Abstract
Background
Liver transplantation is the main treatment for cholestatic liver disease and some metabolic liver diseases in children. However, no accurate prediction model to determine the survival probability of grafts prior to surgery exists. This study aimed to develop an effective prognostic model for allograft survival after pediatric liver transplantation.
Methods
This retrospective cohort study included 2032 patients who underwent pediatric liver transplantation between January 1, 2006, and January 1, 2020. A nomogram was developed using Cox regression and validated based on bootstrap sampling. Predictive and discriminatory accuracies were determined using the concordance index and visualized using calibration curves; net benefits were calculated for model comparison. An online Shiny application was developed for easy access to the model.
Results
Multivariable analysis demonstrated that preoperative diagnosis, recipient age, body weight, graft type, preoperative total bilirubin, interleukin-1β, portal venous blood flow direction, spleen thickness, and the presence of heart disease and cholangitis were independent factors for survival, all of which were selected in the nomogram. Calibration of the nomogram indicated that the 1-, 3-, and 5-year predicted survival rates agreed with the actual survival rate. The concordance indices for graft survival at 1, 3, and 5 years were 0.776, 0.757, and 0.753, respectively, which were significantly higher than those of the Pediatric End-Stage Liver Disease and Child–Pugh scoring systems. The allograft dysfunction risk of a recipient could be easily predicted using the following URL: https://aspelt.shinyapps.io/ASPELT//
Conclusion
The allograft survival after pediatric liver transplantation (ASPELT) score model can effectively predict the graft survival rate after liver transplantation in children, providing a simple and convenient evaluation method for clinicians and patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Pediatric liver transplantation (LT) is the main treatment for cholestatic liver disease and some metabolic liver diseases in children [1]. Since the successful implementation of the first LT by Starzl et al. [2] in 1967, adult and pediatric LTs have been developing continuously. Currently, pediatric LT is a mature treatment for children with end-stage liver diseases, such as acute liver failure, autoimmune diseases, cholestasis, metabolic or genetic diseases, and oncologic and vascular liver diseases [3, 4]. With the development of immunosuppressive drugs and postoperative care, the mortality rate of recipients and grafts has decreased every year. The 1-year and 5-year survival rates after pediatric LT have been reported to exceed 90% and 85%, respectively [4]. However, despite these improvements in treatment techniques, graft dysfunction still poses a significant challenge, directly impacting recipient prognosis. Graft dysfunction in children might result in secondary transplantation or even death [5, 6]. The main causes of dysfunction include chronic rejection, biliary tract or vascular complications, and inflammation or infection [7,8,9].
There are a few clinical methods to predict the pediatric LT survival rate at present, such as Pediatric risk of mortality (PRISM) III, Survival Outcomes Following Pediatric Liver Transplant (SOFT), Pediatric End-Stage Liver Disease (PELD), and Child–Pugh [8, 10], with the last two being the most widely used scoring systems for liver diseases. A higher score in each of these methods indicates a lower prognosis for patients. PELD scores are primarily used in patients younger than 12 years of age, whereas Child–Pugh scores determine the prognosis of patients with liver cirrhosis. However, these scoring tools are not specifically designed for predicting graft survival, although they have been applied to evaluate allograft outcomes in previous studies [11,12,13]. Unfortunately, there is a lack of comprehensive studies analyzing the risk factors associated with allograft outcomes, and as of now, an accurate prediction model for assessing the survival probability of grafts prior to surgery remains elusive. Therefore, the main purposes of this study were to determine the risk factors affecting graft survival and to construct a relatively accurate predictive index using a nomogram to predict allograft survival after pediatric liver transplantation (ASPELT).
