Abstract
Larvae of lacewings (Neuroptera) are known to be fierce predators today. Most characteristic are their prominent piercing-sucking stylets, which are used for venom injection and sucking out the fluids of the prey. Among lacewing larvae, aphidlions (larvae of the groups Chrysopidae and Hemerobiidae, green and brown lacewings) are today highly specialised to feed on aphids and evolved strategies to not be detected and attacked by, e.g., aphid-protecting ants. Fossil relatives of modern aphidlions seem to have also employed other strategies. For the species Pedanoptera arachnophila from about 100-million-year-old Kachin amber, Myanmar, an interaction of its larvae with spiders has been assumed. We present here new specimens resembling these larvae, including one piece of Cretaceous Kachin amber with a syn-inclusion of an aphidlion-like larva and an immature planthopper, indicating planthoppers as potential prey of the group about 100 million years ago. The morphology of the lacewing larva, with a trapezoid head capsule (in dorsal view), simple, toothless stylets, very elongate legs, and a spindle-shaped trunk, indicates that it is conspecific or at least closely related to P. arachnophila. We reconstruct the possible ontogenetic sequence of Pedanoptera arachnophila and discuss its ecology.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Neuroptera, the group of lacewings, is a less species-rich ingroup of Holometabola, the larger group also including beetles (Coleoptera), bees and their relatives (Hymenoptera), butterflies and their relatives (Lepidoptera), flies and their relatives (Diptera), as well as some other minor groups (Trichoptera, Mecoptera, Raphidioptera, Megaloptera, Strepsiptera; Grimaldi & Engel, 2005). As most holometabolans, also lacewings have immature stages that are highly distinct from their corresponding adults concerning morphology and ecology (Aspöck & Aspöck, 1999, 2007), which are hence called larvae (see discussion in Haug, 2020).
Most lacewing larvae are highly specialised predators, with mandible and maxilla (upper and lower jaw) forming a so called stylet, facilitating the injection of venom and saliva as well as sucking out the liquefied prey (Aspöck & Aspöck, 2007; MacLeod, 1964; Zimmermann et al., 2019). In different lineages, these larvae show different specialisation for different strategies: many antlion larvae (Myrmeleontidae) are known to dig trap funnels (e.g., Hollis et al., 2015; Scharf et al., 2011); some aphidlions (larvae of Chrysopidae) use the corpses of their prey for chemical and optical camouflage (e.g., Hayashi & Nomura, 2011; Tauber et al., 2014); larvae of spoon-winged lacewings (Nemopterinae) dig deeper in the soil to hunt, up to 0.3 m (Miller & Stange, 2012).
While Neuroptera is a smaller group today, it is generally considered that it has been more diverse in the past, which also implies that lacewings also fulfilled more ecological roles in the past. This observation is supported by fossil lacewings with now extinct morphologies (e.g., Lu et al., 2020; Shi et al., 2020a, 2020b; Baranov et al., 2022; but see also Badano et al., 2018). Also for the larval forms, highly unusual appearing fossils are known, mostly preserved in amber (Haug et al., 2019a, 2019b; Luo et al., 2022; see discussion in Herrera-Flórez et al., 2020). These seem to have performed ecological functions not seen in the modern fauna (Haug et al., 2021). The difficulties for inferring the exact ecological role of such fossil larvae are coupled with the lack of a modern counterpart.
For fossil larvae strongly resembling modern ones, we can, based on phylogenetic and functional morphological comparison, infer with some reliability what they did back in time. For some exceptional finds we can even infer aspects of their behaviour, based on in situ preservations of interactions. Such finds include for example: (1) different types of lacewing larvae carrying camouflaging items (Hörnig et al., 2020; Liu et al., 2022; Pérez-de la Fuente et al., 2012, 2016, 2018; Wang et al., 2016) as in modern counterparts; (2) small mantis lacewing larvae interacting with spiders, just as in some modern species (Haug et al., 2018; Ohl, 2011); (3) a case of possible group defence (Hörnig et al., 2022) of larvae generally considered to resemble larvae of Ascalaphidae (cf. Wang et al., 2016; Note: currently the status of the group is not settled, some authors use Ascalaphinae; Machado et al., 2019; but see Badano et al., 2021; also the vernacular name “owlfly”, should be substituted by owllacewing) as known for some modern-day species (Henry, 1972); 4) a modern-appearing type of aphidlion (Hemerobiidae) preserved together with its prey in a single piece of amber (Haug et al., 2022a). All these cases represent interactions and principal morphologies also found in the modern fauna.
For now extinct morphologies, it usually proves much more difficult to infer similar details of interaction. Liu et al. (2016) reported unusually long-legged aphidlion-like larvae of the species Pedanoptera arachnophila, preserved in about 100-million-year-old Kachin amber. Due to certain aspects of the find, also of a supposed corresponding adult and in combination with a functional comparison to the likewise long-legged assassin bugs of the group Emesinae (e.g., Castro-Huertas & Forero, 2017; Forero, 2007; Gagné & Howarth, 1974; Resende et al., 2016) it was suggested that the long-legged larvae interacted with web-spinning spiders, either as predators or as kleptoparasites.
We here report new finds of long-legged aphidlion-like larvae. These are potentially conspecific to the known specimens of P. arachnophila, or are at least closely related to this species, but represent different ontogenetic stages. One specimen is preserved together with an immature planthopper and seems to have interacted with it. We discuss implications of these finds.
Material and methods
Material
Two specimens were investigated. Both specimens came from Hukawng Valley, Kachin State, Myanmar. Specimen BUB 3132 is deposited in the collection of one of the co-authors (PM), the other one is part of the PED research collection (Germany, Munich, Ludwig-Maximilians-Universität München, Palaeo-Evo-Devo Research Group Collection of Arthropods) under the accession number PED 2202. The specimen was bought on the online trading site ebay.com from the trader burmite-miner. Myanmar amber is interpreted as originating from the Cretaceous (about 100 mya; for details about locality, see, e.g., Hörnig et al., 2020). The raw amber piece of BUB 3132 was cut with a Dremel 3000 and afterwards polished with wet sandpaper grad 600, 1000 and 5000. Final polishing was done with Sidol metal polish.
