measured area and edge density of seagrass, mangrove, and salt marsh habitats; proportion of altered shorelines; and the location of habitats relative to the passes connecting the embayment to the Gulf of Mexico. We refer to these measures of spatial configuration and position (habitat structure) as seascape metrics. The extent of the spatial influence of a habitat type or an altered shoreline on the estuarine nekton community likely extends a distance away from the habitat or shoreline; however, the distance or area of influence is not known and likely changes seasonally and for different nekton species (Whaley et al. 2007; Pittman and Brown 2011; Olds et al. 2012). Therefore, we quantified habitat structure over multiple distances away from sample locations (100 m, 200 m, 400 m, 800 m, 1600 m), and then used an ordination analysis to objectively identify the scales at which the measurements were the most relevant to nekton assemblage structure. We calculated the area (m2) of mangrove, salt marsh, discontinuous seagrass, continuous seagrass, and combined seagrass (discontinuous and continuous) within circular zones of increasing spatial extent (radii = 100, 200, 400, 800, and 1600 m) around each fish sampling site. Lengths of edges were also summed using the same circular zones, and then divided by area (edge density = m km−2). To calculate altered shorelines, we summed the length of edges classified as either seawall or riprap and then divided by the sum of all edges (both natural and altered). This resulted in percentage of altered shorelines within each circular zone. We also calculated the distance (m) from each sampling site to the nearest pass to the Gulf of Mexico using the cost–distance function provided in the ArcGIS software package (ESRI), using the shoreline as a barrier. These calculations resulted in a series of continuous grid surfaces representing each metric across the estuary, and a subset of these grids was used in subsequent spatial modeling. Seascape metric values at each sample location were log-transformed (Ln(x + 1)) and added to field-collected environment and habitat data to perform Redundancy Analysis.
Nekton Data Collection
Nekton data included in this analysis were collected monthly throughout the year by Florida’s Fisheries-Independent Monitoring Program (FIM, Florida Fish and Wildlife Conservation Commission/Fish and Wildlife Research Institute) during a 6-year period (1998–2003). Although the monitoring program is ongoing, the analysis was restricted to this period to function as a historical “baseline” to be compared with other time periods in the future. Nekton were collected using a 21.3-m bag seine (3.2-mm stretched mesh size) that targets smaller fishes (generally between 15 and 100 mm standard length) and was constrained to deployment in waters of 1.5-m depth or less. Seines are sometimes less efficient in capturing benthic nekton, especially in structurally complex habitats, such as seagrass beds (Rozas and Minello 1997). Therefore, benthivores and the proportion of the benthic contribution to the food web are likely underestimated in this analysis, particularly in seagrass beds. The shallow-water study area represented approximately 46% of the total open-water area of Charlotte Harbor. Seine samples were collected monthly across the estuary using a stratified random sampling methodology. In this methodology, Charlotte Harbor is divided into several large zones. Each of these zones was subdivided into 1 × 1 min cartographic grids. Each of these grid cells was further partitioned into 100 microgrid cells, and several microgrid cells randomly were chosen for sample collection each month. Samples were also stratified within each grid cell based on the presence and absence of seagrass and shoreline vegetation. Although seines were deployed along mangrove and salt marsh edges, seines were not directly deployed within wetland habitats. A detailed description of sampling procedures is described in Whaley et al. (2007). All fish species and invertebrates were identified to the lowest feasible taxonomic level (usually to species) in the field. However, Eucinostomus spp. and Gobiosoma spp. were identified to genus level when < 40 mm standard length (SL) and < 20 mm SL, respectively. For each species collected, a representative sample was retained to verify field identifications in the laboratory. Concurrent with each seine sample, numerous water quality parameters (salinity, dissolved oxygen, depth, and temperature) and habitat parameters (visual estimates of percent seagrass coverage, water depth) were recorded in the sampling area. Locations of nekton sample sites were recorded in the field via a global positioning system, and we used these locations to combine nekton and environmental data with seascape metrics calculated in GIS.
Statistical Analyses
Nekton data were divided into four seasonal time periods to examine taxonomic composition and trophodynamics. Seasons were based on both general wet and dry climate patterns in central Florida (similar to those in Tsou and Matheson 2002) as well as previous analyses of temporal changes in nekton community composition in the lower Peace and Myakka Rivers (Idelberger and Greenwood 2005). Spring was defined as samples from April and May (warm and variable rainfall, depending on year). Summer represented collections from June to October (hot and rainy). Fall represented November and December samples (warm and generally drier). Winter season included samples from January to March (cool and generally dry climate). Many species were divided into size classes using length-frequency diagrams to examine potential ontogenetic shifts in spatial distribution. For example, if a species exhibited a bimodal distribution in a length-frequency diagram, that species was divided into two size classes to represent each mode. Then, each size class of that species was analyzed as a separate “pseudo-species” to examine potential differences in mean density among assemblage types. Very rare species (those never comprising more than 1% in any sample) were removed from the analysis. For the remaining species, density (individuals per 100 m2 area swept by seine) was transformed using the chord transformation to give appropriate weights to rare species and therefore circumvent the problems associated with Euclidean distance as described in detail in Legendre and Gallagher (2001).
