Abstract
Excessive production of reactive oxygen species (ROS) by NADPH oxidase (Nox) resulted in inflammation. The negative regulator of ROS (NRROS) dampens ROS generation during inflammatory responses. 15-Deoxy-∆12,14-prostaglandin J2 (15d-PGJ2) exhibits neuroprotective effects on central nervous system (CNS). However, whether 15d-PGJ2-induced NRROS expression was unknown in rat brain astrocytes (RBA-1). NRROS expression was determined by Western blot, RT/real-time PCR, and promoter activity assays. The signaling components were investigated using pharmacological inhibitors or specific siRNAs. The interaction between transcription factors and the NRROS promoter was investigated by chromatin immunoprecipitation assay. Upregulation of NRROS on the hydrogen peroxide (H2O2)-mediated ROS generation and interleukin 6 (IL-6) secretion was measured. 15d-PGJ2-induced NRROS expression was mediated through PI3K/Akt-dependent activation of Sp1 and FoxO1 and established the essential promoter regions. We demonstrated that 15d-PGJ2 activated PI3K/Akt and following by cooperation between phosphorylated nuclear FoxO1 and Sp1 to initiate the NRROS transcription. In addition, Nrf2 played a key role in NRROS expression induced by 15d-PGJ2 which was mediated through its phosphorylation. Finally, the NRROS stable clones attenuated the H2O2-induced ROS generation and expression of IL-6 through suppressing the Nox-2 activity. These results suggested that 15d-PGJ2-induced NRROS expression is mediated through a PI3K/Akt-dependent FoxO1 and Sp1 phosphorylation, and Nrf2 cascade, which suppresses ROS generation through attenuating the p47phox phosphorylation and gp91phox formation and IL-6 expression in RBA-1 cells. These results confirmed the mechanisms underlying 15d-PGJ2-induced NRROS expression which might be a potential strategy for prevention and management of brain inflammatory and neurodegenerative diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Astrocytes exert several functions including production of growth factors and participating in the immune and repairing responses to brain injuries (Blackburn et al. 2009). In homeostatic maintenance, astrocytes interact with neurons to facilitate ROS detoxification during oxidative insults (Shih et al. 2003) which are involved in neurodegenerative diseases (Ricci et al. 2009). Importantly, astrocytes exert neuroprotection such as uptake of excitotoxic glutamate, protecting against oxidative stress, and limiting the spread of inflammatory cells or infectious agents (Sofroniew and Vinters 2010). Our previous studies indicate that excessive ROS generation in activated astrocytes induces the expression of inflammatory mediators resulting in neuronal apoptosis (Yang et al. 2013).
Excessive ROS could impair cellular functions and enhance inflammatory responses through the expression of inflammatory genes via various signaling pathways (Finkel 2003) in the pathogenesis of human diseases (Chrissobolis and Faraci 2008). The imbalance between ROS generation and antioxidants results in several pathologies of degenerative diseases (Lin and Beal 2006; Poljsak et al. 2013). To protect against the external stresses, ROS are essential for many physiological functions including the killing of invading microorganisms (Chrissobolis and Faraci 2008). The NADPH oxidase (Nox) complex is a major source of ROS in several physiological and pathological processes (Lee and Yang 2012; Rahman et al. 2006). The levels of Nox-dependent ROS generation contribute to the expression of either inflammatory or anti-inflammatory mediators. External stimuli trigger the signal components to initiate the activity of the Nox complex (Lee and Yang 2012). However, the negative regulatory mechanism of Nox activity is still unclear. Until Noubade et al. (2014) identified a novel regulatory gene, negative regulator of ROS (NRROS), limits ROS generation during inflammatory responses. The up-regulation of NRROS interferes with the association between Nox-2 and p22phox, followed by Nox-2 degradation and impediment of the Nox complex formation (Noubade et al. 2014). The zymosan-induced ROS generation is reduced by the expression of NRROS in IFN-γ-primed bone marrow-derived macrophages. Deficiency of NRROS gene shows more susceptible to microglial development and neurological disorders (Wong et al. 2017). These results suggested that NRROS could protect against neuroinflammatory disorders. In addition, NRROS has been shown to inhibit the NF-κB activity through interrupting the function of Toll-like receptors (Kim et al. 2015; Liu et al. 2013). Although the protecting effects of NRROS have been studied, the detail mechanisms of NRROS expression are still unknown in rat brain astrocytes (RBA-1).
15d-PGJ2 plays protecting effects in diverse cell systems (Abdo et al. 2012; Lin et al. 2006) and suppresses the p22phox expression to protect against apoptosis of neurons and brain endothelial cells (Wu et al. 2016, 2014). Moreover, 15d-PGJ2 exerts neuroprotection from oxidative stress in astrocytes (Haskew-Layton et al. 2013). These protecting effects of 15d-PGJ2 are mediated through upregulation of Nrf2-dependent antioxidant proteins (Itoh et al. 2004). Therefore, 15d-PGJ2 might induce NRROS expression and attenuate the ROS-dependent inflammatory responses in RBA-1. We found that 15d-PGJ2 stimulated FoxO1 phosphorylation through PI3K/Akt pathway and activated Sp1 to regulate the NRROS transcription and Nrf2. The constitutive expression of NRROS attenuated the p47phox phosphorylation to suppress ROS generation and IL-6 expression induced by H2O2. Thus, the up-regulation of NRROS by 15d-PGJ2 could protect against brain inflammatory responses.
Materials and Methods
Antibodies and Chemicals
DMEM/F-12 medium, FBS, TRIzol reagent, CM-H2DCFDA, and PLUS-Lipofectamine were from Invitrogen (Carlsbad, CA, USA). Hybond C membrane and enhanced chemiluminescence (ECL) detection system were from GE Healthcare Biosciences (Buckinghamshire, UK). NRROS (AAS08559C) antibody was from Antibody Verify (Las Vegas, NV, USA). Akt (rabbit polyclonal antibody, Cat# sc-8312, RRID:AB_671714), Lamin A (rabbit polyclonal antibody, Cat# sc-20680, RRID:AB_648148), Sp1 (rabbit polyclonal antibody, Cat# sc-14027, RRID:AB_2171049), and p22 (rabbit polyclonal antibody, Cat# sc-20781, RRID:AB_2090309) antibodies were from Santa Cruz (Santa Cruz, CA, USA). FoxO1 (rabbit monoclonal antibody, Cat# ab52857, RRID:AB_869817), Nox2 (rabbit monoclonal antibody, Cat# ab129068, RRID:AB_11144496), p85 (rabbit monoclonal antibody, Cat# ab40755, RRID:AB_777258), DDDDK tag (rabbit polyclonal antibody, Cat# ab1162, RRID:AB_298215), phospho-Sp1 (rabbit polyclonal antibody, Cat# ab37707, RRID:AB_1524434), Nrf2 (mouse monoclonal antibody, Cat# ab89443, RRID:AB_2041334), and phospho-Nrf2 (phospho-Ser40, Rabbit monoclonal antibody, Cat# ab76026, RRID:AB_1524049) antibodies were from Abcam (Cambridge, UK). Phospho-Akt (rabbit polyclonal antibody, Cat# 9271, RRID:AB_329825) and phospho-FoxO1 (rabbit polyclonal antibody, Cat# 9461, RRID:AB_329831, phospho-S256 FoxO1) antibodies were from Cell Signaling (Danvers, MA, USA). Anti-phospho-p47 (rabbit polyclonal antibody, Cat# A1171, RRID:AB_10696129) antibody was from Assay Biotech (Sunnyvale, CA, USA). Anti-glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (Cat# MCA-1D4, RRID:AB_2107599) antibody was from EnCor Biotechnology (Gainesville, FL, USA). 15d-PGJ2 and 15d-PGJ2 antibody (Cat# ADI-915-043-100) were from Enzo (New York, NY, USA). AS1842856 was from Millipore (Billerica, MA, USA). AKT inhibitor VIII was from Cayman (Ann Arbor, MI, USA). SDS-PAGE reagents were from MDBio Inc (Taipei, Taiwan). Actinomycin D, LY294002, mithramycin A, and other chemicals were from Sigma (St. Louis, MO, USA).
Cell Culture and Treatment
Cells from a rat brain astrocytic cell line (RBA-1) were used in this study. The maintenance and subculture of RBA-1 cells were performed as previously described (Lin et al. 2014). The use of the cell lines had been approved by Chang Gung University Institutional Animal Care and Use Committee (IACUC Approval No. CGU16-081). In brief, a primary astrocyte culture was isolated from neonatal rat cerebrum and the cell line was established naturally through successive cell passages. RBA-1 cells were identified with glial fibrillary acid protein (GFAP, an astrocyte-specific marker) staining and exhibited over 95% positive staining cells. Cells were plated onto culture plates and made quiescent at confluence by incubation in serum-free DMEM/F-12 for 24 h and then treated with 15d-PGJ2 at 37 °C for the indicated time intervals. When the inhibitors were used, cells were pretreated with the individual inhibitor for 1 h before exposure to 15d-PGJ2. The cytotoxicity of 15d-PGJ2 and pharmacological inhibitors at the incubation time was checked using an XTT assay kit, showing no significant effect on cell viability. Experiments were performed with cells from passages 7 to 12. No institutional ethical approval was required.