Light-based documentation methods
The amber piece BUB 3132 was documented with a Canon EOS 70D camera, equipped with an MP-E 65 mm macro objective and a Macro Twin Lite MT-24 EX. Light was cross-polarised (polarisation filters in front of objective and light sources, rotated 90 degrees to each other) to reduce reflections within the amber and enhance colour contrast. To compensate for convexities, concavities, and fissures of the amber surface, the amber piece was either (1) submerged in distilled water, fixed with glass blocks and metal nuts, and covered with a cover slip, or (2) a drop of pure glycerine was applied to the amber surface and it was then covered with a cover slip.
Several image stacks were recorded. Images stacks (z-axis) were stacked with CombineZP (Alan Hadley) and resulting images were stitched (x–y-axis) with Adobe Photoshop CS4, resulting in consistently sharp high-resolution images.
Specimen PED 2202 was documented on a Keyence VHX-6000 digital microscope under unpolarised ring light in front of a white background.
X-ray micro-computed tomography (micro-CT imaging)
Micro-CT was performed using a XRadia MicroXCT-200 (Carl Zeiss Microscopy GmbH, Jena, Germany), which is equipped with switchable scintillator-lens units. 1600 projections were recorded under the following settings: 4 × objective; X-ray source settings: 30 kV, 6 W, source distance 60 mm; detector distance 20 mm; binning 2; with an exposure time of 3 s. Recorded projections were reconstructed with binning 1 (full resolution) using the XMReconstructor software (Carl Zeiss Microscopy GmbH, Jena, Germany), resulting in image stacks (TIFF format). System-based calculated pixel size is 5.01 µm, image size 1015 px × 1015 px. Volume renderings based on the micro-CT data were processed using OSIRIX.
Terminology
We briefly summarise the basic body organisation of representatives of the group Insecta in order to improve the communication with non experts (see Haug et al., 2012b). The body is usually organised into the head (ocular segment and post-ocular segments 1–5), and the trunk (post-ocular segments 6–19). The latter is furthermore differentiated into the anterior trunk or thorax (with pro-, meso- and metathorax; post-ocular segments 6–8) and the posterior trunk or abdomen (post-ocular segments 9–19) (compare e.g. Fortey & Thomas, 1997; Haug et al., 2012a for use of this terminology).
The head possesses two types of appendages, the antenna (appendage of post-ocular segment 1) and the mouthparts (appendages of ocular segment and appendages of post-ocular segments 3‒5; see e.g. Krenn, 2019). The head usually bears compound eyes and ocelli (non-holometabolan insects and adult holometabolans) or stemmata (larval holometabolans) (part of the ocular segment). All mouthparts and parts of the head of representatives of Hemiptera form a beak-like structure (unfortunately often called rostrum, which usually indicates an anterior orientation, yet it is backwards oriented).
The mandible and parts of the maxilla (appendages of post-ocular segments 3 and 4 respectively) of the lacewing larvae form so called stylets; the labium (medially conjoined appendages of post-ocular segment 5) of the same is only externally discernible as are the labial palps (distal elements of the labium, on each side respectively).
The thorax has dorsal sclerotisations (pro-, meso- and metanotum; tergites of post-ocular segments 6–8); additionally each segment possesses a pair of appendages, usually for walking (fore-, mid- and hindlegs; appendages of post-ocular segments 6–8). Each leg consists of five major externally discernible elements (from proximal to distal): coxa (most likely the basipod of closely related forms of Euarthropoda), trochanter, femur, tibia and tarsus (all these most likely representing the endopod) (e.g. Bruce & Patel, 2020; Haug et al., 2012a). The tarsus is often further subdivided into multiple, but maximum five tarsus elements, the tarsomeres; the distal most element is sometimes considered to be further subdivided into the distal tarsomere and the praetarsus (which includes the claws, when present). The abdomen also has dorsal sclerotisations (tergites of post-ocular segments 9–19) that are individually numbered within the abdomen (abdomen tergite 1 is the tergite of post-ocular segment 9 and so on).
Results
Description of amber piece BUB 3132
The piece includes two syninclusions, an immature planthopper and an aphidlion-like larva, and some surrounding unidentified plant material or debris. Both syninclusions are well and entirely preserved.
Description of the immature planthopper
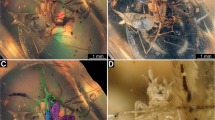
General. Entire specimen overall about 4.7 mm long without appendages and about 1.7 mm wide at its largest width (Fig. 1a, b). Body organised into head and trunk.
Specimen BUB 3132, aphidlion-like larva with immature planthopper in dorsal view. a Overview. b Colour-marked version of a, aphidlion-like larva in purple. a2–6 = abdominal segments 2–6 (post-ocular segments 10–14); be = beak; ce = compound eye; hc = head capsule; ms = mesothorax; mt = metathorax; pt = prothorax; te = trunk end
Head. Head about 0.8 mm wide at largest width and 0.5 mm long medially (dorsally discernible part) (Fig. 1). Laterally with slightly set-off compound eyes, circular in dorsal view and slightly longer than wide, with numerous ommatidia; one possible lateral ocellus discernible, medially to left compound eye, also circular in dorsal view, as wide as long (Fig. 1b); potentially two other ocelli present. Anterior region of head (vertex) triangular in dorsal view, tapering posteriorly, about as long as wide.
Antenna about 0.7 mm long; with two proximal elements and a distal flagellum (Figs. 2a, b, 3a–f). Two proximal elements (scapus, pedicellus) much wider than flagellum. Scapus trapezoid-shaped in overall ventral view, about as long as wide; pedicellus about square-shaped with rounded corners in overall ventral view, as wide as but slightly longer. Proximal element of flagellum square-shaped in ventral view, as long as wide. Flagellum distally with a long seta (arista; aristate-type antenna), longer than wide, about 35 × times, about 0.4 mm long.
