Abstract
Introduction
This study aimed to examine the reasons underlying treatment changes among pediatric patients with attention-deficit/hyperactivity disorder (ADHD).
Methods
Data were obtained through online medical chart abstraction (08/2021–09/2021). Eligible patients with ADHD had initiated a treatment regimen at ages 6–17 and within 1–5 years of chart abstraction. Reasons contributing to treatment changes were analyzed for a randomly selected treatment episode. ADHD/treatment-related complication rate was also described. Results were reported overall and among children (ages 6–12) and adolescents (ages 13–17), separately. Physicians’ perspective on adherence among their child, adolescent, and adult patients was assessed through an online survey.
Results
A total of 156 physicians abstracted 434 patient charts (235 children + 199 adolescents). Mean patient age was 11.3 years, and 68.7% were male. Inadequate/suboptimal symptom management was the most common reason for treatment discontinuation (50/83 [60.2%]), add-on (17/21 [81.0%]), and dose increase (189/237 [79.7%]). Patient/parent/family attitude/dislike of medication and ADHD/treatment-related complications were common reasons for treatment discontinuation, add-on, switch, and dose decrease. Overall, 42.4% of patients had ≥ 1 documented ADHD/treatment-related complication, insomnia/sleep disturbances being the most common (9.7%). Among patients with ≥ 1 complication, 75.5% reported the experience/fear of complications had a negative impact on their treatment adherence. Results were similar among children and adolescents. Physicians reported taking actions toward patients’ non-adherence by further educating patients, closer monitoring, and changing the prescribed ADHD medication.
Conclusion
Lack of effectiveness and ADHD/treatment-related complications are important reasons for treatment changes among children and adolescents with ADHD, highlighting the need for more effective and tolerable treatments to mitigate the burden of ADHD.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Why carry out this study? |
Pediatric patients with attention-deficit/hyperactivity disorder (ADHD) have been shown to have frequent treatment changes and poor adherence to ADHD medications. |
Limited information exists on the reasons underlying different types of treatment changes, the rate of complications experienced by patients treated with prescription ADHD medications and physicians’ perspective on patients’ adherence to treatment. |
Understanding the reasons behind treatment changes among pediatric patients and overall poor adherence exhibited by patients with ADHD (including child, adolescent, and adult patients) may help identify potential strategies to improve ADHD care. |
What was learned from the study? |
This panel-based chart review study found that overall, the lack of effectiveness and ADHD/treatment-related complications are important reasons for treatment changes among children and adolescents with ADHD. |
ADHD/treatment-related complications are common, prompting many patients to have treatment changes and suboptimal adherence to their treatment; both factors may lead to lower effectiveness. |
Introduction
Attention-deficit/hyperactivity disorder (ADHD) is one of the most common neurodevelopmental disorders of childhood, with an estimated prevalence of 10.0% among children and 6.5% among adolescents in the USA [1,2,3]. The management of ADHD often involves medications (i.e., stimulants, non-stimulants) and/or non-pharmacological approaches (e.g., behavioral parental and classroom training). Stimulants remain the most common first-line pharmacological treatment for school-aged patients with ADHD owing to their efficacy in reducing ADHD core symptoms [3, 4].
Treatment outcomes in ADHD rely on several factors, including the type of treatment used as well as adherence and persistence to treatment [5, 6]. However, previous studies have shown poor adherence to ADHD medications [7,8,9] and frequent treatment changes among pediatric patients [10,11,12]. For instance, a recent analysis on ADHD treatment pattern using US claims data [13] found that approximately two-thirds of children and adolescents experienced a treatment change (including discontinuation, switch, add-on, and drop) over a 12-month period, suggesting the presence of an unmet treatment need. Notably, the potential negative consequences of frequent treatment changes and poor adherence may have an impact on multiple aspects of the children and adolescents’ lives, such as poor academic performances [14], impaired quality of life [15], and increased financial burden [13].
Knowledge on the reasons underlying treatment changes among pediatric patients with ADHD is necessary to delineate specific areas of unmet need in patient care and help develop strategies to minimize treatment changes and improve adherence; this may in turn improve symptom management and mitigate the burden of ADHD. Although previous claims-based studies have demonstrated frequent changes of ADHD medications among children and adolescents, the reasons for treatment changes were not captured in these studies because of the lack of clinical information in claims data. Additionally, previous studies have mostly focused on discontinuation [7, 9]. Meanwhile, multiple factors may affect patients’ overall treatment satisfaction (e.g., inadequate symptom control or experience of complications), which may also have an impact on other treatment changes and adherence. However, there is a paucity of comprehensive reports on the reasons underlying different types of treatment changes and the rate of complications experienced by patients treated with prescription ADHD medications in the US [16]. Furthermore, limited information exists on physicians’ perspective on patients’ adherence to treatment, which may be important for understanding treatment decisions and potential strategies to improve adherence.
