Abstract
Ponds and wetlands around the world face anthropogenic pressures that threaten key ecosystem processes such as nutrient and organic matter cycling. Wetlands in arid and semi-arid regions are particularly at risk from uncertainty of water availability and competing pressures for use. Such threats are most acute for non-perennial systems that rely on occasional surface water flows to maintain important ecological functions.
This study investigates the decomposition of an endemic macrophyte Cycnogeton procerum (R.Br.) Buchenau as a key ecosystem process in a chain-of-ponds wetland system, located in the intermittently flowing Mulwaree River, New South Wales, Australia. The aims of this study were to identify spatial and temporal patterns and the relative importance of microbial activity in macrophyte decomposition to improve our understanding of ecological processes in these intermittent systems.
Exponential decomposition rates (proportional mass loss (g)) were highest during spring and summer (0.07-0.10 kd-1) and slower during autumn and winter (0.03-0.04 kd-1), reflecting seasonal drivers. Decomposition was significantly different in 9 mm and 150 μm mesh bags only during spring, suggesting decomposition was mostly performed by microbes, with invertebrate herbivores possibly only a factor during spring lotic conditions.
Mesotrophic conditions, regulated by flow and internal macrophyte and algal dynamics appear to maintain a highly productive, macrophyte-dominated aquatic wetland system. Temperature was a major factor in decomposition rates and expected increases due to climate change will accentuate pressure on the resilience of the macrophyte community. Alterations caused by changing climate and anthropogenic land use place the ponds at high risk.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Globally, aquatic plants are under increasing pressure due to habitat loss, particularly those in arid and semi-arid regions where climate change is expected to modify water availability and increase pressure for the resource (Dunlop and Brown 2008; Turral et al. 2008). The global distribution of wetlands, a major habitat for aquatic plants (macrophytes), has declined by up to 87% following centuries of draining, infilling and conversion for human use (Vorosmarty et al. 2000; Davidson 2014). Increasing global aquatic eutrophication means that macrophytes are under increasing stress as aquatic systems shift from macrophyte- to algal-dominated systems (Grasset et al. 2017). As a consequence of these cumulative stresses, the biodiversity of wetlands is declining at an unparalleled rate (Zedler and Kercher 2005; Kingsford et al. 2016).
Wetlands in southern Australia are particularly at risk due to relative aridity and competition for water (Specht 1990; Larkin et al. 2020) and knowledge of their condition is limited (Australian Government_Environment 2016). While macrophyte communities are common in some Australian rivers (Watson and Barmuta 2011; Paice et al. 2017), high turbidity, altered hydrology, grazing pressure and irrigation development have led to widespread reduction of macrophyte communities in others (Walker 1992). Eutrophication and changes to flow variability have both been influential in stimulating or impeding growth and resilience (Carr et al. 1997; Mackay et al. 2003) and climate change is expected to compound these effects for both riverine and wetland aquatic macrophytes (Balcombe et al. 2011). It is therefore important to document wetland and river ecosystem status and functions to protect and manage them into the future (Finlayson et al. 2017). This is particularly important for rare or iconic river and wetland systems such as chains-of-ponds (Williams and Fryirs 2020).
Macrophytes are intrinsically important in the aquatic food web (Lodge 1991; Bakker et al. 2016). A recent meta-analysis of macrophyte biomass removal by herbivores reported losses of between 44-48%, significantly greater than that reported for terrestrial systems (Wood et al. 2017). Functionally, macrophytes provide protection against erosion (Zierholz et al. 2001), refuge habitat for invertebrates and fish and a substrate for periphyton (Bouchard et al. 2007; Mitsch et al. 2013). They are integral to ecosystem health, because they increase invertebrate abundance and diversity by orders of magnitude compared to unvegetated habitats (Warfe and Barmuta 2004; Reid et al. 2008). Macrophytes have a central role in the carbon cycling of wetlands, through sequestration of carbon in plant tissue, and subsequent release of that carbon as plant material senesces and decays (Bouchard et al. 2007; Mitsch et al. 2013).
Plant litter decomposition follows a complex process of initial leaching of water soluble materials, followed by microbial colonization and conditioning (Bergfur et al. 2007), predominantly by fungal hyphomycetes (Kerr et al. 2013) and then invertebrate shredding and herbivory (Suren and Lake 1989; Graça 2001). Of these steps, microbial decomposition can be dominant, with fungi comprising more than 90% of the active microbial biomass contributing to the process (Komínková et al. 2000). Decomposition rates are variable and depend on individual plant morphology, nutrient concentration and the presence of secondary metabolites (Graça 2001), seasonality, stream flow variability (Dieter et al. 2011), natural and human induced eutrophication (Ferreira et al. 2015) and acidity (Dangles et al. 2004; Holland et al. 2012). There are also differences in decomposition rates related to spatial heterogeneity within lakes (Costantini et al. 2004), and mesocosm studies elsewhere have shown that higher temperatures accelerate macrophyte decomposition and alter heterotrophic microbial structure (Pan et al. 2021). However, many macrophyte decomposition processes have been poorly studied.
Australia has amongst the most highly variable hydrology in the world (Puckridge et al. 1998; Poff et al. 2006) and hosts a highly endemic macrophyte community (Chambers et al. 2008). Climate change is predicted to further increase variability in rainfall and thus river discharges, and under these conditions (Whetton et al. 2015), widespread rhizomatous species such as bulrushes (Typha domingensis PERS.APNI) and the common reed (Phragmites australis (CAV.) Trin ex, Steud) that disperse seed mainly by wind (anemochory), are expected to be more resilient than those such as Water Ribbons (Cycnogeton procerum (R.Br.) Buchenau) and Ribbon Weed (Vallisneria australis S.W.L.Jacobs & Les), that are mostly dispersed by water (hydrochory). Because of their limited dispersal capacity and water requirements, hydrochorous submerged macrophytes are at heightened risk of local extinction.
Mulwaree chain-of-ponds on the southern slopes of NSW, Australia, is an increasingly rare aquatic system (Williams et al. 2020). This system is comprised of deep perennial ponds, with high water clarity, set within the floodplain of the intermittent and relictual Mulwaree River (Abell 1995). The large, deep and steep sided ponds are infrequently connected by surface flows along vegetated preferential flow lines (Mould and Fryirs 2017; Williams et al. 2020). Current hydro-geomorphologic processes are not sufficient to create the ponds but maintain them (Williams et al. 2020) . Contemporary anthropogenic disturbance continues to threaten the structural (geomorphic) integrity of the ponds (Williams et al. 2020). A distinctive aquatic plant community, dominated by C. procerum, is well developed at the margins of the ponds and is particularly important in maintaining the geomorphic integrity of the chain-of-ponds (Williams et al. 2020). Loss of such a dominant macrophyte in this system could destabilize the unique geomorphic structure and impact the ecological value of these riverscapes.
The aims of this study were to document the aquatic macrophyte assemblages in the Mulwaree chains-of-ponds and to explore the spatial and temporal variation in organic matter decomposition as a key ecosystem process. It was hypothesized that macrophyte litter decomposition (sampled through four seasons), would significantly increase and decrease seasonally and vary spatially (within vegetated and mid pond non-vegetated sites). Further, it was hypothesized that, in conditions of high nutrients and comparatively small flow rates, both microbial and herbivore mediated decay would be involved in decomposition. It was expected that microbial plus herbivore mediated decomposition would be significantly higher than microbial decomposition alone.
Methods
Study Sites
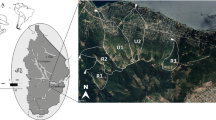
Flow along the Mulwaree system is intermittent and the ponds are only connected (along preferential flow lines) during high flow (Mould and Fryirs 2017; Williams et al. 2020) when the river becomes an active channel. Individual ponds are up to 7.5 m deep, with width and length dimensions of up to 30 to 100 m respectively (Williams et al. 2020). The chain-of-ponds are disconnected by surface flow for the majority of the time, but local aquifers maintain water volume and levels and during drought (Williams et al. 2020). Even during the severest of droughts, the ponds have not been known to dry out. The four adjacent ponds examined in this study were located at Kelburn (35° 52´ 23.41´´S, 149° 39´ 02.31´´E), near Goulburn, New South Wales, Australia (Fig. 1).
