Abstract
A variety of organic feedstocks can be used for anaerobic digestion, resulting in digestates with different compositions, affecting the fertiliser value. Therefore, two experiments were conducted to assess (1) differences in the nitrogen (N) fertiliser value of seven digestates from different feedstocks in a 2-year field experiment with spring wheat, and (2) the degradability of organic matter (OM) in the digestates within an aerobic incubation experiment. In the field, mineral fertiliser equivalents were in a range of 18–60% (1st year) and 39–83% (2nd year). Fertiliser properties could describe 58.9–74.2% of the N offtake variance among digestates. In the incubation experiment, digestates produced 720–1900 mg CO2-C kg−1. After 56 days, 61% of organic C added by food waste digestate has been mineralised, compared to 16–22% for the other digestates. Digestate composition (C/N, Corg/Norg, carbonate, cellulose, lignin, and crude fibre) could explain 90.4% of the CO2 evolution. In both experiments, digested food waste stood out among digestates with the highest N offtake and highest OM mineralisation. In conclusion, differences in fertiliser value and OM degradability could be related to compositional variations. However, apart from food waste, the composition had only minor influence on digestate performance after soil application.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Statement of Novelty
It has been shown in the literature that different feedstocks lead to compositional variations in digestates. Those feedstock-dependent differences have been suggested to affect the fertilising properties. However, it was not clear how strong the effect of digestate composition is, and how this effect relates to measureable differences regarding the fertiliser value in the field. Therefore, the present study characterised common types of digestates from different feedstocks, and related their composition directly to the nitrogen fertiliser value and organic matter degradability. By integration of different digestate properties into models, the influence of certain characteristics could be determined and differentiated. Ultimately, the magnitude of digestate variations was evaluated, promoting an improved understanding for fertiliser management with anaerobic digestates.
Introduction
Anaerobic digestion (AD) of residues and dedicated energy crops is an option to produce renewable energy, which expanded within the last two decades [1]. Beside the benefit of generating renewable energy through AD of biomass, biogas production is related to several advantages for agriculture and environment (hygienisation, stabilisation, mitigation of greenhouse gases etc.), which have been extensively described in several publications [2,3,4,5,6,7,8]. The variety of biomass types, that can be used as feedstock for AD, includes all kind of easily degradable organic matter (OM) such as agricultural feedstocks, municipal organic wastes, industrial organic wastes, sewage sludge, and animal by-products [9, 10]. Feedstock availability, methane yield, microbial degradability or process stability of a feedstock and financial incentives are factors deciding which feedstock is favoured in the biogas sector. The residues of AD, which are referred to as digestates, are commonly used as organic fertiliser. During AD, the initial feedstock undergoes changes in composition which affect the fertilising properties of the digestate [7, 11]. A high share of the feedstock OM is metabolised by microbes to methane (CH4) and carbon dioxide (CO2), decreasing the amount of carbon (C). Hence, digestates have lower carbon to nitrogen ratio (C/N) than the initial feedstock, and contain OM with higher recalcitrance, as mainly the easily available organic C (Corg) fractions are degraded [8, 12]. A lower C/N suggests that less N immobilisation by microbes occurs [13, 14]. Recalcitrant OM, especially lignin, increases soil Corg, due to lower degradability, thereby digestates potentially provide a higher share of recalcitrant Corg for reproduction of the soil OM than the original feedstock [15,16,17]. Thus, digestates are also regarded as soil conditioner, which improve soil properties such as soil structure [8, 18, 19]. The total N content on a dry matter (DM) base is increased through concentration by DM degradation during AD. Mineralisation of organic N (Norg) to ammonium (NH4+-N) increases the share of NH4+-N on total N (NH4+/N), and thereby enhances the immediate N availability for the crops [7, 20, 21].
Advantages of digestates as soil conditioner and fertiliser are mainly attributed to its change in nutrient and OM composition during AD. However, digestates can widely differ in their composition, due to the different nature of the feedstocks [22, 23]. It might be expected that these differences in composition affect fertiliser value, as well as built up of soil OM. Since N is one of the most important plant growth limiting nutrients, it is essential to assess the N fertiliser potential of digestates. Therefore, it is common practice in field and greenhouse experiments to compare manures to mineral N fertiliser to determine the mineral fertiliser equivalent (MFE) [20, 24]. A characterisation of digestate composition depending on feedstock type and the resulting N fertilising and C mineralisation pattern is needed for proper evaluation as organic fertiliser. Therefore, the main objective of this study was to assess properties of digestates obtained from different feedstocks in order to evaluate and predict their N fertiliser value and humus reproduction potential. The following hypotheses were tested in a field and an incubation study:
-
(i)
The higher the NH4+/N of the digestate, the higher the N offtakes and MFE.
-
(ii)
The lower the C/N and Corg/Norg of the digestate, the higher the N offtakes and MFE.
-
(iii)
The lower the share of lignin and cellulose, the higher the N offtakes and MFE.
-
(iv)
The application technique of digestates in the field will directly influence the N fertilising effect: digestate field spreading without incorporation will decrease N offtake, and the higher the NH4+-N concentration, the stronger the fertiliser value losses by omission of digestate incorporation to the soil.
-
(v)
The C/N of digestates negatively affects cumulative CO2 release. Furthermore, the higher the lignin content, the higher the recalcitrance of OM in the digestates and thus, the lower the cumulative CO2 emissions.
Materials and Methods
Fertiliser Types
Seven digestates, obtained from different main feedstocks (Table 1), were collected from on-farm and commercial biogas plants in Baden-Württemberg, Germany and described by their physico-chemical properties (concentration of macro- and micronutrients, potentially toxic elements, pH, Carbonate-C etc.) and OM composition (lignin, cellulose, crude fibre content). The main feedstocks were maize silage (M), clover and grass silage (CG), grass silage (G), food waste (FW), source-separated organic household waste (OW), poultry manure (PM), and cattle slurry (CS) (Table 1). Additionally, undigested cattle slurry (unCS) was included as a kind of control. Total N and NH4+-N were determined by Kjeldhal method [25]. Total C was measured by Dumas combustion method on a macro-elemental analyser (Elementar Vario MAX, Elementar) [26]. Carbonate content (CO3) was determined by the Scheibler gasometrical method [27]. The elements phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), sulphur (S), sodium (Na), boron (B), iron (Fe), copper (Cu), zinc (Zn), chromium (Cr) and nickel (Ni) were analysed by inductively coupled plasma optical emission spectroscopy (ICP-OES VISTA Pro, Varian) after aqua regia digestion. The elements cobalt (Co), molybdenum (Mo), arsenic (As), cadmium (Cd), lead (Pb) and thallium (Tl) were measured on an Inductively Coupled Plasma Mass Spectrometer (ICP-MS) (NexION 300XX, Perkin Elmer). Mercury (Hg) and selenium (Se) were analysed by Hydride-Generation Atomic Absorption and Cold-Vapour Atomic Absorption Spectrometry respectively (AAnalyst 400, Perkin Elmer). Fibre composition was determined after Van Soest [28] and according to VDLUFA guideline 6.5 [29]. The pH and electrical conductivity (EC) (1:10) were measured in fresh samples. The ionic strength (IS) was calculated after Sommer and Husted [30] for the cations NH4+, K+, Na+, Ca2+, and Mg2+ in M l−1. The contribution of anions to ionic strength was assumed to be equal to the cationic strength [31].
