Abstract
Background
International guidance recognizes the shortcomings of the modified Duke Criteria (mDC) in diagnosing infective endocarditis (IE) when transoesophageal echocardiography (TOE) is equivocal. 18F-FDG PET/CT (PET) has proven benefit in prosthetic valve endocarditis (PVE), but is restricted to extracardiac manifestations in native disease (NVE). We investigated the incremental benefit of PET over the mDC in NVE.
Methods
Dual-center retrospective study (2010-2018) of patients undergoing myocardial suppression PET for NVE and PVE. Cases were classified by mDC pre- and post-PET, and evaluated against discharge diagnosis. Receiver Operating Characteristic (ROC) analysis and net reclassification index (NRI) assessed diagnostic performance. Valve standardized uptake value (SUV) was recorded.
Results
69/88 PET studies were evaluated across 668 patients. At discharge, 20/32 had confirmed NVE, 22/37 PVE, and 19/69 patients required surgery. PET accurately re-classified patients from possible, to definite or rejected (NRI: NVE 0.89; PVE 0.90), with significant incremental benefit in both NVE (AUC 0.883 vs 0.750) and PVE (0.877 vs 0.633). Sensitivity and specificity were 75% and 92% in NVE; 87% and 86% in PVE. Duration of antibiotics and C-reactive Protein level did not impact performance. No diagnostic SUV cut-off was identified.
Conclusion
PET improves diagnostic certainty when combined with mDC in NVE and PVE.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Infective endocarditis (IE) is classically viewed as a rare diagnosis, however its incidence has risen since the turn of the century, associated with a surge in cardiovascular intervention in an aging population.1 Diagnosis is based on the modified Duke Criteria (mDC), with patients classified as either definite, possible or rejected for IE based on the clinical, imaging and microbiological features present.2 Foremost among these are typical findings on echocardiography and blood cultures; absence of either of these makes a definite diagnosis of IE difficult to achieve. Despite advances in microbiological and imaging techniques, however, the mDC remain only 80% sensitive and specific, with around a quarter of patients misclassified as possible IE despite pathologically proven disease,3,4,5 even with the use of transoesophageal echocardiography (TOE).6,7 The use of multimodality cross-sectional imaging has therefore gained traction and is now recommended.2
The use of 18F-Fluorodeoxyglucose positron emission tomography with computed tomography (18F-FDG PET/CT, PET) to aid in the diagnosis of prosthetic valve endocarditis (PVE) has been increasingly reported with high sensitivity and specificity,8 especially when using myocardial suppression techniques.9,10,11,12,13 Adding focal tracer avidity around a prosthetic valve as a major criterion to the mDC reduces the number of “possible” cases in a cohort of IE patients, and thus improves the diagnostic utility of these criteria.14 The focus of these studies has been mainly limited to PVE15 and cardiac implantable electronic device (CIED) infection,16 where PET confers incremental benefit over TOE due to elimination of acoustic shielding. By comparison, the role of PET in native valve endocarditis (NVE) is less well studied. When used, it has been limited to identifying extracardiac manifestations in NVE, with low sensitivity and specificity at the valve level, and a failure to meaningfully reclassify patients to confirm or refute the diagnosis.9,12
Barts Heart Centre (BHC) was formed in May 2015 following a merger of the cardiology departments of Barts Health NHS Trust and University College London Hospitals (UCLH) NHS Trust. This merger made BHC the single cardiac surgery referral center for North Central and East London, and resulted in a significant increase in the number of IE cases seen. In line with the European Society of Cardiology (ESC) IE guidelines, this prompted formalization of the UCLH model to form an Endocarditis Team.2 In this context, and with ~ 150 referrals of possible IE per year we sought to investigate the incremental benefit of PET over the mDC in both PVE and NVE using our Endocarditis Team model.
Methods
Patients and Endocarditis Team
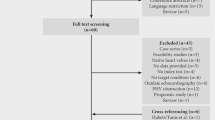
Under terms of an overarching audit, a dual center retrospective review identified all patients undergoing PET for IE from January 2010 to December 2018. Patients imaged early in the post-operative period following valve surgery for IE (< 3 months), those with CIED-only IE, and studies with failure of myocardial suppression were excluded (Figure 1).
