Abstract
Introduction
Daptomycin, a rapid concentration-dependent bactericidal antibiotic, is approved at a dose of 4 mg/kg/day for the treatment of complicated skin and soft tissue infections (cSSTI) and at a dose of 6 mg/kg/day for the treatment of Staphylococcus aureus right-sided infective endocarditis (RIE) and bacteremia associated with cSSTI and RIE. Studies have reported the successful use of high-dose daptomycin (>6 mg/kg/day) in patients with difficult-to-treat infections. The present analysis evaluated the effectiveness and safety of high doses (>6 mg/kg/day) of daptomycin for the treatment of different Gram-positive infections.
Methods
European Cubicin® Outcomes Registry and Experience (EU-CORE) is a non-interventional, multicenter, retrospective, patient registry designed to collect real-world data from patients treated with daptomycin between 2006 and 2012. Clinical outcomes were assessed at the end of daptomycin treatment for three dose groups: ≤6, >6 to <8, and ≥8 mg/kg/day. Safety was assessed for up to 30 days post-daptomycin treatment.
Results
Of the 6075 patients enrolled in EU-CORE, 4892 patients received daptomycin doses ≤6 mg/kg/day, while 1097 patients received high doses (>6 mg/kg/day). The primary infections with the largest proportion of patients treated with a high dose (>6 mg/kg/day) were osteomyelitis (37.1%), foreign body/prosthetic infection (31.6%), and endocarditis (27.6%). S. aureus was identified in 42.9% of patients with positive cultures treated with either ≤6 or >6 mg/kg/day. The overall clinical success rate was 82.0% (899/1097) with high doses (>6 mg/kg/day) and 80.3% (3928/4890) with doses ≤6 mg/kg/day. Numerically higher clinical success rate was observed for endocarditis and foreign body/prosthetic infection, as well as for coagulase-negative staphylococcal and enterococcal infections, with high-dose daptomycin treatment. There were no new or unexpected safety findings at doses >6 mg/kg/day.
Conclusion
These results suggested that daptomycin at doses >6 mg/kg/day was effective and well tolerated. High-dose daptomycin is a potential therapeutic option in patients with difficult-to-treat Gram-positive infections.
Funding
This study was funded by Novartis Pharma AG.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Daptomycin is a cyclic lipopeptide antibiotic that is approved at a dose of 4 mg/kg/day for the treatment of complicated skin and soft tissue infections (cSSTI) and at a dose of 6 mg/kg/day for the treatment of Staphylococcus aureus right-sided infective endocarditis (RIE) and bacteremia associated with cSSTI and RIE [1]. It has a rapid, concentration-dependent bactericidal activity [2]. Daptomycin exhibits a linear pharmacokinetic profile, which is concentration proportional for doses up to 12 mg/kg/day [2–4]. As a result of its concentration-dependent bactericidal activity, higher doses (>6 mg/kg/day) have often been considered for difficult-to-treat infections [5].
Daptomycin is a recognized alternative to vancomycin for the treatment of serious infections [6]. Optimum dosing of daptomycin for serious/deep-seated infection is not yet defined. Based on its concentration-dependent activity, higher doses of daptomycin may lead to a more rapid bacterial clearance and reduce resistance emergence [5, 7]. In infections where there is a difficulty in achieving adequate local antibiotic concentration at the infection site, or in patients with sepsis and high volumes of distribution, high-dose daptomycin may also be advantageous [4, 8]. Additionally, the intrinsic lower susceptibility of enterococci to daptomycin suggests that high doses of daptomycin may be necessary to achieve good results [9].
Several national and international treatment guidelines include high-dose daptomycin (8–10 mg/kg/day) as a therapeutic option for difficult-to-treat infections [6, 10–12]. The Infectious Diseases Society of America guidelines for the treatment of methicillin-resistant S. aureus (MRSA) recommend consideration of high-dose (10 mg/kg/day) daptomycin in patients with persistent MRSA bacteremia associated with vancomycin failure and possible endocarditis [6]. Also, the Spanish Society of Infectious Diseases and Clinical Microbiology has recently addressed the impact of an appropriate antimicrobial treatment (empirical and directed) and its relevance in the management of S. aureus infections, and the role of high-dose daptomycin in these scenarios [11].