Specimen BUB 3132, aphidlion-like larva with immature planthopper in ventral view. a Overview. b Colour-marked version of a, aphidlion-like larva in purple. a2–6 = abdomen segments 2–6 (post-ocular segments 10–14); be = beak; ce = compound eye; fe = femur; ms = mesothorax; mt = metathorax; pt = prothorax; ta = tarsus; ti = tibia; te = trunk end
Specimen BUB 3132, continued. Volume renderings based on micro-CT data. a Overview. b Colour-marked version of a, aphidlion-like larva in blue. c–f Close-up images of volume renderings of aphidlion-like larva in different angles and partly colour-marked. g Detailed photographic image of aphidlion-like larva. at = antenna; hc = head capsule; md = mandible; mx = maxilla; sy = stylet; tu = trunk. a, c, e are presented as red-cyan anaglyphic image, please use red-cyan glasses for 3D
Mouthparts and parts of head capsule form a beak-like structure (rostrum), attached postero-ventrally to head, entire structure about 4.9 mm long and longer than central body (Figs. 2a, b, 3a, b); overall very elongated hexagonal in ventral view, widest anteriorly (about 0.4 mm) and tapering significantly distally. Three overlapping parts more or less externally discernible; anterior part (frons) hexagonal in ventral view, longer than wide (about 2×), reaching to about antenna insertion area (Figs. 2b, 3b); laterally on each side about four circular structures (possible sensory pits), arranged in a rosette-like manner. Middle part (clypeus) rhomboid-shaped in ventral view, longer than wide, more than 2× at largest width, reaching to about insertion area of forelegs (Figs. 2a, b, 3a, b); posterior part (conjoined mouthparts; beak) very elongated hexagonal, many times longer than wide, about 3.9 mm long (Figs. 2a, b, 3a, b).
Anterior trunk. Pronotum trapezoid-shaped in dorsal view, wider than long, about 4×, midline with slightly concave ridge right next to it on each side respectively (median carinae); more pronounced convex ridge (lateral carina) in between midline and lateral edge of pronotum on each side; lateral to ridges two rosette-like arranged clusters of circular structures anteriorly and posteriorly (possible sensory pits); pronotum slightly overlapping subsequent tergite laterally (Fig. 1a, b).
Ventrally with pair of forelegs; about 2.4 mm long and 0.25 mm wide at its largest width, with four discernible elements (five assumed) (Fig. 2a, b). Coxa ovoid-shaped in ventral view, longer than wide, about 2×. Trochanter not discernible. Femur elongated rectangular in antero-median view, wider proximal, tapering slightly distally, longer than wide at maximum width, 2.5×. Tibia also elongated rectangular in anterior view, about as wide as distal part of femur, longer than wide, about 4×; medio-proximally with five elongated lanceolate shaped spines/setae. Tarsus also elongated rectangular in anterior view, about half as wide as tibia; longer than wide, about 8×; probably subdivided into three or four tarsomeres; proximal tarsomere with one possible spine at its latero-distal corner, no more spines discernible on tarsus, tarsomere 2 the longest tarsomere, claw on distal tarsomere not well discernible.
Mesonotum medially elongated rectangular in dorsal view, wider than long at midline length, more than 2×; midline with straight ridges right next to it on each side respectively (median carinae); with slight ridge (lateral carina) lateral to midline (on each side), slightly convex, oblique across the mesonotum, anteriorly at about one third between midline and lateral edge, and posteriorly at slightly less than halfway between midline and lateral edge. Latero-posteriorly on both sides of mesonotum protrusions (developing forewings), lobe-shaped, about as long as anterior part of mesonotum, no venation discernible (Fig. 1a, b).
Ventrally with pair of midlegs overall similar-shaped to foreleg, but longer, about 3.1 mm long and 0.26 mm wide at its largest width (Fig. 2a, b). Coxa elongated rectangular in anterior view, longer than wide, more than 3×, widening slightly distally. Trochanter circular in anterior view, about as long as wide. Femur more ovoid in anterior view, with a more convex lateral edge, and with similar spines or setae on its median edge as the tibia of the foreleg, longer than wide at maximum width, about 2.5×. Tibia largely similar to that of foreleg, but longer, with more spines/setae on its median edge and tapering slightly distally. Tarsus similar to that of foreleg, yet no spines discernible, but potentially the claws.
Metanotum similar to the mesonotum, but without ridges laterally to midline; with posterior protrusions (developing hindwings), lobe-shaped, laterally wider, no venation discernible (Fig. 1a, b).
Ventrally with pair of hindlegs overall similar to midleg, but slightly longer (Fig. 2a, b). Coxa rectangular in anterior view, wider than long, about 1.3×. Trochanter ovoid in anterior view, about as long as wide. Femur similar to that of foreleg, no spines/setae discernible. Tibia similar to that of midleg, also with spines/setae medially. Tarsus longer than that of midleg, subdivided into at least three tarsomeres. Proximal tarsomere longest, over half of total tarsus length, with distinct spines on its distal-most corners, one more medially and two more laterally; middle tarsomere about half the length of proximal one. Distal tarsomere half of the length of middle tarsomere; no claw discernible.
Posterior trunk. Abdomen overall ovoid in dorsal view, wider anteriorly and tapering to about half the anterior width posteriorly (Figs. 1a, b, 2a, b). Anterior tergites all rectangular in dorsal view with slightly convex lateral edges, wider than long, about 3×; with laterally rosette-like cluster of circular structures (possible sensory pits) similar to the structures on the pronotum; discernible on abdomen tergite 3 (left side), abdomen tergite 5 (right side) and on abdomen tergite 6 (right side) (Fig. 1a, b). Other lateral parts of especially anterior abdominal tergites obscured. Abdomen tergite 6 with very concave posterior edge and postero-lateral corners protruding posteriorly about as long as midline. Abdomen tergite 7 (pygofer) U-shaped in dorsal view, with posterior protrusions laterally tapering posteriorly; with smaller setae along its median edge and surrounding the trunk end. Trunk end (possible compound structure of several segments) anteriorly square-shaped in dorsal view, with two posterior protrusions; protrusions elongated triangular, longer than anterior, square-shaped part, about 1.5×.
Description of aphidlion-like larva 1
General. Entire specimen about 1.3 mm long without appendages and 0.4 mm wide at its largest width (Figs. 1, 2, 3a–g). Body organised into head and trunk.
Head. Head capsule rounded trapezoid-shaped in dorsal view, with wider anterior edge and rounded posterior edge; about 0.3 mm long and wide at its largest width anteriorly (without appendages) (Fig. 3). Stemmata not discernible.
Antenna about 0.6 mm long and about 0.02 mm wide at its largest width (Fig. 3a, b); no individual elements discernible.
Stylets directed forwards; sickle-shaped (Fig. 3a–g); about 0.4 mm long and about 0.1 mm wide at its widest most proximally, about as long as the head capsule; tapering slightly distally, no median protrusions (teeth) discernible, stylets medially curved. No labial palps discernible.