Preparation of Cell Extracts, Cellular Fraction Extracts, and Western Blot Analysis
After treatment with 15d-PGJ2, RBA-1 cells were washed with ice-cold PBS, scraped, and collected with SDS-loading buffer (0.1 M Tris–HCl, pH 6.8; 1% SDS; 5% glycerol; 2.5% β-mercaptoethanol; 0.02% bromophenol blue). The nuclear and cytoplasmic fractions were isolated according to the protocol of NE-PER nuclear and cytoplasmic extraction reagents (ThermoFisher, Waltham, MA, USA). Samples were denatured, separated on SDS-PAGE, transferred to nitrocellulose membranes, and then probed with a respective primary antibody overnight at 4 °C. The washed membranes were incubated with an anti-rabbit horseradish peroxidase antibody (1:2000) for 1 h. The immunoreactive bands were detected using ECL reagents and captured by a UVP BioSpectrum 500 Imaging System (Upland, CA, USA). UN-SCAN-IT gel software (Orem, UT, USA) was used to analyze and quantify the image densitometry.
Total RNA Extraction, Real Time-PCR, and PCR Analysis
RBA-1 cells were seeded on 10-cm culture dishes and treated with 15d-PGJ2. Total RNA was extracted with TRIzol reagent (Thermo Fisher, Waltham, MA, USA) according to the protocol of the manufacturer. The cDNA obtained from 5 μg total RNA was used to be a template for real-time PCR amplification. Real-time PCR was performed with KAPA PROBE FAST ABI Prism® qPCR kit (KK4705, Kapa Biosystems, Wilmington, MA, USA) and 7500 Real-Time PCR System (Applied Biosystems, Foster City, CA, USA) using the sequences of primers as follows:
Rat NRROS:
Forward primer: 5′-CTCATGCTTCAGAACCTCTCCTG-3′
Reverse primer: 5′-CAGAGTCGTCGAGCCTCATG-3′
Probe: 5′-TGGAGGTCGTGTCCTTGGCAAGAA-3′
Rat IL-6:
Forward primer: 5′-CGAAAGTCAACTCCATCTGCC-3′
Reverse primer: 5′-GGCAACTGGCTGGAAGTCTCT-3′
Probe: 5′-TCAGGAACAGCTATGAAGTTTCTCTCCG-3′
Human NRROS:
Forward primer: 5′-TTTTCACTTCCTGACCGTGG-3′
Reverse primer: 5′-CACCAACTTGCAGACTCCTTG-3′
Probe: 5′-AGGAACAGAAGCGGAACAGCCACA-3′
GAPDH:
Forward primer: 5′-AACTTTGGCATCGTGGAAGG-3′
Reverse primer: 5′-GTGGATGCAGGGATGATGTTC-3′
Probe: 5′-TGACCACAGTCCATGCCATCACTGC-3′
Relative gene expression was determined by the ΔΔCt method, where Ct meant the threshold cycle. All experiments were performed in triplicate.
Plasmid Constructions, Transfection, and Luciferase Reporter Gene Assays
To study the promoter activity, we constructed the luciferase reporter plasmids to investigate the promoter regulation in RBA-1 cells. The rat NRROS promoter plasmids were inserted into an pGL3-basic vector, respectively. The DNA fragments, rat NRROS promoter regions from − 1716 to + 135 bp, deletion fragments and human NRROS promoter from − 1053 to + 406, were constructed into an pGL3-basic vector. Luciferase plasmid and pCMV-β-gal were co-transfected into RBA-1 cells with Lipofectamine 2000. Promoter activity was detected using a luciferase assay system (Promega, Madison, WI, USA) to analyze the firefly luciferase activities and standardized with β-galactosidase activity. Site-directed mutagenesis of human FoxO1, a Ser256-to-Asp FoxO1 mutant (FoxO1S256D) mutant was constructed by inserting the DNA fragments encoding human FoxO1S256D between the EcoRV and Hind III sites of pCMV-Tag2B (Stratagene, Santa Clara, CA, USA).
Transient Transfection with siRNAs
Quiescent RBA-1 (80% confluence) were washed once with PBS and added 2 ml of serum-free DMEM/F-12 medium to each well (6-well plate). The siRNAs of scrambled, AKT1 (RSS301983, 5′-UUAGGAUGAGCUCGAACAGCUUCUC-3′), and p85 (Pik3r1, RSS303756) were obtained from Thermo Fisher (Waltham, MA, USA); the siRNAs of Sp1 (Rn01_00038010, 5′-CAUUAUUGCUGCUAUGCCA-3′) and FoxO1 (Rn02_00284211, 5′-CUAUUCAUUUGCACCGCCA-3′) were obtained from Sigma (St. Louis, MO, USA). Nrf2 (nfe212-RSS343557) was from Invitrogen (Carlsbad, CA, USA). Transient transfection siRNAs (100 nM) was performed using a GeneMute reagent according to the manufacturer’s instructions (Rockville, MD, USA).
Chromatin Immunoprecipitation Assay
Chromatin immunoprecipitation (ChIP) was performed as previously described with minor modifications (Chi et al. 2011). In brief, RBA-1 cells were fixed with 1% formaldehyde at room temperature for 30 min and then stopped the cross-linkage reaction with 0.125 M glycine. The cells were washed with cold-PBS and harvested with 500 μl ChIP lysis buffer. The fixed chromatins were broken down with sonication to about 500 to 100 bp. The equal concentrations of cell lysates were incubated with 2 μg respective antibody and rotated at 4 °C for 1 h. Twenty microliters ChIP beads (Millipore) was added to the mixture and rotated at 4 °C overnight. After washing, the immunocomplex was eluted with elution buffer and de-cross-linkage at 65 ℃ for overnight. The genomic DNA fragment was extracted from the antibody-absorbed complex. PCR reactions were performed by using response element-specific primers and analyzed with 2% agarose gel electrophoresis.
Stable Clone Construction and Establishment of Human NRROS in RBA-1 Cells
The DNA fragment of the human NRROS amino acid encoding region was inserted between BamHI and HindIII sites of pCMV-Tag2B to construct the expression plasmid, pCMV-Tag2B-hNRROS. RBA-1 cells (about 70% confluence) were plated on a 10-cm dish and transfected with 2 μg of pCMV-Tag2B-hNRROS or pCMV-Tag2B using Xtreme® DNA transfection reagent. Twenty-four hours after transfection, cells were split and selected by cultivated in G418 (500 μg/ml)-containing medium with 5% CO2 at 37 °C. Cells were then sub-cultured every 3 days for 2 weeks to obtain stable clones by collecting drug-resistant colonies. The level of human NRROS expression of the RBA-1 stable clone was analyzed by Western blot and probed with a mouse anti-Flag antibody.
ROS Generation Assay
The levels of ROS were detected using CM-H2DCFDA (C6827, Invitrogen) as previously described (Lee et al. 2013). Briefly, cells were washed twice with warmed serum-free medium and then incubated with CM-H2DCFDA at 37 ℃ for 30 min and then treated with 100 μM H2O2 for 2 h. The cells were washed twice with warm PBS and observed under a fluorescent microscope.
IL-6 ELISA
Cells were treated with 100 μM H2O2 for 2 h. The medium was harvested to analyze the level of IL-6 release using an IL-6 enzyme-linked immunosorbent assay (ELISA) kit (Biolegend, San Diego, CA, USA).
Data and Statistical Analysis
Statistical analysis was performed by using GraphPad Prism Program 7.0 software (RRID:SCR_000306, GraphPad, San Diego, CA, USA). We used one-way ANOVA followed by Dunnett’s post hoc test when comparing more than two groups of data and one-way ANOVA, nonparametric Kruskal–Wallis test, followed by Dunnett’s post hoc test when comparing multiple independent groups. P values of 0.05 were considered to be statistically significant. Post hoc tests were run only if F achieved P < 0.05, and there was no significant in homogeneity of variance. No sample calculation was performed. We did not conduct any data test for outliers, and thus, no single data points were excluded. All the data were expressed as the mean ± SEM, at least three individual experiments (n = number of independent cell culture preparations). The n values are provided in the figure legends. Error bars were omitted when they fell within the dimensions of the symbols.