Although only a limited number of clinical trials have evaluated the safety of high-dose daptomycin [5, 13, 14], several observational studies have reported the successful use of high-dose daptomycin (>6 mg/kg/day) in patients with difficult-to-treat infections [7–9, 15–18]. To date, there are no randomized controlled studies that have evaluated the efficacy and safety of high-dose daptomycin. The objective of this analysis from the European Cubicin® Outcomes Registry and Experience (EU-CORE) study was to evaluate the effectiveness and safety of daptomycin for the treatment of Gram-positive infections in a real-world setting with high doses (>6 mg/kg/day) versus current licensed doses (4 or 6 mg/kg/day).
Methods
Study Design and Data Collection
This was a non-interventional, multicenter, retrospective, patient registry designed to collect real-world outcome data on patients who had received at least one dose of daptomycin for the treatment of a serious Gram-positive bacterial infection. This analysis includes all patients enrolled in the EU-CORE study who received any dose of daptomycin between 2006 and 2012: ≤6 mg/kg/day, >6 to <8 mg/kg/day, or ≥8 mg/kg/day.
Detailed EU-CORE methodology has been described elsewhere [19]. Patients who had received at least one dose of daptomycin, had a minimum of 30 days post-treatment follow-up for safety monitoring, and were not part of a controlled clinical trial, were included. Daptomycin treatment was considered to be first line when there was no prior use of antibiotics and second line if prior antibiotics were used. In this analysis, patients treated with doses >6 mg/kg/day of daptomycin were compared with doses ≤6 mg/kg/day.
Institutional Review Board (IRB) or Ethics Committee (EC) approval in each country was obtained before the start of the study and all procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964, as revised in 2013, and Good Clinical Practices. Informed consent was obtained according to the requirements of the IRB or EC and/or local data privacy regulations.
Effectiveness
Investigators assessed clinical outcomes at the end of daptomycin therapy according to protocol-defined criteria: cured (clinical signs and symptoms resolved, no additional antibiotic therapy was necessary, or infection cleared with a negative culture reported); improved (partial resolution of clinical signs and symptoms and/or additional antibiotic therapy was warranted); failed (inadequate response to daptomycin therapy, worsening or new/recurrent signs and symptoms, need for a change in antibiotic therapy, or positive culture reported at the end of therapy); or non-evaluable (unable to determine response due to insufficient information) [20]. Clinical success was defined as cured or improved outcomes.
Safety
Adverse events (AEs) and serious AEs (SAEs) during daptomycin treatment and the 30-day follow-up period were assessed by the investigators. All reported AEs were recorded, regardless of their relationship to daptomycin.
Statistical Analysis
Statistical analysis was performed using SAS version 9.3 (SAS Institute Inc., Cary, NC, USA). Due to the nature of the trial, inferential analyses were not conducted and no formal statistical methodology other than simple descriptive statistics was used. All analyses were considered to be explanatory.
Numerical variables are summarized as arithmetic mean, standard deviation, median, minimum, first quartile, third quartile, and maximum for continuous variables. Categorical variables were summarized by absolute and relative frequencies.
Simple and multiple post-hoc logistic regression analyses were performed to determine whether any factors could have influenced success rates. These factors included the dose level, duration of treatment, bacteremia, comorbidity, use of prior or concomitant medication, and surgery.