Anterior trunk. Tergites of the thorax elongated rectangular in dorsal view, wider than long, 3–4×. Pronotum with a dorsal protrusion laterally on each side respectively; protrusions tapering distally. Foreleg apparently longest walking appendage, about 2.4 mm long and 0.04 mm wide at its largest width (Fig. 2a, b); no individual elements discernible, but one joint apparent (probably femur-tibia joint). Midleg apparently shortest walking appendage, about 1.8 mm long and 0.03 mm wide at its largest width. Hindleg about 2.1 mm long and 0.04 mm wide at its largest width.
Posterior trunk. Abdomen overall ovoid, widest anteriorly and tapering significantly posteriorly (Fig. 1a, b); individual segments difficult to discern, probably elongated rectangular in dorsal view, wider than long.
Description of amber piece PED 2202
The piece includes one aphidlion-like larva (No. 2), some unknown (organic?) debris and many small droplets of unknown content (comp. Fig. 4a, c, d, f).
Specimen PED 2202. a Ventral view. b Colour-marked version of a. c Dorsal view. d Detail of head in ventral view. e Colour-marked version of d. f. Detail of tip of labial palp. a5 = abdomen segment 5; at = antenna; hc = head capsule; lp = labial palp; ms = mesothorax; metathorax; st = stemmata; sy = stylet; te = trunk end
Description of aphidlion-like larva 2
General. Entire specimen about 2.4 mm long without appendages and 0.5 mm wide at its largest width (Fig. 4). Body organised into head and trunk.
Head. Head capsule rounded trapezoid-shaped in ventral view, with wider anterior edge and rounded posterior edge; about 0.4 mm long and 0.3 mm wide at its largest width anteriorly (without appendages) (Fig. 4a–e); with some prominent scale-shaped processes discernible in ventral view (Fig. 4a, b, d, e). Stemmata antero-laterally discernible, at least three ocelli discernible in ventral view (Fig. 4e, f).
Antenna about 1.2 mm long and about 0.05 mm wide at its largest width most distally (Fig. 4a–c); about 16 individual elements discernible.
Stylets directed forwards; sickle-shaped (Fig. 4a–e); about 0.8 mm long and about 0.08 mm wide at its widest most proximally, about twice as long as the head capsule; tapering slightly distally, no median protrusions (teeth) discernible, stylets medially curved.
Labial palps about 0.8 mm long and 0.05 mm wide at widest; about twice as long as head capsule. About five individual elements discernible; most distal element widening medio-distally and tapering very distally into pointed tip (Fig. 4d–f).
Anterior trunk. Thorax: Pronotum hexagonal in dorsal view, slightly wider than long, and without dorsal protrusion laterally as in aphidlion-like larva 1.
Ventrally with pair of forelegs; about 3 mm long and 0.1 mm wide at its largest width, with five discernible elements (Fig. 4a–c), with some processes (setae?), some of which apparently scale-shaped. Coxa rectangular-shaped in ventral view, longer than wide, about 2×. Trochanter circular-shaped in posterior view. Femur elongated rectangular in posterior view, widening distally, longer than wide at maximum width, 7.6×. Tibia also elongated rectangular in posterior view, about 0.4× the width of femur, longer than wide, about 30×; with two setae/spines at distal-most median corner. Tarsus also elongated rectangular in posterior view, about as wide as tibia; longer than wide, about 16×; apparently not subdivided, distally with two claws discernible, no empodium apparent.
Mesonotum hexagonal in dorsal view, wider than long, about 2×. Ventrally with pair of midlegs overall very similar to forelegs and same length and widest width (Fig. 4a–c). Femur, tibia and tarsus with distinct row of setae along median edge and all around these elements (and the other elements) as well; tibia similar to that of foreleg with two setae/spines at distal-most median corner.
Metanotum similar to the mesonotum, but overall slightly larger, but just 1.7 × wider than long (Fig. 4c). Ventrally with pair of hindlegs overall similar to midlegs, but much longer, 3.8 mm long and about 0.1 mm wide at its largest width (Fig. 4a–c). Tibia and tarsus with distinct row of setae along median edge and all around these elements (and the other elements) as well.
Posterior trunk. Abdomen overall very elongated ovoid in ventral view, wider anteriorly and tapering to about half the anterior width very posteriorly (Fig. 4a–c). Anterior tergites all rectangular in ventral view with slightly convex lateral edges, wider than long, about 2.7–4.5×. Abdomen tergite 6–8 slightly longer than preceding tergites, but still wider than long, 1.6–2.1×. Trunk end (possible compound structure of several segments) trapezoid with very round posterior tip in ventral view, slightly wider than long at its largest width anteriorly.
Discussion
Identity of the fulgoromorphan hemipteran
The larger specimen in amber piece BUB 3132 has a remarkable long beak (“rostrum”) and a rather unusual overall shape, strongly resembling immatures of the group Neazoniidae, an extinct ingroup of Fulgoromorpha known from older, Lebanese amber (Szwedo, 2007), but also reminds of representatives of the group Mimarachnidae. All (immature) representatives of Neazoniidae share an overall flattened body and the rosette-like arranged sensory pits on the head (specifically the frons) and on the tergites of thorax and abdomen. More precisely, the new specimen strongly resembles Neazonia immatura. The diagnostic characters of the latter, like the pronotum shape and that the tibia of the midleg is longer than its femur, can also be observed in the herein described specimen. It additionally also shares a similar-appearing mesonotum and tarsus of the hindleg. Yet, given the 30 million years of time difference between Lebanon amber and Myanmar amber, it is unlikely that the species persisted over this long time span. Still, representatives of Neazoniidae are known from about 100-million-year-old French amber (Szwedo, 2009). We therefore suggest that the new specimen could be a representative of Neazoniidae, closely related to N. immatura.
It is also necessary to consider the similarities to Mimarachnidae. The challenge in this comparison is that the latter group has been largely based on adults (example for exception: Emeljanov & Shcherbakov, 2018). It is therefore well possible that Mimarachnidae and Neazoniidae are either closely related, or even that one group could be an ingroup of the other (just based on species number, it is more likely that Neazoniidae is an ingroup of Mimarachnidae). This makes it well plausible that the new larvae could also be an ingroup of Mimarachnidae. For further interpreting this aspect, it will be necessary to resolve the relationship of Neazoniidae and Mimarachnidae in the future.
