Abstract
Introduction
Propensity score analysis of midterm outcomes after isolated aortic valve replacement through right anterior mini-thoracotomy and partial upper sternotomy could provide information about the most beneficial minimally invasive technique for the patient based on the preoperative risk factors.
Methods
Between March 2015 and February 2021, 694 minimally invasive isolated aortic valve surgeries were performed at our institution. Among these, 441 right anterior mini-thoracotomies and 253 partial upper sternotomies were performed. A propensity score analysis was performed in 202 matched pairs.
Results
Cardiopulmonary bypass time and cross-clamp time were significantly shorter in the right anterior mini-thoracotomy group than in the partial upper sternotomy group (p = 0.001 and p < 0.001, respectively). Time to first mobilization and hospital stay were significantly shorter in the right anterior mini-thoracotomy group than in the partial upper sternotomy group (p = 0.005, p = 0.001, respectively). A significantly lower incidence of revision surgery was noted in the right anterior mini-thoracotomy group than in the partial upper sternotomy group (p = 0.046). No significant differences in 30-day mortality (p = 1.000) and 1-year mortality (p = 0.543) were noted. Kaplan-Meier survival estimates were 96.3% in the right anterior mini-thoracotomy group and 92.7% in the partial upper sternotomy group after 4 years (log rank 0.169), respectively.
Conclusions
Despite the technical challenges, right anterior mini-thoracotomy can be chosen as first-line strategy for isolated aortic valve replacement. For patients unsuitable for this technique, the partial upper sternotomy remains a safe method that can be performed by a wide range of surgeons.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Over the past 30 years, cardiac surgery has increasingly used minimally invasive procedures (MICs), reducing the surgical body trauma and achieving early recovery for the patient |
Innovative approaches to minimally invasive aortic valve replacement (MIC-AVR) such as right anterior mini-thoracotomy (RAMT) and partial upper sternotomy (PUS) concentrate on smaller surgical approaches providing better cosmetic results and shorter hospital stay |
Comparing the perioperative and midterm results of MIC-AVR through RAMT versus PUS can help in making decisions about the most favorable approach for the patient based on their preoperative condition |
The mid-term outcomes of this study showed similar perioperative morbidity and mortality between RAMT and PUS techniques with comparable 4-year estimated survival in both groups |
The RAMT technique is a safe and feasible approach to isolated AVR that does not compromise the surgical quality, postoperative outcome, or patient safety when performed by a team experienced in MIC techniques in a large-volume center |
The PUS procedure provides the best surgical access in MIC-AVR and can be performed by a wide range of surgeons |
Introduction
Over the past 30 years, cardiac surgery has increasingly used minimally invasive procedures (MIC) with the aim of reducing surgical body trauma and achieving early recovery for the patient [1,2,3]. The perioperative mortality rate in isolated aortic valve replacement (AVR) has decreased from 3.9 to 1.9% according to the database of the Society of Thoracic Surgeons (STS) [4,5,6]. Various techniques for MIC-AVR have been established and further developed and compared to median sternotomy (MS) [7, 8]. The innovative MIC-AVR techniques focus on smaller surgical approaches with faster postoperative recovery, which leads to shorter hospital stays [9]. MIC approaches mainly include partial upper sternotomy (PUS) and right anterolateral mini-thoracotomy (RAMT), with considerable advantages compared to MS [10,11,12]. So far, PUS has been the most used MIC access for surgical AVR, allowing better maintenance of the thoracic respiratory mechanics and early mobilization with attractive cosmetic results compared to MS [9, 13,14,15]. The RAMT procedure is also superior to MS, with a lower transfusion rate, shorter duration of mechanical ventilation, lower stroke rate, and shorter length of intensive care and hospital stay [16, 17].
The aim of this study is to compare the perioperative and midterm results of isolated AVR using MIC techniques with RAMT versus PUS.