Field Experiment
A 2-year (2015 and 2016) field experiment with spring wheat (Triticum aestivum L., cultivar “Tybalt”) was conducted in Hohenheim, Germany. The soil type was a Luvisol with silty clay texture, pH 7.3 (H2O), total C in 0–30 cm 1.4% and in 30–60 cm 0.9%, and total N 0.18% (0–30 cm) and 0.13% (30–60 cm). Soil mineral N was 105 and 35 kg N ha−1 (0–60 cm) in 2015 and 2016, respectively. The experiment was designed as two-factorial randomised complete block design with four replicates. Within the experiment, fertiliser treatments and incorporation were tested. The fertiliser treatments can be split into three groups: seven digestates, one cattle slurry (both were denoted as organic fertilisers), mineral fertiliser at different rates and an unfertilised soil (control). Organic fertilisers were applied to soil with and without incorporation. Field plot size was 32 m2 and split into two sampling areas (each 1.75 m width, 8 m length), fitting with the threshing size of a small harvester, separated by a pathway (50 cm). For determination of MFE, the mineral fertiliser calcium ammonium nitrate (CAN, 27% N) was applied at a rate of 0 (control), 25, 50, 75 and 100 kg N ha−1. All organic fertilisers were applied at a rate of 100 kg N ha−1 on the basis of total N. Before fertilisation, a mixed soil sample of five samples per block was taken from 0 to 30 cm and 30 to 60 cm depth. The soil was analysed for total C and N (Elementar Vario MAX CN, Elementar), and mineral N (NH4+ and NO3−) by extraction (1:4) with 0.5 M potassium sulphate (photometer, AutoAnalyzer 3 HR, SEAL). In March, before sowing spring wheat, the mineral N and organic fertilisers were applied and in case of incorporation level “with incorporation” directly incorporated with a harrow. For the incorporation level “without incorporation”, the organic fertilisers were spread in between the plant rows at tillering stage (BBCH stage 25). At ear emergence (BBCH stage 52), a random biomass sample per plot was cut per 0.5 m2 using a circular frame. Dry mass was determined and analysed for total N and total C. At harvest in August, the same sampling area of each plot was used for a second biomass sampling to assess straw yield, number of ear bearing stalks per m2, and grain number per ear. The undisturbed sampling area was threshed to determine grain yield. Two grain sub-samples each of 100 grains were counted and weighed for calculating 1000-grain mass. The C/N was measured in plant biomass, grain, and straw using a macro-elemental analyser (Elementar Vario MAX, Elementar).
Incubation Experiment
Additionally, all seven digestates and the cattle slurry from the field trial were tested in a 56-days aerobic incubation experiment. Two control treatments were included, one unfertilised soil, and one quartz sand blank, the experiment was arranged as randomised complete block design with four replicates. Top soil from an agricultural Luvisol soil with the following characteristics was used: silty-clay loam texture (68% silt, 29% clay, 3% sand), pH 7.0 (CaCl2), 0.17%total N, 1.3% Corg, 4.2 g CO3-C kg−1 soil, and a maximum water holding capacity of 53%. The soil was air-dried and sieved to a particle size of 4 mm. Digestates and undigested cattle slurry were applied as fresh matter (FM) at a rate of 2.65 g Corg kg−1 dry soil, respectively. This application rate increased soil Corg by 20%, which assured sufficient CO2 development. The quartz sand, used as blank, was glued to remove any organic residues. For soil-digestate mixing, the organic amendments were thoroughly mixed into 700 g soil, and then filled into a 2.5 l glass jar. All treatments were moistened to 60% of the water holding capacity, except for the quartz sand blank. One small beaker per jar with 20 ml 1 M sodium hydroxide was used as CO2 trap and put on top of the soil. An additional beaker with deionised water was set inside the jar to maintain soil moisture. The jars were sealed with rubber and lid and aerated at least every 2nd day for 2 min to keep aerobic conditions. The aeration phase was taken into consideration by using the quartz sand blank within CO2 calculation. CO2 evolution was determined by titration of 1 M sodium hydroxide (NaOH) with 0.5 M hydrochloric acid and previous precipitation of carbonates by addition of saturated barium chloride solution. The CO2-C emitted was calculated with the amount of HCl required during NaOH titration and given as mg CO2-C kg−1 soil [32]. The cumulative CO2 evolution or cumulative mineralised CO2 was further depicted as percentage of Corg that was added [33].
Statistical Analysis and Calculations
Data of each year were separately analysed with a mixed model approach. The model can be described by:
where yijklm is the observation of the ith control group in the jth fertiliser group with fertiliser level k and incorporation level l in the mth replicate and μ is the intercept. ci is the effect of the ith control group (with levels control and others), mij is the effect of the jth treatment group within control group level i (with the levels control, N100 and others). \(\tau_{ijk} ,\varphi_{ijl}\) and \(\left( {\tau \varphi } \right)_{ijkl}\) denoted for effects of fertiliser k, incorporation l and their interactions, respectively, again nested within the control and fertiliser group “others”. bm is the complete block effect and eijklm is the error of yijklm. Mixed model analyses for the traits N offtake and grain yield were limited to control and fertilisers applied at a rate of N100. Note that for these analyses, the CAN treatments N25, N50 and N75 were not included. They were used for calculating the MFE only.
Error variances were checked for heterogeneity across treatment levels, but model fit measured via Akaike information criterion (AIC) [34] was not increased by fitting heterogeneous variances. A Tukey test was performed after finding significance at α = 0.05 via F-test. To test whether the effect of fertiliser can be explained by covariables C/N, Corg/Norg, ADF, ADL, and NH4+-N, the model in (1) was extended as following: τijk in (1) was replaced by:
where \(\vartheta_{ijk}\) is the residual fertiliser effect not explained by the four covariables and its slopes β1 to β4. Thus, the model is:
Note that mij and αij as well as \(\varphi_{ijl}\) and αijl are completely confounded. Therefore, arbitrary one of them was dropped from the model. Further note that Corg/Norg will not be affected from incorporation and thus were dropped from (3). To select most important covariates, fertiliser effects (\(\tau_{ijk}\) and \(\left( {\tau \varphi } \right)_{ijkl}\)) were dropped and significant covariates were selected by testing models with all possible subsets of covariates. The best model was selected based on the highest adjusted coefficient of determination (adjusted R2). Additionally, the R2 was determined for the following three models: (i) model (1) or model (3) as both resulted in the same R2, (ii) model (1) after dropping the fertiliser effects and (iii) the final best model selected as described above. These three R2 values allow to calculate the partial R2 values of selected covariates, for fertiliser treatments, and their ratio. The latter can be seen as the part of variance between digestates, which can be explained by the covariates.
Multiplication of total grain N content with factor 5.7 was used to calculate grain protein content [35]. For each sample, N offtake was calculated by N content in aboveground biomass:
The N offtake at harvest was determined by the sum of N offtake by grain and straw. Data of N offtake from the CAN treatments (N0, 25, 50, 75 and 100) were plotted against the amount of N applied by linear regression analysis (Supplemental Fig. S1). The exponential function, that was fitted for the CAN data with increasing N rates, was further used to calculate the equivalent N rate corresponding to N offtake of organic fertilisers, given as MFE (in % of the total N applied) [24, 36]. For the 2nd year, both a linear and polynomial function gave fit, with no change in R2 and same output for MFE calculation.
Gaseous N losses via NH3-N volatilisation were estimated by the ALFAM2 model, which has been developed for animal slurry [37]. Thus, the retrieved output only gives an approximate idea on potential NH3-N losses from the digestates.
Pearson correlation coefficients (r) were generated for digestate properties to determine positive or negative relationships between digestate properties EC with IS, pH with Nt. In addition, digestate characteristics (C/N, Corg/Norg, NH4+/N, ADF, ADL) were correlated with N offtake in spring wheat (field study) and cumulative CO2 evolution (incubation study). Incubation data (mineralised C, n = 4) of organic fertilisers were also correlated with N offtake data from the field experiment (1st year, n = 4) at each incorporation level, as both experiments used the same digestate batches. These correlations from incubation and field experiment were based on raw data, comprising two response variables, and therefore denoted as phenotypic correlations.
In the incubation experiment, mineralised C (%) was calculated by subtracting basal soil respiration from cumulative CO2 production of the respective fertiliser treatment and dividing it by the applied amount of Corg. Carbon mineralisation data for each fertiliser treatment were fitted to a first-order kinetic model Cm = C0 (1 − e−kt) and tested against 0 [38, 39], where Cm is the cumulative amount of mineralised C (% of added Corg) after 56 days of incubation, t is time (days), C0 is the potentially amount of mineralised C (% of Corg), and k is the mineralisation-rate constant (day−1). Kinetic models that can be fitted to C mineralisation data were compared by Sleutel et al. [38], who concluded that all tested models gave similar results when generated for observed data, but when extrapolating data, the model has to be chosen carefully.
The effect of digestate properties (C/N, Corg/Norg, ADF, ADL, crude fibre and CO3-C content) on cumulative CO2 was assessed by using by testing models with all possible subsets of covariates, as described for the field study. Again, the selection of covariates was based on the highest adjusted R2.
where yim is the observation of the ith treatment in the mth complete block, ci is the effect of the ith treatment, bm is the complete block effect and eim is the error of yim. The slopes β1 to β5 were fitted for the five possible covariates. Block and error effects were assumed as random.
All analyses for field and incubation data were performed using the procedure PROC MIXED of the SAS system, version 9.4. For selection of covariates, the procedure PROC REG was used. In this case, the effects of other factors were included via dummy variables per defaults prior to selection of covariates. Significant differences were determined by Tukey test. Letter display of all pairwise mean comparisons was generated by the %MULT macro from Piepho [40].