Identity of the lacewing larva in amber piece BUB 3132 and PED 2202
The here reported lacewing larvae clearly show traits characteristic for aphidlion-like larvae. These are an (in dorsal view) rounded trapezoidal head capsule, strongly forward oriented mouthparts, and each upper and lower jaw forming a curved, toothless stylet.
Quite remarkable in the specimens are the long legs. Such long legs are only known in aphidlion-like larvae, specifically those of Pedanoptera arachnophila (Liu et al., 2016). Not many additional details are accessible in the newly reported larvae.
Both aphidlion-like larvae in amber pieces BUB 3132 and PED 2202 differ in certain aspects from the larvae of P. arachnophila reported by Liu et al. (2016).
Differences of aphidlion-like larva 1 (BUB 3132) to P. arachnophila include: (1) this new larva is significantly smaller. (2) This new larva has two short dorsal processes not present in the larvae of P. arachnophila reported by Liu et al. (2016). (3) This new larva has a relatively shorter antenna, which is longer than the head width, but only about 2×. However, in the larvae of P. arachnophila it is also longer than the head width, but about 3.3×. (4). The three pairs of legs appear to be more or less similar in length, while in the larvae of P. arachnophila reported by Liu et al. (2016) the middle legs are significantly shorter than the other legs. In detail, also the middle legs of the new larva are shorter, but the difference is rather subtle.
Differences of aphidlion-like larva 2 (PED 2202) to the aphidlion-like larva 1 (BUB 3132) and the larvae of P. arachnophila include: (1) The aphidlion-like larva 2 is significantly smaller than P. arachnophila, but slightly larger than the aphidlion-like larva 1. (2) The aphidlion-like larva 2 does not have two short dorsal processes as does the aphidlion-like larva 1 (so it is similar to P. arachnophila in this aspect). Additionally aphidlion-like larva 2 has some scale-shaped processes especially in the head region that are not found in the other larvae. (3) The aphidlion-like larva 2 has equally long fore- and midlegs, but the hindlegs are the longest, which is different from both the aphidlion-like larva 1 and the larvae of P. arachnophila. (4) The antenna of the aphidlion-like larva 2 is longer than the head width, but about 3.5×, which is longer than the antenna of the aphidlion-like larva 1, but almost equal to the antenna length of the larvae of P. arachnophila.
Given the size differences between the known larvae, these morphological differences may represent ontogenetic differences. It is therefore well possible that the larvae are conspecific or at least closely related, but represent different developmental stages. From a formal view, there is no diagnostic character that would allow us to differentiate the new larva from P. arachnophila.
Possible ontogenetic sequence of Pedanoptera arachnophila
Already the four larvae reported by Liu et al. (2016) were interpreted to represent different ontogenetic stages. Two of these were interpreted as final larvae, one as a stage 3 larva, and one as a stage 2 larva. As most lacewings develop through three larval stages (see discussion in Haug, 2020; Haug et al., 2020), three of the specimens reported by Liu et al. (2016) should represent a single stage. Yet, only two of these have similar dimensions, while the third specimen has different proportions of the central body and the first two pairs of walking legs. These differences might therefore not represent ontogenetic differences, but species differences. The supposed stage 2 larva could be indeed conspecific with the first two. The two new larvae are also differing in certain aspects from the four larvae reported by Liu et al. (2016), but clearly represent earlier stages. The small specimen on the fulgoromorphan (BUB 3132) could be conspecific either with the three specimens from Liu et al. (2016) or the single one based on the relative lengths of the walking legs (Fig. 5); concerning its size it would well fit with an interpretation as a stage 1 larva. The other new larva (PED 2202) does not match well with any of the others due to differences in the relative leg lengths; it could represent a stage 2 larva (but this is speculative) of another species. It seems therefore likely that the material includes at least three different species This situation is comparable to that in other specialised larvae (e.g. Badano et al., 2021) where there are certain differences recognisable, indicating the presence of several species, yet properly diagnosing them remains challenging.
Comparison of the specimens possibly representing Pedanoptera arachnophila or a closely related species, all to the same scale. Based on Liu et al. (2016). a PCWT TA-0001; supposed stage 3 larva of P. arachnophila. b PCXJ BA-0005; supposed stage 3 larva of P. arachnophila. c EMTG BU-001113; supposed stage 3 larva, possibly of a different species. d EMTG BU-001111; supposed stage 2 larva of P. arachnophila. e PED 2202; possible stage 2 larva of yet another species. f BUB 3132; possible stage 1 larva of P. arachnophila
Possible interactions
The new aphidlion-like larva 1 (BUB 3132) is apparently in direct contact with the fulgoromorphan. It seems therefore possible that this may represent a syn-vivo interaction, i.e., original interaction, and not a simple effect of the embedding. Smaller animals on larger animals may be understood as cases of parasitism or phoresy. Yet, as the fulgoromorphan is still immature, not possessing wings, its capabilities in providing effective long-distance transport or dispersal for the aphidlion-like larva, an interpretation as a case of phoresis, seems less likely.
There are some cases of parasitism performed by lacewing larvae that also have a fossil record, such as first stage larvae of Mantispidae (Haug et al., 2018; Ohl, 2011) or larvae of Sisyridae (Haug et al., 2022b and references therein). Yet, the sizes of the lacewing larva and the fulgoromorphan make such an interpretation less likely. A comparable case has been discussed for a mite feeding on another lacewing larva (Hörnig et al., 2020). Predators do not necessarily have to be larger than their prey. While explicit data on this aspect becomes rarely published, there is an interesting example in Devetak and Arnett (2010). The authors reported a click beetle larva subduing an only slightly longer, but overall much larger antlion larva. For aphidlions there are several studies on prey preferences, but mainly focusing on pest species (e.g., Mhaske et al., 2019; Pappas et al., 2007; Rana et al., 2017). However, even if it seems less frequently observed, there are reports of aphidlions preying on larger insects than themselves (Huang & Enkegaard, 2010; Michaud & Grant, 2003; Pariser, 1917).
Also, it should be taken into account that most aphidlions indeed prey on aphids. Fulgoromorphans are not only closely related to aphids, but also have a rather similar ecological function (feeding on plant saps) and are to be expected in similar (if not the same) habitat. There are no reliable numbers how often modern aphidlions consume planthoppers, yet it is well known that leafhoppers can make up a major share of the diet of an aphidlion (e.g., Rana et al., 2017). It should therefore not be surprising to see an aphidlion-like larva preying on a planthopper.
