Abstract
Introduction
Injectable therapies such as glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and basal insulin (BI) are well-established agents for people with type 2 diabetes (T2D). This study aimed to investigate real-world effectiveness of GLP-1 RAs or BI in adults with T2D poorly controlled on oral antihyperglycemic drugs (OADs).
Methods
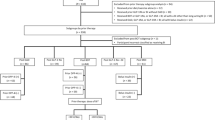
This was a retrospective, observational, longitudinal cohort study of adults with T2D from the US Optum Humedica® database and UK Clinical Practice Research Datalink, who initiated either injectable between January 1, 2010, and June 30, 2016. Baseline characteristics, glycated hemoglobin (HbA1c) change, and cumulative percentage reaching HbA1c < 7% in 24 months after initiation were analyzed in four patient cohorts.
Results
In the US and UK databases, respectively, 20,836 and 5508 patients initiated GLP-1 RAs and 60,598 and 5083 initiated BI. Baseline mean HbA1c at initiation ranged between 8.8% and 10.3% across all cohorts. In all cohorts, a decrease of HbA1c occurred 3–6 months after initiation. The cumulative percentage of patients reaching HbA1c < 7% showed the greatest probability in the first 12 months (15–40% of patients across cohorts at 12 months), particularly in the first 6 months after initiation. The probability of reaching glycemic control diminished after the second quarter. The proportion of patients reaching HbA1c < 7% in both GLP-1 RA and BI cohorts at 12 months was < 25% if baseline HbA1c was ≥ 9%.
Conclusions
For adults with T2D inadequately controlled on OADs, this analysis reveals an unmet clinical need. Initiation of first injectable therapy did not occur until HbA1c was considerably above target, when control is harder to achieve. Results suggest that in individuals with baseline HbA1c ≥ 9.0%, only a minority are likely to achieve an HbA1c < 7% with a GLP-1 RA or BI alone.
Graphic Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Injectable therapies such as glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and basal insulin (BI) are well-established agents for people with type 2 diabetes (T2D) with demonstrated antihyperglycemic efficacy in randomized controlled trials. |
This study was conducted to investigate the effectiveness of initiating therapy with either GLP-1 RA or BI in real-world clinical practice. |
What was learned from the study? |
Initiation of BI or GLP-1 RA was often delayed until the patient had reached high HbA1c values (between 8.8% and 10.3% in this study). |
People on oral antihyperglycemic drugs with glycated hemoglobin (HbA1c) ≥ 9.0% generally were unlikely to achieve control with GLP-1 RA or BI alone. |
If patients had not achieved control within 6–12 months of initiating BI or GLP-1 RA, the probability of reaching control without a treatment change was low |
Treatment intensification should be considered if people are not well controlled after 6–12 months on either injectable. |
Introduction
The benefits of achieving glycemic control (glycated hemoglobin [HbA1c] < 7%) to reduce the risk of complications for people with type 2 diabetes (T2D) are well established [1, 2]. Most people with T2D require pharmacological intervention alongside lifestyle modification with medical nutrition therapy and appropriately prescribed physical activity to attain and maintain glycemic targets [3]. Pharmacologic intervention is usually initiated with a single oral agent followed by sequential addition of oral agents before intensification to injectables [1,2,3].
Injectable therapies such as glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and basal insulin (BI) are well-established agents for people with T2D with demonstrated antihyperglycemic efficacy in randomized clinical trials (RCTs) [4, 5]. Reported ranges of people achieving HbA1c < 7% after initiation are 46–63% for GLP-1 RAs or 39–70% for BI [6,7,8]. However, there are concerns about generalizing RCT evidence of efficacy to effectiveness in routine clinical practice. In the environment of an RCT, participants are highly motivated and selected through many eligibility criteria [9] and closely followed up and monitored. In routine clinical care, the population treated for T2D is much broader, treatment may be initiated later [10, 11], adherence to therapy is variable [9], and interactions between healthcare professionals (HCPs) and patients may be less frequent and comprehensive than in RCTs. For example, differences between RCT and clinical practice patient populations were demonstrated in a real-world study of Danish adults receiving the GLP-1 RA liraglutide, in which only approximately one in four participants would have been eligible for inclusion in the LEAD RCTs [12]. Furthermore, some subpopulations are often underrepresented in RCTs, such as those > 65 years of age, with more comorbidities and/or concomitant medications, and from lower socioeconomic backgrounds [13]. The need for real-world evidence of effectiveness and safety of medications to complement the RCT evidence has increased, along with requests from multiple stakeholders including HCPs, health systems, payers, regulators, and industry [14].