To address the abovementioned knowledge gaps, this study aimed to identify reasons for treatment changes in pediatric patients with ADHD in the US, assess the proportion of pediatric patients receiving ADHD treatment who experienced ADHD/treatment-related complications, and describe physicians’ perspective on adherence to ADHD treatment among their child, adolescent, and adult patients through medical chart abstraction and physician survey.
Methods
Study Design and Data Source
This was a retrospective panel-based chart review study of pediatric patients (children [ages 6–12] and adolescents [ages 13–17]) with ADHD. Online medical chart abstraction was completed by the treating physicians from August 27, 2021, to September 16, 2021. Physicians were recruited from the panel of M3 Global Research, one of the largest panels of healthcare providers in the US that is representative of the American Medical Association Masterfile. An invitation was sent to the panel of physicians who treat patients with ADHD. Eligible physicians who were willing to participate in the study provided their written informed consent. Physicians were then asked to provide clinical information on 1–3 patients with ADHD. The physicians filled in the information of their randomly selected pediatric patients corresponding to a randomly selected treatment regimen initiated between 1 and 5 years prior to the date of chart abstraction via an electronic case report form designed specifically for this study. To reduce potential selection bias, physicians were asked to compile a list of eligible patients and enter information for all ADHD treatments received in the previous 1 to 5 years for an individual patient whose last name began with a letter randomly generated by the survey. The survey then randomly selected one ADHD treatment regimen from among the treatment regimens entered for each selected patient. A randomly selected treatment regimen spanned from initiation of the treatment regimen until the earliest of treatment discontinuation, treatment add-on, treatment switch (i.e., switching ≥ 1 agent in combination therapy), death, or the date of chart abstraction. The index date was the date of the initiation of the randomly selected treatment episode.
Participating physicians were also asked to complete an online survey on their general perspectives on adherence to ADHD treatment for their child, adolescent, and adult patients. This study was conducted in accordance with the applicable ethical regulations; it was exempt from full review and approved through an expedited review by the Western Copernicus Group Institutional Review Board (work order: 1-1458776-1). Participating physicians were compensated for completing the study.
Study Population
Physicians meeting the following eligibility criteria were included in the study: (1) practice medicine in the US; (2) prescribed treatment to ≥ 1 child or adolescent patient with ADHD; (3) had access to patient medical charts, including information on ADHD treatment, reasons for treatment changes, and ADHD/treatment-related complications.
Patients meeting the following eligibility criteria were included in the study: (1) aged 6–17 years inclusively at the index date; (2) had a diagnosis of ADHD prior to the index treatment; (3) had ≥ 1 medical visit in the 12 months prior to data collection; (4) initiated an ADHD treatment between 1 and 5 years prior to data collection.
Study Measures and Outcomes
Physician demographics and practice characteristics (e.g., specialty, practice setting, practice size) were collected. Patient characteristics, including demographic (e.g., age, gender) and clinical (e.g., type and severity of ADHD, comorbidities) variables were also collected. Information on the index treatment, including the type of treatment (e.g., stimulants, non-stimulants, combination therapy [i.e., multiple stimulants, multiple non-stimulants, or combination of stimulants and non-stimulants]), duration of treatment, adherence to planned schedule, and concomitant medications, were summarized.
Treatment changes and modifications related to the index treatment episode were assessed. Key treatment changes at the end of index treatment included treatment discontinuation, add-on, or switch. Data collected included reasons for treatment change, duration of treatment, and ADHD/treatment-related complications that contributed to the end of treatment episode. Data were also collected for treatment modifications, including treatment interruption/drug holiday and dose increase or decrease that occurred during the index treatment but did not constitute the end of the selected treatment episode. The reasons for treatment modifications and ADHD/treatment-related complications that occurred during the treatment were described. Additionally, the degree of negative impact of ADHD/treatment-related complications or fear of potentially experiencing ADHD/treatment-related complications on patients’ adherence to ADHD treatment as well as the reasons for continuing treatment despite the presence of ADHD/treatment-related complications were described.
Data on physicians’ perspective on adherence among child, adolescent, and adult patients and physician strategies to manage non-adherent patients collected through the online survey were summarized.