It therefore seems that at least the early stages of P. arachnophila may have had rather “normal” feeding habits compared to its relatives. If P. arachnophila indeed interacted with spiders, as suggested by Liu et al. (2016), this seems to have been potentially restricted to the later larval stages. Yet, given the possibility that the fulgoromorphan might be a representative of Mimarachnidae, a group of which some of its representatives are thought to mimic spiders, the situation may be even more complex. More syninclusions will be necessary to further enlighten these aspects.
Data availability
Micro-CT data can be shared by the corresponding author upon request. All further data is provided in the text and the figures.
References
Aspöck, U., & H. Aspöck. (1999). Kamelhälse, Schlammfliegen, Ameisenlöwen. Wer sind sie? (Insecta: Neuropterida: Raphidioptera, Megaloptera, Neuroptera). Stapfia 60 / Kataloge der Oberösterreichischen Landesmuseen Neue Folge Nr. 138: 1–34. (in German).
Aspöck, U., & H. Aspöck. (2007). Verbliebene Vielfalt vergangener Blüte. Zur Evolution, Phylogenie und Biodiversität der Neuropterida (Insecta: Endopterygota). Denisia 20 / Kataloge der Oberösterreichischen Landesmuseen Neue Serie 66: 451–516. (in German).
Badano, D., Engel, M. S., Basso, A., Wang, B., & Cerretti, P. (2018). Diverse Cretaceous larvae reveal the evolutionary and behavioural history of antlions and lacewings. Nature Communications, 9(1), 3257. https://doi.org/10.1038/s41467-018-05484-y
Badano, D., Fratini, M., Maugeri, L., Palermo, F., Pieroni, N., Cedola, A., Haug, J. T., Weiterschan, T., Velten, J., Mei, M., Di Giulio, A., & Cerretti, P. (2021). X-ray microtomography and phylogenomics provide insights into the morphology and evolution of an enigmatic Mesozoic insect larva. Systematic Entomology, 46(3), 672–684. https://doi.org/10.1111/syen.12482
Baranov, V., Pérez-de la Fuente, R., Engel, M. S., Hammel, J. U., Kiesmüller, C., Hörnig, M. K., Pazinato, P. G., Stahlecker, C., Haug, C., & Haug, J. T. (2022). The first adult mantis lacewing from Baltic amber, with an evaluation of the post-Cretaceous loss of morphological diversity of raptorial appendages in Mantispidae. Fossil Record, 25(1), 11–24. https://doi.org/10.3897/fr.25.80134
Bruce, H. S., & Patel, N. H. (2020). Knockout of crustacean leg patterning genes suggests that insect wings and body walls evolved from ancient leg segments. Nature Ecology & Evolution, 4(12), 1703–1712. https://doi.org/10.1038/s41559-020-01349-0
Castro-Huertas, V., & Forero, D. (2017). Small range distributions in the high Andes: two new species of Liaghinella (Hemiptera: Heteroptera: Reduviidae: Emesinae) from Colombia. Zootaxa, 4277(3), 399–412. https://doi.org/10.11646/zootaxa.4277.3.5
Devetak, D., & Arnett, A. E. (2010). A click beetle larva (Coleoptera: Elateridae) preying upon an antlion larva (Neuroptera: Myrmeleontidae). Entomological News, 121(5), 514–516. https://doi.org/10.3157/021.121.0515
Emeljanov, A. F., & Shcherbakov, D. E. (2018). The longest-nosed Mesozoic Fulgoroidea (Homoptera): A new family from mid-Cretaceous Burmese amber. Far Eastern Entomologist, 354, 1–14. https://doi.org/10.25221/fee.354.1
Forero, D. (2007). Description of a new species of Liaghinella (Hemiptera: Heteroptera: Reduviidae: Emesinae) from the Colombian Andes, with notes on its feeding habits and conservation status. Zootaxa, 1502(1), 55–68. https://doi.org/10.11646/zootaxa.1502.1.3
Fortey, R. A., & Thomas, R. H. (1997). Arthropod relationships. The Systematics Association Special Volume Series 55 (p. 383). Springer Science & Business Media.
Gagné, W.C., & F.G. Howarth. (1974). The cavernicolous fauna of Hawaiian lava tubes, Part VII. Emesinae or thread-legged bugs (Heteroptera: Redvuiidae). Technical Report 43, Island Ecosystems IRP, U.S. International Biological Program, 1–18. http://hdl.handle.net/10125/26964
Grimaldi, D. A., & Engel, M. S. (2005). Evolution of the Insects (p. 755). Cambridge University Press.