Results
15d-PGJ2 Induces NRROS Expression
NRROS exerts a negatively regulatory effect on ROS generation (Noubade et al. 2014). 15d-PGJ2 has been shown to display neuroprotection against ROS stress (Abdo et al. 2012; Lin et al. 2006). The relationship between 15d-PGJ2 and expression of NRROS is still unknown. Here, we found that 15d-PGJ2 concentration- and time-dependently induced NRROS protein and mRNA expression in RBA-1 cells (p < 0.05, as compared with the control, Fig. 1a, b). Quantification of the Western blotting of 10 μM 15d-PGJ2, we found that NRROS protein was induced with a maximal response within 4–6 h (p < 0.05, as compared with the control) and slightly declined at 24 h (Fig. 1a, lower panel). The levels of NRROS mRNA expression were increased to about sixfold within 1 h (p < 0.05, as compared with the control) and then declined to the basal level within 6 h (Fig. 1b). The lower concentrations (1 and 5 μM) of 15d-PGJ2 induced a little of NRROS expression. Pretreatment with the inhibitor of transcription actinomycin D (Act. D, 1 μM), PPAR-γ (GW9662, 10 nM), Sp1 (mithramycin A, 1 μM), PI3K (LY294002, 10 μM), Akt inhibitor VIII (Akti VIII, 1 μM), or FoxO1 (AS1842856, 0.3 μM) attenuated the 15d-PGJ2-induced NRROS mRNA expression (p < 0.05, as compared with the control, Fig. 1c). These results suggested that 15d-PGJ2-induced transcriptional NRROS mRNA expression is, at least, mediated through PI3K/Akt, Sp1, FoxO1, and PPARγ in RBA-1 cells.
15d-PGJ2 induced NRROS expression in RBA-1 cells. a, b RBA-1 cells were treated with 1, 5, or 10 μM 15d-PGJ2 for the indicated time intervals. The levels of a NRROS protein and b mRNA expression were analyzed by Western blot and RT/real-time PCR, respectively. Lower panel of a indicates the quantitation of 10 μM 15d-PGJ2 treatment. c RBA-1 cells were pretreated with actinomycin D (ActD, 1 μM), mithramycin A (Mith A, 1 μM), GW9662 (GW, 10 nM), LY294002 (10 μM), Akti VIII (1 μM), or AS1842856 (AS, 0.3 μM) for 1 h and then treated with 10 μM 15d-PGJ2 for 2 h. The levels of NRROS mRNA expression were analyzed by RT/real-time PCR. The data are expressed as mean ± SEM, from three independent experiments (n = 3, number of independent cell culture preparations). #p < 0.05, as compared with the respective values significant difference as indicated
Roles of Rat NRROS Promoter and 15d-PGJ2 Response Regions in NRROS Expression
To investigate the regulation of NRROS transcription, we analyzed the candidate sequences of NRROS promoter regions (upstream of transcription initiating site and the exon 1). Figure 2 a shows the sequences of rat NRROS upstream region and consensus motifs for transcription factor binding sites. There are several possibilities of response elements, such as Sp1, FoxO1, transcription factor IIB (TFIIB), and TATA-binding protein (TBP) on the promoter-predicted regions of NRROS. To analyze the DNA sequences of rat NRROS promoters, there was no typical location of TBP binding site (about 30–40 bp to initiation site) but several Sp1 binding sites on rat NRROS promoters. We speculated that NRROS belongs to the TATA-less promoters. Moreover, Sp1 was an important factor in NRROS mRNA expression which was attenuated by mithramycin A (Fig. 1c). To examine the transcriptional regulation, we constructed the luciferase reporter plasmids from rat NRROS promoters. The results of the promoter activity assay indicated that 15d-PGJ2 significantly induced rat NRROS promoter activity in RBA-1 cells (p < 0.05, as compared with the control, Fig. 2b). To investigate the location of 15d-PGJ2-response elements, serial plasmids of deletion mutants were constructed (Fig. 2c). These fragments of the NRROS promoter were transfected into RBA-1 cells to analyze the essential promoter regions for NRROS expression. As shown in Fig. 2d, up630E1 exhibited the maximal promoter activity (p < 0.05, as compared with the control), and the transcription activity of up360E1 was dramatically lower than that of the full-length construction (p < 0.05, as compared with the control, as compared with that of up630E1). These results suggested that the essential promoter region is located between the region of − 630 to − 360 bp to initiate NRROS transcription. Next, to examine the location of 15d-PGJ2-response element, these plasmid-transfected cells were treated with 15d-PGJ2 and analyzed the promoter activities. The results of the promoter activity assay indicated that plasmids of the full-length and − 850 bp obviously exhibited the 15d-PGJ2-induced promoter activities (Fig. 2d). These promoter analyses suggested that 360 bp upstream of transcription initiated site (starting site of exon 1) is the essential promoter region and the region between − 630 and − 360 contained the 15d-PGJ2 response elements for NRROS induction in RBA-1 cells.
Analysis of rat NRTROS promoter and screening for 15d-PGJ2 response elements. a The DNA sequences of rat NRROS promoter and transcription factor binding sites. The exon 1 regions are shown in boldface and (+ 1) indicates the first nucleotide of the mRNA. b RBA-1 cells were transfected with either rat (r)NRROS or (h)NRROS plasmid, and pCMV-β-gal DNA for 24 h, and then treated with 10 μM 15d-PGJ2 for the indicated time intervals. The promoter activity was determined in the cell lysates using a promoter assay kit. c Scheme of the rat NRROS promoter region and reporter constructs. The position of deletion mutant, transcription initiation (+ 1), and location of exon 1 are indicated. Figures were not drawn to scale. d RBA-1 cells transfected with various NRROS constructs were treated with 10 μM 15d-PGJ2 for 1 h. The pGL3B-Luc without the promoter was used as a control (Basic). Deletion mutants were used to analyze the essential promoter regions and 15d-PGJ2 response element. RLU indicates the related luciferase units. Statistical analysis was determined using two-tailed unpaired Student’s t test. The data are presented as mean ± SEM, from three independent experiments (n = 3, number of independent cell culture preparations). #p < 0.05, as compared with the respective values significantly different as indicated
Involvement of PI3K/Akt and FoxO1 in 15d-PGJ2-Induced NRROS Expression
PI3K consisting of a catalytic subunit (p110) and a regulatory subunit (p85) and its downstream component of Akt regulate the expression of several proteins in various types of cells (Lin et al. 2014; Liu et al. 2009). To determine the roles of PI3K/Akt in NRROS induction, pretreatment with the inhibitor of PI3K (LY294002) or Akt (Akti VIII) concentration-dependently attenuated the 15d-PGJ2-induced NRROS protein expression (p < 0.05, as compared with the cells treated with 15d-PGJ2, Fig. 3a, b). The phosphorylation of the FoxO proteins, a subgroup of the Forkhead family of transcription factors, is activated by PI3K/Akt pathway leading to the expression of target genes (Huang and Tindall 2007). To determine the role of FoxO1 in the 15d-PGJ2-induced NRROS protein expression mediated through PI3K/Akt, pretreatment with a FoxO1 inhibitor (AS1842856) concentration-dependently attenuated the 15d-PGJ2-induced NRROS protein expression (p < 0.05, as compared with the cells treated with 15d-PGJ2, Fig. 3c). In addition, the involvement of PI3K/Akt and FoxO1 in the 15d-PGJ2-induced responses was confirmed by determining the phosphorylation of Akt and FoxO1. As shown in Fig. 3d–f, 15d-PGJ2 time-dependently stimulated the phosphorylation of Akt with a maximal response within 60 min (p < 0.05, as compared the control), which was attenuated by either LY294002 or Akti VIII, but not by AS1842856 (p < 0.05, as compared with the cells treated with 15d-PGJ2). Moreover, 15d-PGJ2 stimulated FoxO1 phosphorylation with a maximal response within 45–60 min (p < 0.05, as compared the control), which was attenuated by LY294002, Akti VIII, or AS1842856 (p < 0.05, as compared with the cells treated with 15d-PGJ2), implying that FoxO1 is a downstream component of PI3K/Akt. These results suggested that 15d-PGJ2-induced NRROS expression is mediated through a PI3K/Akt-dependent FoxO1 cascade in RBA-1 cells.
15d-PGJ2-induced NRROS expression is mediated through PI3K, AKT, and FoxO1. RBA-1 cells were pretreated with different concentrations of a, d LY294002, b, e AKTi VIII, or c, f AS1842856 for 1 h and then incubated with 10 μM 15d-PGJ2 for the indicated time intervals. The levels of NRROS, phospho-AKT, phospho-FoxO1, AKT, FoxO1, and GAPDH protein were analyzed by Western blotting. The data are presented as mean ± SEM, from three independent experiments (n = 3, number of independent cell culture preparations). #p < 0.05, as compared with the respective values significantly different as indicated
To ensure the roles of PI3K/Akt and FoxO1 in NRROS expression, transfection with either p85 (Fig. 4a), Akt (Fig. 4b), or FoxO1 (Fig. 4c) siRNA reduced the protein level of p85 (by 55%), Akt (by 47%), or FoxO1 (by 39%), and then attenuated the 15d-PGJ2-induced NRROS protein expression (by 35%, 40%, or 41%, respectively). Moreover, 15d-PGJ2-stimulated Akt and FoxO1 phosphorylation were blocked by transfection with either p85 or Akt siRNA (p < 0.05, as compared with the cells treated with 15d-PGJ2, Fig. 4d, e). Knockdown of FoxO1 reduced the phosphorylation of FoxO1 only, but no effect on the phosphorylation of Akt (Fig. 4f). These results concluded that 15d-PGJ2-induced NRROS expression is mediated through a PI3K/Akt/FoxO1 pathway in RBA-1 cells.