Methods
Compliance with Ethics Guidelines
The study was approved by the ethics committee of the Medical Association of North Rhine in Germany (id. no. 82/2021), and individual patient consent for the study was waived. The study was performed in accordance with the Declaration of Helsinki 1964 and its later amendments.
Study Design and Data Source
This is a retrospective study, and all perioperative data were prospectively collected from the clinic's internal databases. All patients with isolated aortic valve stenosis, insufficiency, and/or endocarditis who underwent MIC-AVR were included in this study. Exclusion criteria were reoperation, aortic valve reconstruction, and need for additional cardiosurgical procedure.
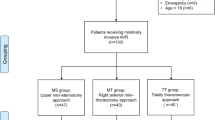
Between March 2015 and February 2021, 768 patients underwent MIC-AVR at our institution. Of those, 63 patients (42 in the RAMT group and 21 in the PUS group) underwent AKE in combination with Morrow resection. Aortic valve reconstruction was performed in seven patients in the PUS group, and four additional patients in the PUS group were excluded from the study because of previous AKE. After application of the inclusion and exclusion criteria, final data from 694 patients were used for statistical analysis. Of these, 441 (63.5%) patients were operated on using RAMT and 253 (36.5%) patients via PUS. After propensity score analysis, 202 matched patients from the RAMT group and 202 from the PUS group were evaluated (Fig. 1).
At the start of the study, PUS was the standard surgical access for isolated AVR surgery in our department. Since March 2017, RAMT has become the first-line strategy for the surgical treatment of all isolated aortic valve pathologies (Supplementary Material). During this period of study, preoperative computer tomography (CT) of the aorta was performed in all patients who required an isolated AVR to determine whether the patient was suitable for the RAMT procedure [18]. In cases of severe calcification of the aorta or pelvic arteries, severe anatomical conditions, or strong adhesions of the lungs, PUS serves as an alternative surgical access benefiting from considerable advantages of the MIC techniques [9, 13,14,15].
The primary endpoint was 30-day mortality. Secondary endpoints were cardiopulmonary bypass (CPB) time and aortic clamping time, conversion to MS, surgical revision, postoperative respiratory failure, hemodialysis, wound healing disorders, length of intensive care and hospital stay, and estimated survival at 4 years.
Follow-Up
Discharged patients were assessed directly in our outpatient clinic after 3 months and yearly thereafter or were contacted by mail or telephone interview during a 3-month late closing interval ending in April 2021 with completion of 98%. Telephone contact with the patients was used to assess the current physical condition and to request further cardiovascular interventions and check-ups by the referring cardiologists and family doctors, who were also contacted if additional information was needed.
Operative Technique
All patients were intubated with a single-lumen endotracheal tube, and transesophageal echocardiography (TEE) was performed in all patients for monitoring heart and valve function during the operation.
A detailed description of the PUS technique has been published previously [9]. The surgical access was performed through a 5–6-cm skin incision followed by J-shaped upper mini-sternotomy at the fourth intercostal space. A central cannulation for CPB through the ascending aorta and right atrium was preferred by most of this patient group (Fig. 2).
Operative site of an aortic valve replacement via the partial superior sternotomy. A For subanular insertion of Ethibon 2–0 U-sutures. B Inserting the U-sutures into the Magna Ease® aortic valve prosthesis. C After closure of the aorta. D Immediately after wound closure. This figure was created entirely by the authors for this article, has never been published before, and was not based in whole or in part on a previously published figure
For the RAMT group, the access was performed through a 3–5-cm skin incision at the second or third right intercostal space. A detailed description of the surgical technique has been published previously [3]. Cannulation for CPB was achieved through the common femoral artery in 436 (98.7%) patients and the common femoral vein in all patients in the unmatched cohort. Additional cannulation of the jugular vein was necessary in 102 (23.1%) patients. Femoral cannulation was performed primarily surgically through a 2-cm skin incision below the inguinal ligament. Since January 2019, percutaneous femoral cannulation for CPB guided by ultrasound sonography was performed in combination with the use of a vascular closure device (VCD) MANTA™ (Essential Medical Inc., Malvern, PA, USA) for femoral artery closure. Except for the automated suturing with Cor-Knot® (Cor-Knot®, LSI Solutions, USA), there is no difference between AVR using RAMT and the traditional sternotomy approach (Fig. 3).