Haug, C., Herrera-Flórez, A. F., Müller, P., & Haug, J. T. (2019a). Cretaceous chimera—an unusual 100-million-year old neuropteran larva from the “experimental phase” of insect evolution. Palaeodiversity, 12(1), 1–11. https://doi.org/10.18476/pale.v12.a1
Haug, J. T. (2020). Why the term “larva” is ambiguous, or what makes a larva? Acta Zoologica, 101(2), 167–188. https://doi.org/10.1111/azo.12283
Haug, J. T., Baranov, V., Müller, P., & Haug, C. (2021). New extreme morphologies as exemplified by 100 million-year-old lacewing larvae. Scientific Reports, 11, 20432. https://doi.org/10.1038/s41598-021-99480-w
Haug, J. T., Baranov, V., Schädel, M., Müller, P., Gröhn, C., & Haug, C. (2020). Challenges for understanding lacewings: how to deal with the incomplete data from extant and fossil larvae of Nevrorthidae? (Neuroptera). Fragmenta Entomologica, 52(2), 137–167. https://doi.org/10.13133/2284-4880/472
Haug, J. T., Briggs, D. E. G., & Haug, C. (2012a). Morphology and function in the Cambrian Burgess Shale megacheiran arthropod Leanchoilia superlata and the application of a descriptive matrix. BMC Evolutionary Biology, 12, 162. https://doi.org/10.1186/1471-2148-12-162
Haug, J. T., Kiesmüller, C., Haug, G. T., Haug, C., & Hörnig, M. K. (2022a). A fossil aphidlion preserved together with its prey in 40 million-year-old Baltic amber. Palaeobiodiversity and Palaeoenvironments. https://doi.org/10.1007/s12549-021-00521-z
Haug, J. T., Maas, A., Haug, C., Waloszek, D., Watling, L., & Thiel, M. (2012b). Evolution of crustacean appendages. In L. Watling & M. Thiel (Eds.), Functional Morphology & Diversity. The Natural History of the Crustacea (Vol 1) (pp. 34–73). Oxford University Press. https://doi.org/10.1093/acprof:osobl/9780195398038.003.0002
Haug, J. T., Müller, P., & Haug, C. (2018). The ride of the parasite: A 100-million-year old mantis lacewing larva captured while mounting its spider host. Zoological Letters, 4, 31. https://doi.org/10.1186/s40851-018-0116-9
Haug, J. T., Müller, P., & Haug, C. (2019b). A 100-million-year old slim insectan predator with massive venom-injecting stylets—a new type of neuropteran larva from Burmese amber. Bulletin of Geosciences, 94(4), 431–440. https://doi.org/10.3140/bull.geosci.1753
Haug, J. T., Zippel, A., van der Wal, S., Gröhn, C., Hoffeins, C., Hoffeins, H.-W., & Haug, C. (2022b). Diversity and fossil record of larvae of three groups of lacewings with unusual ecology and functional morphology: Ithonidae. Coniopterygidae and Sisyridae. Palaeontologia Electronica, 25(2), a14. https://doi.org/10.26879/1212
Hayashi, M., & Nomura, M. (2011). Larvae of the green lacewing Mallada desjardinsi (Neuroptera: Chrysopidae) protect themselves against aphid-tending ants by carrying dead aphids on their backs. Applied Entomology and Zoology, 46(3), 407–413. https://doi.org/10.1007/s13355-011-0053-y
Henry, C. S. (1972). Eggs and rapagula of Ululodes and Ascaloptynx (Neuroptera: Ascalaphidae): a comparative study. Psyche: A Journal of Entomology, 79(1–2), 1–22. https://doi.org/10.1155/1972/54050
Herrera-Flórez, A. F., Braig, F., Haug, C., Neumann, C., Wunderlich, J., Hörnig, M. K., & Haug, J. T. (2020). Identifying the oldest larva of a myrmeleontiformian lacewing—A morphometric approach. Acta Palaeontologica Polonica, 65(2), 235–250. https://doi.org/10.4202/app.00662.2019
Hollis, K. L., Harrsch, F. A., & Nowbahari, E. (2015). Ants vs. antlions: An insect model for studying the role of learned and hard-wired behavior in coevolution. Learning and Motivation, 50, 68–82. https://doi.org/10.1016/j.lmot.2014.11.003
Hörnig, M. K., Haug, C., Müller, P., & Haug, J. T. (2022). Not quite social—possible cases of gregarious behaviour of immatures of various lineages of Insecta preserved in 100 million-year-old amber. Bulletin of Geosciences, 97(1), 69–87. https://doi.org/10.3140/bull.geosci.1818
Hörnig, M. K., Kiesmüller, C., Müller, P., Haug, C., & Haug, J. T. (2020). A new glimpse on trophic interactions of 100-million-year old lacewing larvae. Acta Palaeontologica Polonica, 65(4), 777–786. https://doi.org/10.4202/app.00677.2019
Huang, N., & Enkegaard, A. (2010). Predation capacity and prey preference of Chrysoperla carnea on Pieris brassicae. BioControl, 55(3), 379–385. https://doi.org/10.1007/s10526-009-9254-5
Krenn, H. W. (2019). Form and function of insect mouthparts. In H. Krenn (Ed.), Insect Mouthparts. Form, Function, Development and Performance, Zoological Monographs (Vol. 5, pp. 361–385). Springer Nature Switzerland AG. https://doi.org/10.1007/978-3-030-29654-4_2
Liu, H., Luo, C., Jarzembowski, E. A., & Xiao, C. (2022). Acanthochrysa langae gen. et sp. nov., a new lacewing larva (Neuroptera: Chrysopoidea) from mid-Cretaceous Kachin amber. Cretaceous Research, 133, 105146. https://doi.org/10.1016/j.cretres.2022.105146
Liu, X., Zhang, W., Winterton, S. L., Breitkreuz, L. C., & Engel, M. S. (2016). Early morphological specialization for insect-spider associations in Mesozoic lacewings. Current Biology, 26(12), 1590–1594. https://doi.org/10.1016/j.cub.2016.04.039
Lu, X., Wang, B., Zhang, W., Ohl, M., Engel, M. S., & Liu, X. (2020). Cretaceous diversity and disparity in a lacewing lineage of predators (Neuroptera: Mantispidae). Proceedings of the Royal Society B, 287(1928), 20200629. https://doi.org/10.1098/rspb.2020.0629
Luo, C., Liu, H., & Jarzembowski, E. A. (2022). High morphological disparity of neuropteran larvae during the Cretaceous revealed by a new large species. Geological Magazine, 159(6), 954–962. https://doi.org/10.1017/S0016756822000176
Machado, R. J., Gillung, J. P., Winterton, S. L., Garzón-Orduña, I. J., Lemmon, A. R., Lemmon, E. M., & Oswald, J. D. (2019). Owlflies are derived antlions: Anchored phylogenomics supports a new phylogeny and classification of Myrmeleontidae (Neuroptera). Systematic Entomology, 44(2), 418–450. https://doi.org/10.1111/syen.12334
MacLeod, E.G. (1964). A comparative morphological study of the head capsule and cervix of larval Neuroptera (Insecta). Ph.D. dissertation. Cambridge, Massachusetts, USA: Harvard University, p 528.
Mhaske, S. H., Shetgar, S. S., & Dake, R. B. (2019). Consumption capacity of Chrysoperla zastrowi sillemi (Esben-Peterson) on mealy bugs and aphids. Journal of Entomology and Zoology Studies, 7(3), 810–812.
Michaud, J. P., & Grant, A. K. (2003). Intraguild predation among ladybeetles and a green lacewing: Do the larval spines of Curinus coeruleus (Coleoptera: Coccinellidae) serve a defensive function? Bulletin of Entomological Research, 93(6), 499–505. https://doi.org/10.1079/BER2003269
Miller, R. B., & Stange, L. A. (2012). A new species of Stenorrhachus McLachlan from Chile (Neuroptera: Nemopteridae) with biological notes. Insecta Mundi, 226, 1–8.