Statistical Analyses
All measures and outcomes were descriptively summarized. Means, medians, and standard deviations (SD) were reported for continuous variables. Frequency counts and percentages were reported for categorical variables. Measures and outcomes derived from patient-level information were described overall and stratified by children and adolescent subgroups. No statistical comparisons were conducted.
Results
Sample Size
A total of 156 physicians abstracted 434 patient charts, including 235 for child patients and 199 for adolescent patients with ADHD. The average number of charts abstracted per physician was 2.8.
Physician Characteristics
The characteristics of participating physicians are presented in Table 1. Most physicians were psychiatrists (62.8%), followed by pediatricians (20.5%) and family medicine practitioners (16.0%); 84.6% of physicians were in practice for > 5 years; 75.6% practiced in private or community settings, and 56.4% of the physicians practiced in a suburban setting. Physicians from across all US census regions were included. About three-quarters (75.6%) of physicians reported using a clinical scale rather than clinical judgment to rate ADHD severity. Most physicians had treated a mix of children, adolescents, and adults with ADHD, with 94.2%, 99.4%, and 95.5% of physicians having experience in treating child, adolescent, and adult ADHD patients, respectively.
Patient Characteristics
The characteristics of patients in the overall sample are presented in Table 2. The mean patient age was 11.3 years; 68.7% were male, 71.4% were White Caucasian, and 15.9% were Black or African American. Regarding insurance coverage, 70.3% had commercial insurance and 21.9% were Medicaid beneficiaries. Patients were from across all US census regions. The most common type of ADHD recorded was combined presentation (56.2%), followed by inattentive type (32.5%) and hyperactive type (10.6%); 86.4% of patients had moderate or severe ADHD. The most frequently recorded ADHD-associated comorbidities were anxiety (19.1%), depression (12.2%), learning disability (12.2%), and emotional impulsivity/mood lability (12.0%). Notably, in the child and adolescent subgroups, some differences in the frequencies of ADHD-associated comorbidities at baseline were observed. For example, the proportion of patients with recorded anxiety or depression was higher among adolescents than among children (Supplementary Table S1).
Treatment Characteristics
Prior to the index treatment, 48.2% of patients in the overall sample had received pharmacological therapy and 77.0% had received non-pharmacological therapy. For the index treatment, the majority of patients (83.2%) received stimulants alone, followed by non-stimulants alone (11.3%) and combination therapy (5.1%) (Table 3). The average duration of the index treatment was 23.3 months. The majority (94.9%) of patients were prescribed to take their index medication on a regular schedule, yet 48.3% of these patients had < 80% adherence to their pre-defined treatment schedule, as documented in their medical chart completed by the treating physician. At index treatment initiation, 28.8% of patients were also receiving ≥ 1 other prescribed medication; common medications received included antidepressants (12.4%), antianxiety medications (8.8%), and sleep aids (excluding over-the-counter; 6.7%).
Reasons for Treatment Changes and Modifications
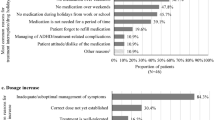
Among patients in the overall sample who discontinued index treatment at the time of chart abstraction (N = 83), the average time to discontinuation was 13.8 months. The most common reasons for treatment discontinuation were inadequate/suboptimal management of symptoms (60.2%), patient attitude/dislike of the medication (30.1%), and the presence of ADHD/treatment-related complications (25.3%; Fig. 1a); the most reported complications contributing to treatment discontinuation included anxiety/panic attacks, emotional impulsivity/mood lability, and insomnia and other sleep disturbances (19.0% each).
Reasons for treatment changes and modifications. a Most common reasons for treatment discontinuation1,2. b Most common reasons for treatment interruption/drug holidays1,3. c Most common reasons for dose increase1,4. d Most common reasons for dose decrease1,5. 1. More than one option could be selected (i.e., not mutually exclusive). 2. Other reasons for treatment discontinuation included cost considerations, inconvenience of dosing, social stigma associated with ADHD medication, treatment monitoring becoming burdensome, and other patient/parent/family/physician-driven reasons. 3. Other reasons for treatment interruption/drug holidays included patient forgot to refill medication, testing treatment efficacy or if treatment is still needed, social stigma associated with ADHD medication, and other patient/parent/family/physician-driven reasons. 4. Other reasons for dose increase included leveraging the treatment's side effects, weight gain, change in insurance coverage, and other patient/physician-driven reasons. 5. Other reasons for dose decrease included inadequate/suboptimal management of symptoms, member of patient's household misuse of medication or addiction, and parent/family attitude/dislike of the medication
Among patients with treatment add-on (N = 21), the average time to add-on was 9.4 months. The most common reasons for treatment add-on were inadequate/suboptimal management of symptoms (81.0%), parent/family attitude/dislike of the medication (14.3%), and the management of ADHD/treatment-related complications (9.5%).