Community Ordination Analysis
We used Redundancy Analysis (RDA, CANOCO™ version 4.5; ter Braak and Smilauer 2002), to examine estuary-wide relationships between nekton species composition, habitat structure metrics measured at multiple scales (100-m to 1600-m radii), and local habitat conditions measured at the sample site. RDA simultaneously relates the distribution of fish species to synthetic gradients, called RDA axes, that are most related to changes in nekton species composition across the analysis area. Identified environmental and habitat variables most closely correlated to key gradients (RDA axes) are then used to interpret patterns in assemblage composition and facilitate spatial modeling of the nekton community. RDA is based on linear relationships between species and environmental (habitat) variables, and ordination axes are restricted to be linear combinations of explanatory variables (ter Braak and Smilauer 2002). The initial RDA for each season included seascape metrics describing area and edge densities for habitats (salt marsh, mangrove, continuous, discontinuous, and all seagrass combined, percentage of altered shorelines) at the five scales (100-m to 1600-m), as well as local-scale environmental parameters taken at sample sites. To reduce the number of variables in the final RDA community model, we retained only those variables that were highly correlated (r ≥ 85%) with the four main RDA axes. If no variable was highly correlated with an axis, we retained the variable that was most correlated with the axis.
Classifying Assemblage Types and Trophic Composition
We performed a cluster analysis using RDA Axis 1 sample scores to identify areas of similar species composition. To determine the most appropriate number of clusters to describe the nekton community in each season, we performed a species indicator analysis (Dufrene and Legendre 1997) across varying numbers of clusters (2–10). We selected the number of clusters that best described the highest number of individual species. For each season, samples were classified into assemblage types using RDA Axis 1 scores and were mapped in a GIS using ESRI software to examine spatial patterns. For each assemblage type, we calculated the mean density (number/100 m2) of all species and size classes. To examine seasonal trophodynamics, we assigned those taxa comprising 1% or more of any sample to trophic guilds (e.g., Elliott et al. 2007; Potter et al. 2015) using existing literature (Electronic Supplemental Material, Table S1). Trophic guilds included the following: planktivores (P) predominately feeding on phytoplankton and zooplankton; benthivores (B) primarily feeding on organisms living above and below the sediment; herbivores (H) mainly feeding directly on living macroalgal and macrophyte material; piscivores (F) primarily feeding on fishes; or omnivores (O) feeding on a variety of planktonic and benthic organisms. We summed mean densities of all species within a trophic guild and divided by the total mean nekton density to calculate the proportion of each trophic guild per sample.
Bootstrapping Seasonal Nekton Densities
We used stratified, non-parametric bootstrapping (i.e., resampling) to assess differences in mean densities of overall nekton as well as the most common species among assemblage types within each season. The bootstrapping simulations were conducted separately for each species and season in the following manner: for each replicated (i.e., re-sampled) data set, the mean density was calculated for each assemblage type, where each assemblage type represented a stratum, and the differences in mean density were calculated for all pairwise combinations of assemblage types. After this process was repeated 10,000 times, the resulting sampling distributions—each representing the difference between any two assemblage types—were summarized by calculating their means and 95% confidence limits. Confidence intervals that did not overlap zero were deemed significantly different. Because of the very large number of possible pairwise comparisons, we focused on comparing the assemblage type with the highest mean nekton density with all other assemblage types to identify those with similarly high densities.
Correlation Analysis Between Habitat and Trophic Composition
We used Spearman’s rank correlation tests for each season to identify potential relationships between seascape metrics (400-m radius) and the proportion and mean density of each trophic guild. The proportion of one trophic guild is dependent on proportions of the other guilds. Despite this non-independence, we included proportions to characterize relationships between trophic dominance and habitat structure. In contrast, densities of each trophic guild are independent, and we use density-habitat correlations to represent the relationship between habitat structure and each trophic guild.
Results
Spatial Habitat Structure Along the Estuarine Gradient
Shoreline vegetation was dominated by salt marshes in the northern portions of the Peace and Myakka Rivers, and gradually transitioned along the main estuarine gradient into mangroves throughout Charlotte Harbor proper. Spatial trends of habitat structure were similar among the radii (100 m to 1600 m); therefore, we report only the 400-m search radius (Electronic Supplemental Material, Table S2). The mean proportion of hardened shoreline (seawall or riprap) around sample sites was generally low throughout the estuary proper (0–10%) and much higher within the rivers (18–43%), especially near the municipalities of Punta Gorda and Port Charlotte. The highest mean area of patchy seagrass as well as mangrove habitat typically occurred in the middle portions of the estuary. The mean area of continuous seagrass and overall seagrass (both types combined) increased substantially from the middle to the lower portions of Charlotte Harbor. Edge metrics for mangroves, salt marshes, and seagrass beds typically followed the same spatial patterns as area metrics.
Environmental Conditions
Salinity was typically very low in the northern-most portions of the Peace and Myakka Rivers throughout the year and increased closer to the mouth of the estuary (Electronic Supplemental Material, Table S3). During the rainy summer season (June–October), the influx of freshwater resulted in lower mean salinities in the rivers and majority of Charlotte Harbor compared with those in the spring. In the fall (November–December) and winter (January–March), mean salinities were generally higher across most of the study area reflecting the drier seasonal climate. Water depth and dissolved oxygen were similar throughout the study area. Water temperature was generally similar across the study area within each season.
Broad-Scale Gradient Related to Nekton Assemblage Composition
Taxonomic composition of nekton assemblages varied along the main estuarine gradient, represented as RDA Axis 1, from the riverine areas to the passes near the Gulf of Mexico throughout all four seasons of the year. Depending upon season, RDA Axis 1 represented between 48 and 67% of the total variation in nekton assemblage composition explained in the analyses (Tables 1, 2, 3, and 4). This gradient was highly correlated with distance to the nearest pass (m) throughout all four seasons (distance, r = − 0.91 to − 0.94). Axis 1 was also highly correlated with area and edge density of continuous seagrass habitat measured at the 400-m (r = 0.89–0.95) and 800-m search radii (r = 0.88–0.96) in the spring, summer, and fall seasonal analyses. Salinity measurements were also highly correlated (summer: r = 0.85; fall: r = 0.90) with Axis 1 in the summer and fall but were uncorrelated with Axis 1 in the spring and winter. In winter, Axis 1 was most highly correlated with area and edge density of continuous seagrass habitat measured at the 800-m and 1600-m search radii. Based on permutation tests, all four axes identified in the final RDA for all four seasonal RDA analyses were significantly related to nekton assemblage composition (p < 0.001). However, RDA Axes 2–4 explained a small fraction (5–15%) of the total variation in nekton assemblage composition in the seasonal analyses. Therefore, we focused on changes in assemblage composition along RDA Axis 1 for each season.