Operative setup of an aortic valve replacement via right anterior mini-thoracotomy. A Before implanting an Inspiris Resilia® aortic valve. B By implanting the aortic valve prosthesis. C Introduction of Cor-Knot® to fix the aortic valve prosthesis. D Femoral artery occlusion with MANTA® System. E Immediately after wound closure. This figure was created entirely by the authors for this article, has never been published before, and was not based in whole or in part on a previously published figure
Statistical Analysis
Propensity score matching was performed. The propensity score for each patient was calculated by logistic regression with adjustment for 12 key baseline variables: age, gender, body mass index (BMI), New York Heart Association score (NYHA) ≥ 3, hypertension, diabetes mellitus, prior stroke, acute endocarditis, urgent indication for surgery, left ventricular ejection fraction (LVEF), aortic valve stenosis, and European System for Cardiac Operative Risk Evaluation II (EuroSCORE II). When matching patients 1:1, a difference in propensity score of 0.7% (0.007) was tolerated.
Data on the matched and unmatched groups were analyzed using descriptive statistics, with categorical variables presented as absolute values and frequencies (%) and the continuous variables presented as the median and interquartile range (IQR) or mean ± standard deviation (SD). Comparisons between the RAMT and PUS groups were carried out using a t-test or Mann-Whitney U test for continuous variables as appropriate depending on distribution and a Fisher's exact or chi-square test for categorical variables. To test for normal distribution of the continuous variables, the Kolmogorov-Smirnov test was used. Cohen's d was used for the continuous variables with significant difference. A Cohen’s d value of > 0.8 indicates a major effect. Survival analyses were presented as Kaplan-Meier curves. In addition, hazard ratios (HRs) were calculated by Cox regression. In all cases, a two-tailed p value of < 0.05 was considered statistically significant. All statistical tests were performed using IBM SPSS Statistics software version 24.0 (IBM Corp., Armonk, NY, USA).
Results
Baseline characteristics and the results in the two patient groups are shown below for the entire patient collective as well as for the corresponding subgroup with propensity score matching.
Demographic Data of Unmatched Group
The demographic characteristics of the unmatched cohort are summarized in Table 1. Patients undergoing PUS technique were significantly older [71.7 (range 63.6–76.8) years] than those in the RAMT group [68.9 (range 60.7–75.3) years; p = 0.005]. There were further significant differences between the two groups concerning the creatinine value (p = 0.049), previous stroke (p = 0.009) or cardiogenic shock (p = 0.019), presence of coronary artery disease (p = 0.031), and NYHA class III/IV (p = 0.016). The EuroSCORE II was significantly higher in the PUS group (p < 0.001).
Demographic Data of Matched Groups
Regarding the age, comorbidities, and EuroSCORE II, there were no significant differences between the two matched subgroups. Apart from a preoperative aortic valve regurgitation (p = 0.036), no further significant differences were detected in the baseline characteristics (Table 2).
Intraoperative Data of Unmatched Groups
No intraoperative mortality was noted in either group. Despite a non-significant longer CPB time (p = 0.794) in the RAMT group, the operation time (p < 0.031) and the cross-clamping time (p = 0.003) were significantly shorter compared to the PUS group. One patient (0.1%; p = 1.000) in the all-patient cohort required an intraoperative conversion from a RAMT access to MS because of bleeding. Furthermore, significantly more biological valve protheses (98.9%; p = 0.001) with larger size (p = 0.001) were implanted in the RAMT group while aortic root enlargement was performed more in the PUS group (p = 0.026) (Table 3).