Among patients who initiated on combination therapy and later had a treatment switch (N = 4), the average time to the switch was 8.2 months. The most common reason for treatment switch was parent/family attitude/dislike of the medication (75.0%).
Additionally, some patients experienced treatment modifications that did not lead to a treatment change. Among patients with treatment interruption/drug holiday (N = 67), the majority (80.6%) reported having treatment interruptions/drug holidays sometimes or frequently, mostly driven by not taking medication during vacations, weekends, and/or holidays from school or work (71.6%; Fig. 1b). Meanwhile, the most common reason for a dose increase was inadequate/suboptimal management of symptoms (189 of 237 patients [79.7%]; Fig. 1c) and that for a dose decrease was presence of ADHD/treatment-related complications (12 of 30 patients [40.0%]; Fig. 1d).
The reasons for treatment changes and modifications were generally similar among the child and adolescent subgroups (Supplementary Table S2).
ADHD/Treatment-Related Complications During Index Treatment and Impact on Adherence
Overall, 42.4% of patients experienced ≥ 1 ADHD/treatment-related complication (Fig. 2a). The most common ADHD/treatment-related complications were insomnia and other sleep disturbances (9.7%), decreased appetite/weight loss (9.4%), and anorexia (9.2%). Among patients with ≥ 1 ADHD/treatment-related complication (n = 184), 75.5% reported the experience or fear of ADHD/treatment-related complications had a negative impact on their adherence to treatment (Fig. 2b), and 66.8% remained on treatment despite experiencing complications. The most commonly reported reasons for remaining on treatment included that the ADHD/treatment-related complications were tolerable (60.2%), patient or family preference (33.3%), other available treatment options were not as effective to manage ADHD symptoms (25.2%), and other available treatment options would also be associated with ADHD/treatment-related complications (22.8%). Results were generally similar among the child and adolescent subgroups (Supplementary Figure S3 and S4).
Physicians’ Perspective on Adherence Among Child, Adolescent, and Adult Patients with ADHD and Management Strategies
Based on the physician survey, 23.8% of the participating physicians reported that their child patients missed a dose of their ADHD treatment about half the time or more; this proportion was 27.7% for adolescent patients and 15.4% for adult patients (Fig. 3a). The physicians’ perspective on the top reasons for patient skipping or missing planned doses of ADHD treatment were forgetting to take it, problems with obtaining prescription, weekend, vacation or special events, and experienced or to avoid potentially experiencing adverse events (Fig. 3b).
Physicians’ perspective on adherence among child, adolescent, and adult patients and management strategies. a Perspective on frequency of patients skipping or missing doses of ADHD treatment. b Perspective on reasons for patients skipping or missing planned doses of ADHD treatment. c Actions taken by physicians for managing non-adherent patients with ADHD
Regarding management strategies for non-adherent patients with ADHD, the most frequent actions taken by physicians to manage patients who reported regularly skipping or missing planned doses of ADHD treatment were patient education, closer monitoring, and change prescribed ADHD medication (i.e., discontinuation, add-on, switch) (Fig. 3c).
Discussion
The current panel-based chart review study of pediatric patients with ADHD found that treatment-related factors were key reasons for treatment changes among children and adolescents with ADHD in the US, despite the multiple US Food and Drug Administration-approved pharmacological treatment options available [17]. While many of the available treatments are associated with favorable efficacy profiles [18], the benefits of treatments can only be realized if patients receive the treatment and adhere to the planned schedule. However, prior real-world evidence has shown that more than a third of children and adolescents with ADHD in the US are not receiving pharmacological treatments [19]; among those who do receive treatments, treatment changes are frequent and adherence tends to be low [8, 9, 13]. Thus, it is crucial to understand the challenges faced by these patients to improve patient care; however, there is a paucity of studies that provide context to interpret these observations. Through analyzing the reasons underlying treatments changes among pediatric patients with ADHD, this study revealed that inadequate/suboptimal management of symptoms was the most common reason for treatment discontinuation (60.2%), and more than one-fourth of patients discontinued medication such as ADHD/treatment-related complications such as sleeping and eating disorders commonly associated with ADHD medications [20, 21]. Furthermore, inadequate/suboptimal management of symptoms and ADHD/treatment-related complications were also among the main reasons for other treatment changes and modifications, including treatment add-on, treatment switch, and dose increase. Over 40% of pediatric patients treated for ADHD experienced at least one complication during treatment. Additionally, adverse events were a common reason for poor adherence among patients, as reported in the physician survey. Overall, physician-perceived patient adherence found in this study appeared to be higher than actual patient adherence reported in the literature [10, 22]. Together, findings of this study fill the gap of existing studies and allow us to better understand the reasons underlying the frequent treatment changes and poor adherence in pediatric ADHD management from the physicians’ perspective. Future studies including patients’ and parents’ perspectives are warranted for a more complete understanding on the challenges associated with ADHD management. Results from these studies may help identify potential solutions to alleviate some of the burdens in ADHD care in the long term.