The current study was conducted to investigate the effectiveness of initiating therapy with either a GLP-1 RA or BI in real-world clinical practice in people with T2D inadequately controlled on oral antihyperglycemic drugs (OADs) by analyzing the electronic medical records (EMR) and claims data from the US Optum Humedica® and UK Clinical Practice Research Datalink (CPRD) databases and to evaluate factors that might predict treatment success.
Materials and Methods
Study Design
This was a retrospective, observational, longitudinal cohort study of US or UK adults with T2D who initiated a GLP-1 RA or BI between January 1, 2010 and June 30, 2016 and were followed up for 2 years up to June 30, 2018 (Figure S1). Data were taken from the US Optum Humedica [15] and UK CPRD [16] databases.
The Optum Humedica database is a large, representative, real-world EMR and claims database in the US, holding data for > 90 million people from multiple academic and community integrated delivery networks, including hospitals, emergency departments, and ambulatory care settings.
The UK CPRD database is a large, real-world research service supporting retrospective and prospective health and clinical studies. It is sponsored by the Medicines and Healthcare Products Regulatory Agency and the National Institute for Health Research as part of the Department of Health and Social Care. The database collects anonymized longitudinal patient data from a network of general practitioner practices across the UK; data are representative of the UK population with respect to demographic factors. These data encompass > 42 million patient lives, including 13 million currently registered patients.
Ethics committee approval was not required as this was a retrospective observational study of existing anonymized electronic medical record data that were collected in daily clinical practice. No intervention was implemented on the patients for the purpose of the study, and no patient-identifiable information was used in the study.
Study Population
Patients were included in this analysis if they had diagnosis of T2D or unspecified diabetes at any time; initiated GLP-1 RA or BI within the identification period of January 1, 2010, to June 30, 2016; were ≥ 18 years of age on the index date (date of first prescription of either injectable); had ≥ 180 days of recorded medical history before and 720 days after the index date; were taking ≥ 1 OAD during the 180-day baseline period; had most recent HbA1c ≥ 7% within 90 days before and 14 days after the index date; and had ≥ 1 valid HbA1c record within 15 and 720 days after GLP-1 RA or BI initiation (Table S1). The GLP-1 RAs included exenatide, liraglutide, lixisenatide, albiglutide, or dulaglutide. The BIs included neutral protamine Hagedorn insulin, insulin detemir, insulin glargine 300 U/ml (Gla-300), and insulin glargine 100 U/ml (Gla-100) or degludec (U-100 and U-200).
Patients were excluded from the study if they had a diagnosis of type 1 diabetes (T1D) at any time, gestational diabetes within 180 days before the index date, or polycystic ovary syndrome at any time before the index date.
Comorbidities and diagnosis of any exclusion criteria (T1D, gestational diabetes, or polycystic ovary syndrome) were identified from patient records using diagnostic codes.
Persistence of BI or GLP-1 RA Use
Persistence was defined as having repeated prescriptions for the index injectable (BI or GLP-1 RA) within an estimated refill period, estimated as the 95th percentile of the number of days between subsequent prescriptions (second up to fifth consecutive prescriptions for patients with ≥ 5 prescriptions). Date of discontinuation was defined as the date of final prescription plus an estimate of the prescription duration. If the provider was changed, the patient was still followed unless the new provider was not covered by the database, in which case the patient was censored. The duration of a prescription was estimated as the median of the distribution of the number of days between subsequent prescriptions (in patients with second up to fifth consecutive prescriptions). Both refill period and prescription duration were estimated separately for each BI or GLP-1 RA brand in each country. A prescription was considered discontinued if there was no prescription of the index medication after the patient’s last prescription plus 90 days. The persistence duration was calculated from the index date until the discontinuation date or until the cutoff date for the study period.