Results
Physico-Chemical Composition of Digestates
Comparison of digestate and cattle slurry composition will be focused on the 1st sampling year. Dry matter content ranged between 3.8 (FW) and 14.1% (OW) (Table 2). For most digestates, pH varied in a narrow range 7.8–8.0, with lowest pH values in CS (7.4) and unCS (6.9). Total N in both years was positively correlated with digestate pH (r = 0.740, p = 0.002). The C/N was highest in unCS with 11.5 and lowest in FW digestate with 2.2. For the field experiment, this means FW provided the lowest input of Corg, and for the incubation study the highest N input compared to the other organic amendments. The Corg/Norg ranged from 7.7 to 24.2 in FW and unCS, respectively. The share of NH4+/N was highest for FW in both sampling years (72 and 66% NH4+). Phosphorus content showed large differences from 6.04 to 18.3 g kg−1 DM. Highest Na and Cl contents were found in FW, with 50.3 g Na and 86.9 g Cl kg−1 DM, followed by OW with 8.26 g Na and 16.2 g kg−1 Cl.
The electrical conductivity (EC) in diluted (1:10) digestates ranged from 1.4 to 3.9 mS cm−1. Considering the tenfold dilution of digestates, the EC increased to 14.0–39.0 mS cm−1 [31]. Ionic strength (IS) of digestates and unCS in 2015 was 0.247–1.06 M l−1. The correlation of EC with IS over both sampling years was intermediate (r = 0.518, p = 0.040). When removing Ca2+ from the IS calculation, as proposed by Sommer and Husted (1995), assuming all Ca to be precipitated with CO32−, NH4+ and Mg2+ with PO43 at a pH above 7, the correlation between IS and EC increased (r = 0.855, p < 0.001). The share of Na within cations was in FW (18.0%) and OW (9.8%) compared to < 4.7% for the other digestates. Similarly, the share of Cl on anionic strength was highest in FW with 20.2% and OW with 12.4% (Table 2).
Second year digestate samples showed nutrient concentrations and ratios mostly in a comparable range as the 1st year, with few exceptions. Clover-grass and PM digestate had higher N concentrations (+ 2.22–2.92% N in DM) in the 2nd year, while CS had a decrease (− 1.72) compared to the 1st year. The C/N were lowered by 0.6–2 units in CG, PM and unCS, while increased by 0.14–0.78 units for the other digestates and highest rise in CS (+ 2.23). Similarly, the Corg/Norg differed in the 2nd year. Largest decrease in Corg/Norg were observed for CG, PM and unCS (− 1.70 to − 2.60), whereas strong increases were noted for OW and CS (+ 4.19 and + 4.65, respectively). All digestate showed higher K concentrations in the 2nd year sampling, particularly CG almost doubled its concentration compared to the 1st year. The concentrations of P showed both, slight increases and decreases, with largest change in PM from 1.83 to 1.00% P.
Regarding micro nutrients, Cu values in PM (85 mg kg−1 DM) were about twice as high as in most other digestates, followed by OW with 73 mg kg−1 DM. Similarly, Zn contents were higher in PM with 339 mg kg−1 DM, while the other digestates were between 124 and 202 mg kg−1 DM. Manganese was in a range of 102–326 mg kg−1 DM, except for BW and PM with 662 and 538 mg kg−1 DM, respectively. In both years, the concentration of potentially toxic elements in digestates and unCS was below the EU threshold limits for EU organic fertiliser products such as compost and digestates (Reg. (EU) 2019/1009). However, the concentrations of heavy metals (As, Cr, Ni and Pb) were always highest in OW.
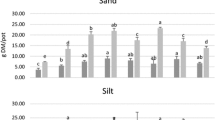
The highest crude fibre content (23.9%) was found in unCS and distinctly lowest in FW (Table 2). In all digestates, the Acid Detergent Fibre (ADF) content was higher than the Neutral Detergent Fibre (NDF). Hemicellulose was calculated according to Van Soest et al. [28] by subtraction of ADF from NDF, leading to negative values for the measured digestates. Therefore, the amount of hemicellulose could not be assessed. Most digestates had similar cellulose and lignin contents, but showed large variations between the two sampling years. FW showed distinctly low amounts of cellulose and lignin compared to the other digestates, with NDF, ADF, and ADL below the detection limit in sampling year 2015.
Incubation Experiment
Carbon Dioxide Evolution
The maximum peak of the daily CO2 release was measured within the first 2 days of incubation (Supplemental Fig. S2, Table S1). No correlation was found with the CO3-C content of fertilisers (data not shown). A second peak was observed between 5 and 10 days after digestate application. After 17 days of incubation, daily CO2-C release was below 20 mg day−1. Food waste digestate showed significantly higher daily CO2 release than all other digestates until day 35. All treatments approximated 5.5–6.2 mg CO2-C after 42 days, being still significantly higher than basal soil respiration (3.8 mg CO2-C) (Supplemental Table S1).
Cumulative CO2 volatilisation ranged from 720 to 1900 mg CO2-C kg−1 soil for the organic amendments, and 286 mg in the untreated soil (Fig. 1, Supplemental Fig. S3). FW digestate showed almost twice as high cumulative CO2 release as all other treatments, followed by M and CG being second highest. In contrast, CS was significantly lower than all other digestates, and not different from unCS.
By equalising the amount of released CO2 as mineralised Corg (Cm) added by the digestates, the share of Corg remaining in the soil could be assessed. The digestates M, CG, G, and PM all emitted around 20–22% of Corg added (Table 3). The lowest mineralised Cm occurred in a comparable range for OW, CS, and unCS (16.4–18.6%). Digested FW had the highest amount of mineralised C with 60.9%.
Cumulative Cm over the 56-day period followed a first-order kinetic model (Table 3). The mineralisation rate constant k was highest for OW with 0.102 day−1 and lowest for PM having 0.087 day−1. The calculated amount of Cm after 56 days closely approximated the modelled amount of potentially mineralised C (C0).
Effect of Digestate Composition on Cumulative CO2 Evolution
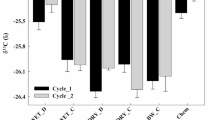
Cumulative CO2 evolution (logarithmically transformed) after 56 days of incubation was negatively correlated with C/N (r = − 0.639, p < 0.001). When omitting unCS and FW, having both, particularly high or low C/N compared to the other digestates, hence distorting the correlation, the coefficient of correlation increased (r = − 0.85, p < 0.001), whereas correlation between Corg/Norg and cumulative CO2 evolution was only intermediate (r = − 0.557, p = 0.005). The correlation of ADF and ADL fraction with CO2 was strongly negative with r = − 0.964 (p < 0.001) and r = − 0.880 (p < 0.001), respectively. Excluding FW decreased the coefficient of ADF to r = − 0.530 (p = 0.004) and lead to a non-significant correlation for ADL (r = − 0.167, p = 0.395). Also digestate CO3 content did not show a significant correlation with CO2 data. Analysing the combination of the digestate properties in a multiple regression, C/N (35.5%), ADF (26.5%), crude fibre (9.2%), ADL (8.7%) Corg/Norg (6.7%) and CO3-C (1.2%) could account for 90.4% of the variance for cumulative CO2 release (model R2 = 0.996) (Table 7).
Field Experiment
Grain Yield, Nitrogen Offtake and Harvest Parameters
Mean grain yield and N offtake of spring wheat varied between the 2 experimental years (Table 4, Supplemental Table S2). In the 1st year, grain yield ranged between 6.01 and 7.89 Mg ha−1, whereas it was about 2 Mg lower in the 2nd year (3.87–5.94 Mg ha−1) (Table 5). Regarding yield structure, the number of ears per m2 was ~ 200 ears higher in the 1st than in the 2nd year. Whereas the remaining harvest indicators appeared rather similar (thousand grain mass, grain number per ear, and grain protein content) between the years (Table 4). In the 1st year, there was no effect of digestate incorporation on grain yield, but N offtake at ear emergence was significantly higher without incorporation. Moreover, there were significant interactions between incorporation and organic fertiliser, where M, CG, OW and FW had higher N offtakes with omission of incorporation (Supplemental Table S3). In contrast, fertiliser incorporation in the 2nd year significantly increased grain yield and number of grains m−2, but not N offtake either at ear emergence or harvest. At the same time, grain protein content was significantly higher without incorporation (except for G). Harvest parameters for each treatment, incorporation level and year are provided in Supplemental Table S4. Both years showed a different pattern when comparing N offtake at ear emergence and harvest (Table 4). In the 1st year, mean N offtake at ear emergence (BBCH 51) was 105 kg N ha−1, meaning on average 52 kg of N was additionally taken up by spring wheat between ear emergence and harvest (Supplemental Table S2). In the 2nd year, most N was already taken up until ear emergence with only minor additional N uptake until harvest, on average 3 and 1.0 kg N ha−1 with or without incorporation respectively (Table 4). However, the variance of N offtake within treatments in in the 2nd year was relatively high.