Methods
Study population and data acquisition
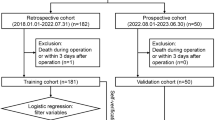
We retrospectively analyzed all the data of children younger than 12 years of age who had undergone LT at the liver surgery department in Renji Hospital affiliated with the Shanghai Jiaotong University School of Medicine from 2006 to 2019. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki, as reflected in a priori approval by the institution’s human research committee (Ethics Committee of Renji Hospital No: KY2020-064). Informed consent to participate in the study was obtained from their parent or legal guardian.
Inclusion and exclusion criteria were applied during the data selection process. Children who met the following criteria were included in the study: age younger than 12 years and underwent liver transplantation at Renji Hospital between 2006 and 2019. Children who met any of the following criteria were excluded from the study: foreign children, orphans, and inadequate follow-up data.
Preoperatively, a thorough and detailed physical examination; laboratory tests including liver and kidney function tests, coagulation profiles, and interleukin (IL)-1β levels; and ultrasound examination and computed tomography were performed routinely before surgery to completely assess the preoperative condition of the recipients and determine the extent of any complications and to determine the PELD and Child–Pugh scores.
All patients were closely monitored in the intensive care unit after surgery. Blood gas, coagulation profile, and liver function tests were assessed daily. Patients were transferred to the general ward after becoming stable. The dosage of the immunosuppressant was adjusted to maintain a safe upper level according to its blood concentration. After discharge, all patients were followed up regularly at the outpatient clinic, and the medication plan was adjusted using a standard protocol.
Study design
Overall, 23 candidate predictors, consisting of 20 indicators of recipients’ characteristics and 3 of donor characteristics, were analyzed during the training of the prediction model. The recipients’ characteristics were as follows: three physical features, namely, age, body weight, and growth failure; diagnosis; presence of any heart disease, portal hypertension, gastrointestinal bleeding, cholangitis, and ascites; preoperative levels of albumin (g/dL), total bilirubin (mg/dL), prothrombin time (s), international normalized ratio (INR), and IL-1β (pg/mL) as per the results of laboratory tests; perioperative features including direction of the portal vein flow, spleen thickness (mm), spleen diameter (mm), surgical method, graft-to-recipient weight ratio; and the year of transplantation. For donor characteristics, body mass index (BMI), relationships to recipients, and ABO compatibility were considered.
The outcome of our prediction model was graft survival, which was defined as the time from transplantation to graft dysfunction.
Statistical analysis
Baseline characteristics were analyzed with descriptive statistics. Continuous variables were described as the mean ± standard deviation, and frequencies and percentages are used to describe categorical variables. Continuous predictors were categorized according to their optimal cutoff points that maximized the sum of sensitivity and specificity in the receiver operating characteristic (ROC) curve analyses. All covariates listed in the previous chapter were included in a univariable analysis using proportional hazards regression, and covariates with less significance (i.e., P value > 0.1) or multicollinearity were excluded from multivariable analysis. Before entering into the multivariable analysis, multiple imputation was implemented for covariates with missing data, and imputation methods were selected based on variable characteristics.
The prediction model was developed for 1-year, 3-year, and 5-year graft survival. Predictors were selected according to the results of multivariable analyses. In addition, the Akaike information criterion, C-index, and area under the ROC curve (AUC) were considered for determining our final predictive model. For internal validation, bootstrapping was conducted, and calibration plots were created to compare the predicted survival probabilities and actual probabilities. Model visualization was achieved using a nomogram, which explains the method of calculating predicted probabilities. To evaluate the model performance and compare it with that of the other existing and well-known scoring systems, such as PELD scores and Child–Pugh, net benefits were calculated and illustrated using decision curves. Moreover, an AUC with precision [i.e., 95% confidence interval (CI)] was generated for all models using nonparametric inverse probability weighting estimation and bootstrapping. All analyses were performed using open-source software R version 4.0. A P value of 0.05 (two-tailed) was considered to indicate statistical significance.
Application development
An interactive web application was built and published based on the R package Shiny to allow easy access to the prediction model. Prediction results can be obtained after inputting patient data.