HbA1c Outcomes
Monthly HbA1c distributions (mean ± standard deviation [SD]) from 12 months before to 24 months after the index date and the HbA1c changes from baseline to each 6-month period in 2 years were analyzed for each cohort in each country. The cumulative percentages of patients reaching HbA1c < 7% during 24 months after initiation were analyzed for the overall patient cohorts for each injectable and country and for subcohorts stratified by baseline HbA1c level, number of OADs, and sulfonylurea versus nonsulfonylurea use within the baseline period. The probability of reaching first HbA1c < 7% was estimated within each quarter across 2 years. The association between baseline characteristics and the glycemic control outcome (defined as reaching first HbA1c < 7.0%) within 2 years post-index date was analyzed for each injectable cohort in either country.
Statistical Analysis
Descriptive statistics were used for baseline characteristics. Categorical variables were summarized as number and percentage; continuous variables were summarized as mean ± SD and/or median.
The 720-day post GLP-1 RA or BI initiation period was divided into consecutive 180-day periods. The mean change in HbA1c from baseline to each 180-day period was calculated. For each time period, data from patients with both a baseline and a follow-up HbA1c within the time period were included. If a patient had more than one measurement within a window, the value closest to the midpoint of that window was selected. The number of patients and summary statistics for the change from baseline (mean ± SD) are presented. Kaplan-Meier curves were used to estimate the cumulative incidence of reaching glycemic control after the index date stratified by each subcohort. The Kaplan-Meier curves included the time from the first GLP-1 RA or BI prescription to the first HbA1c measure < 7% (outcome), end of GLP-1 RA or BI prescription (censoring event), or switch in GLP-1 RA or BI medication to a new non-GLP-1 RA or BI regimen (censoring event). Log-rank tests were applied to compare Kaplan-Meier curves between subcohorts. Within each quarter in 2 years (total of 8 quarters, 4 per year) after the index date, the probabilities of achieving first HbA1c < 7.0% in that quarter were estimated using the percentages of patients who reached glycemic control in the respective quarter among those who remained uncontrolled before that quarter.
A Cox proportional hazards model was applied to identify possible associations between baseline characteristics and the time to reach first HbA1c < 7.0% after initiation of GLP-1 RA or BI, adjusted for potential confounding factors due to the different covariates and distributions. However, given that the objective of this study was not to compare the treatment cohorts (but rather to describe the outcomes in real-world clinical practice), no statistical methodology such as propensity score matching was applied. When data were missing for patients or variables, a missing data category was included. Variable selection in multivariate models was performed using backward elimination. Starting with the full model, each variable was dropped from the model, and a likelihood ratio test performed comparing the full model to each reduced model. The variable with both the highest p value (thus the least informative) and p > 0.10 was dropped from the model. The process was repeated until no variables met the criteria to be dropped from the model. Age, gender, and baseline HbA1c were included a priori in the model.
Subgroup analyses were performed with similar statistical methodology. Statistical analysis was conducted for each of the four cohorts separately. Data from the four cohorts were not merged for statistical testing across cohorts as this was not a study objective, being not feasible owing to the complexities of the two distinct databases nor appropriate because of differences in patient characteristics. Results for each cohort are displayed side by side in tables and figures to present the data efficiently. Any comparisons described between GLP-1 RA and BI, or between the US and the UK, are descriptive only (no statistical testing was carried out between the two countries).
Results
Baseline Demographics and Patient Characteristics
Overall, 97,823,311 patients from the US Optum and 13,066,884 patients from the UK CPRD databases had valid records as of June 30, 2018. After application of inclusion/exclusion criteria, 20,836 and 60,598 patients from the US Optum database were included in the GLP-1 RA and BI cohorts, respectively. From the UK CPRD database, 5508 and 5083 patients were included in the GLP-1 RA and BI cohorts (Table S1).