The roles of PI3K, AKT, and FoxO1 in 15d-PGJ2-induced NRROS expression are confirmed by transfection with respective siRNAs. RBA-1 cells were transfected with siRNA (a, d p85; b, e AKT, or c, f FoxO1) or scramble siRNA for 48 h and then incubated with 10 μM 15d-PGJ2 for 4 h (a–c) and for the indicated time intervals (d–f). The levels of NRROS, phospho-AKT, phospho-FoxO1, AKT, FoxO1, and GAPDH protein were analyzed by Western blotting. The data are presented as mean ± SEM, from three independent experiments (n = 3, number of independent cell culture preparations). #p < 0.05, as compared with the respective values significantly different as indicated
FoxO1 and Sp1 Involve in 15d-PGJ2-Induced NRROS Expression
In the TATA-less promoter, Sp1 facilitates RNA polymerase II association on the promoter to initiate transcription (Smale and Kadonaga 2003). To ensure the role of Sp1 in the 15d-PGJ2-induced NRROS transcription, transfection with Sp1 siRNA knocked down the level of Sp1 protein (by 55%) and then attenuated the 15d-PGJ2-induced NRROS protein expression in RBA-1 cells (by 41%, p < 0.05, as compared with the cells treated with 15d-PGJ2, Fig. 5a). These results indicated that both Sp1 and FoxO1 contributes to regulate NRROS induction in the 15d-PGJ2-treated RBA-1 cells (Figs. 4c and 5a).
15d-PGJ2 stimulates the interaction between Sp1 and FoxO1 leading to NRROS expression. a RBA-1 cells were transfected with Sp1 siRNA and then incubated with 10 μM 15d-PGJ2 for 4 h. The levels of NRROS, Sp1, and GAPDH were analyzed by Western blotting. b Schematic representation of rat NRROS promoter. Locations of primers used for ChIP assays (Sp1 and FoxO1) and transcription factors associated regions are indicated. c, d ChIP assays were performed using an anti-Sp1 or anti-FoxO1 antibody and then amplified the DNA fragments with Sp1 and FoxO1 primers, respectively. e, f RBA-1 cells were pretreated without or with mithramycin A or AS1842856 for 1 h and then incubated with 15d-PGJ2 for 1 h. The association between FoxO1 and Sp1 on promoter was determined by ChIP assays. g Cells were treated with 10 μM 15d-PGJ2 for the indicated time intervals. ChIP assays were performed using an anti-FoxO1 or anti-phospho-FoxO1 antibody. h Cells were treated with 10 μM 15d-PGJ2 for the indicated time intervals. The levels of protein expression in either cytoplasmic or nuclear fractions were analyzed by Western blotting. The data are presented as mean ± SEM, from three independent experiments (n = 3, number of independent cell culture preparations). #p < 0.05, as compared with the respective values significantly different as indicated
Based on our findings of rat NRROS promoter and 15d-PGJ2 induction (Fig. 2), we established that − 630 to − 360 is an essential promoter region and − 850 to − 630 region contained the 15d-PGJ2 response element in NRROS expression. To analyze these regions involved in NRROS expression, we found that the region of − 630 to − 360 contains several Sp1 binding sites and the region of − 850 to − 630 contains the FoxO1 response element. We speculated that both Sp1 and FoxO1 are involved in the 15d-PGJ2-mediated NRROS transcription. As shown in Fig. 5b, the schematic diagram represented the structure of rat NRROS promoter and transcription factors associated regions as indicated. To investigate the roles of these two transcription factors in NRROS expression, ChIP was performed to analyze the association between Sp1 and FoxO1 on rat NRROS promoter. The results indicated that 15d-PGJ2 stimulated the interaction between Sp1 and FoxO1 on NRROS promoter reaching a maximum within 30–60 min (p < 0.05, as compared the control, Fig. 5c, d), which was attenuated by either mithramycin A or AS1842856 (p < 0.05, as compared with the cells treated with 15d-PGJ2, Fig. 5e, f), respectively. These results strongly confirmed that both Sp1 and FoxO1 cooperatively regulate the 15d-PGJ2-induced NRROS transcription in RBA-1 cells.
15d-PGJ2-Induced Nuclear Accumulation and Phosphorylation of FoxO1 Associates with NRROS Promoter to Regulate NRROS Transcription
To examine the role of phosphorylated FoxO1 in the 15d-PGJ2-induced NRROS expression, ChIP assay was performed by using an anti-FoxO1 or anti-phospho-FoxO1 (Ser256) antibody to analyze the interaction between FoxO1 and rat NRROS promoters. We found that 15d-PGJ2 induced FoxO1 accumulation and phosphorylated FoxO1 binding to FoxO1 response element of NRROS promoters (p < 0.05, as compared the control, Fig. 5g). The 15d-PGJ2-stimulated phosphorylation of FoxO1 (Ser256) binding to the promoter was blocked by a FoxO1 inhibitor (AS1842856), but not by a Sp1 inhibitor (mithramycin A) (p < 0.05, as compared with the cells treated with 15d-PGJ2, Supplementary Fig. S1a) on the Sp1 binding region. On the FoxO1-binding regions, the results of ChIP indicated that both phosphorylated FoxO1 and Sp1 bindings were attenuated by AS1842856 or mithramycin A (p < 0.05, as compared with the cells treated with 15d-PGJ2, Supplementary Fig. S1b). Moreover, we determined the localization of NRROS protein in the subcellular fractions. As expected, NRROS accumulation in the cytoplasm occurred within 1–3 h of 15d-PGJ2 treatment (p < 0.05, as compared the control, Fig. 5h). Importantly, the levels of phospho-FoxO1 were obviously detected in the nuclear fraction (p < 0.05, as compared the level of cytoplasmic fraction, Fig. 5h). 15d-PGJ2 stimulated FoxO1 phosphorylation reaching a maximum within 1–2 h and declining at 3–4 h. We also evaluated the effects of phosphorylated FoxO1 on NRROS expression. Overexpression of either constitutively phosphorylated FoxO1 mutant (S256D-FoxO1) or dephosphorylated FoxO1 mutant (S256A-FoxO1) on RBA-1 cells was used to analyze the NRROS expression. We found that S256D-FoxO1 up-regulated NRROS expression and S256A-FoxO1 attenuated the 15d-PGJ2-induced NRROS expression (Supplementary Fig. S2). These findings indicated that the phosphorylated FoxO1 binding on NRROS promoters facilitates the transcriptional initiation in RBA-1 cells challenged with 15d-PGJ2.
15d-PGJ2-Stimulated Phosphorylation of Nrf2 Leading to NRROS Expression
15d-PGJ2 has been shown to induce the expression of antioxidant proteins in an Nrf2-dependent manner (Mochizuki et al. 2005). Thus, the role of Nrf2 in the expression of NRROS was investigated in RBA-1 cells challenged with 15d-PGJ2. We transfected the cells with Nrf2 siRNA which knocked down the levels of Nrf2 protein and attenuated the 15d-PGJ2-induced NRROS protein expression (Fig. 6a). Whether 15d-PGJ2 stimulated Nrf2 phosphorylation was determined in these cells. As shown in Fig. 6b, 15d-PGJ2 stimulated Nrf2 phosphorylation in a time-dependent manner, which was attenuated by transfection with Nrf2 siRNA. 15d-PGJ2 is an endogenous electrophile which could activate Nrf2 and induce the expression of antioxidant proteins (Mochizuki et al. 2005). Thus, the antibody against the 15d-PGJ2 was used to block its inductive effect on NRROS expression. RBA-1 cells were pretreated with 15d-PGJ2 antibody for 1 h and then incubated with 15d-PGJ2 for 6 h. We found that the antibody against the 15d-PGJ2 concentration-dependently attenuated NRROS expression (Fig. 6c).
NRROS expression via Nrf2 activation by 15d-PGJ2 in RBA-1 cells. The cells were transfected with Nrf2 siRNA or scramble siRNA (a, b) for 48 h and then incubated with 10 μM 15d-PGJ2 for 4 h or 6 h (a), and indicated time intervals (b). c The cells were pretreated with various concentration of anti-15d-PGJ2 antibody for 1 h and then incubated with 10 μM 15d-PGJ2 for 6 h. The levels of NRROS, phospho-Nrf2, Nrf2, β-actin, and GAPDH protein were analyzed by Western blotting. The data are presented as mean ± SEM, from three independent experiments (n = 3, number of independent cell culture preparations). #p < 0.05, as compared with the respective values significantly different as indicated
Overexpression of Human NRROS Reduces the H2O2-Induced ROS Generation and IL-6 Expression
15d-PGJ2 has been shown to display neuroprotection against ROS stress (Abdo et al. 2012; Wu et al. 2016). We proposed that 15d-PGJ2 is capable of regulating Nox activity and dampens ROS generation in RBA-1 cells. The results of Western blotting showed that H2O2 time-dependently stimulated p47phox phosphorylation and gp91phox expression, which were attenuated by pretreatment with 15d-PGJ2 through upregulation of NRROS (p < 0.05, as compared the control, Fig. 7a). Moreover, 15d-PGJ2 also inhibited the H2O2-induced IL-6 mRNA expression (p < 0.05, as compared the control, Fig. 7b), suggesting an anti-inflammatory effect of 15d-PGJ2 on the H2O2-mediated responses.