Significant differences in grain yield and N offtake among digestates, unCS, N100 (CAN), and N0 (control) were found throughout both incorporation levels and years (Table 5). In the 1st year, most digestate treatments did not significantly differ in grain yield from the N100. Digestates showed significantly higher grain yields than the control N0, which had only 1.5 Mg ha−1 lower grain yield than the N100. N offtake with digestate application was also significantly higher than N0, but lower than N100. Only FW without incorporation reached comparable N offtake as N100.
In the 2nd year, grain yield of digestates with incorporation approached yield of N100 (Table 5). Only G and unCS with incorporation were significantly below yield of N100. Treatments without incorporation showed all significantly lower yields than N100, except for CG. Irrespective of incorporation, all treatments significantly increased grain yield compared to N0. In both years, there were high variations in grain yield performance throughout the different digestate treatments with, and without incorporation. The MFE ranged from 18 to 60% (1st year) and 30 to 83% (2nd year) (Table 5). Although grain yield and N offtake in the 2nd year was lower, higher MFEs were achieved compared to the 1st year.
Summing up the overall fertiliser performance on spring wheat, Table 6 shows differences among treatments determined by model (3). Considering N offtake, grain yield, and MFE in the 1st year food waste digestates showed the highest fertilisation effect. Nitrogen offtake and MFE were significantly higher with FW application, except for PM, being second highest. In the 2nd year, highest N offtake was achieved again with FW, but also with CG, both were significantly higher than all other organic amendments. For grain yield, the differences among treatments were lower, but still highest yields were achieved with FW and CG. Overall, comparing the highest and lowest N offtake within both years the difference was 17–23 kg N ha−1, while grain yield differed only marginally with 0.66–0.81 Mg ha−1 in the 1st and 2nd year, respectively (Table 6).
Effect of Digestate Composition on Grain Yield and N Offtake
In both years, N offtake data showed a significant negative linear correlation with C/N and Corg/Norg, whereas NH4+-N was positively correlated: with C/N (r = − 0.410, p < 0.001) and Corg/Norg (r = − 0.386, p = 0.002) in the 1st year, and C/N (r = − 0.343, p = 0.006) and Corg/Norg (r = − 0.296, p = 0.018) in the 2nd year. Pearson correlation coefficients of NH4+/N and N offtake were r = 0.307 (p = 0.0137) and r = 0.422 (p < 0.001) in the 1st and 2nd year, respectively.
The combined effect of digestate properties on N offtake was examined by multiple linear regression, including the parameters C/N, Corg/Norg, ADF, ADL, and NH4+/N. Except for Corg/Norg, all parameters were tested for each incorporation level (-in; -without). Due to the strong variations in growth, and the different digestate batches, both years were assessed separately. In the 1st year, C/N-without (28.0%), ADL-in (23.7%), ADF (with and without incorporation 11.6%), NH4+/N-in (8.1%) and Corg/Norg, (2.7%) could account for 74.2% of the N offtake variance among organic fertilisers (model R2 = 0.299) (Table 7). In the 2nd year, C/N (26.5%) Corg/Norg (21.4%), ADL-without (10.4%) and NH4+/N-in (0.6%) were added to the model, explaining N offtake variance among organic fertiliser treatments by 58.9% (model R2 = 0.281) (Table 7). Block effects in the field accounted for 8.20% and 2.96% in the 1st and 2nd year, respectively.
To assess the effect of digestate OM degradability on N fertiliser value, both field (1st year) and incubation data were correlated: Mineralised C of the incubation experiment was phenotypically correlated with N offtake (r = 0.522) in the variant without incorporation. However, there was neither a significant relation between field and incubation results in the variant with incorporation nor with all field data combined.
Discussion
Evaluation of Digestate Characteristics
The higher C/N in unCS compared to digestates (Table 2) can be attributed to the fact that AD typically decreases the C/N due to the unilateral release of C-gases (CH4 and CO2). The narrow C/N of FW digestates (2.2) matches findings from Tampio et al. [41]. The authors (ibid.) assessed three different digestates from food wastes, showing a similar C/N range (1.5–3.3), which was related to the high digestibility and N content on a DM base. The high NH4+-N content of food waste digestates can be explained by the protein-rich nature of kitchen wastes and its high degradability. Results on macro nutrients in digestates were likewise reported in literature [7, 22, 42, 43]. The nutrient stoichiometry, the relation of N to P and K in digestates, better matches the composition of harvested plant products than solid manures. However, the N to P ratio often does not correspond fully to crop nutrient offtakes, still showing a P surplus, and requiring N supplementation (e.g. by mineral N fertiliser, or by biological N2 fixation) within the crop rotation. Digestate N/P was most favorable in FW digestate (N/P 10.9–12.1:1), with balanced amounts of N and P, followed by maize digestate (8.7–9.3:1). The other digestates showed smaller N/P (4.7–8.1:1), increasing the risk to exceeding P loads when applied to soil without complementary sole N inputs. The narrow N to K ratio of digested and undigested CS as well as G would generally supply sufficient amounts of K to the soil, while the other digestates might need additional fertilisation when continuously applied to soil. However, these generalised nutrient ratios do not cover crops with particular high K need, such as potatoes, vegetables and sugar beets.
The EC calculated for undiluted digestates (14.0–39.0 mS cm−1) was probably overestimated when using the dilution factor 10 for calculation (n 2). Stevens et al. [31] compared diluted and undiluted EC measurements and explained, that the dilution dissolves precipitates and separates ion-pairs, hence increases EC. However, Alburquerque et al. [44] reported similar EC values as in our study, measured in undiluted digestates from cattle slurry mixed with agro-industrial wastes ranging from 9.0 to 29.0 mS cm−1. Coelho et al. measured EC values below 1 mS cm−1 and also discussed the wide range for digestate EC values mentioned in the literature [23]. Differences in EC among digestates can be explained by the number of ions, salinity, and physical properties (ibid), and is usually ruled by the main cations contained in the digestate (NH4+, Mg2+, Ca2+, K+, Na+) [31]. Hence EC was related to nutrient concentrations in FM. As expected the rather diluted unCS with its relatively low nutrient concentrations had also the lowest EC. Contrastingly, FW and CG showed the highest EC values and higher concentrations of e.g. NH4+, K+, Mg2+ than other digestates. Regarding FW, the high Na concentrations affected EC, too. This relation between EC and IS for organic fertilisers such as manures has similarly been described in the literature [30, 31]. The data and correlations regarding EC and IS indicated, that most of the Ca in digestates is not diluted, but precipitated in the present pH range of the organic fertilisers.
The high Na and Cl content found in FW and OW were expected, because cooked or processed foods contain large amounts of table salt [45]. Continuous application of digestates with such high rates of Na can pose a risk of accumulation in the soil, mainly in arid and semi-arid regions, potentially affecting salinity or sodicity, and therefore soil structure and plant growth [45]. Regarding micro nutrients (Table 2), molybdenum concentrations did not show ideal levels for methanogenic microorganisms involved in the anaerobic digestion process (≥ 7.5 mg kg−1 DM) [22], except maize digestate in both sampling years, and CG in 2016. Furthermore, none of the investigated digestates contained sufficient Ni needed by microorganisms (≥ 30 mg kg−1 DM). Zirkler et al. [22] found similar results for digestates from sewage sludge, pig slurry and maize, and pointed out that these deficits in micronutrients may reduce CH4 yield during AD. None of the digestates exceeded EU limits on heavy metals, yet highest values were found in OW. Municipal organic household waste is prone to impurities by wrong separation or e.g. by roadside greenery waste polluted with urban dust, which might have caused the elevated concentrations in OW [46, 47].
Concerning results from fibre analysis (Table 2), limitations on hemicellulose detection with Van Soest analysis of certain organic fertilisers were also observed by De Neve et al. [48]. Low amounts of lignocellulose in digested food wastes were likewise reported by Zheng et al. [49]. Such fibre composition indicates higher amount of easily decomposable OM and a potential risk for NH4+-N immobilisation, although the rather low C/N would suggest otherwise [13]. Food wastes comprise highly degradable OM with low amounts of fibres, whereas the other digestates included feedstocks containing more fibrous components with higher recalcitrance (e.g. silage). The low lignin and cellulose content in FW suggests a higher degradability. However, the risk of immobilisation by the high degradability of FW seems low, especially with the low C input by FW, low C/N and high NH4+-share.