Across all cohorts, patients initiating GLP-1 RA or BI had high HbA1c levels (mean 8.8–10.3%), mean age 55.9–64.8 years, long duration of diabetes (mean 8.3 or 10.1 years in the UK for GLP-1 RA or BI, respectively), and various comorbidities and complications (including dyslipidemia, obesity, hypertension, and renal diseases and cardiovascular diseases). The most frequent classes of OADs used during the baseline period were metformin, sulfonylureas, dipeptidyl peptidase-4 inhibitors, and thiazolidinediones (Table 1).
In both countries, GLP-1 RAs were prescribed to patients at a younger age (and shorter diabetes duration in the UK) and higher body mass index (BMI) compared with BI.
Medication Persistence
The persistence of GLP-1 RA or BI appeared to be longer in the UK than in the US (Table S2). Liraglutide and exenatide (daily/weekly) were the most commonly prescribed GLP-1 RAs in both databases. Glargine (Gla-300 and Gla-100) and detemir were the most commonly prescribed BIs in the US, and glargine (Gla-300 and Gla-100) and neutral protamine Hagedorn were the most commonly prescribed BIs in the UK. The estimated persistence of antihyperglycemic drugs that became available during the study period may have been impacted by their shorter duration of availability.
HbA1c Outcomes
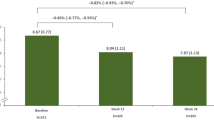
In both databases, there was a trend of increasing HbA1c values leading up to initiation of GLP-1 RA or BI, followed by a significant decrease in the 3–4 months following intensification (Fig. 1); mean HbA1c remained stable after this period. HbA1c change from baseline within each 6-month period is summarized in Table S3. A substantial reduction in the number of patients with HbA1c records occurred over time in the US database.
A higher HbA1c at baseline was associated with a reduced chance of reaching HbA1c < 7% in all cohorts. The proportion of patients reaching HbA1c < 7% at 12 months was < 25% if baseline HbA1c was ≥ 9% in both GLP-1 RA and BI cohorts (Fig. 2a–d). The cumulative percentage of patients reaching HbA1c < 7% showed the greatest increase in the first 12 months, particularly in the first 6 months of this time period (15%–40% of patients across cohorts at 12 months) (Fig. 2e and f). Patients who were on sulfonylureas at baseline were less likely to reach HbA1c < 7% than those who were not, with the largest difference in GLP-1 RA-treated patients (Figure S2).
Proportion of all patients achieving HbA1c < 7% by baseline HbA1c (≥ 7% to < 8% vs. ≥ 8% to < 9% vs. ≥ 9%) for patients initiated on GLP-1 RAs in the US (a) and UK (b), and for patients initiated on BI in the US (c) and UK (d), and for the overall population in the US and UK initiated on GLP-1 RAs (e) and those initiated on BI (f). BI basal insulin, GLP-1 RA glucagon-like peptide-1 receptor agonist, HbA1c glycated hemoglobin, UK United Kingdom, US United States
The quarterly probability of reaching HbA1c < 7% was highest in the second quarter for all cohorts (probability ~ 0.1–0.3 across cohorts; Fig. 3). A trend of decreasing probability of reaching HbA1c < 7% was observed through Quarters 2 to 8.
Among all four cohorts, higher baseline HbA1c level was consistently the strongest predictor of not achieving post-treatment HbA1c < 7% (Table 2). From the other factors, the use of > 1 OAD was a predictor of not achieving HbA1c in both US groups and in the UK group treated by GLP-1 RA. Baseline treatment with sulfonylurea was a predictor of nonachievement of the HbA1c goal in both US cohorts; baseline treatment with insulin was a predictor of nonachievement of the HbA1c goal in UK patients treated with GLP-1 RA.