Riparian zones in the Mulwaree catchment are highly altered (EnvironmentACT. 2004). The once open Eucalyptus woodlands on the low slopes and plains have been replaced by pasture species and exotic weeds that support cattle and sheep grazing (Dodson 1986). The Mulwaree River at the study site generally reflects its agricultural catchment, with locally high electrical conductivity, total nitrogen, and total phosphorus concentrations (EnvironmentACT. 2004; GHD 2013a). There are populations of the pest fish species Gambusia holbrooki Girard 1859, which have been associated with declines in native fauna (Department of Primary Industry NSW. 2020). However, the ponds support a population of mussels (Hyridae) and a range of waterbirds including swans (Cygnus atratus), black ducks (Anas superciliosa) and dusky moorhens (Gallinula tenebrosa) (Hardwick, pers. obs.).
Study Species
Cycnogeton procerum (R.Br.) Buchenau (von Mering and Kadereit 2015) (previously Triglochin procerum R.Br), is a fleshy leaved, rhizomatous and endemic semi-emergent perennial (Rea 1992) which is morphologically variable and widespread across Australia and Malesia (Harden 1993; Brummitt et al. 2001). It is a dominant macrophyte in the Mulwaree chain-of-ponds, occupying the greatest width and depth in the pond margins. Cycnogeton procerum is described functionally as a perennial emergent with water-dispersed seeds (Casanova 2011), while being mostly submerged. It exhibits morphological plasticity to adjust to fluctuating water levels (Rea 1992), producing more, wider and longer leaves following flooding and reverting to smaller leaves with drawdown (Cooling 1996). Flowering mostly between September and March (Aston 1995), dispersal by water (hydrochory) is facilitated by buoyant seeds that can float for up to 5 weeks (James et al. 2013). Cycnogeton procerum has a rapid leaf turnover rate of between 10 and 30 days and hence has a smaller investment in secondary growth during spring and early summer (Rea 1992; Muller et al. 1994; Petherick et al. 2011). It may exhibit plasticity in growth rates under differing nutrient conditions and is adept at sequestering nutrients, in particular, phosphorus (Adcock and Ganf 1994). Functionally, C. procerum may exhibit life history attributes similar to those of other submerged, hydrochorous macrophytes such as V. australis. Submerged macrophytes are important components of lentic systems, importing energy via photosynthesis, increasing availability of nutrients and cycling phosphorus (Wang et al. 2018). They also contribute to dissolved oxygen fluxes, and are influential in altering pH and oxidation-reduction processes in water and sediments (Moss 2006).
Experimental Overview
Ponds were visited on 12 occasions between autumn (March) 2016 and spring (September) 2017 (Fig. 2). Surface water chemistry and nutrient data were collected on 6 occasions between spring (October) 2016 and spring (September) 2017. Flowing water levels in the ponds were measured between June 2016 and September 2017, following previous deployment of a depth logger installed in a shallow piezometer at the edge of pond 1.
Piezometer level at pond 1, 2016-2017 with line denoting zero commence to flow (CTF). Dots denote sampling events and bars, litter decomposition dates used for this paper. For the summer decomposition experiment, water chemistry collection is represented by one dot. Water level data were obtained from the permanently installed water level dataloggers
Vegetation mapping was performed on six occasions when leaf litter bags were deployed. Two in-situ litter decomposition experiments were performed of differing sampling frequency. A seasonal ‘simplified time series’ (Gulis et al. 2006) experiment was used to identify time required for 75-95% mass loss, adjusted to 30 days. Coarse and fine mesh litter bags were deployed on 4 occasions between spring 2016 and spring 2017. In addition, during summer, between January 9 and February 11, 2017, a ‘complete time-series’ (Gulis et al. 2006) litter decomposition rate experiment was conducted across four time points, also using coarse and fine mesh litter bags at the same locations.
Hydrology
Water levels in the ponds were measured to determine flow status within the ponds (Fig. 2) using a Solinst Levelogger depth logger installed in a shallow piezometer constructed of slotted 50 mm diameter PVC pipe that was installed 0.1 m in from the edge of pond 1. The level logger was installed at 0.2 m below cease to flow height (Williams et al. 2020) and recorded water depth hourly from 16 April 2015 to 28 September 2017 (Fig. 2). A Barologger Edge (Solinst Canada Ltd., ON, Canada) was installed in the piezometer to provide data for barometric correction (Fig. 2).
Water Chemistry
Temperature, pH, electrical conductivity (EC), dissolved oxygen and Secchi depth were measured in each pond during each sampling event between November 2016 and September 2017 (Fig. 2), using a pre-calibrated Hydrolab minisonde (Aqualab.com.au). Samples for nutrient and chemical analysis (total phosphorus (TP), dissolved organic carbon (DOC), total nitrogen (TN), ammonium (NH4+), alkalinity as bicarbonate and total alkalinity were collected between 11 November 2016 and 9 September 2017 (Fig. 2) at the centre of each pond at 0.25 m depth into triple rinsed 250 mL PET sample jars. Water samples were filtered (0.2 μm), chilled and frozen as appropriate before analysis (APHA et al. 2005). Analytes and methods for analysis are summarized in Table S1.
Historical nutrient data (1995-2013) from the nearby monitoring station Mulwaree River @ The Towers (2122725 – 149o 41´53.2´´E, 34o 46´54.7´´S) were acquired from Sydney Catchment Authority (GHD 2013b). These data were used to calculate the molar water ratios of total nitrogen and total phosphorus as a measure of nutrient limitation to primary production (Jarvie et al. 2018). Ratios of the macronutrients carbon, nitrogen and phosphorus can affect phytoplankton growth and the ratio of CNP of 106:16:1 – the Redfield ratio (Boulton et al. 2014) is a coarse indication of nutrient limitation and suitability for algal growth. Molar concentrations for TN, TP (converted) and DOC were used to derive ratios. In all analyses it was assumed that organic matter was synonymous to DOC, as is usually the case (Leenheer and Croue 2003; Boulton et al. 2014). Total phosphorus measurements taken during this study were consistently below detection limits of 0.05 mgL-1, so for contemporary analysis purposes were converted to limit of detection/√2 (LOD/√2) (Croghan and Egeghy 2003; Verbovšek 2011).
Vegetation Mapping
Macrophyte species distribution was mapped on six occasions using a rapid assessment technique derived from Gunn et al. (2010) and Wetzel and Likens (1991). Six random transects per pond were surveyed, running perpendicular to the shoreline from the uppermost extent of amphibious vegetation (above the base flow elevation) to the lower depth limit of submersed vegetation. Depth measurements were taken to measure horizontal and vertical species distribution at multiple points. Photographs were taken to verify plant distribution and identification. Identified plant taxa were allocated functional groups, based on growth form and dispersal traits (Brock and Casanova 1997; Casanova 2011; Catford and Jansson 2014).
Leaf Litter Decay
Decay of C. procerum leaves was measured by deploying leaf litter filled mesh bags in the four ponds and determining mass loss on retrieval following Boyero et al. (2016). To prepare the bags, live fresh C. procerum leaves were collected manually from each of the ponds before each deployment and washed in situ to remove periphyton, sediment and invertebrates. Leaf material was air dried for 7 days and then for three days in a drying oven at 30°C to constant mass. Five grams (+/- 0.1 g) of the dried leaves were weighed individually before being placed in numbered litter bags of 100 x 150 mm in size and made of either coarse 9 mm PVC mesh (www.whitesgroup.com.au) or fine 150 μm nylon mesh (Nytex 150 μm SEFAR 03-150/38) (www.sefar.com.au). Coarse (9 mm) mesh bags were used to quantify total organic matter loss, whereas the fine (150 μm) mesh was small enough to exclude invertebrates (Woodward et al. 2012; Mora-Gomez et al. 2015) and thus indicated microbial mediated decomposition.