Consort diagram. All cases referred to the BHC Infective Endocarditis MDT (October 2015 to December 2018) and a search of the UCLH Radiology Database (January 2010 to October 2015) for ‘endocarditis’ and ‘PET’ were assessed for eligibility. Data were collected and categorized for NVE and PVE. BHC, Barts Heart Centre; NVE, native valve endocarditis; PVE, prosthetic valve endocarditis; PET-CT, 18Fluoride fluorodeoxyglucose position emission tomography with computed tomography; UCLH, University College London Hospitals
Data were collected on all relevant clinical and imaging variables, particularly those pertinent to the mDC, duration of antibiotic therapy pre-PET and biochemical markers. Individual cases were scored using the mDC both pre- and post-PET by two independent investigators (CP & TC) in consensus. The definitive discharge diagnosis was recorded by surgical specimen in those who underwent operative intervention, or by Endocarditis Team consensus in those medically managed (excluding the PET findings).
The Endocarditis Team review all cases of IE referred to our Institution on a weekly basis. Prior to the formation of the BHC team, the core members led in the clinical care of patients with IE at UCLH via ad hoc bedside discussion.
Image Acquisition of 18F-FDG PET-CT
All patients underwent myocardial suppression technique to suppress metabolic activity in the myocardium. This was achieved using a > 24-hour high fat, carbohydrate-restricted diet, a > 12-hour fast and intravenous injection of unfractionated heparin (50 IU/kg) 60 minutes prior to assessment with PET.15,16 PET-CT was only undertaken when blood glucose < 11 mmol/L as per local standard operating procedures. Following intravenous administration of 18F-FDG (4.5 MBq/kg) at a mean time of 64 minutes (SD 13 minutes) (mean activity 157 ± 39 MBq), we performed combined imaging with an MI DR PET-CT scanner (GE Healthcare, Chalfont St Giles, UK). An unenhanced, ungated CT was performed (64 detectors, at a pitch of 1.5 and a 2.5 mm collimation) from vertex to thigh for attenuation correction. A subsequent PET was performed at a bed overlap of 49% and a time per bed position of 100s. The reconstruction method was VUE Point FX, with 2 iterations, 12 subsets and a 5 mm Gaussian filter. All studies were standardized for display and reading with an SUV window threshold of 0-1017.
Interpretation of 18F-FDG PET/CT
PET images were read in a blinded fashion by two independent investigators (CP & LM) with joint reading to resolve discrepancies in consensus, using attenuation and non-attenuation corrected images (the latter in particular for PVE). Myocardial suppression was graded as good, fair, poor or non-diagnostic,17 with non-diagnostic cases of unsuppressed diffuse myocardial uptake excluded from further analysis.
Studies were assessed for avidity of the culprit valve. The pattern and distribution of avidity was categorized as focal, heterogenous, homogenous or none (heterogenous and homogenous typically referred to as diffuse). An overall verdict (yes/no) was given on a case-by-case basis as to whether the PET was suspicious for IE or not, with a study considered positive if uptake was either ‘focal’ or ‘heterogenous’. Note was made if PET suggested an alternative diagnosis.
An elliptical region of interest (ROI) was placed over the valve, mediastinal blood pool and liver for semi-quantitative assessment of avidity using absolute mean and maximum standardized uptake values (SUV) allowing target-to-background analysis. In addition to avidity at the level of the cardiac valves, note was made of the presence of extracardiac uptake (including spleen, mediastinal lymph nodes, lungs, spine and sternum).
Studies were analyzed using the freely available Horos (version 3.3.5).
Statistical Analysis
GraphPad Prism (version 7.0) and SPSS (version 25) were used for statistical analyses. Descriptive statistics were calculated for continuous variables, and χ2 for categorical data. Diagnostic performance was evaluated using Receiver Operating Characteristic (ROC) analysis and net reclassification index (NRI). Respective sensitivities, specificities, positive and negative predictive values for PET in both native and prosthetic valve disease were calculated using the discharge diagnoses, categorized as either confirmed or rejected IE as described above.