Discussion
This retrospective observational study evaluated the effectiveness of initiating therapy with either a GLP-1 RA or BI in adults with T2D inadequately controlled on OADs in US and UK real-world clinical practices. The likelihood of achieving control with a single injectable medication added to oral therapy at an HbA1c > 9% was only 25%. In addition, there was a diminishing probability of reaching glycemic control if the target HbA1c was not achieved within the first 6 months on either regimen.
Although there were some differences between the US and UK populations in this study that made a direct comparison between countries inappropriate, they did share some common characteristics. Patients had a mean age of > 55 years and a long duration of T2D (> 8 years; only UK data available), and most had comorbidities. Patients also had a high mean BMI (> 30 kg/m2) and a high mean HbA1c (US 8.8% and 9.5%, and in the UK 9.6% and 10.3%, for patients initiating GLP-1 RAs or BIs, respectively) despite most being treated with ≥ 2 OADs. Given the high mean HbA1c values observed, additional oral agents, such as sodium-glucose cotransporter 2 inhibitors (SGLT2i’s), which have been demonstrated to reduce HbA1c by between −0.21 and −0.78% [17], would be unlikely to achieve glycemic goals of < 7% within the patient populations included in this study. GLP-1 RA use for those already on a SGLT2i has been shown to be effective in helping individuals reach therapeutic targets [18, 19]. These observations may reflect that in clinical practice in both countries, injectable therapies are reserved for patients with relatively advanced disease.
Additionally, injectable therapy in the UK is often provided via secondary care (e.g., diabetes specialists in hospital services); referrals from primary care to secondary care may cause delay in initiating injectable therapy.
In both countries, patients initiating GLP-1 RAs were younger and more obese than those initiating BI. In the UK, these patients had a mean BMI of 38 kg/m2 at initiation of GLP-1 RAs versus 30 kg/m2 at BI initiation. This may result from the UK guidelines that recommend GLP-1 RA therapy for T2D individuals with a BMI ≥ 35 kg/m2, and in those with a BMI < 35 kg/m2, where insulin therapy would have significant occupational implications or weight loss would benefit other significant obesity-related comorbidities [3].
A backward elimination multivariate Cox proportional hazard model was used to identify possible associations between baseline characteristics and glycemic control in each of the four cohorts. Baseline characteristics found to be associated with reduced likelihood of achieving glycemic control included a higher HbA1c level, longer duration of T2D (UK cohorts), increased number of OADs, and use of sulfonylureas (US cohorts), which may all be related to reduced β-cell function.
Of all the baseline characteristics assessed, baseline HbA1c level was the strongest predictor of the likelihood of glycemic control achievement. In the 12 months after initiation of either injectable in both countries, patients with a baseline HbA1c of 7–8% had a 40–60% probability of obtaining an HbA1c < 7%; those with baseline HbA1c of 8–9% had less chance (25–40%), and those with baseline HbA1c ≥ 9% had the lowest probability (< 25% across all cohorts). These results suggest clinicians should consider intensification of therapy with a GLP-1 RA or BI before reaching an HbA1c value of ≥ 9% to maximize the likelihood of achieving the target HbA1c. If an HbA1c < 7% is not achieved 6–12 months after initiating a single injectable, further intensification should be considered, as the probability of achieving control with unchanged therapy is low. For those with HbA1c ≥ 9.0%, prior to first injectable, clinicians should consider a combination of GLP-1 RA and insulin at initiation.
Cumulative incident curves for the proportions of patients reaching HbA1c < 7% demonstrated a sharp increase in the first 6–12 months with little increase thereafter. Additionally, quarterly probabilities showed that the best chance of a person achieving an HbA1c < 7% was shortly after initiation of injectables and diminished quickly thereafter, remaining < 10% in the second year. Blonde et al. revealed similar findings in adults with T2D initiating BI based on data from the US IBM Explorys® database [20]. Together, these results suggest that the chance of achieving glycemic control is much lower after the first year of treatment and that if, despite adequate dosing of GLP-1 RA or BI, people have not reached their target HbA1c after 6–12 months, further intensification with additional medications and/or switching to more potent therapy should be considered.