It can be presumed that the aim of the AD process itself is the main reason for the differences in composition among digestates: AD of agricultural feedstocks are focussed on maximum energy yields, while the main focus of AD of food wastes is stabilisation and hygienisation. The other digestates indicated a rather comparable OM structure. Undigested CS was the only sample, where determination of hemicellulose was possible. Probably, because unCS did not go through AD, thereby retaining OM with low recalcitrance.
Incubation Experiment
The high daily CO2 evolution of all digestates achieved within the first 2 days of incubation has been reported similarly in the literature [33, 50]. Reasons for that can be a priming effect by digestate application and to some extent a release of soil or fertiliser CO3-C. After 17 days, CO2 release of all treatments levelled off (< 20 mg day−1) (Supplemental Fig. S1 and Table S1). The trend was similarly reported by de la Fuente et al. 21 days after digestate application [42].
Organic Matter Mineralisation
The amount of Cm or the total amount of evolved CO2 in present study cannot solely be referred to the OM of the organic fertiliser treatment. Carbon dioxide can evolve from dissolved bicarbonate or CaCO3 dissolution (e.g. after nitrification) and overestimate microbial respiration from OM by additional abiotic C [51, 52]. Bertrand et al. showed in a mineralisation experiment that soils with different texture and CO3-C content release 27–35% abiotic CO2-C from basal soil respiration [53]. Their results suggested that initial soil pH and CO3 content affect basal respiration, rather than C mineralisation [53]. In present incubation experiment, abiotic soil CO2 was not included in the calculation of cumulative CO2 development, and mineralised C was only corrected for basal soil respiration. Native soil CO3-C could have still been released as CO2. Yet, general comparisons of digestates C mineralisation and degradability in the respective soil can still be assessed without considering abiotic soil C sources. Moreover, CO3-C from the digestates showed only minor relevance (1.1%) in the model for cumulative CO2 (model 4) and was not correlated to CO2 evolution.
Regarding C mineralisation (Table 3 and Supplemental Fig. S2) of CS and M digestates tested in our study, other experiments showed higher Cm values (30–33%) after 56 and 76 days [39, 42, 54]. Also Nielsen et al. [55] compared soil respiration of different manure and plant-based digestates and found 20–44% of digestate Corg being mineralised after 178 days in a loamy and sandy soil. The higher amounts of respired CO2 compared to our results (16.4–22.0%) were presumably caused by the longer incubation period [55]. Digested FW showed the highest amount of Cm (Table 3) which indicated the highest OM degradability. Similarly, Köster et al. reported higher amounts of mineralised C when adding food waste digestate (95%) compared to undigested cattle slurry (43%) after 25 days of incubation [56]. Those high amounts of emitted CO2 (Fig. 1) were caused by the low recalcitrance of the OM in FW (Table 2). Thereby, a lower soil humus reproduction potential of FW can be expected compared to digestates derived from agricultural feedstocks like animal manures and dedicated energy plants.
The decomposability of the digestates’ OM is partially driven by the fibre composition (mainly lignin and cellulose content) and partially by the C/N of the digestates. The OW digestate showed an OM composition rather similar to the plant-based digestates, with higher amounts of cellulose and lignin (Table 2), thus OW did not significantly differ in Cm from grass and maize digestate (Table 3). Feedstocks for OW digestate were collected by the municipality and mainly composed of source-separated organic household wastes, thus a higher share of fibrous garden and kitchen wastes can be expected. However, OW had a higher recalcitrance than most plant-based digestates, as shown by significantly lower cumulative CO2 production and mineralised C amounts, although fibre composition was similar to clover-grass digestate. In this case, the higher C/N of OW might explain the slightly lower C mineralisation. Grass digestate was significantly lower in cumulative CO2 release than CG and M (Fig. 1), which also may be attributed to the slightly higher C/N and ADF fraction in grass digestate.
Field Experiment
Yield, fertiliser value, and effect of digestate composition were assessed separately for each year, due to the different digestate sampling batches with compositional variations. Overall trends were discussed for both years and generalised where applicable.
Growth Performance
The generally higher grain yields in the 1st year (Table 4) can be attributed to a higher number of ears m−2. Nerson [57] described a strong positive correlation between grain yield and ear number m−2, and emphasised that the number of ears m−2 was the dominant yield component. As mean N offtake at ear emergence appeared higher in the 2nd year than in the 1st year insufficient N supply in spring could not have affected the tillering of spring wheat in the 2nd year. Regarding weather conditions, the 2nd year exhibited more favourable climate conditions, contrasting the low grain yields in that year. For example, rainfall from sowing to ear emergence (April–June) was 155 and 245 mm in the 1st and 2nd year respectively. It was suspected overall growth conditions for spring wheat in the 2nd year were affected by leaf blotch disease (e.g. Septoria).
The relatively high grain yield for an unfertilised treatment (only 1.5 Mg ha−1 lower grain yield than the N100) might be explained by the high initial soil mineral N content, possibly caused by the relatively warm spring leading to increased mineralisation of soil Norg (Supplemental Fig. S4). Yet, even in the 2nd year, the unfertilised control took up 100 kg N ha−1 by straw and grain at harvest. Therefore, it can be assumed that during the vegetation period additional soil Norg was mineralised in a similar range to the 1st year. This must have occurred later than in the 1st year.
Although grain yield and N offtake in the 2nd year appeared to be lower than in the 1st year, higher MFEs were achieved in the 2nd year (Table 6). This can be explained by the narrow gap in N offtake between mineral and organic fertilisers in 2016. The organic treatments achieved N offtakes closer to the level of N100, hence showed higher MFE. The range between N0 and N100 within the CAN rates was much smaller in the 2nd than in the 1st year (Fig. 1). Consequently, small differences within digestate N offtake lead to pronounced differences for the MFE, especially in the 2nd year. This also explains the higher standard errors in MFEs (2nd year).
Effect of Digestate Incorporation
The effect of application technique was significant in both years, yet not coherent. Significant interactions between organic fertilisers and incorporation in the 1st year, showed varying influence of incorporation among digestates and cattle slurry (Tables 4, 5): most digestates showed higher N offtakes without incorporation (Supplemental Table S3). Only data of the 2nd year showed the hypothesised effect (hypothesis iv) that digestate field spreading without incorporation will negatively affect fertiliser efficiency. However, a decrease with omission of incorporation was only significant for grain yield, not for N offtake (Table 4). The number of grain per ear was significantly higher with incorporation for most digestates (Table 4, Supplemental Table S3) indicating a higher N supply in the early growth stages (BBCH 14–31), when grain number per ear is already predetermined. At the same time, grain protein content in the 2nd year was significantly higher without incorporation (except for G), presumably due to the lower grain yield and thousand grain mass, but similar N offtake as those digestates which were incorporated. Nitrogen offtake from most digestates in the 1st year was significantly higher without incorporation (Table 4, Supplemental Table S3). This effect on N offtake was the contrary to expectations, as fertiliser spreading without incorporation is known to increase the risk of N losses via NH3 volatilisation [58]. However, as incorporation of digestates was delayed for approximately 16 h in the 1st year, NH3 losses from digestates can be expected, which were on average 6.7 kg NH3-N ha−1 within 72 h as estimated by the ALFAM2 model (Supplemental Table S5). The loss of NH3 would thereby increase the C/N of the digestates and could have potentially enhanced N immobilisation after incorporation. Digestate properties added to the models for N offtake in both years partly corroborated this assumption. In the 1st year model (5), the C/N was not included for the incorporated treatment group, presumably due to the loss of N. Yet, within the best fitting model, NH4+-N/N was still added to the incorporated treatment group.
The correlation of N offtake and cumulative CO2 was only significant with omission of incorporation. Hence, the N loss with delayed incorporation in the 1st year might have altered N mineralisation, through immobilisation. All other fertiliser applications were either done with ideal weather conditions (without incorporation) or with immediate incorporation in the 2nd year (< 1 h) leading to negligible NH3-N losses (0.1 kg NH3-N ha−1) (Supplemental Table S5).