Intraoperative Data of Matched Groups
After propensity score analysis, the median operation time (p < 0.001) and median cross-clamping time (p < 0.001) remained significantly shorter in the RAMT group, accounting 124.5 (range 97.0–164.8) min and 40.0 (range 31.0–59.3) min compared to the PUS group with 156.5 (range 130.0–185.0) min and 56.0 (range 47.0–63.0) min, respectively, while the CPB time became significantly shorter in the RAMT group [69.5 (range 55.0–97.0) min vs. 79.0 (range 69.0–97.3) min in PUS, p < 0.001]. Regarding the type (p = 0.048) and size (p < 0.001) of the implanted valve, the difference between the two matched cohorts was also significant without difference in terms of artic root enlargement anymore (p = 0.123). Intraoperative data are detailed in Table 4.
Postoperative Outcomes of Unmatched Groups
The outcomes of unmatched cohorts are summarized in Table 5. The intubation time was significantly shorter in the RAMT group (p < 0.001) accounting 6.0 (range 5.0–10.0) h compared to the PUS group with 10.0 (range 6.0–18) h. Surgical revision was significantly more frequent in the PUS group (9.5%) than in the RAMT group (5.0%) (p = 0.022), and the most common causes were bleeding followed by wound infection, tamponade, prosthesis dysfunction, low cardiac output syndrome (LCOS), and endocarditis. Surgical groin wound revision was noted only in the RAMT group in eight patients (1.8%; p = 0.030). Postoperative acute kidney injury (p = 0.013) and new occurrence of atrial fibrillation (p < 0.001) were significantly lower in the RAMT group affecting 2.3% and 15.4% compared to PUS group with 5.9% and 24.7%, respectively. The number of postoperatively administrated red cell concentrates was significantly lower in the RAMT group than in the PUS group (0.8 ± 2.0 vs. 1.0 ± 1.9; p = 0.020, respectively).
Further significant differences concerning the length of intensive stay, time of the first mobilization, and hospital stay were shown and were lower in the RAMT group [median intensive stay: 1.0 (range 1–2) vs. 2.0 (range 1.0–3.0) days in PUS, p = 0.003; median time to first mobilization: 3.0 (range 2–3) vs. 3.0 (range 2–4) days in PUS, p < 0.001; median hospital stay: 9.0 (range 8 to 12) vs. 11 (range 9–14) days in PUS, p < 0.001].
Thirty-day and 1-year mortality showed no significant difference between the RAMT and PUS group and was 0.9% vs. 1.2% (p = 0.710) and 1.9% vs. 2.8% (p = 0.246), respectively. In the overall patient collective, the estimated survival rates in the RAMT group were significantly higher at 98.6% after 1 year, 97.6% after 2 years, and 97.0% after 3 and 4 years (log rank 0.018) compared with the PUS group (97.2% after 1 year, 95.3% after 2 years, 92.6% at 3 years, and 92.0% after 4, 5, and 6 years, Fig. 4A).
Kaplan-Meier curve for survival after aortic valve replacement. A Unmatched group; B matched group. HR calculated by Cox regression was 0.401 (95% CI 0.183–0.879) for the unmatched and 0.530 (95% CI 0.211–1.330) for the matched group. PUS, partial upper sternotomy; RAMT, right anterior mini-thoracotomy
Postoperative Outcomes of the Matched Groups
Other than persistence of significantly shorter intubation time (p = 0.001), first mobilization time (p = 0.005), and length of hospital stay (p = 0.001) in the RAMT group after the propensity matching, no other significant differences in the postoperative outcomes or 30-day and 1-year mortality were noted between the two groups (Table 6). In the matched cohorts, the estimated survival rate after 4 years was 96.3% in the RAMT group, which was higher compared to the PUS group (92.7%; log rank 0.169) without a significant difference (Fig. 4B).
Discussion
The mid-term outcomes of this study showed similar perioperative morbidity and mortality between RAMT and PUS techniques with comparable 4-year estimated survival in both groups.