Results
Clinical characteristics
A total of 2080 pediatric patients under the age of 12 underwent liver transplantation (LT) at the Liver Surgery Department of Renji Hospital, which is affiliated with the Shanghai Jiaotong University School of Medicine, between the years 2006 and 2019. To enhance the reliability of our follow-up data, we excluded information from 38 foreign pediatric patients and 10 orphaned children. Consequently, our analysis was based on a cohort of 2032 pediatric patients.
Since 2006, the number of pediatric LTs performed at Renji Hospital has been increasing year by year, especially for living donor LT. From 2017 to 2019, the number of pediatric LTs remained at approximately 400 cases annually, and the number of living donor LT cases increased much more dramatically than the number of deceased donor LT cases over the years (Fig. 1a). Supplementary Fig. 1 depicts the geographic distribution of all recipients who have undergone LT at our center.
Number of pediatric liver transplantations from 2006 to 2019 at Renji Hospital and total survival rate for the allograft and patient using the Kaplan–Meier curve. The y-axis indicates the graft survival rate, and the x-axis indicates the number of years after liver transplantation (a). Percentages of recipient survival among the total recipients at different years are shown in the table in the middle (b). DDLT deceased donor liver transplantation, LDLT living donor liver transplantation
The details of the donor and recipient characteristics are summarized in Table 1. This study included 2032 pediatric LT recipients with a median [interquartile range (IQR)] age of 8 months (6–17 months) and body weight of 7.7 kg (6.6–10 kg); there were 1067 (52.50%) girls and 965 (47.50%) boys. Overall, 211 (10.40%) patients were assessed as having comorbid heart disease; 728 (35.80%) had portal hypertension; 244 (12.00%) experienced gastrointestinal bleeding; 579 (28.50%) had cholangitis, and 1131 (55.65%) were diagnosed with ascites. According to ultrasound results, the direction of the portal vein flow was toward-liver in 1413 cases (69.5%) and ex-liver in 332 cases (16.4%), with data missing for the remaining 287 cases (14.1%). The operation methods recorded included 100 cases of split LT (4.9%), 276 cases of orthotopic LT (13.6%), and 1656 cases of living donor LT (81.5%). The median (IQR) follow-up time was 2.69 years (1.45–4.27), and the total number of deaths recorded until 2019 was 170 (8.3%).
The indications for pediatric LT are listed in Supplementary Table 1. Among the 2032 cases, there were 1801 cases of cholestatic liver disease (88.6%) and 1647 cases of biliary atresia (81.1%). There were 154 cases of metabolic diseases (7.6%), 28 cases of tumor diseases (1.4%), 24 cases of retransplantation, 13 cases of acute liver failure, and 12 cases of vascular diseases. All patients received immunosuppressive therapy following the operation, with cyclosporine administered in 282 cases and tacrolimus in 1750 cases.
Postoperative prognosis and univariable and multivariable Cox regression analyses
Among the 2032 patients who underwent pediatric LT, the 1-, 3-, 5-, and 10-year graft survival rates were 93.3%, 90.9%, 89.9%, and 87.3%, respectively. The 1-, 3-, 5-, and 10-year overall survival rates were 94%, 92%, 91%, and 89%, respectively (Fig. 1b).
The results of the univariable and multivariable Cox regression analyses are shown in Table 2. According to the univariable analysis, weight, age, diagnosis, operation type, and all five preoperative comorbidities were selected for the multivariable analysis. Moreover, preoperative laboratory measurements, including serum albumin, serum total bilirubin, prothrombin time, INR, and IL-1β were screened. Other features, including the portal vein flow direction, spleen thickness and diameter, and graft-to-recipient weight ratio along with the BMI of the donors, were also included as independent risk factors in the following analysis.