Results
PET was undertaken in 88/668 patients, with 69 studies (10.3%) eligible for inclusion; 59/404 (14.6%) following the formalization of the Endocarditis Team in October 2015. The cohort featured 48 male patients, with an overall mean age of 61 years (range 21–89 years). Thirty-two (46%) were native valve patients and 37 cases had prosthetic valves, of which 20 were tissue and 17 mechanical prostheses. All patients, except one NVE patient, underwent assessment with TOE (patient refusal). Further baseline characteristics are described in Table 1.
At discharge, 20/32 (63%) had confirmed NVE and 22/37 (59%) confirmed PVE, giving a total prevalence for IE in our cohort of 61%. Nineteen (28%) patients required surgical intervention, 9 (28%) NVE and 10 (27%) PVE, with the remaining 50/69 cases managed medically, as per European Society of Cardiology (ESC) and British Society for Antimicrobial Chemotherapy (BSAC) guidance.2,18Staphylococcus aureus was isolated in 20/69 (29.0%) patients; 18/69 (26.1%) were peripheral blood culture-negative (BCNIE) (Table 1).
The median duration of IE-targeted antibiotic therapy pre-PET was 20.6 (IQR 9.5-25.0) days in 30/32 NVE cases and 17.0 (IQR 11.5-33.0) days in 33/37 PVE cases. Categorical analysis of median duration of antibiotics showed no significant impact on PET performance in NVE, PVE or overall (P > 0.10). Mean CRP at the time of PET was 38.6 (SD ± 29.8) mg/L in 20/32 NVE cases and 39.3 (SD ±4 5.4) mg/L in 29/37 PVE cases. Prolonged duration of antibiotic therapy was associated with a downward trend in CRP at time of PET (β = − 0.11, r2 = 0.004). There was no difference in PET performance with CRP < 40 and ≥ 40 in NVE, PVE or overall (P > 0.10).12
Over a median follow-up of 3.21years (IQR 1.75-3.96, 211.7 patient-years), there were 9 episodes of further IE in 8 patients, with 1 treatment failure in a medically managed patient unfit for surgery. Median IE free duration was 379 days in these individuals (range 28-1095 days). Of these episodes, 5 required surgical intervention during the index admission and four were managed medically. Independent review of mDC post-PET and discharge diagnosis made by the Endocarditis Team showed PET to have correctly confirmed or refuted IE in all cases.
Of the PET studies undertaken, 3/88 (3.4%) were excluded from analysis due to complete failure of myocardial suppression, rendering the scans non-diagnostic (Figure 1). All remaining scans eligible for inclusion were diagnostic, despite variable success in suppressing myocardial uptake (Table 2). Use of PET re-classified patients with possible IE as per mDC to either definite or rejected IE in both NVE and PVE, including the identification of an alternative, non-cardiac source of infection in 12 patients overall (16.9%). Quantification of this reclassification yielded NRI values of 0.89 for NVE and 0.90 for PVE (Table 3), where NRI > 0.5 suggests a beneficial test. ROC curves showed incremental benefit of PET over Duke’s criteria alone in both NVE (AUC 0.883 vs 0.750, P < 0.001) and PVE (AUC 0.877 vs 0.633, P < 0.001) compared to discharge diagnosis (Figure 2). PET sensitivity, specificity, positive and negative predictive values were 75%, 92%, 94% and 69% respectively in NVE, and 87%, 86%, 91% and 80% in PVE.