Results
Patient Demographic and Clinical Characteristics
Of the 6075 patients enrolled in EU-CORE study, 4892 (80.5%) patients received daptomycin doses ≤6 mg/kg/day, 1097 (18.1%) patients received daptomycin doses >6 mg/kg/day, and dosing information was missing for 86 patients. Baseline demographics, clinical characteristics, and significant underlying diseases are summarized in Table 1. The three dose groups (≤6; >6 to <8; ≥8 mg/kg/day) were similar in terms of age, gender, race, and underlying conditions. Of the 5989 patients with reported dosing information, 782 (13.1%) had a baseline creatinine clearance (CrCl) <30 mL/min. Overall, 542 (9.0%) patients were on dialysis at initiation of daptomycin therapy and 526 (8.8%) patients at end of daptomycin therapy. The proportion of patients with CrCl <30 mL/min who received daptomycin every 48 h was 44.0%, while 47.0% received daptomycin every 24 h. Among patients undergoing continuous renal replacement therapy (CRRT), 77.2% received daptomycin every 24 h.
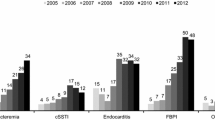
The primary infections with the largest proportion of patients treated with a high dose (>6 mg/kg/day) were osteomyelitis (37.1%), foreign body/prosthetic infection (31.6%; mainly orthopedic, intracardiac and intravascular devices), and endocarditis (27.6%; Fig. 1).
Initial daptomycin dose by type of primary infection. cSSTI complicated skin and soft tissue infection, uSSTI uncomplicated skin and soft tissue infection. a Septic arthritis, urinary tract infection/pyelonephritis, necrotizing infections, necrotizing fasciitis, surgical/non-surgical antibiotic prophylaxis, neutropenic fever, metastatic abscess, central nervous system infection, not otherwise specified
A trend towards use of higher doses over time was observed, most notably in the treatment of osteomyelitis and foreign body/prosthetic infection (Fig. 2). Median duration of daptomycin therapy was 10 (range 1–246), 13 (range 1–300), and 14 (range 1–132) days in the ≤6, >6 to <8, and ≥8 mg/kg/day groups, respectively. The duration of daptomycin therapy was longer in patients with osteomyelitis, endocarditis, and foreign body/prosthetic infection.
Previous and Concomitant Antibiotic Therapy
The majority of patients received other antibiotic therapy before daptomycin in the three dose groups. The most common antibiotics administered prior to daptomycin were penicillins and glycopeptides (Table 1). Treatment failure was the most common reason for discontinuation of prior antibiotic therapy (see Appendix 1 in the supplementary material).
Most inpatients received concomitant antibiotics with daptomycin in the three dose groups; those most commonly used for inpatients included carbapenems, penicillins, and cephalosporins.
Microbiology
Culture samples were available for 4017 (82.1%), 383 (84.7%) and 577 (89.5%) patients treated with ≤6, >6 to <8, and ≥8 mg/kg/day, respectively. Approximately, two-thirds of patients had a positive culture and S. aureus was the most commonly isolated organism (Table 2). Patients in high-dose groups had higher percentage of positive cultures.
Effectiveness
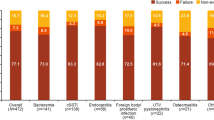
The overall clinical success rate (i.e., cured or improved) at the end of daptomycin therapy was 82.0% (899/1097) in the high-dose (>6 mg/kg/day) group and 80.3% (3928/4890) in the ≤6 mg/kg/day dose group. The clinical success rates were consistent for patients with positive cultures and those with negative cultures or no culture data. At 30 days, the clinical success rate was 91.3% (325/356) in the high-dose (>6 mg/kg/day) group and 90.3% (633/701) in the ≤6 mg/kg/day dose group. Numerically higher clinical success rates were observed with increasing dose for several infections, including endocarditis and foreign body/prosthetic infection (Table 3). Clinical outcome was also higher in coagulase-negative staphylococcal and enterococcal infections (Fig. 3). The clinical success rates tended to be greater when daptomycin was used as first-line therapy [82.1% (≤6 mg/kg), 85.1% (>6 to <8 mg/kg), and 87.9% (≥8 mg/kg)] as compared to second-line therapy [79.2% (≤6 mg/kg), 77.1% (>6 to <8 mg/kg), and 81.9% (≥8 mg/kg)].