To measure decay rate dynamics, a field experiment (a complete time series experiment) was performed beginning on 9 January 2017. A series of litter bags containing C. procerum of each mesh size were placed in the centre of the ponds, suspended from a buoy, at distance from macrophyte associated microbes and fauna and at a random location within the vegetated margins of each pond, all at 0.5 m depth. Bags within the vegetated margins were attached to wooden stakes that were secured in the sediment and amongst the thickly vegetated macrophyte beds to enable maximum availability of macrophyte decomposers, herbivores and detritivores. At each sampling 2 litter bags of each mesh size (in total, 2 bags X 2 mesh sizes in each pond of 4 ponds on 4 occasions) were collected in each pond at 3, 8, 15 and 32 days after deployment. Very little detritus was left beyond 32 days inundation.
To study seasonal variation in decomposition, mesh bags containing seasonally collected C. procerum leaves were deployed on four occasions over the study period from spring 2016 to spring 2017: on 1 November 2016, 7 February 2017, 26 May 2017 and 25 August 2017. Duplicate litter bags of each mesh size were deployed from buoys and wooden stakes at 3 random locations in each pond, with 8 samples of each mesh size per sampling event per pond. To ensure remnants of initial litter were available to measure a decomposition endpoint (a simplified time series experiment), bags were harvested at 23-30 days after deployment, and longer in winter than in summer.
After collection, all litter bags were placed on ice and kept at 4°C overnight. Numbered samples were rinsed and filtered through Whatman Filter, Grade 1, 100 mm diameter, ll μm pore size with absolute ethanol (AR grade) to prevent further decomposition and then rinsed with water to remove residual ethanol. Samples were then dried to constant weight at 30°C for three days and reweighed to determine mass loss over the deployment period. Carbon:Nitrogen:Hydrogen ratios of C. procerum collected in summer were determined by incineration using a LECO CHN900 analyser (LECO, USA).
Data Analysis
Starting and final masses for each vegetation sample were individually weighed for each numbered litter bag. For the simplified time series experiment we assumed exponential loss as experienced elsewhere (Graça et al. 2015). We determined the exponential decay rate (k) of leaf litter in each bag by fitting the exponential decay model, Mfinal=Minitial e-kt, where Mfinal is mass (g) remaining at the time of collection, Minitial is initial mass (g), k is the first order rate constant (day-1) and t is time in days. An individual value of k was determined of each litter bag. Decay constants (k) for replicate samples were averaged within each pond to provide a single value per time. The decay constants were compared between season and mesh sizes using two factor ANOVA with mesh size and season as fixed factors. Data fulfilled the assumptions of homogeneity of variance and normality, which were examined using plots of residuals and Q-Q plots, respectively. Analyses were undertaken in R (RCoreTeam 2020).
Decay rates were correlated with normalized environmental data using RELATE analysis based on Spearman’s Rho Correlation (Clarke and Gorley 2015). Analysis was performed using Primer Ver. 7.0.17. Incomplete data at some events were excluded from the analysis.
For the complete time series experiment, mass loss in individual bags was expressed as a percentage of initial mass. Decay curves were estimated by fitting a three-parameter exponential decay function using nonlinear regression with a binomial error structure. The upper limit parameter of the decay function was set at 1. Data for 9-mm and 150 μm mesh bags were analyzed separately. Decay models were compared using mixed model non-linear regression. Analyses were undertaken using the DRC package (Ritz and Streibig 2005) in R version 3.5.3 (RCoreTeam 2020).
Results
Hydrology and Water Chemistry
The Mulwaree River flowed for around 50% of the study period between winter 2016 and spring 2017 (Fig. 2). Mean (± S.E.) dissolved organic carbon (DOC) was 12.81± 0.16 mgL-1, ranging between 12.0-14.0 mgL-1 over the course of the study. Mean total nitrogen increased from 0.55±0.60 mgL-1 to 1.95±1.95 mgL-1 over the course of the study, while Ammonia, nitrate and nitrite levels were variable over time (Fig. 3, Table S2). Initial Carbon:Nitrogen (C:N) ratios for leaf material collected in summer were 20.24 +/- 5.99 S.E. with nitrogen concentration of 4.1%. Levels of pH increased over time, but were circumneutral, so alkalinity (average 116.3 ± 6.1 mgL-1) was likely present almost entirely as bicarbonate ions (Fig. 3). Electrical conductivity was highly related to flow events with high flows and low EC in Spring 2016 followed by increasing EC over time after the flow ceased (Fig. 3). Dissolved oxygen increased over events to almost saturation during winter (Fig. 3, Table S2). Phosphorus levels were mostly below measured detection limits in this study. Principal Components Analysis revealed that 73.2% of differences in water chemistry were explained by seasonal sampling (Fig. S1).
Long term water quality monitoring at nearby Mulwaree River @ the Towers (State of Catchment (GHD 2013b)) indicates high concentrations for both TN and TP. These include a median TN of 0.91 mgL-1 (n=35), with ranges for previous reporting 1.36 to 1.71 mgL-1. Median TP is reported as 0.03 mgL-1 (n=35) with ranges for previous reporting 0.09 to 1.11 mgL-1. Molar TN:TP ratios varied between 61:1 (2013), with previous calculated ratios of 33:1 and 34:1. More recent monitoring reports a ratio just upstream of the site as 46:1 (EcoLogical. 2020).
Vegetation Mapping
Aquatic macrophytes formed a seasonally dense margin of aquatic vegetation around the ponds, with C. procerum covering the largest surface area. Vegetation communities between the ponds were remarkably consistent, although there was some variation in species in the preferential flow lines. Stands of T. domingensis and P. australis were evident between ponds 2 and 3. Aquatic plant cover extended from above water’s edge to around 3.5 m depth and up to 10 m horizontally at the shallower ends of the ponds (Fig. 3). Gradation from terrestrial to submerged species was evident with Carex spp., Cyperus spp., Eleocharis acuta R.Br. and P. australis (above water level (0 m)) grading to Myriophyllum verrucosum Lindl., Nymphoides geminata R.Br. Kuntze, Nitella sp. (0 to 0.6 m), C. procerum, (0.6 to 1.5 m) to V. australis (to 3 m depth). Vallisneria australis leaves extended on the surface to around 3.5 m depth into the ponds. Other species were scattered along the littoral zone contributing to high biodiversity (Fig. 4).
The preferential flow lines between the ponds were populated variously by Phragmites australis, Crassula helmsii, T. domingensis, Isoetes spp., E. acuta and M. verrucosum. While plant cover appeared greater during summer, most species were perennial and mainly rhizomatous or stoloniferous. Other than localized vegetative reproduction, most species disperse primarily by water (hydrochory) and less often by waterbirds (zoochory) (Table S1).
Cycnogeton procerum exhibited fast and strongly seasonal growth, with leaf development in late winter, covering significant areas of 1 to 2 metres width on the margins of all ponds during summer, less in autumn and winter (Hardwick, pers obs). These results concur with Rea (1992) that C. procerum responds to fluctuating water levels, exhibiting morphological plasticity related to depth. Generally, aquatic plants integrate water column nutrients during the growth phase, particularly nitrogen, but also source sediment detrital carbon and nutrients in complex processes related to oxygen availability (Bornette and Puijalon 2011). The dominance of C. procerum should enable it to play a pivotal role in both maintaining pond physical and functional structure.
Litter Decay Rates of Cycnogeton Procerum
Mass loss for the complete time series experiment followed exponential decay rates (Fig. 5). There was no significant difference in decay curves between locations within ponds for either mesh size (P150 μm = 0.998, P9 mm = 0.979), so data from all locations were pooled. Once pooled by location, there was no significant difference in decay curves between mesh sizes (Fig. 5, P = 0.999).
Decay curves for Cycnogeton procerum litter in mesh bags of 9 mm ( f(x9 mm) = 0.029+ (1-0.029)*e(x/-3.72)) and 150 μm (f(x150 μm) = 0.077+ (1-0.077)*e(x/-3.06) in Mulwaree River, complete time series, January 2017. X axis = time in days. Symbols show mean (± std dev) proportion remaining and are offset slightly for clarity (n=8).