The adjusted odds ratios and their precision indicated that lower body weight (< 7.2 kg), age ≥ 10 years, heart disease, cholangitis, direction of portal vein flow, spleen thickness (≥ 27 mm), retransplantation, split-liver transplantation, and higher levels of total bilirubin (≥ 5.3 mg/dL) significantly increased the risk of allograft dysfunction. The survival curves of significant covariates in each group are demonstrated in Supplementary Fig. 2, panels A-I.
Development and validation of the allograft survival after pediatric liver transplantation nomogram
Based on the multivariable Cox regression results listed in the previous section and combined with a stepwise model selection and clinical consideration, 10 variables were selected and used to construct a visualized nomogram model (Fig. 2) to predict the survival rate of allografts. Considering that IL-1β is not a routine preoperative test in some transplantation centers, we also created a model without IL-1β. Each variable has a corresponding score on the point scale axis of the nomogram. The scores of the different variables are presented in Supplementary Table 2. By adding the scores of different variables, the total score of ASPELT can be easily calculated, and the survival probability of a graft can be subsequently determined. The C-indices of the prediction nomogram, ASPELT, for predicting graft survival at 1, 3, and 5 years were 0.776, 0.757, and 0.753, respectively, whereas those of the version without IL-1β (i.e., ASPELT/IL-1β) were 0.774, 0.751, and 0.749, respectively (Supplementary Table 3).
Nomogram predicting graft survival after a pediatric liver transplantation. To determine the number of points received for each level, a line is drawn upward on each variable axis. Points from every categories were summed together to acquire a total point, which will be marked on the “Total Points” axis. From each point, a line is drawn downward to the survival axes to determine the likelihood of 1-, 3-, or 5-year survival of an allograft. IL interleukin, GS graft survival, LT liver transplantation
The results of internal validation are illustrated by the AUC with the 95% CI and calibration plots. The bootstrap validation exhibited a significant prediction accuracy for both ASPELT and ASPELT/IL-1β. The AUCs for predicting graft survival at 1, 3, and 5 years were 73.27% (95% CI 68.25–77.76%), 67.49% (95% CI 62.27–72.31%), and 60.80% (95% CI 54.87–66.43%), respectively, for ASPELT and 73.44% (95% CI 68.43–77.92%), 67.67% (95% CI 62.58–72.37%), and 61.15% (95% CI 55.25–66.74%), respectively, for ASPELT/IL-1β (Fig. 3a–c). The calibration plots created using bootstrap resampling also suggested that the 1-, 3-, and 5-year predicted survival rates agreed with the actual survival rates (Fig. 4a–c).
ROC curves for ASPELT, ASPELT/IL-1β, PELD, and Child–Pugh scores. Curves present the scores at (a) 1 year, (b) 3 years, and (c) 5 years posttransplantation. ROC receiver operating characteristic, ASPELT allograft survival after pediatric liver transplantation, IL interleukin, PELD Pediatric End-Stage Liver Disease, AUC area under the receiver operating characteristic curve, CI confidence interval
Calibration curves for allograft survival after pediatric liver transplantation scores. Calibration curves showing allograft survival at a 1 year, b 3 years, and c 5 years posttransplantation. The nomogram-predicted probability of OS is plotted on the x-axis, actual OS is plotted on the y-axis. Grey line showed the ideal model where predicted probability fully aligned with the actual probability. OS overall survival
Comparison with other prediction models
According to the results of the ROC and decision curve analyses, our prediction model outperformed the existing prediction scoring systems, PELD and Child–Pugh, at all three predicting points (Figs. 3a–c and 5a–c). Figure 5a–c indicates that the use of ASPELT and ASPELT/IL-1β had a higher net benefit than the other two models over a wide range of threshold probabilities.