Seasonal and Spatial Dynamics of Assemblages
The species indicator analysis divided nekton samples into seven to nine species assemblage types, depending on season (Electronic Supplemental Material, Tables S4–S7). Although many species were divided into size classes to examine potential ontogenetic shifts in diet, distribution patterns were very similar among size classes for most species. Therefore, we report on dominant nekton at the taxon level. To elucidate general spatial patterns in trophic structure, we classified and mapped assemblage types based on dominant trophic guild. All assemblage types were dominated by either benthivores, planktivores, or omnivores. Herbivores and piscivores represented very small proportions (less than 1%) of nekton assemblages throughout the study; therefore, we did not report on these guilds. Trophic dominance within assemblage types showed distinct spatial patterns that were somewhat consistent throughout the year. Based on these patterns, we delineated three distinct regions of the study area (riverine areas, Middle Charlotte Harbor, Lower Charlotte Harbor; Fig. 2). We then summarized percentages of trophic dominance for assemblages within each region over the four seasons to describe general patterns (Table 5).
Spatial distribution of dominant trophic guilds in a spring, b summer, c fall, and d winter seasons. Dominant trophic guild is indicated by color: planktivores (blue), benthivores (red), omnivores (yellow). Some assemblages were dominated by equal proportions of planktivores and benthivores (orange), or planktivores and omnivores (green). Three general regions of trophic dominance included riverine areas, Middle Charlotte Harbor, and Lower Charlotte Harbor
The middle and lower portions of the Peace and Myakka Rivers during spring, summer, and fall were dominated by plankton feeders, mainly bay anchovy (Anchoa mitchilli; Electronic Supplemental Material, Tables S4–S6). Plankton feeders were most dominant during the spring when they represented 94–96% of nekton in the lower rivers (Table 5; Fig. 2a). As freshwater inflow increased in the summer rainy season, planktivore-dominant assemblages expanded into the middle estuary and represented a notable proportion (26%) of samples collected in this area (Fig. 2b). As freshwater inflow subsided in the fall, the spatial extent of these planktivore assemblages retracted back into the rivers (Fig. 2c). As the relatively dry climate persisted into the winter, planktivore-dominated assemblages continued to decline and only represented 50% of the nekton samples collected within the riverine areas (Fig. 2d). Most of the remaining samples (46%) were dominated by omnivores, primarily spot (Leiostomus xanthurus; Electronic Supplemental Material, Table S7). Assemblages in riverine areas were rarely dominated (0–4% samples) by benthivores (Table 5).
In contrast, benthivores generally dominated nekton assemblages in middle and lower portions of Charlotte Harbor throughout most of the year (Table 5; Fig. 2a–d). During the fall, however, the lower estuary contained equal proportions of nekton samples dominated by benthivores (50%) and omnivores (50%; Fig. 2c). The most dominant benthivore species depended upon season and included pinfish (Lagodon rhomboides), rainwater killifish (Lucania parva), clown goby (Microgobius gulosus), tidewater mojarra (Eucinostomus harengulus), and silver jenny (Eucinostomus gula; Electronic Supplemental Material, Tables S4–S7).
Relationships Between Habitat and Trophic Composition
Seascape metrics describing continuous and discontinuous seagrass habitat, distance to the nearest inlet to the Gulf of Mexico, proportion of altered shorelines, and salinity measurements were related to composition of the two main trophic guilds (planktivores and benthivores) during most of the year (Fig. 3). The proportion of planktivores was positively correlated with distance to the inlet, and negatively related to area of both continuous and discontinuous seagrass as well as salinity throughout most of the year; this reflects their typical dominance in nekton assemblages in the lower Peace and Myakka Rivers. For most of the year, the proportion and density of planktivores were positively related to the proportion of altered shorelines which was highest within the lower Peace River and associated with the municipalities of Punta Gorda and Port Charlotte (Electronic Supplemental Material, Table S2). Benthivores generally occurred in higher densities and proportions in the lower estuary, and correlations reflected this general spatial pattern (Fig. 3). During most of the year, the proportion and density of benthivores were positively correlated with the area of continuous seagrass habitat, area of mangrove habitat, and salinity, as well as distance to the nearest pass. The proportion and density of benthivores were also negatively correlated with proportion of altered shorelines during most of year. The proportion of omnivores was negatively correlated with distance to the inlet and positively correlated with area of continuous seagrass habitat during the summer and fall. These relationships reflected the typical spatial pattern of relatively high proportion of omnivores in lower Charlotte Harbor during the summer and fall. In the winter, however, this spatial pattern reversed as relatively high densities of omnivores occurred in the lower rivers resulting in positive relationships with proportion and density of omnivores and distance to the inlet, and negative relationships to salinity and area of continuous seagrass.