Effect of Digestate Composition
Digestate Composition Affecting C Mineralisation
The strong negative correlation between cumulative CO2 evolution and C/N indicates a high predictability of digestate mineralisation and verifies the first part of hypothesis (v), even though the range of C to N ratios (without unCS and FW) was rather narrow (4.1–6.5). The negative correlation between ADF of digestates and CO2 was related to differences in the cellulose to lignin ratio of the amendments. This was supported by findings of Nielsen et al. [55] who determined a strong negative correlation between CO2 respiration and lignin content of the digestates. For the digestates used in our study, the ADF fraction showed a stronger correlation than ADL. But both fractions were incorporated to the regression model of cumulative CO2-evolution after 56 days.
By direct comparison of unCS and CS in cumulative CO2 production, which did not significantly differ from each other (Fig. 1), the influence of the lower C/N in CS was not visible. In this case, the ADF and ADL fractions had to be regarded, showing a higher share of recalcitrant OM in the digested cattle slurry compared to unCS, thus showing the importance to include fibre composition together with C/N in one model. In contrast to our results, Nielsen et al. [55] found undigested cattle slurry to be among the treatments with highest Corg mineralisation (49–51%), e.g. higher than manure-based digestates (20–34%. This might be explained by the larger range in ADL among the batches of digestates and cattle slurry, promoting larger differences in mineralised C compared to present study with narrow range (16–22%). Given the fact that undigested cattle slurry was among the treatments with lowest ADL (ibid.), hence higher degradability, the CO2-release was as expected higher than the digestates. This relationship was also hypothesised (hypothesis v) in our study could be verified by correlation and regression model. Moreover, combined with other digestate properties (C/N, Corg/Norg, CO3-C, crude fibre) the model predictability of cumulative CO2 evolution could be improved. Therefore, emphasising the overall relevance of several digestate properties on soil C mineralisation.
Digestate Composition Affecting N Fertiliser Value
The comparably high N-fertiliser value of FW in both years was related to its composition (Table 2) as discussed for digestate properties. The other waste-based digestate OW was not significantly different in yield and N offtake from most plant-based digestates (Table 6), as nutrient and fibre composition were in a similar range. The plant-based digestates M, CG, and G were also comparable in the field experiment (Table 6), presumably due to compositional similarities. Other factors like NH3 emissions and digestate-soil interaction (N mineralisation vs. N immobilisation) seem to have a stronger effect on the fertiliser value than differences in composition (e.g. C/N, NH4+/N, and fibre composition). Only in the 2nd year CG showed significant higher grain yield and N offtake, corresponding to a higher NH4+-N share, lower C/N and Corg/Norg in the 2nd year sampling compared with M and G. Gutser et al. [20] reported a relatively wide range of MFEs between 40 and 60% for plant-based digestates. Digested PM consisted of many different feedstock components, especially maize silage (Table 1), hence it was closer related to a plant-based digestate. However, PM tended to show a higher N fertiliser value than M, was second highest after FW in the 1st and among the highest yields in the 2nd year. These beneficial fertilising properties in PM might originate from the additional feedstock poultry manure. Moreover, PM showed a high OM degradability, lower C/N and Corg/Norg, as well as lower lignin contents. Digested CS and unCS were not distinctive in their overall N fertiliser value and in a comparable range to the plant-based digestates, also confirmed by similarities in composition.
Data of N offtake and MFE showed a strong positive phenotypic correlation in both years, hence, it was sufficient to assess effects of digestate properties on N offtake data. Within the N offtake models (model R2), the unfertilised control and N100 treatment could describe 56.4% of the experimental variance in the 1st year (model R2 = 0.843) and 55.2% in the 2nd year (model R2 = 0.628). This high impact, as well as the data (Table 4), show that the unfertilised control and N100 encompassed the lower and upper range of the N offtake among treatments, explaining the large share within total N offtake variance. Covariables could describe 74.2 (1st year) and 58.9% (2nd year) of the N offtake variance among digestates and cattle slurry (Table 7). All added properties indicated strong correlation with each other and significant fit in different models. In both years, Corg/Norg was added to the model, a constant variable unaffected by incorporation, while NH4+/N was added only to the incorporated treatment group. Hence NH4+/N was more prone to application method, as previously discussed (cf. 4.3 “Effect of digestate incorporation”). Fibre fractions (ADL, ADF) and C/N showed slightly varying influence among years and incorporation levels, presumably promoted by the different weather conditions and the delayed incorporation of the 1st year altering the influence of digestate properties. In the grain yield model, the NH4+-N effect within the incorporated treatment group increased compared to the N offtake model, particularly in the 2nd year. It seems that the immediate NH4+-N-availability had a stronger effect on grain yield than on N offtake (Table 7). Relevant yield-building factors (grains m−2, ears plant−1, ears m−2 and grains ear−1) could have benefited from a higher share of NH4+-N in the digestates during tillering stage, explaining the increased NH4+ effect. Contrastingly for N offtake, C/N (26.5–28%) was most important together with Corg/Norg and fibre composition, all having a strong effect on soil N mineralisation and immobilisation, hence N supply over the vegetation period and total N uptake potential.
Svensson et al. [59] found in a 4-years field experiment that mineral N contained in digestates or composts was the best predictor for grain yield and N offtake within regression analysis. Yet, in multiple linear regression models, only those covariables which were tested can be accounted for. There might be additional digestate characteristics which have not been taken into consideration that could increase the predictability of the model (model 3). The C/N or Corg/Norg in organic fertilisers is known to significantly affect their fertiliser effect, and a negative correlation between N offtake or MFE with C/N or Corg/Norg has been described in the literature [24, 60]. The highest performing digestates in both years (Table 7) tended to have the lowest C/N or Corg/Norg and highest NH4+-N share. As hypothesised for the field study (hypotheses i–iii), data from N offtake should clearly be related to digestate OM recalcitrance, C/N or Corg/Norg and NH4+-N in the regression model. In both years, digestates with higher NH4+-N tended to show higher N offtakes, as indicated by the significant positive correlation (hypothesis i). Moreover, the C/N indicated a negative correlation (hypothesis ii). Although fibre fractions (ADF, ADL) were not significantly correlated with N offtake, they indicated a significant effect in the linear regression models in both years. That influence of OM degradability could also be corroborated by the significant positive correlation of N offtake data (1st year, without incorporation) with Cm results from the incubation study. Thus, a higher degradability of digestates increased N fertiliser value, partly supporting hypothesis (iii). Digestates did show compositional differences (Table 2). Yet in combination with the high variability (standard error) in the field (Table 5), those properties did not provide a strong predictability on the total experimental variance. But, as indicated in the multiple linear regression models and the feedstock related significant differences, digestate properties did affect fertiliser value, especially N offtake.
Conclusion
Digestate composition may differ in a very broad range based on the feedstocks used for anaerobic digestion, mainly regarding C/N and N content. There is a strong correlation between the electrical conductivity and ionic strength when excluding Ca, probably due to the fact that Ca precipitates at pH > 7.0. In present study, all digestates proved to be suitable organic N fertilisers, as compared to the literature, some (e.g. M, PM, FW, CG) achieving comparable yields and N offtakes as the mineral fertiliser.
The interaction between digestate and (soil) environment overlays larger variations in yield induced by compositional differences among the digestates. The effect of digestate composition on grain yield and N offtake was measureable, but the importance of certain characteristics varied: while C/N was most important for N offtake, the NH4+-N gained impact on grain yield. Furthermore, digestates’ effects on crop growth are also mediated by the time between soil application and incorporation. There are some indications that delayed incorporation of organic fertilisers after field application can even reduce the N fertiliser value, probably due to a stronger effect on N immobilisation than on NH3 emissions. Differences in organic matter degradability among the digestates can be attributed to differences on C/N and fibre fractions (particularly ADF), and therefore depends on the overall degradability of the digestates. Digestate carbonate content seems to be negligible for CO2 evolution in this incubation experiment, but soil carbonate should be considered in future studies (e. g. by selecting a carbonate free soil). To conclude, usual compositional differences among digestates only have a minor influence on digestate performance after soil application. Only in case of major differences (e.g. FW), particularly regarding C/N, NH4+-N concentration and fibre composition, larger variations in C mineralisation pattern and N fertiliser value can be expected.
Data Availability
All analysed and processed data are presented in the manuscript or added in as supplementary information. Additional information and raw data sets can be provided by the author upon request.
Code Availability
Not applicable.