Decision curve analysis for ASPELT. The curves show the analyses at a 1 year, b 3 years, and c 5 years posttransplantation. The y-axis is the net benefit, and the x-axis denotes the threshold probability. ASPELT allograft survival after pediatric liver transplantation, PELD Pediatric End-Stage Liver Disease, IL interleukin, GS graft survival
Clinical use
The ASPELT scores showed a normal distribution (Fig. 6a). Based on the final score, the cutoff values to determine the risk of poor allograft outcomes were estimated to be at scores of 146 and 196. Recipients with a score below 146 were assigned to the low-risk group, those with a score of 146–196 to the median-risk group, and those with a score above 196 to the high-risk group (Supplementary Table 4). With an increase in risk, the graft survival rate at 1, 3, 5, and 10 years after the operation decreased significantly (P < 0.001; Fig. 6b). The ASPELT model can also be accessed on the Internet or via a web application at https://aspelt.shinyapps.io/ASPELT/.
Discussion
In our study, we established a prognostic tool to enhance the prediction of allograft survival after pediatric LT. The evaluation of the performance of our prediction model, ASPELT, suggested that it can precisely categorize LT recipients into low-risk, median-risk, and high-risk groups, wherein a significant difference in the allograft survival rates was noted among the groups. Furthermore, this prediction model has a higher prediction accuracy than the other models previously used.
Our study determined that the preoperative diagnosis, recipient age, body weight, surgical type, preoperative serum total bilirubin level, serum IL-β level, portal venous blood flow direction, spleen thickness, and presence of heart disease and cholangitis were independent risk factors associated with graft survival in pediatric LT. Therefore, we developed ASPELT, a 10-variable nomogram, to predict graft survival at 1, 3, and 5 years. Based on the ASPELT score, the recipients were divided into high-risk, median-risk, and low-risk groups. With an increase in risk, the graft survival rate at 1, 3, 5, and 10 years after the operation decreased significantly (P < 0.0001). As the most widely used tool to evaluate the prognosis of patients with liver disease [14, 15], the PELD and Child–Pugh scoring systems were also compared with ASPELT.
The PELD score was used to determine the priority of pediatric LT candidates based on the risk of death before transplantation [16]. It was developed using bilirubin, INR, serum albumin, age, and growth failure to predict the probability of death on the waiting list. Some studies have shown a correlation between the PELD score and posttransplant survival [15], while others have suggested that the PELD score is not an accurate predictor of the outcomes following transplantation [17, 18]; therefore, the prediction efficiency of PELD has remained undecided. Our study found that the C-indices of PELD for predicting graft survival at 1, 3, and 5 years were 0.548, 0.546, and 0.544, respectively, which were minimally satisfactory.
The Child–Pugh score has been widely used for the assessment of prognosis in liver cirrhosis [19]. In previously reported studies, the variation in survival explained by the Child–Pugh score remains somewhat low (< 50%) [20]. The Child–Pugh score has several limitations [21]. First, it contains subjective variables, such as ascites and encephalopathy, leading to unavoidable deviation in determining the severity of the recipients’ condition. Second, the stratification of the continuous variables also affects the prediction efficiency of the scoring model due to its ceiling or floor effects, which suggests that the Child–Pugh classification may hardly differentiate between patients with an albumin level of 15 g/L versus 24 g/L. In our study, the C-indices of the Child–Pugh scoring system for predicting graft survival at 1, 3, and 5 years were 0.617, 0.618, and 0.615, respectively; these were weaker than those of ASPELT (0.776, 0.757, and 0.755, respectively) and ASPELT/IL-1β (0.774, 0.751, and 0.749, respectively).
Previous prediction models, such as PELD or Child–Pugh, were developed using data either acquired from electronic health records, wherein only preoperative laboratory measurements and physical features were available, or from a single-center study on a small cohort. The ASPELT, however, has been established based on more than 2000 cases, and it considers patient comorbidities, visceral features, and other surgical characteristics apart from those variables used in other models. It also prospectively evaluates the effect of inflammatory responses even though it has not been a regular test before an LT.