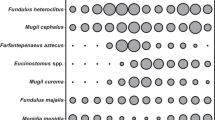
Spearman correlations between seascape metrics and trophic composition in a spring, b summer, c fall, and d winter seasons. Blue indicates positive correlation; red indicates negative correlation. Shade and size of circle are relative to strength of correlation. Blank cells indicate a non-significant correlation (p > 0.05). Variables included are area of mangrove, discontinuous seagrass, continuous seagrass, and salt marsh habitat (m2) within a 400-m radius of sample sites, distance (m) to the nearest inlet to the Gulf of Mexico, altered shorelines represented edge density of altered shorelines within 400-m radius (m km−2), and salinity (ppt). Variables describing nekton and trophic guilds included percent and density (number/100 m2) of planktivores, omnivores, and benthivores
Discussion
The spatial transition in trophic composition along the main estuarine gradient from planktivore-dominated assemblages in the lower rivers to those dominated by benthivores in the middle and lower portions of the estuary reflects the spatial distribution of dominant food resources and habitats across the Charlotte Harbor seascape. Although high concentrations of plankton feeders have previously been documented in the lower Peace and Myakka Rivers (Peebles 2002; Olin et al. 2013), the dominance of trophic guilds throughout the shallow waters of Charlotte Harbor has not been studied. Similar patterns were found in several tropical estuaries in India where the proportion of plankton feeders in nekton assemblages declined from the upper to lower reaches of the estuary (Sreekanth et al. 2020). In a tropical estuary in Brazil, nekton consumers near the mouth of the estuary assimilated nutrients derived mainly from benthic sources (macroalgae, seagrass, and benthic organic material; Claudino et al. 2015). Sheaves et al. (2017) also found spatial differences in nekton trophic composition within Australian estuaries indicating spatial changes in ecosystem processes across different reaches. However, they found a higher contribution of planktivore biomass in the lower reaches of 28 tropical Australian estuaries (Sheaves et al. 2017). The transition of nutrition sources from mainly planktonic to benthic that we found in Charlotte Harbor indicates a spatial change in key ecosystem processes sustaining nekton assemblages along the main estuarine gradient.
Although estuarine habitat structure of seagrasses, mangroves, and salt marshes has been related to taxonomic changes in nekton assemblages in estuaries throughout the world (e.g., Pittman et al. 2004; Whaley et al. 2007; França et al. 2009; Skilleter et al. 2017; Gilby et al. 2018; Santos et al. 2018), this study is one of the first to report relationships between seascape metrics of estuarine habitat structure and nekton trophic composition. The positive relationships between benthic feeders (density and proportion) and area (400-m radius) of both seagrass and mangroves are likely a result of increased benthic productivity typically found in and around these habitats (Heck et al. 2008; Abrantes et al. 2015). A similar positive relationship between seagrass area (800-m radius) and biomass of hyperbenthivorous and piscivorous species was found for nekton assemblages in Venice Lagoon, Italy (Scapin et al. 2018). The negative relationships we found between density of benthic feeders and proportion of human-altered shorelines may reflect reductions in detrital export from natural shorelines (mainly mangroves) that have been replaced by seawalls and riprap in the lower rivers. Lowe and Peterson (2015) also found shifts in nekton diets and reductions in benthic infaunal prey in areas with hardened shorelines in marsh-dominated seascapes in the northern Gulf of Mexico. The influence of hardened shorelines on nekton trophic structure remains unclear in mangrove-dominated estuaries (see Dugan et al. 2018) and may depend on the overall extent of urbanization (Yabsley et al. 2020). We found seascape metrics of both natural and human-altered habitat to be useful in understanding trophodynamics across the riverine/estuarine seascape of Charlotte Harbor. Monitoring trophic processes is emerging as an important component in assessing overall ecosystem health in estuaries throughout the world (Harrison and Whitfield 2006; Borja et al. 2008). Linking trophic processes, particularly benthic pathways, with seascape metrics of estuarine habitats appears to be a useful tool in assessing overall ecosystem health.
Seasonal fluctuations in the contribution of planktonic pathways corresponded to cyclical inputs of freshwater inflow and associated nutrients in Charlotte Harbor. The lower portions of Peace and Myakka Rivers are known to be dominated by the planktivorous bay anchovy, particularly in the wet summer season (Idelberger and Greenwood 2005). In nearby Tampa Bay 90 km to the north, high densities of bay anchovy tend to be in and near the river plume frontal zone because of large aggregations of zooplankton prey in those areas (Peebles 2002). The extremely high densities of bay anchovies in the lower Peace and Myakka Rivers observed in this study during spring likely coincide with the river plume frontal zone containing maximum productivity and chlorophyll-a concentrations (McPherson et al. 1990). The downstream movement and expansion of these planktivore-dominated assemblages into the estuary during the summer rainy season may be a result of similar movement of the nearby river plume frontal zone and planktonic prey aggregations (McPherson et al. 1990). The reduction in spatial distribution of planktivore-dominated assemblages in the dry seasons (fall and winter) may be related to reduced freshwater inflow and nutrient inputs (McPherson et al. 1990). Densities of bay anchovy were closely related to changes in freshwater inflow in both Tampa Bay (Peebles 2002) and Charlotte Harbor (Stevens et al. 2013; Olin et al. 2013). Similar seasonal transitions in trophic dominance from wet to dry seasons were found in many Australian estuaries (Abrantes and Sheaves 2010; Sheaves et al. 2017).
In addition to reduced freshwater and nutrient inputs, increased predation pressure may also play a role in reducing planktivore densities in the lower Peace and Myakka Rivers during the dry season. Seasonal recruitment pulses of several omnivorous species, such as sand seatrout (Cynoscion arenarius, mainly in summer), red drum (Sciaenops ocellatus, mainly in fall), and spot (mainly in winter), commonly occurred in the lower riverine areas where densities of bay anchovies declined dramatically. A previous study (Whaley et al. 2007) also found that juveniles of both red drum and sand seatrout were likely to occur in bay anchovy–dominated assemblages of the lower Peace and Myakka Rivers during the fall (September–November). Juveniles of red drum and spot occasionally feed on fish (Peters and McMicheal 1987, Akin and Winemiller 2015), whereas juvenile sand seatrout sometimes feed on bay anchovies (Sheridan and Livingston 1979). Omnivorous species that occasionally eat fish commonly occur within estuarine littoral zones (Baker and Sheaves 2021), and these species may be important sources of mortality within nekton assemblages in Charlotte Harbor and elsewhere (Baker and Sheaves 2009).