References
Scarlat, N., Dallemand, J.F., Fahl, F.: Biogas: developments and perspectives in Europe. Renew. Energy 129, 457–472 (2018). https://doi.org/10.1016/j.renene.2018.03.006
Ward, A.J., Hobbs, P.J., Holliman, P.J., Jones, D.L.: Optimisation of the anaerobic digestion of agricultural resources. Bioresour. Technol. 99, 7928–7940 (2008). https://doi.org/10.1016/j.biortech.2008.02.044
Arthurson, V.: Closing the global energy and nutrient cycles through application of biogas residue to agricultural land—potential benefits and drawbacks. Energies 2, 226–242 (2009). https://doi.org/10.3390/en20200226
Holm-Nielsen, J.B., Al Seadi, T., Oleskowicz-Popiel, P.: The future of anaerobic digestion and biogas utilization. Bioresour. Technol. 100, 5478–5484 (2009). https://doi.org/10.1016/j.biortech.2008.12.046
Odlare, M., Arthurson, V., Pell, M., Svensson, K., Nehrenheim, E., Abubaker, J.: Land application of organic waste—effects on the soil ecosystem. Appl. Energy 88, 2210–2218 (2011). https://doi.org/10.1016/j.apenergy.2010.12.043
Abubaker, J., Risberg, K., Pell, M.: Biogas residues as fertilisers—effects on wheat growth and soil microbial activities. Appl. Energy 99, 126–134 (2012). https://doi.org/10.1016/j.apenergy.2012.04.050
Möller, K., Müller, T.: Effects of anaerobic digestion on digestate nutrient availability and crop growth: a review. Eng. Life Sci. 12, 242–257 (2012). https://doi.org/10.1002/elsc.201100085
Möller, K.: Effects of anaerobic digestion on soil carbon and nitrogen turnover, N emissions, and soil biological activity. A review. Agron. Sustain. Dev. (2015). https://doi.org/10.1007/s13593-015-0284-3
Gunaseelan, V.N.: Anaerobic digestion of biomass for methane production: a review. Biomass Bioenergy 13, 83–114 (1997). https://doi.org/10.1016/S0961-9534(97)00020-2
Boulamanti, A.K., Maglio, S.D., Giuntoli, J., Agostini, A.: Influence of different practices on biogas sustainability. Biomass Bioenergy 53, 149–161 (2013). https://doi.org/10.1016/j.biombioe.2013.02.020
Gutser, R., Ebertseder, T., Weber, A., Schraml, M., Schmidhalter, U.: Short-term and residual availability of nitrogen after long-term application of organic fertilizers on arable land. J. Plant Nutr. Soil Sci. (2005). https://doi.org/10.1002/jpln.200520510
Tambone, F., Genevini, P., D’Imporzano, G., Adani, F.: Assessing amendment properties of digestate by studying the organic matter composition and the degree of biological stability during the anaerobic digestion of the organic fraction of MSW. Bioresour. Technol. (2009). https://doi.org/10.1016/j.biortech.2009.02.012
Bernal, M.P., Kirchmann, H.: Carbon and nitrogen mineralization and ammonia volatilization from fresh, aerobically and anaerobically treated pig manure during incubation with soil. Biol. Fertil. Soils 13, 135–141 (1992). https://doi.org/10.1007/BF00336268
Johansen, A., Carter, M.S., Jensen, E.S., Hauggard-Nielsen, H., Ambus, P.: Effects of digestate from anaerobically digested cattle slurry and plant materials on soil microbial community and emission of CO2 and N2O. Appl. Soil Ecol. 63, 36–44 (2013). https://doi.org/10.1016/j.apsoil.2012.09.003
Reinhold, G., Klimanek, E., Breitschuh, G.: Zum Einfluss der Biogaserzeugung auf Veränderungen in der Kohlenstoffdynamik von Gülle. Archiv für Acker- und Pflanzenbau und Bodenkd 25(2), 129–137 (1991)
Tambone, F., Scaglia, B., D’Imporzano, G., Schievano, A., Orzi, V., Salati, S., Adani, F.: Assessing amendment and fertilizing properties of digestates from anaerobic digestion through a comparative study with digested sludge and compost. Chemosphere 81, 577–583 (2010). https://doi.org/10.1016/j.chemosphere.2010.08.034
Coban, H., Miltner, A., Elling, F.J., Hinrichs, K.-U., Kästner, M.: The contribution of biogas residues to soil organic matter formation and CO2 emissions in an arable soil. Soil Biol. Biochem. 86, 108–115 (2015). https://doi.org/10.1016/j.soilbio.2015.03.023
Froseth, R.B., Bakken, A.K., Bleken, M.A., Riley, H., Thorup-Kristensen, K., Hansen, S.: Improving N efficiency in barley through green manure management and biogas slurry. In: Nitrogen workshop (2012)
Saveyn, H., Eder, P.: End-of-waste criteria for biodegradable waste subjected to biological treatment (compost & digestate): Technical proposals. (2014)
Gutser, R., Ebertseder, T., Weber, A., Schraml, M., Schmidhalter, U.: Short-term and residual availability of nitrogen after long-term application of organic fertilizers on arable land. J. Plant Nutr. Soil Sci. 168, 439–446 (2005). https://doi.org/10.1002/jpln.200520510
Cavalli, D., Cabassi, G., Borrelli, L., Geromel, G., Bechini, L., Degano, L., Marino Gallina, P.: Nitrogen fertilizer replacement value of undigested liquid cattle manure and digestates. Eur. J. Agron. 73, 34–41 (2016). https://doi.org/10.1016/j.eja.2015.10.007
Zirkler, D., Peters, A., Kaupenjohann, M.: Elemental composition of biogas residues: variability and alteration during anaerobic digestion. Biomass Bioenergy 67, 89–98 (2014). https://doi.org/10.1016/j.biombioe.2014.04.021
Coelho, J.J., Prieto, M.L., Dowling, S., Hennessy, A., Casey, I., Woodcock, T., Kennedy, N.: Physical-chemical traits, phytotoxicity and pathogen detection in liquid anaerobic digestates. Waste Manag. 78, 8–15 (2018). https://doi.org/10.1016/j.wasman.2018.05.017
Delin, S., Stenberg, B., Nyberg, A., Brohede, L.: Potential methods for estimating nitrogen fertilizer value of organic residues. Soil Use Manag. 28, 283–291 (2012). https://doi.org/10.1111/j.1475-2743.2012.00417.x
Jones Jr., J.B.: Kjeldahl method for nitrogen determination. Micro-Macro Publishing, Athens, Ga (1991)
Bremner, J.M., Mulvaney, C.S.: Nitrogen—total. https://doi.org/10.2134/agronmonogr9.2.2ed.c31 (1983)
VDLUFA: VDLUFA-Methodenbuch, Band I: Die Untersuchung von Böden. VDLUFA - Verlag, Darmstadt (1991)
Van Soest, P.J., Robertson, J.B., Lewis, B.A.: Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 74, 3583–3597 (1991). https://doi.org/10.3168/jds.S0022-0302(91)78551-2
VDLUFA: VDLUFA-Methodenhandbuch, Band III: Die chemische Untersuchung von Futtermitteln. VDLUFA-Verlag, Darmstadt (1976)
Sommer, S.G., Husted, S.: The chemical buffer system in raw and digested animal slurry. J. Agric. Sci. 124, 45–53 (1995). https://doi.org/10.1017/S0021859600071239
Stevens, R.J., O’bric, C.J., Carton, O.T.: Estimating nutrient content of animal slurries using electrical conductivity. J. Agric. Sci. 125, 233–238 (1995). https://doi.org/10.1017/S0021859600084367
Isermeyer, H.: Eine einfache Methode zur Bestimmung der Bodenatmung und der Karbonate im Boden. Pflanzenernähr. Bodenkd. 56, 26–38 (1951). https://doi.org/10.1002/jpln.19520560107
Alburquerque, J.A., de la Fuente, C., Bernal, M.P.: Chemical properties of anaerobic digestates affecting C and N dynamics in amended soils. Agric. Ecosyst. Environ. 160, 15–22 (2012). https://doi.org/10.1016/j.agee.2011.03.007
Wolfinger, R.: Covariance structure selection in general mixed models. Commun. Stat. Simul. Comput. 22, 1079–1106 (1993). https://doi.org/10.1080/03610919308813143
Wrigley, C.W., Batey, I.L.: 7—assessing grain quality. In: Cauvain, S.P.B.T.-B. (ed.) Woodhead Publishing Series in Food Science, Technology and Nutrition, 2nd edn., pp. 149–187. Woodhead Publishing, Oxford (2012)
Schröder, J.: Revisiting the agronomic benefits of manure: a correct assessment and exploitation of its fertilizer value spares the environment. Bioresour. Technol. 96, 253–261 (2005). https://doi.org/10.1016/j.biortech.2004.05.015
Hafner, S.D., Pacholski, A., Bittman, S., Carozzi, M., Chantigny, M., Génermont, S., Häni, C., Hansen, M.N., Huijsmans, J., Kupper, T., Misselbrook, T., Neftel, A., Nyord, T., Sommer, S.G.: A flexible semi-empirical model for estimating ammonia volatilization from field-applied slurry. Atmos. Environ. 199, 474–484 (2019). https://doi.org/10.1016/j.atmosenv.2018.11.034