Overexpression of human NRROS reduces the H2O2-induced ROS generation, IL-6 expression, and p47 phosphorylation. a RBA-1 cells were pretreated with 10 μM 15d-PGJ2 for 1 h and then treated with 100 μM H2O2 for the indicated time intervals. The levels of protein expression were analyzed by Western blotting. b RBA-1 cells were pretreated with 15d-PGJ2 for 1 h and then treated with 100 μM H2O2 for the indicated time intervals. The levels of IL-6 mRNA expression were determined by RT/real-time PCR. c, d RBA-1 stable clones of hNRROS and pCMV-Tag2B, or RBA-1 cells were treated with 100 μM H2O2 for 1 h. c The levels of hNRROS mRNA expression were determined by RT/real-time PCR. d The cells were labeled with 10 μM H2DCFDA and then incubated with 100 μM H2O2 for 30 min. The levels of ROS generation were observed using a fluorescence microscope. Scale bar = 100 μm. e RBA-1 stable clones of hNRROS and pCMV-Tag2B, or RBA-1 cells were treated with 100 μM H2O2 for the indicated time intervals. The levels of protein expression were analyzed by Western blotting. f, g RBA-1 stable clones were treated with H2O2 for the indicated time intervals. The levels of IL-6 mRNA expression and secretion of IL-6 were analyzed by RT/real-time PCR and ELISA kit, respectively. The data are presented as mean ± SEM, from three independent experiments (n = 3, number of independent cell culture preparations). *p < 0.05, as compared with the respective values significantly different as indicated
Scheme of signaling pathways is involved in 15d-PGJ2-induced NRROS expression suppressing the H2O2-induced IL-6 expression and ROS generation in RBA-1 cells. NRROS expression induced by 15d-PGJ2 is mediated through a PPARγ-independent activation of PI3K/AKT leading to phosphorylation of FoxO1 and Sp1. These activated transcription factors bind to NRROS promoters and enhance NRROS transcription in RBA-1 cells. Up-regulation of NRROS not only downregulates the H2O2-mediated ROS generation but also inhibits the expression of pro-inflammatory cytokine IL-6. These results elucidate the mechanisms underlying 15d-PGJ2-induced NRROS expression which might be a potential strategy for management of brain inflammatory and neurodegenerative diseases
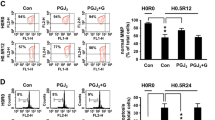
We noticed that H2O2 regulates downstream of signaling components in various types of cells (Miller et al. 2010). To analyze whether NRROS regulated Nox activity in RBA-1 cells, full-length human NRROS was constructed into a pCMV-Tag2B vector. RBA-1 cells were transfected with pCMV-hNRROS (human NRROS) and selected with G418 to establish constitutively expressed clones. The RT/real-time PCR was performed to determine the levels of human NRROS mRNA expression in RBA-1 cells. As shown in Fig. 7c, the levels of overexpressed human NRROS mRNA and protein were detected in pCMV-hNRROS constitutively expressed RBA-1 cells. In addition, H2O2 treatment evoked the levels of ROS generation in RBA-1 cells, which was attenuated by constitutive expression of pCMV-hNRROS stable clone, using H2DCFDA observed under a fluorescent microscope (p < 0.05, as compared the control, Tag2B or RBA-1 cells, Fig. 7d). These results suggested that NRROS attenuates p47phox phosphorylation and suppresses the Nox activity (gp91phox expression) in H2O2-treated RBA-1 cells.
Excessive generation of ROS results in the secretion of pro-inflammatory cytokines such as TNF-α, IL-1, and IL-6 to reflect the proinflammatory responses in M1 macrophages (Mittal et al. 2014). H2O2 activates NF-κB leading to IL-6 transcription and secretion (Zhang et al. 2001), which is involved in the development of several autoimmune diseases (Kobayashi et al. 2016). To further evaluate the effect of NRROS on ROS-mediated inflammatory responses, H2O2-induced IL-6 mRNA transcription and secretion were examined in the constitutively NRROS-expressed RBA-1 cells, determined by using RT/real-time PCR and IL-6 ELISA assays, respectively. We found that H2O2 induced IL-6 mRNA expression and secretion in parental RBA-1 cells and the vector control (Tag2B) clone, which were attenuated in the constitutively NRROS-expressed RBA-1 cells (p < 0.05, as compared the control, Tag2B or RBA-1 cells, Fig. 7e, f). These results indicated that constitutively expressed NRROS reduces the H2O2-induced IL-6 mRNA transcription and secretion in RBA-1 cells.
Discussion
The antioxidant enzymes, including superoxidase dismutase, catalase, glutathione peroxidase, peroxiredoxins, and thioredoxins, cooperate to remove ROS (Mittal et al. 2014). Noubade et al. identified the regulatory activity of NRROS which interferes the Nox complex formation through the interaction of NRROS-Nox-2 and the ER-dependent Nox-2 degradation in macrophages (Noubade et al. 2014). However, there was little information concerning NRROS to regulate Nox activity and ROS generation. Here, we established the essential regions of NRROS transcription and the 15d-PGJ2 response region on NRROS promoters. Figure 7 depicts that transcription factors Sp1 and FoxO1 response elements located on the upstream of exon 1 of rat NRROS gene and contributed to transcriptional regulation mediated through a PI3K/Akt-dependent FoxO1 pathway. In addition, 15d-PGJ2 activated both FoxO1 and Sp1 association with the response elements of NRROS promoter to accelerate NRROS transcription. Upregulation of NRROS attenuated the p47phox phosphorylation and Nox/ROS generation stimulated by H2O2, which downregulated IL-6 expression in RBA-1 cells.
ROS are important signaling molecules in the pathogenesis of inflammatory disorders. Several sources of ROS generation, such as Nox, mitochondria, uncoupled NOS, and xanthine oxidoreductase, compose the ROS generation systems in various types of cells (Mittal et al. 2014). Nox complex generates ROS in various tissues and cells in response to several stresses (Mittal et al. 2014). The regulation of normal physiological functions and the inflammatory responses are dependent on the cellular concentrations of ROS (Kamata and Hirata 1999). Excessive ROS are a causative role in many pathologies of airway diseases (Lee and Yang 2012; Rahman et al. 2006). The stimulated immune cells initiate enzymatic activity of Nox complex to produce superoxide anion during encountering inhaled microorganisms or other mediators. H2O2 can cross cell membranes through aquaporin channels such as AQP3 and AQP8, which also mediate membrane H2O2 uptake and raising the permeability for H2O2 entering into cells (Bienert et al. 2007; Miller et al. 2010). Several reports indicate that H2O2 activates signaling pathways to enhance ROS production in different types of cells (Griendling et al. 2000; Torres and Forman 2003). We observed that constitutively expressed NRROS attenuates the H2O2–induced ROS signal. We also noticed that the role of NRROS is a negative regulatory protein to limit ROS generation. Catalase, superoxide dismutases, and glutathione peroxidase are the ROS-eliminating enzymes. We suggested that constitutively expressed NRROS not only reduces ROS generation but also cooperates with the activities of catalase and glutathione peroxidase to eliminate ROS, such as the exogenous H2O2. Our data indicated that constitutive expression of human NRROS reduced the H2O2-induced Nox/ROS generation, which protected against brain inflammatory diseases (Fig. 8).
NOX complex assembling and activity are mediated by complex formation of gp91phox and phosphorylated p47phox. Regarding the role of the p47phox, many kinases are involved in the phosphorylation of p47phox and it is absolutely required for Nox assembly and activation. We noticed that phosphorylation at Ser370 (the antigen usage of phosphorylated p47phox antibody) had no significant effect on the p47phox interaction with other phox subunits, which was necessary for regulation of Nox activation (El-Benna et al. 2009; Meijles et al. 2014). Several insults, such as pro-inflammatory cytokines, LPS, phorbol ester, and cellular metabolite inducers, trigger p47phox phosphorylation to regulate Nox activity (Mittal et al. 2014). These stimuli initiate the signal transduction to activate downstream kinases. Protein kinase Cs, protein kinase A, Akt, ERK1/2, and p38 MAPK are involved in the regulation of Nox activity via p47phox phosphorylation (El-Benna et al. 2009). The astrocytes activated by pathological stresses produce ROS to protect against microbial infection. On the other side, ROS generation also contributes to neurodegeneration. The astrocytic Nox activity plays an important role in CNS physiology and pathology (Abramov et al. 2005). Furthermore, we found that 15d-PGJ2 reduced the H2O2-induced p47phox phosphorylation, gp91 phox expression, and IL-6 expression. We attempted to establish the relationship among NRROS expression, antioxidative stress, and anti-inflammation in RBA-1 cells. 15d-PGJ2 inhibited the H2O2-induced p47phox phosphorylation and IL-6 mRNA expression through NRROS expression. We found that stable expression of hNRROS reduced the p47phox phosphorylation and pg91phox expression. On the other side, the zymosan-induced ROS generation is reduced by NRROS expression in BMDMs (Noubade et al. 2014). Our results were consistent with this report indicating that stable expression of human NRROS reduced the H2O2-induced ROS generation in RBA-1 cells. Based on these findings, we suggested that NRROS attenuates the phosphorylation of p47phox and Nox-2 activity (gp91phox) in RBA-1 cells.