Implications and Future Research
Understanding how key trophic processes vary across estuarine seascapes, such as Charlotte Harbor, is fundamental in gauging the relative condition and function of these ecosystems through time (Bostrom et al. 2011, Pittman et al. 2021). Like many estuarine ecosystems throughout the world, the Charlotte Harbor watershed is projected to become more urbanized in coming decades (Zwick and Carr 2006). Human encroachment in the watershed will likely result in increased impervious surfaces, nutrient inputs, and a greater human need for freshwater (Flannery et al. 2002). Seasonal increases in freshwater inflow and associated nutrient inputs (nitrogen) stimulate phytoplankton productivity in Charlotte Harbor (McPherson et al. 1990) and likely contribute to high densities of planktivores we found in the lower Peace and Myakka Rivers. However, very high nutrient levels from increased urbanization can lead to hypoxia and reduced water clarity that can greatly reduce overall benthic productivity, as well as the health of seagrass beds (Duarte 2002). Reduced benthic productivity or seagrass loss from poor water quality may cause a decline in benthivores that dominate nekton assemblages throughout most of our Charlotte Harbor study area. Benthic productivity may also be reduced through loss of mangrove shorelines that supply detrital organic material to adjacent shallow-water areas. Benthivorous species can be early indicators of anthropogenic stress in estuaries because they can be highly sensitive to human-induced disturbances (e.g., loss and degradation of important habitats; Teichert et al. 2018). The benthic component of nekton assemblages has been particularly useful in multimetric indices used to gauge estuary health within the European Water Framework Directive (Borja et al. 2004; Coates et al. 2007; Uriarte and Borja 2009; Delpech et al. 2010). In Charlotte Harbor, benthic pathways appear to provide stability to the ecosystem by supplying a consistent food source, particularly in the dry, cool seasons when plankton production is seasonally low. Multiple trophic pathways (e.g., planktonic and benthic) also increase stability of overall trophic production over time, as well as contribute to ecosystem resilience to environmental perturbations (Pimm 1982; Winemiller and Layman 2005; Lobry et al. 2008; Peralta-Maraver et al. 2017). Potential threats from increased human encroachment on estuaries such as the Charlotte Harbor ecosystem highlight the need to monitor spatial changes in trophodynamics, particularly the benthic component, over time.
Spatial trophodynamics described in this study can not only help to monitor changes to ecosystem health, but also develop and refine spatial food web models used in ecosystem-based fishery management applications. This analysis covered the earliest years of Florida’s Fisheries-Independent Monitoring Program (1998–2003) and can therefore serve as a historical baseline to assess trophic composition changes over decadal or longer time scales. Monitoring temporal changes in trophic characteristics of nekton assemblages is increasingly used globally to monitor health of estuaries and coastal ecosystems (Borja et al. 2008). For example, a multimetric nekton index based partially on trophic composition was recently developed to monitor the health of Tampa Bay (Schrandt et al. 2021). Similar indices based on seasonal trophodynamics could be developed for Charlotte Harbor to assess ecosystem health as human development increases in the watershed. Spatial trophodynamics can also provide a better understanding of the energy flows in estuarine food webs for spatial food models (e.g., EcoSpace, Walters et al. 2010; Atlantis, Fulton et al. 2011) needed for ecosystem-based fishery management applications (Cury et al. 2005; Heymans et al. 2016, Gruss et al. 2018). The large contribution of the benthic pathway of the overall food web of Charlotte Harbor demonstrates the need to include both benthic and planktonic production to estimate productivity within estuarine ecosystem models.
References
Abrantes, K.G., R. Johnston, R.M. Connolly, and M. Sheaves. 2015. Importance of mangrove carbon for aquatic food webs in wet–dry tropical estuaries. Estuaries and Coasts 38 (1): 383–399.
Abrantes, K.G., and M. Sheaves. 2010. Importance of freshwater flow in terrestrial–aquatic energetic connectivity in intermittently connected estuaries of tropical Australia. Marine Biology 157 (9): 2071–2086.
Akin, S., and K.O. Winemiller. 2015. Habitat use and diets of juvenile spot (Leiostomus xanthurus) and Atlantic croaker (Micropogonias undulatus) in a small estuary at Mad Island Marsh Texas. Texas Journal of Science 64 (1): 3–31.
Baker, R., and M. Sheaves. 2021. Predation in littoral habitats is a complex process: Comment on Whitfield (2020). Marine Ecology Progress Series 662: 205–208.
Baker, R., and M. Sheaves. 2009. Overlooked small and juvenile piscivores dominate shallow-water estuarine “refuges” in tropical Australia. Estuarine, Coastal and Shelf Science 85 (4): 618–626.
Beck, M.W., K.L. Heck, K.W. Able, D.L. Childers, D.B. Eggleston, B.M. Gillanders, B. Halpern, C.G. Hays, K. Hoshino, T.J. Minello, and R.J. Orth. 2001. The identification, conservation, and management of estuarine and marine nurseries for fish and invertebrates: A better understanding of the habitats that serve as nurseries for marine species and the factors that create site-specific variability in nursery quality will improve conservation and management of these areas. BioScience 51 (8): 633–641.