Incremental benefit of PET-CT with the modified Duke Criteria. ROC curves highlight significant incremental benefit of PET-CT over modified Duke Criteria alone in both PVE and NVE when compared to discharge diagnosis. NVE, native valve endocarditis; PET-CT, 18Fluoride fluorodeoxyglucose position emission tomography with computed tomography; PVE, prosthetic valve endocarditis; ROC, Receiver Operator Characteristic
Focal uptake was observed in 16 (50%) cases of NVE and 18 (49%) in PVE (Table 2). Diffuse uptake was seen in 13 cases (35%) of PVE; 4 were true positive IE with heterogenous uptake, and 7 true negative with homogenous uptake representing post-operative change in the latter category. Illustrative examples are depicted in Figure 3. Embolic phenomena were identified in 14 (20%) patients; splenic avidity and low volume mediastinal lymphadenopathy were not considered here, as while suggestive of infection, they are not specific for IE.19 Semi-quantitative analysis using SUVs showed no clear diagnostic cut-off for SUV, SUVmax or SUVmean, nor when these parameters were normalized to hepatic and mediastinal blood pool uptake (Table 4).
Representative images of Endocarditis on PET-CT. Images acquired following a high fat, low carbohydrate diet, 12-hour fast and intravenous heparin to achieve suppression of physiological myocardial uptake (myocardial suppression). Panel A NVE of mitral valve; Panel B PVE of a mitral valve ring; Panel C NVE of mitral valve with pericardial effusion; Panel D post-surgical uptake in mechanical aortic and mitral valve replacements; Panel E PVE with root abscess in TAVI; Panel F NVE of mitral valve. NVE, native valve endocarditis; PET-CT, 18Fluoride fluorodeoxyglucose position emission tomography with computed tomography; PVE, prosthetic valve endocarditis; TAVI, transcatheter aortic valve implant
Discussion
The climbing incidence of IE, and clear benefits of the Endocarditis Team mandate a guideline-driven approach to the diagnosis of IE.2 The benefit of multimodality imaging is clearly recognized in the current era, with PET having a role where the likelihood of IE is high but TOE remains equivocal. In NVE, this has focussed on the identification of extracardiac emboli.20,21 The recent results of the EURObservational Research Programme of the ESC Endocarditis Registry (EORP EuroENDO Registry, EuroENDO) highlight the increasing use of PET at 16.6%,8 but this has mainly focussed on PVE and CIED-IE.1222 The increasing utilization of PET is reflected in the current study, where PET usage increased from 3.8% (January 2010-September 2015) to 14.6% (October 2015-December 2018) in line with the most recent ESC guidelines and the increasing body of evidence for PET in IE.2,17
IE remains a diagnostic challenge, especially in the absence of positive major criterion of the mDC. In our cohort, all patients had ongoing clinical suspicion of IE following equivocal TOE in 68/69 cases (N = 1, patient refusal), and 26% with likely BCNIE (Table 1). NRI highlights that PET accurately re-classifies individual patients with IE in both NVE (NRI 0.89) and PVE (NRI 0.90) (Table 3), as well as overall by ROC analysis (Figure 2). PET has been used with high specificity in NVE in previous studies,12,23 predominantly through identification of septic emboli.17 Critically, and uniquely to this study, however, NRI highlights the ability of PET to reliably confirm (positive reclassification) and refute (negative reclassification) the diagnosis (positive and negative NRI 0.40-0.50) through evaluation at valve level alone (Table 3).
Our data add further weight to the growing body of evidence advocating the sensitivity and specificity of PET in PVE, and corroborate our ability to utilize PET appropriately in IE. However, we have now been able to additionally demonstrate comparable sensitivity and specificity in NVE, at 75% and 92%, respectively, as with one similar series to date.23 Furthermore, when PET is used in this manner, we see the incremental benefit over the mDC in both NVE and PVE (AUC 0.883 NVE, AUC 0.877 PVE) with a prevalence of confirmed IE in 61% of our cohort at discharge (Figure 2). This is in comparison to similarly-sized series in the literature, with a sensitivity of 17.5%-57% in NVE,9,12,19,20,24,25 and only 28.0% in EuroENDO.8
Given the lack of typical TOE findings of significant valvular insufficiency and/or presence of vegetations in this cohort, it is unsurprising that fifty patients (72%) lacked a surgical indication and were managed medically. This is despite 99% of patients having at least one TOE, a high rate compared to 81%-88% of patients in other studies. 12,13 BCNIE also drives the use of PET in IE, as a lack of positive cultures also makes diagnosis by mDC challenging, with rates ranging from 10%-46% in other series.8,12,13,19 However, correctly diagnosing IE is critical to ensure appropriate inpatient treatment and outpatient monitoring. Follow-up of these patients shows agreement with discharge diagnosis derived by the Endocarditis Team (without use of the PET data reported here) in all patients, with only 3 patients re-presenting with IE within 24 months, over 212 patient-years of follow-up.