The datasets used in the present study are considered well representative of the patient populations in both countries. However, there are a number of limitations including bias inherent to retrospective analyses of EMR and claims data [21]. Retrospective real-world evidence studies do not have initial randomization to fully control for known and unknown confounders. Moreover, patient monitoring and follow-up are not consistent [21]. In both datasets, data were collected from clinical practice and were not specifically designed for research purposes. As a result, often data that would be useful for analysis may be missing, erroneous, or misclassified [22]. Moreover, dosing information of BI was not available, and individual glycemic control goals were not available. Thus, it is not possible to tell for all injectables if they were up-titrated to their maximum effective dose and how well people adhered to treatment. The American Diabetes Association Standards of Care state that a reasonable HbA1c goal for many nonpregnant adults is < 7% [1]; however, local guidelines, factors such as age or comorbidities, or patient/HCP choice may limit the number of people striving for this target.
Additionally, there is clinical practice information that EMR and claims databases do not usually include, such as reasons for therapy choice, or the events that may have resulted in the sharp rise in HbA1c 3–6 months before injectable therapy was initiated (e.g., intercurrent illness, decreased adherence because of medication side effects or cost issues). Lastly, the study period may have limited the inclusion of data about more recently marketed GLP-1 RAs or BIs as well as data with the addition of either drug class on top of the SGLT2 inhibitors that have become more popular in the past several years.
This retrospective, observational, longitudinal cohort study using two large EMR and claims databases revealed the effectiveness in real-world practice of initiating GLP-1 RA or BI medications in the US and UK between 2010 and 2016, with a 2-year follow-up. These medications were mostly initiated in people with T2D with long disease duration, various complications/comorbidities, high BMI, and a high HbA1c despite ongoing OAD treatment. Our results suggest that clinicians should consider earlier intensification of therapy with a GLP-1 RA or BI before reaching an HbA1c value of ≥ 9% to maximize the likelihood of achieving target HbA1c. Regimens with greater antihyperglycemic efficacy including combination injectable therapy may be warranted for those people with HbA1c ≥ 9% despite taking multiple OADs, since in the present study the likelihood of achieving control with a single injectable medication added to oral therapy once HbA1c is > 9% was only about 25%, much less than the likelihood of achieving control when injectable therapy is started at a lower HbA1c. In addition, if glycemic control has not been achieved within the first 6 months of treatment with either regimen, there is a diminishing probability that control will be achieved without further treatment intensification, which should be considered.
References
American Diabetes Association. Standards of medical care in diabetes—2020. Diabetes Care. 2020;43:S1–S224.
Davies MJ, D’Alessio DA, Fradkin J, et al. Management of hyperglycaemia in type 2 diabetes. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia. 2018;2018(61):2461–98. https://doi.org/10.1007/s00125-018-4729-5.
National Institute for Health and Care Excellence (NICE). Type 2 diabetes in adults: management (2015) [November 14]; https://www.nice.org.uk/guidance/ng28
Htike ZZ, Zaccardi F, Papamargaritis D, Webb DR, Khunti K, Davies MJ. Efficacy and safety of glucagon-like peptide-1 receptor agonists in type 2 diabetes: a systematic review and mixed-treatment comparison analysis. Diabetes Obes Metab. 2017;19:524–36. https://doi.org/10.1111/dom.12849.
Wallia A, Molitch ME. Insulin therapy for type 2 diabetes mellitus. JAMA. 2014;311:2315–25. https://doi.org/10.1001/jama.2014.5951.
Esposito K, Chiodini P, Bellastella G, Maiorino MI, Giugliano D. Proportion of patients at HbA1c target <7% with eight classes of antidiabetic drugs in type 2 diabetes: systematic review of 218 randomized controlled trials with 78 945 patients. Diabetes Obes Metab. 2012;14:228–33. https://doi.org/10.1111/j.1463-1326.2011.01512.x.