Ohl, M. (2011). Aboard a spider—a complex developmental strategy fossilized in amber. Naturwissenschaften, 98, 453–456. https://doi.org/10.1007/s00114-011-0783-2
Pappas, M. L., Broufas, G. D., & Koveos, D. S. (2007). Effects of various prey species on development, survival and reproduction of the predatory lacewing Dichochrysa prasina (Neuroptera: Chrysopidae). Biological Control, 43(2), 163–170. https://doi.org/10.1016/j.biocontrol.2007.07.006
Pariser, K. (1917). Beiträge zur Biologie und Morphologie der einheimischen Chrysopiden. Archiv Für Naturgeschichte, Abteilung A, 83(11), 1–57. in German.
Pérez-de la Fuente, R., Delclòs, X., Peñalver, E., & Engel, M. S. (2016). A defensive behavior and plant-insect interaction in Early Cretaceous amber—the case of the immature lacewing Hallucinochrysa diogenesi. Arthropod Structure & Development, 45(2), 133–139. https://doi.org/10.1016/j.asd.2015.08.002
Pérez-de la Fuente, R., Delclòs, X., Peñalver, E., Speranza, M., Wierzchos, J., Ascaso, C., & Engel, M. S. (2012). Early evolution and ecology of camouflage in insects. Proceedings of the National Academy of Sciences, 109(52), 21414–21419. https://doi.org/10.1073/pnas.1213775110
Pérez-de la Fuente, R., Peñalver, E., Azar, D., & Engel, M. S. (2018). A soil-carrying lacewing larva in Early Cretaceous Lebanese amber. Scientific Reports, 8, 16663. https://doi.org/10.1038/s41598-018-34870-1
Rana, L. B., Mainali, R. P., Regmi, H., & RajBhandari, B. P. (2017). Feeding efficiency of green lacewing, Chrysoperla carnea (Stephens) against different species of aphid in laboratory conditions. International Journal of Applied Sciences and Biotechnology, 5(1), 37–41. https://doi.org/10.3126/ijasbt.v5i1.16983
Resende, L. P., Zepon, T., Bichuette, M. E., Pape, R. B., & Gil-Santana, H. (2016). Associations between Emesinae heteropterans and spiders in limestone caves of Minas Gerais, southeastern Brazil. Neotropical Biology and Conservation, 11(3), 114–121. https://doi.org/10.4013/nbc.2016.113.01
Scharf, I., Lubin, Y., & Ovadia, O. (2011). Foraging decisions and behavioural flexibility in trap-building predators: A review. Biological Reviews, 86(3), 626–639. https://doi.org/10.1111/j.1469-185X.2010.00163.x
Shi, C., Yang, Q., Shih, C., Labandeira, C. C., Pang, H., & Ren, D. (2020a). Cretaceous mantid lacewings with specialized raptorial forelegs illuminate modification of prey capture (Insecta: Neuroptera). Zoological Journal of the Linnean Society, 190(3), 1054–1070. https://doi.org/10.1093/zoolinnean/zlaa096
Shi, C., Yang, Q., Winterton, S. L., Pang, H., & Ren, D. (2020b). Stem-group fossils of Symphrasinae shed light on early evolution of Mantispidae (Insecta, Neuroptera). Papers in Palaeontology, 6(1), 143–154. https://doi.org/10.1002/spp2.1265
Szwedo, J. (2007). Nymphs of a new family Neazoniidae fam n (Hemiptera: Fulgoromorpha: Fulgoroidea) from the Lower Cretaceous Lebanese amber. African Invertebrates, 48(1), 127–143.
Szwedo, J. (2009). First discovery of Neazoniidae (Insecta, Hemiptera, Fulgoromorpha) in the Early Cretaceous amber of Archingeay SW France. Geodiversitas, 31(1), 105–116. https://doi.org/10.5252/g2009n1a9
Tauber, C. A., Tauber, M. J., & Albuquerque, G. S. (2014). Debris-carrying in larval Chrysopidae: Unraveling its evolutionary history. Annals of the Entomological Society of America, 107(2), 295–314. https://doi.org/10.1603/AN13163
Wang, B., Xia, F., Engel, M. S., Perrichot, V., Shi, G., Zhang, H., Chen, J., Jarzembowski, E. A., Wappler, T., & Rust, J. (2016). Debris-carrying camouflage among diverse lineages of Cretaceous insects. Science Advances, 2(6), e1501918. https://doi.org/10.1126/sciadv.1501918
Zimmermann, D., Randolf, S., & Aspöck, U. (2019). From chewing to sucking via phylogeny—from sucking to chewing via ontogeny: mouthparts of Neuroptera. In H. Krenn (Ed.), Insect Mouthparts. Form, Function, Development and Performance, Zoological Monographs (Vol. 5, pp. 361–385). Springer Nature Switzerland AG.
Acknowledgements
CK was supported by the Landesgraduiertenförderung MV. JTH is kindly funded by the Volkswagen Foundation in the frame of a Lichtenberg professorship. The German Research Foundation (DFG) supported the study with a grant to JTH (DFG HA 6300/6-1). Micro-computed tomography was performed at the Imaging Center of the Department of Biology, University of Greifswald (DFG INST 292/119-1 FUGG; DFG INST 292/120-1 FUGG). Steffen Harzsch, Greifswald, and J. Matthias Starck, Munich, are thanked for long-term support. We thank all people providing low cost, open access and open source software. This is LEON publication #44.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation was done by Patrick Müller, data collection and analysis were performed by Christine Kiesmüller, Joachim T. Haug and Gideon T. Haug. The first draft of the manuscript was written by Joachim T. Haug, Christine Kiesmüller, Marie K. Hörnig and Carolin Haug and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. This study was supported by the Landesgraduiertenförderung MV (Christine Kiesmüller), the Volkswagen Foundation: Lichtenberg professorship (Joachim T. Haug) and the DFG (DFG HA 6300/6-1 (Joachim T. Haug); DFG INST 292/119-1 FUGG; DFG INST 292/120-1 FUGG).
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Handling Editor: Ulrich Kotthoff.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kiesmüller, C., Haug, G.T., Haug, C. et al. New indications for the life habits of long-legged aphidlion-like larvae in about 100-million-year-old amber. PalZ (2024). https://doi.org/10.1007/s12542-024-00693-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12542-024-00693-x