Following the complete time series experiment results, data from simple time series experiments were analysed using an exponential decay model, with samples pooled within ponds. There was a significant difference in decay rates with mesh size (F(1,24)=19.28, P<0.001), time (F(3,24)=286.84, P<0.001), and their interaction (F(3,24)=20.34, P<0.001)(Fig. 6). Decomposition rates were overall greater in the 9 mm than the 150 μm mesh (P<0.05), and rates in spring (November 2016) and summer (February 2017) (Fig. 6A) were significantly greater than those in autumn and winter (June and September 2017) (P<0.05) (Fig. 6B). The significant interaction term indicates that the difference between mesh sizes was not consistent across sampling periods, which was due to a difference between mesh sizes in November 2016 (P<0.05), but not on other sampling occasions.
Decay Rate Correlations with Water Chemistry
Mass loss from 9 mm mesh bags over 30 days was significantly and positively correlated with temperature (r=0.65, P<0.001), and negatively correlated with electrical conductivity (r = -0.65, P<0.001) and bicarbonate alkalinity (r=-0.63, P<0.001) (Table 1). All other significant correlations were greater than P>0.001 and lower R values (Table 1). Mass loss from the 150 μm mesh bags over 30 days was significantly and positively correlated with temperature (r = 0.69, P<0.001) and negatively correlated with ammonia (r =-0.62, P<0.001) (Table 1).
Discussion
The Mulwaree chain-of-ponds system supports substantial aquatic macrophyte diversity in a highly agricultural landscape. The dominant macrophyte species, C. procerum, is an important source of productivity within the ponds. In addition to its structural role as habitat for periphyton and aquatic fauna, invertebrate grazing and microbial decomposition of C. procerum leaves make it an important part of carbon cycling within the ponds. In this study, decomposition of C. procerum was highly seasonal and dependent particularly on water temperature and quality. Decomposition was driven mostly by microbial decay, which was evidenced by the similarities in mass loss and decay rates of the 9 mm and 150 μm mesh bags. In this highly productive environment with dynamic flow and temperature regimes, the system appears dynamically nutrient limited, and at risk from further agricultural impacts and climate change.
Rates of Litter Decay, Temporal, Spatial and Dynamic Processes
The rates of decay of Cycnogeton procerum were similar to those observed for this species elsewhere (Rea 1992). Exponential decay constants (k) of between 0.027 (range 0.023-0.032) in winter and 0.10 (range 0.068-0.101) in spring, were within global ranges for freshwater plants in lotic systems (median 0.041, 95%, CI 0.015-0.1) (Enriquez et al. 1993) but higher than for lentic systems including temperate (18-20°C) lakes (Belova 1993) (mean 0.021, range 0.001-0.053) and neotropical semi-arid lakes (Rezende et al. 2018) (mean 0.0037, range 0.0031-0.0055). Carbon:Nitrogen ratios for leaf material fall between known levels for emergent (median 28-32:1) and submerged macrophytes (median 8-12:1) (Bakker et al. 2016) and C. procerum elsewhere (25:1) (Roache et al. 2006). The Mulwaree chain-of-ponds are therefore an alternating lotic/lentic aquatic pond system with high relative lentic summer decay rates. Our results suggest that decomposition during summer releases carbon and nutrient stores, which may be recycled in the mixing zone or sink to the bottom of the ponds, into the stratified hypolimnion. This would make them unavailable until the ponds destratify following rainfall or in autumn as the ponds move back to diurnal stratification.
The comparatively high rate of C. procerum mass loss during spring lotic and summer lentic conditions can be explained by several extrinsic and intrinsic factors. Firstly, higher decay rates occurred during periods of increasing temperature and passing flow. Surface temperatures in the persistently stratified ponds were higher during warmer months. Combined with lentic conditions, the warmer temperatures likely accelerated decay processes as reported elsewhere (Graça et al. 2015; Grasset et al. 2017). Concurrently, higher water clarity, combined with higher temperatures is likely to have increased primary productivity by macrophytes, periphyton and phytoplankton to a point of phosphorus limitation, and thereby increasing competition for resources into autumn (Sand-Jensen and Borum 1991).
Secondly, the ponds in this study were mesotrophic, with total nitrogen (0.55-1.95 mgL-1) and historic total phosphorus (4 mgL-1), which were above ANZECC (ANZECC 2000) guidelines for slightly disturbed upland rivers (0.25 mgL-1 TN, 0.020 mgL-1 TP) and recently reported values for the Mulwaree River of 1.03 mg L-1 TN and 0.05 mg L-1 TP (EcoLogical. 2020). Generally, increasing aquatic nutrient levels from oligotrophic to eutrophic status facilitates microbial productivity, decomposition and enhances trophic interactions (Sigee 2005). With disconnection of flows between the ponds, and associated evapotranspiration and evaporation, as well as nearby cattle grazing, conditions also moved towards eutrophication.
Thirdly, nitrogen dynamics in the pond water appear complicated, with total nitrogen increasing over time, but ammonia, nitrates and nitrites being variable. This implies that it is organic nitrogen that is increasing, which may be from that incorporated into phytoplankton or derived from decomposing macrophytes (Shilla et al. 2006). When the ponds are stratified, nitrogen dynamics are further complicated because nutrient cycling occurring above and below the thermocline are separated.
However, electrical conductivity, which was higher during no-flow periods, can negatively influence decomposition (Roache et al. 2006; Fernández-Aláez et al. 2018), impacting microbial facilitated decomposition by inhibiting enzymatic activity. This is complicated by inter pond elevated EC concentrations from upstream ponds filtering downstream over time, suggesting saline groundwater input as observed by Williams et al. (2020). Roache et al. (2006) also suggested synergy between high salinity and dissolved oxygen concentrations affects decomposition of C. procerum. This may further explain some observed seasonality in decomposition rates.
Finally, as leaves were collected seasonally, we were able to match decay with the condition of plants at that time. Generally, the growth of new, more palatable (Elger et al. 2006) leaves in late winter and spring may facilitate greater rates of herbivory (Watson and Barmuta 2011). This was supported by the relatively greater mass loss in the 9 mm compared to the 150 μm mesh bags in the November 2016 (late spring) sampling period. However, the lack of difference between mass loss in 9 mm and 150 μm mesh at other sampling times suggest herbivores such as invertebrate shredders were not influential in litter decay during other seasons of the year. At these times, detritivores dominated litter decomposition. Periphyton growth on the leaves during summer may have led herbivores to have preferentially selected periphyton over C. procerum. Overall, our hypothesis that invertebrate herbivory would have a significant effect on litter decay rates was unproven for most seasons.
Larger herbivores, including waterbirds and cattle, were present in and around the ponds and were observed eating C. procerum (Hardwick, pers. obs.). While these animals would not directly affect the loss of leaf litter in the mesh bags, both waterbirds (Wood et al. 2012) and cattle (Pettit et al. 2012) impact substantially on food chain processes in wetlands elsewhere. Thus, the mass loss estimates using the small bags does not consider all mechanisms for plant matter processing in the ponds. Generally, herbivores of all sizes are influential in processing plant material by ingesting fresh or senescent plant material previously decomposed by heterotrophs, or on the periphyton that commonly colonizes C. procerum (Suren and Lake 1989).
The lack of difference in mass loss rates between mid-pond and vegetated edges was unexpected. Usually, resources within macrophyte beds are functionally disparate to open water habitats (Wang Li 2013; Bodker et al. 2015), with differences in clarity, shade and flow related nutrient availability, species attributes and richness as examples (Engelhardt and Ritchie 2001; Takamura et al. 2003). This implies that the shallower pond edge waters and those mid-pond are relatively well mixed laterally. During summer, temperature stratification may play a part in homogenization of surface waters laterally.
Microbial-mediated decomposition was the primary source of decomposition in this study, evidenced by the similar mass loss rates in the 9 mm and 150 μm mesh bags for summer, autumn and winter. Microbes require environmental C:N ratios that are close to that of their biomass to start mineralization. The ratio required for microbial use of litter is C:N of 15:1, with summer C:N ratios of our leaf material in the ponds of 20.2:1, indicating a mismatch in carbon and nitrogen. Historic median water TN:TP ratios of between 33:1 to 61:1, indicates the ponds were depauperate in TP, considerably greater than the Redfield ratio of TN:TP of 16:1 (Boulton et al. 2014). There are several reasons for comparatively low phosphorus.