Esposito K, Mosca C, Brancario C, Chiodini P, Ceriello A, Giugliano D. GLP-1 receptor agonists and HbA1c target of <7% in type 2 diabetes: meta-analysis of randomized controlled trials. Curr Med Res Opin. 2011;27:1519–28. https://doi.org/10.1185/03007995.2011.590127.
Hermansen K, Davies M, Derezinski T, Martinez Ravn G, Clauson P, Home P. A 26-week, randomized, parallel, treat-to-target trial comparing insulin detemir with NPH insulin as add-on therapy to oral glucose-lowering drugs in insulin-naive people with type 2 diabetes. Diabetes Care. 2006;29:1269–74. https://doi.org/10.2337/dc05-1365.
Edelman SV, Polonsky WH. Type 2 diabetes in the real world: the elusive nature of glycemic control. Diabetes Care. 2017;40:1425–32. https://doi.org/10.2337/dc16-1974.
Khunti K, Nikolajsen A, Thorsted BL, Andersen M, Davies MJ, Paul SK. Clinical inertia with regard to intensifying therapy in people with type 2 diabetes treated with basal insulin. Diabetes Obes Metab. 2016;18:401–9. https://doi.org/10.1111/dom.12626.
Khunti K, Wolden ML, Thorsted BL, Andersen M, Davies MJ. Clinical inertia in people with type 2 diabetes: a retrospective cohort study of more than 80,000 people. Diabetes Care. 2013;36:3411–7. https://doi.org/10.2337/dc13-0331.
Knudsen JS, Thomsen RW, Pottegård A, Knop FK, Sørensen HT. Differences between randomized clinical trial patients and real-world initiators of the glucagon-like peptide 1 receptor agonist liraglutide. Diabetes Care. 2018;41:e133–e135. https://doi.org/10.2337/dc18-0999.
Booth CM, Tannock IF. Randomised controlled trials and population-based observational research: partners in the evolution of medical evidence. Br J Cancer. 2014;110:551–5. https://doi.org/10.1038/bjc.2013.725.
Khosla S, White R, Medina J, et al. Real world evidence (RWE)—a disruptive innovation or the quiet evolution of medical evidence generation? F1000Research. 2018;7:111. https://doi.org/10.12688/f1000research.13585.2.
Optum Inc. Optum [website] (2019). Available from: https://www.optum.com/.
CPRD. Clinical Practice Research Datalink [website] (2019) [Feb 2019]; Available from: https://www.cprd.com/
Yang Y, Zhao C, Ye Y, Yu M, Qu X. Prospect of sodium-glucose co-transporter 2 inhibitors combined with insulin for the treatment of type 2 diabetes. Front Endocrinol (Lausanne). 2020;11:190. https://doi.org/10.3389/fendo.2020.00190.
Ludvik B, Frias JP, Tinahones FJ, et al. Dulaglutide as add-on therapy to SGLT2 inhibitors in patients with inadequately controlled type 2 diabetes (AWARD-10): a 24-week, randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2018;6:370–81. https://doi.org/10.1016/S2213-8587(18)30023-8.
Zinman B, Bhosekar V, Busch R, et al. Semaglutide once weekly as add-on to SGLT-2 inhibitor therapy in type 2 diabetes (SUSTAIN 9): a randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2019;7:356–67. https://doi.org/10.1016/S2213-8587(19)30066-X.
Blonde L, Meneghini L, Peng XV, et al. Probability of achieving glycemic control with basal insulin in patients with type 2 diabetes in real-world practice in the USA. Diabetes Ther. 2018;9:1347–58. https://doi.org/10.1007/s13300-018-0413-5.
Kim HS, Lee S, Kim JH. Real-world evidence versus randomized controlled trial. Clinical research based on electronic medical records. J Korean Med Sci. 2018;33:e213. https://doi.org/10.3346/jkms.2018.33.e213.
Hernandez-Boussard T, Monda KL, Crespo BC, Riskin D. Real world evidence in cardiovascular medicine: ensuring data validity in electronic health record-based studies. J Am Med Inform Assoc. 2019;26:1189–94. https://doi.org/10.1093/jamia/ocz119.