The current chart review study provides a contemporary and comprehensive assessment on the reasons for various types of treatment changes in ADHD and has revealed that the reasons for changes remain mostly treatment-related within the context of the current landscape in the US. This study did not impose specific criteria on patient treatment history (e.g., it did not exclude patients with other psychotropic medications) so that the patient samples could more closely reflect the patient population encountered in real-world routine clinical practice. Comprehensive studies in the US evaluating reasons underlying different types of treatment changes to ADHD medications have been limited, and most studies have focused on treatment discontinuation [9, 23]. Nonetheless, findings in the current study are in line with those found in a systematic literature review that included 31 international studies on reasons for treatment discontinuation among children and adolescents with ADHD. The review found that the most frequently reported reasons for discontinuing ADHD medication in these populations were treatment ineffectiveness/suboptimal response and adverse effects (both reported in 21 of 31 studies). Additionally, poor adherence, parent’s decision to discontinue, and patient attitude were also reported as reasons for discontinuation [9].
It is of note that parent/family and patient attitude/dislike of medication were among the frequently documented reasons for treatment changes in the current study. The negative attitude toward a medication is likely attributable to myriad reasons, which may be treatment-related (e.g., lack of effectiveness, treatment complications) and/or parent/patient-related factors (e.g., children not feeling like themselves, parents concern about long-term effects) [24, 25]. Prior single-center studies on parent/patient attitude toward stimulant treatment for ADHD based on interviews and questionnaires suggested that a major cause of parents’ negative attitude toward stimulants was the concern about adverse and long-term effects of stimulants on their children. Meanwhile, many child and adolescent patients with ADHD felt that stimulant treatment led to changes in their sense of self and depressed mood, and they would choose to discontinue treatment if they could [24, 25]. Future large-scale parent or patient surveys may provide additional insights to the reasons underlying the dislike of medication. Nonetheless, hints from prior studies may point to the potential improvement in parents/patient attitude toward the medication through advancing ADHD treatments with a better safety profile and less negative impact on the lives of child/adolescent patients with ADHD.
Understanding the reasons behind treatment changes is important as it may help identify potential areas of improvement in patient care. A recent US claims analysis [13] found that treatment changes, including discontinuation, switch, add-on, and drop, occurred in 59% of children and 68% of adolescents with ADHD within a year of treatment initiation. That study also found that patients tended to cycle through multiple regimens, with increasing use of combination therapy in later regimens. The observation in the current study was in line with the finding in that study as patients receiving combination therapy appeared to have more severe ADHD at baseline and a higher rate of ADHD/treatment-related complications (data not shown); these patients might have tried other regimens but did not manage to have the disorder controlled. More importantly, the current study has provided context to the findings of the abovementioned claims study by showing the lack of effectiveness and tolerability issues are important reasons for treatment changes among pediatric patients with ADHD, demonstrating the need for effective and tolerable medications to improve patients’ treatment experience. Future prospective studies may be conducted to evaluate whether the use of treatments with more favorable efficacy and safety profiles could lead to reduced treatment changes, for example, through following sequential patients who were started on different types of ADHD medications. Frequent treatment changes or dosage adjustments not only entail additional efforts by both physicians and patients/families (e.g., additional monitoring and clinic visits), but the number of treatment changes has also been shown to be associated with increased healthcare costs among patients with ADHD [13, 26]. Therefore, improved medication may not only reduce the need for treatment change but may also mitigate both the clinical and financial burden associated with ADHD.
This study found that many patients remained on treatment despite experiencing complications because of the lack of better alternatives. It is also notable that if treatment changes were not observed in some patients, it would be unclear how these complications might impact adherence or patients’ attitude toward the treatment; these factors may lead to an eventual treatment change or reduced adherence and ultimately hinder the effectiveness of ADHD management. These findings highlight that existing evidence on the frequent treatment changes and poor adherence among pediatric patients with ADHD likely only represents a small portion of the true unmet needs in this population. As staying on treatment and being adherent while treated are crucial for successful treatment outcomes [25, 27], more tolerable ADHD treatment options are warranted to address some of the management challenges in pediatric ADHD.