Borja, Á., S.B. Bricker, D.M. Dauer, N.T. Demetriades, J.G. Ferreira, A.T. Forbes, P. Hutchings, X. Jia, R. Kenchington, J.C. Marques, and C. Zhu. 2008. Overview of integrative tools and methods in assessing ecological integrity in estuarine and coastal systems worldwide. Marine Pollution Bulletin 56 (9): 1519–1537.
Borja, Á., J. Franco, V. Valencia, J. Bald, I. Muxika, M.J. Belzunce, and O. Solaun. 2004. Implementation of the European water framework directive from the Basque country (northern Spain): A methodological approach. Marine Pollution Bulletin 48 (3–4): 209–218.
Boström, C., S.J. Pittman, C. Simenstad, and R.T. Kneib. 2011. Seascape ecology of coastal biogenic habitats: Advances, gaps, and challenges. Marine Ecology Progress Series 427: 191–217.
Campos, D.M.A.R., A.F.D. Silva, N.D.S. Sales, R.E.M.C.C. Oliveira, and A.L.M. Pessanha. 2015. Trophic relationships among fish assemblages in a mudflat within Brazilian marine protected area. Brazilian Journal of Oceanography 63 (2): 135–146.
Claudino, M.C., A.L.M. Pessanha, F.G. Araújo, and A.M. Garcia. 2015. Trophic connectivity and basal food sources sustaining tropical aquatic consumers along a mangrove to ocean gradient. Estuarine, Coastal and Shelf Science 167: 45–55.
Coates, S., A. Waugh, A. Anwar, and M. Robson. 2007. Efficacy of a multi-metric fish index as an analysis tool for the transitional fish component of the Water Framework Directive. Marine Pollution Bulletin 55 (1–6): 225–240.
Cury, P.M., L.J. Shannon, J.P. Roux, G.M. Daskalov, A. Jarre, C.L. Moloney, and D. Pauly. 2005. Trophodynamic indicators for an ecosystem approach to fisheries. ICES Journal of Marine Science 62 (3): 430–442.
Deegan, L.A., J.T. Finn, S.G. Ayvazian, C.A. Ryder-Kieffer, and J. Buonaccorsi. 1997. Development and validation of an estuarine biotic integrity index. Estuaries 20: 601–617.
Delpech, C., A. Courrat, S. Pasquaud, J. Lobry, O. Le Pape, D. Nicolas, P. Boët, M. Girardin, and M. Lepage. 2010. Development of a fish-based index to assess the ecological quality of transitional waters: The case of French estuaries. Marine Pollution Bulletin 60 (6): 908–918.
Dufrêne, M., and P. Legendre. 1997. Species assemblages and indicator species: The need for a flexible asymmetrical approach. Ecological Monographs 67 (3): 345–366.
Dugan, J.E., K.A. Emery, M. Alber, C.R. Alexander, J.E. Byers, A.M. Gehman, N. McLenaghan, and S.E. Sojka. 2018. Generalizing ecological effects of shoreline armoring across soft sediment environments. Estuaries and Coasts 41 (1): 180–196.
Duarte, C.M. 2002. The future of seagrass meadows. Environmental Conservation 29 (2): 192–206.
Elliott, M., and F. Dewailly. 1995. Structure and components of European estuarine fish assemblages. Netherlands Journal of Aquatic Ecology 29: 397–417.
Elliott, M., A.K. Whitfield, I.C. Potter, S.J. Blaber, D.P. Cyrus, F.G. Nordlie, and T.D. Harrison. 2007. The guild approach to categorizing estuarine fish assemblages: A global review. Fish and Fisheries 8 (3): 241–268.
Flannery, M.S., E.B. Peebles, and R.T. Montgomery. 2002. A percent-of-flow approach for managing reductions of freshwater inflows from unimpounded rivers to southwest Florida estuaries. Estuaries 25 (6): 1318–1332.
Flannery, M.S., J.E. Loper, X. Chen, and K. Hackett. 2010. Watershed and estuarine modeling of the effects of excess freshwater flows resulting from agricultural land use and other watershed alterations on the salinity characteristics of the lower Myakka River estuary, Florida. M. L. Spaulding ed. Proceedings of 11th International Conference on Estuarine and Coastal Modeling, Seattle, Washington, United States, 4–6 November.
Florida Department of Transportation. 1999. Florida land use, cover and forms classification system handbook, 3rd ed. Maps and Publications Office, Tallahassee, FL: Florida Department of Transportation.
França, S., M. Costa, and H. Cabral. 2009. Assessing habitat specific fish assemblages in estuaries along the Portuguese coast. Estuarine, Coastal and Shelf Science 83 (1): 1–12.
Franco, A., M. Elliott, P. Franzoi, and P. Torricelli. 2008. Life strategies of fishes in European estuaries: The functional guild approach. Marine Ecology Progress Series 354: 219–228.
Fulton, E.A., J.S. Link, I.C. Kaplan, M. Savina-Rolland, P. Johnson, C. Ainsworth, P. Horne, R. Gorton, R.J. Gamble, A.D. Smith, and D.C. Smith. 2011. Lessons in modelling and management of marine ecosystems: The Atlantis experience. Fish and Fisheries 12 (2): 171–188.
Gilby, B.L., A.D. Olds, R.M. Connolly, P.S. Maxwell, C.J. Henderson, and T.A. Schlacher. 2018. Seagrass meadows shape fish assemblages across estuarine seascapes. Marine Ecology Progress Series 588: 179–189.
Grüss, A., J.T. Thorson, E.A. Babcock, and J.H. Tarnecki. 2018. Producing distribution maps for informing ecosystem-based fisheries management using a comprehensive survey database and spatio-temporal models. ICES Journal of Marine Science 75 (1): 158–177.
Harrison, T.D., and A.K. Whitfield. 2006. Application of a multimetric fish index to assess the environmental condition of South African estuaries. Estuaries and Coasts 29 (6): 1108–1120.