Australian dryland soils are typically poor in phosphorus (Eldridge et al. 2018) and around 50% of sediment bound phosphorus in similar streams nearby (Croke 2002) derives from gully and stream bank erosion. In the Mulwaree catchment, these sediment sources are disconnected from the main ponds. The highly vegetated ponds and preferential flow lines, presumably contribute to phosphorus adsorption, making it unavailable in the water column. During times when flow is not occurring between the ponds, organic matter, including plant derived phosphorus remain in the ponds, either being integrated into more plant material or trapped beneath the thermocline and oxycline during periods of stratification (Boulton et al. 2014). All these factors contribute to lower TP concentrations in the surface water. Nitrogen, on the other hand is much more labile and present in multiple gaseous and aqueous forms. However, relationships between TN:TP ratios and aquatic plant decomposition are complex and variable. For example, correlations between nitrogen and aquatic decomposition rates may be positive during early phases but negative later in decay processes (Bridgham and Lamberti 2009), so there is much more to learn.
Threats to the Macrophyte Dominated Mulwaree System
The Mulwaree chain-of-ponds are important landscape features but are at risk in a region that is increasingly devoid of natural wetlands and wetland vegetation (Kingsford 2000; Wassens et al. 2017). The system is already mesotrophic with nutrients (Table S2) and dissolved organic carbon concentrations greater than those typically reported in Australian natural waters (Dobson and Frid 1998; Boulton et al. 2014). The Mulwaree chain-of-ponds are macrophyte-dominated, but their depth, nutrient status and high comparative water clarity, enable substantial phytoplankton productivity (Hardwick et al. 2019). Under eutrophic conditions, periphyton and phytoplankton biomass and turbidity would increase, reducing light availability and macrophyte success generally (Phillips et al. 2016), pushing the current macrophyte dominated system to a plankton dominated system.
The relationships that maintain macrophyte dominance as a stable state are complex, dynamic and subject to regime shifts (Scheffer and Carpenter 2003; Williams et al. 2009). Macrophytes remove carbon and nutrients, which are stored in plant material and rhizosphere sediments, and released into the atmosphere (Khan and Ansari 2005). When the Mulwaree ponds are disconnected, the majority of carbon and nutrients are retained within the ponds (Dise 2009), creating a sink for both materials and nutrients. Increasing periods of no-flow in intermittent aquatic systems tends to promote heterotrophy (Acuña et al. 2015) so when connected, the Mulwaree ponds may lose organic material downstream. Passing flows are likely to be important for maintaining the functionality of the current system (Williams et al. 2020).
One of the major changes predicted to occur in aquatic ecosystems under climate change is an increase of aquatic carbon, including DOC (Reitsema et al. 2018). Aquatic DOC concentrations are already rising and it is likely that the reason for these increases are a result of climate change. The quality and composition of DOC is also changing and represents a possible threat to macrophytes. While DOC can be potentially beneficial for macrophyte growth, high concentrations of humic substances limits growth and diversity (Reitsema et al. 2018).
Current modelling predictions for eastern Australia include higher average temperatures in all seasons, more hot days and warm spells, generally less rainfall during cool seasons, and increased intensity of extreme rainfall events (Whetton et al. 2015). The expected effects on Mulwaree chain-of-ponds are longer periods of no-flow and fewer, less frequent flow connection between ponds. These conditions are a threat to macrophytes, particularly during periods of flow disconnection. At these times, carbon is being sequestered during photosynthesis and stored as sedimentary detritus or released as atmospheric CO2. Changes in rainfall variability (Dise 2009) are also likely to threaten the macrophyte dominated status of the Mulwaree chain-of-ponds, resulting in changes to their nutrient and carbon cycling functions.
There were up to 12 macrophyte species in each pond, which is far more than local farm dams that support, on average, only two species (Casanova et al. 1997). The density of this vegetation community plays an important role in protecting the Mulwaree chain-of-ponds from erosion and arguably helps maintain the long term integrity of the ponds (Williams et al. 2020). Elsewhere, macrophytes are known to interact actively by creating hydrogeomorphic feedbacks within river systems (Gurnell et al. 2016). Maintaining the cover and composition of riparian vegetation is critical to minimising erosion and maintaining the physical structure of the system. Fine grained sediment erosion and channel incision is occurring in upstream areas, destroying many of the ponds and transforming them into a fully connected channel system (Williams et al. 2020). The transport of this sediment as suspended load into downstream reaches could further threaten the aquatic vegetation and fauna of the ponds. Marginal and riparian vegetation play an important role in sediment trapping and stabilising stream banks (Eamus et al. 2005). Maintaining and restoring these communities within the Mulwaree catchment will be critical to the preservation of the chain-of-ponds system. The loss of marginal vegetation, particularly macrophyte community structure will be difficult to recover (Capon et al. 2015).
Putting these complex responses into a predictive management system under climate change is difficult. Current flow management policy is unable to restrict agricultural water use, putting long term flows and below flow pond levels at risk (DPIW 2016, 2021). The combined effects of predicted climate change (increased variability and reduced flows) and anthropogenic alteration to flow (reduced replenishment flows, increased relative take and reduction in pond levels during no flow periods), combined with grazing, will likely increase periods of storage of carbon within the ponds and ultimately push macrophyte dominance towards periphyton dominance. Understanding this system and building predictive models of current and future stressors is imperative to protect what is a rare, endangered ecosystem.
Conclusion
The aquatic macrophyte community of the Mulwaree chain-of-ponds is an example of a biodiverse plant community in a rare geomorphic aquatic system. The community is largely rhizomatous and permanent, but at great risk. The reliance of many species in the community on lotic conditions for dissemination of propagules underscores the importance of flow regime in this intermittent system. Decomposition of C. procerum, a dominant aquatic macrophyte in Mulwaree ponds was highly seasonal and mostly by microbial means. Spring and Summer decay rates were higher than in Autumn and Winter, potentially explained by temperature. There were no significant differences in decay rates between the ponds or between the vegetated margins and the mid points of the ponds. The ponds themselves are mesotrophic and are at threat of transforming to algal dominated systems under anthropogenic pressures. Continued agricultural impacts, competing water uses and hydrological changes associated with climate change demand greater monitoring and management of this unique system.
Data Availability
The datasets used and analysed during the current study are available from the authors upon reasonable request.