Meanwhile, the current survey found that physicians believed ~ 75%–85% of their patients with ADHD adhered to the dosing schedule more than half of the time; however, this relatively high physician-perceived patient adherence appears to be inconsistent with actual patient adherence reported in the literature. In previous claims-based analyses in the US that assessed adherence based on how frequently patients refilled their ADHD prescriptions, adherence among children and adolescents ranged between 30 and 60% [10, 22], which was lower than the physician-perceived patient adherence found in the current survey, highlighting the need for enhanced physician–patient communication. While future studies are needed to further explore different avenues for improving ADHD management, we believe the reasons underlying treatment changes identified in the current study (e.g., adverse event experience, patients forgetting to take the medication) can be translated into practical clinical solutions to alleviate some of the management challenges. For instance, physicians should spend more time explaining to their patients and parents/families how the efficacy and safety of the treatments may impact the treatment journey and factor these considerations into the treatment decisions (e.g., can a less effective but more tolerable option be appropriate for some patients to minimize the risk of subsequent treatment changes or lack of adherence?). Physicians should also initiate discussion on adherence or use tools such as Prescription Drug Monitoring Program (PDMP) [28] to monitor prescription fills, to ensure they have a good knowledge of patients’ adherence to their ADHD treatment. Furthermore, pediatric patients and their parents/families should be educated early on during treatment planning to ascertain their understanding on the importance of receiving treatment and being adherent to the treatment schedule. Ongoing discussions should also be conducted to tailor the specific challenges faced by ADHD patients at different developmental stages. Altogether, raising awareness among physicians and pediatric patients as well as their parents/families on the importance of treatment and potential strategies to improve adherence, along with efforts to advance treatment development to minimize the need of treatment changes, would be necessary to alleviate the burden of pediatric ADHD.
The findings from the current study should be considered in respect to certain limitations. First, information captured was limited to that available in the patients’ medical records held by the physicians participating in the study. Thus, information on healthcare services received outside of the physicians’ care settings that were not recorded in the medical charts was not available for this study. Second, the study included only physicians accessible through the panel of M3 Global Research who wished to participate in this study; accordingly, the sample may not be representative of the US physician population treating children and adolescents with ADHD. Third, although participating physicians were instructed to select patients at random, selection bias may still exist (e.g., selection of patients recently seen or with certain outcomes). Fourth, the current survey did not differentiate whether the ADHD/treatment complications were adverse event- or comorbidity-related. Finally, certain findings on treatment patterns and ADHD/treatment-related complications may be limited by the small sample size available.
Conclusions
This chart review study found that overall, the lack of effectiveness and ADHD/treatment-related complications were important reasons for treatment changes among children and adolescents with ADHD. Furthermore, ADHD/treatment-related complications were common among child and adolescent patients, prompting many to have treatment changes and suboptimal adherence to their treatment; both factors may lead to lower effectiveness. These findings highlight the need for more effective and tolerable treatments to mitigate the burden of ADHD.
References
Kessler RC, Avenevoli S, Costello EJ, Georgiades K, Green JG, Gruber MJ, et al. Prevalence, persistence, and sociodemographic correlates of DSM-IV disorders in the National Comorbidity Survey Replication Adolescent Supplement. Arch Gen Psychiatry. 2012;69(4):372–80.
Child and Adolescent Health Measurement Initiative. 2018 National Survey of Children’s Health (NSCH) data query. Data Resource Center for Child and Adolescent Health supported by the U.S. Department of Health and Human Services, Health Resources and Services Administration (HRSA), Maternal and Child Health Bureau (MCHB) 2018. http://www.childhealthdata.org. Accessed 22 Feb 2022.
Wolraich ML, Hagan JF Jr, Allan C, Chan E, Davison D, Earls M, et al. Clinical practice guideline for the diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder in children and adolescents. Pediatrics. 2019;144(4):e20192528.
Posner J, Polanczyk GV, Sonuga-Barke E. Attention-deficit hyperactivity disorder. Lancet. 2020;395(10222):450–62.
Charach A, Ickowicz A, Schachar R. Stimulant treatment over five years: adherence, effectiveness, and adverse effects. J Am Acad Child Adolesc Psychiatry. 2004;43(5):559–67.
Gau SS, Shen HY, Chou MC, Tang CS, Chiu YN, Gau CS. Determinants of adherence to methylphenidate and the impact of poor adherence on maternal and family measures. J Child Adolesc Psychopharmacol. 2006;16(3):286–97.