Heck, K.L., T.J. Carruthers, C.M. Duarte, A.R. Hughes, G. Kendrick, R.J. Orth, and S.W. Williams. 2008. Trophic transfers from seagrass meadows subsidize diverse marine and terrestrial consumers. Ecosystems 11 (7): 1198–1210.
Henderson, C.J., B.L. Gilby, E. Stone, H.P. Borland, and A.D. Olds. 2021. Seascape heterogeneity modifies estuarine fish assemblages in mangrove forests. ICES Journal of Marine Science 78 (3): 1108–1116.
Heymans, J.J., M. Coll, J.S. Link, S. Mackinson, J. Steenbeek, C. Walters, and V. Christensen. 2016. Best practice in Ecopath with Ecosim food-web models for ecosystem-based management. Ecological Modelling 331: 173–184.
Idelberger, C.F., and M.F. Greenwood. 2005. Seasonal variation in fish assemblages within the estuarine portions of the Myakka and Peace Rivers, southwest Florida. Gulf of Mexico Science 23 (2): 224.
Jensen, J.R., J.N. Halls, and J. Michel. 1998. A systems approach to environmental sensitivity index (ESI) mapping for oil spill contingency planning and response. Photogrammetric Engineering and Remote Sensing 64: 1003–1014.
Legendre, P., and E.D. Gallagher. 2001. Ecologically meaningful transformations for ordination of species data. Oecologia 129: 271–280.
Lobry, J., V. David, S. Pasquaud, M. Lepage, B. Sautour, and E. Rochard. 2008. Diversity and stability of an estuarine trophic network. Marine Ecology Progress Series 358: 13–25.
Lowe, M.R., and M.S. Peterson. 2015. Body condition and foraging patterns of nekton from salt marsh habitats arrayed along a gradient of urbanization. Estuaries and Coasts 38 (3): 800–812.
Mathieson, S., A. Cattrijsse, M.J. Costa, P. Drake, M. Elliott, J. Gardner, and J. Marchand. 2000. Fish assemblages of European tidal marshes: A comparison based on species, families and functional guilds. Marine Ecology Progress Series 204: 225–242.
McLusky, D.S., and M. Elliott. 2004. The estuarine ecosystem: Ecology, threats and management, 3rd ed. Oxford: OUP.
McPherson, B.F., R.T. Montgomery, and E.E. Emmons. 1990. Phytoplankton productivity and biomass in the Charlotte Harbor estuarine system, Florida. JAWRA Journal of the American Water Resources Association 26 (5): 787–800.
Nelson, J.A., L. Deegan, and R. Garritt. 2015. Drivers of spatial and temporal variability in estuarine food webs. Marine Ecology Progress Series 533: 67–77.
Olin, J.A., N.E. Hussey, S.A. Rush, G.R. Poulakis, C.A. Simpfendorfer, M.R. Heupel, and A.T. Fisk. 2013. Seasonal variability in stable isotopes of estuarine consumers under different freshwater flow regimes. Marine Ecology Progress Series 487: 55–69.
Olds, A.D., R.M. Connolly, K.A. Pitt, and P.S. Maxwell. 2012. Primacy of seascape connectivity effects in structuring coral reef fish assemblages. Marine Ecology Progress Series 462: 191–203.
Peebles, E.B. 2002. Temporal resolution of biological and physical influences on bay anchovy Anchoa mitchilli egg abundance near a river-plume frontal zone. Marine Ecology Progress Series 237: 257–269.
Peralta-Maraver, I., A.L. Robertson, E.L. Rezende, A.L. Lemes da Silva, D. Tonetta, M. Lopes, R. Schmitt, N.K. Leite, A. Nuñer, and M.M. Petrucio. 2017. Winter is coming: Food web structure and seasonality in a subtropical freshwater coastal lake. Ecology and Evolution 7 (13): 4534–4542.
Peters, K.M., and R.H. McMichael. 1987. Early life history of the red drum, Sciaenops ocellatus (Pisces: Sciaenidae), in Tampa Bay Florida. Estuaries 10 (2): 92–107.
Pimm, S.L. 1982. Food webs. London: Chapman & Hall.
Pittman, S.J., and K.A. Brown. 2011. Multi-scale approach for predicting fish species distributions across coral reef seascapes. PLoS ONE 6: e20583. https://doi.org/10.1371/journal.pone.0020583.
Pittman, S.J., C.A. McAlpine, and K.M. Pittman. 2004. Linking fish and prawns to their environment: A hierarchical landscape approach. Marine Ecology Progress Series 283: 233–254.
Pittman, S.J., K.L. Yates, P.J. Bouchet, D. Alvarez-Berastegui, S. Andréfouët, S.S. Bell, C. Berkström, C. Boström, C.J. Brown, R.M. Connolly, and R. Devillers. 2021. Seascape ecology: Identifying research priorities for an emerging ocean sustainability science. Marine Ecology Progress Series 663: 1–29.
Potter, I.C., J.R. Tweedley, M. Elliott, and A.K. Whitfield. 2015. The ways in which fish use estuaries: A refinement and expansion of the guild approach. Fish and Fisheries 16 (2): 230–239.
Rozas, L.P., and T.J. Minello. 1997. Estimating densities of small fishes and decapod crustaceans in shallow estuarine habitats: A review of sampling design with focus on gear selection. Estuaries 20 (1): 199–213.
Santos, R.O., D. Lirman, S.J. Pittman, and J.E. Serafy. 2018. Spatial patterns of seagrasses and salinity regimes interact to structure marine faunal assemblages in a subtropical bay. Marine Ecology Progress Series 594: 21–38.