Code Availability
Not applicable
References
Abell RS (1995) Geology, geomorphology and hydrology of the Lake Bathurst drainage basin, New South Wales. In: Organisation AGS (ed) Department of Primary Industries and Energy. Department of Primary Industries and Energy, Canberra
Acuña V, Casellas M, Corcoll N, Timoner X, Sabater S (2015) Increasing extent of periods of no flow in intermittent waterways promotes heterotrophy. Freshw Biol 60:1810–1823
Adcock PW, Ganf GG (1994) Growth Characteristics of Three Macrophyte Species Growing in a Natural and Constructed Wetland System. Water Sci Technol 29:95–102
ANZECC (2000) Australian and New Zealand Guidelines for Fresh and Marine Water Quality. Australian Water Association, Artarmon NSW
APHA, AWA, WEF (2005) Standard Methods for the Examination of Water and Wastewater, 21st edn. APHA, Washington DC
Aston HI (1995) A revision of the tuberous-rooted speciesof Triglochin L. Juncaginaceae in Australia. Muelleria 8:331–364
Australian Government Department of the Environment (2016) State of the Environment Biodiversity 2016 (Inland Waters) Report, Australian Government, Canberra, ACT Australia
Bakker ES, Wood KA, Pagès JF, Veen GF, Christianen MJA, Santamaría L, Nolet BA, Hilt S (2016) Herbivory on freshwater and marine macrophytes: A review and perspective. Aquat Bot 135:18–36
Balcombe SR, Sheldon F, Capon SJ, Bond NR, Hadwen WL, Marsh N, Bernays SJ (2011) Climate-change threats to native fish in degraded rivers and floodplains of the Murray–Darling Basin, Australia. Mar Freshw Res 62:1099–1114
Belova M (1993) Microbial decomposition of freshwater macrophytes in the littoral zone of lakes. Hydrobiologia 251:59–64
Bergfur J, Johnson RK, Sandin L, Goedkoop W, Nygren K (2007) Effects of nutrient enrichment on boreal streams: invertebrates, fungi and leaf-litter breakdown. Freshw Biol 52:1618–1633
Bodker JE, Turner RE, Tweel A, Schulz C, Swarzenski C (2015) Nutrient-enhanced decomposition of plant biomass in a freshwater wetland. Aquat Bot 127:44–52
Bornette G, Puijalon S (2011) Response of aquatic plants to abiotic factors: a review. Aquat Sci 73:1–14
Bouchard V, Frey SD, Gilbert JM, Reed SE (2007) Effects of macrophyte functional group richness on emergent freshwater wetland functions. Ecology 88:2903–2914
Boulton AJ, Brock M, Robson B, Ryder D, Chambers J, Davis J (2014) Australian Freshwater Ecology: Processes and Management. Wiley Blackwell
Boyero L et al (2016) Biotic and abiotic variables influencing plant litter breakdown in streams: a global study. Proc R Soc B Biol Sci 283:1–10
Bridgham SD, Lamberti GA (2009) Ecological Dynamics III: Decomposition in Wetlands. In: Maltby E, Barker T (eds) The Wetlands Handbook. Blackwell Publishing, Oxford UK, pp 326–345
Brock MA, Casanova MT (1997) Plant life at the edge of wetlands: ecological responses to wetting and drying patterns. In: Klomp N, Lunt I (eds) Frontiers in Ecology: Building the Links. Elsevier Science, Oxford, pp 181–192
Brummitt RKP, Hollis FS, Brummitt NA (2001) World Geographical Scheme for Recording Plant Distributions: Edition 2. In: Brummitt RK (ed) International Working Group on Taxonomic Databases For Plant Sciences (TDWG) http://www.tdwg.org/standards/109
Capon SJ, Lynch AJJ, Bond N, Chessman BC, Davis J, Davidson N, Finlayson M, Gell PA, Hohnberg D, Humphrey C, Kingsford RT, Nielsen D, Thomson JR, Taylor MP, Fryirs KA, Brierley GJ, Ward K, MacNally R (2015) Regime shifts, thresholds and multiple stable states in freshwater ecosystems; a critical appraisal of the evidence. Sci Total Environ 534:122–130
Carr GM, Duthie HC, Taylor WD (1997) Models of aquatic plant productivity: a review of the factors that influence growth. Aquat Bot 59:195–215
Casanova MT (2011) Using water plant functional groups to investigate environmental water requirements. Freshw Biol 56:2637–2652
Casanova MT, Douglas-Hill A, Brock MA, Muschal M, Bales M (1997) Farm ponds in New South Wales, Australia: relationships between macrophyte and phytoplankton abundances. Mar Freshw Res 48:353–360
Catford JA, Jansson R (2014) Drowned, buried and carried away: effects of plant traits on the distribution of native and alien species in riparian ecosystems. New Phytol 204:19–36
Chambers PA, Lacoul P, Murphy KJ, Thomaz SM (2008) Global diversity of aquatic macrophytes in freshwater. In: Balian EV, Lévêque C, Segers H, Martens K (eds) Freshwater Animal Diversity Assessment. Springer, Netherlands, Dordrecht, pp 9–26
Clarke KR, Gorley RN (2015) Primer v7: User Manual/Tutorial. PRIMER-E, Plymouth, UK, p 296
Cooling M (1996) Adaptations of aquatic macrophytes to seasonally fluctuating water levels. PhD, University of Adelaide, Adelaide, South Australia
Costantini ML, Sabetta L, Mancinelli G, Loreto R (2004) Spatial Variability of the Decomposition Rate of Schoenoplectus tatora in a Polluted Area of Lake Titicaca. J Trop Ecol 20:325–335
Croghan, C, Egeghy P (2003) Methods of dealing with values below the limit of detection using SAS Presented at Southeastern SAS User Group. St. Petersburg, FL, September 22-24,2003. https://cfpub.epa.gov/si/si_public_record_report.cfm?Lab=NERL&dirEntryId=64046
Croke J (2002) Managing phosphorus in catchments,. Fact Sheet 11. Land & Water Australia, Canberra
Dangles O, Gessner MO, Guerold F, Chauvet E (2004) Impacts of stream acidification on litter breakdown: implications for assessing ecosystem functioning. J Appl Ecol 100:1–12
Davidson NC (2014) How much wetland has the world lost? Long-term and recent trends in global wetland area. Mar Freshw Res 65:934
Department of Primary Industry NSW (2020) Eastern Gambusia, available at https://www.dpi.nsw.gov.au/fishing/aquatic-biosecurity/pests-diseases/freshwater-pests/finfish-species/gambusia
Dieter D, von Schiller D, García-roger EM, Sánchez-montoya MM, Gómez R, Mora-gómez J, Sangiorgio F, Gelbrecht J, Tockner K (2011) Preconditioning effects of intermittent stream flow on leaf litter decomposition. Aquat Sci 73:599–609
Dise NB (2009) Biogeochemical Dynamics III: The Critical Role of Carbon in Wetlands p. In: Maltby BET (ed) The Wetlands Handbook. Blackwell Scientific Ltd., Oxford UK, pp 249–265
Dobson M, Frid C (1998) Ecology of Aquatic Systems. Longman, Harlow, UK
Dodson J (1986) Holocene Vegetation and Environments Near Goulburn, New South Wales. Aust J Bot 34:231–249
DPIW (2016) Water Sharing Plan Greater Sydney Metropolitan Region Unregulated River Water Sources: Background document for amended plan 2016 incorporating the Kangaroo River Management Zone. Department of Primary Industries, Water, Sydney NSW
DPIW (2021) NSW Water Management ACT 2000 - SECT 52. NSW Department of Planning Industry and Environment-Water, Sydney NSW
Dunlop M, Brown PR (2008) Implications of climate change for Australia’s National Reserve System: A preliminary assessment., Report to the Department of Climate Change, 2008. Department of Climate Change, Canberra, Australia
Eamus D, Macinnis-Ng CM, Hose GC, Zeppel MJ, Taylor DT, Murray BR (2005) Ecosystem services: an ecophysiological examination. Aust J Bot 53:1–19
EcoLogical. (2020) Sydney Drinking Water Catchment Audit 2019 - Volume 3. Water, NSW
Eldridge DJ, Maestre FT, Koen TB, Delgado-Baquerizo M (2018) Australian dryland soils are acidic and nutrient-depleted, and have unique microbial communities compared with other drylands. J Biogeogr 45:2803–2814
Elger A, Barrat-Segretain MH, Willby NJ (2006) Seasonal variability in the palatability of freshwater macrophytes: a case study. In: Caffrey JM, Dutartre A, Haury J, Murphy KJ, Wade PM (eds) Macrophytes in Aquatic Ecosystems: From Biology to Management: Proceedings of the 11th International Symposium on Aquatic Weeds, European Weed Research Society. Springer, Netherlands, Dordrecht, pp 89–93