15d-PGJ2, an endogenous ligand of PPARγ, induces PPARγ-dependent or independent cell apoptosis in cancer cells (Ray et al. 2006; Shimada et al. 2002) and regulates the inflammatory responses (Kim et al. 2007). In the regulation of inflammatory and immune responses, 15d-PGJ2 blocks IKK activity and association of NF-κB to κB sites of promoter to inhibit expression of genes via a PPARγ-independent pathway (Giri et al. 2004; Straus et al. 2000). 15d-PGJ2 also reduced expression of several pro-inflammatory cytokines and exhibited neuronal protecting ability against CNS inflammation (Giri et al. 2004; Wu et al. 2014). Our studies suggested that both of the PPARγ-dependent and PPARγ-independent pathways are involved in the regulation of 15d-PGJ2-induced NRROS to attenuate H2O2-induced pro-inflammatory cytokine (IL-6) expression. In addition, we found that 15d-PGJ2 induced serial phosphorylation of signal molecules through PI3K/AKT/FoxO1. Pretreatment with inhibitors and gene-specific knockdown (siRNA transfection) obtained the consistent results, suggesting that 15d-PGJ2 triggers phosphorylation cascade through PI3K/AKT/FoxO1 in RBA-1 cells to induce NRROS expression. Furthermore, the PPARγ response element was not present on rat NRROS promoter region. However, pretreatment with GW9662, a PPARγ antagonist, reduced the 15d-PGJ2-induced NRROS mRNA expression. Based on these results, we suggested that cooperative effects of PPARγ-independent and PPARγ-dependent pathways were involved in the regulation of 15d-PGJ2-induced NRROS expression. In addition, we also noticed that PPARγ agonists trigger several signal components to regulate gene expression in different types of cells or tissues (Mix et al. 2004; Phulwani et al. 2006; Wei et al. 2014). Therefore, the role of PPARγ response element in these responses is needed for further studies.
The transcription factor FoxOs contain the FoxO consensus motif and associated ability to response element of promoter in regulation of transcriptional activation or repression. To regulate FoxO activities, posttranslational modifications affect the FoxO-mediated transcriptional activity, subcellular localization, and protein stability (Fu and Tindall 2008). Previous reports indicate that FoxO1 affects cellular responses including metabolism, differentiation, and apoptosis by the Akt-dependent phosphorylation at Ser256 of FoxO1 (Fu and Tindall 2008; Savai et al. 2014). Pretreatment with inhibitor, AS1842856, reduced the phosphorylation of FoxO1. In this report, we found that pretreatment with respective inhibitors or transfection with siRNA of PI3K, Akt, or FoxO1 attenuated the 15d-PGJ2-induced NRROS expression in RBA-1 cells. The phosphorylation of these signal molecules indicated the similar results that PI3K/Akt-phosphorylated Ser256 of FoxO1 was involved in the 15d-PGJ2-mediated responses. This PPARγ-independent pathway may be mediated through the TATA-less promoter activity of NRROS. We noticed that 15d-PGJ2-stimulated phosphorylated FoxO1 (Ser256) binding to the promoter was blocked by a FoxO1 inhibitor, but not by a Sp1 inhibitor (Supplementary Fig. S1a) on the Sp1 binding region. On the FoxO1-binding regions, both phosphorylated FoxO1 and Sp1 bindings were attenuated by AS1842856 and mithramycin A (Supplementary Fig. S1b). The results of ChIP assay suggested that the phosphorylated FoxO1 association on FoxO1-binding regions are cooperated with Sp1 to accelerate NRROS transcription in RBA-1 cells treated with 15d-PGJ2. In addition to FoxO1 and Sp1, Nrf2 has been known to be a master transcription factor for up-regulation of antioxidant proteins including heme oxygenase-1 in various types of cells (Haskew-Layton et al. 2013; Lin et al. 2018; Shih et al. 2003). In this study, we also found that 15d-PGJ2 time-dependently stimulated Nrf2 phosphorylation which was attenuated by transfection with Nrf2 siRNA which knocked down the level of Nrf2 protein in RBA-1 cells (Supplementary Fig. 3a). Because transfection with Nrf2 siRNA knocked down the level of Nrf2 protein and could be applied to evaluate its role in the 15d-PGJ2-mediated responses. Thus, the role of Nrf2 in NRROS expression was further investigated by transfection with Nrf2 siRNA which downregulated the level of Nrf2 protein and subsequently attenuated the 15d-PGJ2-induced NRROS expression (Fig. 6a). These results suggested that Nrf2 also plays an important role in the NRROS expression induced by 15d-PGJ2.
Previous reports also indicated that Akt phosphorylates FoxO1 at Ser256 and promotes the association of 14-3-3 and Foxo1 degradation. The posttranslational modification of FoxO1 induces gene expression to regulate cellular functions in several types of cells (Tzivion et al. 2011; Zhang et al. 2011). FoxOs could mediate ROS detoxification through upregulation of catalase or MnSOD to reduce the oxidative stress (Akasaki et al. 2014; Zhang et al. 2011). Based on our results, we found the novel characters in Akt-mediated FoxO1 phosphorylation in 15d-PGJ2-treated RBA-1 cells, confirmed by transfection with either Akt or FoxO1 siRNA which downregulated the NRROS induction. We also noted that 15d-PGJ2-stimulated Akt phosphorylation may be due to the covalent modification of cysteine residues on phosphatase and tensin homolog and inactivation of its activity (Suh et al. 2018). This is an important issue for further study in the future. Moreover, the results of ChIP assay indicated that 15d-PGJ2-induced Ser256 phosphorylation of FoxO1 bound on NRROS promoter. The Western blotting of subcellular fraction also demonstrated that abundant Ser256 phosphorylation of FoxO1 was detected in the nuclear fraction. Overexpression of the phosphorylation-mimic mutant, S256D-FoxO1, also upregulated the NRROS expression. These results suggested that 15d-PGJ2-induced NRROS expression reduces the ROS stress through Akt-dependent FoxO1 phosphorylation in RBA-1 cells. Further, 15d-PGJ2 exerts neuroprotection from oxidative stress in astrocytes (Haskew-Layton et al. 2013). These protecting effects of 15d-PGJ2 are mediated through upregulation of Nrf2-dependent antioxidant proteins (Itoh et al. 2004). In this study, we found that Nrf2 played a key role in the NRROS expression induced by 15d-PGJ2 through its phosphorylation. The inductive effect of 15d-PGJ2 on NRROS expression was blocked by its respective antibody. Therefore, Nrf2 also plays an important in the 15d-PGJ2-mediated responses in RBA-1 cells.
Our findings proposed evidence to support the anti-inflammatory ability of 15d-PGJ2 in brain astrocytes. 15d-PGJ2 has been shown to reduce the expression of pro-inflammatory cytokines (such as IL-1β, IL-6, and TNF-α) and proteins (such as iNOS, COX-2, and ICAM-1) (Giri et al. 2004; Jiang et al. 1998; Ricote et al. 1998). Astrocyte-derived cytokines and chemokines also play important roles in neuroprotection or neurotoxicity in brain lesions and neurological diseases (Choi et al. 2014). Especially, we focused on the ROS-mediated expression of astrocyte-secreted cytokines such as IL-6. The elevated IL-6 by brain injury or inflammation is detected in the cerebrospinal fluid of patients with several neuronal diseases (Van Wagoner and Benveniste 1999; Van Wagoner et al. 1999). The IL-6 is a typical marker for inflammatory and immunological reaction (Kobayashi et al. 2016). Previous report indicates that IL-6 has beneficial potential by neurotrophic and neuroprotective effects in CNS or accelerates the pathophysiological responses in CNS disorders (Van Wagoner et al. 1999). The oxidative stresses induce IL-6 expression in several types of cells (Giri et al. 2004; Zhang et al. 2001). We obtained the similar results in RBA-1 cells showing that H2O2 induced IL-6 mRNA and protein expression. The constitutive expression of human NRROS inhibited p47phox phosphorylation, gp91phox expression, and attenuated ROS generation and IL-6 mRNA expression. These results suggested that NRROS plays a regulatory role in ROS-dependent IL-6 expression and astrocyte reaction.
In summary, we found that 15d-PGJ2-induced NRROS expression is mediated through a PPAR-γ-independent pathway and established in the essential regions of the transcriptional initiation and 15d-PGJ2 response element in RBA-1 cells. 15d-PGJ2 stimulated FoxO1 phosphorylation mediated through PI3K/Akt pathway and activated Sp1 to regulate the NRROS transcription. In addition, Nrf2 played a key role in NRROS expression induced by 15d-PGJ2 which was mediated through its phosphorylation. Upregulation of NRROS attenuated the p47phox phosphorylation and suppressed ROS generation leading to a decrease in the expression of IL-6. Based on these findings, we found the first time that up-regulation of NRROS by 15d-PGJ2 provides useful therapeutic strategies for brain injury, inflammation, and neurodegenerative diseases.