Scapin, L., M. Zucchetta, A. Sfriso, and P. Franzoi. 2018. Local habitat and seascape structure influence seagrass fish assemblages in the Venice Lagoon: The importance of conservation at multiple spatial scales. Estuaries and Coasts 41 (8): 2410–2425.
Schrandt, M.N., T.C. MacDonald, E.T. Sherwood, and M.W. Beck. 2021. A multimetric nekton index for monitoring, managing and communicating ecosystem health status in an urbanized Gulf of Mexico estuary. Ecological Indicators 123: 107310.
Skilleter, G.A., N.R. Loneragan, A. Olds, Y. Zharikov, and B. Cameron. 2017. Connectivity between seagrass and mangroves influences nekton assemblages using nearshore habitats. Marine Ecology Progress Series 573: 25–43.
Sheaves, M., R. Johnston, and R.M. Connolly. 2012. Fish assemblages as indicators of estuary ecosystem health. Wetland Ecology and Management 20: 477–490.
Sheaves, M., R. Baker, K.G. Abrantes, and R.M. Connolly. 2017. Fish biomass in tropical estuaries: Substantial variation in food web structure, sources of nutrition and ecosystem-supporting processes. Estuaries and Coasts 40 (2): 580–593.
Sheridan, Peter F., and R.J. Livingston. 1979. Cyclic trophic relationships of fishes in an unpolluted, river-dominated estuary in north Florida. In Ecological processes in coastal and marine systems, ed. R.J. Livingston, 143–161. New York: Springer.
Sreekanth, G.B., A.K. Jaiswar, H.B. Shivkumar, B. Manikandan, and E.B. Chakurkar. 2020. Fish composition and assemblage structure in tropical monsoonal estuaries: Estuarine use and feeding guild approach. Estuarine, Coastal and Shelf Science 244: 106911.
Stevens, P.W., M.F. Greenwood, and D.A. Blewett. 2013. Fish assemblages in the oligohaline stretch of a southwest Florida river during periods of extreme freshwater inflow variation. Transactions of the American Fisheries Society 142 (6): 1644–1658.
ter Braak C.J.F., and P. Smilauer. (2002) CANOCO reference manual and CanoDraw for Windows user’s guide: software for canonical community ordination (version 4.5). New York: Microcomputer Power.
Teichert, N., M. Lepage, and J. Lobry. 2018. Beyond classic ecological assessment: The use of functional indices to indicate fish assemblages’ sensitivity to human disturbance in estuaries. Science of the Total Environment 639: 465–475.
Tsou, T.S., and R.E. Matheson. 2002. Seasonal changes in the nekton community of the Suwannee River estuary and the potential impacts of freshwater withdrawal. Estuaries 25 (6): 1372–1381.
Uriarte, A., and A. Borja. 2009. Assessing fish quality status in transitional waters, within the European Water Framework Directive: Setting boundary classes and responding to anthropogenic pressures. Estuarine, Coastal and Shelf Science 82 (2): 214–224.
Walters, C., V. Christensen, W. Walters, and K. Rose. 2010. Representation of multistanza life histories in Ecospace models for spatial organization of ecosystem trophic interaction patterns. Bulletin of Marine Science 86 (2): 439–459.
Whaley, S.D., J.J. Burd Jr., and B.A. Robertson. 2007. Using estuarine landscape structure to model distribution patterns in nekton communities and in juveniles of fishery species. Marine Ecology Progress Series 330: 83–99.
Whitfield, A.K., and M. Elliott. 2002. Fishes as indicators of environmental and ecological changes within estuaries: A review of progress and some suggestions for the future. Journal of Fish Biology 61: 229–250.
Whitfield, A.K. 2017. The role of seagrass meadows, mangrove forests, salt marshes and reed beds as nursery areas and food sources for fishes in estuaries. Reviews in Fish Biology and Fisheries 27 (1): 75–110.
Winemiller, K.O., and C.A. Layman. 2005. Food web science: Moving on the path from abstraction to prediction. In Dynamic food webs: Multispecies assemblages, ecosystem development and environmental change, ed. P.C. Ruiter, V. Wolters, and J.C. Moore, 10–23. Amsterdam: Elsevier.
Yabsley, N.A., Gilby, B.L., Schlacher, T.A., Henderson, C.J., Connolly, R.M., Maxwell, P.S. and Olds, A.D. 2020. Landscape context and nutrients modify the effects of coastal urbanization. Marine Environmental Research 158: 104936.
Zwick, P.D., and M.H. Carr. 2006. Florida 2060: A population distribution scenario for the State of Florida, 25. Gainesville, FL: Geoplan Center, University of Florida.
Acknowledgements
We are grateful to the Florida Fish and Wildlife Research Institute’s Fisheries-Independent Monitoring Program for collecting the field data and analyzing species-size classes. Our appreciation also goes to the Southwest and South Florida Water Management District for use of their land use/land cover and seagrass GIS coverages. In addition, we greatly appreciate support by senior staff (R. Baumstark, R. Flamm, and K. O’Keife) and the entire Center for Spatial Analysis team at FWRI (partially supported by the US Fish and Wildlife Service, Sport Fish Restoration Grant F-66), and Latosha Thompson for creating map figures. We appreciate A. Adams, E. Matheson, and three anonymous reviewers for providing helpful input on earlier drafts of the manuscript. A State Wildlife Grant (Grant Number T312) from the Florida Fish and Wildlife Conservation Commission generously supported this analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Mark S. Peterson
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Whaley, S.D., Shea, C.P., Burd, J.J. et al. Trophodynamics of Nekton Assemblages and Relationships with Estuarine Habitat Structure Across a Subtropical Estuary. Estuaries and Coasts 46, 580–593 (2023). https://doi.org/10.1007/s12237-022-01134-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-022-01134-1