Acknowledgements
Funding
The authors received writing and editorial support in the preparation of this manuscript, provided by Barrie Anthony of Evidence Scientific Solutions, and funded by Sanofi US, Inc. This study was funded by Sanofi US, Inc. Open access fees are funded by Sanofi US, Inc.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
Rory J. McCrimmon, Anders Boss, Terry Dex, Neil Skolnik, Linong Ji, Angelo Avogaro, and Lawrence Blonde contributed to data interpretation and the drafting, critical review, and revision of the manuscript. Xuejun Victor Peng designed the study and contributed to data interpretation and the drafting, critical review, and revision of the manuscript. Robert Lubwama and Leah Shepherd acquired and analyzed the data and contributed to data interpretation and the drafting, critical review, and revision of the manuscript. All authors approved the final draft of the manuscript. Xuejun Victor Peng is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Disclosures
Xuejun Victor Peng was an employee of Sanofi US, Inc., at the time of the study and is now an employee of Takeda Pharmaceutical Company Limited (Cambridge, MA). Anders Boss, Robert Lubwama, and Terry Dex are employees of Sanofi US, Inc. Rory J. McCrimmon has received honoraria for advisory board activity and educational lectures from Eli Lilly, Novo Nordisk, and Sanofi. Leah Shepherd was an employee of Evidera for the duration of this study. Neil Skolnik reported receiving nonfinancial support from AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, and Sanofi; and receiving personal fees and serving on advisory boards of AstraZeneca, Boehringer Ingelheim, Eli Lilly, GlaxoSmithKline, Intarcia, Janssen Pharmaceuticals, Merck, Mylan, Sanofi, and Teva Pharmaceutical. Linong Ji has received consulting and lecture fees from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Eli Lilly, GlaxoSmithKline, Guangzhou Zhongyi Pharmaceutical, Johnson & Johnson, Merck, Novartis, Novo Nordisk, Roche, Sanofi-Aventis, Takeda, and research funding from Bayer and Guangzhou Zhongyi Pharmaceutical. Angelo Avogaro has received honoraria or consulting fees from AstraZeneca, Boehringer Ingelheim, Eli Lilly, Mundipharma, Novartis, Novo Nordisk, Sanofi, Servier, and Takeda, and research funding from AstraZeneca. Lawrence Blonde has received grant/research support for himself and/or his institution from Janssen Pharmaceuticals, Inc., Lexicon Pharmaceuticals, Inc., Merck & Co., Novo Nordisk, and Sanofi; has received support as a speaker from Janssen Pharmaceuticals, Inc., Novo Nordisk, and Sanofi; and has acted as a consultant for AstraZeneca, Gilead Sciences, Inc., Janssen Pharmaceuticals, Inc., Merck & Co., Inc., Novo Nordisk, and Sanofi.
Compliance with Ethics Guidelines
Ethics committee approval was not required as this was a retrospective observational study of existing anonymized electronic medical record data that were collected in daily clinical practice. No intervention was implemented on the patients for the purpose of the study, and no patient-identifiable information was used in the study.
Data Availability
Qualified researchers may request access to patient-level data and related study documents including the clinical study report, study protocol with any amendments, blank case report form, statistical analysis plan, and dataset specifications. Patient-level data will be anonymized and study documents will be redacted to protect the privacy of our trial participants. Further details on Sanofi’s data-sharing criteria, eligible studies, and process for requesting access can be found at: https://www.clinicalstudydatarequest.com/.
Author information
Authors and Affiliations
Corresponding author
Additional information
Digital Features
To view digital features for this article go to https://doi.org/10.6084/m9.figshare.12759614.
Xuejun Victor Peng affiliated at the time of the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Peng, X.V., McCrimmon, R.J., Shepherd, L. et al. Glycemic Control Following GLP-1 RA or Basal Insulin Initiation in Real-World Practice: A Retrospective, Observational, Longitudinal Cohort Study. Diabetes Ther 11, 2629–2645 (2020). https://doi.org/10.1007/s13300-020-00905-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-020-00905-y