Ahmed R, Aslani P. Attention-deficit/hyperactivity disorder: an update on medication adherence and persistence in children, adolescents and adults. Expert Rev Pharmacoecon Outcomes Res. 2013;13(6):791–815.
Biederman J, Fried R, DiSalvo M, Storch B, Pulli A, Woodworth KY, et al. Evidence of low adherence to stimulant medication among children and youths with ADHD: an electronic health records study. Psychiatr Serv. 2019;70(10):874–80.
Gajria K, Lu M, Sikirica V, Greven P, Zhong Y, Qin P, et al. Adherence, persistence, and medication discontinuation in patients with attention-deficit/hyperactivity disorder—a systematic literature review. Neuropsychiatr Dis Treat. 2014;10:1543–69.
Christensen L, Sasane R, Hodgkins P, Harley C, Tetali S. Pharmacological treatment patterns among patients with attention-deficit/hyperactivity disorder: retrospective claims-based analysis of a managed care population. Curr Med Res Opin. 2010;26(4):977–89.
Guo L, Danielson M, Cogan L, Hines L, Armour B. Treatment patterns and costs among children aged 2 to 17 years with ADHD in New York State Medicaid in 2013. J Atten Disord. 2021;25(4):463–72.
Hodgkins P, Sasane R, Christensen L, Harley C, Liu F. Treatment outcomes with methylphenidate formulations among patients with ADHD: retrospective claims analysis of a managed care population. Curr Med Res Opin. 2011;27(Suppl 2):53–62.
Schein J, Childress A, Adams J, Cloutier M, Gagnon-Sanschagrin P, Maitland J, et al. Treatment patterns among children and adolescents with attention-deficit/hyperactivity disorder in the United States—a retrospective claims analysis. APSARD annual meeting; January 13–16; Virtual2022
Keilow M, Holm A, Fallesen P. Medical treatment of attention deficit/hyperactivity disorder (ADHD) and children’s academic performance. PLoS ONE. 2018;13(11): e0207905.
Tsujii N, Okada T, Usami M, Kuwabara H, Fujita J, Negoro H, et al. Effect of continuing and discontinuing medications on quality of life after symptomatic remission in attention-deficit/hyperactivity disorder: a systematic review and meta-analysis. J Clin Psychiatry. 2020;81(3):19r13015.
Faraone SV, Banaschewski T, Coghill D, Zheng Y, Biederman J, Bellgrove MA, et al. The World Federation of ADHD international consensus statement: 208 evidence-based conclusions about the disorder. Neurosci Biobehav Rev. 2021;128:789–818.
Children and Adults with Attention-Deficit/Hyperactivity Disorder (CHADD). ADHD medications approved by the US FDA 2021. https://chadd.org/about-adhd/adhd-medications-approved-by-the-us-fda/. Accessed 7 Aug 2022.
Cortese S, Adamo N, Del Giovane C, Mohr-Jensen C, Hayes AJ, Carucci S, et al. Comparative efficacy and tolerability of medications for attention-deficit hyperactivity disorder in children, adolescents, and adults: a systematic review and network meta-analysis. Lancet Psychiatry. 2018;5(9):727–38.
Danielson ML, Bitsko RH, Ghandour RM, Holbrook JR, Kogan MD, Blumberg SJ. Prevalence of parent-reported ADHD diagnosis and associated treatment among U.S. children and adolescents, 2016. J Clin Child Adolesc Psychol. 2018;47(2):199–212.
Centers for Medicare & Medicaid Services (CMS) Medicaid Program Integrity Education (MPIE). Stimulant and related medications: use in pediatric patients. 2015. https://www.cms.gov/Medicare-Medicaid-Coordination/Fraud-Prevention/Medicaid-Integrity-Education/Pharmacy-Education-Materials/Downloads/stim-pediatric-factsheet11-14.pdf. Accessed 8 Aug 2022.
Clavenna A, Bonati M. Pediatric pharmacoepidemiology—safety and effectiveness of medicines for ADHD. Expert Opin Drug Saf. 2017;16(12):1335–45.
Setyawan J, Hodgkins P, Guerin A, Gauthier G, Cloutier M, Wu EQ, et al. Comparing treatment adherence of lisdexamfetamine and other medications for the treatment of attention deficit/hyperactivity disorder: a retrospective analysis. J Med Econ. 2013;16(7):962–75.
Brinkman WB, Simon JO, Epstein JN. Reasons why children and adolescents with attention-deficit/hyperactivity disorder stop and restart taking medicine. Acad Pediatr. 2018;18(3):273–80.