At our department, the final decision on the operative strategy for each patient requiring AVR was made individually during a preoperative medical staff meeting considering the patient's preoperative data such as age, comorbidities, vascular status, and EuroSCORE II. The choice of the surgical technique was a result of application of an internal policy recommendation, which was tailored to the individual patient. In our unmatched cohorts, the patients in the PUS group had significantly higher EuroSCORE II than the RAMT group, 2.8 ± 2.5 and 2.3 ± 2.4, respectively (p < 0.001). These preoperative data are similar to those reported in previous studies comparing the two subgroups [11, 18,19,20]. An increased EuroSCORE II is a predictive factor for a higher risk of intra- and postoperative complications as well as postoperative morbidity and mortality [20]. Accordingly, the PUS procedure for these patients was a safe surgical technique that could be performed by all cardiac surgeons to reduce the surgical risk. To avoid selection bias between the two subgroups, these differences in demographic characteristics were adjusted and eliminated by developing a propensity score matching in line with previous published studies comparing RAMT and PUS techniques in AVR [11, 19, 20].
In our cohort, MIC-AVR via RAMT was significantly less time-consuming than the PUS technique with significantly shorter mean operative, CPB, and cross-clamping times. Similar results have already been reported in other studies [18, 19]. In 2018, Mikus et al. revealed significant difference in terms of shorter CPB and cross-clamping time in the RAMT group compared to PUS group, (p < 0.001) and hypothesized that the high standardization of the procedure by an expert team could make RAMT as fast and effective as the PUS procedure [19]. However, other retrospective studies report significantly longer CPB and cross-clamping time in the RAMT compared to PUS group. They justified their results with the learning curve reflected in their studies, since both MIC techniques were performed by all surgeons [11, 20]. In our RAMT cohort, the operating times could be reduced by using a 3D camera, long surgical instruments, and an automated suture closure system [3]. Furthermore, RAMT was performed exclusively by surgeons with excellent surgical skills using minimally invasive surgery. We believe that effective and clear communication among the surgical team as well as a careful planning and preparation of the operation favors the reduction of operating times.
In our matched cohort, the median intubation time in the RAMT group was 4 h shorter with significantly shorter hospital stay than in the PUS group. The length of intensive care stay showed no significant difference in the matched groups. Miceli et al. reported the median intubation time was 1 h less in the RAMT group with significantly shorter intensive care and hospital stay [18]. Reducing the length of hospital stay is an important aspect of resource use, since intensive care and hospital stays are the main determinants of cost after cardiac surgery [21].
Contrary to previous studies, which reported a high incidence of conversion to MS with the RAMT procedure, we found no conversion to MS in either the RAMT or the PUS group after propensity score analysis [11, 20]. Furthermore, Semsroth and colleagues emphasize that the extension of the PUS to MS is uncomplicated, while the conversion from RAMT to MS is technically challenging [11, 20]. The absence of conversion to MS in our matched cohorts could be explained by the fact that the RAMT procedure was performed exclusively by experienced surgeons.
In our matched cohort, 10.9% of the patients in the PUS group and 5.4% of the patients in the RAMT group required surgical revision mostly because of bleeding, p = 0.046. Mikus et al. also reported an increased occurrence of tamponade requiring surgical revision with the PUS procedure compared to RAMT [19]. Further studies report no significant difference in terms of surgical revision due to bleeding when comparing these two MIC techniques [11, 18, 20]. The small number of surgical revisions in our RAMT group is due to the expertise of the surgeons in minimally invasive techniques and the tight perioperative coagulation management.
In the literature, intraoperative surgical complications such as an increased occurrence of stroke, vascular and wound complications related to surgical peripheral cannulation were correlated to the RAMT technique [11, 20, 22]. In contrast, groin wound complications occurred in three patients (1.5%) in the RAMT group without significant difference compared to PUS. Since January 2019, ultrasound-guided percutaneous femoral cannulation for CPB was used in our department in combination with the use of MANTA™ VCD to avoid surgical complications in the groin. MANTA™ is described as an effective, fast, and safe device, which also has a positive effect on the operating time compared to surgical access in cardiac surgery [23].