Results of both simple and multiple logistic regression analyses were consistent. As shown in Fig. 4, the clinical success rate was greater in patients treated with daptomycin doses ≥8 mg/kg/day as compared to ≤6 mg/kg/day, those who were treated for >28 days as compared to <14 days, and patients who underwent surgery as compared to no surgery. All the corresponding odds ratios were larger than 1.
Safety
AEs, SAEs, and 30-day overall mortality rate and mortality due to infections are presented in Table 4. Musculoskeletal and connective tissue disorders were reported as AEs in 25 (0.4%) patients. A total of 12 cases were considered by the investigator as related to daptomycin and among them 5 were SAEs (Table 4). Rhabdomyolysis and myalgia were the most reported AEs possibly related to daptomycin in the ≤6 mg/kg/day group or with higher doses of daptomycin but they were rare (0.2%). Discontinuation of daptomycin was observed in all patients with AEs of musculoskeletal and connective tissue disorders reported as related to daptomycin.
Rates of AEs leading to daptomycin discontinuation were similar between dose groups (4.0, 5.1, and 4.7% in the ≤6, >6 to <8, and ≥8 mg/kg/day groups, respectively). There were no new or unexpected safety findings at doses >6 mg/kg/day and no episodes of eosinophilic pneumonia were reported.
Increased blood creatine phosphokinase (CPK) was reported as an AE in 49 (1.0%), 9 (2.0%), and 18 (2.8%) patients in the ≤6, >6 to <8, and ≥8 mg/kg/day groups, respectively (Table 4). Shift to worsening CrCl from initiation to end of daptomycin therapy by daptomycin dose was 10.4% or less for all dose ranges [≤6 mg/kg/day (8.2%), >6 and <8 mg/kg/day (10.4%), ≥8 mg/kg/day (9.5%)].
A total of 782 (13.1%) patients had a baseline CrCl <30 mL/min. Overall AEs or SAEs were reported at greater frequencies in patients with CrCl <30 mL/min who received daptomycin every 24 h as compared with the total patient population (Table 4). There were no safety signals associated with daptomycin therapy in these patients.
Discussion
High-dose daptomycin treatment has been recommended for difficult-to-treat Gram-positive infections within various national and international guidelines [6, 10–12]. Although higher doses of daptomycin are increasingly prescribed for severe infections [21], there are a few studies evaluating the effectiveness of high dose. Patients within EU-CORE were more likely to be treated with high-dose daptomycin, if they had bacteremia, endocarditis, osteomyelitis, or foreign body/prosthetic infection. Numerically higher clinical success rate was observed with increasing dose in endocarditis and foreign body/prosthetic infection. As previously observed, success rates were higher when daptomycin was used as first-line treatment and this difference was observed across all dosing groups [22]. Logistic regression analyses revealed that factors contributing to statistically significant higher success rates were dose level (≥8 mg/kg/day), treatment duration (>28 days), and surgery. The higher success rate in patients treated for >28 days could be explained by the management of serious infections (e.g., osteomyelitis, endocarditis, and foreign body/prosthetic infection) that typically require a prolonged course of antibiotic therapy.
Effectiveness and safety of high-dose daptomycin treatment has been observed in several difficult-to-treat infections including mediastinitis after cardiac surgery and osteomyelitis [23, 24]. A retrospective evaluation of the effectiveness and safety of high-dose daptomycin therapy (>6 mg/kg/day) in a multicenter cohort of 245 adult patients with enterococcal infections showed that high-dose daptomycin was effective and safe [9]. Furthermore, high-dose daptomycin was shown to be an effective and safe alternative to standard-of-care in the treatment of left-sided endocarditis due to common Gram-positive pathogens [15, 16]. The clinical success rates observed in the present study for endocarditis are consistent with results from these prior studies where numerically higher clinical success rates were observed with daptomycin doses of at least 8 mg/kg/day.