Abbreviations
- BMDMs:
-
Bone marrow-derived macrophages
- ChIP:
-
Chromatin immunoprecipitation
- CNS:
-
Central nervous system
- 15d-PGJ2 :
-
15-Deoxy-∆12,14-prostaglandin J2
- ECL:
-
Enhanced chemiluminescence
- ELISA:
-
Enzyme-linked immunosorbent assay
- FBS:
-
Fetal bovine serum
- GFAP:
-
Glial fibrillary acid protein
- GPx:
-
Glutathione peroxidase
- IFN:
-
Interferon
- IL:
-
Interleukin
- Nox:
-
NADPH oxidase
- NRROS:
-
Negative regulator of ROS
- PPAR:
-
Peroxisome proliferator-activated receptor
- RBA-1:
-
Rat brain astrocytes
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxidase dismutase
- TBP:
-
TATA-binding protein
- TFIIB:
-
Transcription factor IIB
- Trxs:
-
Thioredoxins
References
Abdo H, Mahe MM, Derkinderen P, Bach-Ngohou K, Neunlist M, Lardeux B et al (2012) The omega-6 fatty acid derivative 15-deoxy-Delta-Δ12,14-prostaglandin J2 is involved in neuroprotection by enteric glial cells against oxidative stress. J Physiol 590:2739–2750. https://doi.org/10.1113/jphysiol.2011.222935
Abramov AY, Jacobson J, Wientjes F, Hothersall J, Canevari L, Duchen MR et al (2005) Expression and modulation of an NADPH oxidase in mammalian astrocytes. J Neurosci 25:9176–9184. https://doi.org/10.1523/JNEUROSCI.1632-05.2005
Akasaki Y, Alvarez-Garcia O, Saito M, Carames B, Iwamoto Y, Lotz MK et al (2014) FoxO transcription factors support oxidative stress resistance in human chondrocytes. Arthritis Rheumatol 66:3349–3358. https://doi.org/10.1002/art.38868
Bienert GP, Moller AL, Kristiansen KA, Schulz A, Moller IM, Schjoerring JK, Jahn TP et al (2007) Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J Biol Chem 282:1183–1192. https://doi.org/10.1074/jbc.M603761200
Blackburn D, Sargsyan S, Monk PN, Shaw PJ et al (2009) Astrocyte function and role in motor neuron disease: a future therapeutic target? Glia 57:1251–1264. https://doi.org/10.1002/glia.20848
Chi PL, Luo SF, Hsieh HL, Lee IT, Hsiao LD, Chen YL, Yang CM et al (2011) Cytosolic phospholipase A2 induction and prostaglandin E2 release by interleukin-1beta via the myeloid differentiation factor 88-dependent pathway and cooperation of p300, Akt, and NF-κB activity in human rheumatoid arthritis synovial fibroblasts. Arthritis Rheum 63:2905–2917. https://doi.org/10.1002/art.30504
Choi SS, Lee HJ, Lim I, Satoh J, Kim SU et al (2014) Human astrocytes: secretome profiles of cytokines and chemokines. PLoS ONE 9:e92325. https://doi.org/10.1371/journal.pone.0092325
Chrissobolis S, Faraci FM (2008) The role of oxidative stress and NADPH oxidase in cerebrovascular disease. Trends Mol Med 14:495–502. https://doi.org/10.1016/j.molmed.2008.09.003
El-Benna J, Dang PM, Gougerot-Pocidalo MA, Marie JC, Braut-Boucher F et al (2009) p47phox, the phagocyte NADPH oxidase/NOX2 organizer: structure, phosphorylation and implication in diseases. Exp Mol Med 41:217–225. https://doi.org/10.3858/emm.2009.41.4.058
Finkel T (2003) Oxidant signals and oxidative stress. Curr Opin Cell Biol 15:247–254
Fu Z, Tindall DJ (2008) FOXOs, cancer and regulation of apoptosis. Oncogene 27:2312–2319. https://doi.org/10.1038/onc.2008.24
Giri S, Rattan R, Singh AK, Singh I et al (2004) The 15-deoxy-δ12,14-prostaglandin J2 inhibits the inflammatory response in primary rat astrocytes via down-regulating multiple steps in phosphatidylinositol 3-kinase-Akt-NF-κB-p300 pathway independent of peroxisome proliferator-activated receptor γ. J Immunol 173:5196–5208
Griendling KK, Sorescu D, Lassegue B, Ushio-Fukai M et al (2000) Modulation of protein kinase activity and gene expression by reactive oxygen species and their role in vascular physiology and pathophysiology. Arterioscler Thromb Vasc Biol 20:2175–2183
Haskew-Layton RE, Payappilly JB, Xu H, Bennett SA, Ratan RR et al (2013) 15-Deoxy-Δ12,14-prostaglandin J2 (15d-PGJ2) protects neurons from oxidative death via an Nrf2 astrocyte-specific mechanism independent of PPARγ. J Neurochem 124:536–547. https://doi.org/10.1111/jnc.12107
Huang H, Tindall DJ (2007) Dynamic FoxO transcription factors. J Cell Sci 120:2479–2487. https://doi.org/10.1242/jcs.001222
Itoh K, Tong KI, Yamamoto M et al (2004) Molecular mechanism activating Nrf2-Keap1 pathway in regulation of adaptive response to electrophiles. Free Radic Biol Med. 15;36(10):1208–13. doi: https://doi.org/10.1016/j.freeradbiomed.2004.02.075
Jiang C, Ting AT, Seed B et al (1998) PPAR-γ agonists inhibit production of monocyte inflammatory cytokines. Nature 391:82–86
Kamata H, Hirata H (1999) Redox regulation of cellular signalling. Cell signal 11:1–14
Kim JH, Kim K, Kim I, Seong S, Kim N et al (2015) NRROS negatively regulates osteoclast differentiation by inhibiting RANKL-mediated NF-κB and reactive oxygen species pathways. Mol Cells 38:904–910. https://doi.org/10.14348/molcells.2015.0177
Kim WJ, Kim JH, Jang SK et al (2007) Anti-inflammatory lipid mediator 15d-PGJ2 inhibits translation through inactivation of eIF4A. EMBO J 26:5020–5032
Kobayashi EH, Suzuki T, Funayama R, Nagashima T, Hayashi M, Sekine H, Tanaka N, Moriguchi T, Motohashi H, Nakayama K, Yamamoto M et al (2016) Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat Commun 7:11624. https://doi.org/10.1038/ncomms11624
Lee IT, Lin CC, Lee CY, Hsieh PW, Yang CM et al (2013) Protective effects of (-)-epigallocatechin-3-gallate against TNF-α-induced lung inflammation via ROS-dependent ICAM-1 inhibition. J Nutr Biochem 24:124–136. https://doi.org/10.1016/j.jnutbio.2012.03.009
Lee IT, Yang CM (2012) Role of NADPH oxidase/ROS in pro-inflammatory mediators-induced airway and pulmonary diseases. Biochem Pharmacol 84:581–590. https://doi.org/10.1016/j.bcp.2012.05.005
Lin CC, Lee IT, Chi PL, Hsieh HL, Cheng SE, Hsiao LD, Liu CJ, Yang CM et al (2014) C-Src/Jak2/PDGFR/PKCδ-dependent MMP-9 induction is required for thrombin-stimulated rat brain astrocytes migration. Mol Neurobiol 49:658–672
Lin CC, Yang CC, Chen YW, Hsiao LD, Yang CM et al (2018) Arachidonic acid induces ARE/Nrf2-dependent heme oxygenase-1 transcription in rat brain astrocytes. Mol Neurobiol 55:3328–3343. https://doi.org/10.1007/s12035-017-0590-7
Lin MT, Beal MF (2006) Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443:787–795. https://doi.org/10.1038/nature05292
Lin TN, Cheung WM, Wu JS, Chen JJ, Lin H, Chen JJ, Liou JY, Shyue SK, Wu KK et al (2006) 15d-prostaglandin J2 protects brain from ischemia-reperfusion injury. Arterioscler Thromb Vasc Biol 26:481–487. https://doi.org/10.1161/01.ATV.0000201933.53964.5b
Liu J, Zhang Z, Chai L, Che Y, Min S, Yang R et al (2013) Identification and characterization of a unique leucine-rich repeat protein (LRRC33) that inhibits Toll-like receptor-mediated NF-κB activation. Biochem Biophys Res Commun 434:28–34. https://doi.org/10.1016/j.bbrc.2013.03.071
Liu P, Cheng H, Roberts TM, Zhao JJ et al (2009) Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov 8:627–644. https://doi.org/10.1038/nrd2926
Meijles DN, Fan LM, Howlin BJ, Li JM (2014) Molecular insights of p47phox phosphorylation dynamics in the regulation of NADPH oxidase activation and superoxide production. J Biol Chem 289:22759–22770
Miller EW, Dickinson BC, Chang CJ et al (2010) Aquaporin-3 mediates hydrogen peroxide uptake to regulate downstream intracellular signaling. Proc Natl Acad Sci USA 107:15681–15686. https://doi.org/10.1073/pnas.1005776107