In terms of 30-day and 1-year mortality, there were no significant differences in our cohort when comparing the two matched groups (p = 1.000 and p = 0.543, respectively). The trend for estimated survival after 4 years was better in the RAMT group compared to the PUS group (96.3% vs. 92.7%, log rank 0.169). Miceli et al. also reported higher 1- and 5-year survival rates in the RAMT group than in the PUS group [18]. However, Semsroth et al. described a trend toward better survival rates with PUS, recording at the same time longer CPB and cross-clamping times in the RAMT group [11, 20]. In contrast, we noted significantly shorter operative, CPB, and cross-clamping times in our RAMT cohort, which also had reduced risks of postoperative morbidity and mortality, explaining the better estimated survival rates in this group [18, 22].
Study Limitation
The main limitation of our study is its retrospective and non-randomized design. Furthermore, the interpretation of Kaplan-Meier's survival rate may have been difficult because of the different follow-up periods of both groups due to the timing of the procedures. Besides, the generalizability of our findings is questionable, given the single-center design.
Conclusion
The RAMT is a safe and feasible technique for isolated AVR that does not compromise surgical quality, postoperative outcomes, or patient safety when performed by a team experienced in MIC techniques in a large-volume center. However, the PUS procedure provides the best surgical access in MIC-AVR that can be performed by a wide range of surgeons.
References
Cosgrove DM, Sabik JF. Minimally invasive approach for aortic valve operations. Ann Thorac Surg. 1996;62:596–7.
Wells WJ, Arroyo H, Bremner RM, et al. Homograft conduit failure in infants is not due to somatic outgrowth. J Thorac Cardiovasc Surg. 2002;124:88–96.
Bakhtiary F, El-Sayed Ahmad A, Amer M, et al. Video-assisted minimally invasive aortic valve replacement through right anterior minithoracotomy for all comers with aortic valve disease. Innovations (Phila). 2021;16:169–74.
Brown JM, O’Brien SM, Wu C, et al. Isolated aortic valve replacement in North America comprising 108,687 patients in 10 years: changes in risks, valve types, and outcomes in the Society of Thoracic Surgeons National Database. J Thorac Cardiovasc Surg. 2009;137:82–90.
Brennan JM, Edwards FH, Zhao Y, et al. Long-term survival after aortic valve replacement among high-risk elderly patients in the United States: insights from the Society of Thoracic Surgeons Adult Cardiac Surgery Database, 1991 to 2007. Circulation. 2012;126:1621–9.
Ghoreishi M, Thourani VH, Badhwar V, et al. Less-invasive aortic valve replacement: trends and outcomes from the society of thoracic surgeons database. Ann Thorac Surg. 2021;111:1216–23.
Nishimura RA, Carabello BA, Faxon DP, et al. ACC/AHA 2008 guideline update on valvular heart disease: focused update on infective endocarditis: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines: endorsed by the Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. Circulation. 2008;118:887–96.
Iribarne A, Easterwood R, Chan EY, et al. The golden age of minimally invasive cardiothoracic surgery: current and future perspectives. Future Cardiol. 2011;7:333–46.
Bakir I, Casselman FP, Wellens F, et al. Minimally invasive versus standard approach aortic valve replacement: a study in 506 patients. Ann Thorac Surg. 2006;81:1599–604.
Phan K, Xie A, Di Eusanio M, Yan TD. A meta-analysis of minimally invasive versus conventional sternotomy for aortic valve replacement. Ann Thorac Surg. 2014;98:1499–511.
Semsroth S, Matteucci-Gothe R, Heinz A, et al. Comparison of anterolateral minithoracotomy versus partial upper hemisternotomy in aortic valve replacement. Ann Thorac Surg. 2015;100:868–73.
Woo A, Williams WG, Choi R, et al. Clinical and echocardiographic determinants of long-term survival after surgical myectomy in obstructive hypertrophic cardiomyopathy. Circulation. 2005;111:2033–41.