Identifying the factors responsible for the high performance of PET in the current cohort is critical. Factors thought responsible for the low sensitivity of PET in NVE are well summarized in the literature, from both technical limitations of the modality as well as the pathophysiology of NVE.12,13,17,23 These include discrete vegetations with less common paravalvular complications in NVE, a less aggressive local inflammatory response compared to PVE resulting in lower FDG uptake, duration of antibiotics pre-PET and CRP at time of PET.
Achieving adequate myocardial suppression is imperative to the successful use of PET in IE, with ~ 85% of suppression graded as good or fair in our cohort, and only 3/72 (4%) studies found to be non-diagnostic and therefore excluded from analysis. Even when myocardial suppression was poor, meaningful valvular assessment was still possible in this cohort based on visual analysis. This is in comparison to failure of myocardial suppression in 5%-32% of the cohort in similar studies of NVE and PVE.12,13,23
In our practice, PET is used where the diagnosis remains unclear despite 99% utilization of high-quality TOE (with lower rates in other series), including in patients with NVE. This is particularly important as this group of patients typically lack valve findings that mandate early surgery, hence diagnostic equipoise. Our approach is to therefore repeat echocardiography prior to PET, in order to easily identify those in whom valve dysfunction may have developed. However, this is reflected in the long median duration of antibiotics (19 days, IQR 10-30 days) prior to PET and downward trend in CRP (mean 39.9 mg/L, SD 39.8 mg/L) when compared to other studies.12,13,19,23 Our data suggest that when PET is targeted to those patients in whom the possibility of IE is high, but the mDC are equivocal, PET can add meaningful information irrespective of inflammatory marker levels or duration of antibiotic therapy.12 This finding may be explained by the pathophysiology of the disease; the long time-to-PET in the present study may allow for sufficient valvular inflammation to develop meeting the threshold for detection using 18F-FDG.17 This is however in contrast to a previous study where a lack of inflammatory response, or suppression of that response with antibiotics, has been associated with poor sensitivity of PET in NVE, with a proposed CRP cut-off of 40 mg/L.13 This relative delay to PET reflects real-world practice of a high-throughput nuclear medicine department in the United Kingdom’s National Health Service, in comparison to other series’ where PET was typically undertaken in < 10 days.8,12,13,19,23 with not all patients receiving broad-spectrum antibiotics for possible IE.23
We would suggest the use of PET in IE requires involvement of an Endocarditis Team with high volume throughput. This will both optimize case selection and improve technical reading of the study. This is particularly important to correctly distinguish valve from myocardial tracer uptake, and recognize patterns of uptake typical and atypical for IE, especially following previous cardiac surgery (Figure 3). Our analyses did not identify a clear semi-quantitative indicator to confirm or refute IE based on SUV, though this has been posited in other studies.13,20 In our experience, this failure to identify a semi-quantitative cut-off is a consequence of often low-grade focal avidity in NVE even with good myocardial suppression, and diffuse patchy uptake with a high standard deviation of SUV in PVE. Alternative analyses, such as doughnut-shaped ROIs or machine learning, or scanning techniques, such as ECG- and/or respiratory-gating may yet highlight a computed cut-off suggestive of valve infection.
The results of this study suggest that the real-world application of PET to patients with IE has meaningful benefit. Nonetheless, a formal prospective multicenter diagnostic accuracy study with hard endpoints is warranted, and we would argue should include patients with both NVE and PVE.