Daptomycin exhibits concentration-dependent activity [3] and higher doses of daptomycin are associated with increased degree and speed of bactericidal activity [25]. Furthermore, since Enterococcus species usually have higher minimum inhibitory concentrations (MICs) of daptomycin than other Gram-positive pathogens, patients with enterococcal infections may require higher doses of daptomycin. Increasing prevalence and recognition of reduced susceptibility to glycopeptides amongst staphylococcal species is another reason why high-dose daptomycin can be a therapeutic option of interest. Others have argued that in view of the variability in pharmacokinetic parameters, MIC distribution and pharmacodynamics of important bacterial populations, high-dose daptomycin (10 mg/kg/day) could enhance clinical outcomes in complex infections [26]. In a retrospective cohort case–control analysis, clinical outcomes of patients treated with either daptomycin (8–10 mg/kg/day) or vancomycin for MRSA bacteremia with vancomycin MIC values ≥1.5 mg/L were compared [17]. The daptomycin group had a favorable outcome at 14 and 30 days. In agreement with this study, our results confirm that daptomycin doses >6 mg/kg/day with a median duration of approximately 14 days was effective in the treatment of patients with infections including endocarditis, bacteremia, skin and soft tissue infection, foreign body/prosthetic infection, and osteomyelitis. In patients with renal impairment (CrCl < 30 mL/min), the recommended dosing interval for daptomycin is every 48 h. Unlike in patients undergoing hemodialysis or continuous ambulatory peritoneal dialysis, the pharmacokinetics of daptomycin in patients receiving CRRT are similar to healthy volunteers. Therefore, dosing frequency in these patients should be every 24 h [27, 28], as reported in this registry where daptomycin was given every 24 h in 77.8% of patients on CRRT.
This study showed that daptomycin at doses >6 mg/kg/day was well tolerated with no new or unexpected safety findings. CPK elevation is a well-known safety signal for daptomycin, and baseline and weekly CPK measurement is recommended during high-dose daptomycin therapy. A few studies have reported CPK elevations that led to drug discontinuation due to musculoskeletal symptoms or related AEs [13, 14, 25], while others have reported high-dose daptomycin to be safe with no elevations of CPK [29, 30]. Fowler et al. [31] observed CPK elevations of 6.7% for daptomycin (6 mg/kg every 24 h) versus 0.9% for the comparator group in the complex S. aureus bacteremia and endocarditis study. In contrast, CPK elevations observed in the current study in patients treated with high-dose daptomycin were lower (2.0% and 3.0% in the >6 to <8 and ≥8 mg/kg/day groups, respectively). Muscular events were rare and seemed to be similar in the three dose groups.
The limitations of this study include the inherent limitations that are expected in real-world observational studies, which include non-randomized trial design, non-blinded interpretation of results, and the subjective assessments of clinical outcomes as determined by individual investigators.
Conclusion
High-dose daptomycin (>6 mg/kg/day) exhibited good safety and tolerability profile. These findings, combined with high success rates in a large spectrum of infections and against a wide range of pathogens, suggest that high-dose daptomycin (>6 mg/kg/day) is a therapeutic option in patients with difficult-to-treat infections and may provide better outcome for some infections.
References
Novartis Europharm Ltd. Cubicin® (daptomycin) summary of product characteristics. 2012.
Safdar N, Andes D, Craig WA. In vivo pharmacodynamic activity of daptomycin. Antimicrob Agents Chemother. 2004;48(1):63–8.
Benvenuto M, Benziger DP, Yankelev S, Vigliani G. Pharmacokinetics and tolerability of daptomycin at doses up to 12 milligrams per kilogram of body weight once daily in healthy volunteers. Antimicrob Agents Chemother. 2006;50(10):3245–9.
Gould IM, Miro JM, Rybak MJ. Daptomycin: the role of high-dose and combination therapy for Gram-positive infections. Int J Antimicrob Agents. 2013;42(3):202–10.
Wu G, Abraham T, Rapp J, Vastey F, Saad N, Balmir E. Daptomycin: evaluation of a high-dose treatment strategy. Int J Antimicrob Agents. 2011;38(3):192–6.
Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin Infect Dis. 2011;52(3):285–92.
Bassetti M, Nicco E, Ginocchio F, Ansaldi F, de Florentiis D, Viscoli C. High-dose daptomycin in documented Staphylococcus aureus infections. Int J Antimicrob Agents. 2010;36(5):459–61.
Falcone M, Russo A, Venditti M, Novelli A, Pai MP. Considerations for higher doses of daptomycin in critically ill patients with methicillin-resistant Staphylococcus aureus bacteremia. Clin Infect Dis. 2013;57(11):1568–76.
Casapao AM, Kullar R, Davis SL, et al. Multicenter study of high-dose daptomycin for treatment of enterococcal infections. Antimicrob Agents Chemother. 2013;57(9):4190–6.
Mensa J, Barberan J, Llinares P, et al. Guidelines for the treatment on infections caused by methicillin-resistant Staphylococcus aureus. Rev Esp Quimioter. 2008;21(4):234–58.
Gudiol F, Aguado JM, Almirante B, et al. Diagnosis and treatment of bacteremia and endocarditis due to Staphylococcus aureus. A clinical guideline from the Spanish Society of Clinical Microbiology and Infectious Diseases (SEIMC). Enferm Infecc Microbiol Clin. 2015;33(9):625.e1–625.e23.
Gudiol F, Aguado JM, Pascual A, et al. Consensus document for the treatment of bacteremia and endocarditis caused by methicillin-resistent Staphylococcus aureus. Sociedad Espanola de Enfermedades Infecciosas y Microbiologia Clinica. Enferm Infecc Microbiol Clin. 2009;27(2):105–15.
Katz DE, Lindfield KC, Steenbergen JN, et al. A pilot study of high-dose short duration daptomycin for the treatment of patients with complicated skin and skin structure infections caused by gram-positive bacteria. Int J Clin Pract. 2008;62(9):1455–64.
Figueroa DA, Mangini E, Amodio-Groton M, et al. Safety of high-dose intravenous daptomycin treatment: three-year cumulative experience in a clinical program. Clin Infect Dis. 2009;49(2):177–80.
Murray KP, Zhao JJ, Davis SL, et al. Early use of daptomycin versus vancomycin for methicillin-resistant Staphylococcus aureus bacteremia with vancomycin minimum inhibitory concentration >1 mg/L: a matched cohort study. Clin Infect Dis. 2013;56(11):1562–9.
Carugati M, Bayer AS, Miro JM, et al. High-dose daptomycin therapy for left-sided infective endocarditis: a prospective study from the international collaboration on endocarditis. Antimicrob Agents Chemother. 2013;57(12):6213–22.
Cheng CW, Hsu PC, Yang CC, et al. Influence of early daptomycin therapy on treatment outcome of methicillin-resistant Staphylococcus aureus bacteraemia with high vancomycin minimum inhibitory concentrations. Int J Antimicrob Agents. 2013;41(3):293–4.
Kullar R, Davis SL, Levine DP, et al. High-dose daptomycin for treatment of complicated gram-positive infections: a large, multicenter, retrospective study. Pharmacotherapy. 2011;31(6):527–36.
Gonzalez-Ruiz A, Beiras-Fernandez A, Lehmkuhl H, Seaton RA, Loeffler J, Chaves RL. Clinical experience with daptomycin in Europe: the first 2.5 years. J Antimicrob Chemother. 2011;66(4):912–9.
Dohmen PM, Guleri A, Capone A, et al. Daptomycin for the treatment of infective endocarditis: results from a European registry. J Antimicrob Chemother. 2013;68(4):936–42.
Parra-Ruiz J, Pena-Monje A, Tomas-Jimenez C, Pomares-Mora J, Hernandez-Quero J. Efficacy and safety of high dose (>/=8 mg/kg/day) daptomycin. Enferm Infecc Microbiol Clin. 2011;29(6):425–7.