Walther T, Falk V, Metz S, et al. Pain and quality of life after minimally invasive versus conventional cardiac surgery. Ann Thorac Surg. 1999;67:1643–7.
Gutermann H, Pettinari M, Van Kerrebroeck C, et al. Myectomy and mitral repair through the left atrium in hypertrophic obstructive cardiomyopathy: the preferred approach for contemporary surgical candidates. J Thorac Cardiovasc Surg. 2014;147:1833–6.
Candaele S, Herijgers P, Demeyere R, et al. Chest pain after partial upper versus complete sternotomy for aortic valve surgery. Acta Cardiol. 2003;58:17–21.
Del Giglio M, Mikus E, Nerla R, et al. Right anterior mini-thoracotomy vs. conventional sternotomy for aortic valve replacement: a propensity-matched comparison. J Thorac Dis 2018;10:1588–95.
Olds A, Saadat S, Azzolini A, et al. Improved operative and recovery times with mini-thoracotomy aortic valve replacement. J Cardiothorac Surg. 2019;14:91.
Miceli A, Murzi M, Gilmanov D, et al. Minimally invasive aortic valve replacement using right minithoracotomy is associated with better outcomes than ministernotomy. J Thorac Cardiovasc Surg. 2014;148:133–7.
Mikus E, Calvi S, Campo G, et al. Full sternotomy, hemisternotomy, and minithoracotomy for aortic valve surgery: is there a difference. Ann Thorac Surg. 2018;106:1782–8.
Semsroth S, Matteucci, Gothe R, et al. Comparison of two minimally invasive techniques and median sternotomy in aortic valve replacement. Ann Thorac Surg 2017;104:877–883.
Hamilton A, Norris C, Wensel R, Koshal A. Cost reduction in cardiac surgery. Can J Cardiol. 1994;10:721–7.
Murzi M, Cerillo AG, Miceli A, et al. Antegrade and retrograde arterial perfusion strategy in minimally invasive mitral-valve surgery: a propensity score analysis on 1280 patients. Eur J Cardiothorac Surg. 2013;43:e167–72.
El-Sayed Ahmad A, Bayram A, Salamate S, et al. Percutaneous versus surgical femoral access in minimally invasive cardiac operations. Eur J Cardiothorac Surg. 2022 Apr 20:ezab520.
Acknowledgements
Funding
Open Access funding was enabled and organized by Projekt DEAL. No funding or sponsorship was received for this study or publication of this article. The journal’s Rapid Service Fee was funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICME) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Conception and design: Saad Salamate, Ali El-Sayed Ahmad. Acquisition of data: Saad Salamate, Mohamed Amer. Analysis of data: Saad Salamate, Ali Bayram, Sami Sirat. Interpretation of data: Ali El-Sayed Ahmad, Farhad Bakhtiary. Drafting article: Saad Salamate, Ali Bayram. Final revision of the manuscript to be published: Farhad Bakhtiary, Mirko Doss, Ali El-Sayed Ahmad.
Disclosures
Farhad Bakhtiary discloses speakers’ honoraria and/or consulting fees from Edwards Lifesciences, LIS, and Abbott. Saad Salamate, Mohamed Amer, Sami Sirat, Ali Bayram, Mirko Doss, and Ali El-Sayed Ahmad all have nothing to disclose with regard to personal, financial, commercial, or academic conflicts of interest.
Compliance with Ethics Guidelines
The study was approved by the ethics committee of the Medical Association of North Rhine, Germany (id. no. 2019201/2019), and individual patient consent for the study was waived. The study was performed in accordance with the Declaration of Helsinki 1964 and its later amendments.
Data Availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Bakhtiary, F., Salamate, S., Amer, M. et al. Comparison of Right Anterior Mini-Thoracotomy Versus Partial Upper Sternotomy in Aortic Valve Replacement. Adv Ther 39, 4266–4284 (2022). https://doi.org/10.1007/s12325-022-02263-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-022-02263-6