The ASPELT score model offers several key advantages in clinical practice. By considering multiple risk factors, the ASPELT score provides a comprehensive assessment of graft survival probability for individual patients. This enables clinicians to develop personalized treatment plans based on each patient’s unique risk profile. Tailored interventions, such as intensified immunosuppressive therapy or alternative treatment strategies, can be implemented, optimizing posttransplant care and improving outcomes. The accurate prediction of graft survival offered by the ASPELT score assists transplant centers in prioritizing candidates based on their individual risk profiles. The ASPELT score allows for precise risk categorization of LT recipients into low-risk, median-risk, and high-risk groups based on preoperative variables. This enables clinicians to identify patients at higher risk of graft failure and tailor their management accordingly. By closely monitoring high-risk patients and adjusting treatment strategies, clinicians can potentially improve graft survival rates and overall patient outcomes.
The limitation of this retrospective study mainly lies in the lack of external validation. If any record from a different transplantation center can be used for model validation, the model performance evaluation can be more robust. Second, this study was conducted at a single center and may, therefore, be weaker in methodology than studies using randomly sampled populations, especially when the prognosis of transplantation may depend on the clinical logistics or responsible surgeon that varies from center to center and is difficult to quantify. Another drawback is the nontraceable missing data, which is inherent in this type of research.
In conclusion, the ASPELT score model can effectively predict the graft survival rate after LT in children, providing a simple and convenient evaluation method for clinicians and patients. The ASPELT score model is not limited to research applications but has direct implications for clinical practice in pediatric LT. By utilizing the ASPELT score, clinicians can enhance patient care, improve outcomes, and contribute to advancements in the field of pediatric liver transplantation.
Data availability
The authors confirmed that the data supporting the findings of this study are available within the article and/or supplementary materials.
References
Chung PHY, Chok KSH, Wong KKY, Tam PKH, Lo CM. Determining the optimal timing of liver transplant for pediatric patients after Kasai portoenterostomy based on disease severity scores. J Pediatr Surg. 2020;55:1892–6.
Starzl TE, Iwatsuki S, Van Thiel DH, Gartner JC, Zitelli BJ, Malatack JJ, et al. Evolution of liver transplantation. Hepatology. 1982;2:614–36.
Chen YH, Lou JG, Yang ZH, Chen QJ, Hua CZ, Ye S, Zhang CM, Chen J, Huang ZW, Yu JD, Gao ZG, Shu Q. Diagnosis, treatment, and prevention of severe acute hepatitis of unknown etiology in children. World J Pediatr. 2022;18(8):538–44.
Ng VL, Fecteau A, Shepherd R, Magee J, Bucuvalas J, Alonso E, et al. Outcomes of 5-year survivors of pediatric liver transplantation: report on 461 children from a North American multicenter registry. Pediatrics. 2008;122:e1128–35.
Sieders E, Peeters PM, TenVergert EM, de Jong KP, Porte RJ, Zwaveling JH, et al. Graft loss after pediatric liver transplantation. Ann Surg. 2002;235:125–32.
Huang M, Xu YS, Yan S, Shi YJ, Pan SB, Chen YB, et al. Use of veno-arterial extracorporeal membrane oxygenation in the first successful combined lung-liver transplantation patient in China. World J Emerg Med. 2022;13:412–4.
Wallot MA, Mathot M, Janssen M, Hölter T, Paul K, Buts JP, et al. Long-term survival and late graft loss in pediatric liver transplant recipients—a 15-year single-center experience. Liver Transpl. 2002;8:615–22.
Rana A, Pallister ZS, Guiteau JJ, Cotton RT, Halazun K, Nalty CC, et al. Survival outcomes following pediatric liver transplantation (Pedi-SOFT) score: a novel predictive index. Am J Transplant. 2015;15:1855–63.
Fan YC, Wang XQ, Zhu DY, Huai XR, Yu WF, Su DS, Pan ZY. Association of different central venous pressure levels with outcome of living-donor liver transplantation in children under 12 years. World J Pediatr. 2023;19:170–9.