Berger I, Dor T, Nevo Y, Goldzweig G. Attitudes toward attention-deficit hyperactivity disorder (ADHD) treatment: parents’ and children’s perspectives. J Child Neurol. 2008;23(9):1036–42.
Charach A, Yeung E, Volpe T, Goodale T, Dosreis S. Exploring stimulant treatment in ADHD: narratives of young adolescents and their parents. BMC Psychiatry. 2014;14:110.
Schein J, Childress A, Adams J, Cloutier M, Gagnon-Sanschagrin P, Maitland J, et al. Treatment patterns among adults with attention-deficit/hyperactivity disorder in the United States: a retrospective claims study. Curr Med Res Opin. 2021;37(11):2007–14.
Wolraich ML, Chan E, Froehlich T, Lynch RL, Bax A, Redwine ST, et al. ADHD diagnosis and treatment guidelines: a historical perspective. Pediatrics. 2019;144(4):e20191682.
Centers for Disease Control and Prevention. Prescription Drug Monitoring Programs (PDMPs): U.S. Department of Health & Human Services; 2021. https://www.cdc.gov/drugoverdose/pdmp/index.html. Accessed 24 Feb 2022.
Acknowledgements
Funding
This work was supported by Otsuka Pharmaceutical Development & Commercialization, Inc. The study sponsor also funded the journal’s Rapid Service and Open Access Fees. The study sponsor was involved in several aspects of the research, including the study design, the interpretation of data, the writing of the manuscript, and the decision to submit the manuscript for publication.
Medical Writing Assistance
Medical writing assistance was provided by Flora Chik, PhD, an employee of Analysis Group, Inc., a consulting company that has provided paid consulting services to Otsuka Pharmaceutical Development & Commercialization, Inc.
Author Contributions
Martin Cloutier, Marjolaine Gauthier-Loiselle, Rebecca Bungay, and Annie Guerin contributed to study conception and design, collection and assembly of data, and data analysis and interpretation. Jeff Schein and Ann Childress contributed to study conception and design, data analysis and interpretation. All authors reviewed and approved the final content of this manuscript.
Prior Presentation
Part of the material in this manuscript was presented at the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) 2022 In-Person and Virtual Conference, May 15–18, 2022, as a podium presentation.
Disclosures
Jeff Schein is an employee of Otsuka Pharmaceutical Development & Commercialization, Inc. Ann Childress received research support from Allergan, Takeda/Shire, Emalex, Akili, Ironshore, Arbor, Aevi Genomic Medicine, Neos Therapeutics, Otsuka, Pfizer, Purdue, Rhodes, Sunovion, Tris, KemPharm, Supernus, and the US Food and Drug Administration; was on the advisory board of Takeda/Shire, Akili, Arbor, Cingulate, Ironshore, Neos Therapeutics, Otsuka, Pfizer, Purdue, Adlon, Rhodes, Sunovion, Tris, Supernus, and Corium; received consulting fees from Arbor, Ironshore, Neos Therapeutics, Purdue, Rhodes, Sunovion, Tris, KemPharm, Supernus, Corium, Jazz, Tulex Pharma, and Lumos Pharma; received speaker fees from Takeda/Shire, Arbor, Ironshore, Neos Therapeutics, Pfizer, Tris, and Supernus; and received writing support from Takeda /Shire, Arbor, Ironshore, Neos Therapeutics, Pfizer, Purdue, Rhodes, Sunovion, and Tris. Martin Cloutier, Marjolaine Gauthier-Loiselle, Rebecca Bungay, and Annie Guerin are employees of Analysis Group, Inc., a consulting company that has provided paid consulting services to Otsuka Pharmaceutical Development & Commercialization, Inc.
Compliance with Ethics Guidelines
This study was conducted in accordance with the applicable ethical regulations; it was exempt from full review and approved through an expedited review by the Western Copernicus Group Institutional Review Board (work order: 1-1458776-1). Eligible physicians who were willing to participate in the study provided their written informed consent.
Data Availability
The data analyzed in this study are subject to Health Insurance Portability and Accountability Act privacy restrictions and are not publicly available. De-identified data could be made available by the corresponding author upon reasonable request.
Author information
Authors and Affiliations
Corresponding author
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Schein, J., Cloutier, M., Gauthier-Loiselle, M. et al. Reasons for Treatment Changes in Children and Adolescents with Attention-Deficit/Hyperactivity Disorder: A Chart Review Study. Adv Ther 39, 5487–5503 (2022). https://doi.org/10.1007/s12325-022-02329-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-022-02329-5