Limitations
Advances in IE have been hindered by low incidence and a lack of randomized trials as a result.26 This retrospective analysis is based on real-world application of PET in a cohort of ‘all-comers’ referred to an Endocarditis Team discussing approximately 150 cases per annum, and therefore mitigates patient selection bias. Furthermore, our approach to multimodality imaging in patients with possible IE has been driven by experience in the context of international guidance, with a fixed pathway for PET utilization in both NVE and PVE as described earlier in the manuscript. Despite the high throughput of our center, utilization rates are similar to those seen in the current EuroENDO registry,8 and explains the limited patient cohort presented here. While similar in size to other studies in the literature, this study adds a unique perspective in NVE and adds to the growing body of evidence for the use of PET in IE.
The incidence of IE and the limitations of the mDC may cause bias when relying on expert consensus to confirm or refute IE in medically managed patients, and is a significant issue in all IE studies without surgical specimens. However, this is where the guidance currently supports the use of PET, where the diagnosis is not clear, and therefore surgical intervention is not necessarily mandated. Blinded scoring of the mDC and imaging analysis by the research team reduce the limitations of expert consensus in our study, and is supported by net reclassification following PET, highlighting the benefit of PET overall and for reclassification of individual patients. This impact is further mitigated by follow-up data that suggest the correct diagnosis was made, especially given a low incidence of recurrent episodes of IE over 212 patient-years.
Despite these limitations, the incremental benefit of PET in both NVE and PVE described herein suggests meaningful benefit. However, we would only advocate the routine use of PET where the diagnosis is equivocal after high-quality TOE and surgery is not mandated for another indication.
Conclusions
In this retrospective analysis, we highlight the incremental benefit of PET for the diagnosis of IE in both native and prosthetic disease. PET performs well irrespective of inflammatory markers or duration of IE-focussed antibiotic treatment. We advocate the use of PET by expert Endocarditis Teams where both NVE and PVE is suspected, but TOE remains equivocal.
New Knowledge Gained
The literature consistently identify poor sensitivity of PET/CT for NVE. We highlight that in a high-volume center, PET can be used to contribute to the diagnosis of both NVE and PVE in a meaningful manner. PET provides meaningful information at valve level in PVE and NVE, to help confirm and refute the diagnosis (NRI), outperforms mDC alone (AUC) and has higher than reported sensitivity in NVE. We further explore duration of antibiotics, CRP at time of PET and time to PET to explain why our findings differ to the rest of the literature.
Abbreviations
- BCNIE:
-
Blood culture-negative infective endocarditis
- ESC:
-
European Society of Cardiology
- IE:
-
Infective endocarditis
- mDC:
-
Modified Duke Criteria
- NRI:
-
Net reclassification index
- NVE:
-
Native valve endocarditis
- PET-CT:
-
18F-Fluorodeoxyglucose positron emission tomography with computed tomography
- PVE:
-
Prosthetic valve endocarditis
- SUV:
-
Standardized uptake values
- TOE:
-
Transoesophageal echocardiogram
References
Dayer MJ, Jones S, Prendergast B, Baddour LM, Lockhart PB, Thornhill MH. Incidence of infective endocarditis in England, 2000–13: A secular trend, interrupted time-series analysis. Lancet 2015; 385:1219-28. https://doi.org/10.1016/S0140-6736(14)62007-9
Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta J-P, Del Zotti F et al. 2015 ESC Guidelines for the management of infective endocarditis. Eur Heart J 2015; 36:3075-28. https://doi.org/10.1093/eurheartj/ehv319
Habib G, Derumeaux G, Avierinos JF, Casalta JP, Jamal F, Volot F et al. Value and limitations of the Duke criteria for the diagnosis of infective endocarditis. J Am Coll Cardiol 1999; 33:2023-29
Li JS, Sexton DJ, Mick N, Nettles R, Fowler VG, Ryan T et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis 2000; 30:633-38. https://doi.org/10.1086/313753
Gomes A, Glaudemans AWJM, Touw DJ, van Melle JP, Willems TP, Maass AH et al. Diagnostic value of imaging in infective endocarditis: A systematic review. Lancet Infect Dis 2017; 17:e1-e14. https://doi.org/10.1016/S1473-3099(16)30141-4
Koo HJ, Yang DH, Kang JW, Lee JY, Kim DH, Song JM et al. Demonstration of infective endocarditis by cardiac CT and transoesophageal echocardiography: Comparison with intra-operative findings. Eur Heart J Cardiovasc Imaging 2018; 19:199-07. https://doi.org/10.1093/ehjci/jex010
Bruun NE, Habib G, Thuny F, Sogaard P. Cardiac imaging in infectious endocarditis. Eur Heart J 2014; 35:624-32. https://doi.org/10.1093/eurheartj/eht274
Habib G, Erba PA, Iung B, Donal E, Cosyns B, Laroche C et al. Clinical presentation, aetiology and outcome of infective endocarditis. Results of the ESC-EORP EURO-ENDO (European infective endocarditis) registry: A prospective cohort study. Eur Heart J 2019; 40:3222. https://doi.org/10.1093/eurheartj/ehz620
Ricciardi A, Sordillo P, Ceccarelli L, Maffongelli G, Calisti G, Di Pietro B et al. 18-Fluoro-2-deoxyglucose positron emission tomography-computed tomography: An additional tool in the diagnosis of prosthetic valve endocarditis. Int J Infect Dis 2014; 28:219-24. https://doi.org/10.1016/j.ijid.2014.04.028
Graziosi M, Nanni C, Lorenzini M, Diemberger I, Bonfiglioli R, Pasquale F et al. Role of18F-FDG PET/CT in the diagnosis of infective endocarditis in patients with an implanted cardiac device: A prospective study. Eur J Nucl Med Mol Imaging 2014; 41:1617-23. https://doi.org/10.1007/s00259-014-2773-z
Bensimhon L, Lavergne T, Hugonnet F, Mainardi J-L, Latremouille C, Maunoury C et al. Whole body [18F]fluorodeoxyglucose positron emission tomography imaging for the diagnosis of pacemaker or implantable cardioverter defibrillator infection: A preliminary prospective study. Clin Microbiol Infect 2011; 17:836-44. https://doi.org/10.1111/j.1469-0691.2010.03312.x
de Camargo RA, Sommer Bitencourt M, Meneghetti JC, Soares J, Gonçalves LFT, Buchpiguel CA et al. The role of 18F-fluorodeoxyglucose positron emission tomography/computed tomography in the diagnosis of left-sided endocarditis: native vs prosthetic valves endocarditis. Clin Infect Dis. 2019. https://doi.org/10.1093/cid/ciz267
Swart LE, Gomes A, Scholtens AM, Sinha B, Tanis W, Lam MGEH et al. Improving the diagnostic performance of 18 F-fluorodeoxyglucose positron-emission tomography/computed tomography in prosthetic heart valve endocarditis. Circulation 2018; 138:1412-27. https://doi.org/10.1161/CIRCULATIONAHA.118.035032
Saby L, Laas O, Habib G, Cammilleri S, Mancini J, Tessonnier L et al. Positron emission tomography/computed tomography for diagnosis of prosthetic valve endocarditis: Increased valvular18F- fluorodeoxyglucose uptake as a novel major criterion. J Am Coll Cardiol 2013; 61:2374-82. https://doi.org/10.1016/j.jacc.2013.01.092
San S, Ravis E, Tessonier L, Philip M, Cammilleri S, Lavagna F et al. Prognostic value of 18F-fluorodeoxyglucose positron emission tomography/computed tomography in infective endocarditis. J Am Coll Cardiol. 2019 https://doi.org/10.1016/j.jacc.2019.06.050