Mittal M, Siddiqui MR, Tran K, Reddy SP, Malik AB et al (2014) Reactive oxygen species in inflammation and tissue injury. Antioxid Redox Signal 20:1126–1167. https://doi.org/10.1089/ars.2012.5149
Mix KS, Coon CI, Rosen ED, Suh N, Sporn MB, Brinckerhoff CE et al (2004) Peroxisome proliferator-activated receptor-γ-independent repression of collagenase gene expression by 2-cyano-3,12-dioxooleana-1,9-dien-28-oic acid and prostaglandin 15-deoxy-delta-Δ(12,14) J2: a role for Smad signaling. Mol Pharmacol 65:309–318. https://doi.org/10.1124/mol.65.2.309
Mochizuki M, Ishii Y, Itoh K, Iizuka T, Morishima Y, Kimura T, Kiwamoto T, Matsuno Y, Hegab AE, Nomura A, Sakamoto T, Uchida K, Yamamoto M, Sekizawa K et al (2005) Role of 15-deoxy delta (12,14) prostaglandin J2 and Nrf2 pathways in protection against acute lung injury. Am J Respir Crit Care Med 171(11):1260–1266. https://doi.org/10.1164/rccm.200406-755OC
Noubade R, Wong K, Ota N, Rutz S, Eidenschenk C, Valdez PA, Ding J, Peng I, Sebrell A, Caplazi P, DeVoss J, Soriano RH, Sai T, Lu R, Modrusan Z, Hackney J, Ouyang W et al (2014) NRROS negatively regulates reactive oxygen species during host defence and autoimmunity. Nature 509:235–239
Phulwani NK, Feinstein DL, Gavrilyuk V, Akar C, Kielian T et al (2006) 15-deoxy-Delta-Δ12,14-prostaglandin J2 (15d-PGJ2) and ciglitazone modulate Staphylococcus aureus-dependent astrocyte activation primarily through a PPAR-γ-independent pathway. J Neurochem 99:1389–1402. https://doi.org/10.1111/j.1471-4159.2006.04183.x
Poljsak B, Suput D, Milisav I et al (2013) Achieving the balance between ROS and antioxidants: when to use the synthetic antioxidants. Oxid Med Cell Longev 2013:956792
Rahman I, Biswas SK, Kode A et al (2006) Oxidant and antioxidant balance in the airways and airway diseases. Eur J Pharmacol 533:222–239. https://doi.org/10.1016/j.ejphar.2005.12.087
Ray DM, Akbiyik F, Phipps RP et al (2006) The peroxisome proliferator-activated receptor γ (PPARγ) ligands 15-deoxy-Delta-Δ12,14-Prostaglandin J2 and ciglitazone induce human B lymphocyte and B cell lymphoma apoptosis by PPARγ-independent mechanisms. J Immunol 177:5068–5076
Ricci G, Volpi L, Pasquali L, Petrozzi L, Siciliano G et al (2009) Astrocyte-neuron interactions in neurological disorders. J Biol Phys 35:317–336. https://doi.org/10.1007/s10867-009-9157-9
Ricote M, Li AC, Willson TM, Kelly CJ, Glass CK et al (1998) The peroxisome proliferator-activated receptor-γ is a negative regulator of macrophage activation. Nature 391:79–82
Savai R, Al-Tamari HM, Sedding D, Kojonazarov B, Muecke C, Teske R, Capecchi MR, Weissmann N, Grimminger F, Seeger W, Schermuly RT, Pullamsetti SS et al (2014) Pro-proliferative and inflammatory signaling converge on FoxO1 transcription factor in pulmonary hypertension. Nat Med 20:1289–1300. https://doi.org/10.1038/nm.3695
Shih AY, Johnson DA, Wong G, Kraft AD, Jiang L, Erb H, Johnson JA, Murphy TH et al (2003) Coordinate regulation of glutathione biosynthesis and release by Nrf2-expressing glia potently protects neurons from oxidative stress. J Neurosci 23:3394–3406
Shimada T, Kojima K, Yoshiura K, Hiraishi H, Terano A et al (2002) Characteristics of the peroxisome proliferator activated receptor γ (PPARγ) ligand induced apoptosis in colon cancer cells. Gut 50:658–664
Smale ST, Kadonaga JT (2003) The RNA polymerase II core promoter. Annu Rev Biochem 72:449–479. https://doi.org/10.1146/annurev.biochem.72.121801.161520
Sofroniew MV, Vinters HV (2010) Astrocytes: biology and pathology. Acta neuropathol 119:7–35. https://doi.org/10.1007/s00401-009-0619-8
Straus DS, Pascual G, Li M, Welch JS, Ricote M, Hsiang CH, Sengchanthalangsy LL, Ghosh G, Glass CK et al (2000) 15-deoxy-delta-Δ12,14-prostaglandin J2 inhibits multiple steps in the NF-κB signaling pathway. Proc Natl Acad Sci USA 97:4844–4849
Suh J, Kim DH, Kim EH, Park SA, Park JM, Jang JH, Kim SJ, Na HK, Kim ND, Kim NJ, Suh YG, Surh YJ et al (2018) 15-Deoxy-Δ12,14-prostaglandin J2 activates PI3K-Akt signaling in human breast cancer cells through covalent modification of the tumor suppressor PTEN at cysteine 136. Cancer Lett 424:30–45. https://doi.org/10.1016/j.canlet.2018.03.016
Torres M, Forman HJ (2003) Redox signaling and the MAP kinase pathways. BioFactors 17:287–296
Tzivion G, Dobson M, Ramakrishnan G et al (2011) FoxO transcription factors; regulation by AKT and 14-3-3 proteins. Biochim Biophys Acta 1813:1938–1945
Van Wagoner NJ, Benveniste EN (1999) Interleukin-6 expression and regulation in astrocytes. J Neuroimmunol 100:124–139
Van Wagoner NJ, Oh JW, Repovic P, Benveniste EN et al (1999) Interleukin-6 (IL-6) production by astrocytes: autocrine regulation by IL-6 and the soluble IL-6 receptor. J Neurosci 19:5236–5244
Wei J, Zhu H, Komura K, Lord G, Tomcik M, Wang W, Doniparthi S, Tamaki Z, Hinchcliff M, Distler JH, Varga J et al (2014) A synthetic PPAR-γ agonist triterpenoid ameliorates experimental fibrosis: PPAR-γ-independent suppression of fibrotic responses. Ann Rheum Dis 73:446–454. https://doi.org/10.1136/annrheumdis-2012-202716
Wong K, Noubade R, Manzanillo P, Ota N, Foreman O, Hackney JA, Friedman BA, Pappu R, Scearce-Levie K, Ouyang W et al (2017) Mice deficient in NRROS show abnormal microglial development and neurological disorders. Nat Immunol 18:633–641
Wu JS, Tsai HD, Cheung WM, Hsu CY, Lin TN et al (2016) PPAR-γ ameliorates neuronal apoptosis and ischemic brain injury via suppressing NF-κB-driven p22phox transcription. Mol Neurobiol 53:3626–3645. https://doi.org/10.1007/s12035-015-9294-z
Wu JS, Tsai HD, Huang CY, Chen JJ, Lin TN et al (2014) 15-Deoxy-Δ12,14-PGJ 2, by activating peroxisome proliferator-activated receptor-gamma, suppresses p22phox transcription to protect brain endothelial cells against hypoxia-induced apoptosis. Mol Neurobiol 50:221–238
Yang CM, Hsieh HL, Lin CC, Shih RH, Chi PL, Cheng SE, Hsiao LD et al (2013) Multiple factors from bradykinin-challenged astrocytes contribute to the neuronal apoptosis: involvement of astroglial ROS, MMP-9, and HO-1/CO system. Mol Neurobiol 47:1020–1033. https://doi.org/10.1007/s12035-013-8402-1
Zhang J, Johnston G, Stebler B, Keller ET et al (2001) Hydrogen peroxide activates NF-κB and the interleukin-6 promoter through NF-κB-inducing kinase. Antioxid Redox Signal 3:493–504
Zhang X, Tang N, Hadden TJ, Rishi AK et al (2011) Akt, FoxO and regulation of apoptosis. Biochim Biophys Acta 1813:1978–1986. https://doi.org/10.1016/j.bbamcr.2011.03.010
Acknowledgments
We appreciated Ya-Fang Shi for her technical assistance.
Funding
This work was supported by the Ministry of Science and Technology, Taiwan [grant numbers: MOST107-2320-B-039-071-MY2, MOST108-2320-B-039-061, MOST109-2320-B-039-061, and MOST108-2320-B-182-014]; China Medical University, Taiwan [grant numbers: CMU108-MF-08]; Chang Gung Medical Research Foundation, Taiwan [grant numbers: CMRPG5F0203, CMRPG5J0142, CMRPG5J0143].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, CY., Yang, CC., Hsiao, LD. et al. Involvement of FoxO1, Sp1, and Nrf2 in Upregulation of Negative Regulator of ROS by 15d-PGJ2 Attenuates H2O2-Induced IL-6 Expression in Rat Brain Astrocytes. Neurotox Res 40, 154–172 (2022). https://doi.org/10.1007/s12640-020-00318-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00318-6