Engelhardt KAM, Ritchie ME (2001) Effects of macrophyte species richness on wetland ecosystem functioning and services. Nature 411:687
Enriquez S, Duarte CM, Sand-Jensen K (1993) Patterns in Decomposition Rates among Photosynthetic Organisms: The Importance of Detritus C:N:P Content. Oecologia 94:457–471
Environment ACT. (2004) Goulburn Mulwaree Riparian condition. State of Environment Reporting ACT Government Canberra, Australia
Fernández-Aláez C, Fernández-Aláez M, García-Criado F, García-Girón J (2018) Environmental drivers of aquatic macrophyte assemblages in ponds along an altitudinal gradient. Hydrobiologia 812:79–98
Ferreira V, Castagneyrol B, Koricheva J, Gulis V, Chauvet E, Graça MA (2015) A meta-analysis of the effects of nutrient enrichment on litter decomposition in streams. Biol Rev 90:669–688
Finlayson CM, Capon SJ, Rissik D, Pittock J, Fisk G, Davidson NC, Bodmin KA, Papas P, Robertson HA, Schallenberg M, Saintilan N, Edyvane K, Bino G (2017) Policy considerations for managing wetlands under a changing climate Journal of. Mar Freshw Res 68:1803–1815
GHD (2013a) 2013 Audit of the Sydney Drinking Water Catchment: Volume 1 - Main Report 23/14960 (eds) GHD Water Sciences Group Parramatta NSW 2150 Australia
GHD (2013b) 2013 Audit of the Sydney Drinking Water Catchment: Volume 2 - appendices A_H Report 23/14960 (eds) GHD Water Sciences Group Parramatta NSW 2150 Australia
Graça MS (2001) The role of invertebrates on leaf litter decomposition in streams - a Review. Int Rev Hydrobiol 86:383–393
Graça MA, Ferreira V, Canhoto C, Encalada AC, Guerrero-Bolaño F, Wantzen KM, Boyero L (2015) A conceptual model of litter breakdown in low order streams. Int Rev Hydrobiol 100:1–12
Grasset C, Levrey LH, Delolme C, Arthaud F, Bornette G (2017) The interaction between wetland nutrient content and plant quality controls aquatic plant decomposition. Wetl Ecol Manag 25:211–219
Gulis V, Ferriera V, Graca MAS (2006) Stimulation of leaf litter decomposition and associated fungi and invertebrates by moderate eutrophication: implications for stream assessment. Freshw Biol 51:1655–1669
Gunn IDM, O’Hare M, Carvalho L, Roy DB, Rothery P, Darwell AM (2010) Assessing the condition of lake habitats: a test of methods for surveying aquatic macrophyte communities. Hydrobiologia 656:87–97
Gurnell A, Corenblit D, García de Jalón D, González del Tánago M, Grabowski R, O’hare M, Szewczyk M (2016) A conceptual model of vegetation–hydrogeomorphology interactions within river corridors. River Res Appl 32:142–163
Harden GJ (1993) Flora of New South Wales - Volume 4, 1 edition. NSW University Press, Sydney NSW
Hardwick L, Hose G, Fryirs K (2019) Functional ecology: stories of Mulwaree Chain-of- Ponds, a rare geomorphic relic and aquatic enigma. ESA19: Science for Practical Solutions. Ecological Society of Australia, Launceston, Tasmania, Australia
Holland A, Duivenvoorden LJ, Kinnear SHW (2012) Naturally acidic waterways: conceptual food webs for better management and understanding of ecological functioning. Aquat Conserv Mar Freshwat Ecosyst 22:836–847
James EA, Jordan R, Griffin PC (2013) Spatial genetic analysis of two polyploid macrophytes reveals high connectivity in a modified wetland. Freshw Biol 58:2102–2113
Jarvie HP, Smith DR, Norton LR, Edwards FK, Bowes MJ, King SM, Scarlett P, Davies S, Dils RM, Bachiller-Jareno N (2018) Phosphorus and nitrogen limitation and impairment of headwater streams relative to rivers in Great Britain: A national perspective on eutrophication. Sci Total Environ 621:849–862
Kerr JL, Baldwin DS, Tobin MJ, Puskar L, Kappen P, Rees GN, Silvester E (2013) High Spatial Resolution Infrared Micro-Spectroscopy Reveals the Mechanism of Leaf Lignin Decomposition by Aquatic Fungi. PLoS ONE 8:e60857
Khan FA, Ansari AA (2005) Eutrophication: An Ecological Vision. Bot Rev 71:449–482
Kingsford RT (2000) Ecological impacts of dams, water diversions and river management on floodplain wetlands in Australia. Austral Ecol 25:109–127
Kingsford RT, Basset A, Jackson L (2016) Wetlands: conservation's poor cousins. Aquat Conserv Mar Freshwat Ecosyst 26:892–916
Komínková D, Kuehn KA, Büsing N, Steiner D, Gessner MO (2000) Microbial biomass, growth, and respiration associated with submerged litter of Phragmites australis decomposing in a littoral reed stand of a large lake. Aquat Microb Ecol 22:271–282
Larkin ZT, Ralph TJ, Tooth S, Fryirs KA, Carthey AJR (2020) Identifying threshold responses of Australian dryland rivers to future hydroclimatic change. Sci Rep 10:6653
Leenheer JA, Croue J-P (2003) Characterising Dissolved Aquatic Organic Matter. Environ Sci Technol 0103:19–26
Lodge DM (1991) Herbivory on freshwater macrophytes. Aquat Bot 41:195–224
Mackay SJ, Arthington AH, Kennard MJ, Pusey BJ (2003) Spatial variation in the distribution and abundance of submersed macrophytes in an Australian subtropical river. Aquat Bot 77:169–186
von Mering S, Kadereit JW (2015) Phylogeny, biogeography and evolution of Triglochin L. (Juncaginaceae). Mol Phylogenet Evol 83:200–212
Mitsch WJ, Bernal B, Nahlik AM, Mander Ü, Zhang L, Anderson CJ, Jørgensen SE, Brix H (2013) Wetlands, carbon, and climate change. Landsc Ecol 28:583–597
Mora-Gomez J, Elosegi A, Mas-Marti E, Romani AM (2015) Factors controlling seasonality in leaf-litter breakdown in a Mediterranean stream. Freshwat Sci 34:1245–1258
Moss B (2006) Engineering and biological approaches to the restoration from eutrophication of shallow lakes in which aquatic plant communities are important components. Hydrobiologia 200–201:367–377
Mould S, Fryirs K (2017) The Holocene evolution and geomorphology of a chain of ponds, southeast Australia: Establishing a physical template for river management. Catena 149:349–362
Muller KL, Ganf GG, Boon PI (1994) Methane flux from beds of Baumea arthrophylla (Nees) Boeckeler and Triglochin procerum R. Br. at Bool Lagoon, South Australia. Mar Freshw Res 45:1543–1553
Paice RL, Chambers JM, Robson BJ (2017) Native submerged macrophyte distribution in seasonally-flowing, south-western Australian streams in relation to stream condition. Aquat Sci 79:171–185
Pan M, Wang T, Hu B, Shi P, Xu J, Zhang M (2021) Mesocosm Experiments Reveal Global Warming Accelerates Macrophytes Litter Decomposition and Alters Decomposition-Related Bacteria Community Structure. Water 13:1940
Petherick LM, Moss PT, McGowan HA (2011) Climatic and environmental variability during the termination of the Last Glacial Stage in coastal eastern Australia-A review. Aust J Earth Sci 58:563–577
Pettit NE, Jardine TD, Hamilton SK, Sinnamon V, Valdez D, Davies PM, Douglas MM, Bunn SE (2012) Seasonal changes in water quality and macrophytes and the impact of cattle on tropical floodplain waterholes. Mar Freshw Res 63:788–800
Phillips G, Willby N, Moss B (2016) Submerged macrophyte decline in shallow lakes: What have we learnt in the last forty years? Aquat Bot 135:37–45
Poff NL, Olden JD, Pepin DM, Bledsoe BP (2006) Placing global stream flow variability in geographic and geomorphic contexts. River Res Appl 22:149–166
Puckridge JT, Sheldon J, Walker KF, Boulton AJ (1998) Flow variability and the ecology of large rivers. Mar Freshw Res 49:55–72
RCoreTeam (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Rea N (1992) The influence of water regime on the population ecology of two emergent macrophytes in South Australia. Ph.D. Thesis, The University of Adelaide, Adelaide
Reid DJ, Quinn GP, Lake PS, Reich P (2008) Terrestrial detritus supports the food webs in lowland intermittent streams of south-eastern Australia: a stable isotope study. Freshw Biol 53:2036–2050