Konychev A, Heep M, Moritz RK, et al. Safety and efficacy of daptomycin as first-line treatment for complicated skin and soft tissue infections in elderly patients: an open-label, multicentre, randomized phase IIIb trial. Drugs Aging. 2013;30(10):829–36.
Weis F, Heyn J, Hinske CL, et al. Daptomycin as supportive treatment option in patients developing mediastinitis after open cardiac surgery. J Cardiothorac Surg. 2012;7:81.
Lamp KC, Friedrich LV, Mendez-Vigo L, Russo R. Clinical experience with daptomycin for the treatment of patients with osteomyelitis. Am J Med. 2007;120(10 Suppl 1):S13–20.
Moise PA, Hershberger E, Amodio-Groton MI, Lamp KC. Safety and clinical outcomes when utilizing high-dose (> or = 8 mg/kg) daptomycin therapy. Ann Pharmacother. 2009;43(7):1211–9.
Soon RL, Turner SJ, Forrest A, Tsuji BT, Brown J. Pharmacokinetic/pharmacodynamic evaluation of the efficacy and safety of daptomycin against Staphylococcus aureus. Int J Antimicrob Agents. 2013;42(1):53–8.
Corti N, Rudiger A, Chiesa A, et al. Pharmacokinetics of daily daptomycin in critically ill patients undergoing continuous renal replacement therapy. Chemotherapy. 2013;59(2):143–51.
Khadzhynov D, Slowinski T, Lieker I, et al. Plasma pharmacokinetics of daptomycin in critically ill patients with renal failure and undergoing CVVHD. Int J Clin Pharmacol Ther. 2011;49(11):656–65.
King EA, McCoy D, Desai S, Nyirenda T, Bicking K. Vancomycin-resistant enterococcal bacteraemia and daptomycin: are higher doses necessary? J Antimicrob Chemother. 2011;66(9):2112–8.
De Rosa FG, Mollaretti O, Cometto C, Pagani N, Montrucchio C, Di Perri G. Early experience with high-dosage daptomycin for prosthetic infections. Clin Infect Dis. 2009;49(11):1772–3.
Fowler VG Jr, Boucher HW, Corey GR, et al. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med. 2006;355(7):653–65.
Acknowledgments
Sponsorship, article processing charges, and the open access charge for this study were funded by Novartis Pharma AG. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship of this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval to the version to be published. These data were presented in part at the 25th European Congress of Clinical Microbiology and Infectious Diseases, Copenhagen, Denmark, 2015 (abstract 1448). Medical writing support was provided by Jitendriya Mishra (Novartis Healthcare Pvt. Ltd., Hyderabad, India), Sai Krishnaveni Chevooru (Novartis Healthcare Pvt. Ltd., Hyderabad, India), and Farid Khalfi (Novartis Ireland Ltd., Dublin, Ireland). Funding for writing support was provided by Novartis Pharma AG.
Disclosures
R Andrew Seaton received consultancy fees and honoraria for speaking at Novartis sponsored symposia. Francesco Menichetti has nothing to declare. Georgios Dalekos has nothing to declare. Andres Beiras-Fernandez received fees for speakers panels from Orion Pharma, Novartis, Fresenius, Neovii, Merck, and Sanofi-Aventis. He is also a member of the SAB Sanofi-Aventis. Francisco Nacinovich has nothing to declare. Rashidkhan Pathan is an employee of Novartis Healthcare Pvt. Ltd. Kamal Hamed is an employee of Novartis Pharmaceuticals Corporation.
Compliance with ethics guidelines
The study has been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The protocol was approved by the health authority and the Institutional Review Board (IRB) or Ethics Committee (EC) in each country and written informed consent was obtained according to the requirements of the IRB or EC and/or the local data privacy regulations.
Open Access
This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Seaton, R.A., Menichetti, F., Dalekos, G. et al. Evaluation of Effectiveness and Safety of High-Dose Daptomycin: Results from Patients Included in the European Cubicin® Outcomes Registry and Experience. Adv Ther 32, 1192–1205 (2015). https://doi.org/10.1007/s12325-015-0267-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-015-0267-4