Sleutel, S., De Neve, S., Prat Roibás, M.R., Hofman, G.: The influence of model type and incubation time on the estimation of stable organic carbon in organic materials. Eur. J. Soil Sci. 56, 505–514 (2005). https://doi.org/10.1111/j.1365-2389.2004.00685.x
Alburquerque, J.A., de la Fuente, C., Carrasco, L., Cegarra, J., Bernal, M.P.: Carbon and nitrogen mineralisation in soil amended with digestates from anaerobic co-digestion processes. In: Proceedings of the 14th Ramiran International Conference “Treatment and use of organic residues in agriculture: Challenges and opportunities towards sustainable management.”, Lisbon, Portugal (2010)
Piepho, H.P.: A SAS macro for generating letter displays of pairwise mean comparisons. Commun. Biometry Crop Sci. 7, 4–13 (2012)
Tampio, E., Salo, T., Rintala, J.: Agronomic characteristics of five different urban waste digestates. J. Environ. Manag. 169, 293–302 (2016). https://doi.org/10.1016/j.jenvman.2016.01.001
de la Fuente, C., Alburquerque, J.A., Clemente, R., Bernal, M.P.: Soil C and N mineralisation and agricultural value of the products of an anaerobic digestion system. Biol. Fertil. Soils 49, 313–322 (2013). https://doi.org/10.1007/s00374-012-0719-9
Risberg, K.: Quality and function of anaerobic digestion residues. http://pub.epsilon.slu.se/12017/ (2015)
Alburquerque, J.A., de la Fuente, C., Ferrer-Costa, A., Carrasco, L., Cegarra, J., Abad, M., Bernal, M.P.: Assessment of the fertiliser potential of digestates from farm and agroindustrial residues. Biomass Bioenergy 40, 181–189 (2012). https://doi.org/10.1016/j.biombioe.2012.02.018
Pawlett, M., Tibbett, M.: Is sodium in anaerobically digested food waste a potential risk to soils? Sustain. Environ. Res. 25, 235–239 (2015)
Kupper, T., Bürge, D., Bachmann, H.J., Güsewell, S., Mayer, J.: Heavy metals in source-separated compost and digestates. Waste Manag. 34, 867–874 (2014). https://doi.org/10.1016/j.wasman.2014.02.007
Sahakyan, L., Maghakyan, N., Belyaeva, O., Tepanosyan, G., Kafyan, M., Saghatelyan, A.: Heavy metals in urban dust: contamination and health risk assessment: a case study from Gyumri, Armenia. Arab. J. Geosci. 9, 142 (2016). https://doi.org/10.1007/s12517-015-2159-y
De Neve, S., Sleutel, S., Hofman, G.: Carbon mineralization from composts and food industry wastes added to soil. Nutr. Cycl. Agroecosyst. 67, 13–20 (2003). https://doi.org/10.1023/A:1025113425069
Zheng, W., Lü, F., Phoungthong, K., He, P.: Relationship between anaerobic digestion of biodegradable solid waste and spectral characteristics of the derived liquid digestate. Bioresour. Technol. 161, 69–77 (2014). https://doi.org/10.1016/j.biortech.2014.03.016
Chen, R., Blagodatskaya, E., Senbayram, M., Blagodatsky, S., Myachina, O., Dittert, K., Kuzyakov, Y.: Decomposition of biogas residues in soil and their effects on microbial growth kinetics and enzyme activities. Biomass Bioenergy 45, 221–229 (2012). https://doi.org/10.1016/j.biombioe.2012.06.014
Ramnarine, R., Wagner-Riddle, C., Dunfield, K.E., Voroney, R.P.: Contributions of carbonates to soil CO2 emissions. Can. J. Soil Sci. 92, 599–607 (2012). https://doi.org/10.4141/CJSS2011-025
Tamir, G., Shenker, M., Heller, H., Bloom, P.R., Fine, P., Bar-Tal, A.: Can soil carbonate dissolution lead to overestimation of soil respiration? Soil Sci. Soc. Am. J. 75, 1414 (2011). https://doi.org/10.2136/sssaj2010.0396
Bertrand, I., Delfosse, O., Mary, B.: Carbon and nitrogen mineralization in acidic, limed and calcareous agricultural soils: apparent and actual effects. Soil Biol. Biochem. 39, 276–288 (2007). https://doi.org/10.1016/j.soilbio.2006.07.016
Wolf, U.: Emission of NH3, N2O and CO2 following the application of differently treated digestates from biogas production. Dissertation, Braunschweig, Technische Universität Braunschweig (2014)
Nielsen, K., Roß, C.L., Hoffmann, M., Muskolus, A., Ellmer, F., Kautz, T.: The chemical composition of biogas digestates determines their effect on soil microbial activity. Agriculture 10, 1–19 (2020). https://doi.org/10.3390/agriculture10060244
Köster, J.R., Cárdenas, L.M., Bol, R., Lewicka-Szczebak, D., Senbayram, M., Well, R., Giesemann, A., Dittert, K.: Anaerobic digestates lower N2O emissions compared to cattle slurry by affecting rate and product stoichiometry of denitrification—an N2O isotopomer case study. Soil Biol. Biochem. 84, 65–74 (2015). https://doi.org/10.1016/j.soilbio.2015.01.021
Nerson, H.: Effects of population density and number of ears on wheat yield and its components. Field Crops Res. 3, 225–234 (1980). https://doi.org/10.1016/0378-4290(80)90031-3
Sommer, S.G., Hutchings, N.J.: Ammonia emission from field applied manure and its reduction—invited paper. Eur. J. Agron. 15, 1–15 (2001). https://doi.org/10.1016/S1161-0301(01)00112-5
Svensson, K., Odlare, M., Pell, M.: The fertilizing effect of compost and biogas residues from source separated household waste. J. Agric. Sci. 142, 461–467 (2004). https://doi.org/10.1017/S0021859604004514
Sørensen, P., Fernández, J.A.: Dietary effects on the composition of pig slurry and on the plant utilization of pig slurry nitrogen. J. Agric. Sci. 140, 343–355 (2003). https://doi.org/10.1017/S0021859603003113
Acknowledgements
The authors want to thank Ingrid Claß-Mahler for her support during the fieldwork, sample preparation and analyses. We also thank Heidi Zimmermann for her practical field work and her technical expertise and guidance. Our gratitude to the experimental facility at the “Meiereihof” in Hohenheim, particularly Florian Liebelt and Thomas Ruopp, for their technical support within the field experiment. We also thank the various students assisting in the field work. Sanjib Sunwar conducted the incubation study for his master thesis and we thank him for his good work. We also would like to thank the collaborating farms for providing the digestates and the SAATEN-UNION GmbH for providing the spring wheat seeds (TYBALT). We also thank the BMEL and the Fachagentur Nachwachsende Rohstoffe (FNR) e.V. for the funding.
Funding
Open Access funding enabled and organized by Projekt DEAL. The research leading to these results received financial support from the BMEL and the Fachagentur Nachwachsende Rohstoffe (FNR) e.V. within the project FNR Gärresteigenschaften.
Author information
Authors and Affiliations
Contributions
FH conducted the experiment, evaluated the data and wrote the first draft and final manuscript. KM came up with the research focus and coordinated the project, where the experiments were part of. JH supported statistical analysis, developed the model for the field study and wrote sections of the Material and Method part. All co-authors edited, revised and supported the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interests to declare. The manuscript is part of the PhD thesis from the main author and the experiments were conducted within the project FNR Gärresteigenschaften. Financial or non-financial interests: The authors have no relevant financial or non-financial interests to disclose. The research was part of the doctoral thesis of the main author.
Ethical Approval
Not applicable.
Informed Consent
All authors gave their consent to submit the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Häfner, F., Hartung, J. & Möller, K. Digestate Composition Affecting N Fertiliser Value and C Mineralisation. Waste Biomass Valor 13, 3445–3462 (2022). https://doi.org/10.1007/s12649-022-01723-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-022-01723-y