Reitsema RE, Meire P, Schoelynck J (2018) The future of freshwater macrophytes in a changing world: dissolved organic carbon quantity and quality and its interactions with macrophytes. Front Plant Sci 9:629
Rezende RDS, Costa Novaes JL, Queiroz de Albuquerque C, Da Costa RS, Gonçalves Junior JF (2018) Aquatic invertebrates increase litter breakdown in Neotropical shallow semi-arid lakes. J Arid Environ 154:8–14
Ritz C, Streibig JC (2005) Bioassay analysis using R. J Stat Softw 12:1–22
Roache MC, Bailey PC, Boon PI (2006) Effects of salinity on the decay of the freshwater macrophyte, Triglochin procerum Aquat Bot 84:45–52
Sand-Jensen K, Borum J (1991) Interactions among phytoplankton, periphyton, and macrophytes in temperate freshwaters and estuaries. Aquat Bot 41:137–175
Scheffer M, Carpenter SR (2003) Catastrophic regime shifts in ecosystems: linking theory to observation. Trends Ecol Evol 18:648–656
Shilla D, Asaeda T, Fujino T, Sanderson B (2006) Decomposition of dominant submerged macrophytes: Implications for nutrient release in Myall Lake NSW Australia Wetlands. Ecology and Management 14(5):427
Sigee DC (2005) Eutrophication: The Microbial Response to High Nutrient Levels, Freshwater Microbiology: biodiversity and dynamic interactions of microorganisms in the freshwater environment. John Wiley & Sons Ltd., Chichester UK, pp 443–482
Specht RL (1990) Forested wetlands of Australia. In: Lugo AE, Brinson EP, Brown S (eds) Forested Wetlands. Elsevier, Amsterdam, the Netherlands, pp 387–406
Suren AM, Lake PS (1989) Edibility of fresh and decomposing macrophytes to three species of freshwater invertebrate herbivores. Hydrobiologia 178:165–178
Takamura N, Kadono Y, Fukushima M, Nakagawa M, Kim B-HO (2003) Effects of aquatic macrophytes on water quality and phytoplankton communities in shallow lakes. Ecol Res 18:381–395
Turral H, Burker J, Faurès J-M (2008) Climate change, water and food security. FAO Water Reports. Food and Agriculture Organization of the United Nations, Viale delle Terme di Caracalla, 00100 Rome Italy. Available at https://www.fao.org/3/i2096e/i2096e00.pdf
Verbovšek T (2011) A comparison of parameters below the limit of detection in geochemical analyses by substitution methods = Primerjava ocenitev parametrov pod mejo določljivosti pri geokemičnih analizah z metodo nadomeščanja. RMZ-Mater Geoenviron 58:393–404
Vorosmarty CJ, Green P, Salisbury J, Lammers RB (2000) Global water resources: vulnerability from climate change and population growth. Science 289:284–288
Walker KF (1992) The River Murray, Australia: A Semiarid Lowland River. In: Calow P, Petts GE (eds) The Rivers Handbook. Blackwell Scientific, Oxford UK, pp 472–492
Wang Li WG (2013) Influence of submerged macrophytes on phosphorus transference between sediment and overlying water in decomposition period. Acta Ecol Sin 33:5426–5437
Wang L, Liu Q, Hu C, Liang R, Qiu J, Wang Y (2018) Phosphorus release during decomposition of the submerged macrophyte Potamogeton crispus. Limnology 19:355–366
Warfe DM, Barmuta LA (2004) Habitat structural complexity mediates the foraging success of multiple predator species. Oecologia 141:171–178
Wassens S, Ning N, Hardwick L, Bino G, Maguire J (2017) Long-term changes in freshwater aquatic plant communities following extreme drought. Hydrobiologia 799:233–247
Watson A, Barmuta LA (2011) Feeding-preference trials confirm unexpected stable isotope analysis results: freshwater macroinvertebrates do consume macrophytes. Mar Freshw Res 62:1248–1257
Wetzel RG, Likens GE (1991) Limnological Analysis, Second edition. Springer-Verlag, New York
Whetton, P, Holper P, Clarke J, Webb L, Hennessy K, Colman R, Moise A, Power S, Braganza K, Watterson I, Murphy B, Timbal B, Hope P, Dowdy A, Bhend J, Kirono D, Wilson L, Grose M, Ekstrom M, Rafter T, Heady C, Narsey S, Bathols J, McInnes Kath, Monselesan D, Church J, Lenton A, O'Grady J, Bedin T, Erwin T, Li Y (2015) Climate change in Australia Projections for Australia's NRM regions CSIRO and Bureau of Meteorology, Melbourne Australia
Williams RT, Fryirs KA (2020) The morphology and geomorphic evolution of a large chain-of-ponds river system. Earth Surf Process Landf 45:1732–1748
Williams DD, Febria CM, Schriever TA (2009) Structure and mechanics of intermittent wetland communities: bacteria to anacondas. In: Herrara JR (ed) International wetlands: ecology, conservation, restoration. Nova Science 17–55
Williams RT, Fryirs KA, Hose GC (2020) The hydrological function of a large chain-of-ponds: a wetland system with intermittent surface flows. Aquat Sci 82:61
Wood KA, Stillman RA, Clarke RT, Daunt F, O’Hare MT (2012) The impact of waterfowl herbivory on plant standing crop: a meta-analysis. Hydrobiologia 686:157–167
Wood KA, O’Hare MT, McDonald C, Searle KR, Daunt F, Stillman RA (2017) Herbivore regulation of plant abundance in aquatic ecosystems. Biol Rev 92:1128–1141
Woodward G, Gessner MO, Giller PS, Gulis V, Hladyz S, Lecerf A, Malmqvist B, McKie BG, Tiegs SD, Cariss H, Dobson M, Elosegi A, Ferreira V, Graça MAS, Fleituch T, Lacoursière JO, Nistorescu M, Pozo J, Risnoveanu G, Schindler M, Vadineanu A, Vought LB-M, Chauvet E (2012) Continental-Scale Effects of Nutrient Pollution on Stream Ecosystem Functioning. Science 336:1438–1440
Zedler JB, Kercher S (2005) Wetland Resources: Status, Trends, Ecosystem Services, and Restorability. Annu Rev Environ Resour 30:39–74
Zierholz C, Prosser IP, Fogarty PJ, Rustomji P (2001) In-stream wetlands and their significance for channel filling and the catchment sediment budget, Jugiong Creek, New South Wales. Geomorphology 38:221–235
Acknowledgements
This project was supported by an Australian Research Council Linkage grant (LP130100120) awarded to Kirstie Fryirs (orcid 0000-0003-0541-3384) and Grant Hose (orcid 0000-0003-2106-5543) . Thank you to T and K Delves for field and laboratory assistance and Australian Laboratory Services and Envirolab Australia for nutrient analysis; also the owners of ‘Kelburn’ for hospitality and site access. We would like to thank anonymous reviewers who provided valuable comments on the paper.
Lorraine Hardwick is currently an honorary postdoctoral associate at Macquarie University, and an honorary adjunct research fellow at the Gulbali Institute, Charles Sturt University.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions This project was supported by an Australian Research Council Linkage grant (LP130100120) awarded to Kirstie Fryirs (orcid 0000-0003-0541-3384) and Grant Hose (orcid 0000-0003-2106-5543). Lorraine Hardwick was supported by an Australian Postgraduate Research Scholarship and a Macquarie University Higher Degree Research Funds.
Author information
Authors and Affiliations
Contributions
Research and methodology were conceived by LH, supervised by GH and KF. Manuscript was written by LH and edited by LH, GH and KF. Data collection and analysis was performed by LH, with assistance from GH. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests Statement
The authors declare no conflicts of interest.
Ethics Approval
No ethics approvals were required for this research
Consent to Participate
Not applicable
Consent for Publication
Not applicable
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 32.2 kb)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hardwick, L.J., Fryirs, K.A. & Hose, G.C. Spatial and Temporal Variation in Macrophyte Litter Decomposition in a Rare Chain-of-ponds, an Intermittent Stream and Wetland System. Wetlands 42, 33 (2022). https://doi.org/10.1007/s13157-022-01550-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-022-01550-w