Ferah O, Akbulut A, Açık ME, Gökkaya Z, Acar U, Yenidünya Ö, et al. Scoring systems and postoperative outcomes in pediatric liver transplantation. Transplant Proc. 2019;51:2430–3.
Spitzer AL, Dick AA, Bakthavatsalam R, Halldorson JB, Salvalaggio PR, Reyes JD, et al. Intraoperative portal vein blood flow predicts allograft and patient survival following liver transplantation. HPB (Oxford). 2010;12:166–73.
Chul Yoon K, Song S, Jwa EK, Lee S, Man Kim J, Kim OK, et al. Survival outcomes in split compared with whole liver transplantation. Liver Transpl. 2018;24:1411–24.
Alexopoulos SP, Nekrasov V, Cao S, Groshen S, Kaur N, Genyk YS, et al. Effects of recipient size and allograft type on pediatric liver transplantation for biliary atresia. Liver Transpl. 2017;23:221–33.
Abbott WJ, Thomson A, Steadman C, Gatton ML, Bothwell C, Kerlin P, et al. Child-Pugh class, nutritional indicators and early liver transplant outcomes. Hepatogastroenterology. 2001;48:823–7.
Barshes NR, Lee TC, Udell IW, O’mahoney CA, Karpen SJ, Carter BA, et al. The pediatric end-stage liver disease (PELD) model as a predictor of survival benefit and posttransplant survival in pediatric liver transplant recipients. Liver Transpl. 2006;12:475–80.
McDiarmid SV, Anand R, Lindblad AS, Principal Investigators and Institutions of the Studies of Pediatric Liver Transplantation (SPLIT) Research Group. Development of a pediatric end-stage liver disease score to predict poor outcome in children awaiting liver transplantation. Transplantation. 2002;74:173–81.
Bourdeaux C, Tri TT, Gras J, Sokal E, Otte JB, de Ville de Goyet J, et al. PELD score and posttransplant outcome in pediatric liver transplantation: a retrospective study of 100 recipients. Transplantation. 2005;79:1273–6.
McDiarmid SV, Merion RM, Dykstra DM, Harper AM. Selection of pediatric candidates under the PELD system. Liver Transpl. 2004;10(10 Suppl 2):S23–30.
Peng Y, Qi X, Guo X. Child-Pugh versus MELD score for the assessment of prognosis in liver cirrhosis: a systematic review and meta-analysis of observational studies. Med (Baltimore). 2016;95: e2877.
Christensen E. Prognostic models including the Child-Pugh, MELD and Mayo risk scores–where are we and where should we go? J Hepatol. 2004;41:344–50.
Kim HJ, Lee HW. Important predictor of mortality in patients with end-stage liver disease. Clin Mol Hepatol. 2013;19:105–15.
Acknowledgements
The authors wish to acknowledge all those who performed the pediatric liver transplantation in this study.
Funding
The study was supported by the National Natural Science Foundation of China (grant number 81700570).
Author information
Authors and Affiliations
Contributions
GGX: conceptualization, data curation, writing—original draft, formal analysis, methodology, visualization, writing–review and editing. PST: formal analysis, methodology, visualization. CC, FYC: conceptualization, data curation, writing–original draft. XQ: conceptualization, data curation, writing–original draft, supervision, validation, writing–review and editing. GGX and XZF contributed equally to this article. All the authors contributed to the critical review and final approval of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
No financial or non-financial benefits have been received or will be received from any party related directly or indirectly to the subject of this article.
Ethical approval
The study protocol was approval by the institution’s human research committee (Ethics Committee of Renji Hospital, No: KY2020-064). Informed consent to participate in the study has been obtained from their parent or legal guardian.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gu, GX., Pan, ST., Fan, YC. et al. Development and validation of a nomogram to predict allograft survival after pediatric liver transplantation. World J Pediatr 20, 239–249 (2024). https://doi.org/10.1007/s12519-023-00766-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12519-023-00766-y