Calais J, Touati A, Grall N, Laouénan C, Benali K, Mahida B et al. Diagnostic impact of 18 F-fluorodeoxyglucose positron emission tomography/computed tomography and white blood cell SPECT/computed tomography in patients with suspected cardiac implantable electronic device chronic infection. Circ Cardiovasc Imaging. 2019 https://doi.org/10.1161/circimaging.117.007188
Slart RHJA, Glaudemans AWJM, Gheysens O, Lubberink M, Kero T, Dweck MR et al. Procedural recommendations of cardiac PET/CT imaging: standardization in inflammatory-, infective-, infiltrative-, and innervation (4Is)-related cardiovascular diseases: A joint collaboration of the EACVI and the EANM. Eur J Nucl Med Mol Imaging 2020; 2020:1-24. https://doi.org/10.1007/s00259-020-05066-5
Gould FK, Denning DW, Elliott TSJ, Foweraker J, Perry JD, Prendergast BD et al. Guidelines for the diagnosis and antibiotic treatment of endocarditis in adults: A report of the working party of the british society for antimicrobial chemotherapy. J Antimicrob Chemother. 2012. https://doi.org/10.1093/jac/dkr450
Philip M, Delcourt S, Mancini J, Tessonnier L, Cammilleri S, Arregle F et al. 18F-fluorodeoxyglucose positron emission tomography/computed tomography for the diagnosis of native valve infective endocarditis: A prospective study. Arch Cardiovasc Dis 2021; 114:211-20. https://doi.org/10.1016/j.acvd.2020.10.005
Mahmood M, Kendi AT, Ajmal S, Farid S, O’Horo JC, Chareonthaitawee P et al. Meta-analysis of 18F-FDG PET/CT in the diagnosis of infective endocarditis. J Nucl Cardiol. 2019. https://doi.org/10.1007/s12350-017-1092-8
Van Riet J, Hill EE, Gheysens O, Dymarkowski S, Herregods MC, Herijgers P et al. 18F-FDG PET/CT for early detection of embolism and metastatic infection in patients with infective endocarditis. Eur J Nucl Med Mol Imaging 2010; 37:1189-97. https://doi.org/10.1007/s00259-010-1380-x
Orvin K, Goldberg E, Bernstine H, Groshar D, Sagie A, Kornowski R et al. The role of FDG-PET/CT imaging in early detection of extra-cardiac complications of infective endocarditis. Clin Microbiol Infect 2015; 21:69-76. https://doi.org/10.1016/j.cmi.2014.08.012
Abikhzer G, Martineau P, Grégoire J, Finnerty V, Harel F, Pelletier-Galarneau M. [18F]FDG-PET CT for the evaluation of native valve endocarditis. J Nucl Cardiol. 2020. https://doi.org/10.1007/s12350-020-02092-6
Salomäki SP, Saraste A, Kemppainen J, Bax JJ, Knuuti J, Nuutila P et al. 18F-FDG positron emission tomography/computed tomography in infective endocarditis. J Nucl Cardiol 2017; 24:195-06. https://doi.org/10.1007/s12350-015-0325-y
Kouijzer IJE, Berrevoets MAH, Aarntzen EHJG, De Vries J, Van Dijk APJ, Oyen WJG et al. 18F-fluorodeoxyglucose positron-emission tomography combined with computed tomography as a diagnostic tool in native valve endocarditis. Nucl Med Commun 2018; 39:747-52. https://doi.org/10.1097/MNM.0000000000000864
Cahill TJ, Prendergast BD. Infective endocarditis. Lancet 2016; 387:882-93. https://doi.org/10.1016/S0140-6736(15)00067-7
Acknowledgements
This retrospective analysis was supported by researchers at the National Institute for Health Research Biomedical Research Centres at University College London and Queen Mary, University of London. CP is funded by the Derek Willoughby Trust and Barts Charity. The authors acknowledge the contribution of the Barts Heart Centre Endocarditis Team.
Disclosures
The authors have no relationships or activities that are relevant or conflict with the content of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Funding
No funding to declare.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarizes the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Primus, C.P., Clay, T.A., McCue, M.S. et al. 18F-FDG PET/CT improves diagnostic certainty in native and prosthetic valve Infective Endocarditis over the modified Duke Criteria. J. Nucl. Cardiol. 29, 2119–2128 (2022). https://doi.org/10.1007/s12350-021-02689-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-021-02689-5